Prey Survival and Vulnerability of Juvenile Rhynchocypris oxycephalus in Juvenile Fish Shelters under Predation by Korean Native Piscivorous Fish (Coreoperca herzi)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Installation of Experimental Equipment
2.2. Water Quality Analysis
2.3. Experimental Fish
2.4. Quantification of the Predation Sequence
2.5. Statistical Analysis
3. Results and Discussion
3.1. Changes in the Water Quality
3.2. Changes in Body Size and Weight
3.3. Prey Survival Results
3.4. Statistical Analysis Results
3.5. Performance Evaluation of the Developed JFS in Terms of Vulnerability
3.6. Correlation of Vulnerability and Prey Survival
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bailey, K.M.; Duffy-Anderson, J.T. Fish predation and mortality. In Elements of Physical Oceanography: A Derivative of the Encyclopedia of Ocean Sciences; Steele, J.H., Thorpe, S.A., Turekian, K.K., Eds.; Elsevier Ltd.: London, UK, 2009; pp. 322–329. [Google Scholar]
- Grorud-Colvert, K.; Sponaugle, S. Larval supply and juvenile recruitment of coral reef fishes to marine reserves and non-reserves of the upper Florida Keys, USA. Mar. Biol. 2009, 156, 277–288. [Google Scholar] [CrossRef]
- Michael, J.H.; Hickford, M.C.; David, R.S. Predation, vegetation and habitat-specific survival of terrestrial eggs of a diadromous fish, Galaxias maculatus (Jenyns, 1842). J. Exp. Mar. Biol. Ecol. 2010, 385, 66–72. [Google Scholar]
- Sogard, S.M. Size-selective mortality in the juvenile stage of teleost fishes: A review. Bull. Mar. Sci. 1997, 60, 1129–1157. [Google Scholar]
- Damsgard, B.; Dill, L. Risk-taking behavior in weight-compensating coho salmon, Oncorhynchus kisutch. Behav. Ecol. 1998, 9, 26–32. [Google Scholar] [CrossRef]
- Magnhagen, C. Predation risk and foraging in juvenile pink (Oncorhynchus gorbusha) and chum salmon (O. keta). Can. J. Fish. Aquat. Sci. 1988, 45, 592–596. [Google Scholar] [CrossRef]
- Pettersson, L.B.; Brönmark, C. Trading off safety against food: State dependent habitat choice and foraging in crucian carp. Oecologia 1993, 95, 353–357. [Google Scholar] [CrossRef]
- Holmlund, C.M.; Hammer, M. Ecosystem services generated by fish populations. Ecol. Econ. 1999, 29, 253–268. [Google Scholar] [CrossRef]
- Choi, J.; Kumar, H.K.; Han, J.; An, K. The development of a regional multimetric fish model based on biological integrity in lotic ecosystems and some factors influencing the stream hearth. Water Air Soil Pollut. 2011, 271, 3–24. [Google Scholar] [CrossRef]
- Han, J.; Paek, W.K.; An, K. Exotic species, Micropterus salmoides as a key bioindicator influencing the reservoir health and fish community structure. J. Asia-Pac. Biodivers. 2016, 9, 403–411. [Google Scholar] [CrossRef]
- MacRae, P.S.D.; Jackson, D.A. The influence of smallmouth bass (Micropterus dolomieu) predation and habitat complexity on the structure of littoral zone fish assemblages. Can. J. Fish. Aquat. Sci. 2001, 58, 342–351. [Google Scholar] [CrossRef]
- Garcia-Berthou, E.; Moreno-Amich, R. Introduction of exotic fish into a mediterranean lake over a 90-year period. Arch. Hydrobiol. 2000, 149, 271–284. [Google Scholar]
- Suttle, K.B.; Power, M.E.; Levine, J.M.; McNeely, C. How fine sediment in riverbed impairs growth and survival of juvenile salmonids. Ecol. Appl. 2004, 14, 969–974. [Google Scholar] [CrossRef]
- Finstad, A.G.; Einum, S.; Forseth, T.; Ugedal, O. Shelter availability affects behaviour, size-dependent and mean growth of juvenile Atlantic salmon. Freshw. Biol. 2007, 52, 1710–1718. [Google Scholar] [CrossRef]
- Fischer, P. An experimental test of metabolic and behavioural responses of benthic fish species to different types of substrate. Can. J. Fish. Aquat. Sci. 2000, 57, 2336–2344. [Google Scholar] [CrossRef]
- Matsuzaki, S.S.; Sakamoto, M.; Kawabe, K.; Takamura, N. A laboratory study of the effects of shelter availability and invasive crayfish on the growth of native stream fish. Freshw. Biol. 2012, 57, 874–882. [Google Scholar] [CrossRef]
- Kieffer, J.D.; Colgan, P.W. Differences in learning by foraging juvenile pumpkinseed and bluegill sunfish in a structured habitat. Environ. Biol. Fish. 1992, 33, 359–366. [Google Scholar] [CrossRef]
- Layman, C.A.; Carmen, G.M.; Jacob, E.A. Linking fish colonization rates and water level change in littoral habitats of a Venezuelan floodplain river. Aquat. Ecol. 2010, 44, 269–273. [Google Scholar] [CrossRef]
- D'Anna, G.; Giacalone, V.M.; Fernádez, T.V.; Vaccaro, A.M.; Pipitone, C.; Mirto, S.; Mazzola, S.; Badalamenti, F. Effects of predator and shelter conditioning on hatchery-reared white seabream Diplodus sargus (L., 1758) released at sea. Aquaculture 2012, 356, 91–97. [Google Scholar] [CrossRef]
- Moriniére, E.C.; Nagelkerken, I.; Meij, H.; Velde, G. What attracts juvenile coral reef fish to mangroves: Habitat complexity or shade? Mar. Biol. 2004, 144, 139–145. [Google Scholar] [CrossRef]
- Ferter, K.; Meyer-Rochow, V.B. Turning night into day: Effects of stress on the self-feeding behavior of the eurasian perch Perca fluviatilis. Zool. Stud. 2010, 49, 176–181. [Google Scholar]
- Fraser, D.F.; Cerri, R.D. Experimental evaluation of predator–prey relationships in a patchy environment: Consequences for habitat use patterns in minnows. Ecology 1982, 63, 307–313. [Google Scholar] [CrossRef]
- Mittelbach, G.G. Predator-mediated habitat use: Some consequences for species interactions. Environ. Biol. Fish. 1986, 7, 159–170. [Google Scholar] [CrossRef]
- Werner, E.E.; Mittlebach, G.G.; Hall, D.J.; Gilliam, J.F. Experimental tests of optimal habitat use in fish: The role of relative habitat profitability. Ecology 1983, 64, 1525–1539. [Google Scholar] [CrossRef]
- Savino, J.F.; Stein, R.A. Behavioural interactions between fish predators and their prey: Effects of plant density. Anim. Behav. 1989, 37, 311–321. [Google Scholar] [CrossRef]
- Richmond, H.E.; Hrabik, T.R.; Mensinger, A.F. Light intensity, prey detection and foraging mechanisms of age 0 year yellow perch. J. Fish. Biol. 2004, 65, 195–205. [Google Scholar] [CrossRef]
- American Public Health Association (APHA). Standard Methods for the Examination of Water and Wastewater, 21th ed.; APHA: Washington, DC, USA, 2005; pp. 9–72. [Google Scholar]
- Yu, D.; Chen, M.; Zhou, Z.; Eric, R.; Tang, Q.; Liu, H. Global climate change will severely decrease potential distribution of the East Asian coldwater fish Rhynchocypris oxycephalus (Actinopterygii, Cyprinidae). Hydrobiologia 2013, 700, 23–32. [Google Scholar] [CrossRef]
- Kim, H.C.; Kim, M.S.; Yu, H.S. Biological control of vector mosquitoes by the use of fish predators, Moroco oxycephalus and Misgurnus anguillicaudatus in the laboratory and semi–field rice paddy. Korean J. Entomol. 1994, 24, 269–284. [Google Scholar]
- Jang, S.H.; Ryu, H.S.; Lee, J.H. A study on the stock assessment and management implications of the Korean aucha perch (Coreoperca herzi) in freshwater: (1) estimation of population ecological characteristics of Coreoperca herzi in the mid–upper system of the Seomjin River. Korean J. Limnol. 2003, 43, 82–90. [Google Scholar]
- Gye, M.C. Spermatogenesis of Coreoperca herzi (Perciformes; Percichthyidae). Korean J. Limnol. 2002, 35, 232–236. [Google Scholar]
- Fuiman, L.A.; Magurran, A.E. Development of predator defences in fishes. Rev. Fish. Biol. Fish. 1994, 4, 145–183. [Google Scholar] [CrossRef]
- Fuiman, L.A. Vulnerability of Atlantic herring larvae to predation by yearling herring. Mar. Ecol. Prog. Ser. 1989, 51, 291–299. [Google Scholar] [CrossRef]
- Scharf, F.S.; Buckel, J.A.; McGinn, P.A.; Juanes, F. Vulnerability of marine forage fishes to piscivory: Effects of prey behavior on susceptibility to attack and capture. J. Exp. Mar. Biol. Ecol. 2003, 294, 41–59. [Google Scholar] [CrossRef]
- Gates, C.E. What really is experimental error in block designs? Am. Stat. 1995, 49, 362–363. [Google Scholar] [CrossRef]
- Bailey, R.A. Design of Comparative Experiments; Cambridge University Press: Cambridge, UK, 2008; pp. 53–71. [Google Scholar]
- Xu, H.; Paerl, H.W.; Qin, B.; Zhu, G.; Gao, G. Nitrogen and phosphorus inputs control phytoplankton growth in eutrophic lake Taihu, China. Limnol. Oceanogr. 2010, 55, 420–432. [Google Scholar] [CrossRef]
- Donald, D.B.; Bogard, M.J.; Finlay, K.; Bunting, L.; Leavitt, P.R. Phytoplankton-specific response to enrichment of phosphorus-rich surface waters with ammonium, nitrate, and urea. PLoS ONE 2013, 8, e53277. [Google Scholar] [CrossRef] [PubMed]
- Lazzaro, X. A review of planktivorous fishes: Their evolution, feeding behaviours, selectivities, and impacts. Hydrobiologia 1987, 146, 97–167. [Google Scholar] [CrossRef]
- Tonn, W.M.; Paszkowski, C.A.; Holopainen, I.J. Piscivory and recruitment: Mechanisms structuring prey populations in small lakes. Ecology 1992, 73, 951–958. [Google Scholar] [CrossRef]
- Van Wassenbergh, S.; De Rechter, D. Piscivorous cyprinid fish modulates suction feeding kinematics to capture elusive prey. Zoology 2011, 114, 46–52. [Google Scholar] [CrossRef] [PubMed]
- Eby, L.A.; Rudstam, L.G.; Kitchell, J.F. Predator responses to prey population dynamics: An empirical analysis based on lake trout growth rates. Can. J. Fish. Aquat. Sci. 1995, 52, 1564–1571. [Google Scholar] [CrossRef]
- Sullivan, M.G. Handbook of Fish Biology and Fisheries. Volume 1: Fish Biology. Volume 2: Fisheries. Fish and Fisheries; Blackwell Science Ltd.: Oxford, UK, 2004; pp. 1–96. [Google Scholar]
- Stefan, H.G.; Fang, X.; Eaton, J.G. Simulated fish habitat changes in North American lakes in response to projected climate warming. Trans. Am. Fish. Soc. 2001, 130, 459–477. [Google Scholar] [CrossRef]
- Olson, M.H. Ontogenetic shifts in largemouth bass: Variability and consequences for first-year growth. Ecology 1996, 77, 179–190. [Google Scholar] [CrossRef]
- Buijse, A.D.; Houthuijzen, R.P. Piscivory, growth, and size-selective mortality of age 0 pikeperch (Stizostedion lucioperca). Can. J. Fish. Aquat. Sci. 1992, 49, 894–902. [Google Scholar] [CrossRef]
- Kerfoot, W.C.; Sih, A. Predation: Direct and Indirect Impacts on Aquatic Communities; University Press of New England: Hanover, NH, USA, 1987; pp. 1–386. [Google Scholar]
- Resetarits, W.J. Ecological interactions among predators in experimental stream communities. Ecology 1991, 72, 1782–1793. [Google Scholar] [CrossRef]
- Werner, E.E.; Gilliam, J.F.; Hall, D.J.; Mittelbach, G.M. An experimental test of the effects of predation risk on habitat use in fish. Ecology 1983, 64, 1540–1548. [Google Scholar] [CrossRef]
- Sih, A. Foraging strategies and the avoidance of predation by an aquatic insect, Notonecta hoffmanni. Ecology 1982, 63, 786–796. [Google Scholar] [CrossRef]
- Lundvall, D.; Svanbäk, R.; Persson, L.; Byström, P. Size-dependent predation in piscivores: Interactions between predator foraging and prey avoidance abilities. Can. J. Fish. Aquat. Sci. 1999, 56, 1285–1292. [Google Scholar] [CrossRef]
- Hershey, A.E. Effects of predatory sculpin on the chironomid communities in an arctic lake. Ecology 1985, 66, 1131–1138. [Google Scholar] [CrossRef]
- Coull, B.C.; Wells, J.B.J. Refuges from fish predation: Experiments with phytal meiofauna from the New Zealand rocky intertidal. Ecology 1983, 64, 1599–1609. [Google Scholar] [CrossRef]
- Savino, J.F.; Stein, R.A. Predator-prey interaction between largemouth bass and bluegills as influenced by simulated, submerged vegetation. Trans. Am. Fish. Soc. 1982, 111, 255–266. [Google Scholar] [CrossRef]
- Laurel, B.J.; Gregory, R.S.; Brown, J.A. Predator distribution and habitat patch area determine predation rates on Age-0 juvenile cod Gadus spp. Mar. Ecol. Prog. Ser. 2003, 251, 245–254. [Google Scholar] [CrossRef]
- Main, K.L. Predator avoidance in seagrass meadows: Prey behavior, microhabitat selection, and cryptic coloration. Ecology 1987, 68, 170–180. [Google Scholar] [CrossRef]
- Savino, J.F.; Stein, R.A. Behavior of fish predators and their prey: Habitat choice between open water and dense vegetation. Environ. Biol. Fish. 1989, 24, 287–293. [Google Scholar] [CrossRef]
- Magurran, A.E. The adaptive significance of schooling as an anti-predator defense in fish. Ann. Zool. Fenni. 1990, 27, 51–66. [Google Scholar]

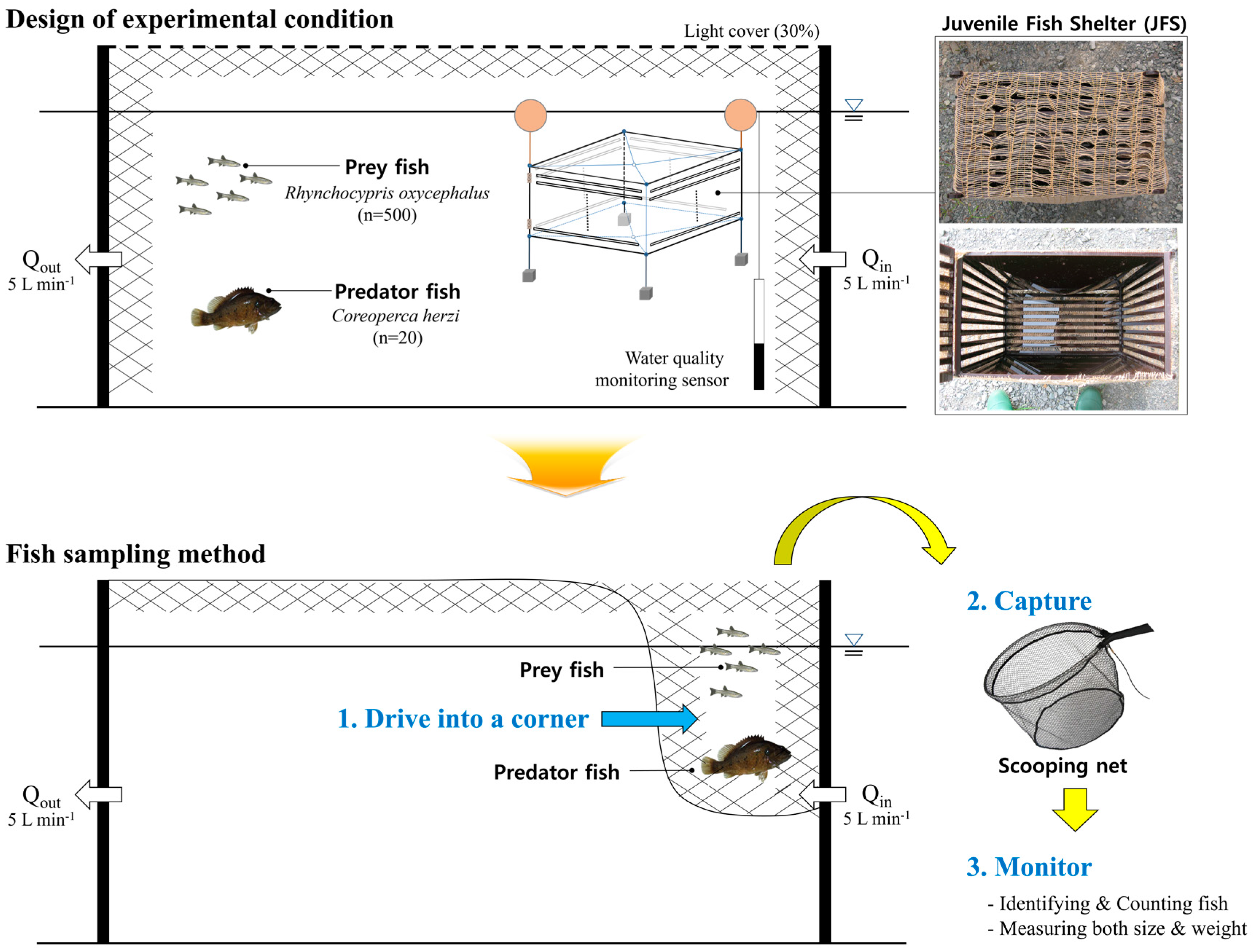

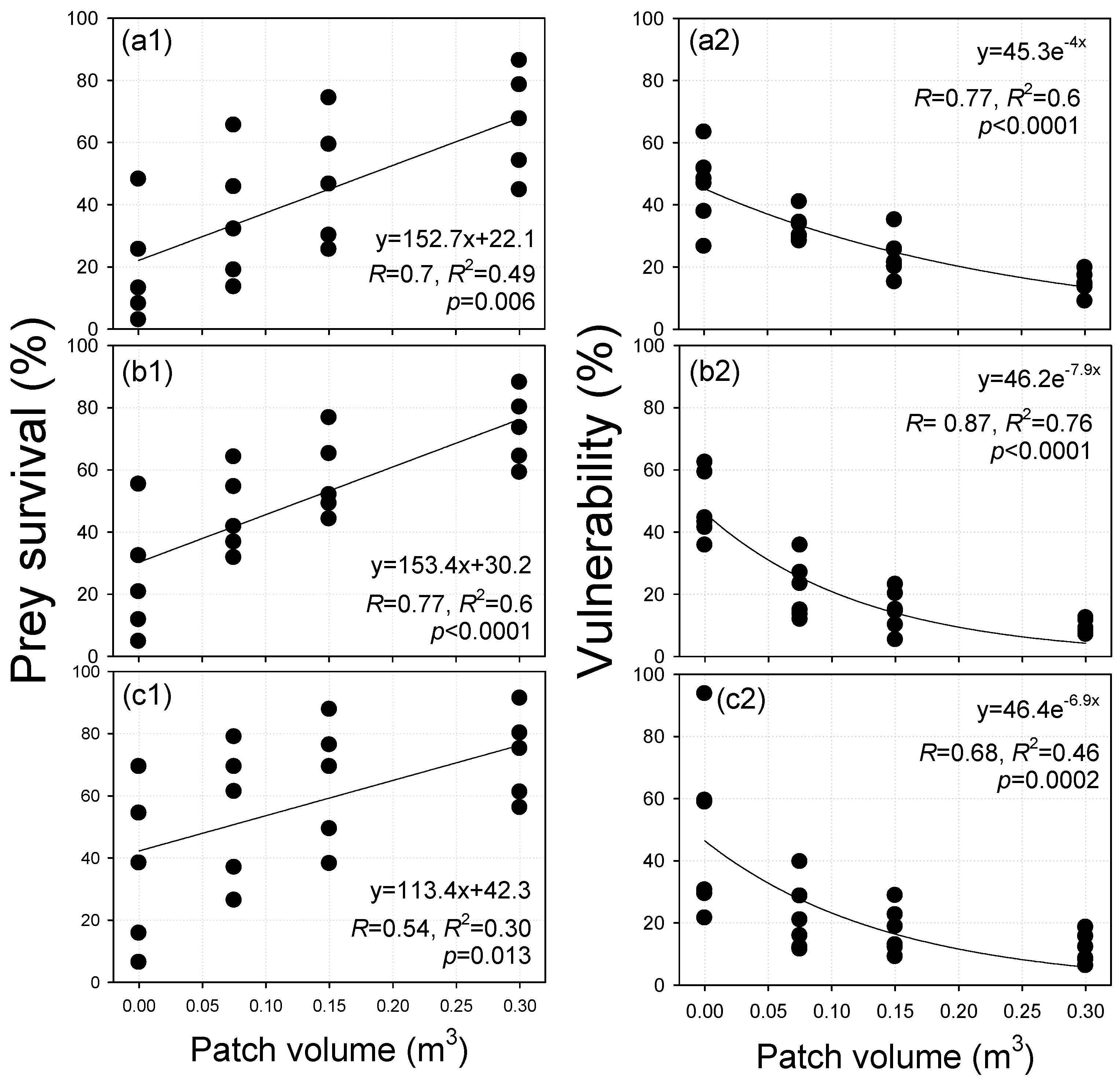
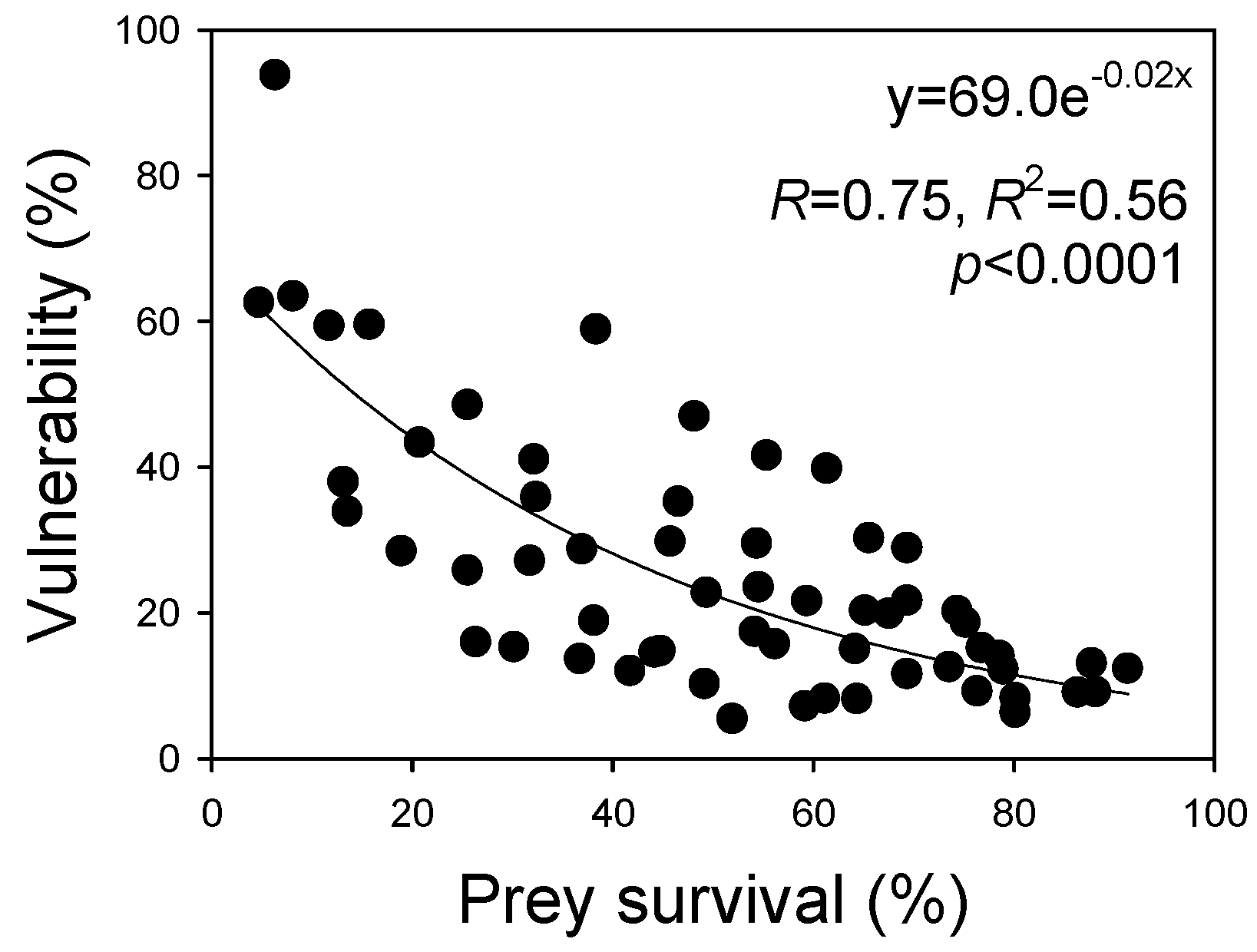
| Description | Control | T1 | T2 | T3 | |
|---|---|---|---|---|---|
| Test bed Physical condition | Water volume (m3) | 8.0 | 8.0 | 8.0 | 8.0 |
| Water depth (cm) | 80 | 80 | 80 | 80 | |
| Input & output flow rate (L·min−1) | 5.0 | 5.0 | 5.0 | 5.0 | |
| Net size (mm) | 2 | 2 | 2 | 2 | |
| Light transmissivity (%) | 30 | 30 | 30 | 30 | |
| Fish shelter | Number of patches (JFS) | 0 | 1 | 2 | 4 |
| Patch volume (m3) | 0 | 0.08 | 0.15 | 0.30 | |
| Patch area (m2) | 0 | 0.25 | 0.50 | 1.00 | |
| Experimental fish | Number of prey fish (individual) b | 500 | 500 | 500 | 500 |
| Size of prey fish (mm) b | 28.9 ± 1.0 | 29.3 ± 0.9 | 29.2 ± 0.9 | 29.3 ± 0.9 | |
| Number of predator fish (individual) c | 20 | 20 | 20 | 20 | |
| Size of predator fish (mm) c | 88.9 ± 2.0 | 88.8 ± 1.5 | 88.6 ± 1.3 | 88.8 ± 2.1 | |
| Description | Period 1 c | Period 2 d | Period 3 e |
|---|---|---|---|
| Rainfall (mm) | 108 | 460 | 98 |
| Water depth (cm) a | 84 ± 4 | 85 ± 5 | 84 ± 3 |
| Water temperature (°C) a | 25.6 ± 2.0 | 16.0 ± 2.5 | 19.7 ± 3.3 |
| pH a | 8.7 ± 0.6 | 9.2 ± 0.3 | 9.1 ± 0.3 |
| DO (mg O2 L−1) a | 11.4 ± 1.3 | 13.5 ± 1.7 | 12.7 ± 0.5 |
| EC (μS·cm−1) a | 155 ± 17 | 187 ± 10 | 163 ± 11 |
| SS (mg·L−1) b | 1.0 ± 0.2 | 0.9 ± 0.1 | 1.7 ± 0.6 |
| BOD (mg O2 L−1) b | 1.3 ± 0.1 | 1.1 ± 0.1 | 1.1 ± 0.1 |
| TN (mg N L−1) b | 1.590 ± 0.090 | 1.510 ± 0.080 | 1.520 ± 0.050 |
| NH4+ (mg N L−1) b | 0.114 ± 0.012 | 0.119 ± 0.009 | 0.131 ± 0.010 |
| NO3− (mg N L−1) b | 1.390 ± 0.021 | 1.403 ± 0.040 | 1.419 ± 0.008 |
| TP (mg P L−1) b | 0.050 ± 0.004 | 0.049 ± 0.005 | 0.056 ± 0.001 |
| PO43− (mg P L−1) b | 0.026 ± 0.001 | 0.023 ± 0.001 | 0.024 ± 0.002 |
| Phytoplankton (cells mL−1) b | 908 ± 184 | 526 ± 159 | 712 ± 63 |
| Zooplankton (ind L−1) b | 5.3 ± 2.8 | 2.8 ± 2.2 | 1.5 ± 0.6 |
| Description | R. oxycephalus | C. herzi | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| IBS a (mm) | FBS b (mm) | GR c (%) | IBW d (g) | FBW e (g) | GR c (%) | IBS a (mm) | FBS b (mm) | GR c (%) | IBW d (g) | FBW e (g) | GR c (%) | ||
| Period 1 | Control | 28.5 ± 1.0 | 29.3 ± 0.7 | 2.6 | 0.39 ± 0.01 | 0.44 ± 0.01 | 10.4 | 88.7 ± 2.0 | 90.8 ± 2.8 | 2.4 | 18.5 ± 0.5 | 21.2 ± 1.7 | 12.4 |
| T1 | 28.7 ± 0.9 | 29.9 ± 0.7 | 3.9 | 0.39 ± 0.01 | 0.44 ± 0.01 | 10.0 | 89.2 ± 1.7 | 91.5 ± 2.2 | 2.6 | 18.7 ± 0.4 | 20.8 ± 1.2 | 9.8 | |
| T2 | 28.6 ± 1.0 | 30.1 ± 0.6 | 5.1 | 0.39 ± 0.01 | 0.44 ± 0.01 | 10.1 | 89.0 ± 1.6 | 90.8 ± 2.0 | 2.0 | 18.3 ± 0.4 | 20.6 ± 1.3 | 11.0 | |
| T3 | 28.6 ± 0.9 | 30.2 ± 0.7 | 5.1 | 0.39 ± 0.01 | 0.45 ± 0.01 | 12.2 | 88.9 ± 2.3 | 90.7 ± 2.2 | 2.0 | 18.5 ± 0.6 | 20.3 ± 1.5 | 8.9 | |
| Period 2 | Control | 30.7 ± 1.0 | 31.3 ± 0.9 | 2.0 | 0.40 ± 0.01 | 0.45 ± 0.01 | 10.8 | 93.4 ± 1.8 | 94.5 ± 2.3 | 1.2 | 22.1 ± 0.9 | 23.5 ± 1.5 | 6.1 |
| T1 | 30.5 ± 0.9 | 31.5 ± 0.9 | 3.2 | 0.40 ± 0.01 | 0.45 ± 0.01 | 11.2 | 92.7 ± 1.4 | 94.9 ± 1.7 | 2.2 | 22.2 ± 0.8 | 23.4 ± 0.9 | 5.0 | |
| T2 | 30.5 ± 0.7 | 32.1 ± 0.8 | 5.0 | 0.40 ± 0.01 | 0.45 ± 0.01 | 10.7 | 92.1 ± 1.6 | 94.8 ± 2.1 | 2.9 | 22.0 ± 1.0 | 23.3 ± 1.3 | 5.7 | |
| T3 | 30.7 ± 0.9 | 31.7 ± 0.7 | 3.4 | 0.40 ± 0.01 | 0.45 ± 0.01 | 10.5 | 92.3 ± 2.0 | 95.3 ± 2.1 | 3.1 | 22.2 ± 0.6 | 23.4 ± 1.0 | 5.0 | |
| Period 3 | Control | 27.4 ± 1.0 | 28.0 ± 0.8 | 2.2 | 0.36 ± 0.01 | 0.39 ± 0.01 | 7.6 | 84.7 ± 3.0 | 87.4 ± 2.2 | 3.1 | 15.0 ± 0.5 | 15.9 ± 0.6 | 5.5 |
| T1 | 28.7 ± 0.9 | 29.9 ± 0.7 | 3.9 | 0.37 ± 0.01 | 0.40 ± 0.01 | 6.8 | 84.5 ± 1.5 | 86.9 ± 1.2 | 2.8 | 14.9 ± 0.4 | 15.8 ± 0.6 | 5.2 | |
| T2 | 28.6 ± 1.0 | 30.1 ± 0.6 | 5.1 | 0.37 ± 0.01 | 0.41 ± 0.01 | 8.1 | 84.8 ± 0.7 | 87.2 ± 1.3 | 2.7 | 15.1 ± 0.4 | 15.7 ± 0.5 | 3.6 | |
| T3 | 28.6 ± 0.9 | 31.1 ± 0.8 | 8.0 | 0.37 ± 0.01 | 0.41 ± 0.01 | 8.6 | 85.1 ± 2.0 | 87.2 ± 1.8 | 2.4 | 15.2 ± 0.6 | 15.6 ± 0.6 | 2.7 | |
| Sources of Variation | Prey Survival | Vulnerability | ||||||
|---|---|---|---|---|---|---|---|---|
| df | MS | F | Pr > F | df | MS | F | Pr > F | |
| Seasonal effects | 2 | 5069.8 | 181.04 | <0.0001 | 2 | 3050.9 | 42.95 | <0.0001 |
| Shelter effects | 3 | 1134.7 | 40.52 | <0.0001 | 3 | 390.7 | 5.50 | 0.0074 |
| Interaction | 6 | 55.4 | 1.98 | 0.089 | 6 | 29.5 | 0.42 | 0.8645 |
| Elapsed time | 4 | 3836.6 | 137.01 | <0.0001 | 4 | 185.2 | 2.61 | 0.0484 |
| Error | 44 | 28.0 | 44 | 71.0 | ||||
| Total | 59 | 59 | ||||||
© 2017 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, S.; Ahn, C.H.; Song, H.M.; Park, J.R.; Joo, J.C. Prey Survival and Vulnerability of Juvenile Rhynchocypris oxycephalus in Juvenile Fish Shelters under Predation by Korean Native Piscivorous Fish (Coreoperca herzi). Water 2017, 9, 36. https://doi.org/10.3390/w9010036
Lee S, Ahn CH, Song HM, Park JR, Joo JC. Prey Survival and Vulnerability of Juvenile Rhynchocypris oxycephalus in Juvenile Fish Shelters under Predation by Korean Native Piscivorous Fish (Coreoperca herzi). Water. 2017; 9(1):36. https://doi.org/10.3390/w9010036
Chicago/Turabian StyleLee, Saeromi, Chang Hyuk Ahn, Ho Myeon Song, Jae Roh Park, and Jin Chul Joo. 2017. "Prey Survival and Vulnerability of Juvenile Rhynchocypris oxycephalus in Juvenile Fish Shelters under Predation by Korean Native Piscivorous Fish (Coreoperca herzi)" Water 9, no. 1: 36. https://doi.org/10.3390/w9010036
APA StyleLee, S., Ahn, C. H., Song, H. M., Park, J. R., & Joo, J. C. (2017). Prey Survival and Vulnerability of Juvenile Rhynchocypris oxycephalus in Juvenile Fish Shelters under Predation by Korean Native Piscivorous Fish (Coreoperca herzi). Water, 9(1), 36. https://doi.org/10.3390/w9010036






