Removal of Nitrogen and COD from Reclaimed Water during Long-Term Simulated Soil Aquifer Treatment System under Different Hydraulic Conditions
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experiment Setup
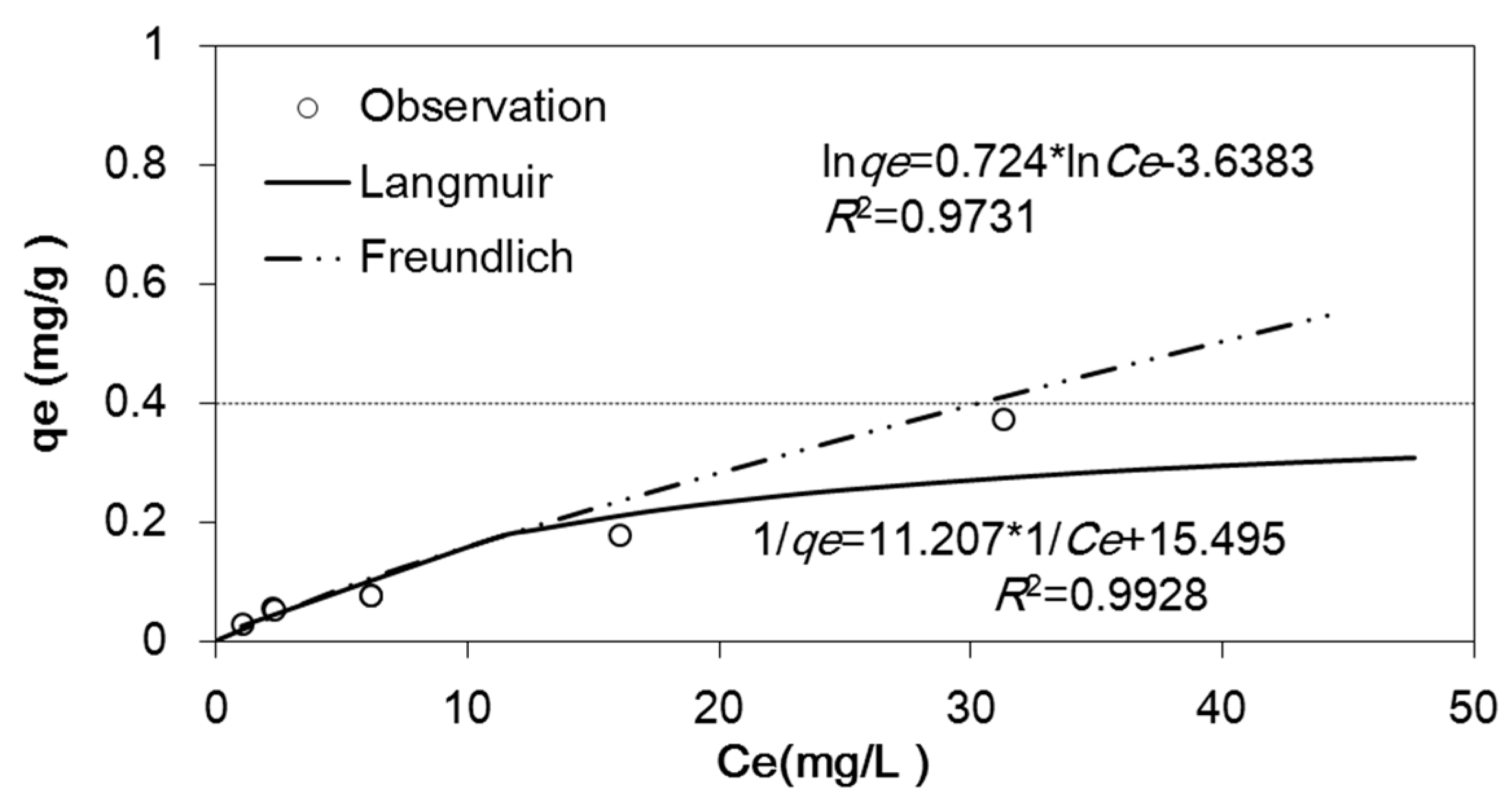
2.2. Adsorption Experiment
3. Results
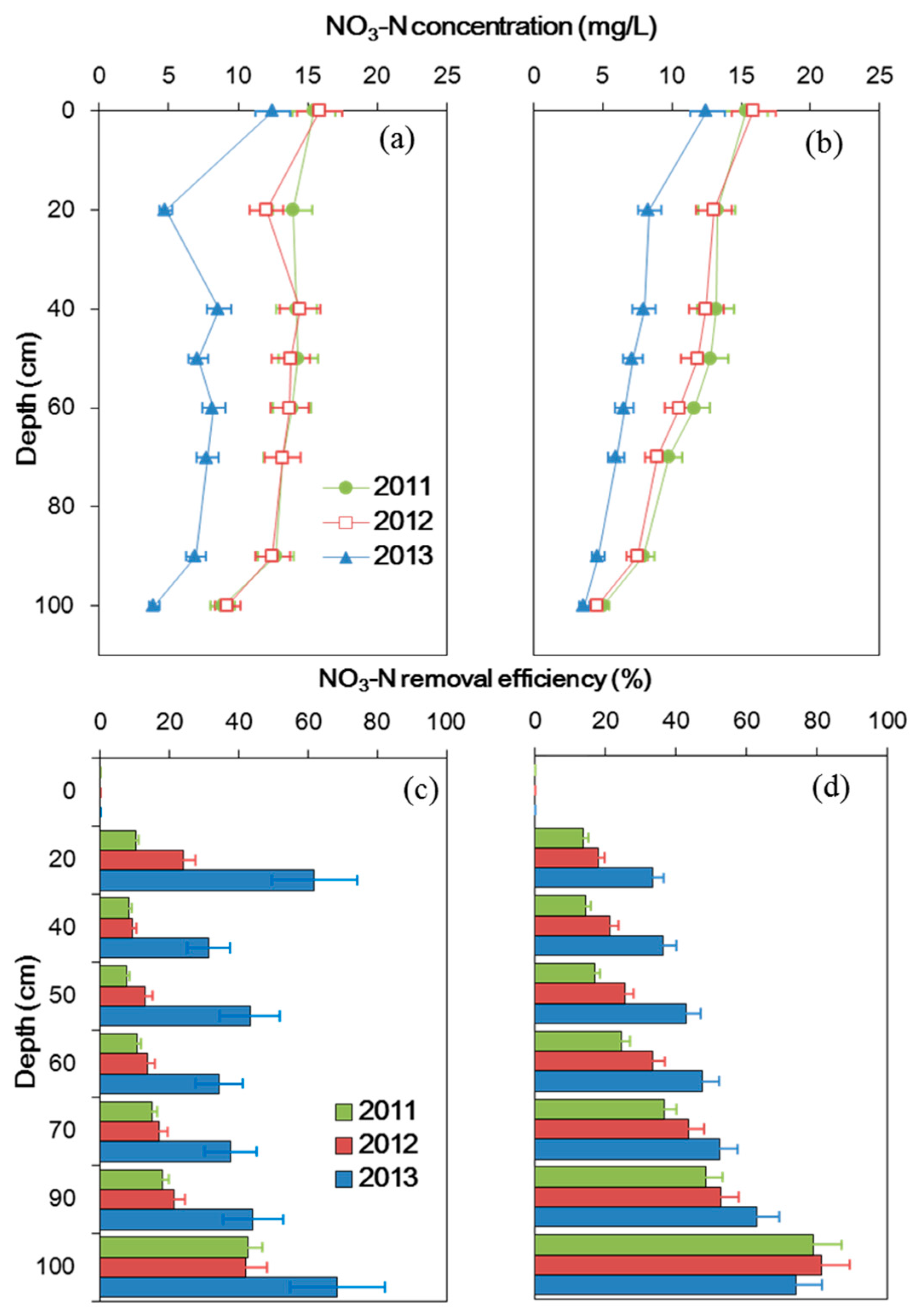
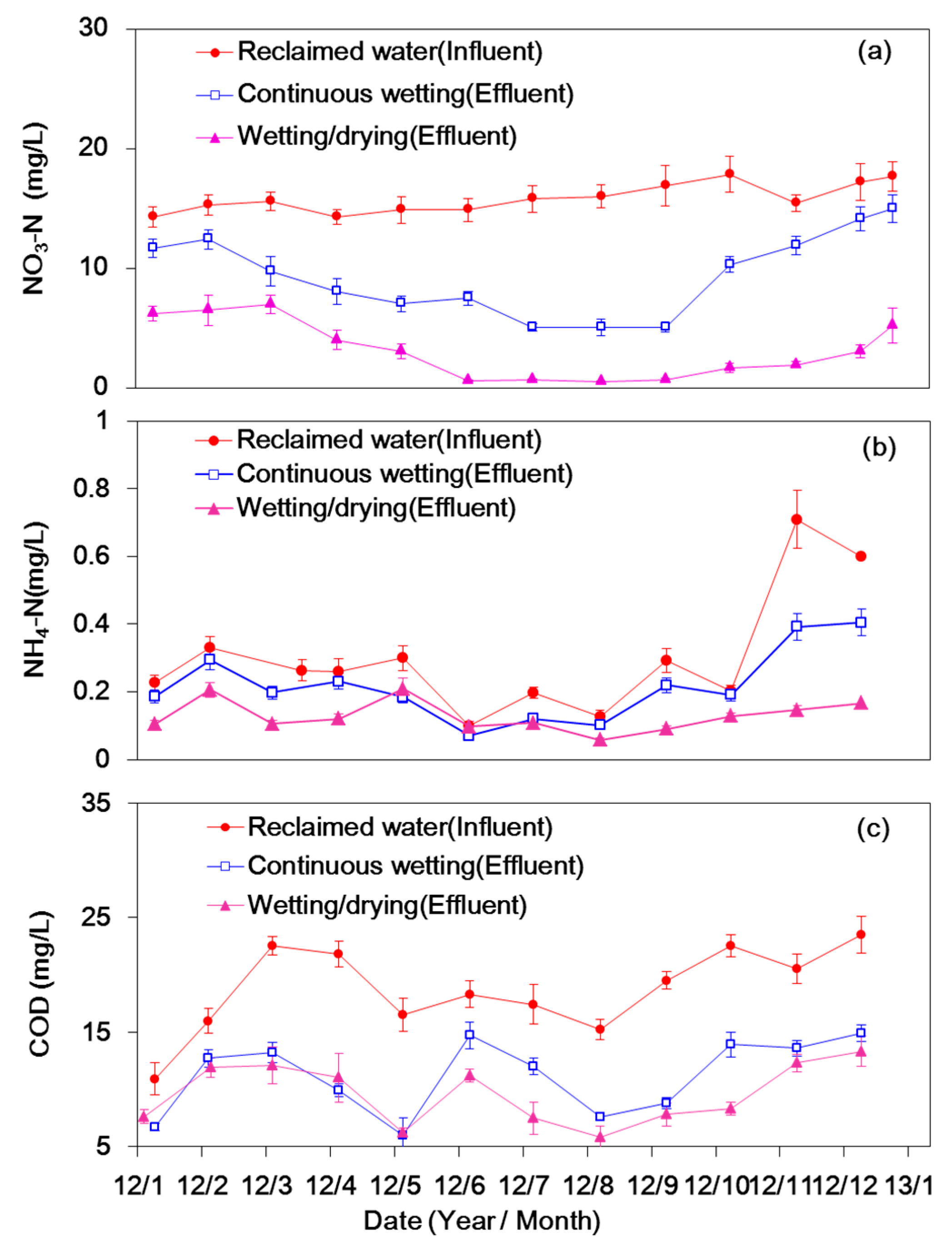
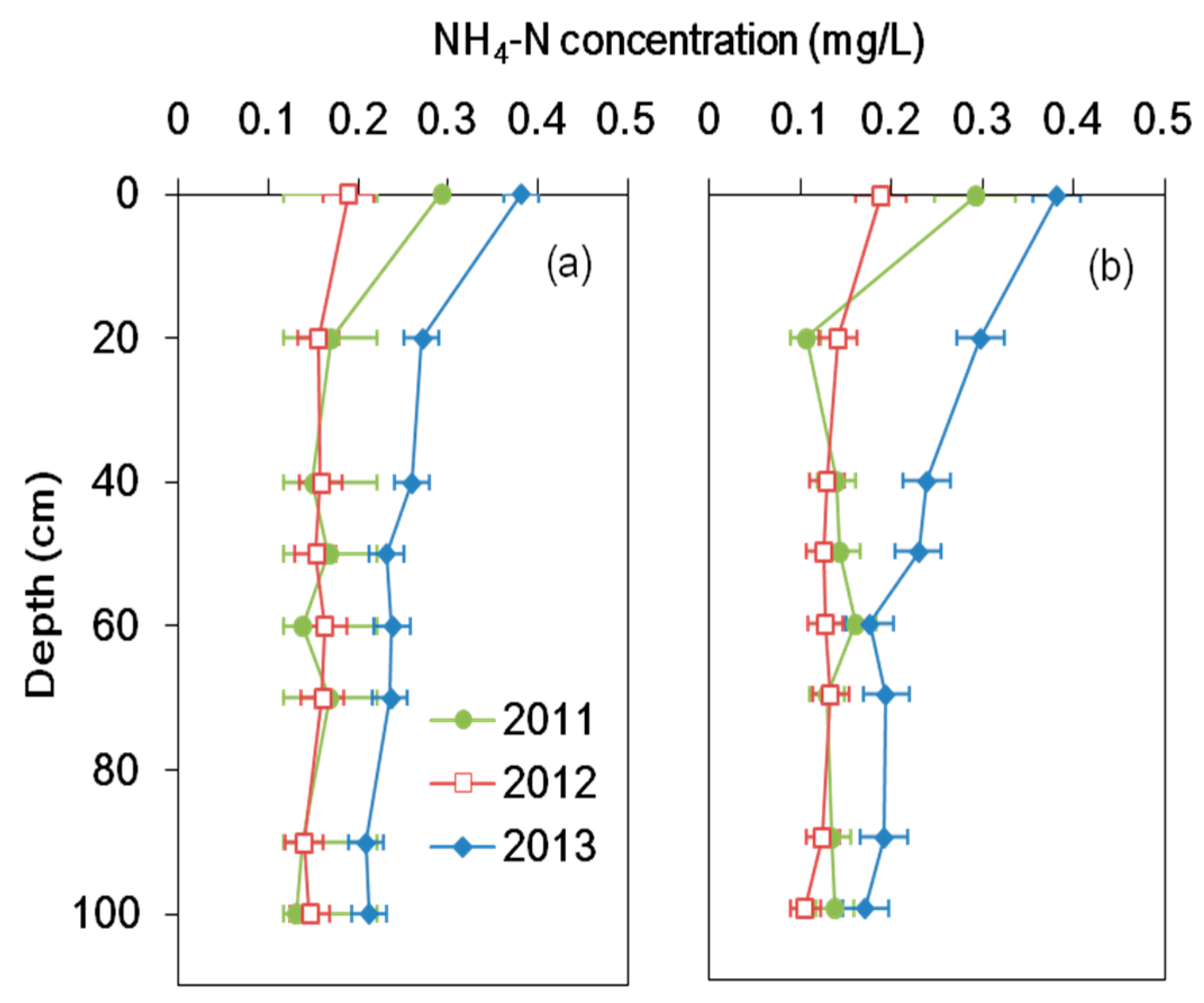
3.1. Nitrogen Concentration and Its Removal Efficiency
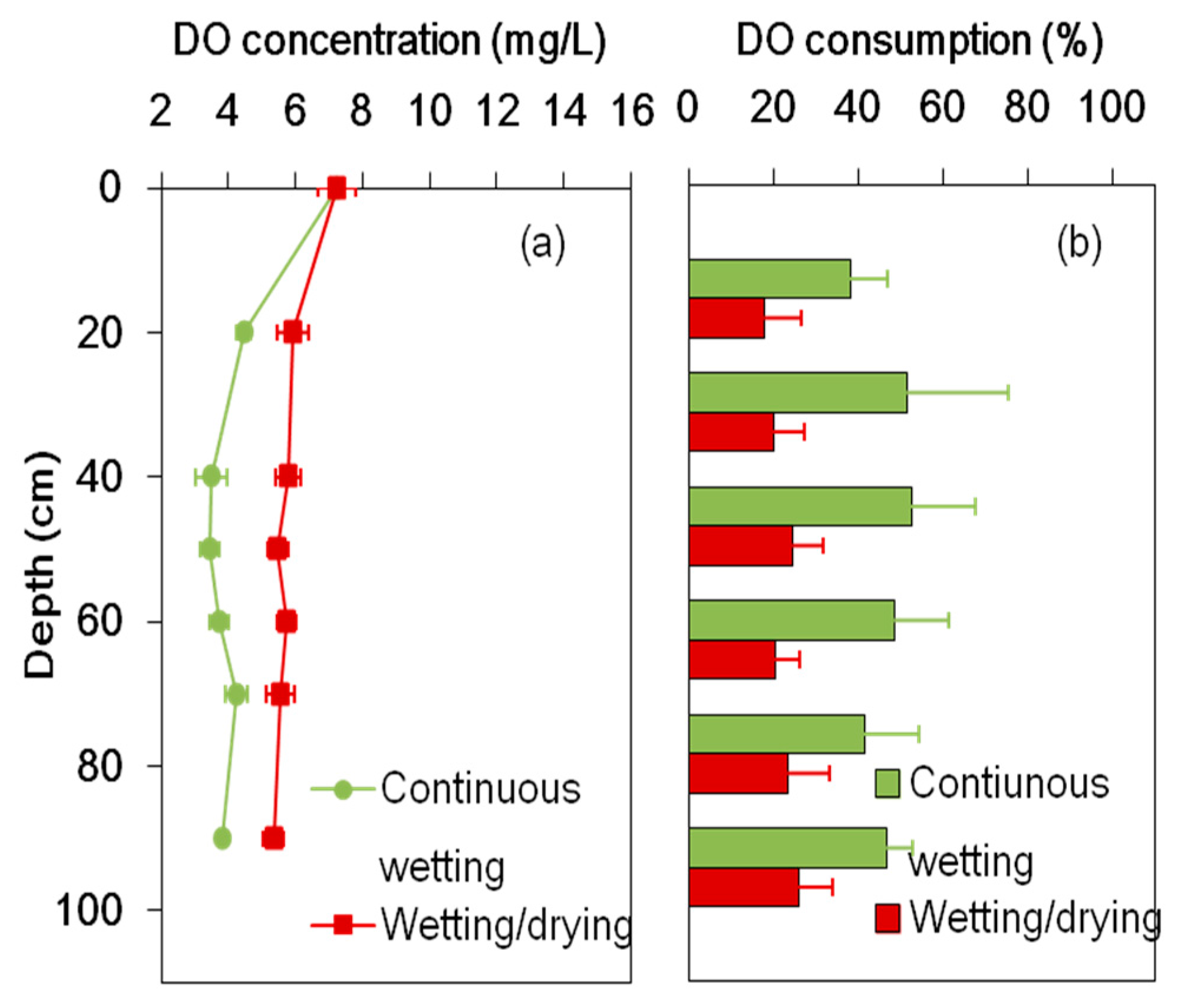
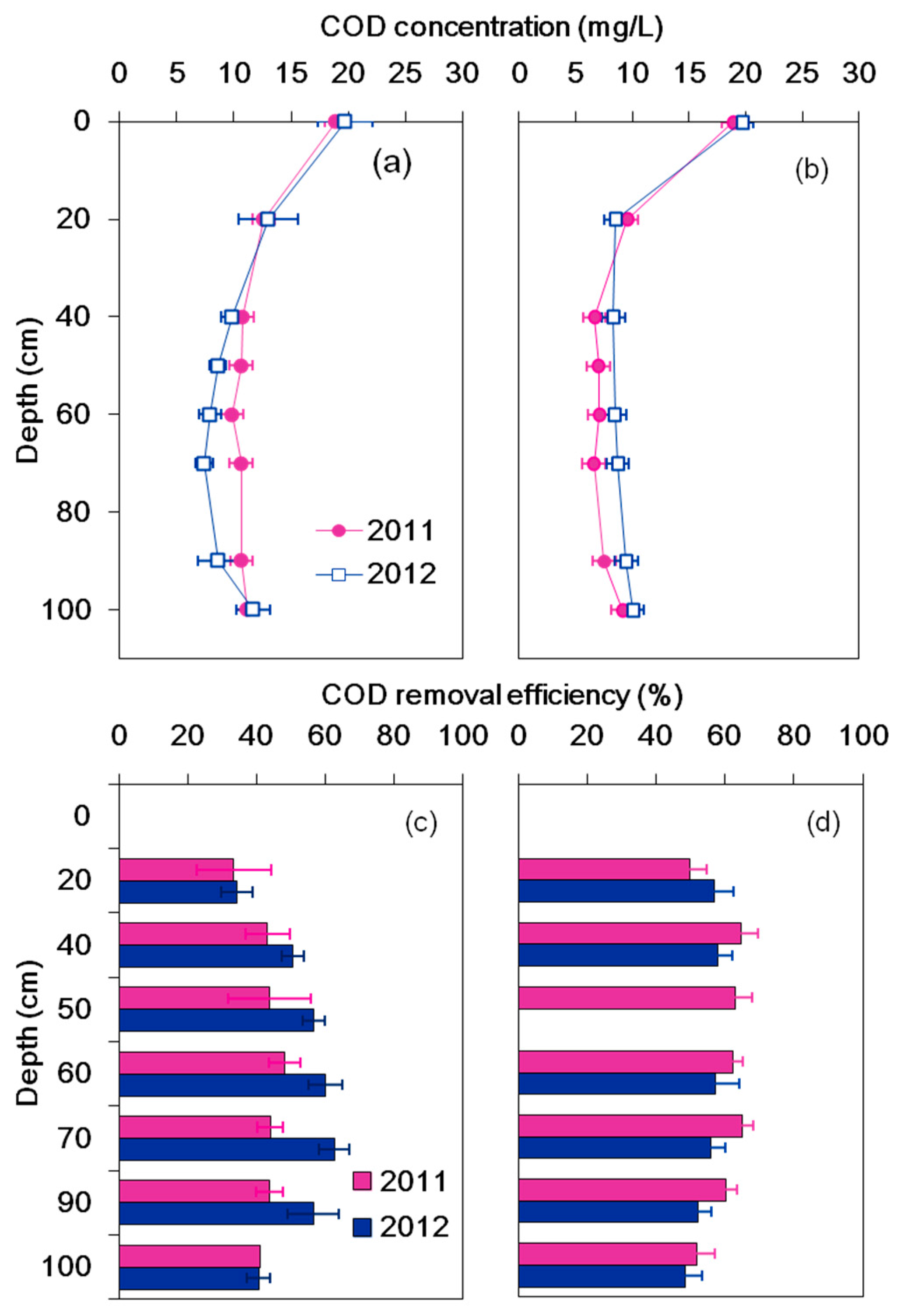
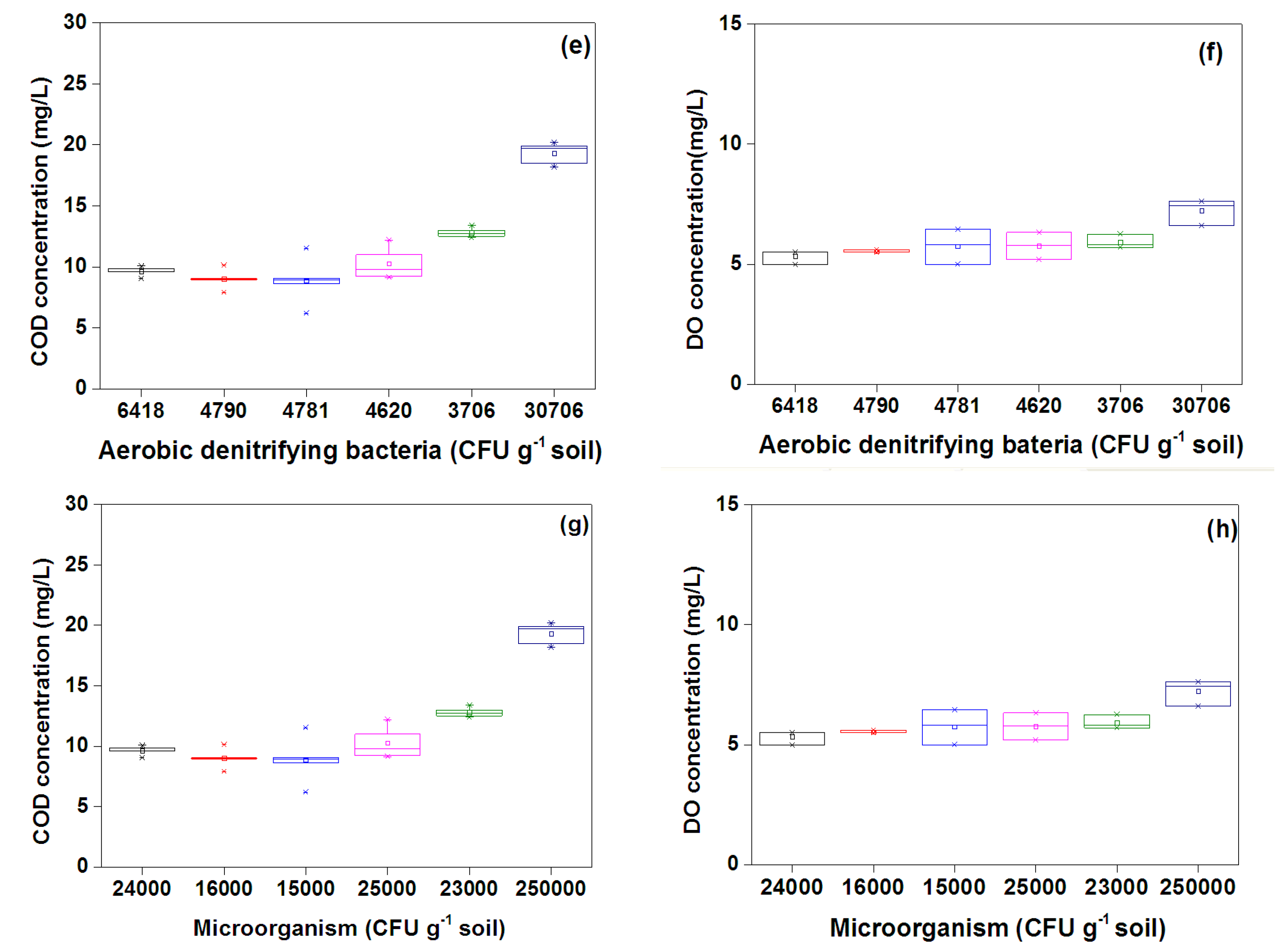
3.2. DO Consumption and COD Removal
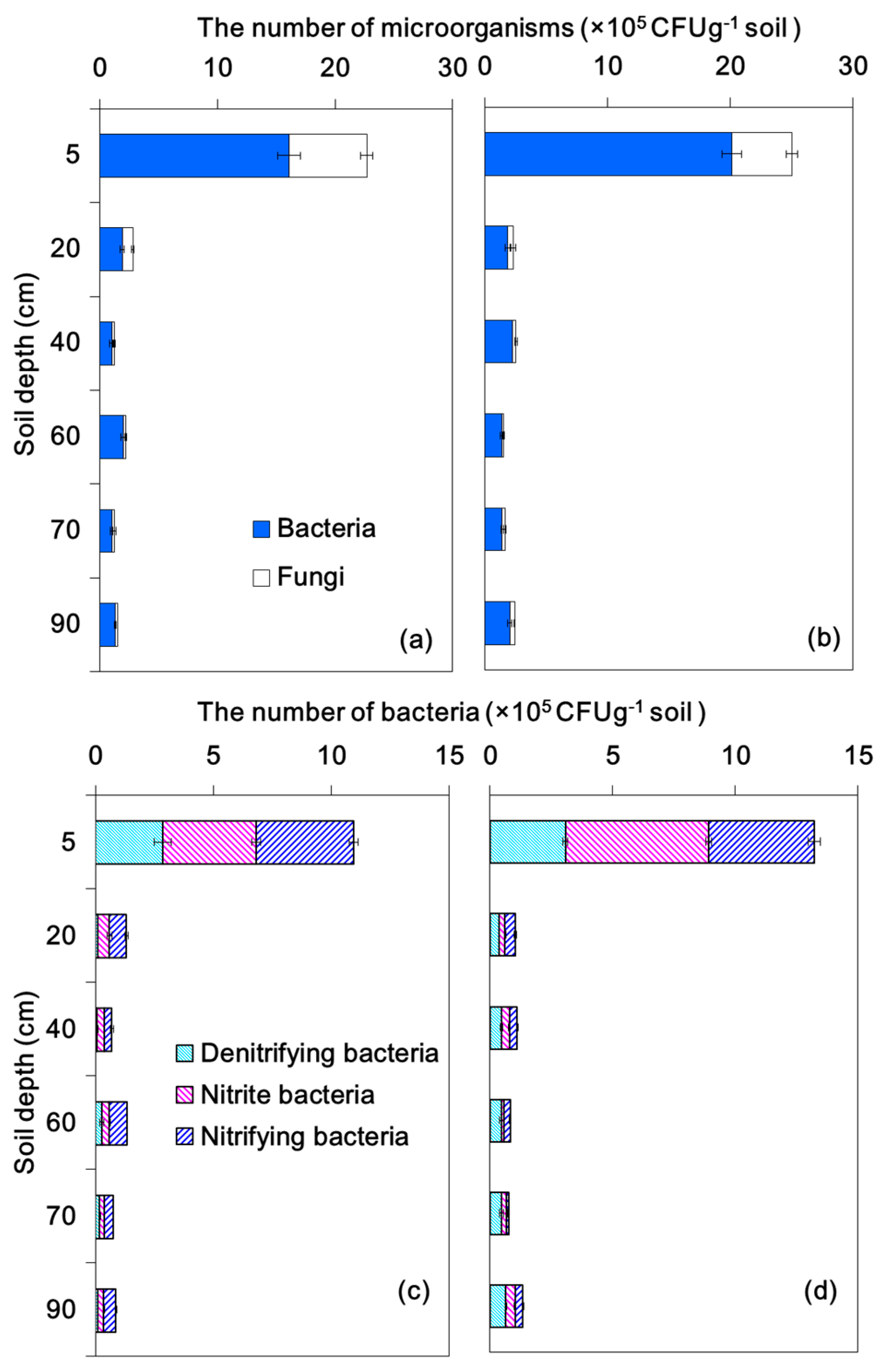
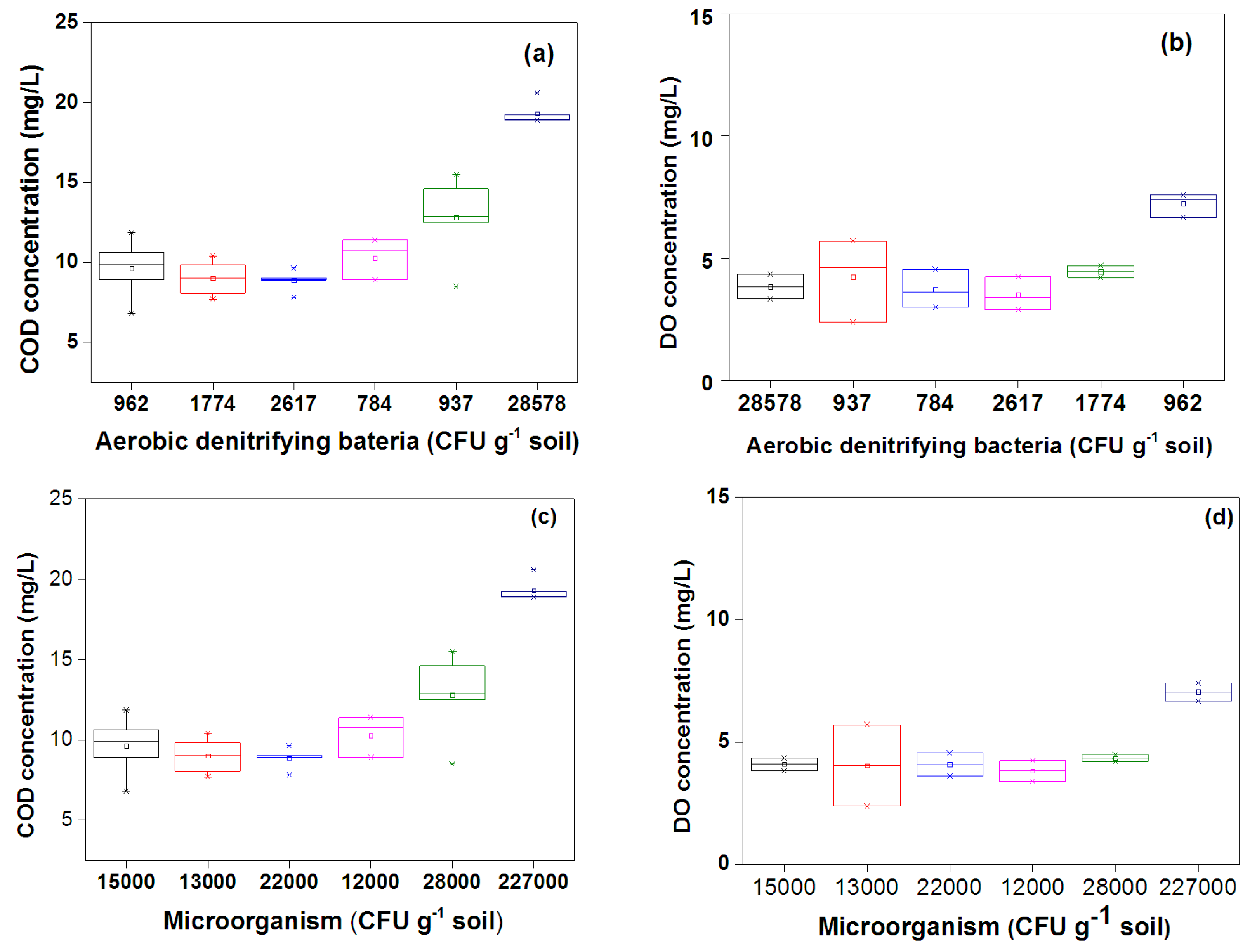
3.3. Microorganisms
4. Discussion
4.1. Nitrogen Removal
4.2. COD Removal
5. Summary and Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Chen, W.P.; Lu, S.D.; Jiao, W.T.; Wang, M.E.; Chang, A.C. Reclaimed water: A safe irrigation water source? Environ. Dev. 2013, 8, 74–83. [Google Scholar] [CrossRef]
- Chen, Y.; Chen, C. The optimal reuse of reclaimed water: A mathematical model analysis. Water Resour. Manag. 2014, 28, 2035–2048. [Google Scholar] [CrossRef]
- Wang, X.P.; Huang, G.H. Evaluation on the irrigation and fertilization management practices under the application of treated sewage water in Beijing, China. Agric. Water Manag. 2008, 95, 1011–1027. [Google Scholar] [CrossRef]
- Zheng, F.D.; Liu, L.C.; Li, B.H.; Yang, Y.; Guo, M.L. Effects of reclaimed water use for scenic water on groundwater environment in a multilayered aquifer system beneath the Chaobai River, Beijing, China: Case Study. J. Environ. Eng. 2015, 20. [Google Scholar] [CrossRef]
- Yu, Y.L.; Ma, M.Y.; Zheng, F.D.; Liu, L.C.; Zhao, N.N.; Li, X.X.; Yang, Y.M.; Guo, J. Spatio-temporal variation and controlling factors of water quality in Yongding River replenished by reclaimed water in Beijing, North China. Water 2017, 9, 453. [Google Scholar] [CrossRef]
- Dash, R.R.; Bhanu Prakash, E.V.P.; Kumar, P.; Mehrotra, I.; Sandhu, C.; Grischek, T. River bank filtration in Haridwar, India: Removal of turbidity, organics and bacteria. Hydrogeol. J. 2010, 18, 973–983. [Google Scholar] [CrossRef]
- Goren, O.; Burg, A.; Gavrieli, I.; Negev, I.; Guttman, J.; Kraitzer, T.; Kloppmann, W.; Lazar, B. Biogeochemical processes in infiltration basins and their impact on the recharging effluent, the soil aquifer treatment (SAT) system of the Shafdan plant, Israel. Appl. Geochem. 2014, 48, 58–69. [Google Scholar] [CrossRef]
- Nadav, I.; Arye, G.; Tarchitzky, J.; Chen, Y. Enhanced infiltration regime for treated-wastewater purification in soil aquifer treatment (SAT). J. Hydrol. 2012, 420–421, 275–283. [Google Scholar] [CrossRef]
- Filter, J.; Jekel, M.; Ruhl, A.S. Impacts of Accumulated Particulate Organic Matter on Oxygen Consumption and Organic Micro-Pollutant Elimination in Bank Filtration and Soil Aquifer Treatment. Water 2017, 9, 349. [Google Scholar] [CrossRef]
- Regnery, J.; Wing, A.D.; Kautz, J.; Drewes, J.E. Introducing sequential managed aquifer recharge technology (SMART)—From laboratory to full-scale application. Chemosphere 2016, 154, 8–16. [Google Scholar] [CrossRef] [PubMed]
- Wei, L.L.; Li, S.L.; Noguera, D.R.; Qin, K.; Jiang, J.Q.; Zhao, Q.L.; Kong, X.J.; Cui, F.Y. Dissolved organic matter removal during coal slag additive soil aquifer treatment for secondary effluent recharging: Contribution of aerobic biodegradation. J. Environ. Manag. 2015, 156, 158–166. [Google Scholar] [CrossRef] [PubMed]
- Abel, C.D.T.; Sharma, S.K.; Buçpapaj, E.; Kennedy, M.D. Impact of hydraulic loading rate and media type on removal of bulk organic matter and nitrogen from primary effluent in a laboratory-scale soil aquifer treatment system. Water Sci. Technol. 2013, 68, 217–226. [Google Scholar] [CrossRef] [PubMed]
- Bertelkamp, C.; Reungoat, J.; Cornelissen, E.R.; Singhal, N.; Reynisson, J.; Cabo, A.J.; van der Hoek, J.P.; Verliefde, A.R.D. Sorption and biodegradation of organic micropollutants during river bank filtration: A laboratory column study. Water Res. 2014, 52, 231–241. [Google Scholar] [CrossRef] [PubMed]
- Maeng, S.K.; Sharma, S.K.; Abel, C.D.T.; Knezev, A.M.; Song, K.G.; Amy, G.L. Effects of effluent organic matter characteristics on the removal of bulk organic matter and selected pharmaceutically active compounds during managed aquifer recharge: Column study. J. Contam. Hydrol. 2012, 140–141, 139–149. [Google Scholar] [CrossRef] [PubMed]
- Essandoh, H.M.K.; Tizaoui, C.; Mohamed, M.H.A. Removal of dissolved organic carbon and nitrogen during simulated soil aquifer treatment. Water Res. 2013, 47, 3559–3572. [Google Scholar] [CrossRef] [PubMed]
- Von Rohr, M.R.; Hering, J.G.; Kohler, H.P.E.; von Gunten, U. Column studies to assess the effects of climate variables on redox processes during riverbank filtration. Water Res. 2014, 61, 263–275. [Google Scholar] [CrossRef] [PubMed]
- Dillon, P.K.; Kumar, A.; Kookana, R.; Leijs, R.; Reed, D.; Parsons, S.; Ingleton, G. Managed Aquifer Recharge-Risks to Groundwater Dependent Ecosystems: A Review. In Water for a Healthy Country Flagship Report to Land & Water Australia; CSIRO: Canberra, Australia, 2009. [Google Scholar]
- Rauch-Williams, T.; Drewes, J.E. Using soil biomass as an indicator for the biological removal of effluent-derived organic carbon during soil infiltration. Water Res. 2006, 40, 961–968. [Google Scholar] [CrossRef] [PubMed]
- Abel, C.D.T. Soil Aquifer Treatment: Assessment and Applicability of Primary Effluent Reuse in Developing Countries. Ph.D. Thesis, Delft University of Technology, Delft, The Netherlands, 2014. [Google Scholar]
- Meng, J.; Li, J.L.; Li, J.Z.; Sun, K.; Antwi, P.; Deng, K.W.; Wang, C.; Buelna, G. Efficiency and bacterial populations related to pollutant removal in an upflow microaerobic sludge reactor treating manure-free piggery wastewater with low COD/TN ratio. Bioresour. Technol. 2016, 201, 166–173. [Google Scholar] [CrossRef] [PubMed]
- Calderer, M.; Martí, V.; de Pablo, J.; Guivernau, M.; Prenafeta-Boldú, F.X.; Viñas, M. Effects of enhanced denitrification on hydrodynamics and microbial community structure in a soil column system. Chemosphere 2014, 111, 112–119. [Google Scholar] [CrossRef] [PubMed]
- Gao, H.; Schreiber, F.; Collins, G.; Jensen, M.M.; Kostka, J.E.; Lavik, G.; de Beer, D.; Zhou, H.Y.; Kuypers, M.M.M. Aerobic denitrification in permeable Wadden Sea sediments. ISME J. 2010, 4, 417–426. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Yang, K.; Wang, H.Y.; Lv, B.; Ma, F. Characteristics of nitrate removal in a bio-ceramsite reactor by aerobic denitrification. Environ. Technol. 2015, 36, 1457–1463. [Google Scholar] [CrossRef] [PubMed]
- Wan, C.L.; Yang, X.; Lee, D.J.; Du, M.A.; Wan, F.; Chen, C. Aerobic denitrification by novel isolated strain using NO2-N as nitrogen source. Bioresour. Technol. 2011, 102, 7244–7248. [Google Scholar] [CrossRef] [PubMed]
- Patureau, D.; Davison, J.; Bernet, N.; Moletta, R. Denitrification under various aeration conditions in Comamonas sp., strain SGLYS2. FEMS Microbiol. Ecol. 1994, 14, 71–78. [Google Scholar] [CrossRef]
- Huang, H.K.; Tseng, S.K. Nitrate reduction by Citrobacter diversus under aerobic environment. Appl. Microbiol. Biotechnol. 2001, 55, 90–94. [Google Scholar] [CrossRef] [PubMed]
- Patureau, D.; Bernet, N.; Delgenès, J.P.; Moletta, R. Effect of dissolved oxygen and carbon-nitrogen loads on denitrification by an aerobic consortium. Appl. Microbiol. Biotechnol. 2000, 54, 535–542. [Google Scholar] [CrossRef] [PubMed]
- Chiu, Y.C.; Chung, M.S. Determination of optimal COD/nitrate ratio for biological denitrification. Int. Biodeterior. Biodegrad. 2003, 51, 43–49. [Google Scholar] [CrossRef]
- Kim, M.; Jeong, S.Y.; Yoon, S.J.; Cho, S.J.; Kim, Y.H.; Kim, M.J.; Ryu, E.Y.; Lee, S.J. Aerobic denitrification of Pseudomonas putida AD-21 at different C/N ration. J. Biosci. Bioeng. 2008, 106, 498–502. [Google Scholar] [CrossRef] [PubMed]
- Essandoh, H.M.K.; Tizaoui, C.; Mohamed, M.H.A.; Amy, G.; Brdjanovic, D. Soil aquifer treatment of artificial wastewater under saturated conditions. Water Res. 2011, 45, 4211–4226. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.S.; Jin, M.G.; Sun, Q. Experiment and numerical simulation on transportation of ammonia nitrogen in saturated soil column with steady flow. Procedia Environ. Sci. 2011, 10, 1327–1332. [Google Scholar] [CrossRef]
- Paus, K.H.; Morgan, J.; Gulliver, J.S.; Asce, F.; Hozalski, R.M. Effects of bioretention media compost volume fraction on toxic metals removal, hydraulic conductivity, and phosphorous release. J. Environ. Eng. 2014, 140. [Google Scholar] [CrossRef]
- Brown, P.; Ong, S.K.; Lee, Y. Influence of anoxic and anaerobic hydraulic retention time on biological nitrogen and phosphorus removal in a membrane bioreactor. Desalination 2011, 270, 227–232. [Google Scholar] [CrossRef]
- Güngör, K.; Ünlü, K. Nitrite and nitrate removal efficiencies of soil aquifer treatment columns. Turkish J. Eng. Environ. Sci. 2005, 29, 159–170. [Google Scholar]
- Abel, C.D.T.; Sharma, S.K.; Mersha, S.A.; Kennedy, M.D. Influence of intermittent infiltration of primary effluent on removal of suspended solids, bulk organic matter, nitrogen and pathogens indicators in a simulated managed aquifer recharge system. Ecol. Eng. 2014, 64, 100–107. [Google Scholar] [CrossRef]
- Ak, M.; Gunduz, O. Comparison of organic matter removal from synthetic and real wastewater in a laboratory-scale soil aquifer treatment system. Water Air Soil Pollut. 2013, 224, 1–16. [Google Scholar] [CrossRef]
- Wei, F.S.; Qi, W.Q.; Sun, Z.G.; Huang, Y.R.; Shen, Y.W. Water and Wastewater Monitoring and Analysis Method; China Environmental Science Press: Beijing, China, 2002; ISBN 978-7-8016-3400-9. [Google Scholar]
- Dong, X.Z.; Cai, M. Manual of System Determinative Common Bacteriology; Science Press: Beijing, China, 2001; ISBN 978-7-0300-8460-6. [Google Scholar]
- Schmidt, C.A.; Clark, M.W. Efficacy of a denitrification wall to treat continuously high nitrate loads. Ecol. Eng. 2012, 42, 203–211. [Google Scholar] [CrossRef]
- Münch, E.V.; Lant, P.; Keller, J. Simultaneous nitrification and denitrification in bench-scale sequencing batch reactors. Water Res. 1996, 30, 277–284. [Google Scholar] [CrossRef]
- Yao, S.; Ni, J.R.; Ma, T.; Li, C. Heterotrophic nitrification and aerobic denitrification at low temperature by a newly isolated bacterium, Acinetobacter sp. HA2. Bioresour. Technol. 2013, 139, 80–86. [Google Scholar] [CrossRef] [PubMed]
- Hayatsu, M.; Tago, K.; Saito, M. Various players in the nitrogen cycle: Diversity and functions of the microorganisms involved in nitrification and denitrification. Soil Sci. Plant Nutr. 2008, 54, 33–45. [Google Scholar] [CrossRef]
- Ryu, H.D.; Kim, D.; Lim, H.E.; Lee, S.I. Nitrogen removal from low carbon-to-nitrogen wastewater in four-stage biological aerated filter system. Process Biochem. 2008, 43, 729–735. [Google Scholar] [CrossRef]
- Zhang, L.Y.; Ye, Y.B.; Wang, L.J.; Xi, B.D.; Wang, H.Q.; Li, Y. Nitrogen removal processes in deep subsurface wastewater infiltration systems. Ecol. Eng. 2015, 77, 275–283. [Google Scholar] [CrossRef]
- Willems, H.P.; Rotelli, M.D.; Berry, D.F.; Smith, E.P.; Reneau, R.B., Jr.; Mostaghimi, S. Nitrate removal in riparian wetland soils: Effects of flow rate, temperature, nitrate concentration and soil depth. Water Res. 1997, 31, 841–849. [Google Scholar] [CrossRef]
- Fox, P.; Makam, R. Surface area and travel time relationships in aquifer treatment systems. Water Environ. Res. 2009, 81, 2337–2343. [Google Scholar] [CrossRef] [PubMed]
- Pavelic, P.; Dillon, P.J.; Mucha, M.; Nakai, T.; Barry, K.E.; Bestland, E. Laboratory assessment of factors affecting soil clogging of soil aquifer treatment systems. Water Res. 2011, 45, 3153–3163. [Google Scholar] [CrossRef] [PubMed]
- Quanrud, D.M.; Hafer, J.; Karpiscak, M.M.; Zhang, J.M.; Lansey, K.E.; Arnold, R.G. Fate of organics during soil-aquifer treatment: Sustainability of removals in the field. Water Res. 2003, 37, 3401–3411. [Google Scholar] [CrossRef]
- Bunsri, T.; Sivakumar, M.; Hagare, D. Transport and biotransformation of organic carbon and nitrate compounds in unsaturated soil conditions. Water Sci. Technol. 2008, 58, 2143–2153. [Google Scholar] [CrossRef] [PubMed]
- Gray, N.F. Biology of Wastewater Treatment; World Scientific: London, UK, 2004; ISBN 1-86094-332-2. [Google Scholar]
- Metcalf and Eddy, Inc. Wastewater Engineering: Treatment and Reuse; McGraw-Hill: New York, NY, USA, 2003; ISBN 978-0-0734-0118-8. [Google Scholar]
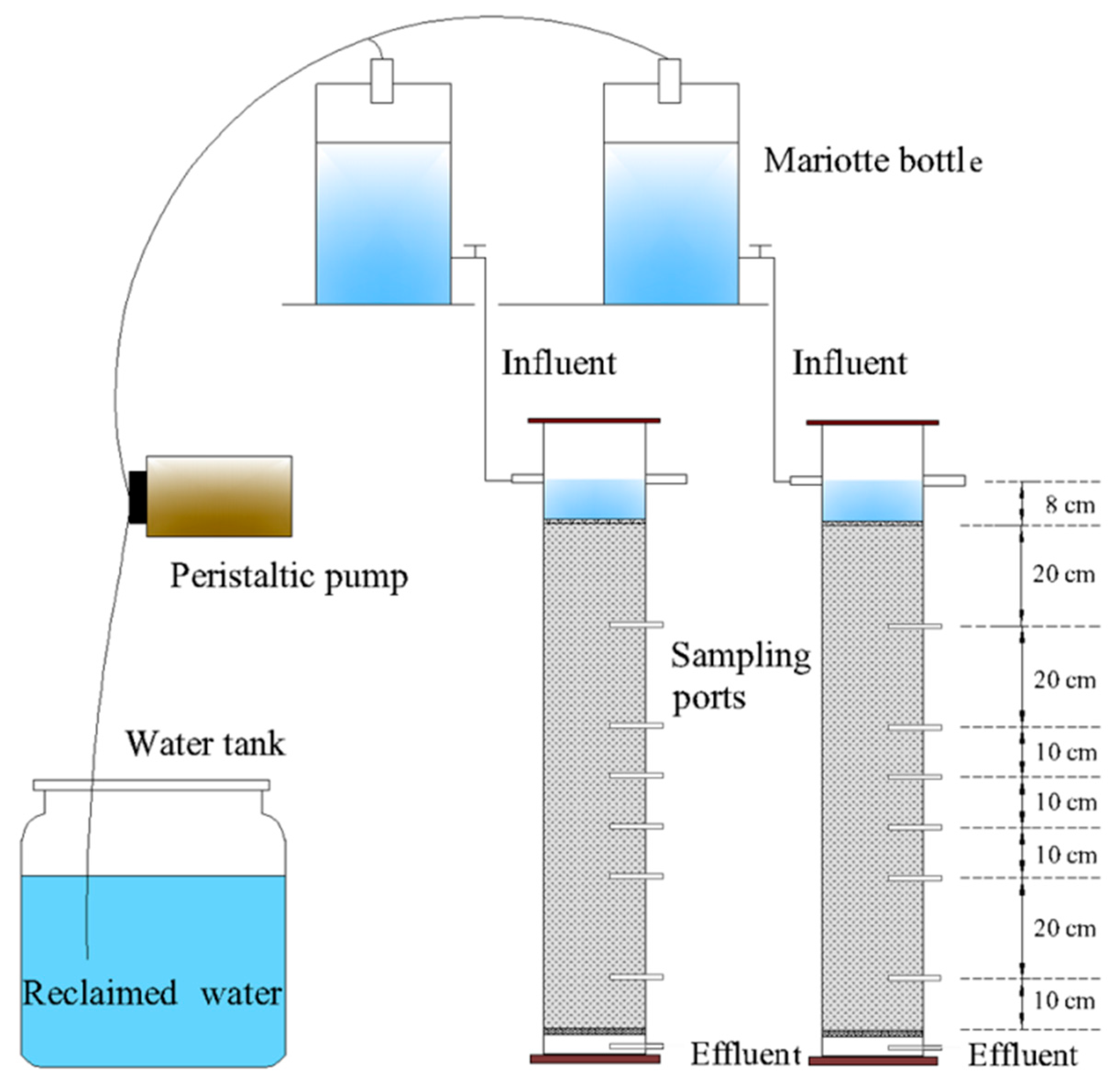
| Soil Depth | Bulk Density | Sand | Silt | Clay | Organic | NO3–N | NH4–N |
|---|---|---|---|---|---|---|---|
| (cm) | (g/cm3) | (%) | (%) | (%) | Matter (%) | (mg/kg) | (mg/kg) |
| 5 | 1.47 | 35 | 57 | 8 | 1.24 | 100 | 0.7 |
| 15 | 1.47 | 35 | 57 | 8 | 0.83 | 25 | 0.7 |
| 35 | 1.49 | 28 | 61 | 11 | 0.48 | 48 | 1 |
| 55 | 1.49 | 12 | 72 | 16 | 0.64 | 66 | 0.5 |
| 75 | 1.52 | 19 | 67 | 14 | 0.54 | 68 | 1.1 |
| 95 | 1.54 | 20 | 66 | 14 | 0.27 | 78 | 0.4 |
| Parameters | Average Concentration (mg/L) | The Number of Samplings (n) |
|---|---|---|
| Chemical oxygen demand (COD) | 19.3 ± 2.6 | 71 |
| Ammonia nitrogen (NH4–N) | 0.29 ± 0.1 | 228 |
| Nitrate nitrogen (NO3–N) | 15.6 ± 1.6 | 228 |
| Total nitrogen (TN) | 17.3 ± 2.0 | 93 |
| Dissolved oxygen (DO) | 7.25 ± 0.6 | 10 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pan, W.; Xiong, Y.; Huang, Q.; Huang, G. Removal of Nitrogen and COD from Reclaimed Water during Long-Term Simulated Soil Aquifer Treatment System under Different Hydraulic Conditions. Water 2017, 9, 786. https://doi.org/10.3390/w9100786
Pan W, Xiong Y, Huang Q, Huang G. Removal of Nitrogen and COD from Reclaimed Water during Long-Term Simulated Soil Aquifer Treatment System under Different Hydraulic Conditions. Water. 2017; 9(10):786. https://doi.org/10.3390/w9100786
Chicago/Turabian StylePan, Weiyan, Yunwu Xiong, Quanzhong Huang, and Guanhua Huang. 2017. "Removal of Nitrogen and COD from Reclaimed Water during Long-Term Simulated Soil Aquifer Treatment System under Different Hydraulic Conditions" Water 9, no. 10: 786. https://doi.org/10.3390/w9100786
APA StylePan, W., Xiong, Y., Huang, Q., & Huang, G. (2017). Removal of Nitrogen and COD from Reclaimed Water during Long-Term Simulated Soil Aquifer Treatment System under Different Hydraulic Conditions. Water, 9(10), 786. https://doi.org/10.3390/w9100786






