Response of Zooplankton to Climate Variability: Droughts Create a Perfect Storm for Cladocerans in Shallow Eutrophic Lakes
Abstract
1. Introduction
2. Materials and Methods
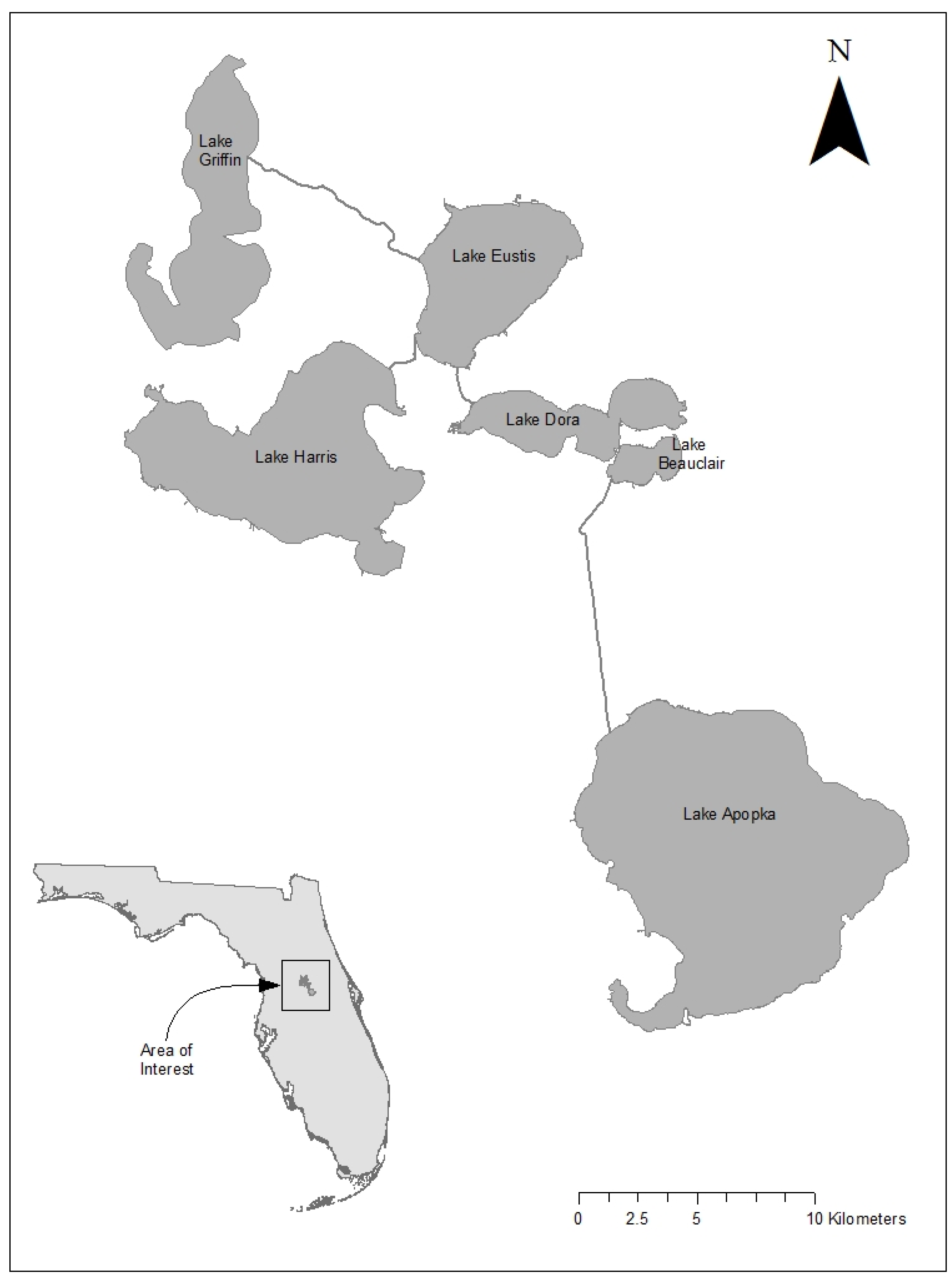
2.1. Description of the Study Lakes
2.2. Sampling and Laboratory Methods
2.3. Statistical Analysesiagram
2.4. Data
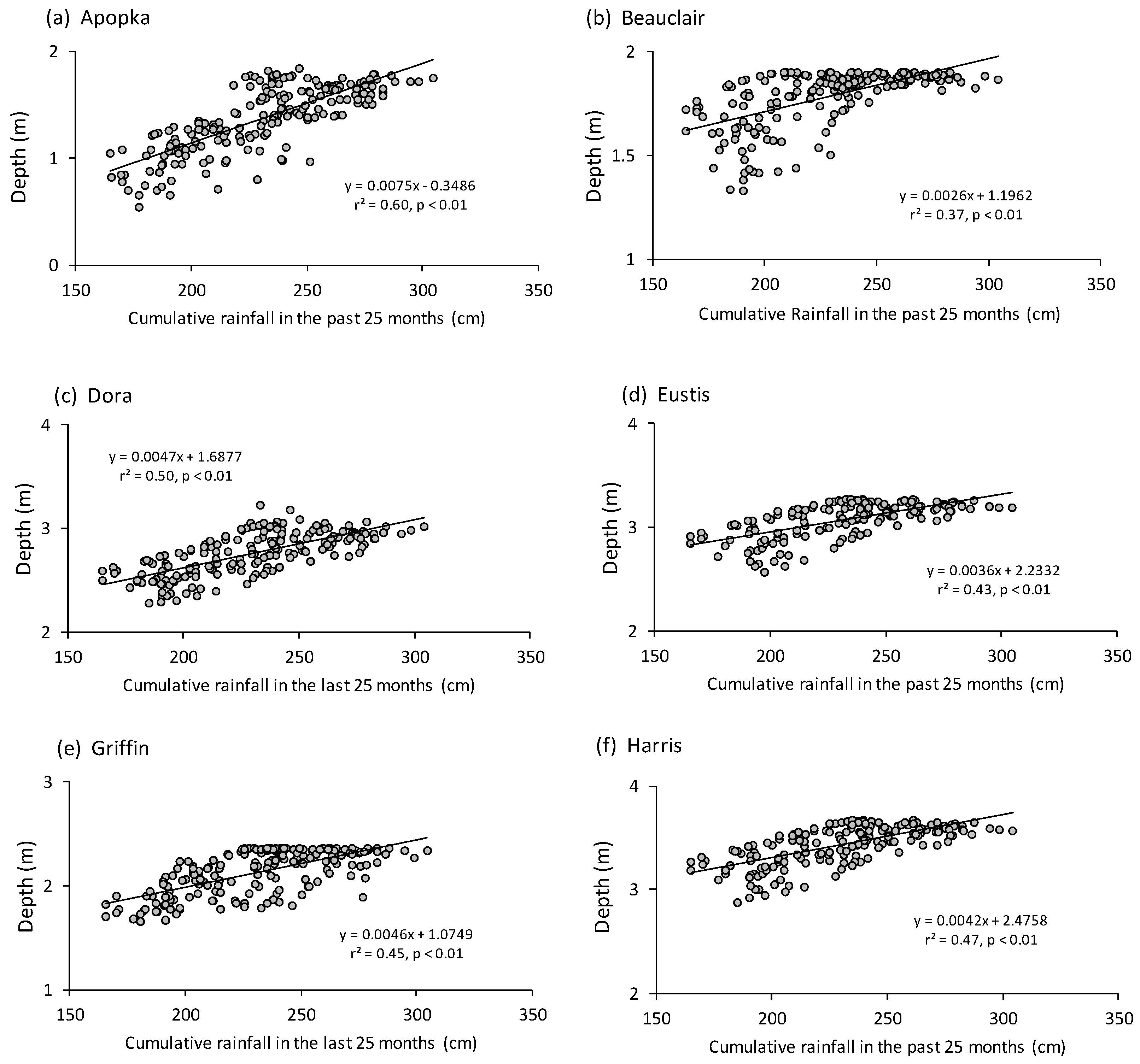
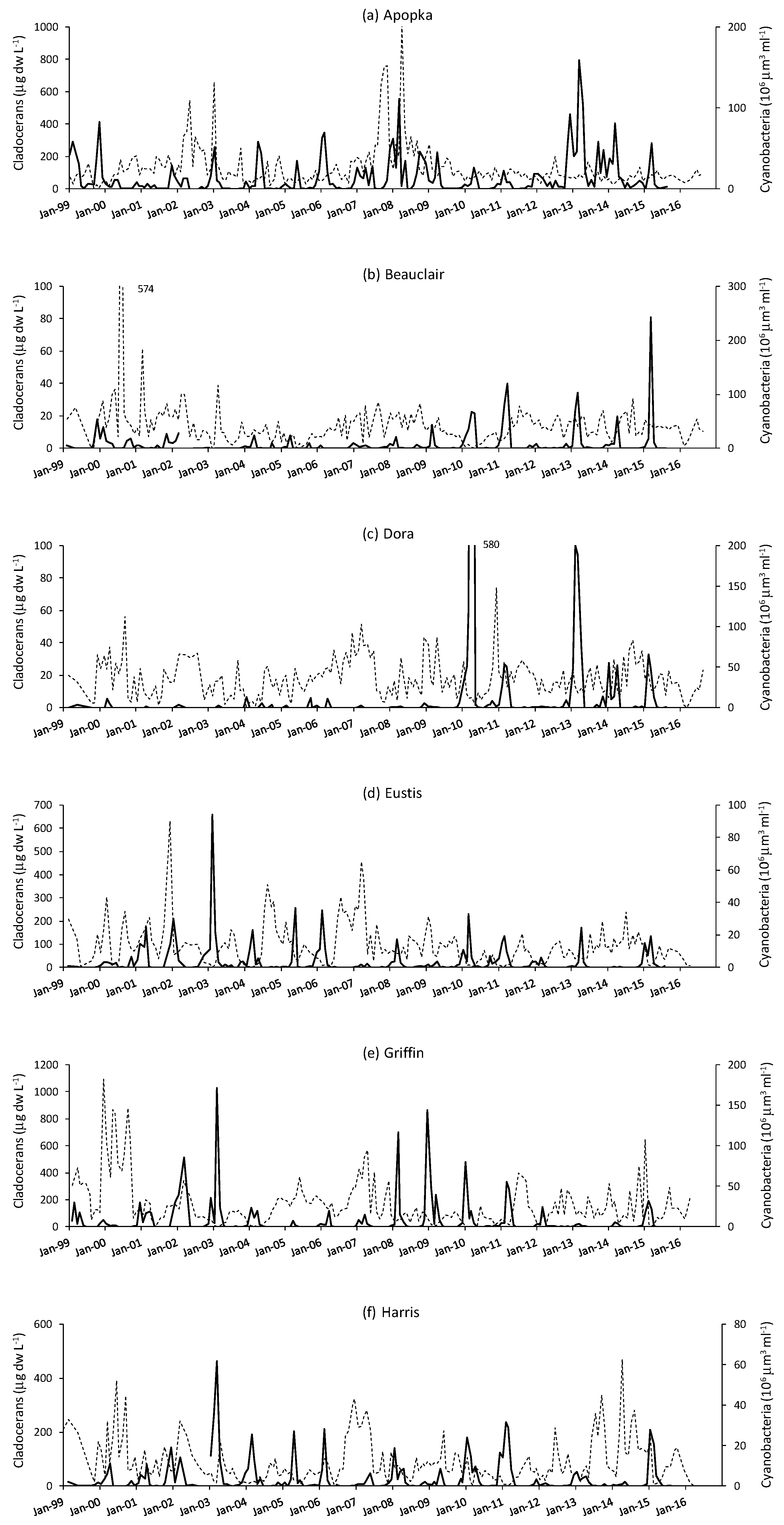
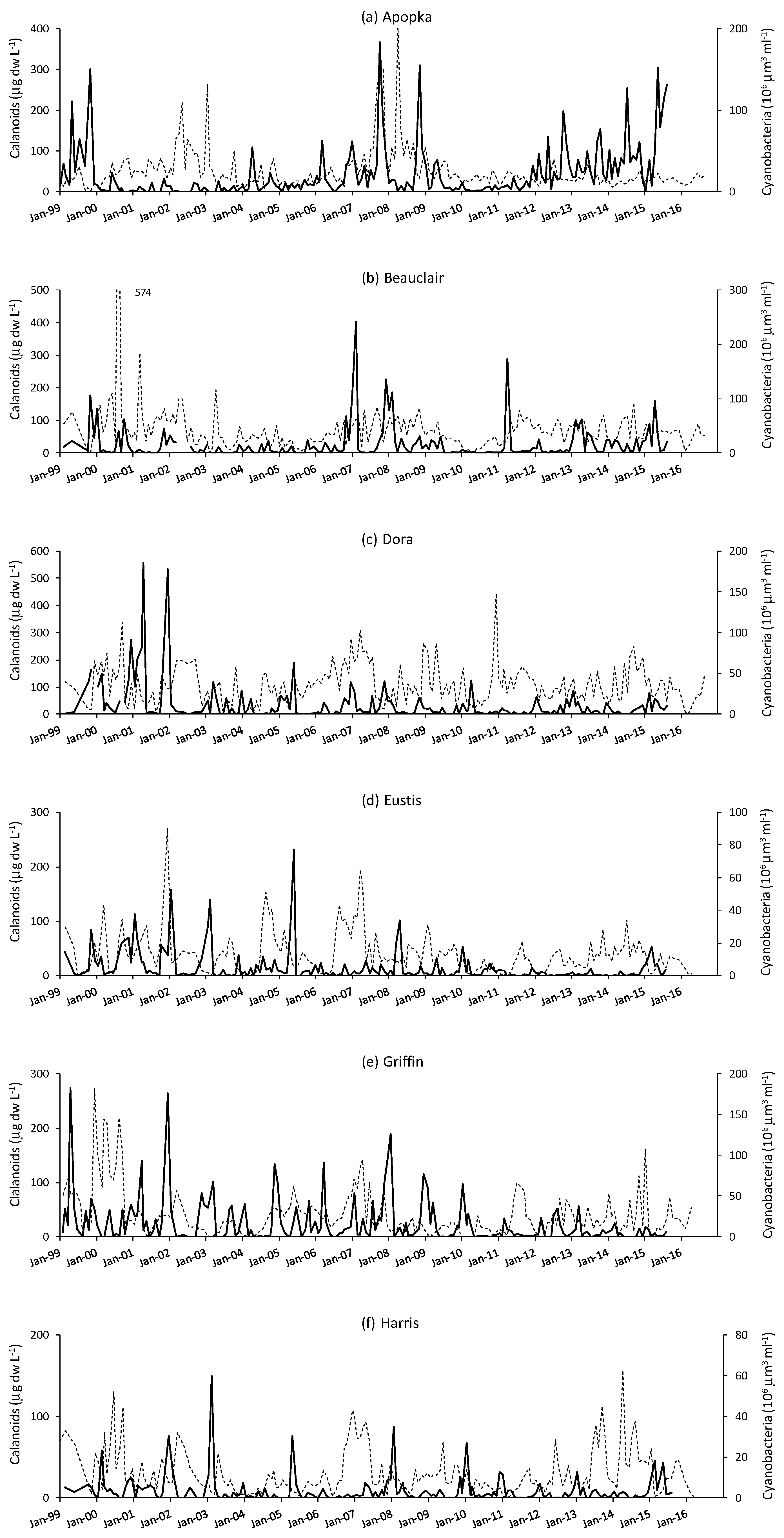
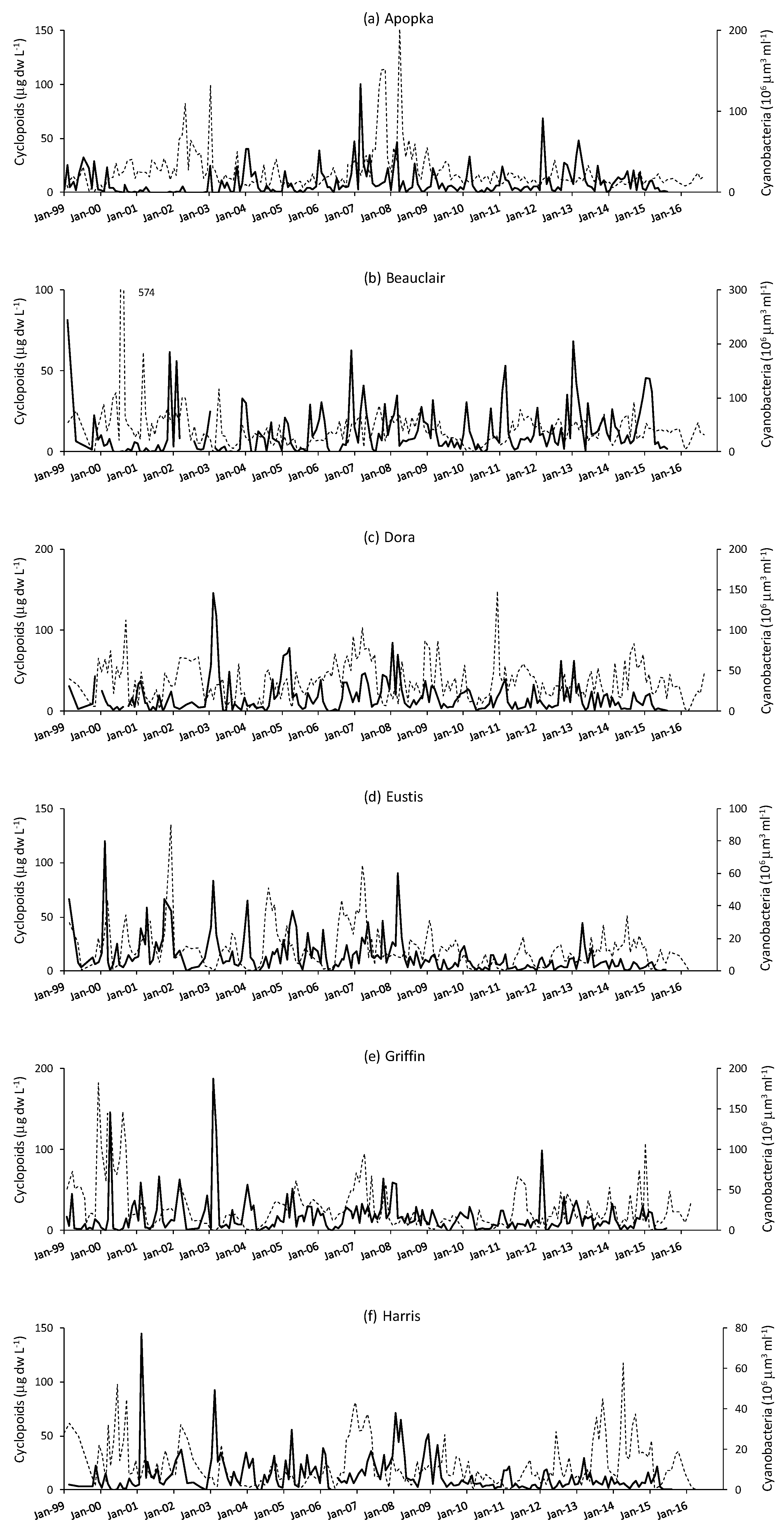
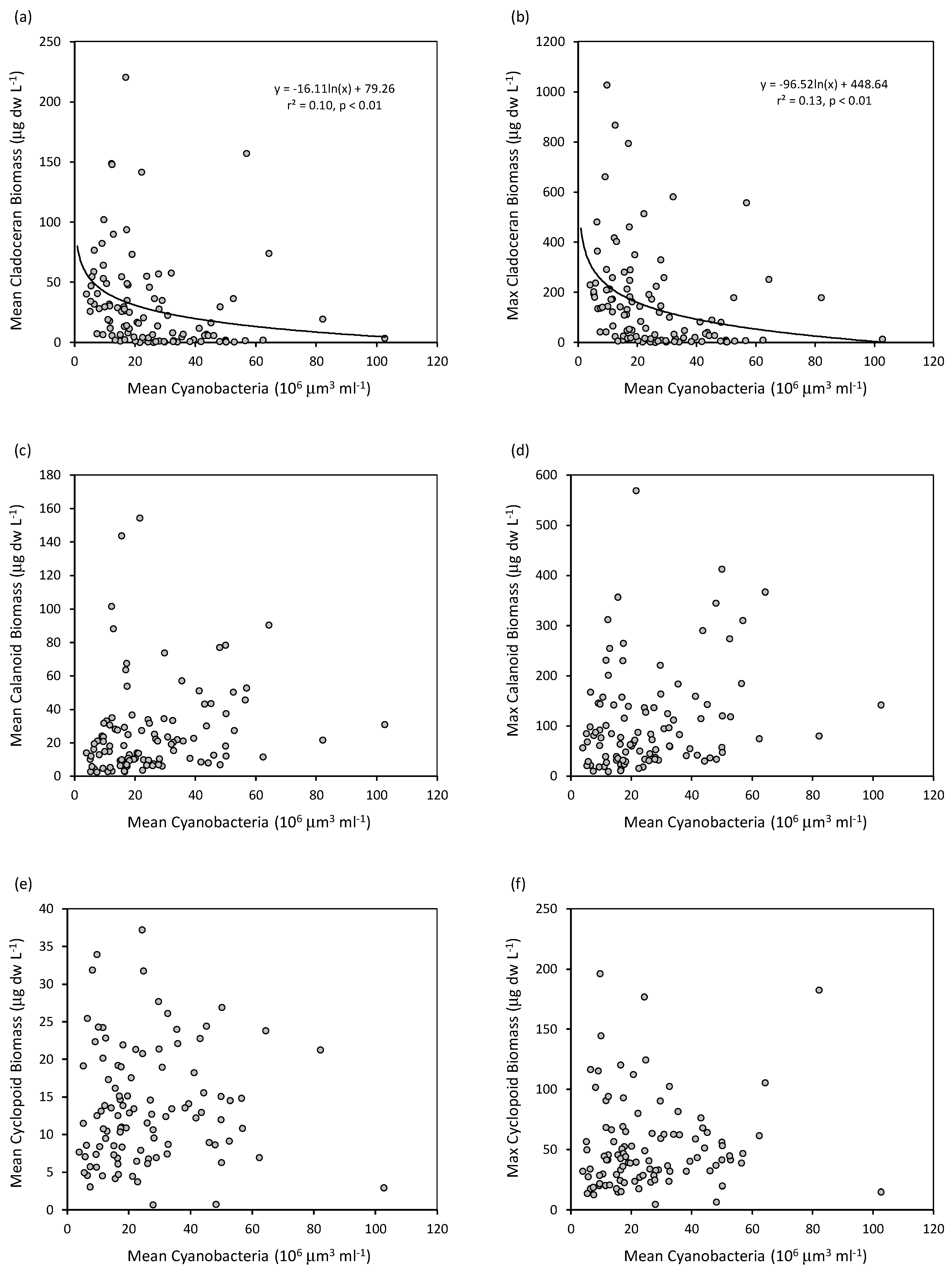
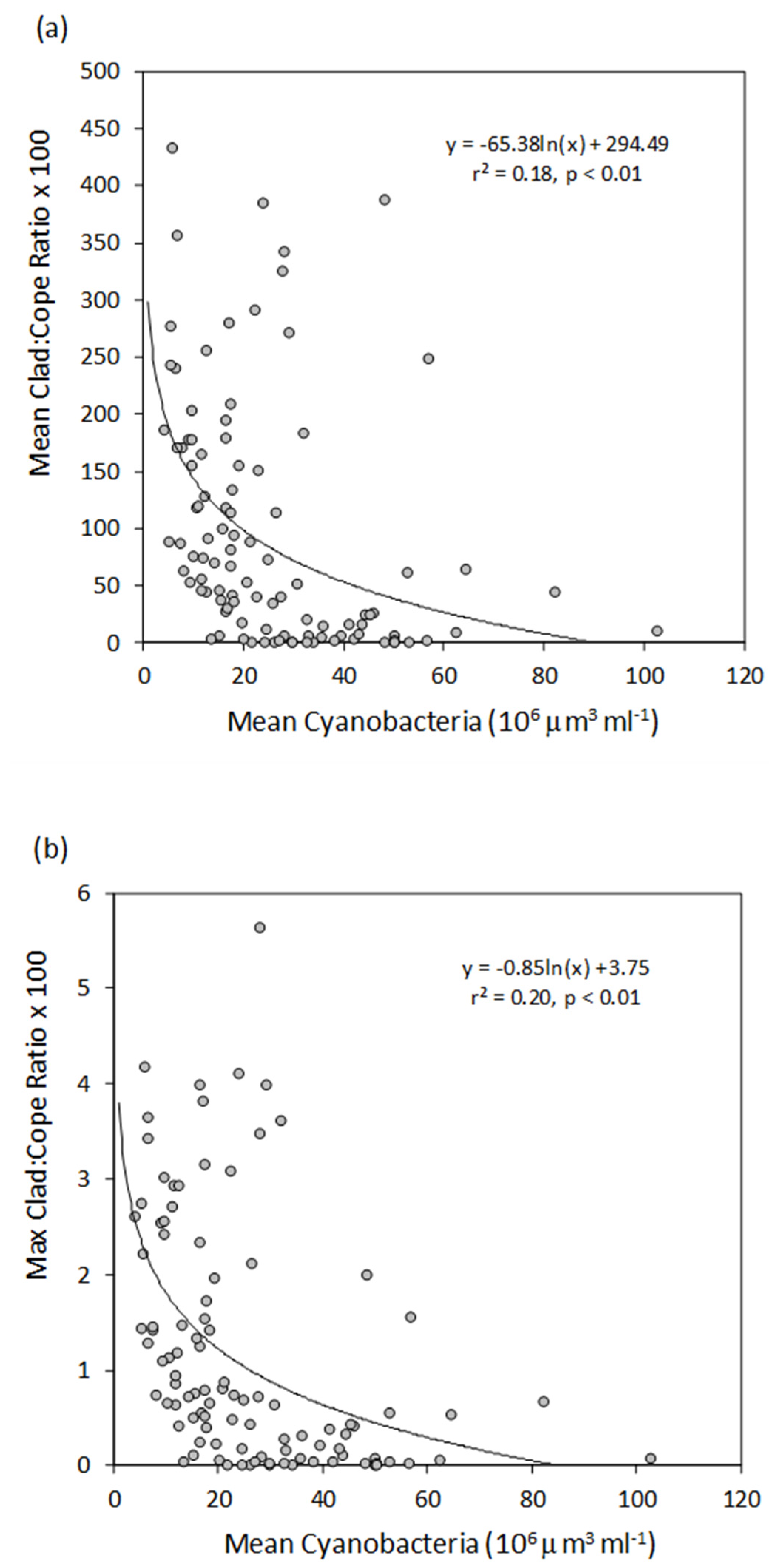
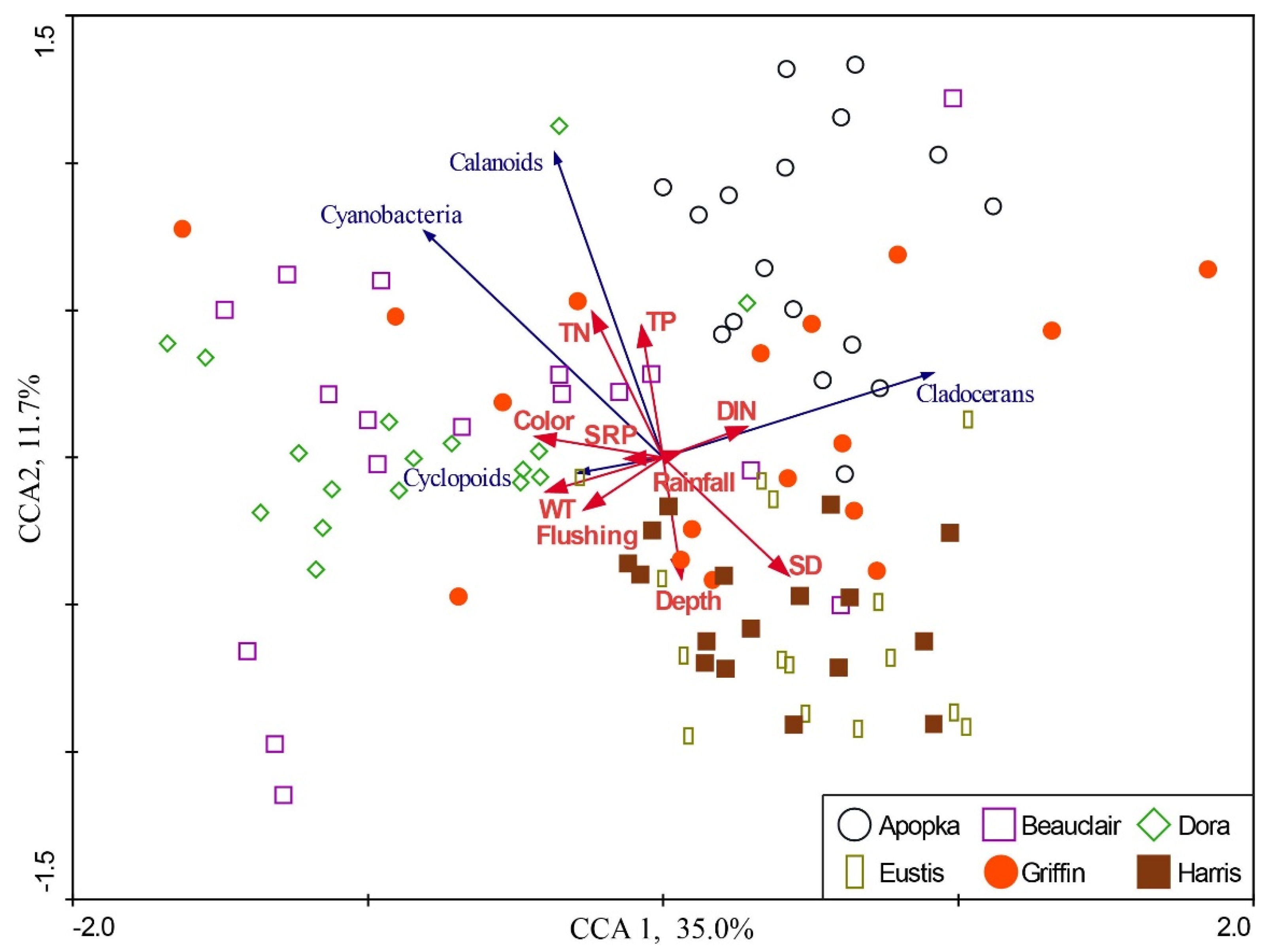
3. Results
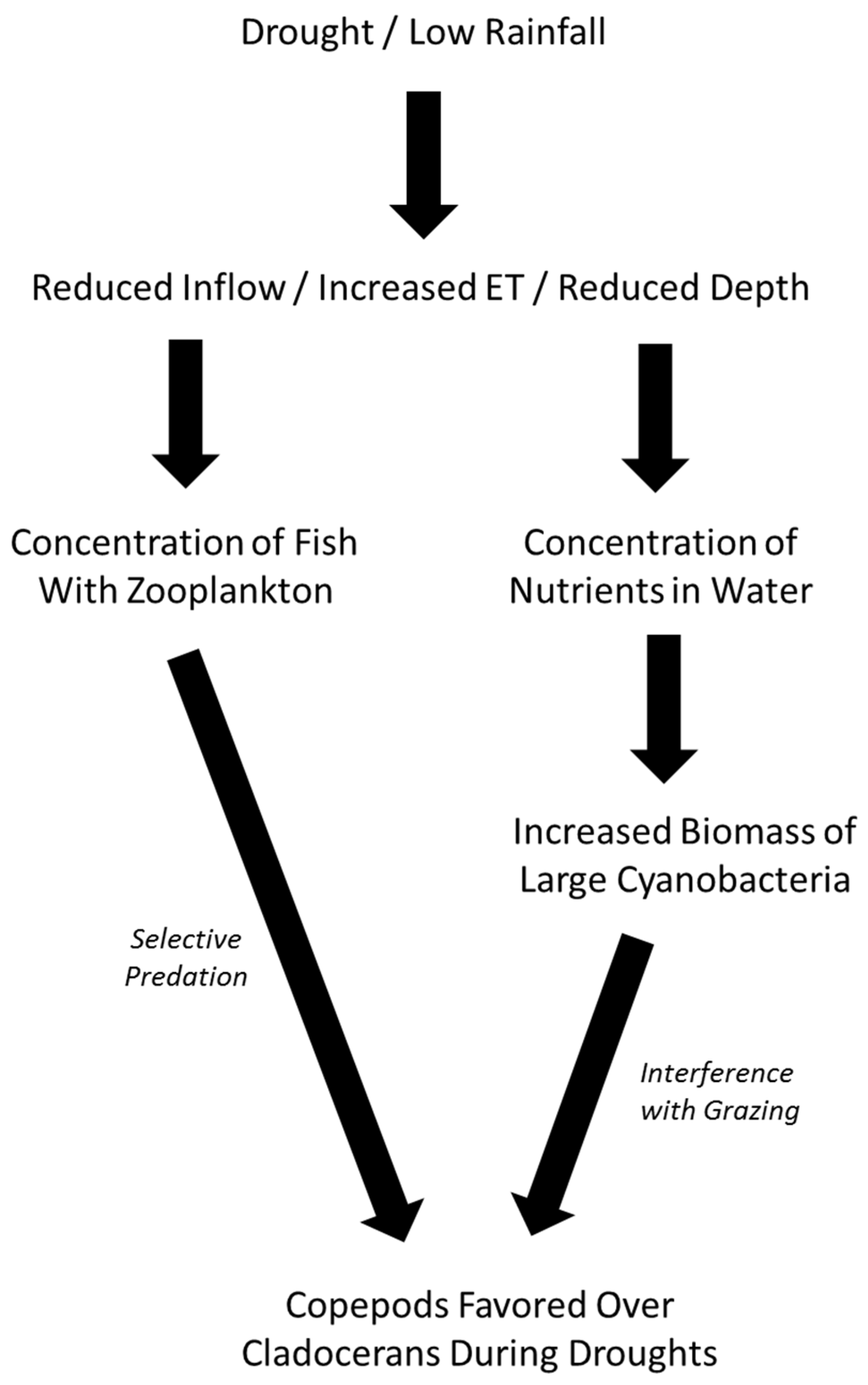
4. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Jeppesen, E.; Meerhoff, M.; Jacobsen, B.A.; Hansen, R.S.; Sondergaard, M.; Jensen, J.P.; Lauridsen, T.L.; Mazzeo, N.; Branco, C.W.C. Restoration of shallow lakes by nutrient control and biomanipulation-the successful strategy varies with lake size and climate. Hydrobiologia 2007, 581, 269–285. [Google Scholar] [CrossRef]
- Havens, K.E.; Beaver, J.R. Composition, size, and biomass of zooplankton in large productive Florida lakes. Hydrobiologia 2011, 668, 49–60. [Google Scholar] [CrossRef]
- Sommer, U.; Adrian, R.; De Senerpont Domis, L.; Elser, J.J.; Gaedke, U.; Ibelings, B.; Jeppesen, E.; Lürling, M.; Molinero, J.C.; Mooij, W.M.; et al. Beyond the plankton ecology group (PEG) model: Mechanisms driving plankton succession. Annu. Rev. Ecol. Evolut. Syst. 2012, 43, 429–448. [Google Scholar] [CrossRef]
- Straile, D. Zooplankton biomass dynamics in oligotrophic versus eutrophic conditions: A test of the PEG model. Freshw. Biol. 2015, 60, 174–183. [Google Scholar] [CrossRef]
- Havens, K.E.; Schelske, C.L. The importance of considering biological processes when setting total maximum daily loads (TMDL) for phosphorus in shallow lakes and reservoirs. Environ. Pollut. 2001, 113, 1–9. [Google Scholar] [CrossRef]
- Marc, B.; Diogo, F.Â.O.; Mauro, M.; Marc, P.; Ariadne, M. Occurrence of Cylindrospermopsis (cyanobacteria) in 39 Brazilian tropical reservoirs during the 1998 drought. Aquat. Microb. Ecol. 2000, 23, 13–27. [Google Scholar]
- Ha, K.; Cho, E.-A.; Kim, H.-W.; Joo, G.-J. Microcystis bloom formation in the lower Nakdong River, South Korea: Importance of hydrodynamics and nutrient loading. Mar. Freshw. Res. 1999, 50, 89–94. [Google Scholar] [CrossRef]
- Van Cleave, K.; Lenters, J.D.; Wang, J.; Verhamme, E.M. A regime shift in Lake Superior ice cover, evaporation, and water temperature following the warm El Niño winter of 1997–1998. Limnol. Oceanogr. 2014, 59, 1889–1898. [Google Scholar] [CrossRef]
- Darchambeau, F.; Borges, A.V.; Sarmento, H.; Leporcq, B.; Isumbisho, P.M.; Alunga, G.; Masilya, P.M.; Descy, J.-P. Teleconnections between ecosystem productivity and climate indices in a tropical great lake. In Proceedings of the EGU General Assembly 2013, Vienna, Austria, 7–12 April 2013. [Google Scholar]
- Abtew, W.; Trimble, P. El Niño–Southern Oscillation link to South Florida hydrology and water management applications. Water Resour. Manag. 2010, 24, 4255–4271. [Google Scholar] [CrossRef]
- Havens, K.E.; Fulton, R.S.; Beaver, J.R.; Samples, E.E.; Colee, J. Effects of climate variability on cladoceran zooplankton and cyanobacteria in a shallow subtropical lake. J. Plankton Res. 2016, 38, 418–430. [Google Scholar] [CrossRef]
- Havens, K.E.; Ji, G.; Beaver, J.R.; Fulton, R.S., III; Teacher, C.E. Dynamics of cyanobacteria blooms are linked to the hydrology of shallow Florida lakes and provide insight into possible impacts of climate change. Hydrobiologia 2017. in review. [Google Scholar]
- Fulton, R.S. Nutrient Loading and Water Quality Trends in the Upper Ocklawaha Basin Lakes through 2014; St. Johns River Water Management District: Palatka, FL, USA, 2016; p. 97. [Google Scholar]
- Coveney, M.F.; Lowe, E.F.; Battoe, L.E.; Marzolf, E.R.; Conrow, R. Response of a eutrophic, shallow subtropical lake to reduced nutrient loading. Freshw. Biol. 2005, 50, 1718–1730. [Google Scholar] [CrossRef]
- Catalano, M.J.; Allen, M.S.; Schaus, M.H.; Buck, D.G.; Beaver, J.R. Evaluating short-term effects of omnivorous fish removal on water quality and zooplankton at a subtropical lake. Hydrobiologia 2010, 655, 159–169. [Google Scholar] [CrossRef]
- Catalano, M.J.; Allen, M.S. A whole-lake density reduction to assess compensatory responses of gizzard shad Dorosoma cepedianum. Can. J. Fish. Aquat. Sci. 2011, 68, 955–968. [Google Scholar] [CrossRef]
- Meerhoff, M.; Iglesias, C.; De Mello, F.T.; Clemente, J.M.; Jensen, E.; Lauridsen, T.L.; Jeppesen, E. Effects of habitat complexity on community structure and predator avoidance behaviour of littoral zooplankton in temperate versus subtropical shallow lakes. Freshw. Biol. 2007, 52, 1009–1021. [Google Scholar] [CrossRef]
- Iglesias, C.; Mazzeo, N.; Meerhoff, M.; Lacerot, G.; Clemente, J.M.; Scasso, F.; Kruk, C.; Goyenola, G.; García-Alonso, J.; Amsinck, S.L.; et al. High predation is of key importance for dominance of small-bodied zooplankton in warm shallow lakes: Evidence from lakes, fish exclosures and surface sediments. Hydrobiologia 2011, 667, 133–147. [Google Scholar] [CrossRef]
- Kopp, J.F.; McKee, G.D. Methods of Chemical Analysis of Water and Waste; Section 9.3, USEPA/600/4-79/020; United States Environmental Protection Agency: Cincinnati, OH, USA, 1983.
- Havens, K.E.; East, T.L.; Beaver, J.R. Experimental studies of zooplankton-phytoplankton-nutrient interactions in a large subtropical lake (Lake Okeechobee, Florida, USA). Freshw. Biol. 1996, 36, 579–597. [Google Scholar] [CrossRef]
- McNabb, C.D. Enumeration of freshwater phytoplankton concentrated on the membrane filter1. Limnol. Oceanogr. 1960, 5, 57–61. [Google Scholar] [CrossRef]
- Hillebrand, H.; Dürselen, C.-D.; Kirschtel, D.; Pollingher, U.; Zohary, T. Biovolume calculation for pelagic and benthic microalgae. J. Phycol. 1999, 35, 403–424. [Google Scholar] [CrossRef]
- McCauley, E. The estimation of the abundance and biomass of zooplankton in samples. In A Manual on Methods for the Assessment of Secondary Productivity in Fresh Waters; Downing, J.A., Rigler, F.H., Eds.; Blackwell Scientific: Oxford, UK, 1984; pp. 228–265. [Google Scholar]
- Ter Braak, C.J.F.; Smilauer, P. Canoco Reference Manual and User’s Guide to Canoco for Windows: Software for Canonical Community Ordination (Version 4.5); Microcomputer Power: Ithaca, NY, USA, 2002. [Google Scholar]
- Jeppesen, E.; Meerhoff, M.; Davidson, T.A.; Trolle, D.; Sondergaard, M.; Lauridsen, T.L.; Beklioglu, M.; Brucet, S.; Volta, P.; Gonzalez-Bergonzoni, I.; et al. Climate change impacts on lakes: An integrated ecological perspective based on a multi-faceted approach, with special focus on shallow lakes. J. Limnol. 2014, 73, 88–111. [Google Scholar] [CrossRef]
- Havens, K.E.; Sharfstein, B.; Brady, M.A.; East, T.L.; Harwell, M.C.; Maki, R.P.; Rodusky, A.J. Recovery of submerged plants from high water stress in a large subtropical lake in Florida, USA. Aquat. Bot. 2004, 78, 67–82. [Google Scholar] [CrossRef]
- Cai, W.; Santoso, A.; Wang, G.; Yeh, S.-W.; An, S.-I.; Cobb, K.M.; Collins, M.; Guilyardi, E.; Jin, F.-F.; Kug, J.-S.; et al. ENSO and greenhouse warming. Nat. Clim. Chang. 2015, 5, 849–859. [Google Scholar] [CrossRef]
- Yen, J.; Murphy, D.W.; Fan, L.; Webster, D.R. Sensory-motor systems of copepods involved in their escape from suction feeding. Integr. Comp. Biol. 2015, 55, 121–133. [Google Scholar] [CrossRef] [PubMed]
- Fulton, R.S.; Godwin, W.F.; Schaus, M.H. Water quality changes following nutrient loading reduction and biomanipulation in a large shallow subtropical lake, Lake Griffin, Florida, USA. Hydrobiologia 2015, 753, 243–263. [Google Scholar] [CrossRef]
- Schaus, M.H.; Vanni, M.J.; Wissing, T.E.; Bremigan, M.T.; Garvey, J.E.; Stein, R.A. Nitrogen and phosphorus excretion by detritivorous gizzard shad in a reservoir ecosystem. Limnol. Oceanogr. 1997, 42, 1386–1397. [Google Scholar] [CrossRef]
- DeMott, W.R.; Moxter, F. Foraging cyanobacteria by copepods: Responses to chemical defense and resource abundance. Ecology 1991, 72, 1820–1834. [Google Scholar] [CrossRef]
- Marc, B.; Marc, P.; Marc, T. Effects of a cyanobacterial bloom (Cylindrospermopsis raciborskii) on bacteria and zooplankton communities in ingazeira reservoir (Northeast Brazil). Aquat. Microb. Ecol. 2001, 25, 215–227. [Google Scholar]
- Koski, M.; Schmidt, K.; Engstrom-Ost, J.; Viitasalo, M.; Jonasdottir, S.; Repka, S.; Sivonen, K. Calanoid copepods feed and produce eggs in the presence of toxic cyanobacteria Nodularia spumigena. Limnol. Oceanogr. 2002, 47, 878–885. [Google Scholar]
- Ger, K.A.; Urrutia-Cordero, P.; Frost, P.C.; Hansson, L.-A.; Sarnelle, O.; Wilson, A.E.; Lürling, M. The interaction between cyanobacteria and zooplankton in a more eutrophic world. Harmful Algae 2016, 54, 128–144. [Google Scholar] [CrossRef] [PubMed]
- DeMott, W.R. The role of taste in food selection by freshwater zooplankton. Oecologia 1986, 69, 334–340. [Google Scholar] [CrossRef] [PubMed]
- Fabre, A.; Lacerot, G.; de Paiva, R.R.; Soares, M.C.S.; de Magalhães, V.F.; Bonilla, S. South American PSP toxin-producing Cylindrospermopsis raciborskii (Cyanobacteria) decreases clearance rates of cladocerans more than copepods. Hydrobiologia 2017, 785, 61–69. [Google Scholar] [CrossRef]
- Rohrlack, T.; Dittmann, E.; Borner, T.; Christoffersen, K. Effects of cell-bound microcystins on survival and feeding of Daphnia spp. Appl. Environ. Microbiol. 2001, 67, 3523–3529. [Google Scholar] [CrossRef] [PubMed]
- Ghadouani, A.; Pinel-Alloul, B.; Prepas, E.E. Effects of experimentally induced cyanobacterial blooms on crustacean zooplankton communities. Freshw. Biol. 2003, 48, 363–381. [Google Scholar] [CrossRef]
- Paerl, H.W.; Fulton, R.S. Ecology of harmful cyanobacteria. In Ecology of Harmful Algae; Granéli, E., Turner, J.T., Eds.; Springer: Berlin/Heidelberg, Germany, 2006; pp. 95–109. [Google Scholar]
- Lurling, M.; van der Grinten, E. Life-history characteristics of Daphnia exposed to dissolved microcystin-LR and to the cyanobacterium Microcystis aeruginosa with and without microcystins. Environ. Toxicol. Chem. 2003, 22, 1281–1287. [Google Scholar] [CrossRef] [PubMed]
- Hansson, L.-A.; Gustafsson, S.; Rengefors, K.; Bomark, L. Cyanobacterial chemical warfare affects zooplankton community composition. Freshw. Biol. 2007, 52, 1290–1301. [Google Scholar] [CrossRef]
- Perga, M.E.; Domaizon, I.; Guillard, J.; Hamelet, V.; Anneville, O. Are cyanobacterial blooms trophic dead ends? Oecologia 2013, 172, 551–562. [Google Scholar] [CrossRef] [PubMed]

| Lake | Area (km2) | Mean/Max Depth (m) | TP (μg L−1) | TN (mg L−1) | Chl-a (μg L−1) | Secchi (m) |
|---|---|---|---|---|---|---|
| Apopka | 127 | 1.4/5.2 | 131 | 4.7 | 65 | 0.3 |
| Beauclair | 5 | 1.8/4.4 | 97 | 3.7 | 115 | 0.4 |
| Dora | 17 | 2.8/5.2 | 56 | 3.3 | 97 | 0.4 |
| Eustis | 32 | 3.1/6.6 | 38 | 2.0 | 35 | 0.7 |
| Griffin | 36 | 2.1/6.1 | 54 | 2.8 | 66 | 0.5 |
| Harris | 75 | 3.4/7.9 | 38 | 1.8 | 37 | 0.6 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ji, G.; Havens, K.E.; Beaver, J.R.; Fulton, R.S., III. Response of Zooplankton to Climate Variability: Droughts Create a Perfect Storm for Cladocerans in Shallow Eutrophic Lakes. Water 2017, 9, 764. https://doi.org/10.3390/w9100764
Ji G, Havens KE, Beaver JR, Fulton RS III. Response of Zooplankton to Climate Variability: Droughts Create a Perfect Storm for Cladocerans in Shallow Eutrophic Lakes. Water. 2017; 9(10):764. https://doi.org/10.3390/w9100764
Chicago/Turabian StyleJi, Gaohua, Karl E. Havens, John R. Beaver, and Rolland S. Fulton, III. 2017. "Response of Zooplankton to Climate Variability: Droughts Create a Perfect Storm for Cladocerans in Shallow Eutrophic Lakes" Water 9, no. 10: 764. https://doi.org/10.3390/w9100764
APA StyleJi, G., Havens, K. E., Beaver, J. R., & Fulton, R. S., III. (2017). Response of Zooplankton to Climate Variability: Droughts Create a Perfect Storm for Cladocerans in Shallow Eutrophic Lakes. Water, 9(10), 764. https://doi.org/10.3390/w9100764






