Intraoperative Guidance Using Hyperspectral Imaging: A Review for Surgeons
Abstract
:1. Introduction
2. Theoretical Overview
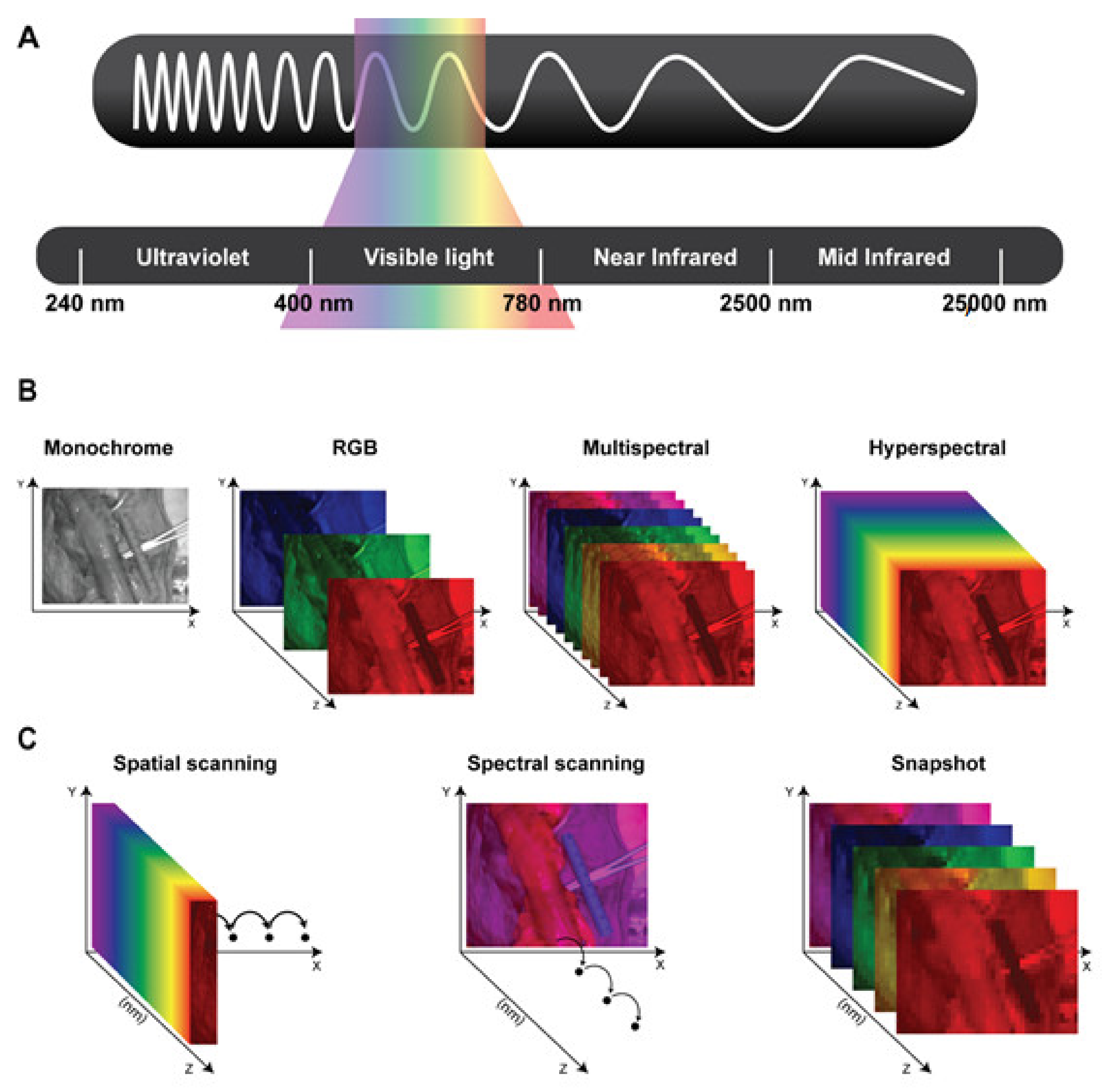
2.1. The Electromagnetic Spectrum and the Hypercube
2.2. Types of Hyperspectral Imaging Hardware
3. Hyperspectral Imaging as an Intraoperative Imaging Tool
4. Tissue Recognition
4.1. Cancer Recognition
4.2. Recognition of Anatomical Structures
4.3. Thermal Ablation Efficacy Recognition
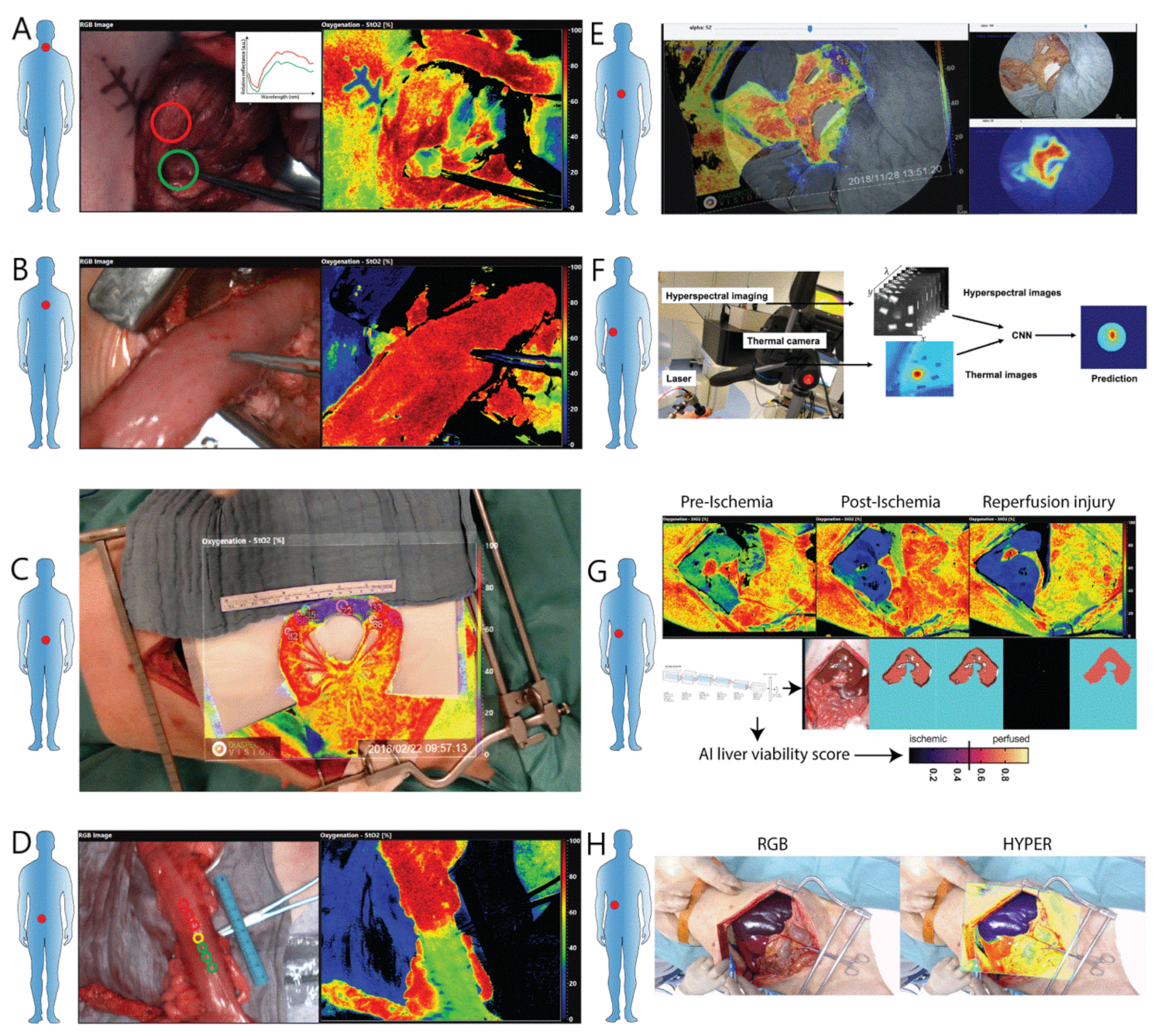
5. Perfusion Assessment
5.1. Perfusion Assessment in Colorectal Surgery
5.2. Perfusion Assessment in Upper Gastrointestinal Surgery
5.3. Perfusion Assessment in Hepatopancreaticobiliary Surgery (HPB)
5.4. Perfusion Assessment in Reconstructive Surgery
5.5. Perfusion Assessment in Urology
5.6. Perfusion Assessment in Neurosurgery
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Collins, F.S.; Varmus, H. A new initiative on precision medicine. N. Engl. J. Med. 2015, 372, 793–795. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mascagni, P.; Longo, F.; Barberio, M.; Seeliger, B.; Agnus, V.; Saccomandi, P.; Hostettler, A.; Marescaux, J.; Diana, M. New intraoperative imaging technologies: Innovating the surgeon’s eye toward surgical precision. J. Surg. Oncol. 2018, 118, 265–282. [Google Scholar] [CrossRef] [PubMed]
- Sarantopoulos, A.; Beziere, N.; Ntziachristos, V. Optical and opto-acoustic interventional imaging. Ann. Biomed. Eng. 2012, 40, 346–366. [Google Scholar] [CrossRef]
- Boppart, S.A.; Brown, J.Q.; Farah, C.S.; Kho, E.; Marcu, L.; Saunders, C.M.; Sterenborg, H.J. Label-free optical imaging technologies for rapid translation and use during intraoperative surgical and tumor margin assessment. J. Biomed. Opt. 2017, 23, 021104. [Google Scholar] [CrossRef] [Green Version]
- Goetz, A.F. Three decades of hyperspectral remote sensing of the Earth: A personal view. Remote Sens. Environ. 2009, 113, S5–S16. [Google Scholar] [CrossRef]
- Feng, Y.-Z.; Sun, D.-W. Application of hyperspectral imaging in food safety inspection and control: A review. Crit. Rev. Food Sci. Nutr. 2012, 52, 1039–1058. [Google Scholar] [CrossRef] [PubMed]
- Govender, M.; Chetty, K.; Bulcock, H. A review of hyperspectral remote sensing and its application in vegetation and water resource studies. Water SA 2007, 33, 145–151. [Google Scholar] [CrossRef] [Green Version]
- Kuula, J.; Pölönen, I.; Puupponen, H.-H.; Selander, T.; Reinikainen, T.; Kalenius, T.; Saari, H. Using VIS/NIR and IR spectral cameras for detecting and separating crime scene details. In Proceedings of the Sensors and Command, Control, Communications, and Intelligence (C3I) Technologies for Homeland Security and Homeland Defense XI, SPIE Defense, Security and Sensing, Baltimore, MD, USA, 23–27 April 2012; p. 83590P. [Google Scholar]
- Tatzer, P.; Wolf, M.; Panner, T. Industrial application for inline material sorting using hyperspectral imaging in the NIR range. Real-Time Imaging 2005, 11, 99–107. [Google Scholar] [CrossRef]
- Cucci, C.; Delaney, J.K.; Picollo, M. Reflectance hyperspectral imaging for investigation of works of art: Old master paintings and illuminated manuscripts. Acc. Chem. Res. 2016, 49, 2070–2079. [Google Scholar] [CrossRef] [PubMed]
- Jacques, S.L. Optical properties of biological tissues: A review. Phys. Med. Biol. 2013, 58, R37. [Google Scholar] [CrossRef] [PubMed]
- Lu, G.; Fei, B. Medical hyperspectral imaging: A review. J. Biomed. Opt. 2014, 19, 010901. [Google Scholar] [CrossRef] [PubMed]
- Calin, M.A.; Parasca, S.V.; Savastru, D.; Manea, D. Hyperspectral imaging in the medical field: Present and future. Appl. Spectrosc. Rev. 2014, 49, 435–447. [Google Scholar] [CrossRef]
- Ortega, S.; Fabelo, H.; Iakovidis, D.K.; Koulaouzidis, A.; Callico, G.M. Use of hyperspectral/multispectral imaging in gastroenterology. Shedding some–different–light into the dark. J. Clin. Med. 2019, 8, 36. [Google Scholar] [CrossRef] [Green Version]
- Clancy, N.T.; Jones, G.; Maier-Hein, L.; Elson, D.S.; Stoyanov, D. Surgical spectral imaging. Med. Image Anal. 2020, 63, 101699. [Google Scholar] [CrossRef] [PubMed]
- Shapey, J.; Xie, Y.; Nabavi, E.; Bradford, R.; Saeed, S.R.; Ourselin, S.; Vercauteren, T. Intraoperative multispectral and hyperspectral label-free imaging: A systematic review of in vivo clinical studies. J. Biophotonics 2019, 12, e201800455. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ghamisi, P.; Plaza, J.; Chen, Y.; Li, J.; Plaza, A.J. Advanced spectral classifiers for hyperspectral images: A review. IEEE Geosci. Remote. Sens. Mag. 2017, 5, 8–32. [Google Scholar] [CrossRef] [Green Version]
- Köhler, H.; Kulcke, A.; Maktabi, M.; Moulla, Y.; Jansen-Winkeln, B.; Barberio, M.; Diana, M.; Gockel, I.; Neumuth, T.; Chalopin, C. Laparoscopic system for simultaneous high-resolution video and rapid hyperspectral imaging in the visible and near-infrared spectral range. J. Biomed. Opt. 2020, 25, 086004. [Google Scholar] [CrossRef] [PubMed]
- Pichette, J.; Charle, W.; Lambrechts, A. Fast and compact internal scanning CMOS-based hyperspectral camera: The Snapscan. In Proceedings of the Photonic Instrumentation Engineering IV, San Francisco, CA, USA, 28 January–2 February 2017; p. 1011014. [Google Scholar]
- Kulcke, A.; Holmer, A.; Wahl, P.; Siemers, F.; Wild, T.; Daeschlein, G. A compact hyperspectral camera for measurement of perfusion parameters in medicine. Biomed. Eng./Biomed. Tech. 2018, 63, 519–527. [Google Scholar] [CrossRef]
- Sorg, B.S.; Moeller, B.J.; Donovan, O.; Cao, Y.; Dewhirst, M.W. Hyperspectral imaging of hemoglobin saturation in tumor microvasculature and tumor hypoxia development. J. Biomed. Opt. 2005, 10, 044004. [Google Scholar] [CrossRef] [PubMed]
- Zuzak, K.J.; Naik, S.C.; Alexandrakis, G.; Hawkins, D.; Behbehani, K.; Livingston, E.H. Characterization of a near-infrared laparoscopic hyperspectral imaging system for minimally invasive surgery. Anal. Chem. 2007, 79, 4709–4715. [Google Scholar] [CrossRef]
- Han, Z.; Zhang, A.; Wang, X.; Sun, Z.; Wang, M.D.; Xie, T. In vivo use of hyperspectral imaging to develop a noncontact endoscopic diagnosis support system for malignant colorectal tumors. J. Biomed. Opt. 2016, 21, 016001. [Google Scholar] [CrossRef] [Green Version]
- Fabelo, H.; Ortega, S.; Lazcano, R.; Madroñal, D.; Callicó, G.; Juárez, E.; Salvador, R.; Bulters, D.; Bulstrode, H.; Szolna, A. An intraoperative visualization system using hyperspectral imaging to aid in brain tumor delineation. Sensors 2018, 18, 430. [Google Scholar] [CrossRef] [Green Version]
- Fabelo, H.; Halicek, M.; Ortega, S.; Shahedi, M.; Szolna, A.; Piñeiro, J.F.; Sosa, C.; O’Shanahan, A.J.; Bisshopp, S.; Espino, C.; et al. Deep learning-based framework for in vivo identification of glioblastoma tumor using hyperspectral images of human brain. Sensors 2019, 19, 920. [Google Scholar] [CrossRef] [Green Version]
- Martinez, B.; Leon, R.; Fabelo, H.; Ortega, S.; Piñeiro, J.F.; Szolna, A.; Hernandez, M.; Espino, C.; O’Shanahan, A.; Carrera, D.; et al. Most relevant spectral bands identification for brain cancer detection using hyperspectral imaging. Sensors 2019, 19, 5481. [Google Scholar] [CrossRef] [Green Version]
- Jansen-Winkeln, B.; Barberio, M.; Chalopin, C.; Schierle, K.; Diana, M.; Köhler, H.; Gockel, I.; Maktabi, M. Feedforward artificial neural network-based colorectal Cancer detection using Hyperspectral imaging: A step towards automatic optical biopsy. Cancers 2021, 13, 967. [Google Scholar] [CrossRef]
- Collins, T.; Maktabi, M.; Barberio, M.; Bencteux, V.; Jansen-Winkeln, B.; Chalopin, C.; Marescaux, J.; Hostettler, A.; Diana, M.; Gockel, I. Automatic Recognition of Colon and Esophagogastric Cancer with Machine Learning and Hyperspectral Imaging. Diagnostics 2021, 11, 1810. [Google Scholar] [CrossRef] [PubMed]
- Zuzak, K.J.; Naik, S.C.; Alexandrakis, G.; Hawkins, D.; Behbehani, K.; Livingston, E. Intraoperative bile duct visualization using near-infrared hyperspectral video imaging. Am. J. Surg. 2008, 195, 491–497. [Google Scholar] [CrossRef] [PubMed]
- Nouri, D.; Lucas, Y.; Treuillet, S. Hyperspectral interventional imaging for enhanced tissue visualization and discrimination combining band selection methods. Int. J. Comput. Assist. Radiol. Surg. 2016, 11, 2185–2197. [Google Scholar] [CrossRef] [PubMed]
- Wisotzky, E.L.; Uecker, F.C.; Arens, P.; Dommerich, S.; Hilsmann, A.; Eisert, P. Intraoperative hyperspectral determination of human tissue properties. J. Biomed. Opt. 2018, 23, 091409. [Google Scholar] [CrossRef] [Green Version]
- Barberio, M.; Maktabi, M.; Gockel, I.; Rayes, N.; Jansen-Winkeln, B.; Köhler, H.; Rabe, S.M.; Seidemann, L.; Takoh, J.P.; Diana, M.; et al. Hyperspectral based discrimination of thyroid and parathyroid during surgery. Curr. Dir. Biomed. Eng. 2018, 4, 399–402. [Google Scholar] [CrossRef]
- Maktabi, M.; Köhler, H.; Ivanova, M.; Neumuth, T.; Rayes, N.; Seidemann, L.; Sucher, R.; Jansen-Winkeln, B.; Gockel, I.; Barberio, M. Classification of hyperspectral endocrine tissue images using support vector machines. Int. J. Med Robot. Comput. Assist. Surg. 2020, 16, 1–10. [Google Scholar] [CrossRef]
- Barberio, M.; Collins, T.; Bencteux, V.; Nkusi, R.; Felli, E.; Viola, M.G.; Marescaux, J.; Hostettler, A.; Diana, M. Deep Learning Analysis of In Vivo Hyperspectral Images for Automated Intraoperative Nerve Detection. Diagnostics 2021, 11, 1508. [Google Scholar] [CrossRef]
- De Landro, M.; Saccomandi, P.; Barberio, M.; Schena, E.; Marescaux, M.; Diana, M. Hyperspectral imaging for thermal effect monitoring in in vivo liver during laser ablation. In Proceedings of the 2019 41st Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Berlin, Germany, 23–27 July 2019; pp. 1851–1854. [Google Scholar]
- De Landro, M.; Espíritu García-Molina, I.; Barberio, M.; Felli, E.; Agnus, V.; Pizzicannella, M.; Diana, M.; Zappa, E.; Saccomandi, P. Hyperspectral Imagery for Assessing Laser-Induced Thermal State Change in Liver. Sensors 2021, 21, 643. [Google Scholar] [CrossRef] [PubMed]
- Akbari, H.; Kosugi, Y.; Kojima, K.; Tanaka, N. Detection and analysis of the intestinal ischemia using visible and invisible hyperspectral imaging. IEEE Trans. Biomed. Eng. 2010, 57, 2011–2017. [Google Scholar] [CrossRef] [PubMed]
- Barberio, M.; Longo, F.; Fiorillo, C.; Seeliger, B.; Mascagni, P.; Agnus, V.; Lindner, V.; Geny, B.; Charles, A.-L.; Gockel, I. HYPerspectral Enhanced Reality (HYPER): A physiology-based surgical guidance tool. Surg. Endosc. 2019, 34, 1736–1744. [Google Scholar] [CrossRef] [PubMed]
- Jansen-Winkeln, B.; Holfert, N.; Köhler, H.; Moulla, Y.; Takoh, J.; Rabe, S.; Mehdorn, M.; Barberio, M.; Chalopin, C.; Neumuth, T.; et al. Determination of the transection margin during colorectal resection with hyperspectral imaging (HSI). Int. J. Colorectal Dis. 2019, 34, 731–739. [Google Scholar] [CrossRef] [PubMed]
- Jansen-Winkeln, B.; Germann, I.; Köhler, H.; Mehdorn, M.; Maktabi, M.; Sucher, R.; Barberio, M.; Chalopin, C.; Diana, M.; Moulla, Y.; et al. Comparison of hyperspectral imaging and fluorescence angiography for the determination of the transection margin in colorectal resections—A comparative study. Int. J. Colorectal Dis. 2021, 36, 283–291. [Google Scholar] [CrossRef] [PubMed]
- Mehdorn, M.; Köhler, H.; Rabe, S.M.; Niebisch, S.; Lyros, O.; Chalopin, C.; Gockel, I.; Jansen-Winkeln, B. Hyperspectral imaging (HSI) in acute mesenteric ischemia to detect intestinal perfusion deficits. J. Surg. Res. 2020, 254, 7–15. [Google Scholar] [CrossRef]
- Barberio, M.; Felli, E.; Pizzicannella, M.; Agnus, V.; Al-Taher, M.; Seyller, E.; Moulla, Y.; Jansen-Winkeln, B.; Gockel, I.; Marescaux, J. Quantitative serosal and mucosal optical imaging perfusion assessment in gastric conduits for esophageal surgery: An experimental study in enhanced reality. Surg. Endosc. 2020, 35, 5827–5835. [Google Scholar] [CrossRef]
- Barberio, M.; Felli, E.; Pop, R.; Pizzicannella, M.; Geny, B.; Lindner, V.; Baiocchini, A.; Jansen-Winkeln, B.; Moulla, Y.; Agnus, V.; et al. A novel technique to improve anastomotic perfusion prior to esophageal surgery: Hybrid ischemic preconditioning of the stomach. Preclinical efficacy proof in a porcine survival model. Cancers 2020, 12, 2977. [Google Scholar] [CrossRef]
- Köhler, H.; Jansen-Winkeln, B.; Maktabi, M.; Barberio, M.; Takoh, J.; Holfert, N.; Moulla, Y.; Niebisch, S.; Diana, M.; Neumuth, T.; et al. Evaluation of hyperspectral imaging (HSI) for the measurement of ischemic conditioning effects of the gastric conduit during esophagectomy. Surg. Endosc. 2019, 33, 3775–3782. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moulla, Y.; Buchloh, D.C.; Köhler, H.; Rademacher, S.; Denecke, T.; Meyer, H.-J.; Mehdorn, M.; Lange, U.G.; Sucher, R.; Seehofer, D. Hyperspectral Imaging (HSI)—A New Tool to Estimate the Perfusion of Upper Abdominal Organs during Pancreatoduodenectomy. Cancers 2021, 13, 2846. [Google Scholar] [CrossRef] [PubMed]
- Wakabayashi, T.; Barberio, M.; Urade, T.; Pop, R.; Seyller, E.; Pizzicannella, M.; Mascagni, P.; Charles, A.-L.; Abe, Y.; Geny, B. Intraoperative perfusion assessment in enhanced reality using quantitative optical imaging: An experimental study in a pancreatic partial ischemia model. Diagnostics 2021, 11, 93. [Google Scholar] [CrossRef] [PubMed]
- Felli, E.; Al-Taher, M.; Collins, T.; Baiocchini, A.; Felli, E.; Barberio, M.; Ettorre, G.M.; Mutter, D.; Lindner, V.; Hostettler, A.; et al. Hyperspectral evaluation of hepatic oxygenation in a model of total vs. arterial liver ischaemia. Sci. Rep. 2020, 10, 15441. [Google Scholar] [CrossRef] [PubMed]
- Felli, E.; Al-Taher, M.; Collins, T.; Nkusi, R.; Felli, E.; Baiocchini, A.; Lindner, V.; Vincent, C.; Barberio, M.; Geny, B.; et al. Automatic Liver Viability Scoring with Deep Learning and Hyperspectral Imaging. Diagnostics 2021, 11, 1527. [Google Scholar] [CrossRef] [PubMed]
- Urade, T.; Felli, E.; Barberio, M.; Al-Taher, M.; Felli, E.; Goffin, L.; Agnus, V.; Ettorre, G.M.; Marescaux, J.; Mutter, D. Hyperspectral enhanced reality (HYPER) for anatomical liver resection. Surg. Endosc. 2021, 35, 1844–1850. [Google Scholar] [CrossRef]
- Kohler, L.H.; Köhler, H.; Kohler, S.; Langer, S.; Nuwayhid, R.; Gockel, I.; Spindler, N.; Osterhoff, G. Hyperspectral Imaging (HSI) as a new diagnostic tool in free flap monitoring for soft tissue reconstruction: A proof of concept study. BMC Surg. 2021, 21, 222. [Google Scholar] [CrossRef]
- Thiem, D.G.; Frick, R.W.; Goetze, E.; Gielisch, M.; Al-Nawas, B.; Kämmerer, P.W. Hyperspectral analysis for perioperative perfusion monitoring—A clinical feasibility study on free and pedicled flaps. Clin. Oral Investig. 2021, 25, 933–945. [Google Scholar] [CrossRef]
- Tracy, C.R.; Terrell, J.D.; Francis, R.P.; Wehner, E.F.; Smith, J.; Litorja, M.; Hawkins, D.L.; Pearle, M.S.; Cadeddu, J.A.; Zuzak, K.J. Characterization of renal ischemia using DLP hyperspectral imaging: A pilot study comparing artery-only occlusion versus artery and vein occlusion. J. Endourol. 2010, 24, 321–325. [Google Scholar] [CrossRef]
- Best, S.L.; Thapa, A.; Holzer, M.J.; Jackson, N.; Mir, S.A.; Cadeddu, J.A.; Zuzak, K.J. Minimal arterial in-flow protects renal oxygenation and function during porcine partial nephrectomy: Confirmation by hyperspectral imaging. Urology 2011, 78, 961–966. [Google Scholar] [CrossRef]
- Holzer, M.S.; Best, S.L.; Jackson, N.; Thapa, A.; Raj, G.V.; Cadeddu, J.A.; Zuzak, K.J. Assessment of renal oxygenation during partial nephrectomy using hyperspectral imaging. J. Urol. 2011, 186, 400–404. [Google Scholar] [CrossRef]
- Best, S.L.; Thapa, A.; Jackson, N.; Olweny, E.; Holzer, M.; Park, S.; Wehner, E.; Zuzak, K.; Cadeddu, J.A. Renal oxygenation measurement during partial nephrectomy using hyperspectral imaging may predict acute postoperative renal function. J. Endourol. 2013, 27, 1037–1040. [Google Scholar] [CrossRef]
- Liu, Z.-W.; Faddegon, S.; Olweny, E.O.; Best, S.L.; Jackson, N.; Raj, G.V.; Zuzak, K.J.; Cadeddu, J.A. Renal oxygenation during partial nephrectomy: A comparison between artery-only occlusion versus artery and vein occlusion. J. Endourol. 2013, 27, 470–474. [Google Scholar] [CrossRef] [PubMed]
- Sucher, R.; Wagner, T.; Köhler, H.; Sucher, E.; Guice, H.; Recknagel, S.; Lederer, A.; Hau, H.M.; Rademacher, S.; Schneeberger, S.; et al. Hyperspectral Imaging (HSI) of Human Kidney Allografts. Ann. Surg. 2020. [Google Scholar] [CrossRef] [PubMed]
- Mori, M.; Chiba, T.; Nakamizo, A.; Kumashiro, R.; Murata, M.; Akahoshi, T.; Tomikawa, M.; Kikkawa, Y.; Yoshimoto, K.; Mizoguchi, M.; et al. Intraoperative visualization of cerebral oxygenation using hyperspectral image data: A two-dimensional mapping method. Int. J. Comput. Assist. Radiol. Surg. 2014, 9, 1059–1072. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cahill, R.; O’shea, D.; Khan, M.; Khokhar, H.; Epperlein, J.; Mac Aonghusa, P.; Nair, R.; Zhuk, S. Artificial intelligence indocyanine green (ICG) perfusion for colorectal cancer intra-operative tissue classification. Br. J. Surg. 2021, 108, 5–9. [Google Scholar] [CrossRef] [PubMed]
- Van Dam, G.M.; Themelis, G.; Crane, L.M.; Harlaar, N.J.; Pleijhuis, R.G.; Kelder, W.; Sarantopoulos, A.; De Jong, J.S.; Arts, H.J.; Van Der Zee, A.G.; et al. Intraoperative tumor-specific fluorescence imaging in ovarian cancer by folate receptor-α targeting: First in-human results. Nat. Med. 2011, 17, 1315–1319. [Google Scholar] [CrossRef]
- Ishizawa, T.; Bandai, Y.; Ijichi, M.; Kaneko, J.; Hasegawa, K.; Kokudo, N. Fluorescent cholangiography illuminating the biliary tree during laparoscopic cholecystectomy. J. Br. Surg. 2010, 97, 1369–1377. [Google Scholar] [CrossRef] [PubMed]
- Van den Bos, J.; Al-Taher, M.; Bouvy, N.D.; Stassen, L.P. Near-infrared fluorescence laparoscopy of the ureter with three preclinical dyes in a pig model. Surg. Endosc. 2019, 33, 986–991. [Google Scholar] [CrossRef] [PubMed]
- Al-Taher, M.; Barberio, M.; Felli, E.; Agnus, V.; Ashoka, A.H.; Gioux, S.; Klymchenko, A.; Bouvy, N.; Stassen, L.; Marescaux, J.; et al. Simultaneous multipurpose fluorescence imaging with IRDye® 800BK during laparoscopic surgery. Surg. Endosc. 2020, 35, 4840–4848. [Google Scholar] [CrossRef]
- Ashoka, A.H.; Kong, S.-H.; Seeliger, B.; Andreiuk, B.; Soares, R.V.; Barberio, M.; Diana, M.; Klymchenko, A.S. Near-infrared fluorescent coatings of medical devices for image-guided surgery. Biomaterials 2020, 261, 120306. [Google Scholar] [CrossRef] [PubMed]
- Barberio, M.; Al-Taher, M.; Forgione, A.; Hoskere Ashoka, A.; Felli, E.; Agnus, V.; Marescaux, J.; Klymchenko, A.; Diana, M. A novel method for near-infrared fluorescence imaging of the urethra during perineal and transanal surgery: Demonstration in a cadaveric model. Colorectal Dis. 2020, 22, 1749–1753. [Google Scholar] [CrossRef] [PubMed]
- Barberio, M.; Al-Taher, M.; Felli, E.; Ashoka, A.H.; Marescaux, J.; Klymchenko, A.; Diana, M. Intraoperative ureter identification with a novel fluorescent catheter. Sci. Rep. 2021, 11, 4501. [Google Scholar] [CrossRef] [PubMed]
- Halicek, M.; Fabelo, H.; Ortega, S.; Callico, G.M.; Fei, B. In-vivo and ex-vivo tissue analysis through hyperspectral imaging techniques: Revealing the invisible features of cancer. Cancers 2019, 11, 756. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Di Costanzo, G.G.; Francica, G.; Pacella, C.M. Laser ablation for small hepatocellular carcinoma: State of the art and future perspectives. World J. Hepatol. 2014, 6, 704. [Google Scholar] [CrossRef]
- Karliczek, A.; Harlaar, N.; Zeebregts, C.; Wiggers, T.; Baas, P.; Van Dam, G. Surgeons lack predictive accuracy for anastomotic leakage in gastrointestinal surgery. Int. J. Colorectal Dis. 2009, 24, 569–576. [Google Scholar] [CrossRef] [Green Version]
- Degett, T.H.; Andersen, H.S.; Gögenur, I. Indocyanine green fluorescence angiography for intraoperative assessment of gastrointestinal anastomotic perfusion: A systematic review of clinical trials. Langenbeck’s Arch. Surg. 2016, 401, 767–775. [Google Scholar] [CrossRef] [PubMed]
- Hope-Ross, M.; Yannuzzi, L.A.; Gragoudas, E.S.; Guyer, D.R.; Slakter, J.S.; Sorenson, J.A.; Krupsky, S.; Orlock, D.A.; Puliafito, C.A. Adverse reactions due to indocyanine green. Ophthalmology 1994, 101, 529–533. [Google Scholar] [CrossRef]
- Blanco-Colino, R.; Espin-Basany, E. Intraoperative use of ICG fluorescence imaging to reduce the risk of anastomotic leakage in colorectal surgery: A systematic review and meta-analysis. Tech. Coloproctol. 2018, 22, 15–23. [Google Scholar] [CrossRef]
- Osterkamp, J.; Strandby, R.; Nerup, N.; Svendsen, M.; Svendsen, L.; Achiam, M. Quantitative fluorescence angiography detects dynamic changes in gastric perfusion. Surg. Endosc. 2020, 1–10. [Google Scholar] [CrossRef]
- D’Urso, A.; Agnus, V.; Barberio, M.; Seeliger, B.; Marchegiani, F.; Charles, A.-L.; Geny, B.; Marescaux, J.; Mutter, D.; Diana, M. Computer-assisted quantification and visualization of bowel perfusion using fluorescence-based enhanced reality in left-sided colonic resections. Surg. Endosc. 2020, 35, 4321–4331. [Google Scholar] [CrossRef]
- Barberio, M.; Felli, E.; Seyller, E.; Longo, F.; Chand, M.; Gockel, I.; Geny, B.; Swanström, L.; Marescaux, J.; Agnus, V. Quantitative fluorescence angiography versus hyperspectral imaging to assess bowel ischemia: A comparative study in enhanced reality. Surgery 2020, 168, 178–184. [Google Scholar] [CrossRef]
- Diana, M.; Noll, E.; Diemunsch, P.; Dallemagne, B.; Benahmed, M.A.; Agnus, V.; Soler, L.; Barry, B.; Namer, I.J.; Demartines, N. Enhanced-reality video fluorescence: A real-time assessment of intestinal viability. Ann. Surg. 2014, 259, 700–707. [Google Scholar] [CrossRef]
- Diana, M.; Agnus, V.; Halvax, P.; Liu, Y.-Y.; Dallemagne, B.; Schlagowski, A.-I.; Geny, B.; Diemunsch, P.; Lindner, V.; Marescaux, J. Intraoperative fluorescence-based enhanced reality laparoscopic real-time imaging to assess bowel perfusion at the anastomotic site in an experimental model. J. Br. Surg. 2015, 102, e169–e176. [Google Scholar] [CrossRef] [PubMed]
- Diana, M.; Halvax, P.; Dallemagne, B.; Nagao, Y.; Diemunsch, P.; Charles, A.-L.; Agnus, V.; Soler, L.; Demartines, N.; Lindner, V.; et al. Real-time navigation by fluorescence-based enhanced reality for precise estimation of future anastomotic site in digestive surgery. Surg. Endosc. 2014, 28, 3108–3118. [Google Scholar] [CrossRef] [PubMed]
- Quero, G.; Lapergola, A.; Barberio, M.; Seeliger, B.; Saccomandi, P.; Guerriero, L.; Mutter, D.; Saadi, A.; Worreth, M.; Marescaux, J.; et al. Discrimination between arterial and venous bowel ischemia by computer-assisted analysis of the fluorescent signal. Surg. Endosc. 2019, 33, 1988–1997. [Google Scholar] [CrossRef] [PubMed]
- Low, D.E.; Kuppusamy, M.K.; Alderson, D.; Cecconello, I.; Chang, A.C.; Darling, G.; Davies, A.; D’Journo, X.B.; Gisbertz, S.S.; Griffin, S.M.; et al. Benchmarking complications associated with esophagectomy. Ann. Surg. 2019, 269, 291–298. [Google Scholar] [CrossRef] [Green Version]
- Morse, B.C.; Simpson, J.P.; Jones, Y.R.; Johnson, B.L.; Knott, B.M.; Kotrady, J.A. Determination of independent predictive factors for anastomotic leak: Analysis of 682 intestinal anastomoses. Am. J. Surg. 2013, 206, 950–956. [Google Scholar] [CrossRef] [PubMed]
- Laemmel, E.; Genet, M.; Le Goualher, G.; Perchant, A.; Le Gargasson, J.-F.; Vicaut, E. Fibered confocal fluorescence microscopy (Cell-viZio™) facilitates extended imaging in the field of microcirculation. J. Vasc. Res. 2004, 41, 400–411. [Google Scholar] [CrossRef]
- Diana, M.; Dallemagne, B.; Chung, H.; Nagao, Y.; Halvax, P.; Agnus, V.; Soler, L.; Lindner, V.; Demartines, N.; Diemunsch, P.; et al. Probe-based confocal laser endomicroscopy and fluorescence-based enhanced reality for real-time assessment of intestinal microcirculation in a porcine model of sigmoid ischemia. Surg. Endosc. 2014, 28, 3224–3233. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Smits, F.J.; Molenaar, I.Q.; Besselink, M.G.; Rinkes, I.H.B.; van Eijck, C.H.; Busch, O.R.; van Santvoort, H.C. Early recognition of clinically relevant postoperative pancreatic fistula: A systematic review. HPB 2020, 22, 1–11. [Google Scholar] [CrossRef]
- Strasberg, S.M.; Drebin, J.A.; Mokadam, N.A.; Green, D.W.; Jones, K.L.; Ehlers, J.P.; Linehan, D. Prospective trial of a blood supply-based technique of pancreaticojejunostomy: Effect on anastomotic failure in the Whipple procedure. J. Am. Coll. Surg. 2002, 194, 746–758. [Google Scholar] [CrossRef]
- Ishizawa, T.; Zuker, N.B.; Kokudo, N.; Gayet, B. Positive and negative staining of hepatic segments by use of fluorescent imaging techniques during laparoscopic hepatectomy. Arch. Surg. 2012, 147, 393–394. [Google Scholar] [CrossRef] [PubMed]
- Aoki, T.; Koizumi, T.; Mansour, D.A.; Fujimori, A.; Kusano, T.; Matsuda, K.; Tashiro, Y.; Watanabe, M.; Otsuka, K.; Murakami, M. Ultrasound-guided preoperative positive percutaneous indocyanine green fluorescence staining for laparoscopic anatomical liver resection. J. Am. Coll. Surg. 2020, 230, e7–e12. [Google Scholar] [CrossRef] [PubMed]
- Mizuno, T.; Sheth, R.; Yamamoto, M.; Kang, H.S.C.; Yamashita, S.; Aloia, T.A.; Chun, Y.S.; Lee, J.E.; Vauthey, J.-N.; Conrad, C. Laparoscopic Glissonean pedicle transection (Takasaki) for negative fluorescent counterstaining of segment 6. Ann. Surg. Oncol. 2017, 24, 1046–1047. [Google Scholar] [CrossRef] [PubMed]
- Terasawa, M.; Ishizawa, T.; Mise, Y.; Inoue, Y.; Ito, H.; Takahashi, Y.; Saiura, A. Applications of fusion-fluorescence imaging using indocyanine green in laparoscopic hepatectomy. Surg. Endosc. 2017, 31, 5111–5118. [Google Scholar] [CrossRef] [PubMed]
- Nomi, T.; Hokuto, D.; Yoshikawa, T.; Matsuo, Y.; Sho, M. A novel navigation for laparoscopic anatomic liver resection using indocyanine green fluorescence. Ann. Surg. Oncol. 2018, 25, 3982. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Noordmans, H.J.; Ferrier, C.; De Roode, R.; Leijten, F.; Van Rijen, P.; Gosselaar, P.; Klaessens, J.; Verdaasdonk, R. Imaging the seizure during surgery with a hyperspectral camera. Epilepsia 2013, 54, e150–e154. [Google Scholar] [CrossRef] [Green Version]
- Pichette, J.; Laurence, A.; Angulo, L.; Lesage, F.; Bouthillier, A.; Nguyen, D.K.; Leblond, F. Intraoperative video-rate hemodynamic response assessment in human cortex using snapshot hyperspectral optical imaging. Neurophotonics 2016, 3, 045003. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Application Category | Application/ Subcategory | Target | Subject (n) | Device Type | Acquisition Time | Spatial Resolution | Spectral Range | Reference |
|---|---|---|---|---|---|---|---|---|
| Tissue recognition | Cancer recognition | brain tumor | human (22) | Spatial scanning (two cameras) | 40 + 80 s for both cameras | 1004 × 1787 pixels | 400 to 1700 nm | Fabelo H. et al. 2018 [24] |
| brain tumor | human (16) | spatial scanning | ~1 min | 1004 × 1787 pixels | 400 to 1000 nm | Fabelo H. et al. 2019 [25] | ||
| brain tumor | human (16) | spatial scanning | ND | 1004 × 1787 pixels | 400 to 1000 nm | Martinez I. et al. 2019 [26] | ||
| colorectal cancer | human (54) | spatial scanning | ~6 s | 640 × 480 pixels | 500 to 1000 nm | Jansen-W., B et al. 2021 [27] | ||
| colorectal cancer/esophageal cancer | human (22) | spatial scanning | ~6 s | 640 × 480 pixels | 500 to 1000 nm | Collins T. et al. 2021 [28] | ||
| Anatomical structures recognition | biliary structure | pig (3) | spectral scanning | ~90 s | ND | 650 to 1100 nm | Zuzak, K.J et al. 2008 [29] | |
| ureters, facial nerve | pig (3) | spectral scanning (two cameras) | ND | 1392 × 1040 pixels and 640× 12 pixels | 400–1100 nm and 850–1800 nm | Nouri, D et al. 2016 [30] | ||
| artery, vein, bone, muscle, fat, connective tissue, parotid gland, and nerve | human (6) | spectral scanning | ND | 1920 × 1080 pixels | 380 to 1100 nm | Wisotzky, L. et al. 2018 [31] | ||
| parathyroid, thyroid, and recurrent laryngeal nerve recognition | human (7) | spatial scanning | ~6 s | 640 × 480 pixels | 500 to 1000 nm | Barberio, M. et al. 2018 [32] | ||
| parathyroid, thyroid, and recurrent laryngeal nerve recognition | human (9) | spatial scanning | ~6 s | 640 × 480 pixels | 500 to 1000 nm | Maktabi, M. et al. 2020 [33] | ||
| artery, vein, nerve, muscle, fat, skin | pigs (8) | spatial scanning | ~6 s | 640 × 480 pixels | 500 to 1000 nm | Barberio, M. et al. 2021 [34] | ||
| Thermal ablation efficacy recognition | thermal effect monitoring during hepatic laser ablation | pig (1) | spatial scanning | ~6 s | 640×480 pixels | 500 to 1000nm | De Landro, M et al. 2019 [35] | |
| thermal effect monitoring during hepatic laser ablation | pig (1) | spatial scanning | ~6 s | 640 × 480 pixels | 50 to 1000 nm | De Landro, M et al. 2021 [36] | ||
| Perfusion assessment | Colorectal surgery | small bowel perfusion | pig (1) | spatial scanning (two devices) | ND | 484 × 700 and 240 × 420 pixels | 400–1000 and 900-1700 nm | Akbari, H. et al. 2010 [37] |
| small bowel perfusion | pig (6) | spatial scanning | ~6 s | 640 × 480 pixels | 500 to 1000 nm | Barberio, M. et al. 2019 [38] | ||
| colonic perfusion | human (24) | spatial scanning | ~6 s | 640 × 480 pixels | 500 to 1000 nm | Jansen-W., B et al. 2019 [39] | ||
| colonic perfusion | human (32) | spatial scanning | ~6 s | 640 × 480 pixels | 500 to 1000 nm | Jansen-W., B et al. 2021 [40] | ||
| acute mesenteric ischemia | human (11) | spatial scanning | ~6 s | 640 × 480 pixels | 500 to 1000 nm | Mehdorn, M. et al. 2020 [41] | ||
| Upper-gastrointestinal surgery | gastric conduit perfusion | pig (5) | spatial scanning | ~6 s | 640 × 480 pixels | 500 to 1000 nm | Barberio M. et al. 2020 [42] | |
| gastric conduit perfusion | pig (17) | spatial scanning | ~6 s | 640 × 480 pixels | 500 to 1000 nm | Barberio M. et al. 2020 [43] | ||
| gastric conduit perfusion | human (22) | spatial scanning | ~6 s | 640 × 480 pixels | 500 to 1000 nm | Köhler, H. et al. 2019 [44] | ||
| perfusion of upper abdominal organs | human (20) | spatial scanning | ~6 s | 640 × 480 pixels | 500 to 1000 nm | Moulla, Y. et al. 2021 [45] | ||
| Hepatopancreaticobiliary surgery | pancreatic perfusion | pig (6) | spatial scanning | ~6 s | 640 × 480 pixels | 500 to 1000 nm | Wakabayashi, T, et al. 2021 [46] | |
| hepatic ischemia differentiation | pig (6) | spatial scanning | ~6 s | 640 × 480 pixels | 500 to 1000 nm | Felli, E. et al. 2020 [47] | ||
| hepatic ischemia/reperfusion injury | pig (5) | Spatial scanning | ~6 s | 640 × 480 pixels | 500 to1000 nm | Felli, E. et al. 2021 [48] | ||
| hepatic resection guidance | porcine (3) | spatial scanning | ~6 s | 640 × 480 pixels | 500 to 1000 nm | Urade, T. et al. 2021 [49] | ||
| Reconstructive surgery | flap perfusion | human (22) | spatial scanning | ~6 s | 640 × 480 pixels | 500 to 1000 nm | Kohler, L.H. et al. 2021 [50] | |
| perfusion of free and pedicled flap | human (30) | spatial scanning | ~6 s | 640 × 480 pixels | 500 to 1000 nm | Thiem, D.G et al. 2021 [51] | ||
| Urology | renal perfusion | pig (7) | spectral scanning | <30 s | ND | 520 to 645 nm | Tracy, C.R. et al. 2010 [52] | |
| renal perfusion (partial nephrectomies) | pig (14) | spectral scanning | <30 s | ND | 520 to 645 nm | Best, S.L. et al. 2011 [53] | ||
| renal perfusion (partial nephrectomies) | human (21) | spectral scanning | <30 s | ND | 520 to 645 nm | Holzer, M.S. et al. 2011 [54] | ||
| renal perfusion (partial nephrectomies) | human (26) | spectral scanning | <30 s | ND | 520 to 645 nm | Best, S.L. et al. 2013 [55] | ||
| renal perfusion (partial nephrectomies) | human (37) | spectral scanning | <30 s | ND | 520 to 645 nm | Liu, Z.W. et al. 2013 [56] | ||
| graft perfusion (kidney transplant) | human (17) | spatial scanning | ~6 s | 640 × 480 pixels | 500 to 1000 nm | Sucher, R. et al. 2020 [57] | ||
| Neurosurgery | brain perfusion | human (4) | ND | 5–16 s | 640 × 480 data points (pixels) | 400 to 800 nm | Mori, M. et al. 2014 [58] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barberio, M.; Benedicenti, S.; Pizzicannella, M.; Felli, E.; Collins, T.; Jansen-Winkeln, B.; Marescaux, J.; Viola, M.G.; Diana, M. Intraoperative Guidance Using Hyperspectral Imaging: A Review for Surgeons. Diagnostics 2021, 11, 2066. https://doi.org/10.3390/diagnostics11112066
Barberio M, Benedicenti S, Pizzicannella M, Felli E, Collins T, Jansen-Winkeln B, Marescaux J, Viola MG, Diana M. Intraoperative Guidance Using Hyperspectral Imaging: A Review for Surgeons. Diagnostics. 2021; 11(11):2066. https://doi.org/10.3390/diagnostics11112066
Chicago/Turabian StyleBarberio, Manuel, Sara Benedicenti, Margherita Pizzicannella, Eric Felli, Toby Collins, Boris Jansen-Winkeln, Jacques Marescaux, Massimo Giuseppe Viola, and Michele Diana. 2021. "Intraoperative Guidance Using Hyperspectral Imaging: A Review for Surgeons" Diagnostics 11, no. 11: 2066. https://doi.org/10.3390/diagnostics11112066
APA StyleBarberio, M., Benedicenti, S., Pizzicannella, M., Felli, E., Collins, T., Jansen-Winkeln, B., Marescaux, J., Viola, M. G., & Diana, M. (2021). Intraoperative Guidance Using Hyperspectral Imaging: A Review for Surgeons. Diagnostics, 11(11), 2066. https://doi.org/10.3390/diagnostics11112066











