1. Introduction
Human habitation and disposal of wastes affect the water supply sources and leads to new environmental issues. Metals enter into surface water and groundwater through weathering of rocks [
1,
2,
3] or from human activities including mining, industrial wastes or airborne particulate matter [
4,
5]. Groundwater pollution due to heavy metals where it is used as a source of drinking water [
6,
7,
8] and the possible risk to human health has been studied widely [
9,
10]. This is because certain heavy metals are required for normal functioning of the human body, but exposure to excess concentration of heavy metals may lead to health risks. World Health Organisation (WHO) lists four heavy metals (arsenic, lead, mercury and cadmium) in its list of ten chemicals as a major public health concern [
11]. Arsenic is a much studied heavy metal in groundwater affecting larger parts of Asia [
12,
13,
14,
15] and is mainly geogenic in origin.
Many industrial practices like combustion, extraction and processing use heavy metals for production. Wastewater from these industries will contain heavy metals that pollute soil, surface water and groundwater if proper care is not taken in the disposal [
16,
17]. Tanneries causing chromium pollution due to use of chromium sulphate during the tanning process is a major concern in Bangladesh and India [
18,
19,
20]. Due to poor management, landfills [
21,
22] and municipal wastewater [
23] have been the source of groundwater and soil pollution especially in developing countries. Occurrence of metals in groundwater in areas possessing mineral deposits and associated mining activities are common [
24,
25,
26,
27]. Potential risk of heavy metal contamination due to application of fertilisers was reported by many others [
28,
29,
30].
South Africa is experiencing a water crisis and the problem is more aligned towards water quality than the quantity. Challenges to water shortage and access to water of desirable quality include population increase, economic growth and climate change. Agricultural practices mostly depend on groundwater in many parts of the country apart from public water supply. Almost two-thirds of the country’s population rely on groundwater for domestic needs [
31]. Hence, there is increasing stress to maintain the quality of water so that it does not have any negative impact on human health or flora and fauna. Deterioration of water quality will have wider economic implications on various sectors including agriculture, industry and recreation [
32]. The Department of Water Affairs and Forestry (DWAF) of the Republic of South Africa has reported that effluent discharge from industries had led to salinisation and contamination due to heavy metals in groundwater, but the exact contaminant levels are not available [
32]. There are several studies on heavy metals in groundwater in several parts of the world including some urban areas in South Africa. More studies on regional water quality based on heavy metals are essential to quantify the extent of pollution and plan for possible measures of mitigation. Thus, in this study, water quality assessment was carried out in Richards Bay and Empangeni towns of South Africa. The demand for water in these areas is increasing to satisfy the needs of industries, mining, agriculture and urban water supply. Being a highly industrialised and commercial region of South Africa, there is ample scope to study and improve the groundwater quality of this region, which is considered a highly vulnerable area to pollution [
32].
Studies that involve large datasets or involve assessment of many parameters rely on statistical techniques to interpret and group the common or independent source of the parameters [
33]. Such techniques serve as a rapid and powerful tool for scientists and decision-makers to arrive at a management solution [
34]. This study was carried out with an aim to quantify the heavy metals in groundwater and to evaluate the impact of human-induced activities on groundwater in South Africa using multivariate statistical analysis and human exposure risk analysis through the drinking water pathway.
3. Results
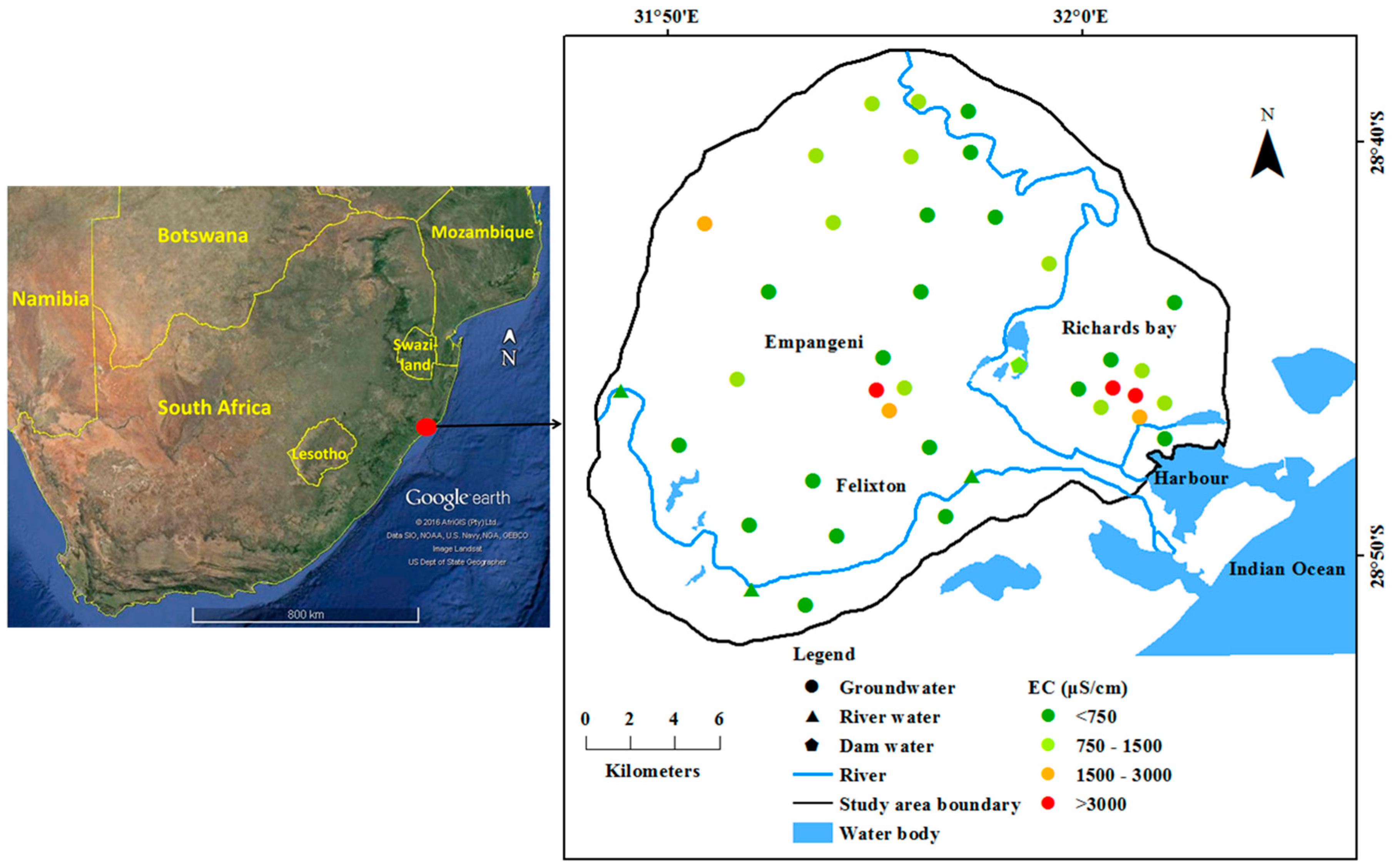
EC is a good indicator of total salinity or total amount of dissolved solids in water. Though it does not provide detailed information about the ionic composition in water, this can be used to determine the suitability of water for drinking and irrigation use. Desirable EC in drinking water is <750 μS/cm and the range between 750 and 1500 μS/cm is permissible. Groundwater quality at eighteen locations was desirable and eleven locations was permissible for drinking (
Figure 1). Three groundwater samples were not permissible i.e., ranging from 1500 to 3000 μS/cm and three samples were hazardous with high EC exceeding above 3000 μS/cm. Surface water samples from the Mhlathuze River have EC < 750 μS/cm. Though this is suitable for drinking, it is not used for a domestic purpose in this area. Water stored in the dam for irrigation use had permissible EC.
TDS also helps to determine the suitability of water for drinking and irrigation. Groundwater at twenty-nine locations was either desirable or permissible for drinking, whereas, at four locations, the water could be used only for irrigation. As a worst case, at two locations, the TDS was higher than 3000 mg/L (same locations as EC because TDS is calculated from EC) and unfit for any intended use [
47]. Groundwater will require treatment if it has to be used for domestic water supply. If this water was used for irrigation, it will increase the salinity of the soil. Prolonged use of high TDS water will turn the soil unsuitable for plant growth and irrigation will have to shift to more salt-resistant crops.
The unsuitable groundwater samples based on TDS were located near the Richards Bay harbour and in the central part near Empangeni. River water samples were within 500 mg/L of TDS and do not pose threat to human health due to consumption. Dam water was also permissible for drinking and not harmful for irrigation (
Table 1). TDS indicates that surface water from the river and the dam are fresh [
48]. Twenty-nine groundwater samples were fresh while five locations with TDS between 1000 and 10,000 mg/L were brackish. One groundwater sample having TDS of 16,806 mg/L is classified as saline (
Table 1). This sample is located close to the active landfill and the dumping of wastes from the Empangeni town have increased the groundwater salinity.
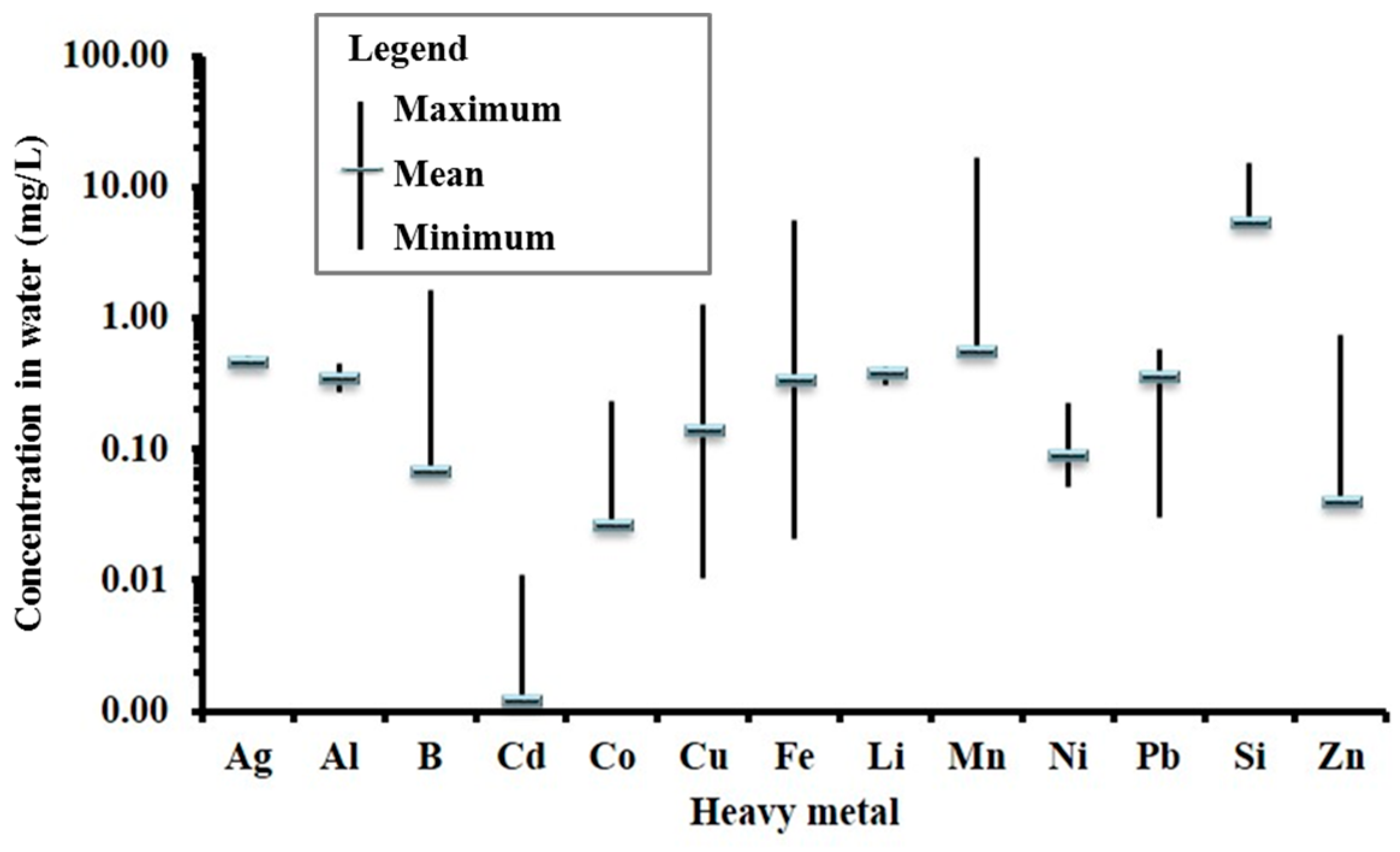
Range of heavy metals measured in the water samples is given in
Figure 2. Concentration of metals in water was compared against the water quality standards for drinking and domestic use. South African water quality guidelines for domestic use proposes a target water quality range, which is described as ‘what is considered good or ideal water quality’. Water quality outside of this range may, under certain circumstances, still be acceptable [
49]. These ranges are available only for eight of the heavy metals studied and, of these, aluminium, cadmium and chromium (VI) have no standard range, but has a no-effect range as the target guideline (
Table 2). Comparison of the target water quality range of South Africa with the health based guidelines by the WHO [
50] are given in
Table 2.
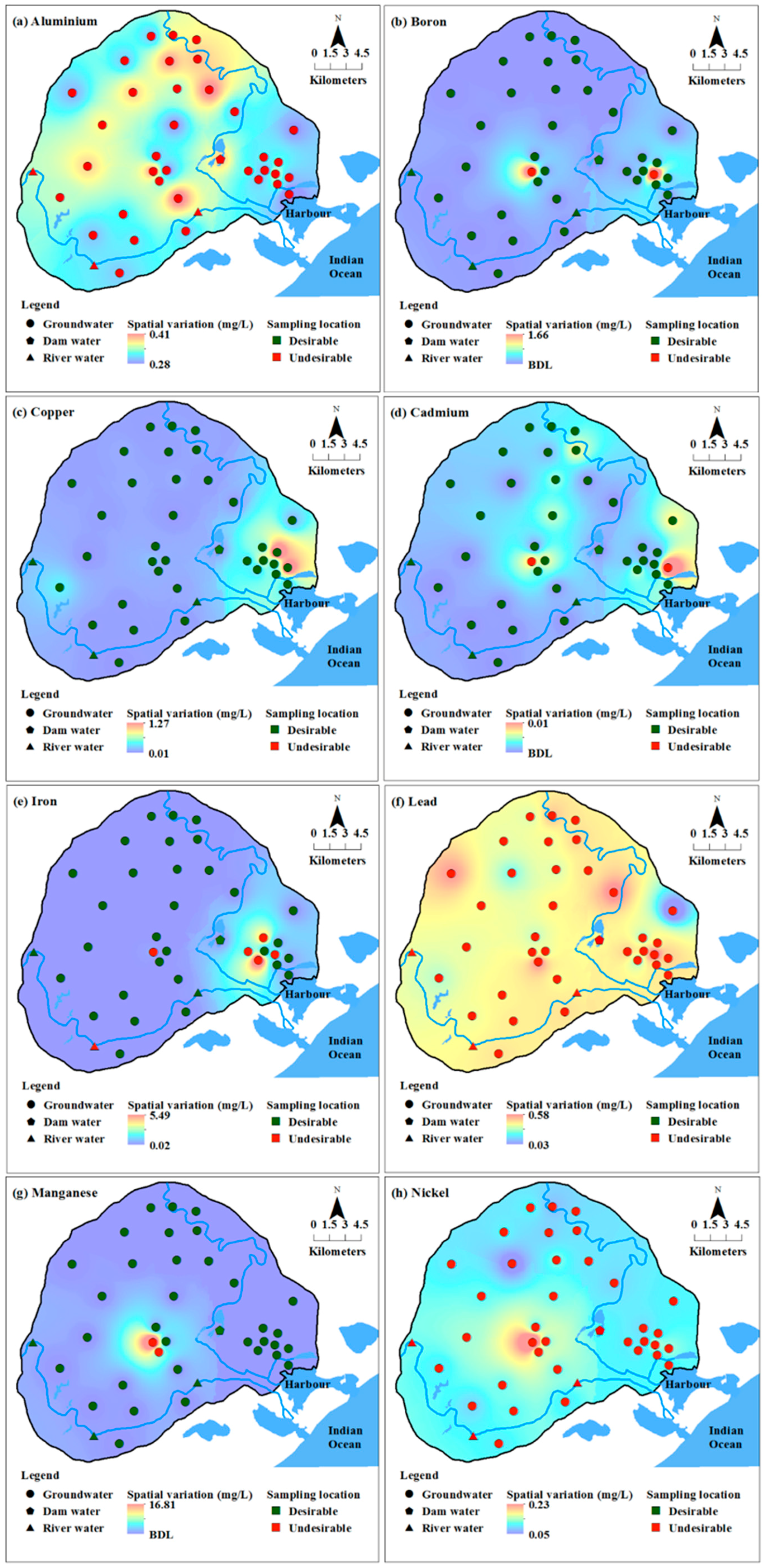
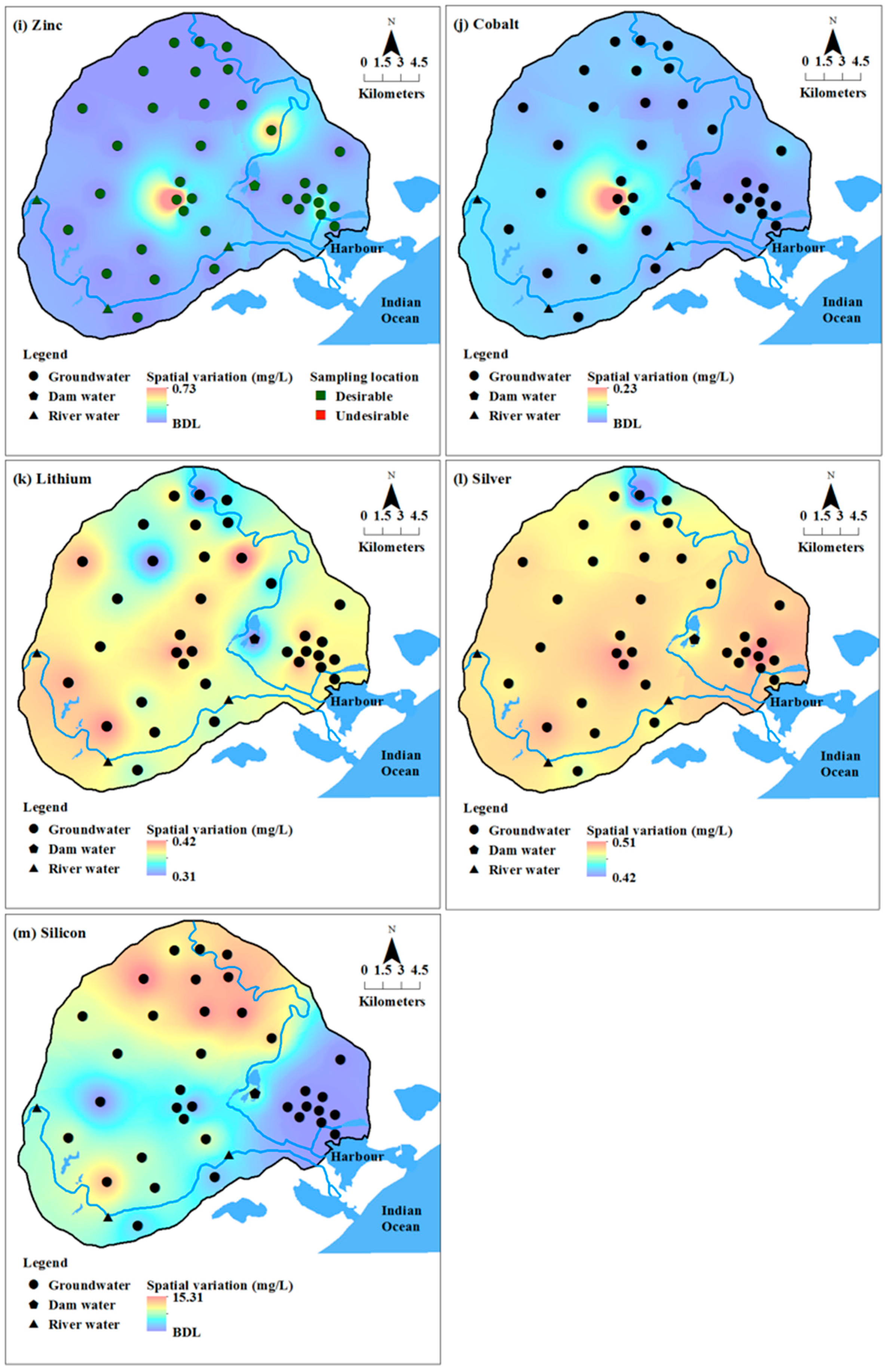
Concentration of chromium, titanium and zirconium were below detection limits (BDL) in all the groundwater samples. In addition to these metals, silicon was also BDL in surface water. Cobalt, lithium, silicon, titanium and zirconium do not have any health-based guidelines or standard limits proposed by various organisations [
49,
51,
52,
53]. Copper and zinc were within WHO limits [
50] in all samples. Aluminium, nickel and lead were above the limits in all the surface and groundwater samples. In groundwater, boron and cadmium exceeded WHO health guidelines in two groundwater sampling locations. Iron exceeds DWAF limits [
49] of 0.1 mg/L in five groundwater and one surface water sample collected (
Figure 3). Manganese was above WHO limits i.e., 0.5 mg/L in two groundwater samples and above DWAF limits i.e., 0.05 in eight groundwater locations. Though limits are not proposed for silver, concentrations <0.05 mg/L normally occurs in fresh water [
50] (
Table 2) and concentrations above this can be indicative of pollution. All the samples of this area had above 0.05 mg/L of silver in groundwater as well as surface water. Spatial distribution of these heavy metals in groundwater (
Figure 3) showed a high concentration of most heavy metals in the eastern and central parts of the study area.
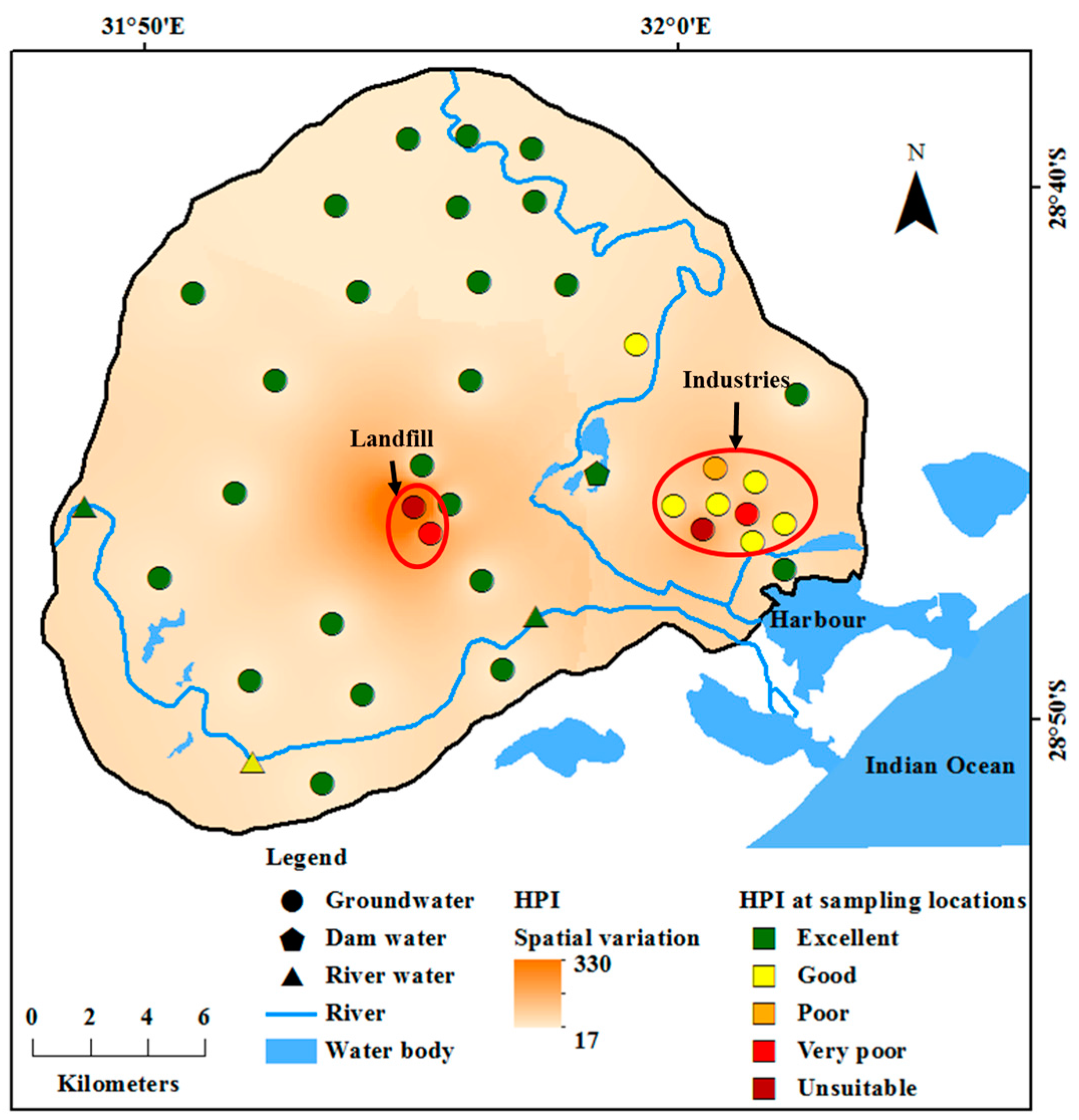
HPI was calculated for ten trace metals. Since chromium was BDL and five metals (cobalt, lithium, silicon, titanium and zirconium) do not have standard limits, they were not included in the HPI calculation. Standard values in Equations (2) to (4) are based on the maximum permissible limits of WHO prescribed in
Table 2 for all heavy metals except for iron and silver. Concentration of 0.05 mg/L was considered as the standard limit for silver. DWAF [
49] limit for iron (0.1 mg/L) was considered, as WHO has not suggested any guideline value for iron. Since the presence of these ions in drinking water is not desired, the ideal values for these ions is considered as 0 mg/L. HPI in groundwater varied from 17 to 330, river water was from 21 to 26 and dam water had the least HPI of 17. Classification of water samples based on HPI is given in
Table 3. Groundwater is suitable in 68% of the area and the spatial variation in HPI shown in
Figure 4.
Human exposure risk due to heavy metals was calculated separately for infants (0 to 6 months), children and adults as the intake of water differs among different age range. IR was 250 mL/day for infants, 1.5 L/day for children and 3 L/day for adults and body weight was 6 kg, 20 kg [
54] and 60.7 kg [
55] for infants, children and adults, respectively. Exposure risk (HE
DW) was above 1 for manganese in children with the rest of the heavy metals being <1 for all human age groups. Statistical summary of HE
DW for different metals are given in
Table 4. The sum of HE
DW also does not exceed 1 for all metals except for manganese under the children age group. The results do not provide conclusive evidence for determining the potential risk due to exposure of these metals through the drinking water pathway.
HQ takes into consideration the RfD specific to each metal and, based on the concentration level of the metal to cause acute or chronic disorders in humans, it can help to ascertain the risk in a better way. RfD values were available for most heavy metals (
Table 5) except for aluminium, cobalt, iron, silver, titanium, zirconium and zinc. Though chromium has an RfD proposed by Integrated Risk Information System (IRIS) from US EPA [
56], due to the low concentration (BDL) in the water samples, it was not included in the assessment of non-carcinogenic risk. Statistical details of the HQ for different metals are given in
Table 6.
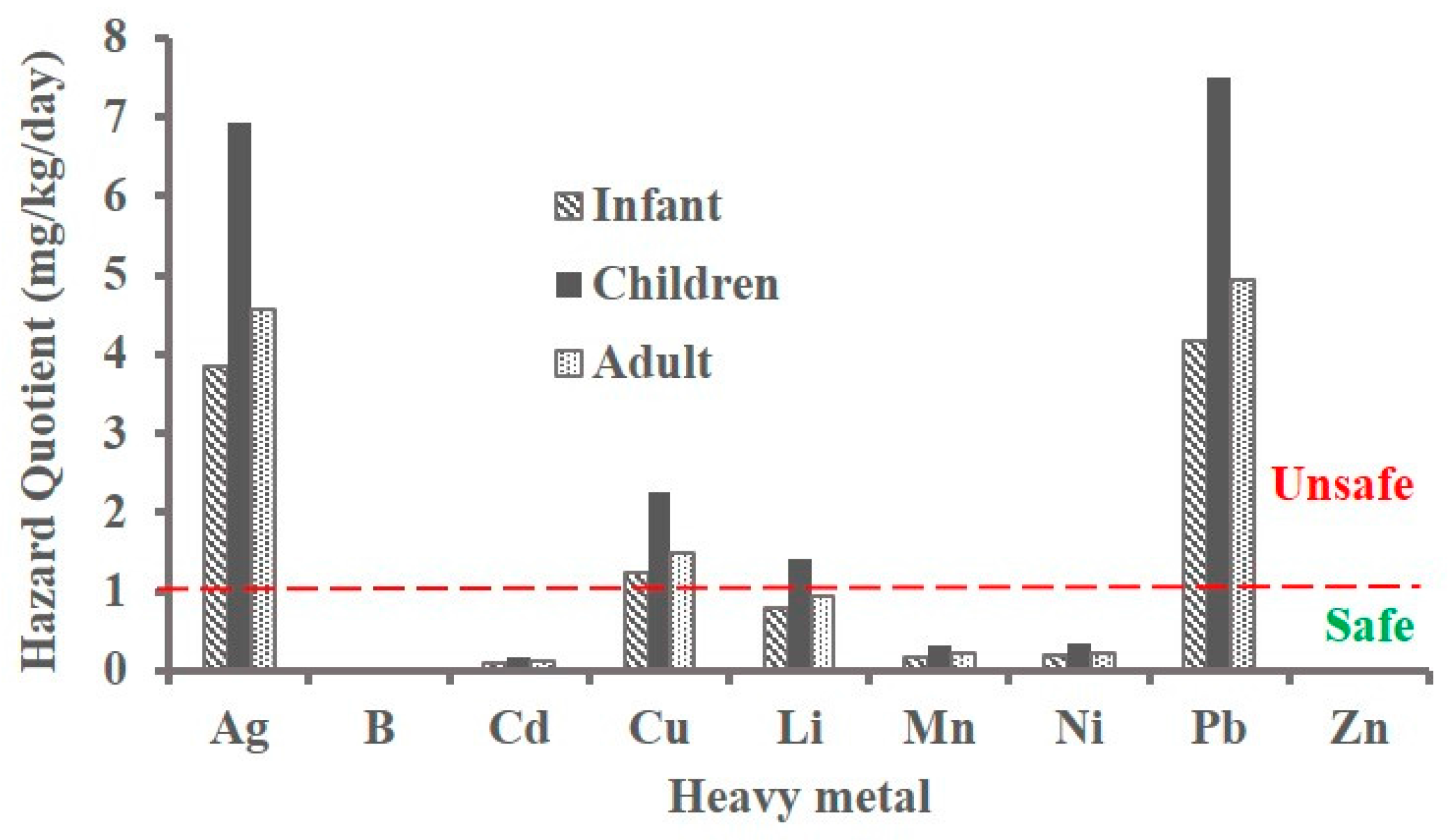
Considering the HQ of each heavy metal individually, boron, nickel and zinc do not pose any potential risk to human health. Concentrations of silver, copper, manganese and lead imply a major threat with HQ above 1 (
Table 6). Cadmium and lithium are at the border of high risk level (based on maximum value in
Table 6) for infants, but are potential risks to children and adults. Mean HQ of heavy metals (
Figure 5) indicate the low risk due to boron, cadmium, manganese, nickel and zinc.
Figure 5 shows that greater health risk from heavy metals is for children than infants and adults. Due to high exposure risk from silver, copper and lead, the cumulative HQ in all locations sampled exceeds 1 with even the minimum values being 6.2, 11.2 and 7.4 mg/kg/day for infants, children and adults, respectively (
Table 6). Surface water also shows a high potential risk if consumed following similar variation in the presence of heavy metals as in groundwater (
Table 7).
Inter-metal relationship can help to identify the source and the pathway of these metals to the environment. Correlation and factor analysis were performed to recognise independent and multiple sources. Cobalt had significant positive correlation (
r2 > 0.8) with manganese and nickel (
Table 8). Zinc also showed significant correlation with cobalt, manganese and nickel. Though strong correlation was not observed i.e.,
r2 > 0.8, zinc and boron as well as silver and boron exhibit moderate association. Weak positive correlations were found between lithium–silver, boron–cobalt, boron–manganese, boron–nickel and cadmium–copper. To further support the co-existence of metals from similar sources, factor analysis was performed. Four significant factors with eigenvalues above 1 and cumulative variance of 72% (
Table 9) were attained, which are discussed in the next section.
4. Discussion
Abundance of metals in groundwater was in the following order: silicon > manganese > silver > lithium > iron > lead > aluminium > copper > nickel > boron > zinc > cobalt > cadmium > chromium = titanium = zirconium. In surface water, concentrations were: silver > lithium > aluminium > lead > iron > nickel > copper > cobalt > manganese > zinc > cadmium > silicon = boron = chromium = titanium = zirconium. Spatial variation of HPI in
Figure 4 shows two distinct areas with a high concentration in groundwater. These are the industrial areas located near the harbour on the eastern side of the study area and the landfill in the central part. Spatial distribution in HPI also closely follows the distribution of EC (
Figure 1) and heavy metals (
Figure 3) in groundwater, indicating the possibility of occurrence of high concentrations of major and minor ions due to these activities.
Statistical analysis had the highest variance of 34% for factor 1 and show strong positive loadings between cobalt, manganese, nickel and zinc with moderate positive loading from boron. These metals do not occur normally in fresh water (
Table 2, [
50]) and so their origin is from external sources and not geogenic. Most of these metals are used widely in different industrial processes like electroplating smelting etc., and wastewater from these industries contain some concentration of these metals [
64]. Hence, factor 1 is largely influenced by industrial activities. Negative loadings of silica in factor 2 with positive correlation of lithium, iron and silver specify that they are not derived directly from soil. Silicon, the chemical element in silica contributes to 60% of the earth crust composition [
65,
66], and it is also present in seawater (2.9 mg/L) [
67]. Thus, a negative relation with silica rules out the possibility of geogenic sources as the source for factor 2. Iron ores are mined and processed in this area and related human processes may contribute to iron in groundwater. Due to the positive affinity, lithium and silver may also have been contributed from similar source i.e., mining and associated activities.
Factor 3 has a total variance of 12.3% with positive loadings for lead, silver and boron. Lead is a common pollutant from paint industries, apart from combustion of fossil fuels, industrial smelting operations and fertilizers [
68]. Lead can also be found in groundwater through dissolution of minerals from soil. Boron could be derived from earth crust as well as due to seawater intrusion in the coastal areas [
60]. Occurrence of silver in groundwater could not be ascertained clearly, but co-existence with lead and boron imply that they have a common source(s). Lead and silver occurring at high concentrations than that in natural fresh water (
Table 2) clearly indicates an external pollution source. This factor is from multiple sources associated with geogenic processes as well as anthropogenic activities including industrial operations and seawater intrusion due to over-pumping in the coastal areas. Fertilizers used for agriculture are a well-known source for cadmium and copper, which show positive loadings in factor 4 and exhibit the lowest variance of 9.5%. Presence of fertilizer plants in this area could also have contributed to these metals apart from their application to irrigation fields [
29,
30]. Factor analysis has helped as a useful tool for identifying the multiple sources of these metals in groundwater, which is grouped in
Table 9. Pollution due to heavy metals is significant in these areas, and it is important to continuously monitor the water quality to prevent further deterioration of water resources. More detailed analysis of the occurrence and sources of silver in water is needed.
5. Conclusions
Water issues in South Africa focus more on water quality than quantity and more studies are needed to identify the causes and extent of pollution. This study was carried out with an aim of determining the heavy metal contamination in water and assess the impact of human activities on groundwater in and around two major towns, Empangeni and Richards Bay. Based on EC and TDS, the water is suitable for drinking purposes in most parts of the area except for a few locations. Order of dominance of heavy metals were silicon > manganese > silver > lithium > iron > lead > aluminium > copper > nickel > boron > zinc > cobalt > cadmium, while chromium, titanium and zirconium were BDL in water. Contamination due to aluminium, lead and nickel was prevalent in the entire area and boron, cadmium, iron and manganese exceeded the limit at a few locations. HPI varied from 17 to 330 indicating high contamination in two zones within the study area. HQ indicated potential risk to human health through the drinking water pathway due to consumption of groundwater for all age groups i.e., infants, children and adults.
Statistical examination indicated four factors both as independent and a combination of multiple sources for the presence of these heavy metals in groundwater. The inter-metal relationship between cobalt, manganese, nickel, zinc and boron were indicative of industrial activities. Lithium, iron and silver in groundwater were related to mining influenced operations and processes. Application of fertilizers is likely to have contributed to the presence of cadmium and copper in groundwater. Multiple sources including geogenic industries and seawater intrusion have contributed to lead, silver and boron. This study located the two zones of pollution: (1) the industrial site near the coast in the eastern part and (2) a landfill site at the central part of the study area. It is important to prevent further pollution from these sites by adopting proper management measures that include treatment of wastes from industries and should comply with wastewater disposal standards before dumping. Landfill site should be monitored for leachate percolation, and measures to reduce this must be implemented. Consumption of groundwater by the public may result in health issues and hence proper treatment of water is necessary to make it suitable for public water supply.