Biotic Integrity, Water Quality, and Landscape Characteristics of a Subtropical River
Abstract
:1. Introduction
2. Materials and Methods
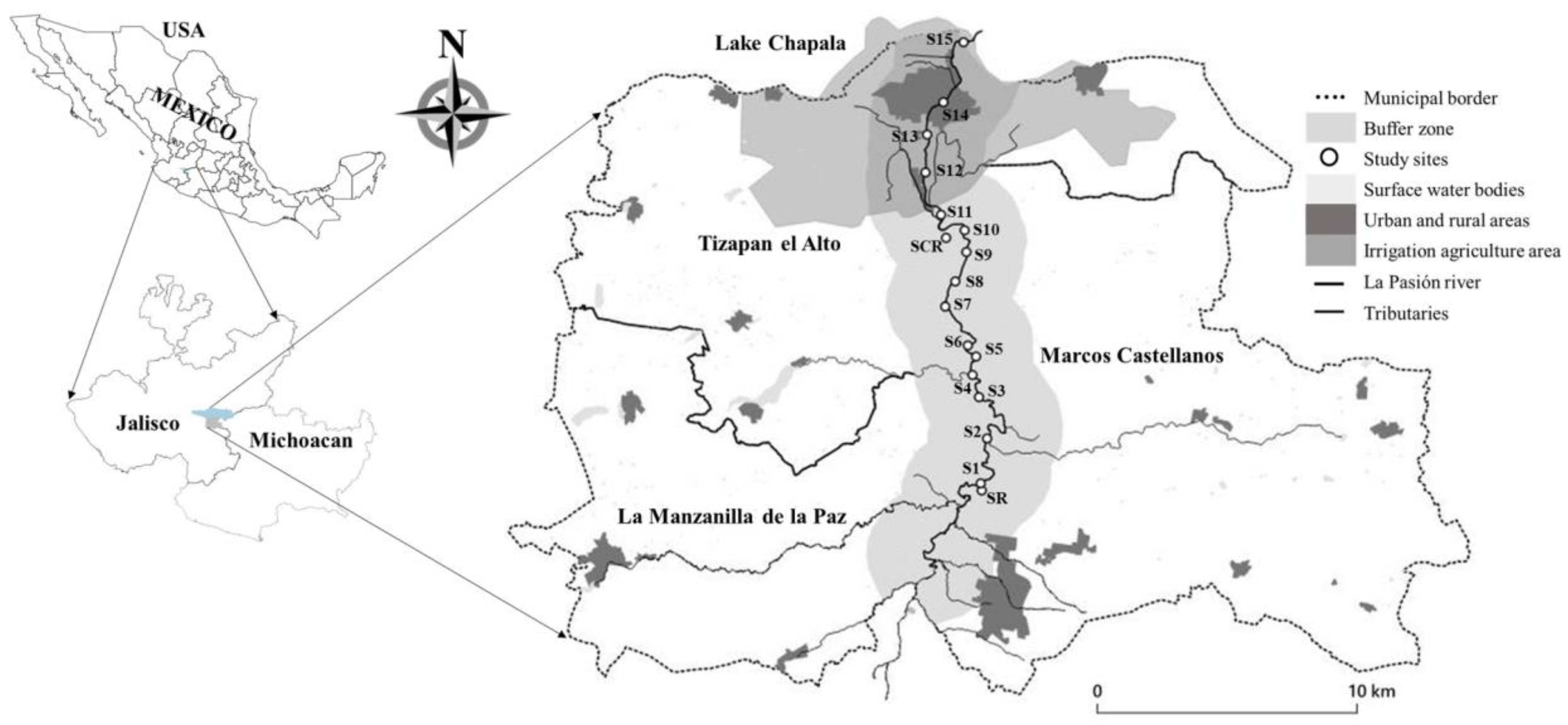
2.1. Study Area
2.2. Fieldwork and Sample Analysis
2.3. Hydrogeomorphological Characteristics and Indices
2.4. Abiotic and Biotic Indices
2.5. Statistical Analyses
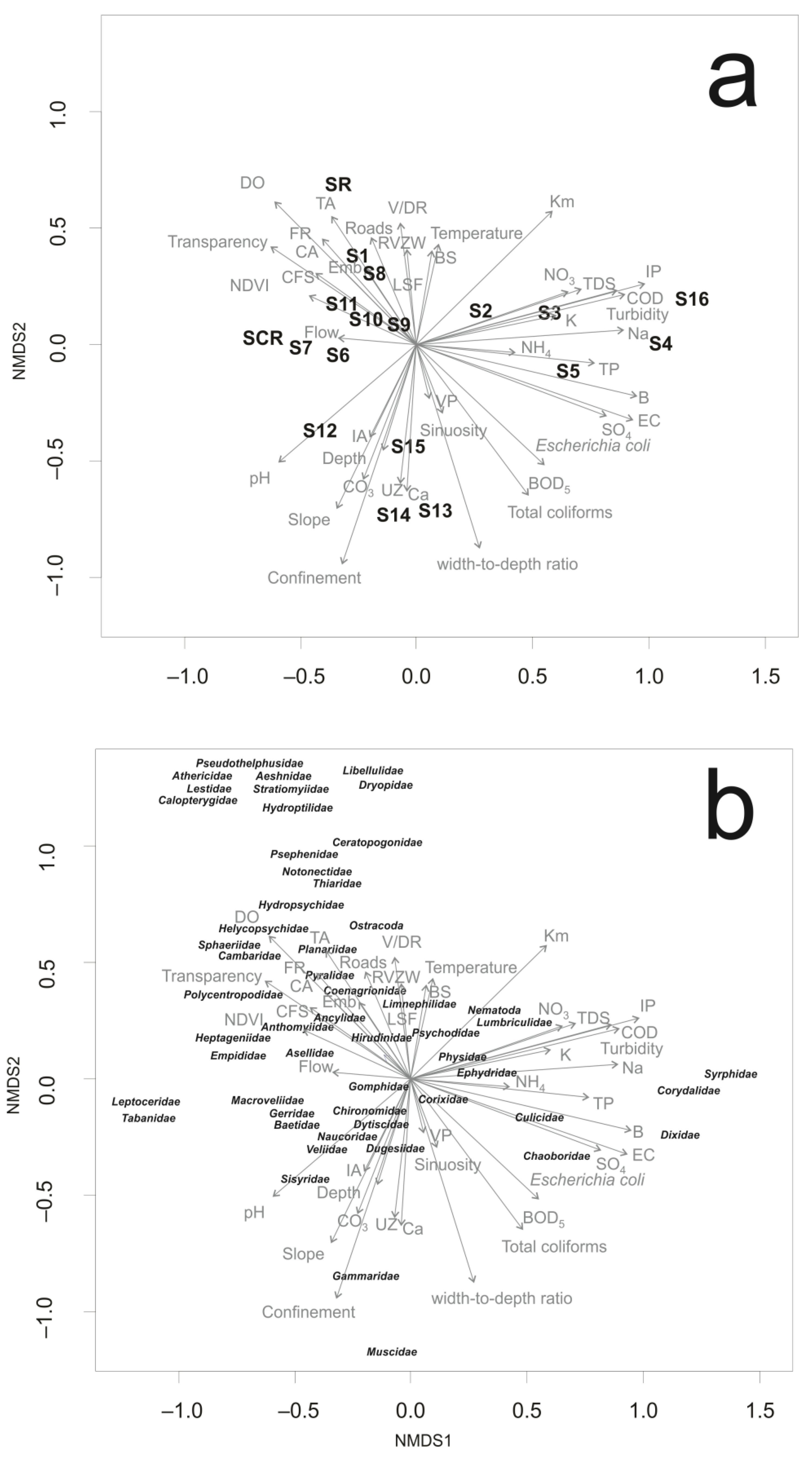
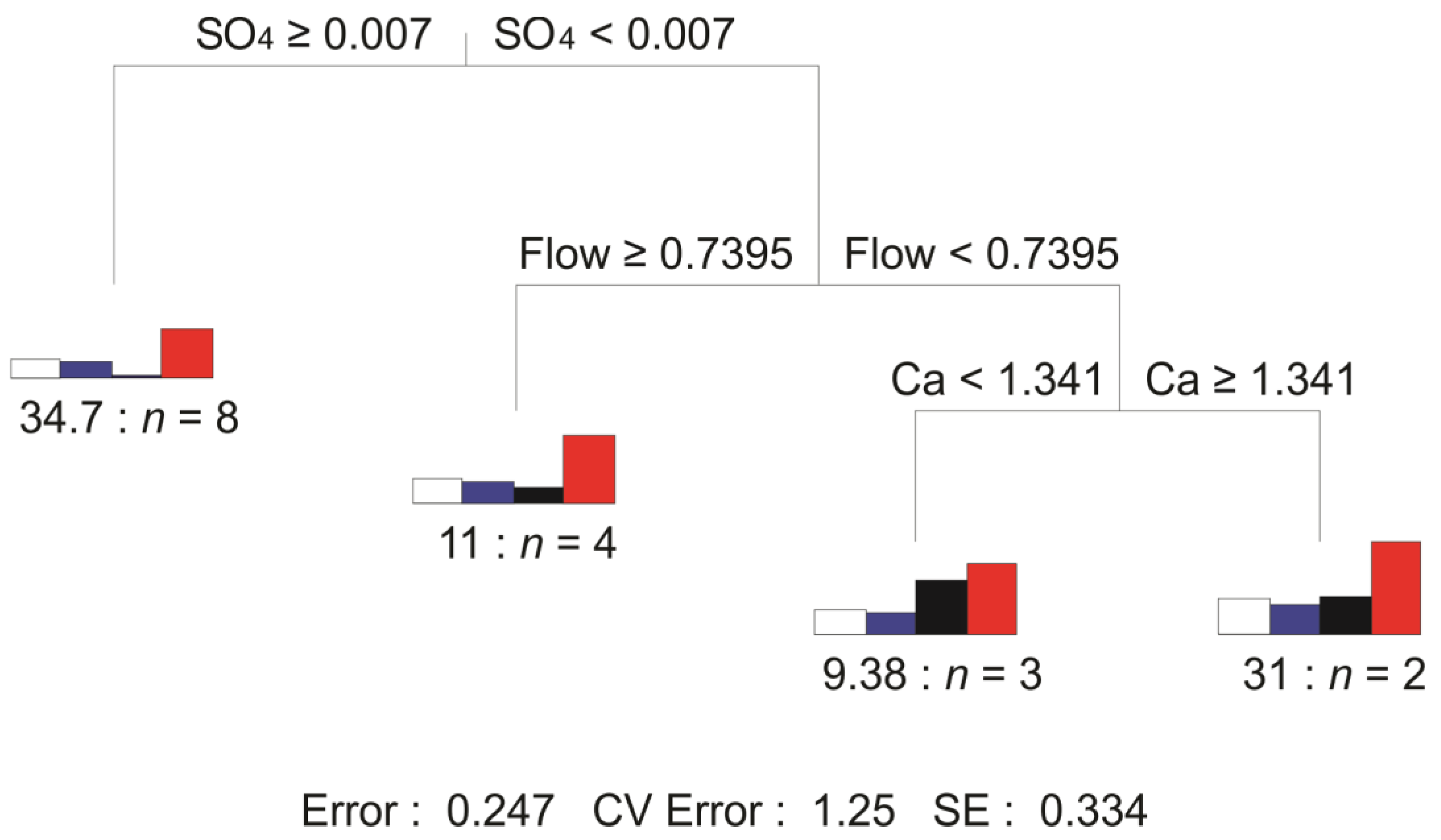
3. Results
3.1. Landscape Characteristics
3.2. Water and Habitat Characteristics
3.3. Macroinvertebrates Characteristics
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| MLR | Machine learning regression algorithm |
| NDVI | Normalized difference vegetation index |
| NSF-WQI | National Sanitation Foundation index of water quality |
| EPT-B % | Percentage of Ephemeroptera, Plecoptera, and Trichoptera (minus Baetidae) |
| BMWP | Biological Monitoring Working Party index |
| NMDS | Non-metric multidimensional scaling |
| MRT | Multivariate regression tree analysis |
| HQI | Habitat quality index |
| B | Boron |
| BS | Bank Stability |
| CA | Channel alteration |
| CFS | Channel flow status |
| Emb | Embeddedness |
| FE | Frequency of riffles |
| IA | Irrigated agriculture |
| IP | Induced pastureland |
| LSF | Lowland semideciduous forest |
| RVZW | Riparian vegetative zone width |
| RA | Rainfed agriculture |
| UZ | Urban zone |
| VP | Vegetative protection |
| V/DR | Velocity/depth regime |
Appendix A
| Sites | pH | EC (µS/cm) | Ca2+ (mg/L) | Mg2+ (mg/L) | Na+ (mg/L) | K+ (mg/L) | CO3 (mg/L) | HCO3 (mg/L) | Cl (mg/L) | SO42− (mg/L) | B (mg/L) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| S1 | 7.73 | 264.3 | 1.44 | 3.32 | 1.77 | 2.95 | 0.13 | 3.13 | 15.09 | 0.0002 | 0 |
| S2 | 7.98 | 423 | 1.49 | 2.28 | 2.02 | 3.33 | 0 | 4.20 | 26.41 | 0.011 | 0.249 |
| S3 | 8.24 | 502 | 1.64 | 2.25 | 2.12 | 4.48 | 0 | 4.85 | 33.95 | 0.008 | 0.21 |
| S4 | 6.87 | 1664 | 2.21 | 3.00 | 7.18 | 5.65 | 0 | 9.51 | 192.39 | 0.073 | 0.843 |
| S5 | 7.68 | 690 | 1.12 | 3.83 | 1.22 | 3.31 | 0 | 4.35 | 18.86 | 0.047 | 0 |
| S6 | 7.18 | 368 | 1.24 | 3.58 | 2.05 | 3.81 | 0 | 3.62 | 22.63 | 0.006 | 0 |
| S7 | 7.64 | 321 | 1.04 | 0.49 | 0.80 | 2.69 | 0 | 3.49 | 18.86 | 0.0003 | 0 |
| S8 | 7.65 | 340 | 1.69 | 0.74 | 1.05 | 3.82 | 0 | 3.54 | 18.86 | 0.002 | 0 |
| S9 | 7.65 | 347 | 1.32 | 1.36 | 2.47 | 3.27 | 0 | 3.56 | 18.86 | 0.0018 | 0 |
| S10 | 7.57 | 341 | 1.54 | 1.15 | 1.26 | 3.88 | 0 | 3.56 | 18.86 | 0.004 | 0 |
| S11 | 8.04 | 332 | 1.11 | 3.94 | 1.82 | 4.76 | 0.25 | 3.54 | 18.86 | 0.006 | 0 |
| S12 | 8.08 | 360.2 | 1.99 | 0.56 | 1.15 | 4.08 | 0 | 3.62 | 15.09 | 0.008 | 0 |
| S13 | 8.49 | 464 | 3.51 | 3.36 | 1.86 | 3.28 | 0.56 | 5.18 | 26.41 | 0.012 | 0.2235 |
| S14 | 8.29 | 546 | 3.93 | 3.88 | 2.89 | 3.83 | 0.33 | 5.59 | 30.18 | 0.015 | 0.275 |
| S15 | 7.71 | 1004 | 0.08 | 5.22 | 3.39 | 2.73 | 0 | 9.58 | 49.04 | 0.102 | 0.468 |
| SR | 7.73 | 264.3 | 1.44 | 3.32 | 1.77 | 2.95 | 0.13 | 3.13 | 15.09 | 0.0002 | 0 |
| SCR | 8.04 | 332 | 1.11 | 3.94 | 1.82 | 4.76 | 0.25 | 3.54 | 18.86 | 0.006 | 0 |
| Sites | NH4 (mg/L) | PO43−TP (mg/L) | Hardness (mg/L CaCO3) | NO3 (mg/L) | COD (mg/L) | O and G (mg/L) | BOD5 (mg/L) | TN (mg/L) | ET (°C) | WT (°C) | DOPS (%) |
| S1 | 4.71 | 0.12 | 100 | 0 | 12.33 | 0 | 2.12 | 4.71 | 17.7 | 25.8 | 95.9 |
| S2 | 1.17 | 1.23 | 115 | 0.04 | 15.67 | 0.25 | 2.7 | 1.21 | 18 | 25 | 61.7 |
| S3 | 1.42 | 0.55 | 160 | 0.32 | 149 | 0.04 | 27.21 | 1.74 | 17.3 | 17.9 | 47.3 |
| S4 | 2.38 | 25.87 | 340 | 2.16 | 1065.67 | 0.27 | 477.19 | 4.55 | 16.7 | 18.1 | 44.2 |
| S5 | 1.25 | 0.18 | 130 | 0 | 42.33 | 0 | 369.05 | 1.25 | 17.3 | 18.2 | 39.8 |
| S6 | 11.57 | 3.65 | 130 | 0.10 | 12.33 | 0 | 188.54 | 11.66 | 19.2 | 18.7 | 46.4 |
| S7 | 2.39 | 0.00 | 135 | 0.10 | 159 | 0 | 27.4 | 2.50 | 19.1 | 18.5 | 38.9 |
| S8 | 2.04 | 3.33 | 110 | 0 | 185.67 | 0.24 | 81.9 | 2.04 | 19.4 | 18.6 | 51.5 |
| S9 | 3.25 | 3.01 | 125 | 0.10 | 29 | 0.13 | 62.83 | 3.35 | 19 | 18.5 | 59.2 |
| S10 | 1.88 | 0.37 | 125 | 0 | 2.33 | 0.24 | 25.04 | 1.88 | 19.2 | 18.7 | 54.6 |
| S11 | 3.10 | 2.83 | 125 | 0.72 | 12.33 | 0.01 | 107.3 | 3.82 | 19.2 | 18.7 | 60.5 |
| S12 | 2.04 | 3.20 | 130 | 0.35 | 39 | 0 | 596.36 | 2.39 | 23.1 | 19.7 | 59.7 |
| S13 | 6.59 | 0.12 | 210 | 0.10 | 12.33 | 0 | 188.54 | 6.68 | 24.7 | 19.7 | 44.3 |
| S14 | 2.27 | 1.28 | 230 | 0.01 | 19 | 0.22 | 165.65 | 2.29 | 23.2 | 21 | 53.6 |
| S15 | 13.96 | 22.07 | 305 | 0.44 | 142.33 | 0.03 | 667.02 | 14.40 | 28.7 | 21.1 | 45.9 |
| SR | 4.71 | 0.12 | 100 | 0 | 12.33 | 0 | 2.12 | 4.71 | 17.7 | 25.8 | 95.9 |
| SCR | 3.10 | 2.83 | 125 | 0.72 | 12.33 | 0.01 | 107.3 | 3.82 | 19.2 | 18.7 | 60.5 |
| Sites | DO (mg/L) | Sal (‰) | Trans (cm) | Turb (NTU) | TDS (mg/L) | TotCol (MPN 100 mL) | FecCol (MPN 100 mL) | E. coli (MPN 100 mL) | Flow (m3/s) | ||
| S1 | 6.27 | 0.13 | 0.61 | 19 | 166.4 | 40 | 30 | 30 | 0.27 | ||
| S2 | 4.11 | 0.11 | 0.13 | 65 | 147.2 | 230 | 150 | 100 | 0.64 | ||
| S3 | 3.64 | 0.14 | 0.05 | 100 | 205.8 | 2100 | 430 | 110 | 0.69 | ||
| S4 | 3.39 | 0.14 | 0.25 | 50 | 205.8 | 930 | 280 | 40 | 0.70 | ||
| S5 | 3.04 | 0.13 | 0.11 | 50 | 198.45 | 1500 | 930 | 280 | 1.19 | ||
| S6 | 3.52 | 0.12 | 0.16 | 40 | 183.75 | 2400 | 1500 | 150 | 0.49 | ||
| S7 | 2.97 | 0.13 | 0.16 | 35 | 198.45 | 930 | 150 | 90 | 0.69 | ||
| S8 | 3.92 | 0.09 | 0.38 | 13 | 132.3 | 280 | 210 | 90 | 0.95 | ||
| S9 | 4.52 | 0.11 | 0.55 | 14 | 169.05 | 230 | 110 | 30 | 0.79 | ||
| S10 | 4.16 | 0.12 | 0.65 | 10 | 176.4 | 210 | 110 | 90 | 1.01 | ||
| S11 | 4.63 | 0.09 | 0.39 | 11 | 139.65 | 430 | 90 | 40 | 0.69 | ||
| S12 | 4.5 | 0.11 | 0.55 | 9 | 169.05 | 210 | 110 | 70 | 0.30 | ||
| S13 | 3.34 | 0.10 | 0.56 | 30 | 147 | 4600 | 1500 | 90 | 0.62 | ||
| S14 | 3.95 | 0.10 | 0.24 | 24 | 154.35 | 1500 | 230 | 110 | 0.51 | ||
| S15 | 3.38 | 0.11 | 0.13 | 24 | 169.05 | 2100 | 930 | 280 | 0.64 | ||
| SR | 6.27 | 0.13 | 0.93 | 19 | 166.4 | 40 | 30 | 30 | 0.27 | ||
| SCR | 4.63 | 0.09 | 0.54 | 11 | 139.65 | 430 | 90 | 40 | 6.97 | ||
References
- Best, J. Anthropogenic stresses on the world’s big rivers. Nat. Geosci. 2019, 12, 7–21. [Google Scholar] [CrossRef]
- Anderson, E.P.; Jackson, S.; Tharme, R.E.; Douglas, M.; Flotemersch, J.E.; Zwarteveen, M.; Lokgariwar, C.; Montoya, M.; Wali, A.; Tipa, G.T.; et al. Understanding rivers and their social relations: A critical step to advance environmental water management. WIREs Water 2019, 6, 1–32. [Google Scholar] [CrossRef] [PubMed]
- Tickner, D.; Parker, H.; Moncrieff, C.R.; Oates, N.E.; Ludi, E.; Acreman, M. Managing rivers for multiple benefits–a coherent approach to research, policy and planning. Front. Environ. Sci. 2017, 5, 1–8. [Google Scholar] [CrossRef]
- Moyle, P.B.; Leidy, R.A. Loss of Biodiversity in Aquatic Ecosystems: Evidence from Fish Faunas. In Conservation Biology; Fiedler, P.L., Jain, S.K., Eds.; Springer: Boston, MA, USA, 1992; pp. 127–169. [Google Scholar] [CrossRef]
- Rico-Sánchez, A.E.; Rodríguez-Romero, A.J.; Sedeño-Díaz, J.E.; López-López, E.; Sundermann, A. Aquatic macroinvertebrate assemblages in rivers influenced by mining activities. Sci. Rep. 2022, 12, 3209. [Google Scholar] [CrossRef]
- Roe, D.; Seddon, N.; Elliott, J. Biodiversity Loss is a Development Issue: A Rapid Review of Evidence; IIED: London, UK, 2019; pp. 1–23. [Google Scholar]
- Dopelt, K.; Radon, P.; Davidovitch, N. Environmental effects of the livestock industry: The relationship between knowledge, attitudes, and behavior among students in Israel. Int. J. Environ. 2019, 16, 1359. [Google Scholar] [CrossRef]
- Landrigan, P.J.; Fuller, R. Global health and environmental pollution. Int. J. Public Health 2015, 60, 761–762. [Google Scholar] [CrossRef]
- Chen, F.L.; Yang, B.C.; Peng, S.Y.; Lin, T.C. Applying a deployment strategy and data analysis model for water quality continuous monitoring and management. IJDSN 2020, 16, 1–15. [Google Scholar] [CrossRef]
- Soltani, A.A.; Oukil, A.; Boutaghane, H.; Bermad, A.; Boulassel, M.R. A new methodology for assessing water quality based on data envelopment analysis: Application to Algerian dams. Ecol. Indic. 2021, 121, 106952. [Google Scholar] [CrossRef]
- Sumudumali, R.G.I.; Jayawardana, J.M.C.K. A review of biological monitoring of aquatic ecosystems approaches: With special reference to macroinvertebrates and pesticide pollution. Environ. Manag. 2021, 67, 263–276. [Google Scholar] [CrossRef]
- Cruz-García, L.M.; Arreola-Lizarraga, J.A.; Mendoza-Salgado, R.A.; Galina-Tessaro, P.; Beltrán-Morales, L.F.; Ortega-Rubio, A. Applying ecological diversity indices with ecosystem approach at ecoregional level and prioritizing the decree of new protected natural areas. Interciencia 2015, 40, 179–184. [Google Scholar]
- Liquete, C.; Cid, N.; Lanzanova, D.; Grizzetti, B.; Reynaud, A. Perspectives on the link between ecosystem services and biodiversity: The assessment of the nursery function. Ecol. Indic. 2016, 63, 249–257. [Google Scholar] [CrossRef]
- Hernández-García, A.; Sandoval-Moreno, A. Regionalización y cambios territoriales en las Ciénegas de Jalisco-Michoacán, México (1990–2015). Agua y Territ. 2018, 12, 49–58. [Google Scholar] [CrossRef]
- QGIS Development Team. QGIS Geographic Information System. Open Source Geospatial Foundation Project. Available online: http://qgis.osgeo.org (accessed on 10 October 2021).
- INEGI Digital Elevation Model Archives (DEM). Available online: https://www.inegi.org.mx/app/geo2/elevacionesmex/ (accessed on 9 February 2020).
- Wang, L.; Liu, H. An efficient method for identifying and filling surface depressions in digital elevation models for hydrologic analysis and modelling. Int. J. Geogr. Inf. Syst. 2006, 20, 193–213. [Google Scholar] [CrossRef]
- Wu, T.; Li, J.; Li, T.; Sivakumar, B.; Zhang, G.; Wang, G. High-efficient extraction of drainage networks from digital elevation models constrained by enhanced flow enforcement from known river maps. Geomorphology 2019, 340, 184–201. [Google Scholar] [CrossRef]
- Barbour, M.T.; Gerritsen, J.; Snyder, B.D.; Stribling, J.B. Rapid Bioassessment Protocols for Use in Streams and Wadeable Rivers: Periphyton, Benthic Macroinvertebrates and Fish, 2nd ed.; US Environmental Protection Agency, Office of Water: Washington, DC, USA, 1999; pp. 1–337. [Google Scholar]
- Stenert, C.; de Mello, Í.C.; Pires, M.M.; Knauth, D.S.; Katayama, N.; Maltchik, L. Responses of macroinvertebrate communities to pesticide application in irrigated rice fields. Environ. Monit. Assess. 2018, 190, 1–13. [Google Scholar] [CrossRef]
- Arabameri, A.; Pradhan, B.; Pourghasemi, H.R.; Rezaei, K.; Kerle, N. Spatial Modelling of Gully Erosion Using GIS and R Programing: A Comparison among Three Data Mining Algorithms. Appl. Sci. 2018, 8, 1369. [Google Scholar] [CrossRef]
- Baird, R.B.; Eaton, A.D.; Rice, E.W. Methods for the Examination of Water and Wastewater, 23rd ed.; American Public Health Association (APHA), AWWA & WEF: Washington, DC, USA, 2017; pp. 43–1537. [Google Scholar]
- Barbour, M.T.; Stribling, J.B.; Verdonschot, P.F.M. The multihabitat approach of USEPA’s rapid bioassessment protocols: Benthic macroinvertebrates. Limnetica 2006, 25, 839–850. [Google Scholar] [CrossRef]
- Norma DOF Para Análisis de Agua (NMX-AA-005-SCFI-2013). Medición de Grasas y Aceites Recuperables en Aguas Naturales, Residuales y Residuales Tratadas. Método de Prueba. Available online: https://www.gob.mx/cms/uploads/attachment/file/166764/nmx-aa-005-scfi-2013.pdf (accessed on 22 November 2020).
- Norma DOF Para Análisis de Agua (NOM-112-SSA1-1994). Determinación de Bacterias Coliformes por la Técnica del Número Más Probable. Available online: http://www.ordenjuridico.gob.mx/Documentos/Federal/wo69535.pdf (accessed on 15 October 2020).
- Velázquez-Machuca, M.A.; Pimentel-Equihua, J.L.; Ortega-Escobar, M. Variaciones longitudinales y temporales en la hidroquímica del río Duero. Agrociencia 2010, 44, 599–609. [Google Scholar]
- Pineda-López, R.; Pérez-Munguia, R.M.; Mathuriau, C.; Villalobos-Hiriart, J.L.; Barba-Álvarez, R.; Bernal, T.; Barba-Macías, E. Protocolo de Muestreo de Macroinvertebrados en Aguas Continentales para la Aplicación de la Norma de Caudal Ecológico (NMX-AA-159-SCFI-2012). 2014, PNRA, México. Available online: https://agua.org.mx/biblioteca/protocolo-de-muestreo-de-macroinvertebrados-en-aguas-continentales-para-la-aplicacion-de-la-norma-de-caudal-ecologico-nmx%E2%80%90aa%E2%80%90159%E2%80%90scfi%E2%80%902012/ (accessed on 2 October 2022).
- Flotemersch, J.E.; Saavedra, J.M.; Laffitte, L.; Laurenzano, B.; Bonardi, M.A.; Blocksom, K.A. Benthic macroinvertebrate field sampling effort required to produce a sample adequate for the assessment of rivers and streams of Neuquén Province, Argentina. Limnologica 2017, 65, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Oscoz, J.; Galicia, D.; Miranda, R. Identification Guide of Freshwater Macroinvertebrates of Spain; Springer Science & Business Media: Dordrecht, The Netherlands, 2011; pp. 1–140. ISBN 978-94-007-1554-7. [Google Scholar]
- Pennak, R.W. Fresh-Water Invertebrates of United States, 2nd ed.; Wiley-Interscience Pub.: New York, NY, USA, 1978; pp. 1–782. [Google Scholar]
- Rodríguez-Romero, A.J.; Rico-Sánchez, A.E.; Mendoza-Martínez, E.; Gómez-Ruiz, A.; Sedeño-Díaz, J.E.; López-López, E. Impact of Changes of Land Use on Water Quality, from Tropical Forest to Anthropogenic Occupation: A Multivariate Approach. Water 2018, 10, 1518. [Google Scholar] [CrossRef]
- Cornejo, A.; López-López, E.; Bernal, J. Protocolo de Biomonitoreo para la Vigilancia de la Calidad del Agua en Afluentes Superficiales de Panama; Instituto Conmemorativo Gorgas de Estudios de la Salud: Panamá, Panamá, 2019; pp. 1–81. [Google Scholar]
- Rosgen, D.L. A classification of natural rivers. Catena 1994, 22, 169–199. [Google Scholar] [CrossRef]
- Dhingra, S.; Kumar, D. A review of remotely sensed satellite image classification. IJECE 2019, 9, 1720–1731. [Google Scholar] [CrossRef]
- Li, M.; Zang, S.; Zhang, B.; Li, S.; Wu, C. A Review of Remote Sensing Image Classification Techniques: The Role of Spatio-contextual Information. Eur. J. Remote Sens. 2014, 47, 389–411. [Google Scholar] [CrossRef]
- Salinas-Zavala, M.V.; Martínez-Rincón, R.O.; Morales-Zárate, M.V. Tendencia en el siglo XXI del Índice de Diferencias Normalizadas de Vegetación (NDVI) en la parte sur de la península de Baja California. Investig. Geogr. 2017, 94, 82–90. [Google Scholar] [CrossRef]
- Luvai, A.; Obiero, J.; Omuto, C. Soil Loss Assessment Using the Revised Universal Soil Loss Equation (RUSLE) Model. Appl. Environ. Soil. Sci. 2022, 2022, 2122554. [Google Scholar] [CrossRef]
- Prontuarios Municipales INEGI. Available online: https://www.inegi.org.mx/app/areasgeograficas/#collapse-Resumen (accessed on 17 January 2020).
- Kachroud, M.; Trolard, F.; Kefi, M.; Jebari, S.; Bourrié, G. Water quality indices: Challenges and application limits in the literature. Water 2019, 11, 361. [Google Scholar] [CrossRef]
- Noori, R.; Berndtsson, R.; Hosseinzadeh, M.; Adamowski, J.F.; Abyaneh, M.R. A critical review on the application of the National Sanitation Foundation Water Quality Index. Environ. Pollut. 2019, 244, 575–587. [Google Scholar] [CrossRef]
- Water Quality Index. Field Manual for Water Quality Monitoring, the National Sanitation Foundation. Available online: https://www.knowyourh2o.com/outdoor-3/water-quality-index-calculator-for-surface-water (accessed on 20 March 2022).
- Budka, A.; Łacka, A.; Szoszkiewicz, K. The use of rarefaction and extrapolation as methods of estimating the effects of river eutrophication on macrophyte diversity. Biodivers. Conserv. 2019, 28, 385–400. [Google Scholar] [CrossRef]
- Chao, A.; Gotelli, N.J.; Hsieh, T.C.; Sander, E.L.; Ma, K.H.; Colwell, R.K.; Ellison, A.M. Rarefaction and extrapolation with Hill numbers: A framework for sampling and estimation in species diversity studies. Ecol. Monogr. 2014, 84, 45–67. [Google Scholar] [CrossRef]
- Masese, F.O.; Raburu, P.O. Improving the performance of the EPT Index to accommodate multiple stressors in Afrotropical streams. Afr. J. Aquat. Sci. 2017, 42, 219–233. [Google Scholar] [CrossRef]
- Magallón-Ortega, G.; Escalera-Gallardo, C.; López-López, E.; Sedeño-Díaz, J.E.; López-Hernández, M.; Arroyo-Damián, M.; Moncayo-Estrada, R. Water Quality Analysis in a Subtropical River with an Adapted Biomonitoring Working Party (BMWP) Index. Diversity 2021, 13, 606. [Google Scholar] [CrossRef]
- Ruiz-Picos, R.A.; Sedeño-Díaz, J.E.; López-López, E. Calibrating and validating the Biomonitoring Working Party (BMWP) index for the Bioassessment of water quality in neotropical streams. Water Qual. 2017, 3, 39–58. [Google Scholar] [CrossRef]
- Kennen, J.G.; Sullivan, D.J.; May, J.T.; Bell, A.H.; Beaulieu, K.M.; Rice, D.E. Temporal changes in aquatic-invertebrate and fish assemblages in streams of the north-central and northeastern US. Ecol. Indic. 2012, 18, 312–329. [Google Scholar] [CrossRef]
- Jia, Y.; Sui, X.; Chen, Y. Development of a Fish-Based Index of Biotic Integrity for Wadeable Streams in Southern China. Environ. Manag. 2013, 52, 995–1008. [Google Scholar] [CrossRef]
- Borcard, D.; Gillet, F.; Legendre, P. Numerical Ecology with R, 2nd ed.; Springer: New York, NY, USA, 2011; pp. 1–292. [Google Scholar] [CrossRef]
- McCune, B.; Grace, J.; Urban, D. Analysis of Ecological Communities; MjM Software Design: Gleneden Beach, OR, USA, 2002; pp. 1–304. ISBN 0-9721290-0-6. [Google Scholar]
- De’ath, G. Multivariate regression trees: A new technique for modeling species-environment relationships. Ecology 2002, 83, 1105–1117. [Google Scholar]
- McCune, B.; Mefford, M.J. PC-ORD. Multivariate Analysis of Ecological Data. Available online: www.bockytech.com.tw/PDF-File/PC-ORD.pdf (accessed on 5 January 2021).
- Peck, J.E. Multivariate Analysis for Ecologists: Step-by-Step, 2nd ed.; MjM Software Design: Gleneden Beach, OR, USA, 2016; pp. 1–192. [Google Scholar]
- Breiman, L. Random forests. Mach. Learn. 2001, 45, 5–32. [Google Scholar] [CrossRef]
- Dinno, A. Dunn. Test: Dunn’s Test of Multiple Comparisons Using Rank Sums. R Package Version 1.3.5. Available online: http://cran.stat.unipd.it/web/packages/dunn.test/dunn.test.pdf (accessed on 15 October 2020).
- Oksanen, J.; Blanchet, F.G.; Kindt, R.; Legendre, P.; O’Hara, R.B.; Simpson, G.L.; Solymos, P.; Stevens, M.H.H.; Wagner, H. Vegan: Community Ecology Package. R Package Version 2.5-7. Available online: https://cran.r-project.org/web/packages/vegan/index.html (accessed on 7 October 2021).
- De’ath, G.; Therneau, T.M.; Atkinson, B.; Ripley, B.; Oksanen, J. Mvpart: Multivariate Partitioning. Available online: https://mran.microsoft.com/snapshot/2014-12-11/web/packages/mvpart/index.html (accessed on 12 March 2020).
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2021; Available online: https://www.R-project.or.rg/ (accessed on 16 August 2021).
- Aazami, J.; Esmaili-Sari, A.; Abdoli, A.; Sohrabi, H.; Van den Brink, P.J. Monitoring and assessment of water health quality in the Tajan River, Iran using physicochemical, fish and macroinvertebrates indices. J. Environ. Health Sci. Eng. 2015, 13, 1–12. [Google Scholar] [CrossRef]
- Englert, D.; Zubrop, J.P.; Schulz, R.; Bundschun, M. Variability in ecosystem structure and functioning in a low order strem: Implications of land use and season. Sci. Total Environ. 2015, 538, 341–349. [Google Scholar] [CrossRef]
- López-López, E.; Sedeño-Díaz, J.E.; Mendoza-Martínez, E.; Gómez-Ruiz, A.; Martínez-Ramírez, E. Water quality and macroinvertebrate community in dryland streams: The case of the Tehuacán-Cuicatlán Biosphere Reserve (México) facing climate change. Water 2019, 11, 1376. [Google Scholar] [CrossRef]
- El Sayed, S.M.; Hegab, M.H.; Mola, H.R.A.; Ahmed, N.M.; Goher, M.E. An integrated water quality assessment of Damietta and Rosetta branches (Nile River, Egypt) using chemical and biological indices. Environ. Monit. Assess. 2020, 192, 228. [Google Scholar] [CrossRef]
- Kaelin, K.; Altermatt, F. Landscape-level predictions of diversity in river networks reveal opposing patterns for different groups of macroinvertebrates. Aquat. Ecol. 2016, 50, 283–295. [Google Scholar] [CrossRef]
- Robinson, C.T.; Schweizer, P.; Larsen, A.; Schubert, C.J.; Siebers, A.R. Beaver effects on macroinvertebrate assemblages in two streams with contrasting morphology. Sci. Total Environ. 2020, 722, 137899. [Google Scholar] [CrossRef]
- Upadhyay, K.D.; Mainali, J.; Ghimire, N.P. Diversity of aquatic plants and macroinvertebrates and their spatial patterns in a Himalayan Watershed, Central Nepal. Aquat. Bot. 2022, 180, 103529. [Google Scholar] [CrossRef]
- Niculae, M.I.; Avram, S.; Corpade, A.M.; Dedu, S.; Gheorghe, C.A.; Pascu, I.S.; Ontel, I.; Rodino, S. Evaluation of the quality of lentic ecosystems in Romania by a GIS based WRASTIC model. Sci. Rep. 2021, 11, 5361. [Google Scholar] [CrossRef] [PubMed]
- Robinson, C.T.; Tockner, K.; Ward, J.V. The fauna of dynamic riverine landscapes. Freshw. Biol. 2002, 47, 661–677. [Google Scholar] [CrossRef]
- Calderón-Cendejas, J.; Madrid-Ramírez, L.; Ramírez-Zierold, J.; Díaz-Valenzuela, J.; Merino-Ibarra, M.; Morató-Sánchez de Tagle, S.; Chino-Téllez, A. Evaluation of the impacts of land use in water quality and the role of nature-based solutions: A citizen science-based study. Sustainability 2021, 13, 10519. [Google Scholar] [CrossRef]
- Crooks, E.C.; Harris, I.M.; Patil, S.D. Influence of Land Use Land Cover on River Water Quality in Rural North Wales, UK. JAWRA 2021, 57, 357–373. [Google Scholar] [CrossRef]
- Fierro, P.; Bertrán, C.; Tapia, J.; Hauenstein, E.; Peña-Cortés, F.; Vergara, C.; Cerna, C.; Vargas-Chacoff, L. Effects of local land-use on riparian vegetation, water quality, and the functional organization of macroinvertebrate assemblages. Sci. Total Environ. 2017, 609, 724–734. [Google Scholar] [CrossRef]
- Árva, D.; Specziár, A.; Erős, T.; Tóth, M. Effects of habitat types and within lake environmental gradients on the diversity of chironomid assemblages. Limnologica 2015, 53, 26–34. [Google Scholar] [CrossRef]
- Gething, K.J.; Ripley, M.C.; Mathers, K.L.; Chadd, R.P.; Wood, P.J. The influence of substrate type on macroinvertebrate assemblages within agricultural drainage ditches. Hydrobiologia 2020, 847, 4273–4284. [Google Scholar] [CrossRef]
- Porst, G.; Brauns, M.; Irvine, K.; Solimini, A.; Sandin, L.; Pusch, M.; Miler, O. Effects of shoreline alteration and habitat heterogeneity on macroinvertebrate community composition across European lakes. Ecol. Ind. 2019, 98, 285–296. [Google Scholar] [CrossRef]
- Medupin, C. Spatial and temporal variation of benthic macroinvertebrate communities along an urban river in Greater Manchester, UK. Environ. Monit. Assess. 2020, 192, 84. [Google Scholar] [CrossRef] [PubMed]
- Humphries, P. Aquatic macrophytes, macroinvertebrate associations and levels in a lowland Tasmanian river. Hydrobiologia 1996, 321, 219–233. [Google Scholar] [CrossRef]
- Law, A.; Baker, A.; Sayer, C.; Foster, G.; Gunn, I.D.M.; Taylor, P.; Pattison, Z.; Blaikie, J.; Willby, N.J. The effectiveness of aquatic plants as surrogates for wider biodiversity in standing fresh waters. Fresh. Biol. 2019, 64, 1664–1675. [Google Scholar] [CrossRef]
- Heino, J.; Ilmonen, J.; Paasivirta, L. Continuous variation of macroinvertebrate communities along environmental gradients in northern streams. BER 2014, 19, 21–38. [Google Scholar]
- Bartošová, M.; Schenková, J.; Polášková, V.; Bojková, J.; Šorfová, V.; Horsák, M. Macroinvertebrate assemblages of the post-mining calcareous stream habitats: Are they similar to those inhabiting the natural calcareous springs? Ecol. Eng. 2019, 136, 38–45. [Google Scholar] [CrossRef]
- Moncayo-Estrada, R.; Escalera-Gallardo, C.; Arroyo-Damián, M.; Campos-Campos, O.; Silva-García, J.T. Nitrogen nutrients in a subtropical river: Temporal variation and analysis at different spatial scales. Mar. Freshw. Res. 2017, 68, 1935–1949. [Google Scholar] [CrossRef]

| Sites | Human Settlements | Agriculture | Grassland | Semideciduous Forest |
|---|---|---|---|---|
| SR | 0 | 0 | 2.19 | 0 |
| S1 | 0 | 0 | 2.19 | 0 |
| S2 | 0 | 0 | 218.53 | 0 |
| S3 | 0 | 0 | 999.74 | 0 |
| S4 | 0 | 0 | 639.02 | 218.68 |
| S5 | 0 | 0 | 639.02 | 218.68 |
| S6 | 0 | 0 | 81.22 | 487.37 |
| S7 | 0 | 0 | 210.17 | 639.19 |
| S8 | 0 | 0 | 114.49 | 885.27 |
| S9 | 0 | 0.07 | 23.67 | 975.27 |
| S10 | 0 | 502.68 | 0 | 409.96 |
| S11 | 92.73 | 626.82 | 0 | 144.46 |
| S12 | 16.95 | 93.07 | 0 | 0.57 |
| S13 | 0 | 543.85 | 0 | 455.83 |
| S14 | 999.77 | 0 | 0 | 0 |
| S15 | 0.08 | 378.54 | 0 | 0 |
| SCR | 16.95 | 93.07 | 0 | 0.57 |
| Total | 1126.48 | 2238.1 | 4109.33 | 2930.24 |
| Sites | pH | CO3 (mg/L) | HCO3 (mg/L) | SO42− (mg/L) | PO43−TP (mg/L) | NH4 (mg/L) | NO3 (mg/L) | COD (mg/L) | BOD5 (mg/L) | Turb (NTU) | Flow (m3/s) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| S1 | 7.73 | 0.13 | 3.13 | 0.0002 | 0.12 | 4.71 | 0 | 12.33 | 2.12 | 19 | 0.27 |
| S2 | 7.98 | 0 | 4.20 | 0.011 | 1.23 | 1.17 | 0.04 | 15.67 | 2.7 | 65 | 0.64 |
| S3 | 8.24 | 0 | 4.85 | 0.008 | 0.55 | 1.42 | 0.32 | 149 | 27.21 | 100 | 0.69 |
| S4 | 6.87 | 0 | 9.51 | 0.073 | 25.87 | 2.38 | 2.16 | 1065.67 | 477.19 | 50 | 0.70 |
| S5 | 7.68 | 0 | 4.35 | 0.047 | 0.18 | 1.25 | 0 | 42.33 | 369.05 | 50 | 1.19 |
| S6 | 7.18 | 0 | 3.62 | 0.006 | 3.65 | 11.57 | 0.10 | 12.33 | 188.54 | 40 | 0.49 |
| S7 | 7.64 | 0 | 3.49 | 0.0003 | 0.00 | 2.39 | 0.10 | 159 | 27.4 | 35 | 0.69 |
| S8 | 7.65 | 0 | 3.54 | 0.002 | 3.33 | 2.04 | 0 | 185.67 | 81.9 | 13 | 0.95 |
| S9 | 7.65 | 0 | 3.56 | 0.0018 | 3.01 | 3.25 | 0.10 | 29 | 62.83 | 14 | 0.79 |
| S10 | 7.57 | 0 | 3.56 | 0.004 | 0.37 | 1.88 | 0 | 2.33 | 25.04 | 10 | 1.01 |
| S11 | 8.04 | 0.25 | 3.54 | 0.006 | 2.83 | 3.10 | 0.72 | 12.33 | 107.3 | 11 | 0.69 |
| S12 | 8.08 | 0 | 3.62 | 0.008 | 3.20 | 2.04 | 0.35 | 39 | 596.36 | 9 | 0.30 |
| S13 | 8.49 | 0.56 | 5.18 | 0.012 | 0.12 | 6.59 | 0.10 | 12.33 | 188.54 | 30 | 0.62 |
| S14 | 8.29 | 0.33 | 5.59 | 0.015 | 1.28 | 2.27 | 0.01 | 19 | 165.65 | 24 | 0.51 |
| S15 | 7.71 | 0 | 9.58 | 0.102 | 22.07 | 13.96 | 0.44 | 142.33 | 667.02 | 24 | 0.64 |
| SR | 7.73 | 0.13 | 3.13 | 0.0002 | 0.12 | 4.71 | 0 | 12.33 | 2.12 | 19 | 0.27 |
| SCR | 8.04 | 0.25 | 3.54 | 0.006 | 2.83 | 3.10 | 0.72 | 12.33 | 107.3 | 11 | 6.97 |
| Sites | NSF-WQI | Significance | HQI | Significance | EPT% | BMWP | Significance |
|---|---|---|---|---|---|---|---|
| SR | 86 | Good | 179 | Optimum | 88.5 | 269 | Excellent |
| S1 | 86 | Good | 173 | Optimum | 6.2 | 172 | Excellent |
| S2 | 64 | Medium | 163 | Suboptimum | 0 | 68 | Regular |
| S3 | 49 | Bad | 157 | Suboptimum | 0 | 70 | Regular |
| S4 | 48 | Bad | 160 | Suboptimum | 0 | 15 | Very polluted |
| S5 | 56 | Medium | 156 | Suboptimum | 0 | 54 | Polluted |
| S6 | 50 | Medium | 152 | Suboptimum | 107.1 | 139 | Excellent |
| S7 | 60 | Medium | 155 | Suboptimum | 63.9 | 88 | Regular |
| S8 | 57 | Medium | 155 | Suboptimum | 11.1 | 127 | Excellent |
| S9 | 59 | Medium | 167 | Optimum | 6.9 | 106 | Good |
| S10 | 64 | Medium | 172 | Optimum | 5.9 | 154 | Excellent |
| S11 | 59 | Medium | 163 | Suboptimum | 51.5 | 135 | Excellent |
| S12 | 58 | Medium | 152 | Suboptimum | 1.9 | 95 | Regular |
| S13 | 56 | Medium | 127 | Suboptimum | 2.3 | 69 | Regular |
| S14 | 55 | Medium | 140 | Suboptimum | 0 | 67 | Regular |
| S15 | 50 | Medium | 132 | Suboptimum | 0 | 104 | Good |
| S16 | 22 | Very bad | 54 | Marginal | 0 | 22 | Very polluted |
| SCR | 59 | Medium | 74 | Marginal | 6.8 | 71 | Regular |
| Sites | Culicidae | Chironomidae | Lumbriculidae | Baetidae | Polycentropodidae | Physidae | Asellidae |
|---|---|---|---|---|---|---|---|
| S1 | 10 | 277 | 8 | 15 | 127 | 26 | 17 |
| S2 | 20 | 18 | 15 | 49 | 29 | 16 | 94 |
| S3 | 2230 | 17 | 0 | 0 | 0 | 219 | 0 |
| S4 | 633 | 41 | 1254 | 0 | 0 | 7 | 1 |
| S5 | 786 | 3 | 5 | 0 | 0 | 36 | 0 |
| S6 | 276 | 26 | 27 | 0 | 0 | 130 | 0 |
| S7 | 1 | 59 | 0 | 220 | 1 | 7 | 56 |
| S8 | 0 | 166 | 1 | 41 | 84 | 2 | 8 |
| S9 | 3 | 231 | 20 | 45 | 38 | 7 | 26 |
| S10 | 17 | 533 | 0 | 15 | 39 | 3 | 23 |
| S11 | 6 | 310 | 6 | 30 | 28 | 6 | 41 |
| S12 | 4 | 270 | 6 | 84 | 378 | 13 | 7 |
| S13 | 4 | 41 | 0 | 14 | 1 | 0 | 3 |
| S14 | 18 | 39 | 0 | 26 | 0 | 0 | 0 |
| S15 | 48 | 48 | 0 | 196 | 0 | 0 | 0 |
| SR | 8 | 95 | 0 | 2 | 0 | 10 | 14 |
| SCR | 0 | 67 | 0 | 112 | 1 | 4 | 0 |
| S16 | 15 | 0 | 2 | 0 | 0 | 0 | 0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gudiño-Sosa, L.F.; Moncayo-Estrada, R.; Velázquez-Machuca, M.A.; Cruz-Cárdenas, G.; Ávila-Meléndez, L.A.; Pimentel-Equihua, J.L. Biotic Integrity, Water Quality, and Landscape Characteristics of a Subtropical River. Water 2023, 15, 1748. https://doi.org/10.3390/w15091748
Gudiño-Sosa LF, Moncayo-Estrada R, Velázquez-Machuca MA, Cruz-Cárdenas G, Ávila-Meléndez LA, Pimentel-Equihua JL. Biotic Integrity, Water Quality, and Landscape Characteristics of a Subtropical River. Water. 2023; 15(9):1748. https://doi.org/10.3390/w15091748
Chicago/Turabian StyleGudiño-Sosa, Luis Fernando, Rodrigo Moncayo-Estrada, Martha Alicia Velázquez-Machuca, Gustavo Cruz-Cárdenas, Luis Arturo Ávila-Meléndez, and José Luis Pimentel-Equihua. 2023. "Biotic Integrity, Water Quality, and Landscape Characteristics of a Subtropical River" Water 15, no. 9: 1748. https://doi.org/10.3390/w15091748





