TiO2/Activated Carbon/2D Selenides Composite Photocatalysts for Industrial Wastewater Treatment
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Activation of Charcoal
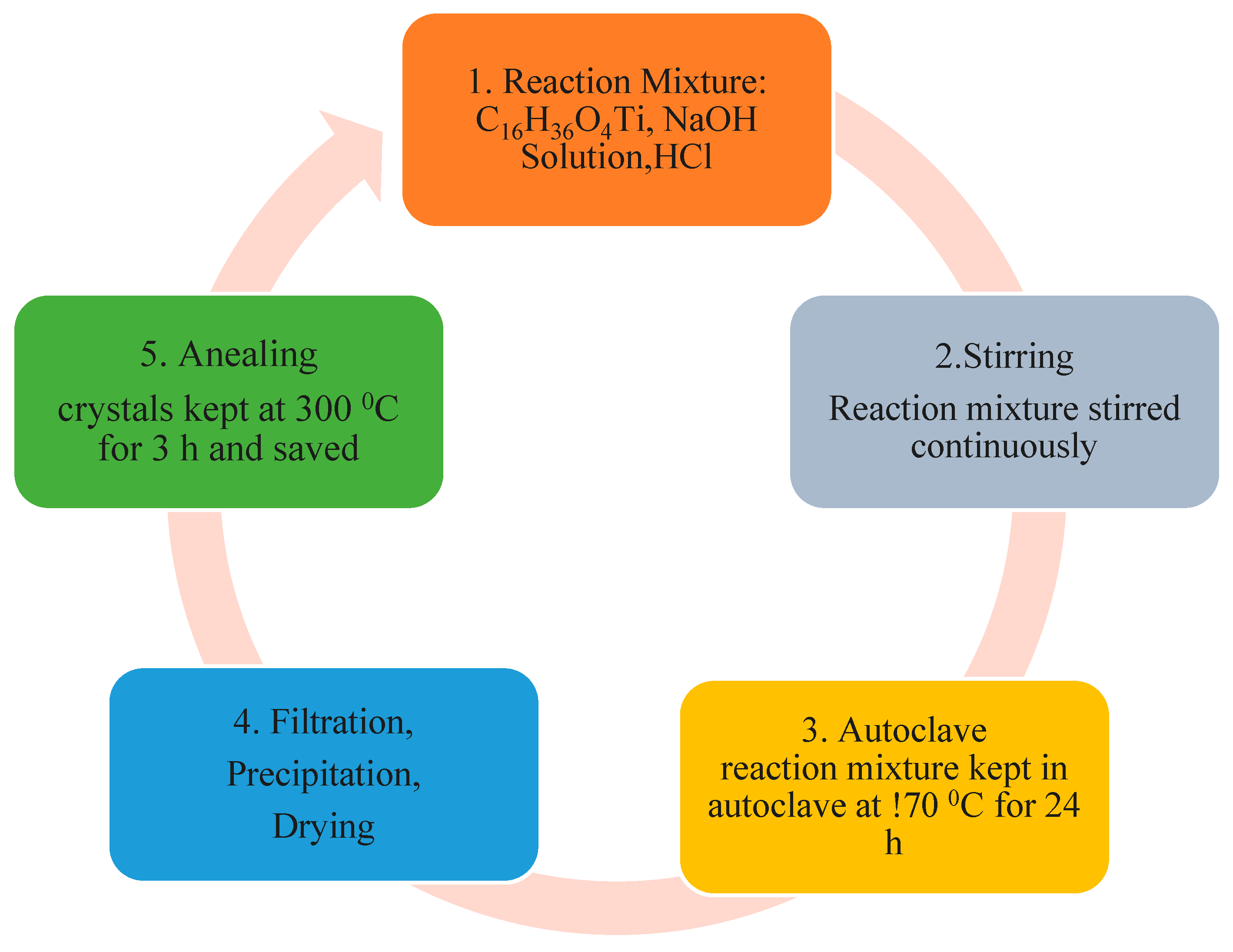
2.3. Synthesis of TiO2
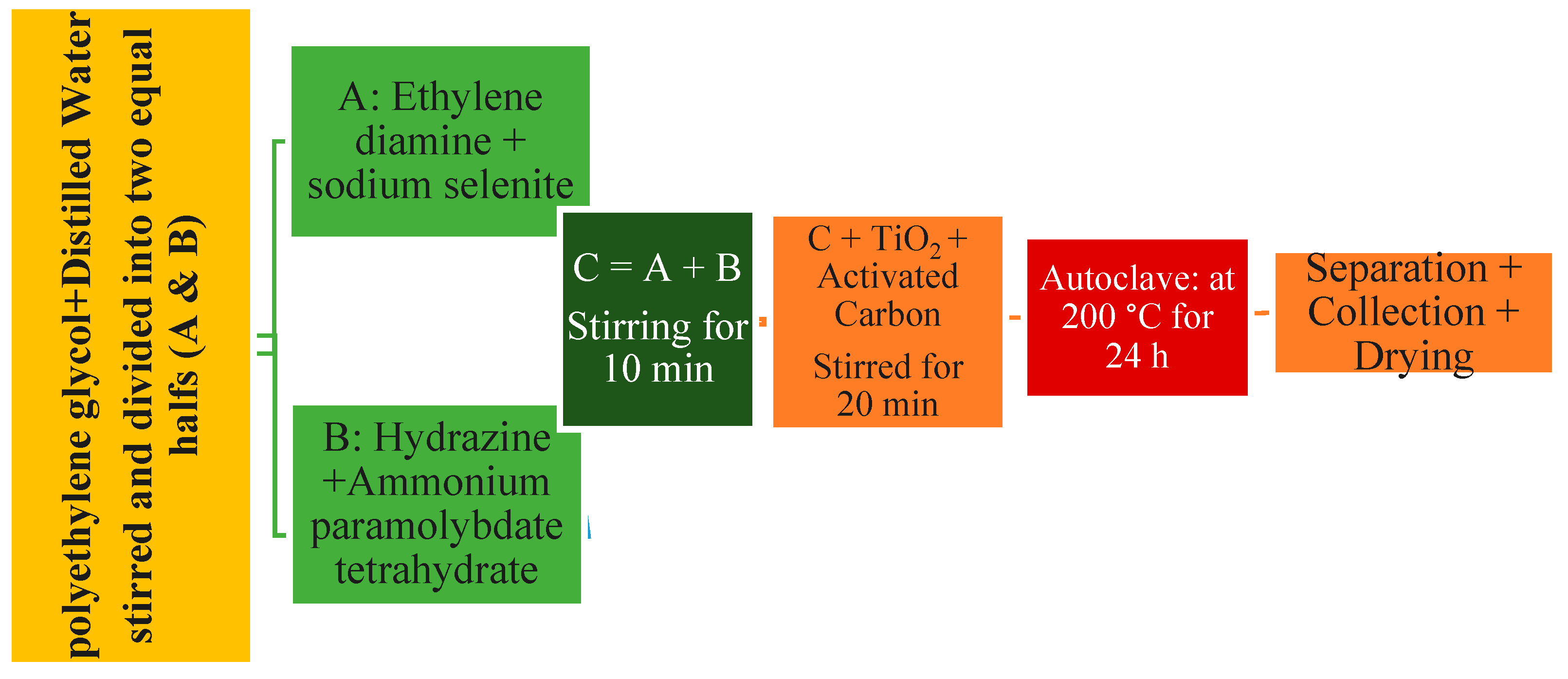
2.4. Synthesis of TiO2/xAC/2DMoSe2
2.5. Characterization
3. Results and Discussion
3.1. FTIR Study of Composites
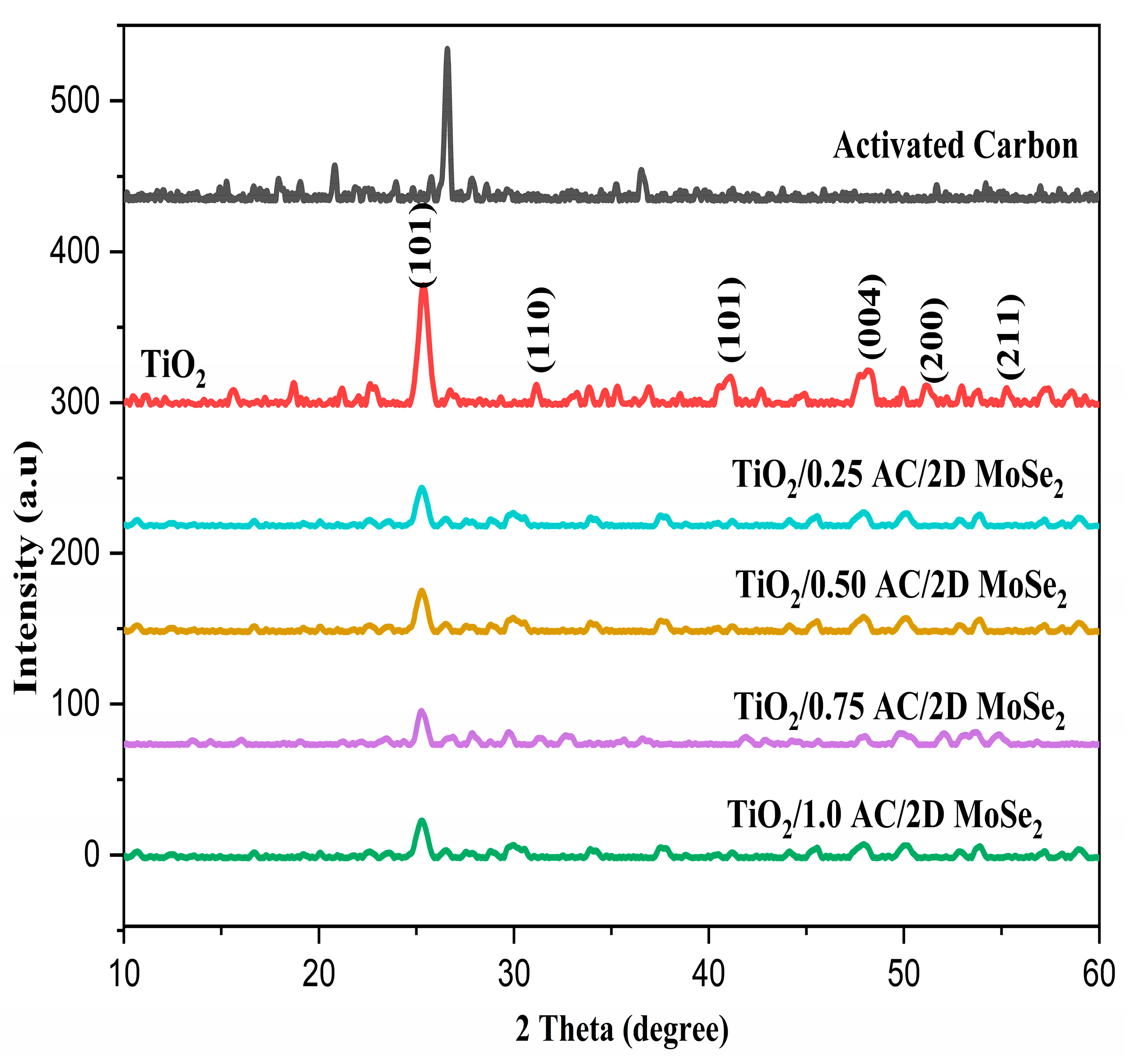
3.2. Structural Properties
3.3. Morphological Properties
3.4. Optical Properties
4. Parameters Affecting Photodegradation of Methylene Blue
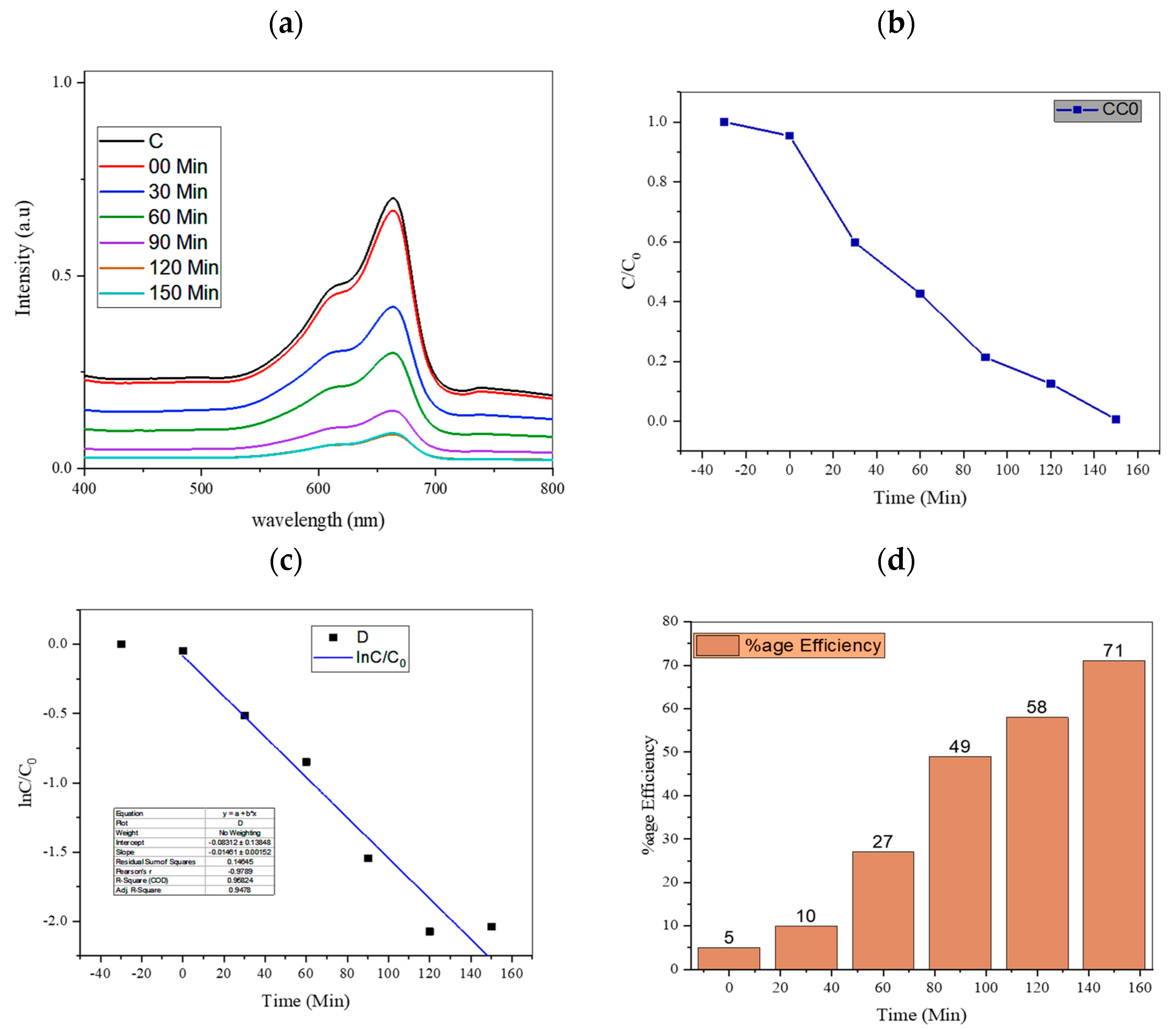
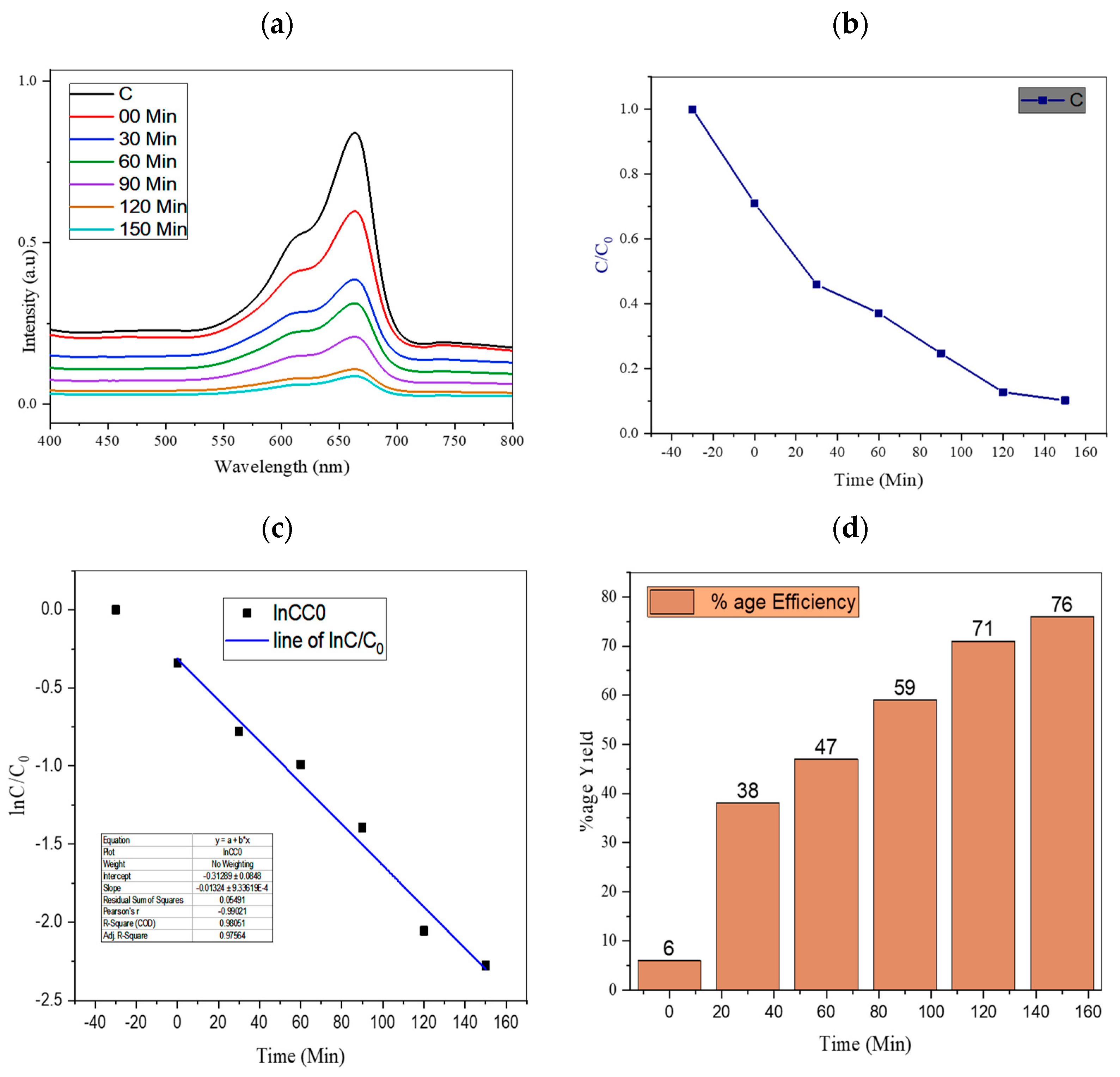
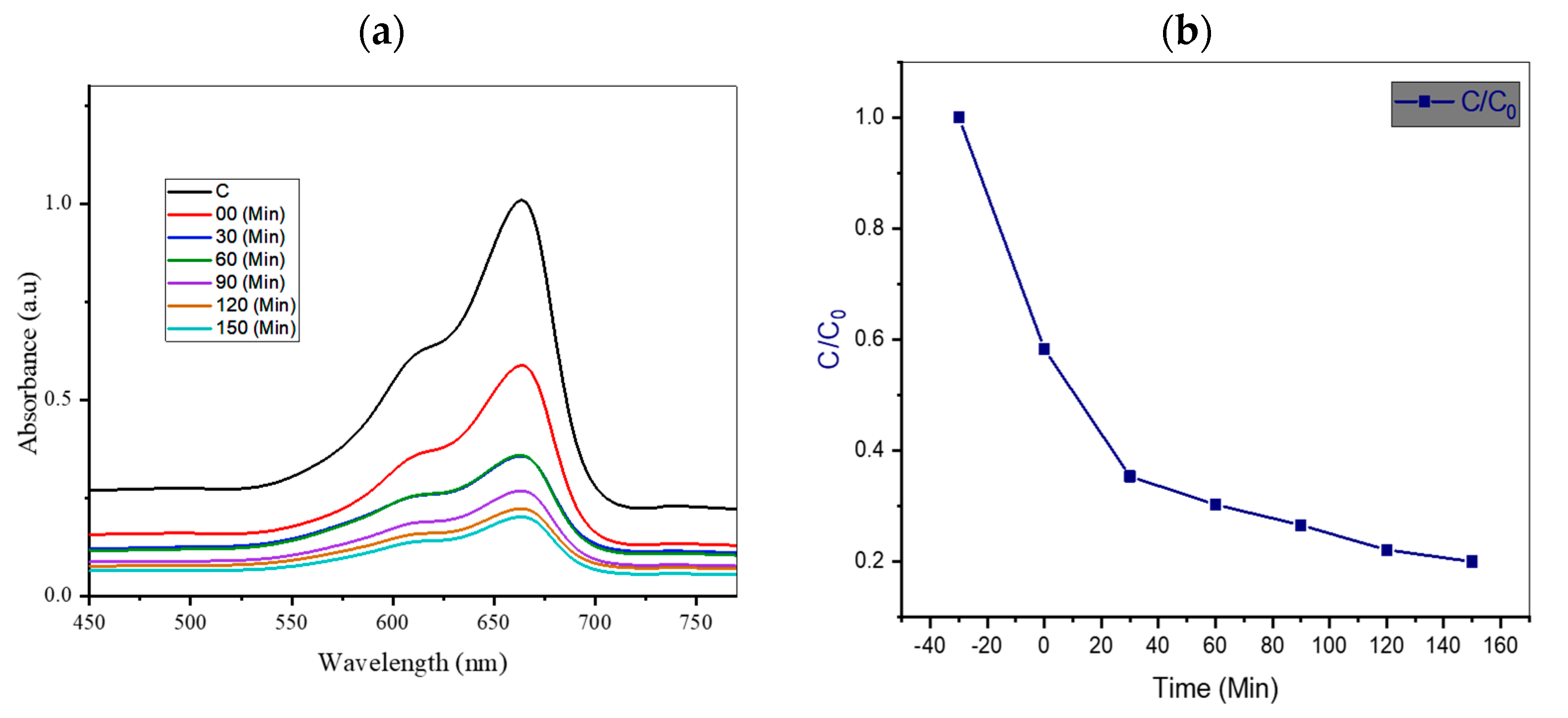
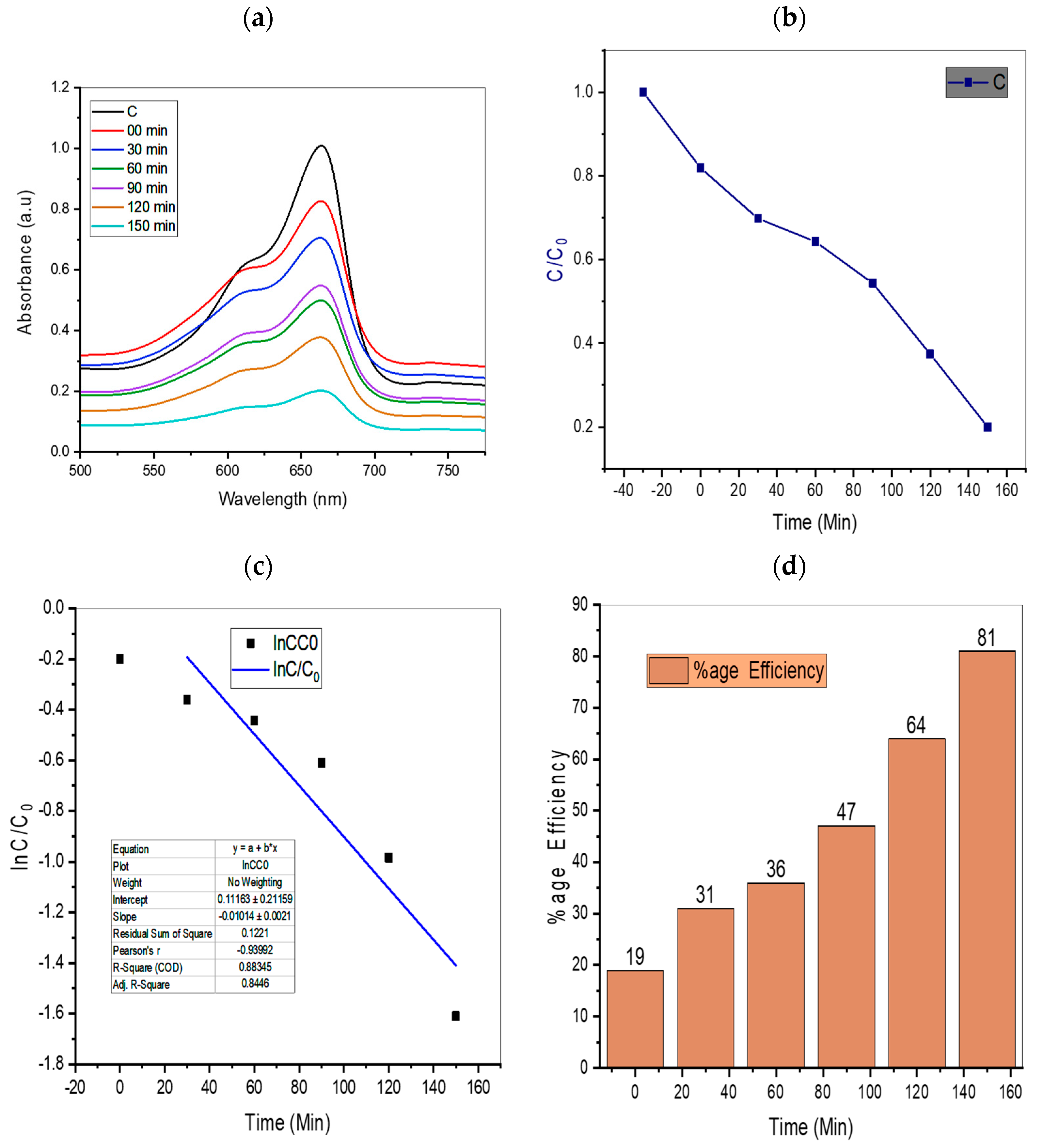
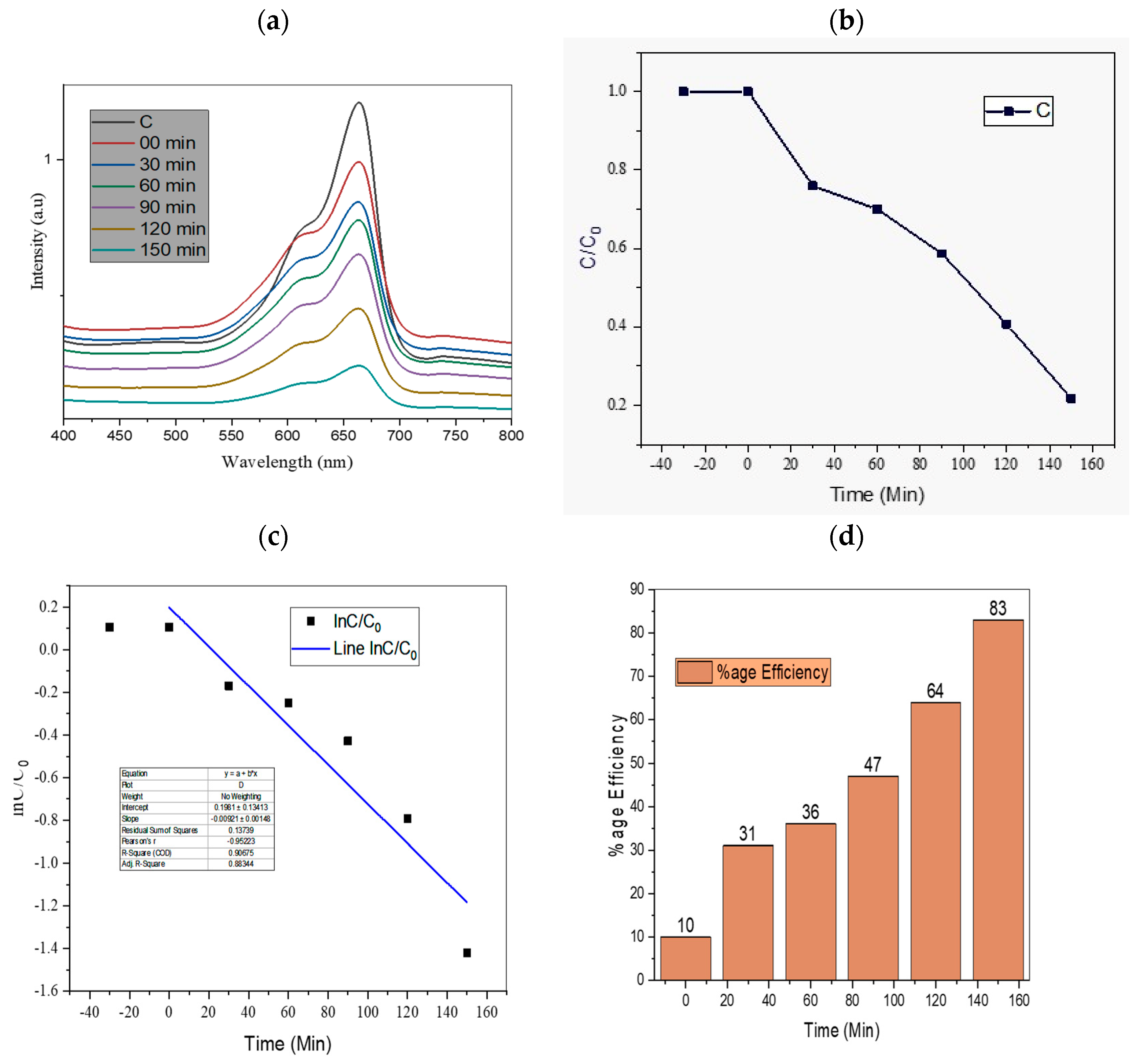
4.1. Effect of Irradiation Time
4.2. Effect of Initial Dye Concentration
4.3. Effect of pH
4.4. Effect of Catalyst Loading
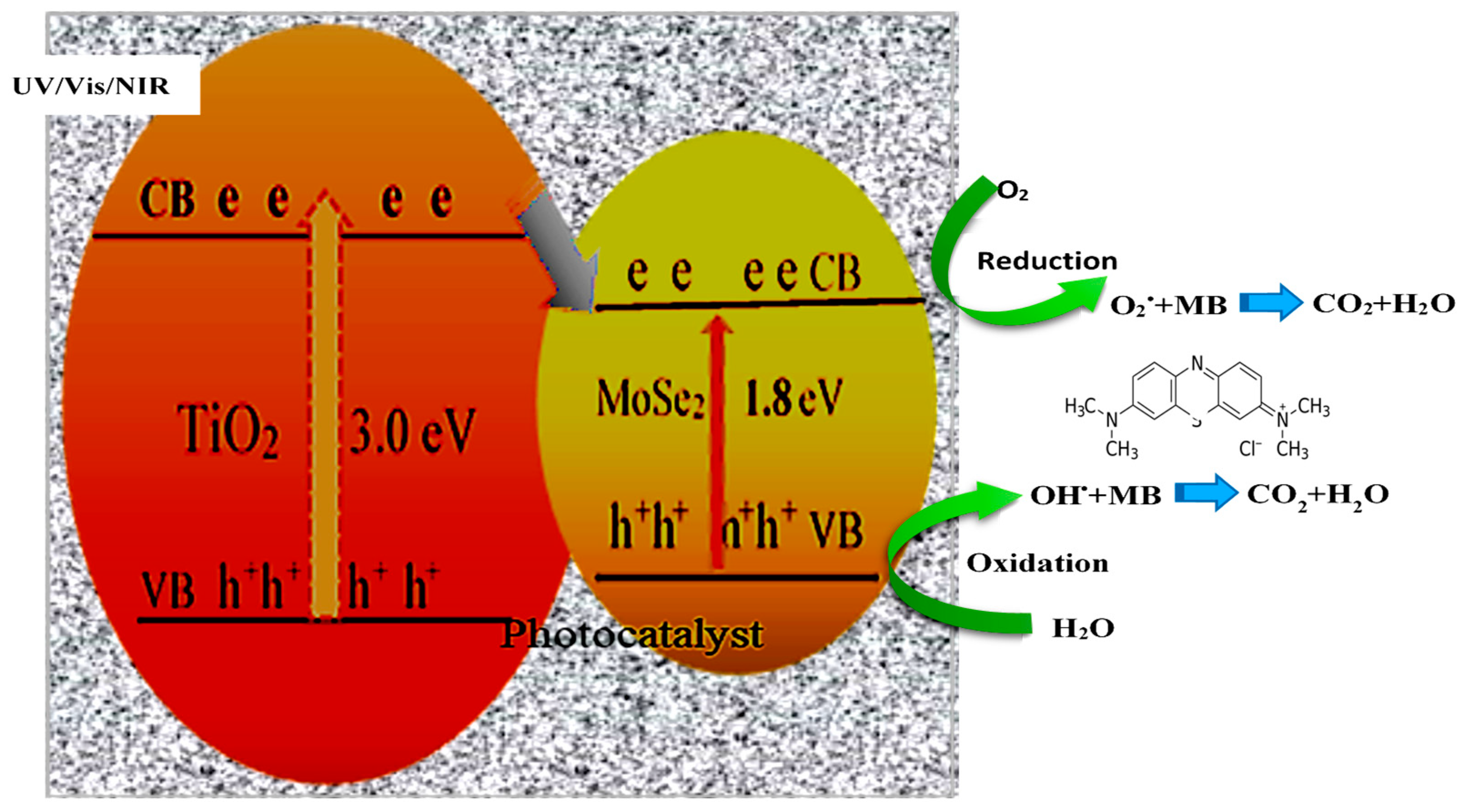
5. Proposed Photocatalytic Mechanism
6. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Krishna, R.; Mishra, J.; Ighalo, J.O. Rising demand for rain water harvesting system in the world: A case study of Joda Town, India. World Sci. News 2020, 146, 47–59. [Google Scholar]
- Olvera, R.C.; Silva, S.L.; Robles-Belmont, E.; Lau, E.Z. Review of nanotechnology value chain for water treatment applications in Mexico. Resour.-Effic. Technol. 2017, 3, 1–11. [Google Scholar] [CrossRef]
- Singha, I.; Mishrab, P.K. Nano-membrane filtration a novel application of nanotechnology for waste water treatment. Mater. Today Proc. 2020, 29, 327–332. [Google Scholar] [CrossRef]
- Kamali, M.; Persson, K.M.; Costa, M.E.; Capela, I. Sustainability criteria for assessing nanotechnology applicability in industrial wastewater treatment: Current status and future outlook. Environ. Int. 2019, 125, 261–276. [Google Scholar] [CrossRef]
- Das, A.; Dey, A. P-Nitrophenol-Bioremediation using potent Pseudomonas strain from the textile dye industry effluent. J. Environ. Chem. Eng. 2020, 8, 103830. [Google Scholar] [CrossRef]
- Anas, M.; Han, D.S.; Mahmoud, K.; Park, H.; Abdel-Wahab, A. Photocatalytic degradation of organic dye using titanium dioxide modified with metal and non-metal deposition. Mater. Sci. Semicond. Process. 2016, 41, 209–218. [Google Scholar] [CrossRef]
- de Vidales, M.J.M.; Nieto-Márquez, A.; Morcuende, D.; Atanes, E.; Blaya, F.; Soriano, E.; Fernández-Martínez, F. 3D printed floating photocatalysts for wastewater treatment. Catal. Today 2019, 328, 157–163. [Google Scholar] [CrossRef]
- Ighalo, J.O.; Adeniyi, A.G.; Adeniran, J.A.; Ogunniyi, S. A systematic literature analysis of the nature and regional distribution of water pollution sources in Nigeria. J. Clean. Prod. 2021, 283, 124566. [Google Scholar] [CrossRef]
- Kumar, S.; Ahlawat, W.; Bhanjana, G.; Heydarifard, S.; Nazhad, M.M.; Dilbaghi, N. Nanotechnology-based water treatment strategies. J. Nanosci. Nanotechnol. 2014, 14, 1838–1858. [Google Scholar] [CrossRef]
- Qu, X.; Brame, J.; Li, Q.; Alvarez, P.J. Nanotechnology for a safe and sustainable water supply: Enabling integrated water treatment and reuse. Acc. Chem. Res. 2013, 46, 834–843. [Google Scholar] [CrossRef]
- Nwabor, O.F.; Nnamonu, E.; Martins, P.; Ani, O. Water and waterborne diseases: A review. Int. J. Trop. Dis. Health 2016, 12, 1–14. [Google Scholar]
- Munir, H.M.S.; Feroze, N.; Ramzan, N.; Sagir, M.; Babar, M.; Tahir, M.S.; Shamshad, J.; Mubashir, M.; Khoo, K.S. Fe-zeolite catalyst for ozonation of pulp and paper wastewater for sustainable water resources. Chemosphere 2022, 297, 134031. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Caro, F.; Corbett, C.J.; Ding, X. Estimating the environmental and economic impacts of widespread adoption of potential technology solutions to reduce water use and pollution: Application to China’s textile industry. Environ. Impact Assess. Rev. 2019, 79, 106293. [Google Scholar] [CrossRef]
- Mittal, H.; Kumar, A.; Khanuja, M. In-situ oxidative polymerization of aniline on hydrothermally synthesized MoSe2 for enhanced photocatalytic degradation of organic dyes. J. Saudi Chem. Soc. 2019, 23, 836–845. [Google Scholar] [CrossRef]
- Khan, S.; Malik, A. Environmental and health effects of textile industry wastewater. In Environmental Deterioration and Human Health; Springer: Berlin/Heidelberg, Germany, 2014; pp. 55–71. [Google Scholar]
- Tounsadi, H.; Metarfi, Y.; Taleb, M.; El Rhazi, K.; Rais, Z. Impact of chemical substances used in textile industry on the employee's health: Epidemiological study. Ecotoxicol. Environ. Saf. 2020, 197, 110594. [Google Scholar] [CrossRef]
- Bener, S.; Atalay, S.; Ersöz, G. The hybrid process with eco-friendly materials for the treatment of the real textile industry wastewater. Ecol. Eng. 2020, 148, 105789. [Google Scholar] [CrossRef]
- Gupta, R.; Pandit, C.; Pandit, S.; Gupta, P.K.; Lahiri, D.; Agarwal, D.; Pandey, S. Potential and future prospects of biochar-based materials and their applications in removal of organic contaminants from industrial wastewater. J. Mater. Cycles Waste Manag. 2022, 24, 852–876. [Google Scholar] [CrossRef]
- Jaganathan, V.; Cherurveettil, P.; Chellasamy, A.; Premapriya, M. Environmental pollution risk analysis and management in textile industry: A preventive mechanism. In Proceedings of the 1st International Conference on Social Sciences and Humanities, ICSH 2014, Gaborone, Botswana, 4–7 June 2014. [Google Scholar]
- Khan, I.; Saeed, K.; Zekker, I.; Zhang, B.; Hendi, A.H.; Ahmad, A.; Ahmad, S.; Zada, N.; Ahmad, H.; Shah, L.A. Review on methylene blue: Its properties, uses, toxicity and photodegradation. Water 2022, 14, 242. [Google Scholar] [CrossRef]
- Santoso, E.; Ediati, R.; Kusumawati, Y.; Bahruji, H.; Sulistiono, D.; Prasetyoko, D. Review on recent advances of carbon based adsorbent for methylene blue removal from waste water. Mater. Today Chem. 2020, 16, 100233. [Google Scholar] [CrossRef]
- Abdelrahman, E.A.; Hegazey, R.; El-Azabawy, R.E. Efficient removal of methylene blue dye from aqueous media using Fe/Si, Cr/Si, Ni/Si, and Zn/Si amorphous novel adsorbents. J. Mater. Res. Technol. 2019, 8, 5301–5313. [Google Scholar] [CrossRef]
- Shandilya, P.; Mittal, D.; Soni, M.; Raizada, P.; Hosseini-Bandegharaei, A.; Saini, A.K.; Singh, P. Fabrication of fluorine doped graphene and SmVO4 based dispersed and adsorptive photocatalyst for abatement of phenolic compounds from water and bacterial disinfection. J. Clean. Prod. 2018, 203, 386–399. [Google Scholar] [CrossRef]
- Raizada, P.; Sudhaik, A.; Singh, P.; Shandilya, P.; Saini, A.K.; Gupta, V.K.; Lim, J.-H.; Jung, H.; Hosseini-Bandegharaei, A. Fabrication of Ag3VO4 decorated phosphorus and sulphur co-doped graphitic carbon nitride as a high-dispersed photocatalyst for phenol mineralization and E. coli disinfection. Sep. Purif. Technol. 2019, 212, 887–900. [Google Scholar] [CrossRef]
- Pare, B.; Singh, P.; Jonnalgadda, S. Degradation and Mineralization of Victoria Blue B Dye in a Slurry Photoreactor Using Advanced Oxidation Process; CSIR: New Delhi, India, 2009. [Google Scholar]
- Pare, B.; Singh, P.; Jonnalagadda, S. Visible Light Induced Heterogeneous Advanced Oxidation Process to Degrade Pararosanilin Dye in Aqueous Suspension of ZnO; CSIR: New Delhi, India, 2008. [Google Scholar]
- Watanabe, N.; Nakamura, S.; Liu, B.; Wang, N. Utilization of Structure from Motion for processing CORONA satellite images: Application to mapping and interpretation of archaeological features in Liangzhu Culture, China. Archaeol. Res. Asia 2017, 11, 38–50. [Google Scholar] [CrossRef]
- Sharma, K.; Dutta, V.; Sharma, S.; Raizada, P.; Hosseini-Bandegharaei, A.; Thakur, P.; Singh, P. Recent advances in enhanced photocatalytic activity of bismuth oxyhalides for efficient photocatalysis of organic pollutants in water: A review. J. Ind. Eng. Chem. 2019, 78, 1–20. [Google Scholar] [CrossRef]
- Priya, B.; Raizada, P.; Singh, N.; Thakur, P.; Singh, P. Adsorptional photocatalytic mineralization of oxytetracycline and ampicillin antibiotics using Bi2O3/BiOCl supported on graphene sand composite and chitosan. J. Colloid Interface Sci. 2016, 479, 271–283. [Google Scholar] [CrossRef]
- Raizada, P.; Singh, P.; Kumar, A.; Pare, B.; Jonnalagadda, S.B. Zero valent iron-brick grain nanocomposite for enhanced solar-Fenton removal of malachite green. Sep. Purif. Technol. 2014, 133, 429–437. [Google Scholar] [CrossRef]
- Sudhaik, A.; Raizada, P.; Shandilya, P.; Singh, P. Magnetically recoverable graphitic carbon nitride and NiFe2O4 based magnetic photocatalyst for degradation of oxytetracycline antibiotic in simulated wastewater under solar light. J. Environ. Chem. Eng. 2018, 6, 3874–3883. [Google Scholar] [CrossRef]
- Raizada, P.; Kumari, J.; Shandilya, P.; Dhiman, R.; Singh, V.P.; Singh, P. Magnetically retrievable Bi2WO6/Fe3O4 immobilized on graphene sand composite for investigation of photocatalytic mineralization of oxytetracycline and ampicillin. Process Saf. Environ. Prot. 2017, 106, 104–116. [Google Scholar] [CrossRef]
- Chen, D.; Cheng, Y.; Zhou, N.; Chen, P.; Wang, Y.; Li, K.; Huo, S.; Cheng, P.; Peng, P.; Zhang, R. Photocatalytic degradation of organic pollutants using TiO2-based photocatalysts: A review. J. Clean. Prod. 2020, 268, 121725. [Google Scholar] [CrossRef]
- Jafari, S.; Zhao, F.; Zhao, D.; Lahtinen, M.; Bhatnagar, A.; Sillanpää, M. A comparative study for the removal of methylene blue dye by N and S modified TiO2 adsorbents. J. Mol. Liq. 2015, 207, 90–98. [Google Scholar] [CrossRef]
- Beiki, H.; Esfahany, M.N.; Etesami, N. Turbulent mass transfer of Al2O3 and TiO2 electrolyte nanofluids in circular tube. Microfluid. Nanofluid. 2013, 15, 501–508. [Google Scholar] [CrossRef]
- Kusiak-Nejman, E.; Morawski, A.W. TiO2/graphene-based nanocomposites for water treatment: A brief overview of charge carrier transfer, antimicrobial and photocatalytic performance. Appl. Catal. B Environ. 2019, 253, 179–186. [Google Scholar] [CrossRef]
- Lee, C.-G.; Javed, H.; Zhang, D.; Kim, J.-H.; Westerhoff, P.; Li, Q.; Alvarez, P.J. Porous electrospun fibers embedding TiO2 for adsorption and photocatalytic degradation of water pollutants. Environ. Sci. Technol. 2018, 52, 4285–4293. [Google Scholar] [CrossRef] [PubMed]
- Mallakpour, S.; Nikkhoo, E. Surface modification of nano-TiO2 with trimellitylimido-amino acid-based diacids for preventing aggregation of nanoparticles. Adv. Powder Technol. 2014, 25, 348–353. [Google Scholar] [CrossRef]
- Raza, N.; Raza, W.; Gul, H.; Azam, M.; Lee, J.; Vikrant, K.; Kim, K.-H. Solar-light-active silver phosphate/titanium dioxide/silica heterostructures for photocatalytic removal of organic dye. J. Clean. Prod. 2020, 254, 120031. [Google Scholar] [CrossRef]
- Alamelu, K.; Ali, B.J. Au nanoparticles decorated sulfonated graphene-TiO2 nanocomposite for sunlight driven photocatalytic degradation of recalcitrant compound. Sol. Energy 2020, 211, 1194–1205. [Google Scholar] [CrossRef]
- Moradi, M.; Khorasheh, F.; Larimi, A. Pt nanoparticles decorated Bi-doped TiO2 as an efficient photocatalyst for CO2 photo-reduction into CH4. Sol. Energy 2020, 211, 100–110. [Google Scholar] [CrossRef]
- Xie, Z.; Zhao, F.; Zou, S.; Zhu, F.; Zhang, Z.; Wang, W. TiO2 nanorod arrays decorated with Au nanoparticles as sensitive and recyclable SERS substrates. J. Alloy. Compd. 2021, 861, 157999. [Google Scholar] [CrossRef]
- Xiao, W.-Z.; Xu, L.; Rong, Q.-Y.; Dai, X.-Y.; Cheng, C.-P.; Wang, L.-L. Two-dimensional H-TiO2/MoS2 (WS2) van der Waals heterostructures for visible-light photocatalysis and energy conversion. Appl. Surf. Sci. 2020, 504, 144425. [Google Scholar] [CrossRef]
- Wang, S.; Yun, J.-H.; Luo, B.; Butburee, T.; Peerakiatkhajohn, P.; Thaweesak, S.; Xiao, M.; Wang, L. Recent progress on visible light responsive heterojunctions for photocatalytic applications. J. Mater. Sci. Technol. 2017, 33, 1–22. [Google Scholar] [CrossRef]
- Yang, L.; Hao, X.; Yu, D.; Zhou, P.; Peng, Y.; Jia, Y.; Zhao, C.; He, J.; Zhan, C.; Lai, B. High visible-light catalytic activity of Bis-PDI-T@ TiO2 for activating persulfate toward efficient degradation of carbamazepine. Sep. Purif. Technol. 2021, 263, 118384. [Google Scholar] [CrossRef]
- Kang, L.; Zhang, M.; Zhang, J.; Liu, S.; Zhang, N.; Yao, W.; Ye, Y.; Luo, C.; Gong, Z.; Wang, C. Dual-defect surface engineering of bimetallic sulfide nanotubes towards flexible asymmetric solid-state supercapacitors. J. Mater. Chem. A 2020, 8, 24053–24064. [Google Scholar] [CrossRef]
- Wang, S.; Li, Y.; Wang, X.; Zi, G.; Zhou, C.; Liu, B.; Liu, G.; Wang, L.; Huang, W. One-step supramolecular preorganization constructed crinkly graphitic carbon nitride nanosheets with enhanced photocatalytic activity. J. Mater. Sci. Technol. 2022, 104, 155–162. [Google Scholar] [CrossRef]
- Wu, L.; Shi, S.; Li, Q.; Zhang, X.; Cui, X. TiO2 nanoparticles modified with 2D MoSe2 for enhanced photocatalytic activity on hydrogen evolution. Int. J. Hydrog. Energy 2019, 44, 720–728. [Google Scholar] [CrossRef]
- Hu, Z.; Wu, Z.; Han, C.; He, J.; Ni, Z.; Chen, W. Two-dimensional transition metal dichalcogenides: Interface and defect engineering. Chem. Soc. Rev. 2018, 47, 3100–3128. [Google Scholar] [CrossRef]
- Wang, D.; Xu, Y.; Sun, F.; Zhang, Q.; Wang, P.; Wang, X. Enhanced photocatalytic activity of TiO2 under sunlight by MoS2 nanodots modification. Appl. Surf. Sci. 2016, 377, 221–227. [Google Scholar] [CrossRef]
- Liang, Z.; Bai, X.; Hao, P.; Guo, Y.; Xue, Y.; Tian, J.; Cui, H. Full solar spectrum photocatalytic oxygen evolution by carbon-coated TiO2 hierarchical nanotubes. Appl. Catal. B Environ. 2019, 243, 711–720. [Google Scholar] [CrossRef]
- Coehoorn, R.; Haas, C.; Dijkstra, J.; Flipse, C.d.; De Groot, R.; Wold, A. Electronic structure of MoSe2, MoS2, and WSe2. I. Band-structure calculations and photoelectron spectroscopy. Phys. Rev. B 1987, 35, 6195. [Google Scholar] [CrossRef]
- Boyjoo, Y.; Sun, H.; Liu, J.; Pareek, V.K.; Wang, S. A review on photocatalysis for air treatment: From catalyst development to reactor design. Chem. Eng. J. 2017, 310, 537–559. [Google Scholar] [CrossRef]
- Ren, Z.; Liu, X.; Zhuge, Z.; Gong, Y.; Sun, C.Q. MoSe2/ZnO/ZnSe hybrids for efficient Cr (VI) reduction under visible light irradiation. Chin. J. Catal. 2020, 41, 180–187. [Google Scholar] [CrossRef]
- Rivera, P.; Schaibley, J.R.; Jones, A.M.; Ross, J.S.; Wu, S.; Aivazian, G.; Klement, P.; Seyler, K.; Clark, G.; Ghimire, N.J. Observation of long-lived interlayer excitons in monolayer MoSe2–WSe2 heterostructures. Nat. Commun. 2015, 6, 6242. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Liu, B.; Cai, H.; Liu, S.; Yamauchi, Y.; Jun, S.C. Covalently Interlayer-Confined Organic–Inorganic Heterostructures for Aqueous Potassium Ion Supercapacitors. Small 2023, 19, 2204275. [Google Scholar] [CrossRef] [PubMed]
- Xie, T.; Liu, Y.; Wang, H.; Wu, Z. Layered MoSe2/Bi2WO6 composite with PN heterojunctions as a promising visible-light induced photocatalyst. Appl. Surf. Sci. 2018, 444, 320–329. [Google Scholar] [CrossRef]
- Leary, R.; Westwood, A. Carbonaceous nanomaterials for the enhancement of TiO2 photocatalysis. Carbon 2011, 49, 741–772. [Google Scholar] [CrossRef]
- Yan, Y.; Kuang, W.; Shi, L.; Ye, X.; Yang, Y.; Xie, X.; Shi, Q.; Tan, S. Carbon quantum dot-decorated TiO2 for fast and sustainable antibacterial properties under visible-light. J. Alloy. Compd. 2019, 777, 234–243. [Google Scholar] [CrossRef]
- Moosavi, S.; Lai, C.W.; Gan, S.; Zamiri, G.; Akbarzadeh Pivehzhani, O.; Johan, M.R. Application of efficient magnetic particles and activated carbon for dye removal from wastewater. ACS Omega 2020, 5, 20684–20697. [Google Scholar] [CrossRef]
- Siddiqui, I.; Mittal, H.; Kohli, V.K.; Gautam, P.; Ali, M.; Khanuja, M. Hydrothermally synthesized micron sized, broom-shaped MoSe2 nanostructures for superior photocatalytic water purification. Mater. Res. Express 2018, 5, 125020. [Google Scholar] [CrossRef]
- Pan, J.; Zhu, X.; Chen, X.; Zhao, Y.; Liu, J. Gd3+-Doped MoSe2 nanosheets used as a theranostic agent for bimodal imaging and highly efficient photothermal cancer therapy. Biomater. Sci. 2018, 6, 372–387. [Google Scholar] [CrossRef]
- Subramani, A.; Byrappa, K.; Kumaraswamy, G.; Ravikumar, H.; Ranganathaiah, C.; Lokanatha Rai, K.; Ananda, S.; Yoshimura, M. Hydrothermal preparation and characterization of TiO2: AC composites. Mater. Lett. 2007, 61, 4828–4831. [Google Scholar] [CrossRef]
- Alkorbi, A.S.; Javed, H.M.A.; Hussain, S.; Latif, S.; Mahr, M.S.; Mustafa, M.S.; Alsaiari, R.; Alhemiary, N.A. Solar light-driven photocatalytic degradation of methyl blue by carbon-doped TiO2 nanoparticles. Opt. Mater. 2022, 127, 112259. [Google Scholar] [CrossRef]
- Thinkohkaew, K.; Piroonpan, T.; Jiraborvornpongsa, N.; Potiyaraj, P. Development of multifunctional polypropylene nonwoven fabric by radiation induced grafting of TiO2 nanoparticles and trifluoroethyl methacrylate for protective textile applications. Materialia 2022, 21, 101355. [Google Scholar] [CrossRef]
- Jimoh, A.; Akpeji, B.; Azeez, S.; Ayipo, Y.; Abdulsalam, Z.; Adebayo, Z.; Ajao, A.; Zakariyah, A.; Elemike, E. Biosynthesis of Ag and TiO2 nanoparticles and the evaluation of their antibacterial activities. Inorg. Chem. Commun. 2022, 141, 109503. [Google Scholar] [CrossRef]
- Dessai, S.; Ayyanar, M.; Amalraj, S.; Khanal, P.; Vijayakumar, S.; Gurav, N.; Rarokar, N.; Kalaskar, M.; Nadaf, S.; Gurav, S. Bioflavonoid mediated synthesis of TiO2 nanoparticles: Characterization and their biomedical applications. Mater. Lett. 2022, 311, 131639. [Google Scholar] [CrossRef]
- Liu, S.; Chen, X.; Chen, X. A TiO2/AC composite photocatalyst with high activity and easy separation prepared by a hydrothermal method. J. Hazard. Mater. 2007, 143, 257–263. [Google Scholar] [CrossRef] [PubMed]
- Cai, Q.; Wang, F.; Xiang, J.; Dan, M.; Yu, S.; Zhou, Y. Layered MoS2 Grown on Anatase TiO2 {001} Promoting Interfacial Electron Transfer to Enhance Photocatalytic Evolution of H2 From H2S. Front. Environ. Chem. 2020, 1, 591645. [Google Scholar] [CrossRef]
- Yuan, Y.-J.; Ye, Z.-J.; Lu, H.-W.; Hu, B.; Li, Y.-H.; Chen, D.-Q.; Zhong, J.-S.; Yu, Z.-T.; Zou, Z.-G. Constructing anatase TiO2 nanosheets with exposed (001) facets/layered MoS2 two-dimensional nanojunctions for enhanced solar hydrogen generation. ACS Catal. 2016, 6, 532–541. [Google Scholar] [CrossRef]
- Wang, S.; Yi, L.; Halpert, J.E.; Lai, X.; Liu, Y.; Cao, H.; Yu, R.; Wang, D.; Li, Y. A novel and highly efficient photocatalyst based on P25–graphdiyne nanocomposite. Small 2012, 8, 265–271. [Google Scholar] [CrossRef]
- Dashora, A.; Ahuja, U.; Venugopalan, K. Electronic and optical properties of MoS2 (0 0 0 1) thin films: Feasibility for solar cells. Comput. Mater. Sci. 2013, 69, 216–221. [Google Scholar] [CrossRef]
- Mohammad, N.H.; EL-Sherbiny, G.M.; Hammad, A.A.; Askar, A.A.; El-Nour, S.A.A. Gamma-ray and sunlight-induced synthesis of silver nanoparticles using bacterial cellulose and cell-free filtrate produced by Komagataeibacter rhaeticus N1 MW322708 strain. Cellulose 2022, 29, 1791–1805. [Google Scholar] [CrossRef]
- Shah, I.H.; Ashraf, M.; Sabir, I.A.; Manzoor, M.A.; Malik, M.S.; Gulzar, S.; Ashraf, F.; Iqbal, J.; Niu, Q.; Zhang, Y. Green synthesis and Characterization of Copper oxide nanoparticles using Calotropis procera leaf extract and their different biological potentials. J. Mol. Struct. 2022, 1259, 132696. [Google Scholar] [CrossRef]
- Parvathiraja, C.; Katheria, S.; Siddiqui, M.R.; Wabaidur, S.M.; Islam, M.A.; Lai, W.-C. Activated Carbon-Loaded Titanium Dioxide Nanoparticles and Their Photocatalytic and Antibacterial Investigations. Catalysts 2022, 12, 834. [Google Scholar] [CrossRef]
- Dai, C.; Zhou, Z.; Tian, C.; Li, Y.; Yang, C.; Gao, X.; Tian, X. Large-scale synthesis of graphene-like MoSe2 nanosheets for efficient hydrogen evolution reaction. J. Phys. Chem. C 2017, 121, 1974–1981. [Google Scholar] [CrossRef]
- Tichapondwa, S.M.; Newman, J.; Kubheka, O. Effect of TiO2 phase on the photocatalytic degradation of methylene blue dye. Phys. Chem. Earth Parts A/B/C 2020, 118, 102900. [Google Scholar] [CrossRef]
- Small, J.M.; Hintelmann, H. Methylene blue derivatization then LC–MS analysis for measurement of trace levels of sulfide in aquatic samples. Anal. Bioanal. Chem. 2007, 387, 2881–2886. [Google Scholar] [CrossRef]
- Joseph, S.; Mathew, B. Microwave-assisted green synthesis of silver nanoparticles and the study on catalytic activity in the degradation of dyes. J. Mol. Liq. 2015, 204, 184–191. [Google Scholar] [CrossRef]
- Al-Hamoud, K.; Shaik, M.R.; Khan, M.; Alkhathlan, H.Z.; Adil, S.F.; Kuniyil, M.; Assal, M.E.; Al-Warthan, A.; Siddiqui, M.R.H.; Tahir, M.N. Pulicaria undulata Extract-Mediated Eco-Friendly Preparation of TiO2 Nanoparticles for Photocatalytic Degradation of Methylene Blue and Methyl Orange. ACS Omega 2022, 7, 4812–4820. [Google Scholar] [CrossRef]
- Rahman, Q.I.; Ali, A.; Ahmad, N.; Lohani, M.B.; Mehta, S.; Muddassir, M. Synthesis and characterization of CuO rods for enhanced visible light driven dye degradation. J. Nanosci. Nanotechnol. 2020, 20, 7716–7723. [Google Scholar] [CrossRef]
- Rahman, Q.I.; Hasan, S.; Ali, A.; Mehta, S.; Raja, M.A.; Ahmad, N.; Khan, A.R.; Muddassir, M. Synthesis and Characterizations of Nitrogen (N) Doped Strontium Titanate (SrTiO3) Nanoparticles for Enhanced Visible Light Driven Photocatalytic Degradation. J. Nanosci. Nanotechnol. 2020, 20, 6475–6481. [Google Scholar] [CrossRef]
- Askari, M.B.; Banizi, Z.T.; Seifi, M.; Dehaghi, S.B.; Veisi, P. Synthesis of TiO2 nanoparticles and decorated multi-wall carbon nanotube (MWCNT) with anatase TiO2 nanoparticles and study of optical properties and structural characterization of TiO2/MWCNT nanocomposite. Optik 2017, 149, 447–454. [Google Scholar] [CrossRef]
- Yoon, C.-J.; Lee, S.-H.; Kwon, Y.-B.; Kim, K.; Lee, K.-H.; Kim, S.M.; Kim, Y.-K. Fabrication of sustainable and multifunctional TiO2@ carbon nanotube nanocomposite fibers. Appl. Surf. Sci. 2021, 541, 148332. [Google Scholar] [CrossRef]
- Alghamdi, Y.G.; Krishnakumar, B.; Malik, M.A.; Alhayyani, S. Design and preparation of biomass-derived activated carbon loaded TiO2 photocatalyst for photocatalytic degradation of reactive red 120 and ofloxacin. Polymers 2022, 14, 880. [Google Scholar] [CrossRef] [PubMed]
- Justh, N.; Mikula, G.J.; Bakos, L.P.; Nagy, B.; László, K.; Parditka, B.; Erdélyi, Z.; Takáts, V.; Mizsei, J.; Szilágyi, I.M. Photocatalytic properties of TiO2@ polymer and TiO2@ carbon aerogel composites prepared by atomic layer deposition. Carbon 2019, 147, 476–482. [Google Scholar] [CrossRef]
- Saravanan, L.; Patil, R.A.; Gultom, P.; Kumar, B.; Manikandan, A.; Fu, Y.-P.; Chueh, Y.-L.; Cheng, C.-L.; Yeh, W.-C.; Ma, Y.-R. Rutile-phase TiO2@ carbon core-shell nanowires and their photoactivation in visible light region. Carbon 2021, 181, 280–289. [Google Scholar] [CrossRef]
- Salama, A.; Mohamed, A.; Aboamera, N.M.; Osman, T.; Khattab, A. Photocatalytic degradation of organic dyes using composite nanofibers under UV irradiation. Appl. Nanosci. 2018, 8, 155–161. [Google Scholar] [CrossRef]
- Elsayed, E.M.; Elnouby, M.S.; Gouda, M.; Elessawy, N.A.; Santos, D. Effect of the morphology of tungsten oxide embedded in sodium alginate/polyvinylpyrrolidone composite beads on the photocatalytic degradation of methylene blue dye solution. Materials 2020, 13, 1905. [Google Scholar] [CrossRef]
- Abdellah, M.; Nosier, S.; El-Shazly, A.; Mubarak, A. Photocatalytic decolorization of methylene blue using TiO2/UV system enhanced by air sparging. Alex. Eng. J. 2018, 57, 3727–3735. [Google Scholar] [CrossRef]
- Azeez, F.; Al-Hetlani, E.; Arafa, M.; Abdelmonem, Y.; Nazeer, A.A.; Amin, M.O.; Madkour, M. The effect of surface charge on photocatalytic degradation of methylene blue dye using chargeable titania nanoparticles. Sci. Rep. 2018, 8, 7104. [Google Scholar] [CrossRef]
- Nguyen Thi Thu, T.; Nguyen Thi, N.; Tran Quang, V.; Nguyen Hong, K.; Nguyen Minh, T.; Le Thi Hoai, N. Synthesis, characterisation, and effect of pH on degradation of dyes of copper-doped TiO2. J. Exp. Nanosci. 2016, 11, 226–238. [Google Scholar] [CrossRef]
- Hejazi, R.; Mahjoub, A.R.; Khavar, A.H.C.; Khazaee, Z. Fabrication of novel type visible-light-driven TiO2@ MIL-100 (Fe) microspheres with high photocatalytic performance for removal of organic pollutants. J. Photochem. Photobiol. A Chem. 2020, 400, 112644. [Google Scholar] [CrossRef]
- Hendekhale, N.R.; Mohammad-Khah, A. A novel synthesis of Co2ZrO5 and m-ZrO2 nanoparticles by sono-precipitation and hydrothermal methods and their application in UV/Visible-photocatalytic studies. J. Environ. Chem. Eng. 2020, 8, 104065. [Google Scholar] [CrossRef]
- Jia, Z.; La, L.; Zhang, W.; Liang, S.; Jiang, B.; Xie, S.; Habibi, D.; Zhang, L. Strong enhancement on dye photocatalytic degradation by ball-milled TiO2: A study of cationic and anionic dyes. J. Mater. Sci. Technol. 2017, 33, 856–863. [Google Scholar] [CrossRef]
- Khade, G.; Gavade, N.; Suwarnkar, M.; Dhanavade, M.; Sonawane, K.; Garadkar, K. Enhanced photocatalytic activity of europium doped TiO2 under sunlight for the degradation of methyl orange. J. Mater. Sci. Mater. Electron. 2017, 28, 11002–11011. [Google Scholar] [CrossRef]





| Sr. No. | Source | No. of Publications in Years | |||||
|---|---|---|---|---|---|---|---|
| 2019 | 2020 | 2021 | 2022 | 2023 | 2019–2023 | ||
| 1 | Google Scholar | 545 | 783 | 951 | 1580 | 668 | 4527 |
| 2 | Science Direct | 6 | 6 | 20 | 15 | 10 | 57 |
| Sr. No. | Composite | Ratio | Photodegradation %Age Efficiency |
|---|---|---|---|
| 1 | AC | pure | 55% [75] |
| 2 | TiO2 | Pure | 71–73% [75] |
| 3 | TiO2/AC | NA | 55% [86] |
| 4 | TiO2@AC | NA | 65% [87] |
| 5 | TiO2/MWCNT | NA | 61.1% [83] |
| 6 | TiO2@carbon nanotube | NA | 85% [84] |
| 7 | AC-TiO2/(OFL) | NA | 82% [85] |
| 8 | TiO2/AC/2D MoSe2 | 1:0.25:1 | 76% present work |
| 9 | TiO2/AC/2D MoSe2 | 1:0.5:1 | 80% present work |
| 10 | TiO2/AC/2D MoSe2 | 1:0.75:1 | 81% present work |
| 11 | TiO2/AC/2D MoSe2 | 1:1:1 | 83% present work |
| Photocatalyst | Time (min) | 0 | 30 | 60 | 90 | 120 | 150 |
|---|---|---|---|---|---|---|---|
| Pure TiO2 | MB mg/L | 10 | 9 | 7.3 | 5.1 | 4.2 | 2.9 |
| TiO2/0.25AC/2DMoSe2 | 10 | 6.2 | 5.3 | 4.1 | 2.9 | 2.4 | |
| TiO2/0.50AC/2DMoSe2 | 10 | 7.6 | 6.6 | 4.8 | 3.3 | 2.0 | |
| TiO2/0.75AC/2DMoSe2 | 10 | 7.0 | 6.5 | 5.5 | 3.5 | 1.9 | |
| TiO2/AC/2DMoSe2 | 10 | 6.9 | 6.4 | 5.3 | 3.6 | 1.7 |
| Sr. No. | MB (mg/L) | % Degradation |
|---|---|---|
| 1 | 5 | 87 |
| 2 | 10 | 83 |
| 3 | 15 | 74 |
| 4 | 20 | 69 |
| 6 | 25 | 65 |
| Sr. No. | pH | Percentage Degradation |
|---|---|---|
| 1 | 7.0 | 72 |
| 2 | 7.5 | 74 |
| 3 | 8.0 | 79 |
| 4 | 8.5 | 80 |
| 5 | 9.0 | 82 |
| 6 | 9.5 | 83 |
| 7 | 10.0 | 81 |
| Sr. No. | Catalyst Loading (g/dm3) | Percentage Degradation |
|---|---|---|
| 1 | 0.01 | 79 |
| 2 | 0.10 | 83 |
| 3 | 0.15 | 73 |
| 4 | 0.20 | 66 |
| 5 | 0.25 | 62 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ahmad, S.; Tahir, M.S.; Kamal, G.M.; Zhang, X.; Nazir, S.; Tahir, M.B.; Jiang, B.; Safdar, M. TiO2/Activated Carbon/2D Selenides Composite Photocatalysts for Industrial Wastewater Treatment. Water 2023, 15, 1788. https://doi.org/10.3390/w15091788
Ahmad S, Tahir MS, Kamal GM, Zhang X, Nazir S, Tahir MB, Jiang B, Safdar M. TiO2/Activated Carbon/2D Selenides Composite Photocatalysts for Industrial Wastewater Treatment. Water. 2023; 15(9):1788. https://doi.org/10.3390/w15091788
Chicago/Turabian StyleAhmad, Shehzad, Muhammad Suleman Tahir, Ghulam Mustafa Kamal, Xu Zhang, Saima Nazir, Muhammad Bilal Tahir, Bin Jiang, and Muhammad Safdar. 2023. "TiO2/Activated Carbon/2D Selenides Composite Photocatalysts for Industrial Wastewater Treatment" Water 15, no. 9: 1788. https://doi.org/10.3390/w15091788





