Natural Radioactivity in Drinking Water in the Surroundings of a Metamorphic Outcrop in Hungary: The Hydrogeological Answer to Practical Problems
Abstract
:1. Introduction
Methods Rationale
2. Materials and Methods
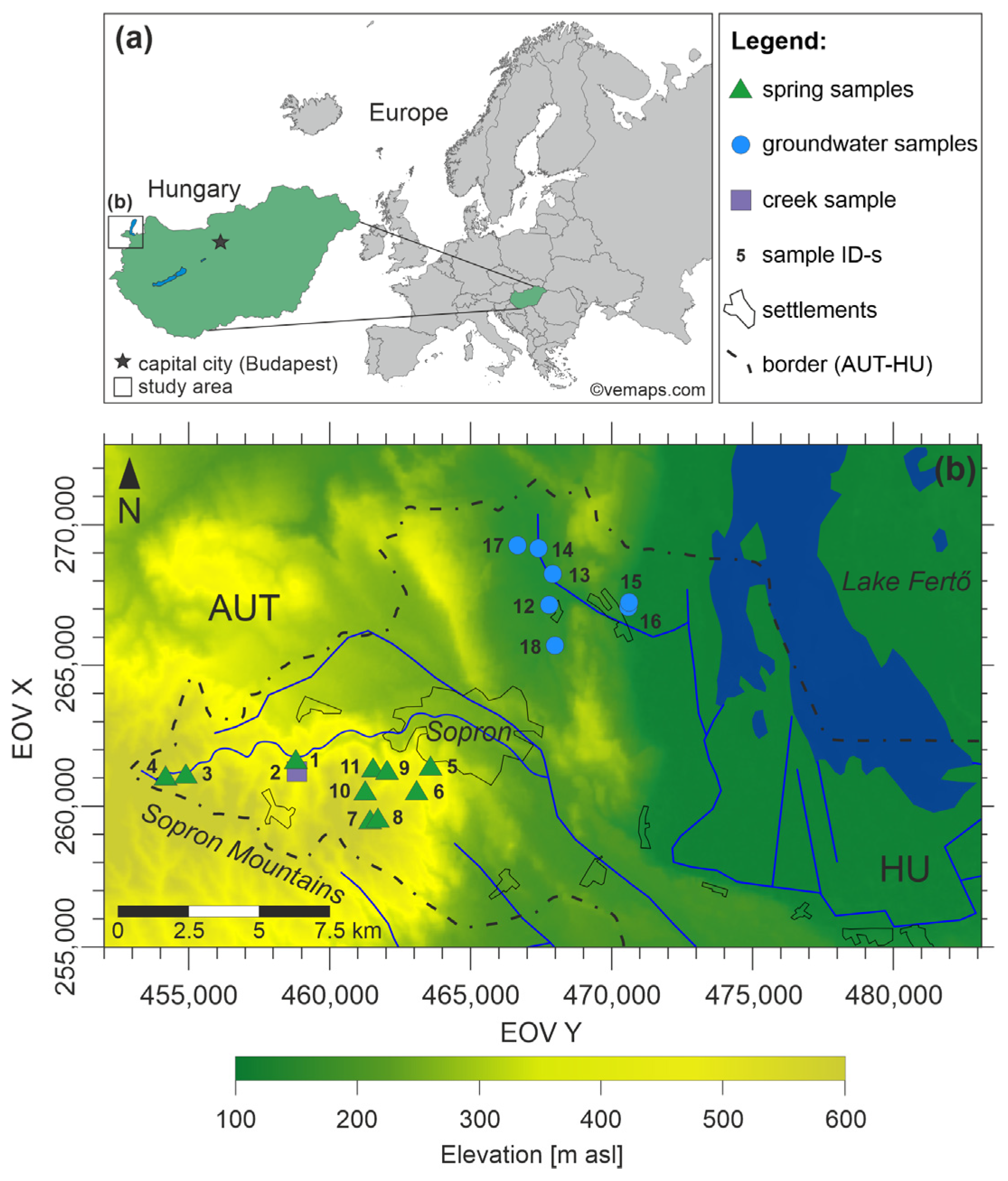
2.1. Study Area
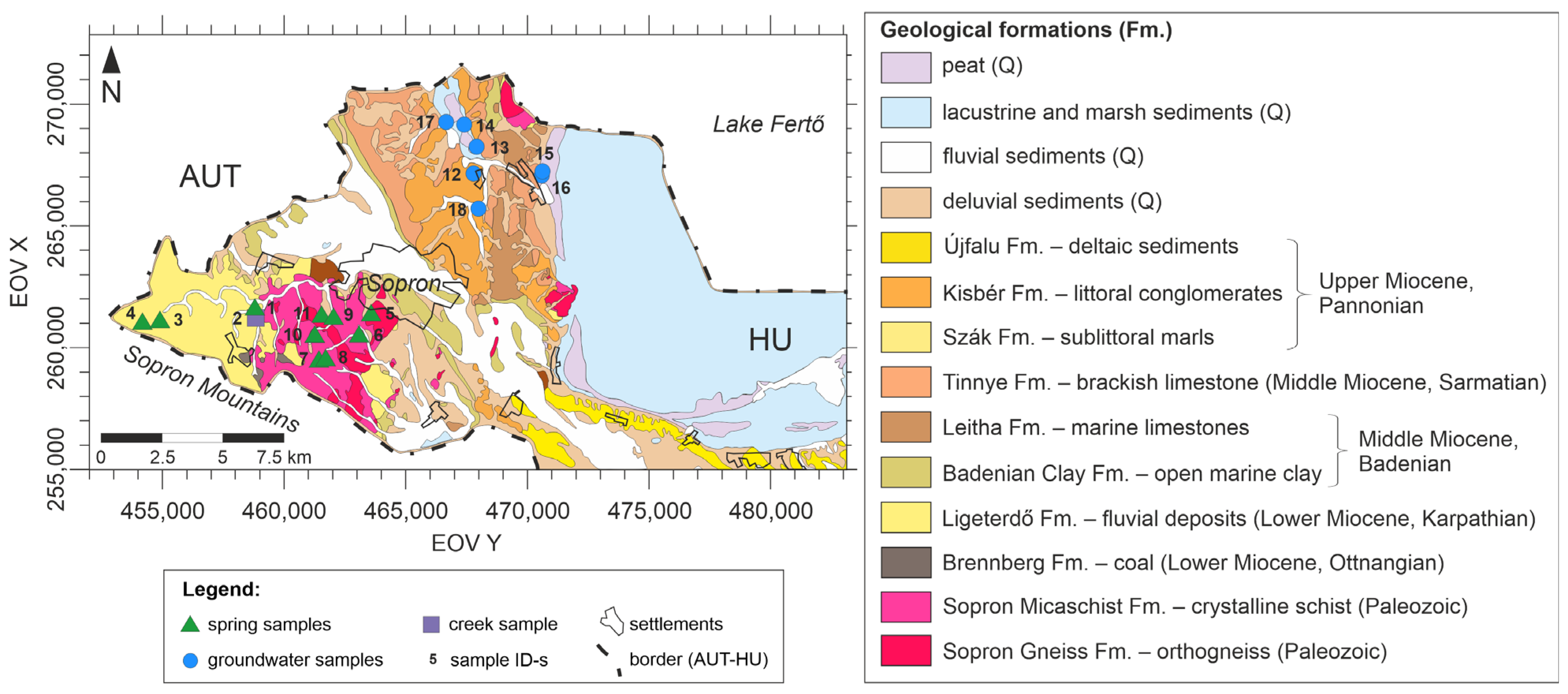
2.2. Geological Settings
2.3. Hydrogeological Characterisation
2.4. Sampling and Laboratory Measurements
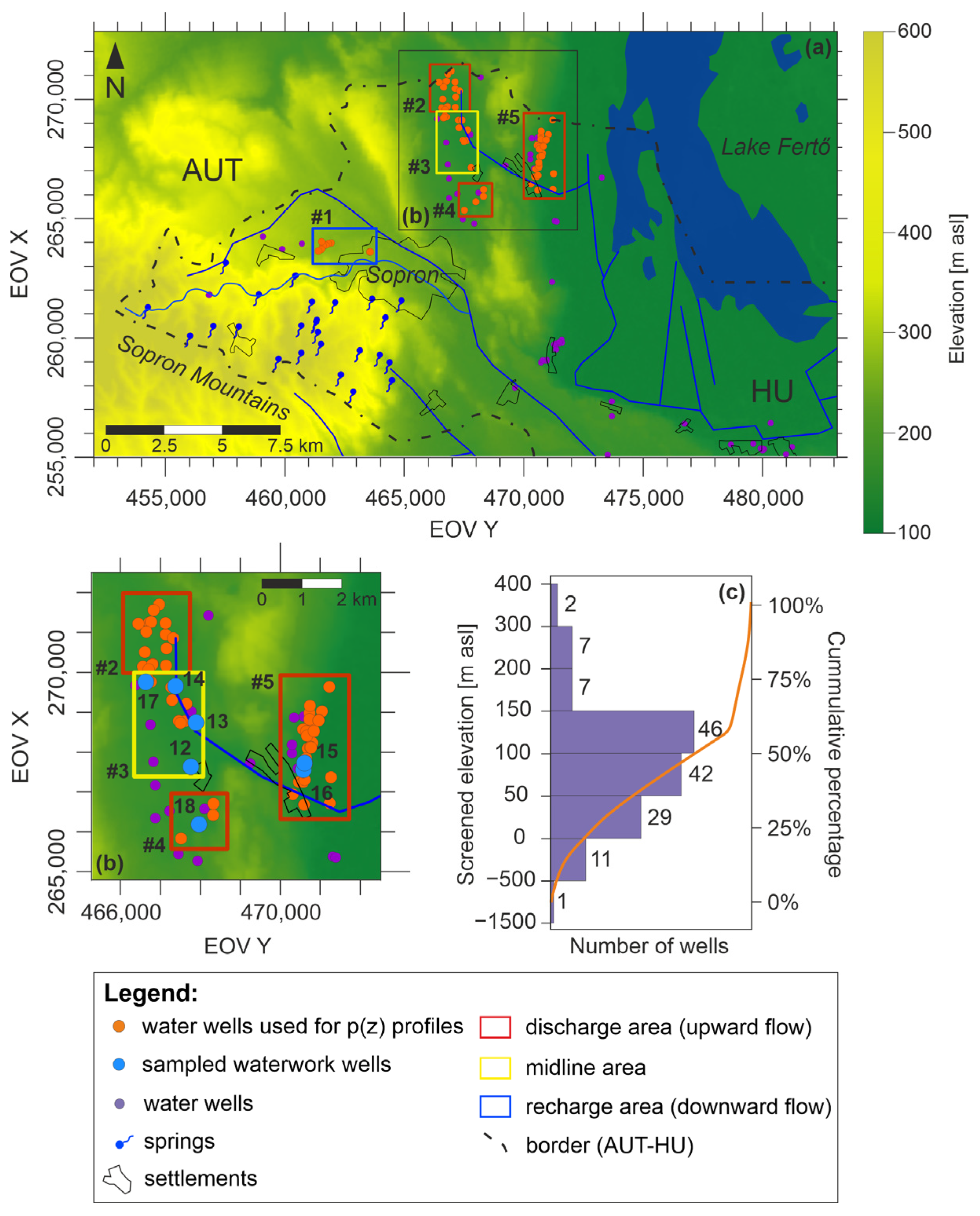
2.5. Hydraulic Evaluation
3. Results
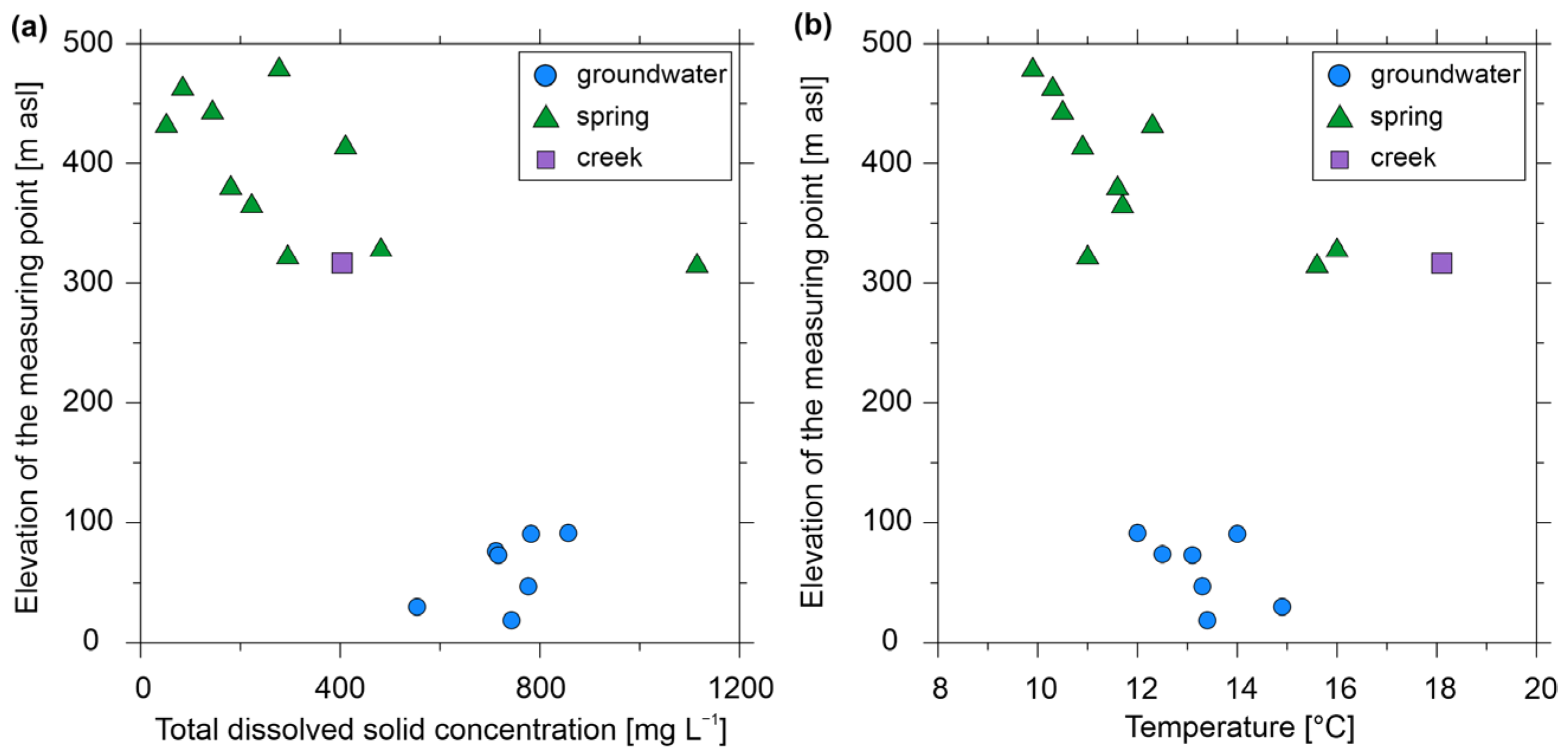
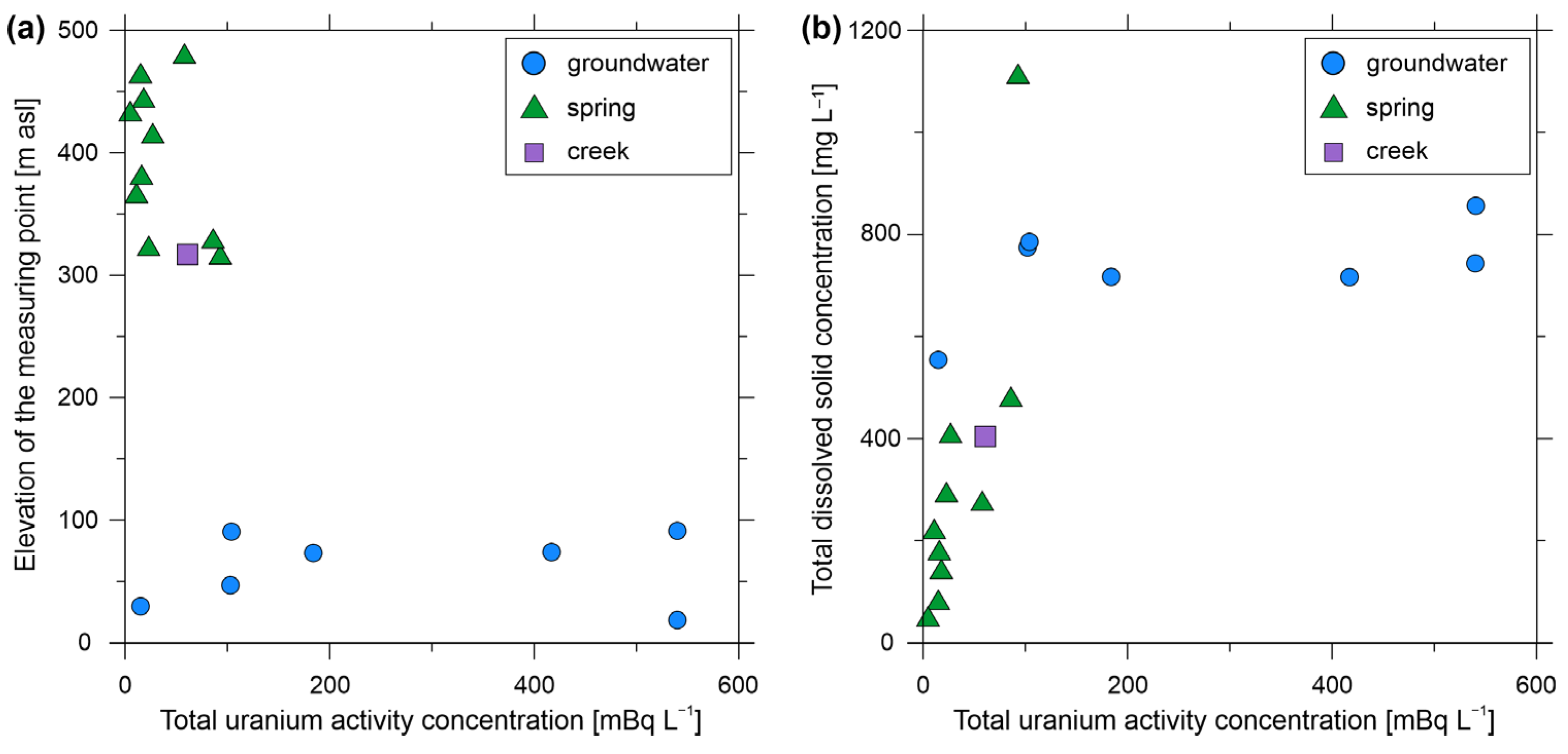
3.1. Physicochemical Properties Measured On-Site and Hydrochemistry
3.2. Radionuclide Measurements
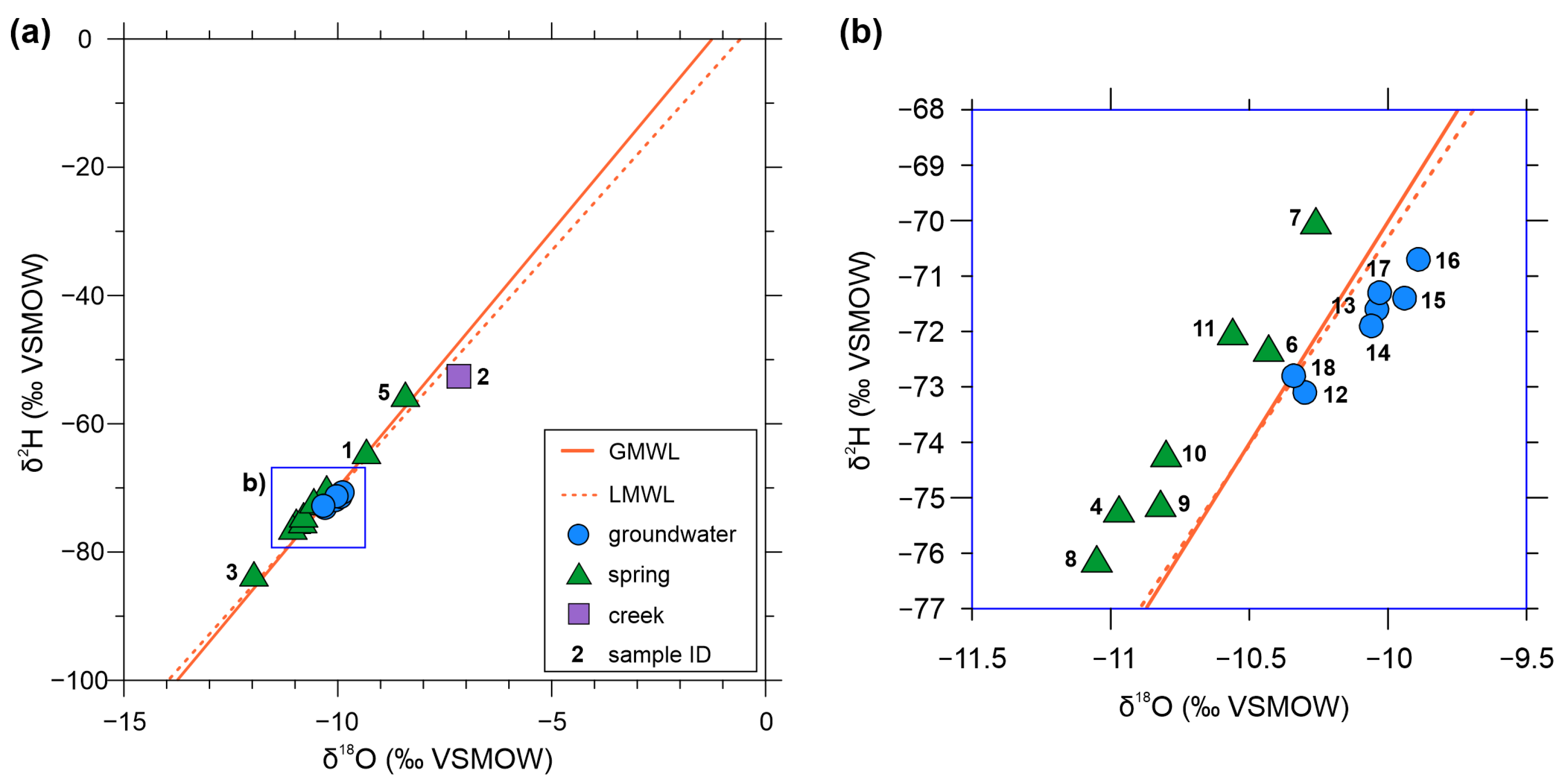
3.3. Stable Isotope Measurements
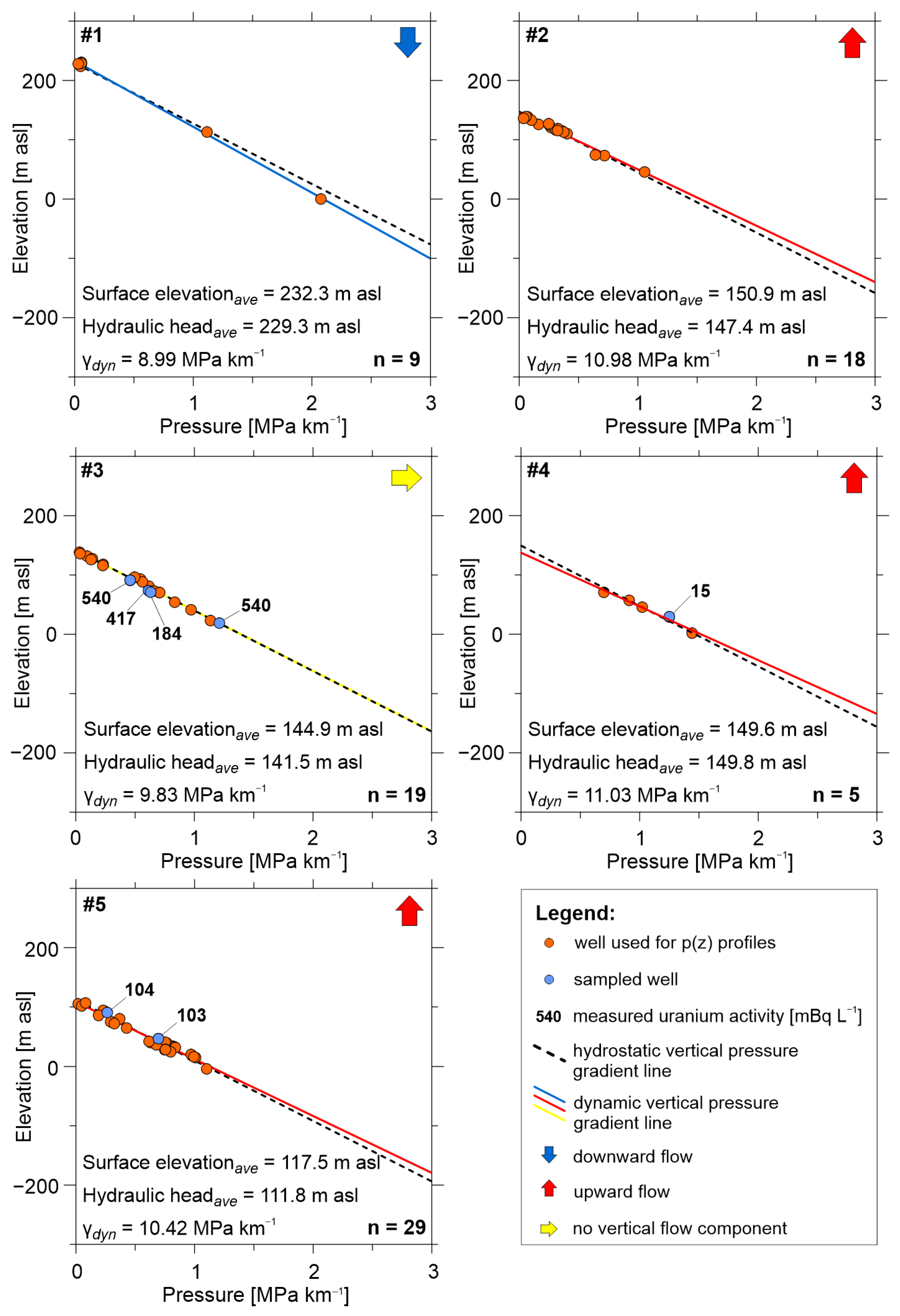
3.4. Hydraulic Evaluation
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lapworth, D.J.; Boving, T.B.; Kreamer, D.K.; Kebede, S.; Smedley, P.L. Groundwater Quality: Global Threats, Opportunities and Realising the Potential of Groundwater. Sci. Total Environ. 2022, 811, 152471. [Google Scholar] [CrossRef] [PubMed]
- Erőss, A.; Csondor, K.; Izsák, B.; Vargha, M.; Horváth, Á.; Pándics, T. Uranium in Groundwater—The Importance of Hydraulic Regime and Groundwater Flow System’s Understanding. J. Environ. Radioact. 2018, 195, 90–96. [Google Scholar] [CrossRef] [PubMed]
- Erőss, A. Natürliche Radioaktivität Im Grundwasser—Neue Parameter Und Herausforderungen Für Die Trinkwasserversorgung. Grundwasser 2020, 25, 111–112. [Google Scholar] [CrossRef]
- Edmunds, W.M.; Smedley, P.L. Groundwater Geochemistry and Health: An Overview. Geol. Soc. Lond. Spec. Publ. 1996, 113, 91–105. [Google Scholar] [CrossRef]
- Vengosh, A.; Coyte, R.M.; Podgorski, J.; Johnson, T.M. A Critical Review on the Occurrence and Distribution of the Uranium- and Thorium-Decay Nuclides and Their Effect on the Quality of Groundwater. Sci. Total Environ. 2022, 808, 151914. [Google Scholar] [CrossRef]
- Hoehn, E. Radionuclides in groundwaters: Contaminants and tracers. In Groundwater Quality: Remediation and Protection; Herbert, M., Kovar, K., Eds.; IAHS, Publication no. 250; IAHS Press: Wallingford, UK, 1998; pp. 3–9. [Google Scholar]
- Nuccetelli, C.; Rusconi, R.; Forte, M. Radioactivity in Drinking Water: Regulations, Monitoring Results and Radiation Protection Issues. Ann. Dell’istituto Super. Sanità 2012, 48, 362–373. [Google Scholar] [CrossRef]
- Banning, A.; Benfer, M. Drinking Water Uranium and Potential Health Effects in the German Federal State of Bavaria. Int. J. Environ. Res. Public Health 2017, 14, 927. [Google Scholar] [CrossRef]
- Skeppström, K.; Olofsson, B. Uranium and Radon in Groundwater—An Overview of the Problem. Eur. Water 2007, 17/18, 51–62. [Google Scholar]
- Tóth, J. Gravitational Systems of Groundwater Flow: Theory, Evaluation, Utilization; Cambridge University Press: Cambridge, UK, 2009. [Google Scholar]
- Finch, W.I.; Davis, J.F. Sandstone-Type Uranium Deposits; IAEA-TECDOC-328; International Atomic Energy Agency: Vienna, Austria, 1985. [Google Scholar]
- Grenthe, I.; Drożdżynński, J.; Fujino, T.; Buck, E.C.; Albrecht-Schmitt, T.E.; Wolf, S.F. Uranium. In The Chemistry of the Actinide and Transactinide Elements; Springer Netherlands: Dordrecht, The Netherlands, 2018; pp. 253–698. [Google Scholar]
- Cuthbert, M.O.; Gleeson, T.; Moosdorf, N.; Befus, K.M.; Schneider, A.; Hartmann, J.; Lehner, B. Global Patterns and Dynamics of Climate–Groundwater Interactions. Nat. Clim. Chang. 2019, 9, 137–141. [Google Scholar] [CrossRef]
- COUNCIL DIRECTIVE 2013/51/EURATOM. Available online: https://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2013:296:0012:0021:EN:PDF (accessed on 14 March 2023).
- 5/2023 Government Decree (in Hungarian: 5/2023 (I. 12) Korm. Rendelet Az Ivóvíz Minőségi Követelményeiről És Az Ellenőrzés Rendjéről). Available online: https://net.jogtar.hu/jogszabaly?docid=A2300005.KOR&searchUrl=/gyorskereso?keyword%3D5/2023 (accessed on 19 April 2023).
- Baják, P.; Hegedűs-Csondor, K.; Tiljander, M.; Korkka-Niemi, K.; Surbeck, H.; Izsák, B.; Vargha, M.; Horváth, Á.; Pándics, T.; Erőss, A. Integration of a Shallow Soda Lake into the Groundwater Flow System by Using Hydraulic Evaluation and Environmental Tracers. Water 2022, 14, 951. [Google Scholar] [CrossRef]
- Csondor, K.; Baják, P.; Surbeck, H.; Izsák, B.; Horváth, Á.; Vargha, M.; Erőss, A. Transient Nature of Riverbank Filtered Drinking Water Supply Systems—A New Challenge of Natural Radioactivity Assessment. J. Environ. Radioact. 2020, 211, 106072. [Google Scholar] [CrossRef]
- Ayotte, J.D.; Szabo, Z.; Focazio, M.J.; Eberts, S.M. Effects of Human-Induced Alteration of Groundwater Flow on Concentrations of Naturally-Occurring Trace Elements at Water-Supply Wells. Appl. Geochem. 2011, 26, 747–762. [Google Scholar] [CrossRef]
- Gómez, M.; Suursoo, S.; Martin-Sanchez, N.; Vaasma, T.; Leier, M. Natural Radioactivity in European Drinking Water: A Review. Crit. Rev. Environ. Sci. Technol. 2023, 53, 198–215. [Google Scholar] [CrossRef]
- Kovács-Bodor, P.; Csondor, K.; Erőss, A.; Szieberth, D.; Freiler-Nagy, Á.; Horváth, Á.; Bihari, Á.; Mádl-Szőnyi, J. Natural Radioactivity of Thermal Springs and Related Precipitates in Gellért Hill Area, Buda Thermal Karst, Hungary. J. Environ. Radioact. 2019, 201, 32–42. [Google Scholar] [CrossRef]
- Eisenlohr, L.; Surbeck, H. Radon as a Natural Tracer to Study Transport Processes in a Karst System. An Example in the Swiss Jura. Comptes Rendus L’academie Sci. Ser. 2 Sci. Terre Planetes 1995, 321, 761–767. [Google Scholar]
- Erőss, A.; Mádl-Szőnyi, J.; Surbeck, H.; Horváth, Á.; Goldscheider, N.; Csoma, A.É. Radionuclides as Natural Tracers for the Characterization of Fluids in Regional Discharge Areas, Buda Thermal Karst, Hungary. J. Hydrol. 2012, 426–427, 124–137. [Google Scholar] [CrossRef]
- Gainon, F.; Surbeck, H.; Zwahlen, F. Natural Radionuclides in Groundwater as Pollutants and as Useful Tracers. In Proceedings of the 12th Symposium on Water Rock Interaction, Kunming, China, 31 July–5 August 2007. [Google Scholar]
- Gainon, F.; Goldscheider, N.; Surbeck, H. Conceptual Model for the Origin of High Radon Levels in Spring Waters—The Example of the St. Placidus Spring, Grisons, Swiss Alps. Swiss J. Geosci. 2007, 100, 251–262. [Google Scholar] [CrossRef]
- Gainon, F. Les Isotopes Radioactifs de LA Série de l’uranium-238 (222Rn, 226Ra, 234U et 238U) Dans Les Eaux Thermales de Suisse: Sites d’Yverdon-Les-Bains, Moiry, Loèche-Les-Bains, Saxon, Val d’Illiez, Bad Ragaz, Delémont, Lavey-Les-Bains, Brigerbad et Combioula. Ph.D. Thesis, Université de Neuchâtel, Neuchâtel, Switzerland, 2008. [Google Scholar]
- Swarzenski, P.W. U/Th Series Radionuclides as Coastal Groundwater Tracers. Chem. Rev. 2007, 107, 663–674. [Google Scholar] [CrossRef]
- Smith, C.G.; Swarzenski, P.W.; Dimova, N.T.; Zhang, J. Natural radium and radon tracers to quantify water exchange and movement in reservoirs. In Handbook of Environmental Isotope Geochemistry: Volume I; Springer: Heidelberg, Germany, 2012; pp. 345–365. [Google Scholar] [CrossRef]
- Bourdon, B.; Turner, S.; Henderson, G.M.; Lundstrom, C.C. Introduction to U-Series Geochemistry. Rev. Miner. Geochem. 2003, 52, 1–21. [Google Scholar] [CrossRef]
- Curtis, G.P.; Davis, J.A.; Naftz, D.L. Simulation of Reactive Transport of Uranium(VI) in Groundwater with Variable Chemical Conditions. Water Resour. Res. 2006, 42, W04404. [Google Scholar] [CrossRef]
- Grundl, T.; Cape, M. Geochemical Factors Controlling Radium Activity in a Sandstone Aquifer. Ground Water 2006, 44, 518–527. [Google Scholar] [CrossRef] [PubMed]
- Cherdynt︠s︡ev, V.V.; Viktor, V.; Ivanovich, M.; Harmon, R.S.; Russell, S. Uranium Series Disequilibrium: Applications to Environmental Problems; Clarendon Press: Oxford, UK, 1982; ISBN 0198544235. [Google Scholar]
- Burow, K.R.; Belitz, K.; Dubrovsky, N.M.; Jurgens, B.C. Large Decadal-Scale Changes in Uranium and Bicarbonate in Groundwater of the Irrigated Western U.S. Sci. Total Environ. 2017, 586, 87–95. [Google Scholar] [CrossRef]
- Coyte, R.M.; Jain, R.C.; Srivastava, S.K.; Sharma, K.C.; Khalil, A.; Ma, L.; Vengosh, A. Large-Scale Uranium Contamination of Groundwater Resources in India. Environ. Sci. Technol. Lett. 2018, 5, 341–347. [Google Scholar] [CrossRef]
- Alam, M.d.S.; Cheng, T. Uranium Release from Sediment to Groundwater: Influence of Water Chemistry and Insights into Release Mechanisms. J. Contam. Hydrol. 2014, 164, 72–87. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, F.; Chambers, D.; Fernandes, S.; Fesenko, S.; Goulet, R.; Howard, B.; Kim, C.-K.; Martin, P.; Moore, W.S.; Phaneuf, M.; et al. The Environmental Behaviour of Radium: Revised Edition; International Atomic Energy Agency: Vienna, Austria, 2014. [Google Scholar]
- Chałupnik, S.; Wysocka, M.; Janson, E.; Chmielewska, I.; Wiesner, M. Long Term Changes in the Concentration of Radium in Discharge Waters of Coal Mines and Upper Silesian Rivers. J. Environ. Radioact. 2017, 171, 117–123. [Google Scholar] [CrossRef]
- McMahon, P.B.; Vengosh, A.; Davis, T.A.; Landon, M.K.; Tyne, R.L.; Wright, M.T.; Kulongoski, J.T.; Hunt, A.G.; Barry, P.H.; Kondash, A.J.; et al. Occurrence and Sources of Radium in Groundwater Associated with Oil Fields in the Southern San Joaquin Valley, California. Environ. Sci. Technol. 2019, 53, 9398–9406. [Google Scholar] [CrossRef]
- Miklyaev, P.S.; Petrova, T.B.; Shchitov, D.V.; Sidyakin, P.A.; Murzabekov, M.A.; Marennyy, A.M.; Nefedov, N.A.; Sapozhnikov, Y.A. The Results of Long-Term Simultaneous Measurements of Radon Exhalation Rate, Radon Concentrations in Soil Gas and Groundwater in the Fault Zone. Appl. Radiat. Isot. 2021, 167, 109460. [Google Scholar] [CrossRef]
- Sukanya, S.; Noble, J.; Joseph, S. Factors Controlling the Distribution of Radon (222Rn) in Groundwater of a Tropical Mountainous River Basin in Southwest India. Chemosphere 2021, 263, 128096. [Google Scholar] [CrossRef]
- Kovács-Bodor, P.; Anda, D.; Jurecska, L.; Óvári, M.; Horváth, Á.; Makk, J.; Post, V.; Müller, I.; Mádl-Szőnyi, J. Integration of In Situ Experiments and Numerical Simulations to Reveal the Physicochemical Circumstances of Organic and Inorganic Precipitation at a Thermal Spring. Aquat. Geochem. 2018, 24, 231–255. [Google Scholar] [CrossRef]
- Erőss, A.; Surbeck, H.; Csondor, K.; Horváth, Á.; Mádl-Szőnyi, J.; Lénárt, L. Radionuclides in the Waters of the Bükk Region, Hungary. J. Radioanal. Nucl. Chem. 2015, 303, 2529–2533. [Google Scholar] [CrossRef]
- El-Aassy, I.E.; El-Feky, M.G.; Issa, F.A.; Ibrahim, N.M.; Desouky, O.A.; Khattab, M.R. Uranium and 234U/238U Isotopic Ratios in Some Groundwater Wells at Southwestern Sinai, Egypt. J. Radioanal. Nucl. Chem. 2015, 303, 357–362. [Google Scholar] [CrossRef]
- Suksi, J.; Rasilainen, K.; Pitkänen, P. Variations in 234U/238U Activity Ratios in Groundwater—A Key to Flow System Characterisation? Phys. Chem. Earth Parts A/B/C 2006, 31, 556–571. [Google Scholar] [CrossRef]
- Grabowski, P.; Bem, H. Uranium Isotopes as a Tracer of Groundwater Transport Studies. J. Radioanal. Nucl. Chem. 2012, 292, 1043–1048. [Google Scholar] [CrossRef] [PubMed]
- Gilkeson, R.; Cowart, J.B. Radium, Radon and Uranium Isotopes in Groundwater from Cambrian-Ordovician Sandstone Aquifers in Illinois. In Radon in Ground Water; CRC Press: Boca Raton, FL, USA, 1987; pp. 403–422. [Google Scholar]
- Osmond, J.K.; Cowart, J.B. The Theory and Uses of Natural Uranium Isotopic Variations in Hydrology. Energy Rev. 1976, 14, 621–679. [Google Scholar]
- Gascoyne, M. Geochemistry of the Actinides and Their Daughters. In Uranium-Series Disequilibrium: Applications to Earth, Marine, and Environmental Sciences; Ivanovich, M., Harmon, R.S., Eds.; Clarendon Press: Oxford, UK, 1992; pp. 34–61. [Google Scholar]
- Srivastava, S.K.; Balbudhe, A.Y.; Vishwa Prasad, K.; Padma Savithri, P.; Tripathi, R.M.; Puranik, V.D. Variation in the Uranium Isotopic Ratios 234U/238U, 238U/Total-U and 234U/Total-U in Indian Soil Samples: Application to Environmental Monitoring. Radioprotection 2013, 48, 231–242. [Google Scholar] [CrossRef]
- Bouchaou, L.; Michelot, J.L.; Vengosh, A.; Hsissou, Y.; Qurtobi, M.; Gaye, C.B.; Bullen, T.D.; Zuppi, G.M. Application of Multiple Isotopic and Geochemical Tracers for Investigation of Recharge, Salinization, and Residence Time of Water in the Souss–Massa Aquifer, Southwest of Morocco. J. Hydrol. 2008, 352, 267–287. [Google Scholar] [CrossRef]
- Heilweil, V.M.; Solomon, D.K.; Gingerich, S.B.; Verstraeten, I.M. Oxygen, Hydrogen, and Helium Isotopes for Investigating Groundwater Systems of the Cape Verde Islands, West Africa. Hydrogeol. J. 2009, 17, 1157–1174. [Google Scholar] [CrossRef]
- Negrel, P.; Pauwels, H.; Dewandel, B.; Gandolfi, J.M.; Mascré, C.; Ahmed, S. Understanding Groundwater Systems and Their Functioning through the Study of Stable Water Isotopes in a Hard-Rock Aquifer (Maheshwaram Watershed, India). J. Hydrol. 2011, 397, 55–70. [Google Scholar] [CrossRef]
- Sun, Z.; Ma, R.; Wang, Y.; Ma, T.; Liu, Y. Using Isotopic, Hydrogeochemical-Tracer and Temperature Data to Characterize Recharge and Flow Paths in a Complex Karst Groundwater Flow System in Northern China. Hydrogeol. J. 2016, 24, 1393–1412. [Google Scholar] [CrossRef]
- Folch, A.; Menció, A.; Puig, R.; Soler, A.; Mas-Pla, J. Groundwater Development Effects on Different Scale Hydrogeological Systems Using Head, Hydrochemical and Isotopic Data and Implications for Water Resources Management: The Selva Basin (NE Spain). J. Hydrol. 2011, 403, 83–102. [Google Scholar] [CrossRef]
- Antal, E. A Fertő tó Éghajlata. In A Fertő tó Természeti Adottságai; Kováts, Z., Kozmáné Tóth, E., Eds.; Országos Meteorológiai Szolgálat: Budapest, Hungary, 1982; pp. 44–124. [Google Scholar]
- Dövényi, Z. Magyarország Kistájainak Katasztere, 2nd ed.; Dövényi, Z., Ed.; MTA Földrajztudományi Kutatóintézet: Budapest, Hungary, 2010. [Google Scholar]
- Kovács, Z. Kapuvár Szénhidrogén Koncesszióra Javasolt Terület Komplex Érzékenységi És Terhelhetőségi Vizsgálati Jelentés Tervezete; Magyar Bányászati és Földtani Szolgálat: Budapest, Hungary, 2019. [Google Scholar]
- Földessy, J.; Molnár, F.; Biró, L. Ércföldtan Magyarországon a Földtani Közlöny 150 Évének Tükrében. Földtani Közlöny 2020, 150, 315–334. [Google Scholar] [CrossRef]
- Török, K. Multiple Fluid Migration Events in the Sopron Gneisses during the Alpine High-Pressure Metamorphism, as Recorded by Bulk-Rock and Mineral Chemistry and Fluid Inclusions. Neues Jahrb. Mineral. Abh. 2002, 177, 1–36. [Google Scholar] [CrossRef]
- Kósa, L. A Soproni-Hegység Uránkutatásának Felújítása; Mecseki Ércbányászati Vállalat: Pécs, Hungary, 1968. [Google Scholar]
- Mentes, G.; Eper-Pápai, I. Investigation of Temperature and Barometric Pressure Variation Effects on Radon Concentration in the Sopronbánfalva Geodynamic Observatory, Hungary. J. Environ. Radioact. 2015, 149, 64–72. [Google Scholar] [CrossRef] [PubMed]
- Freiler, Á.; Horváth, Á.; Török, K.; Földes, T. Origin of Radon Concentration of Csalóka Spring in the Sopron Mountains (West Hungary). J. Environ. Radioact. 2016, 151, 174–184. [Google Scholar] [CrossRef]
- Kisházi, P.; Ivancsics, J. A Soproni Gneisz Formáció Genetikai Kőzettana. Földtani Közlöny 1989, 119, 153–166. [Google Scholar]
- Kisházi, P.; Ivancsics, J. A Soproni Csillámpala Formáció Genetikai Kőzettana. Földtani Közlöny 1987, 117, 203–221. [Google Scholar]
- Draganits, E. Two Crystalline Series of the Sopron Hills (Burgenland) and Their Correlation to the Lower Austroalpine in Eastern Australia. Jahrb. Geol. Bundesanst. 1998, 141, 113–146. [Google Scholar]
- Török, K. Multiple Fluid Migration Events and REE+Th Mineralisation during Alpine Metamorphism in the Sopron Micaschist from the Eastern-Alps (Sopron Area, Western Hungary). Földtani Közlöny 2020, 150, 45. [Google Scholar] [CrossRef]
- Haas, J.; Budai, T.; Csontos, L.; Fodor, L.; Konrád, G.; Koroknai, B. Magyarország Prekainozoós Medencealjzatának Földtana Magyarázó “Magyarország Pre-Kainozoos Földtani Térképéhez”; 2014; ISBN 9789636712983. Available online: https://eles.mbfsz.gov.hu/sites/default/files/file/2018/03/23/aljzat_magyarazo.pdf (accessed on 29 March 2023).
- Freeze, R.A.; Cherry, J.A. Groundwater; Prentice-Hall: Hoboken, NJ, USA, 1979; Volume 39, ISBN 0133653129. [Google Scholar]
- Németh, L.; Elsholtz, L. A VI. Számú Kutatócsoport 1959. In Évi Jelentése a Soproni-Hegység Területén Végzett Radiohidrológiai Kutatásokról, 1960. [Google Scholar]
- Castanea Környezetvédelmi Egyesület. Sopron Környékének Forrásai, Sopron, Hungary, 2003.
- Herzig, A.; Dokulil, M. Neusiedler See–ein Steppensee in Europa. In Ökologie und Schutz von Seen. Dokulil, M.; Hamm, A., Kohl, J.G., Eds.; Facultas: Wien, Austria, 2001; pp. 401–415. [Google Scholar]
- Löffler, H. Der Neusiedlersee: Naturgeschichte Eines Steppensees, 1st ed.; Molden: Graz, Austria, 1974. [Google Scholar]
- Magyar, N.; Hatvani, I.G.; Székely, I.K.; Herzig, A.; Dinka, M.; Kovács, J. Application of Multivariate Statistical Methods in Determining Spatial Changes in Water Quality in the Austrian Part of Neusiedler See. Ecol. Eng. 2013, 55, 82–92. [Google Scholar] [CrossRef]
- Tóth, J. Groundwater discharge: A common generator of diverse geologic and morphologic phenomena. Int. Assoc. Sci. Hydrol. Bull. 1971, 16, 7–24. [Google Scholar] [CrossRef]
- Surbeck, H. Alpha Spectrometry Sample Preparation Using Selectively Adsorbing Thin Films. Appl. Radiat. Isot. 2000, 53, 97–100. [Google Scholar] [CrossRef] [PubMed]
- Baják, P.; Csondor, K.; Pedretti, D.; Muniruzzaman, M.; Surbeck, H.; Izsák, B.; Vargha, M.; Horváth, Á.; Pándics, T.; Erőss, A. Refining the Conceptual Model for Radionuclide Mobility in Groundwater in the Vicinity of a Hungarian Granitic Complex Using Geochemical Modeling. Appl. Geochem. 2022, 137, 105201. [Google Scholar] [CrossRef]
- Jobbágy, V.; Dirican, A.; Wätjen, U. Radiochemical Characterization of Mineral Waters for a European Interlaboratory Comparison. Microchem. J. 2013, 110, 675–680. [Google Scholar] [CrossRef]
- Kendall, C.; Caldwell, E.A. Fundamentals of Isotope Geochemistry. In Isotope Tracers in Catchment Hydrology; Elsevier: Amsterdam, The Netherlands, 1998; pp. 51–86. [Google Scholar]
- Craig, H. Isotopic Variations in Meteoric Waters. Science 1961, 133, 1702–1703. [Google Scholar] [CrossRef]
- Tóth, J. Hydraulic Continuity In Large Sedimentary Basins. Hydrogeol. J. 1995, 3, 4–16. [Google Scholar] [CrossRef]
- Jablánczy, S.; Firbás, O. A Soproni Hegyvidéki Erdők Vízrajzi Felvétele. Az Erdő 1956, 5, 16–20. [Google Scholar]
- Back, W. Hydrochemical Facies and Ground-Water Flow Patterns in Northern Part of Atlantic Coastal Plain; USGS: Reston, VA, USA, 1966. [Google Scholar]
- Gat, J.R. Oxygen and hydrogen isotopes in the hydrologic cycle. Annu. Rev. Earth Planet. Sci. 1996, 24, 225–262. [Google Scholar] [CrossRef]
- Czuppon, G.; Demény, A.; Leél-Őssy, S.; Óvari, M.; Molnár, M.; Stieber, J.; Kiss, K.; Kármán, K.; Surányi, G.; Haszpra, L. Cave Monitoring in the Béke and Baradla Caves (Northeastern Hungary): Implications for the Conditions for the Formation Cave Carbonates. Int. J. Speleol. 2018, 47, 13–28. [Google Scholar] [CrossRef]
- Deák, J. Environmental Isotopes and Water Chemical Studies in Hungary for Groundwater Research. In Proceedings of the International Symposium on Isotope Hydrology, Neuherberg, Germany, 19–23 June 1978; IAEA-SM: Neuherberg/Muenchen, Germany, 1978. [Google Scholar]
- Fórizs, I.; Berecz, T.; Molnár, Z.; Süveges, M. Origin of Shallow Groundwater of Csepel Island (South of Budapest, Hungary, River Danube): Isotopic and Chemical Approach. Hydrol. Process. 2005, 19, 3299–3312. [Google Scholar] [CrossRef]
- Izápy, G. Magyarország Forrásainak Katasztere [Natural Spring Cadastral of Hungary]; OVF-VITUKI Rt. Hidrológiai Intézete: Budapest, Hungary, 2002. [Google Scholar]
- Deák, J.; Coplen, T. Identification of Holocene and Pleistocene Groundwaters in Hungary Using Oxygen and Hydrogen Isotopic Ratios. In Proceedings of the Symposium on Isotopes in Water Resources Management, Vienna, Austria, 20–24 March 1995; IAEA: Vienna, Austria, 1996. [Google Scholar]
- Pregler, A.; Surbeck, H.; Eikenberg, J.; Werthmüller, S.; Szidat, S.; Türler, A. Increased Uranium Concentrations in Ground and Surface Waters of the Swiss Plateau: A Result of Uranium Accumulation and Leaching in the Molasse Basin and (Ancient) Wetlands? J. Environ. Radioact. 2019, 208–209, 106026. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baják, P.; Molnár, B.; Hegedűs-Csondor, K.; Tiljander, M.; Jobbágy, V.; Kohuth-Ötvös, V.; Izsák, B.; Vargha, M.; Horváth, Á.; Csipa, E.; et al. Natural Radioactivity in Drinking Water in the Surroundings of a Metamorphic Outcrop in Hungary: The Hydrogeological Answer to Practical Problems. Water 2023, 15, 1637. https://doi.org/10.3390/w15091637
Baják P, Molnár B, Hegedűs-Csondor K, Tiljander M, Jobbágy V, Kohuth-Ötvös V, Izsák B, Vargha M, Horváth Á, Csipa E, et al. Natural Radioactivity in Drinking Water in the Surroundings of a Metamorphic Outcrop in Hungary: The Hydrogeological Answer to Practical Problems. Water. 2023; 15(9):1637. https://doi.org/10.3390/w15091637
Chicago/Turabian StyleBaják, Petra, Bence Molnár, Katalin Hegedűs-Csondor, Mia Tiljander, Viktor Jobbágy, Viktória Kohuth-Ötvös, Bálint Izsák, Márta Vargha, Ákos Horváth, Emese Csipa, and et al. 2023. "Natural Radioactivity in Drinking Water in the Surroundings of a Metamorphic Outcrop in Hungary: The Hydrogeological Answer to Practical Problems" Water 15, no. 9: 1637. https://doi.org/10.3390/w15091637





