Selenium Removal by Sediments and Plants at the Constructed Pariette Wetlands, Utah (USA)
Abstract
:1. Introduction
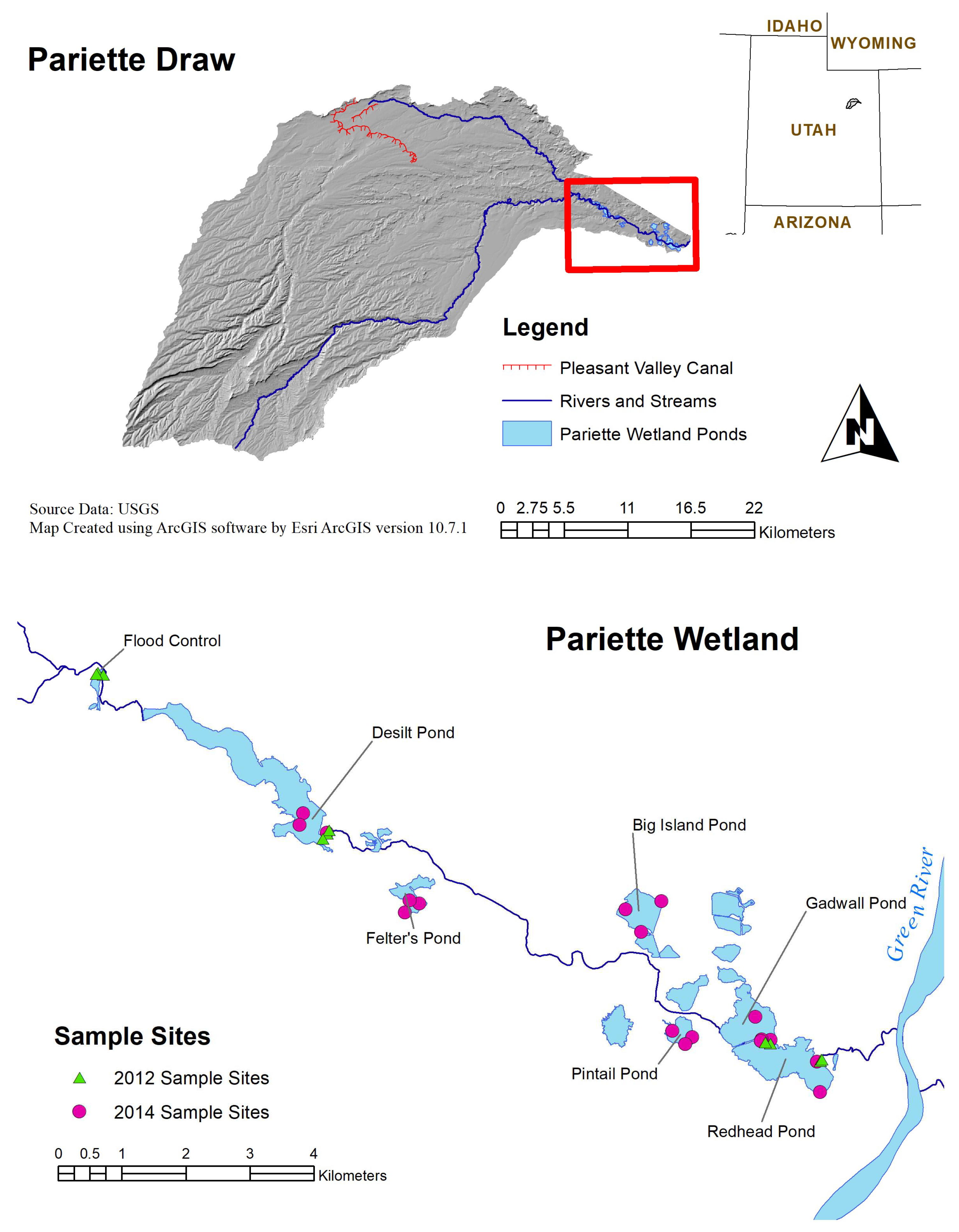
2. Materials and Methods
2.1. Description of Study Area
2.2. Field Methods
2.3. Laboratory and Statistical Analysis
3. Results
3.1. Temporal Variation of Plant Se Concentration
3.2. Spatial Distribution of Se in Water, Sediments and Plants
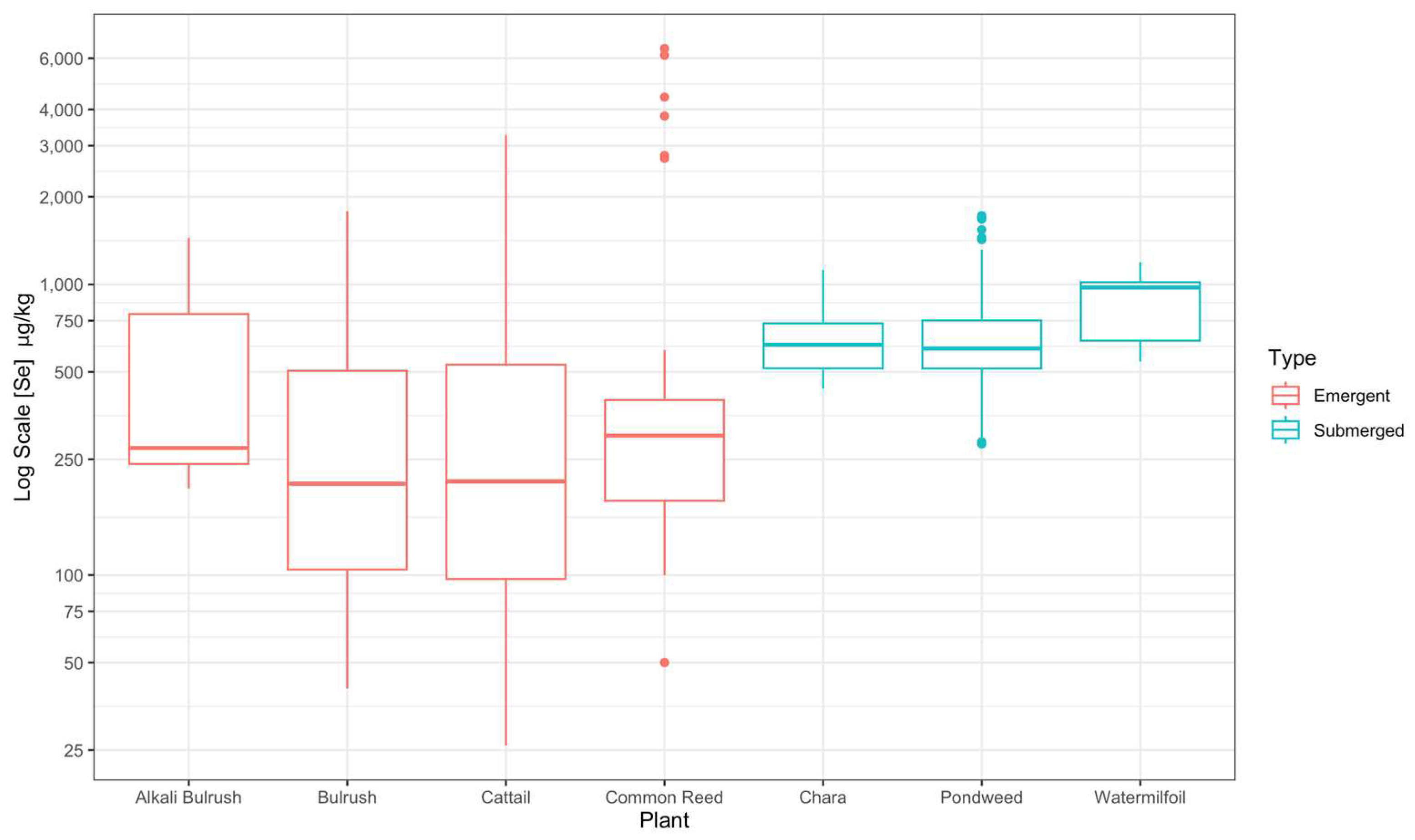
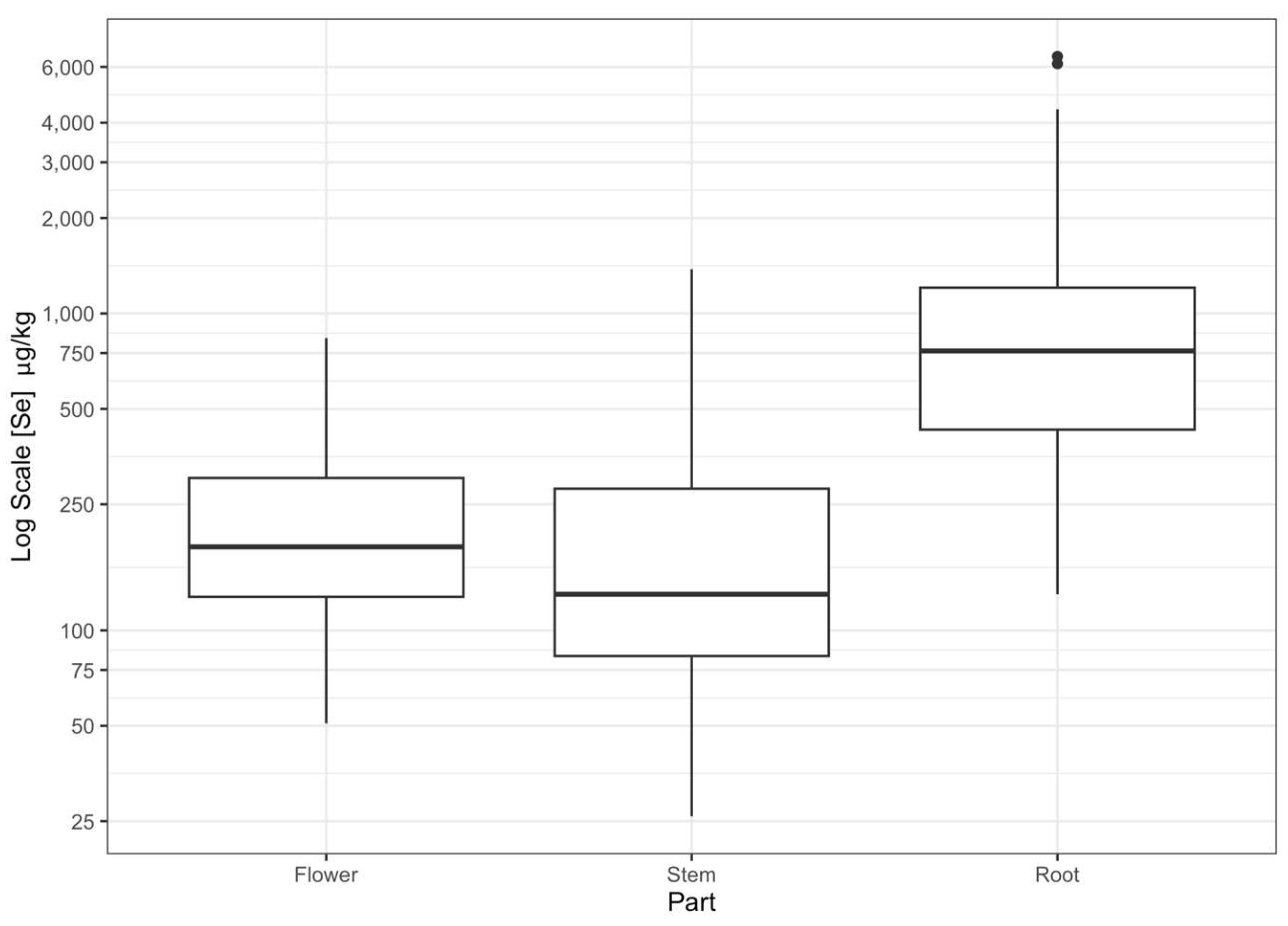
3.3. Plant Species and Part Se Concentration
4. Discussion
4.1. Mass Balance of Selenium
4.2. Selenium Uptake in Plant Tissue
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hasanuzzaman, M.; Bhuyan, M.H.M.B.; Raza, A.; Hawrylak-Nowak, B.; Matraszek-Gawron, R.; Nahar, K.; Fujita, M. Selenium Toxicity in Plants and Environment: Biogeochemistry and Remediation Possibilities. Plants 2020, 9, 1711. [Google Scholar] [CrossRef] [PubMed]
- Bodnar, M.; Konieczka, P.; Namiesnik, J. The properties, functions, and use of selenium compounds in living organisms. J. Environ. Sci. Health Part C 2012, 30, 225–252. [Google Scholar] [CrossRef] [PubMed]
- Rizwan, M.; Ali, S.; Rehman, M.Z.u.; Rinklebe, J.; Tsang, D.C.W.; Tack, F.M.G.; Abbasi, G.H.; Hussain, A.; Igalavithana, A.D.; Lee, B.C.; et al. Effects of selenium on the uptake of toxic trace elements by crop plants: A review. Crit. Rev. Environ. Sci. Technol. 2021, 51, 2531–2566. [Google Scholar] [CrossRef]
- White, P.J.; Broadley, M.R. Biofortification of crops with seven mineral elements often lacking in human diets–iron, zinc, copper, calcium, magnesium, selenium and iodine. New Phytol. 2009, 182, 49–84. [Google Scholar] [CrossRef] [PubMed]
- Hartikainen, H.; Xue, T.; Piironen, V. Selenium as an anti-oxidant and pro-oxidant in ryegrass. Plant Soil 2000, 225, 193–200. [Google Scholar] [CrossRef]
- Bellinger, F.P.; Raman, A.V.; Reeves, M.A.; Berry, M.J. Regulation and function of selenoproteins in human disease. Biochem. J. 2009, 422, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Lanza, M.G.D.B.; Reis, A.R.d. Roles of selenium in mineral plant nutrition: ROS scavenging responses against abiotic stresses. Plant Physiol. Biochem. 2021, 164, 27–43. [Google Scholar] [CrossRef]
- Gui, J.-Y.; Rao, S.; Huang, X.; Liu, X.; Cheng, S.; Xu, F. Interaction between selenium and essential micronutrient elements in plants: A systematic review. Sci. Total Environ. 2022, 853, 158673. [Google Scholar] [CrossRef]
- Gupta, M.; Gupta, S. An overview of selenium uptake, metabolism, and toxicity in plants. Front. Plant Sci. 2017, 7, 2074. [Google Scholar] [CrossRef]
- Ohlendorf, H.M.; Kilness, A.W.; Simmons, J.L.; Stroud, R.K.; Hoffman, D.J.; Moore, J.F. Selenium Toxicosis in Wild Aquatic Birds. J. Toxicol. Environ. Health 1988, 24, 67–92. [Google Scholar] [CrossRef]
- Lemly, A.D. Environmental hazard of selenium in the Animas La Plata water development project. Ecotox. Environ. Safe. 1997, 37, 92–96. [Google Scholar] [CrossRef] [PubMed]
- US EPA. Aquatic Life Ambient Water Quality Criteria for Selenium—Freshwater; US EPA: Washington, DC, USA, 2016.
- Levander, O.A.; Burk, R.F. Update of human dietary standars for selenium. In Selenium: Its Molecular Biology and Role in Human Health, 2nd ed.; Hatfield, D.L., Berry, M.J., Gladyshev, V.N., Eds.; Springer: New York, NY, USA, 2006. [Google Scholar]
- Ge, K.Y.; Yang, G.Q. The Epidemiology of Selenium Deficiency in the Etiologic Study of Endemic Diseases in China. Am. J. Clin. Nutr. 1993, 57, S259–S263. [Google Scholar] [CrossRef] [PubMed]
- Tiwary, A.K.; Stegelmeier, B.L.; Panter, K.E.; James, L.F.; Hall, J.O. Comparative toxicosis of sodium selenite and selenomethionine in lambs. J. Vet. Diagn. Investig. 2006, 18, 61–70. [Google Scholar] [CrossRef]
- Mroczek-Zdyrska, M.; Strubińska, J.; Hanaka, A. Selenium improves physiological parameters and alleviates oxidative stress in shoots of lead-exposed Vicia faba L. minor plants grown under phosphorus-deficient conditions. J. Plant Growth Regul. 2017, 36, 186–199. [Google Scholar] [CrossRef]
- Molnár, Á.; Kolbert, Z.; Kéri, K.; Feigl, G.; Ördög, A.; Szőllősi, R.; Erdei, L. Selenite-induced nitro-oxidative stress processes in Arabidopsis thaliana and Brassica juncea. Ecotox. Environ. Saf. 2018, 148, 664–674. [Google Scholar] [CrossRef] [PubMed]
- Terry, N.; Zayed, A.M.; de Souza, M.P.; Tarun, A.S. Selenium in higher plants. Annu. Rev. Plant Physiol. Plant Mol. Biol. 2000, 51, 401–432. [Google Scholar] [CrossRef]
- Seiler, R.L. Prediction of Areas Where Irrigation Drainage May Induce Selenium Contamination of Water. J. Environ. Qual. 1995, 24, 973–979. [Google Scholar] [CrossRef]
- Gao, S.; Tanji, K.K.; Dahlgren, R.A.; Ryu, J.; Herbel, M.J.; Higashi, R.M. Chemical status of selenium in evaporation basins for disposal of agricultural drainage. Chemosphere 2007, 69, 585–594. [Google Scholar] [CrossRef] [PubMed]
- Bañuelos, G.S.; Lin, Z.Q. Acceleration of selenium volatilization in seleniferous agricultural drainage sediments amended with methionine and casein. Environ. Pollut. 2007, 150, 306–312. [Google Scholar] [CrossRef]
- Ohlendorf, H.M.; Covington, S.M.; Byron, E.R.; Arenal, C.A. Conducting site-specific assessments of selenium bioaccumulation in aquatic systems. Integr. Environ. Assess. Manag. 2011, 7, 314, Erratum in Integr. Environ. Assess. Manag. 2012, 8, 210. [Google Scholar] [CrossRef]
- Zhang, Y.Q.; Zahir, Z.A.; Frankenberger, W.T. Fate of colloidal-particulate elemental selenium in aquatic systems. J. Environ. Qual. 2004, 33, 559–564. [Google Scholar] [CrossRef] [PubMed]
- Ohlendorf, H.M.; Hoffman, D.J.; Saiki, M.K.; Aldrich, T.W. Embryonic mortality and abnormalities of aquatic birds: Apparent impacts of selenium from irrigation drainwater. Sci. Total Environ. 1986, 52, 49–63. [Google Scholar] [CrossRef]
- Presser, T.S. The Kesterson effect. Environ. Manag. 1994, 18, 437–454. [Google Scholar] [CrossRef]
- Stephens, D.W.; Waddell, B.; Peltz, L.A.; Miller, J.B. Detailed Study of Selenium and Selected Elements in Water, Bottom Sediment, and Biota Associated with Irrigation Drainage in the Middle Green River Basin, Utah 1988–90; U.S. Geological Survey: Salt Lake City, UT, USA, 1992.
- Williams, D. Pariette Wetland Manager of the Bureau of Land Management Green River District—Vernal Field Office. 2011. Available online: https://www.blm.gov/press-release/bureau-land-management-conduct-pariette-wetlands-prescribed-fire (accessed on 12 December 2022).
- Jones, C.P.; Grossl, P.R.; Amacher, M.C.; Boettinger, J.L.; Jacobson, A.R.; Lawley, J.R. Selenium and salt mobilization in wetland and arid upland soils of Pariette Draw, Utah (USA). Geoderma 2017, 305, 363–373. [Google Scholar] [CrossRef]
- Pilon-Smits, E.A.H. Selenium in Plants. In Progress in Botany: Vol. 76; Lüttge, U., Beyschlag, W., Eds.; Springer International Publishing: Cham, Switzerland, 2015; pp. 93–107. [Google Scholar]
- Wingert, S.; Adams, C. TMDLs for Total Dissolved Solids, Selenium, and Boron in the Pariette Draw Watershed; US EPA: Washington, DC, USA, 2011.
- Jones, C.P.; Amacher, M.C.; Grossl, P.R.; Jacobson, A.R. Selenium mass balance and flux in water of Pariette Wetlands, Utah (USA). Appl. Geochem. 2020, 113, 104517. [Google Scholar] [CrossRef]
- Jones, C.P.; Isanhart, J.P.; Jackson, A.K. Hazard Assessment of Born, Mercury, and Selenium in Biota from Pairette Wetland Complex, Utah; Final Report for Utah Department of Water Quality; Utah State University: Logan, UT, USA, 2015; p. 128. [Google Scholar]
- Zalunardo, D. Myton Habitat Management Plan—Diamond Mountain Resource Area Vernal District; Diamond Mountain Resource Area Resource Management Plan and Environmental Impact Statement; United States Bureau of Land Management: Washington, DC, USA, 1979.
- Wilde, F.D. Water-Quality Sampling by the US Geological Survey: Standard Protocols and Procedures; U.S. Geological Survey: Salt Lake City, UT, USA, 2010; Volume 312.
- Cutter, G.A. Speciation of Selenium and Arsenic in Natural Waters and Sediments; EPRI EA-4641; Electric Power Research Institute: Palo Alto, CA, USA, 1986. [Google Scholar]
- US Environmental Protection Agency (USEPA). Method 7741A—Selenium (Atomic Absorption, Gaseous Hydride). Available online: https://www.epa.gov/sites/production/files/2015-12/documents/7741a.pdf (accessed on 12 December 2022).
- Pilon-Smits, E.A.H.; de Souza, M.P.; Hong, G.; Amini, A.; Bravo, R.C.; Payabyab, S.T.; Terry, N. Selenium volatilization and accumulation by twenty aquatic plant species. J. Environ. Qual. 1999, 28, 1011–1018. [Google Scholar] [CrossRef]
- Zasoski, R.J.; Burau, R.G. Rapid nitric-perchloric acid digestion method for multi-element tissue analysis. Commun. Soil Sci. Plant Anal. 1977, 8, 425–436. [Google Scholar] [CrossRef]
- Fellows, I. Deducer: A data analysis GUI for R. J. Stat. Softw. 2012, 49, 1–15. [Google Scholar] [CrossRef]
- Authority, S.N.W.; Acharya, K.; Adhikari, A. A Comparison of Water Quality Improvements from Three Different Wetland Types in the Las Vegas Valley Watershed. 2010. Available online: https://www.lvwash.org/assets/pdf/resources-wqresearch-wetlands.pdf (accessed on 12 December 2022).
- Azaizeh, H.; Salhani, N.; Sebesvari, Z.; Shardendu, S.; Emons, H. Phytoremediation of selenium using subsurface-flow constructed wetland. Int. J. Phytorem. 2006, 8, 187–198. [Google Scholar] [CrossRef]
- Dicataldo, G.; Hayes, D.F.; Miller, T.G.; Scanlan, L. Selenium speciation and distribution in a wetland system of the Great Salt Lake, Utah. Environ. Eng. Sci. 2010, 27, 777–788. [Google Scholar] [CrossRef]
- Hansen, D.; Duda, P.J.; Zayed, A.; Terry, N. Selenium removal by constructed wetlands: Role of biological volatilization. Environ. Sci. Technol. 1998, 32, 591–597. [Google Scholar] [CrossRef]
- Karlson, U.; Parker; Frankenberger, W.T.; Calderone, S.J. Influence of temperature and organic amendments on the mobilization of selenium in sediments. Soil Biol. Biochem. 1990, 22, 615–620. [Google Scholar]
- Garcia-Hernandez, J.; Glenn, E.P.; Artiola, J.; Baumgartner, D.J. Bioaccumulation of selenium (Se) in the Cienega de Santa Clara Wetland, Sonora, Mexico. Ecotoxicol. Environ. Saf. 2000, 46, 298–304. [Google Scholar] [CrossRef]
- Pollard, J.; Cizdziel, J.; Stave, K.; Reid, M. Selenium concentrations in water and plant tissues of a newly formed arid wetland in Las Vegas, Nevada. Environ. Monit. Assess. 2007, 135, 447–457. [Google Scholar] [CrossRef] [PubMed]
- White, P.J. Selenium metabolism in plants. Biochim. Biophys. Acta BBA-Gen. Subj. 2018, 1862, 2333–2342. [Google Scholar] [CrossRef]
- Zayed, A.; Lytle, C.M.; Terry, N. Accumulation and volatilization of different chemical species of selenium by plants. Planta 1998, 206, 284–292. [Google Scholar] [CrossRef]
- Feng, R.; Wang, L.; Yang, J.; Zhao, P.; Zhu, Y.; Li, Y.; Yu, Y.; Liu, H.; Rensing, C.; Wu, Z. Underlying mechanisms responsible for restriction of uptake and translocation of heavy metals (metalloids) by selenium via root application in plants. J. Hazard. Mater. 2021, 402, 123570. [Google Scholar] [CrossRef] [PubMed]
- Bitterli, C.; Bañuelos, G.S.; Schulin, R. Use of transfer factors to characterize uptake of selenium by plants. J. Geochem. Explor. 2010, 107, 206–216. [Google Scholar] [CrossRef]
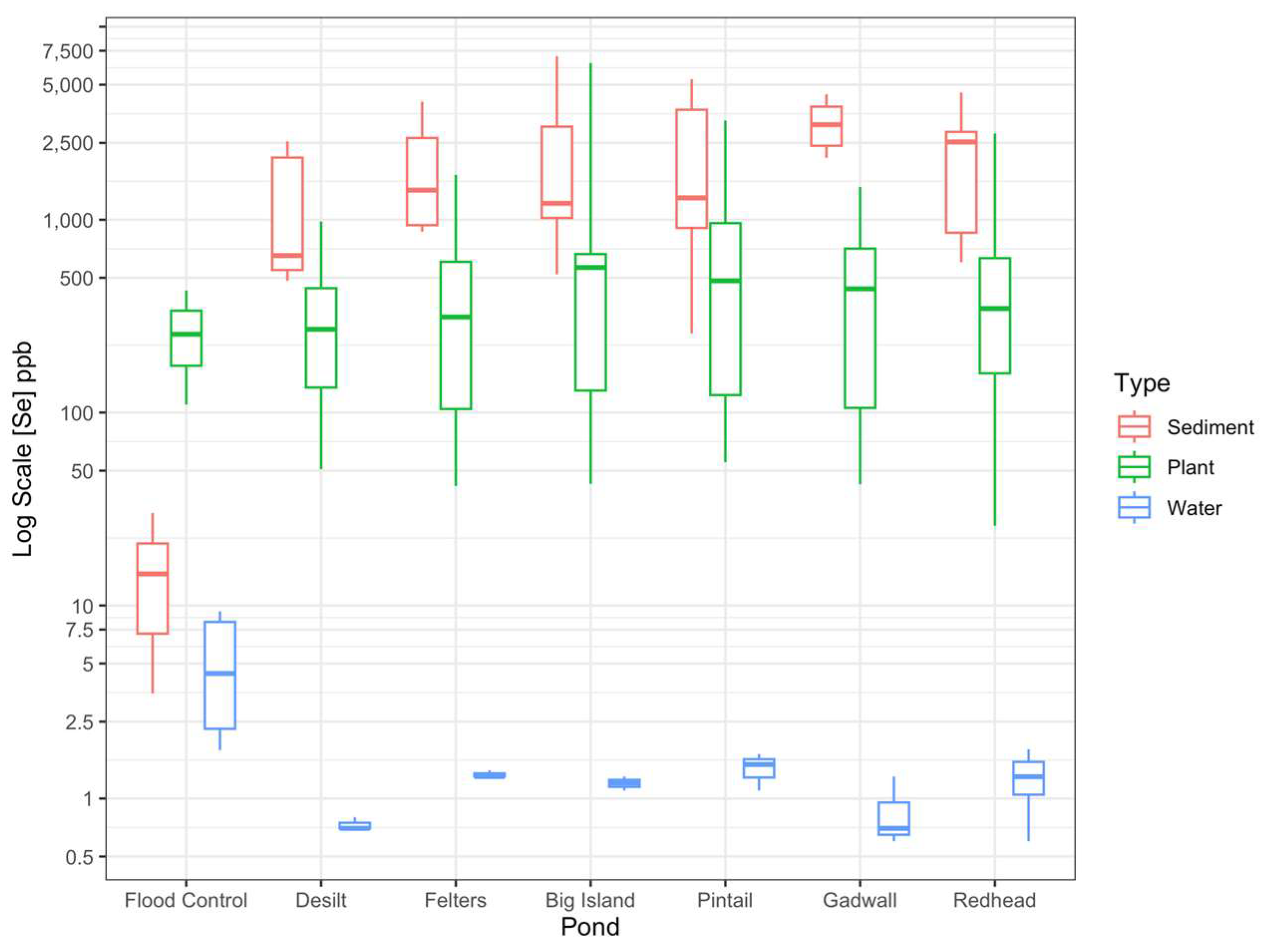
| Summary Statistics | n | Mean ± Std Err μg L−1 or μg kg−1 | Min μg L−1 or μg kg−1 | Max μg L−1 or μg kg−1 |
|---|---|---|---|---|
| Water | ||||
| Flood Control | 4 | 5.4 ± 1.9 | 1.8 | 9.3 |
| Desilt | 3 | 0.7 ± 0.03 | 0.7 | 0.8 |
| Felters | 3 | 1.3 ± 0.03 | 1.3 | 1.4 |
| Big Island | 3 | 1.2 ± 0.06 | 1.1 | 1.3 |
| Pintail | 3 | 1.4 ± 0.2 | 1.1 | 1.7 |
| Gadwall | 3 | 0.9 ± 0.2 | 0.6 | 1.3 |
| Redhead | 3 | 1.0 ± 0.2 | 0.6 | 1.4 |
| Sediment | ||||
| Flood Control | 3 | 16.1 ± 7.8 | 3.5 | 30.2 |
| Desilt | 9 | 1186 ± 786 | 482 | 7031 |
| Felters | 9 | 1881 ± 399 | 868 | 4090 |
| Big Island | 9 | 2206 ± 786 | 132 | 7031 |
| Pintail | 9 | 2070 ± 609 | 257 | 5344 |
| Gadwall | 9 | 3154 ± 283 | 2092 | 4458 |
| Redhead | 9 | 2252 ± 436 | 602 | 4559 |
| Plant * | ||||
| Flood Control | 24 | 259 ± 21 | 110 | 430 |
| Desilt | 67 | 319 ± 26 | 50 | 980 |
| Felters | 45 | 439 ± 57 | 41 | 1709 |
| Big Island | 57 | 906 ± 180 | 43 | 6476 |
| Pintail | 45 | 680 ± 101 | 55 | 3262 |
| Gadwall | 65 | 449 ± 47 | 0.6 | 1479 |
| Redhead | 100 | 502 ± 49 | 26 | 2800 |
| Species | Root (μg kg−1) | Stem (μg kg−1) | Flower (μg kg−1) | Whole Plant (μg kg−1) |
|---|---|---|---|---|
| Emergent Vegetation | ||||
| S. maritimus | 1188 ± 128 | 278 ± 25 | 235 ± 20 | 534 ± 147 * |
| S. acutus | 743 ± 72 | 227 ± 35 | 152 ± 15 | 368 ± 37 * |
| Typha | 897 ± 97 | 196.0 ± 30.0 | 207 ± 22 | 420 ± 43 * |
| P. australis | 2755 ± 768 | 240 ± 28 | 376 ± 42 | 832 ± 226 * |
| Submerged Vegetation | ||||
| Chara spp. | 929 ± 194 | 656 ± 173 | 675 ± 162 | 637 ± 26 |
| P. filiformis | NA | NA | NA | 775 ± 68 |
| M. spicatum | NA | NA | NA | 874 ± 122 |
| Variable | Chevron Refinery Marsh, California [43] | Cienega de Santa Clara Wetland, Mexico [45] | Clark County Wetlands Park, Nevada [46] | Pariette Wetlands, Utah [31] |
|---|---|---|---|---|
| Mean annual temp, deg C | 14.4 | 14.4 | 20.8 | 8.2 |
| Mean annual Precip, mm | 274 | 63 | 106 | 178 |
| Area, ha | 36 | 4200 | 53 | 1023 |
| Principal plant taxa | Scirpus spp. Scirpus fornicus Distichlis spicata Grindelia stricta | Typha spp. Phragmites spp. Distichlis palmeri | Scirpus spp. Typha spp. Najas spp. Chara spp. | Scirpus spp. Typha spp. Phragmites spp. Chara spp. |
| Plant Se Concentration mg kg−1 | 5 to 20 | 0.03 to 0.17 | 0.3 to 2.81 | 0.03 to 6.5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jones, C.; Amacher, M.; Grossl, P.; Jacobson, A. Selenium Removal by Sediments and Plants at the Constructed Pariette Wetlands, Utah (USA). Water 2023, 15, 1728. https://doi.org/10.3390/w15091728
Jones C, Amacher M, Grossl P, Jacobson A. Selenium Removal by Sediments and Plants at the Constructed Pariette Wetlands, Utah (USA). Water. 2023; 15(9):1728. https://doi.org/10.3390/w15091728
Chicago/Turabian StyleJones, Colleen, Michael Amacher, Paul Grossl, and Astrid Jacobson. 2023. "Selenium Removal by Sediments and Plants at the Constructed Pariette Wetlands, Utah (USA)" Water 15, no. 9: 1728. https://doi.org/10.3390/w15091728






