Removal of Carbamazepine in Aqueous Solution by TiO2 Ceramic Photo-Catalyst under Simulated Solar Light: Kinetics, Effects of Environmental Factors and Degradation Pathways
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemical Reagents
2.2. Solution Preparation
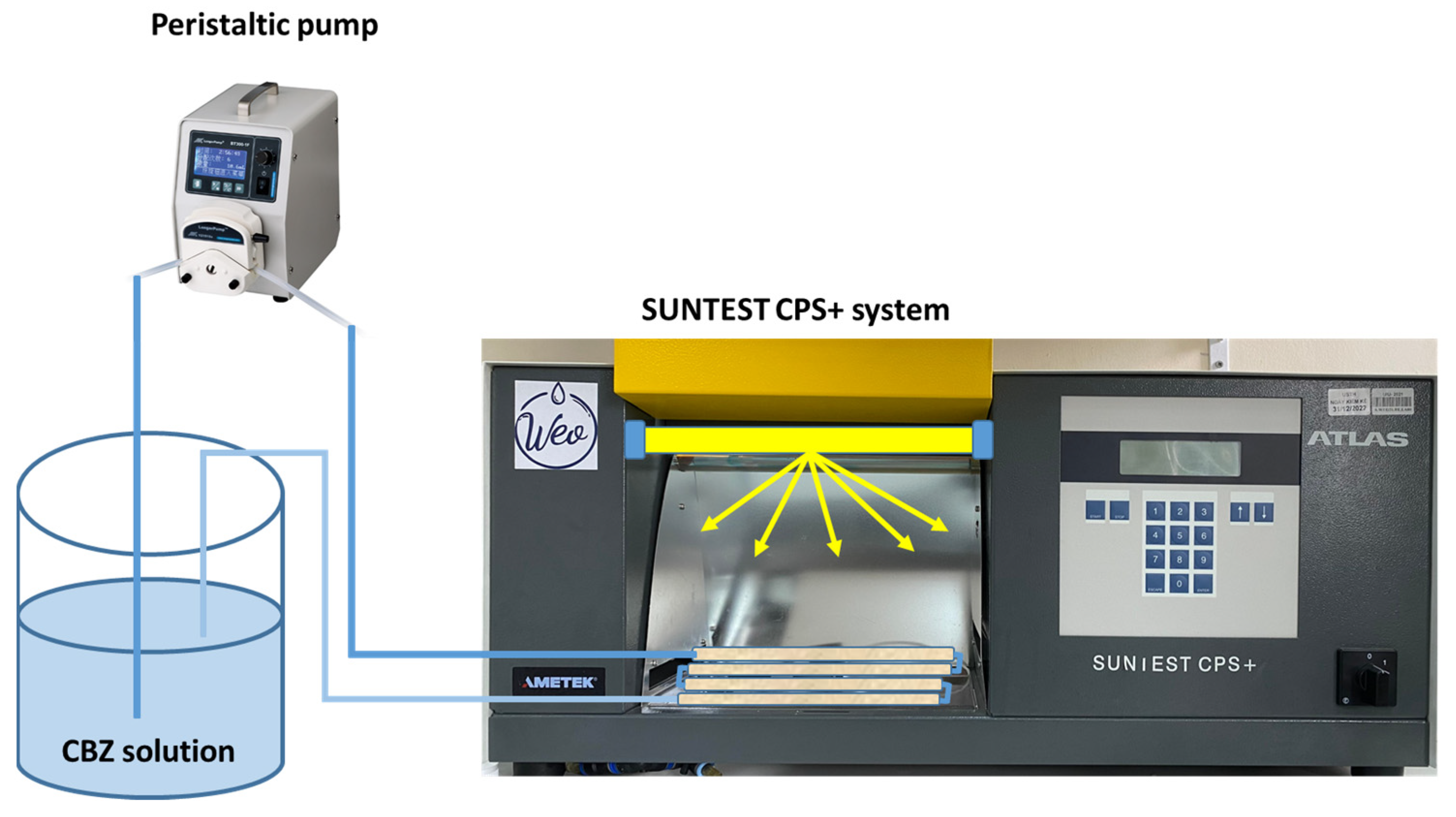
2.3. Experimental Setup
2.4. Analysis
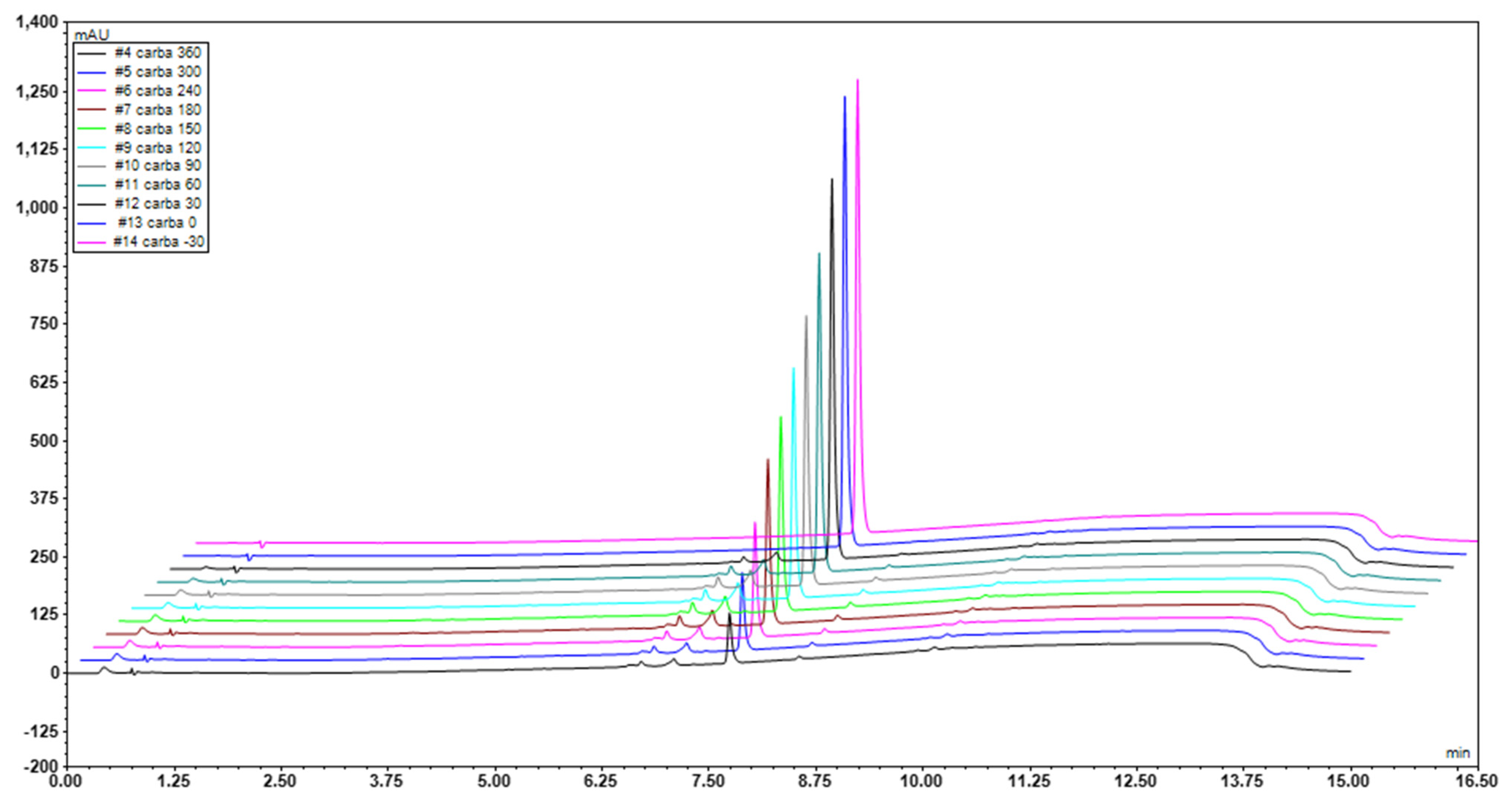
2.4.1. High-Performance Liquid Chromatography (HPLC)
2.4.2. Total Organic Carbon Analyzer (TOC)
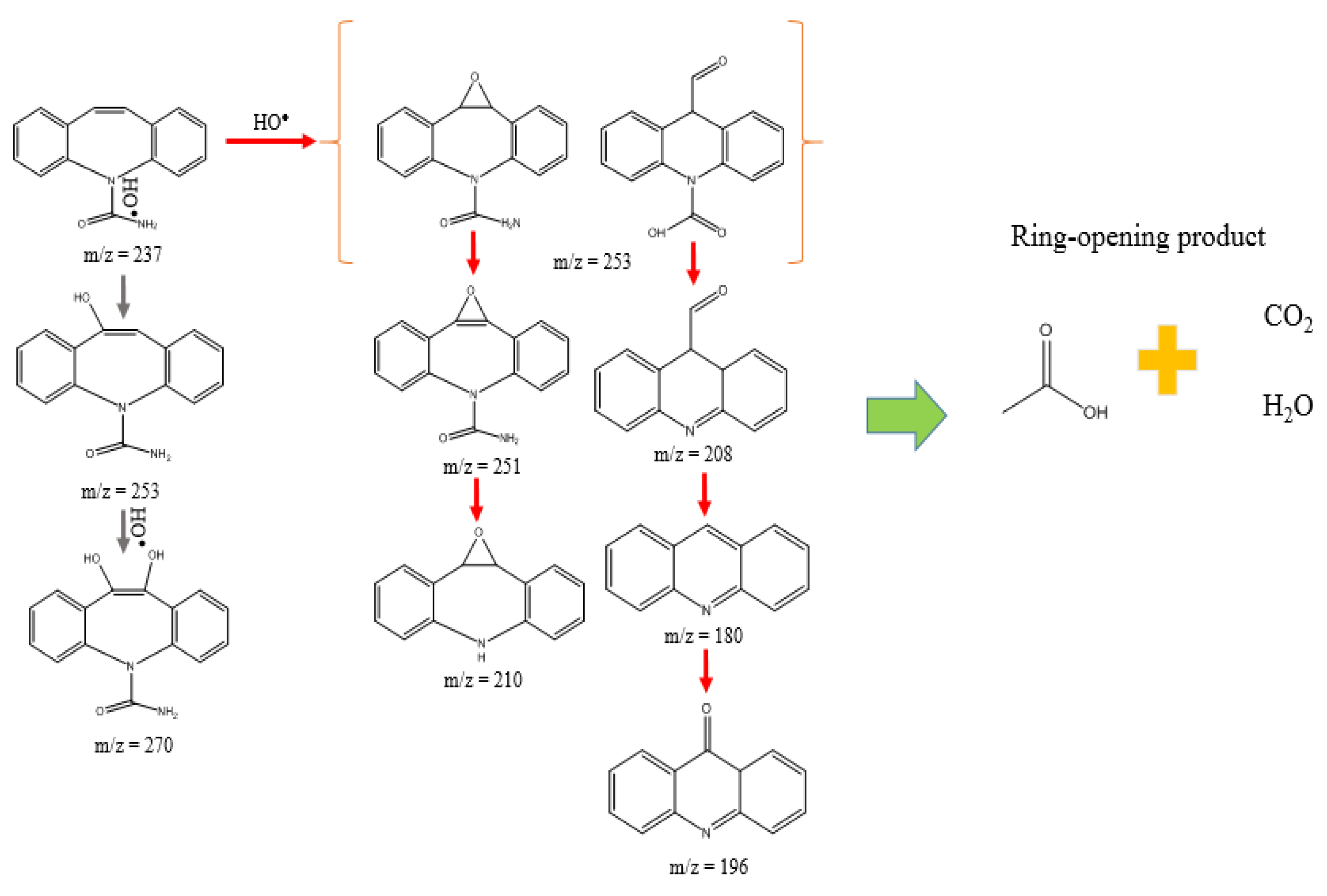
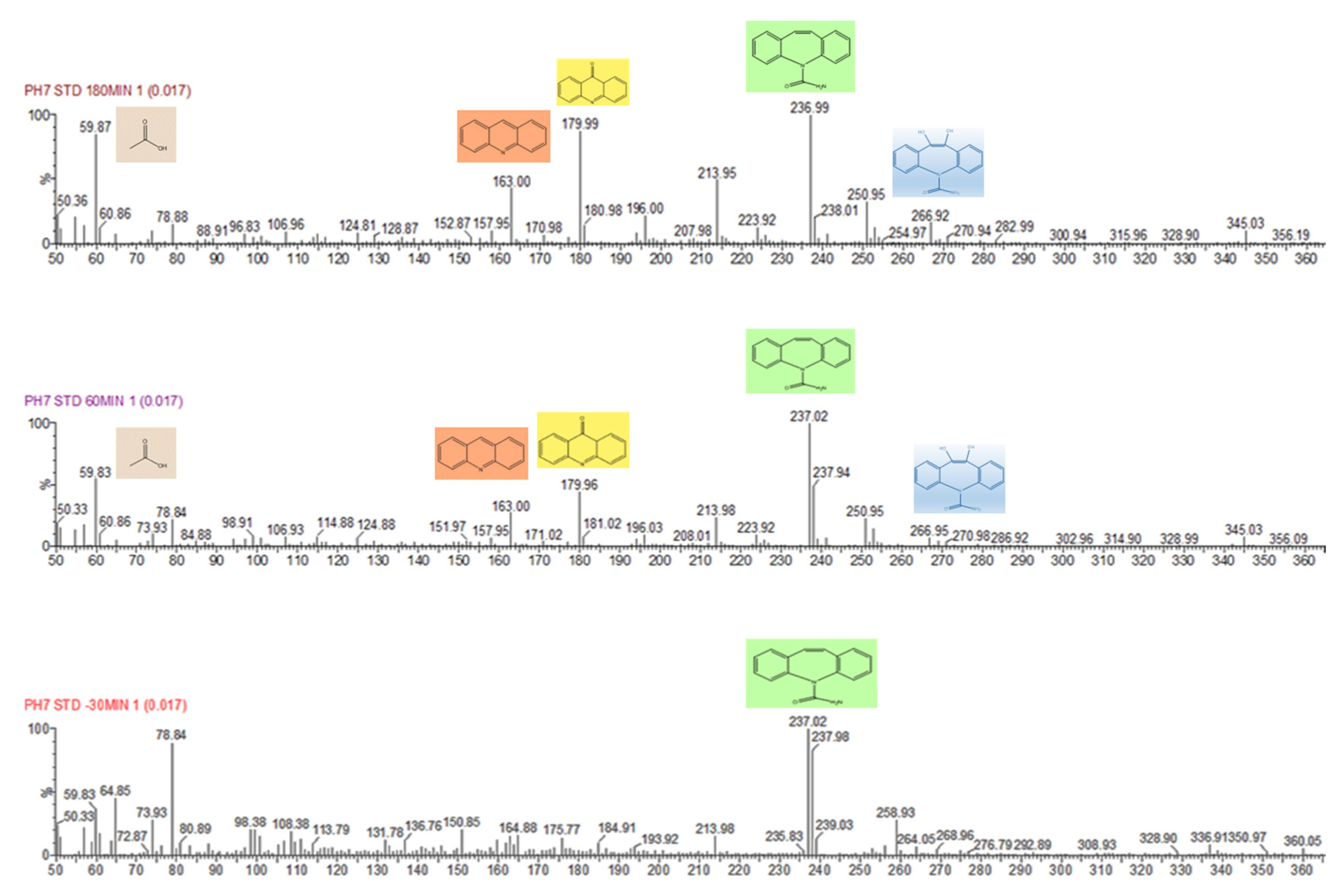
2.4.3. Identification of By-Products
2.5. Toxicity of By-Products
3. Results and Discussion
3.1. Photocatalytic Test and Kinetic Degradation
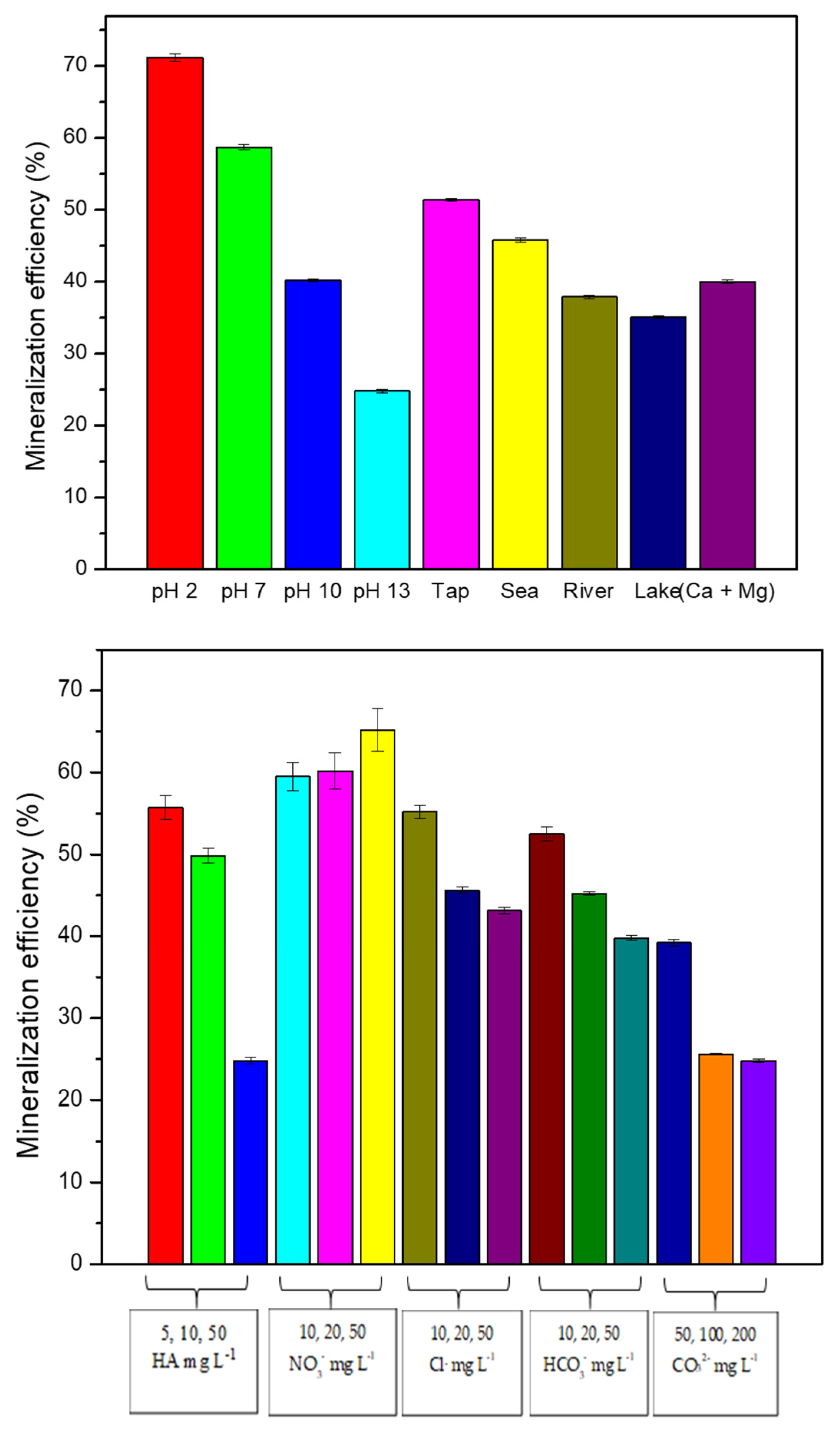
3.1.1. Effect of pH

3.1.2. Effect of Anions and Humic Acid (HA)

3.1.3. Effect of Cations Ca2+, Mg2+, and Water Matrices

3.2. Mineralization Studies
3.3. Mechanism and Proposed Pathways
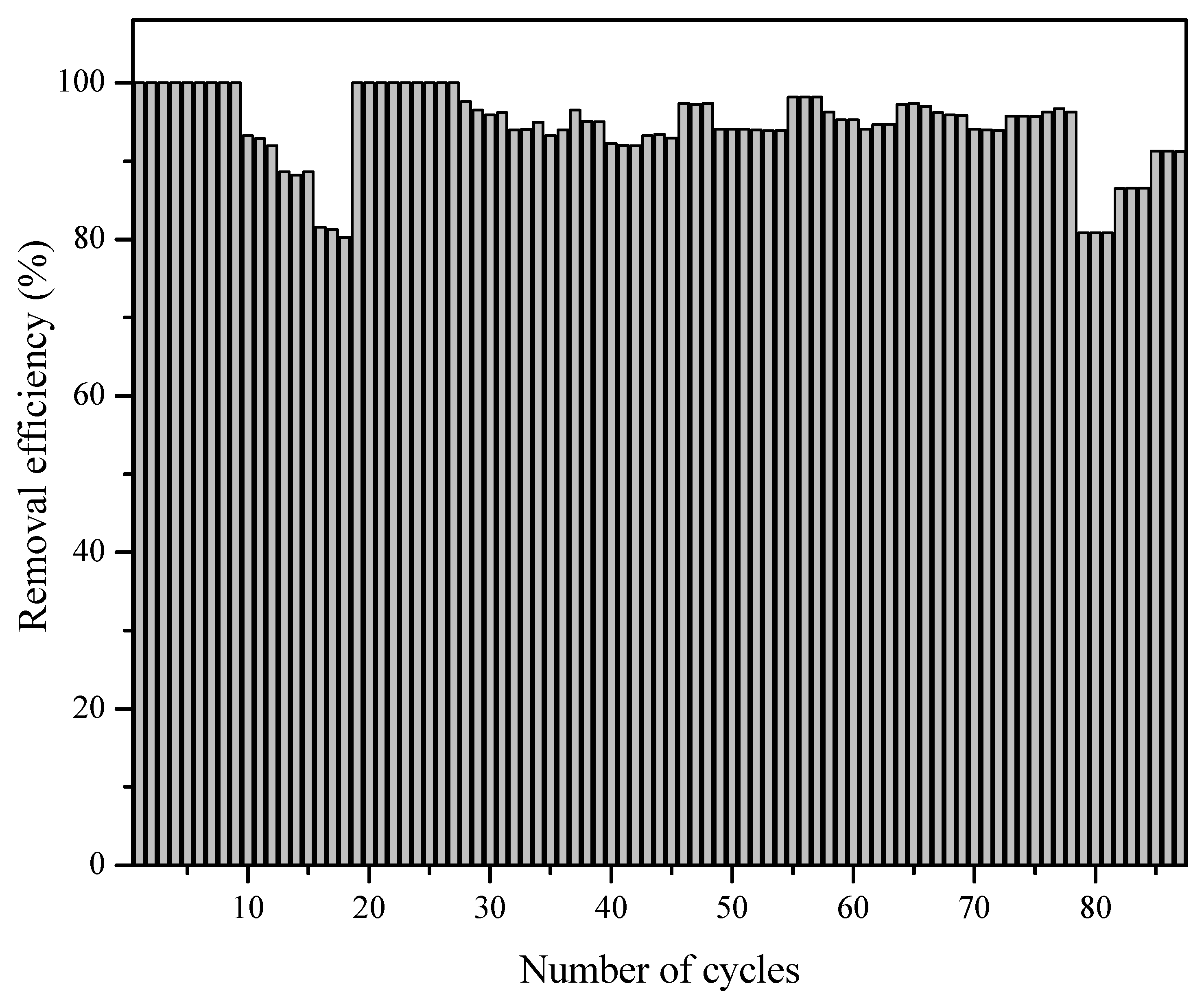
3.4. Reusability of Photocatalyst
3.5. Prediction of By-Product Toxicity
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Abbas, K.K.; Shabeeb, K.M.; Aljanabi, A.A.A.; Al-Ghaban, A.M.H.A. Photocatalytic degradation of Cefazolin over spherical nanoparticles of TiO2/ZSM-5 mesoporous nanoheterojunction under simulated solar light. Environ. Technol. Innov. 2020, 20, 101070. [Google Scholar] [CrossRef]
- Akter, S.; Islam, M.S.; Kabir, M.H.; Shaikh, M.A.A.; Gafur, M.A. UV/TiO2 photodegradation of metronidazole, ciprofloxacin and sulfamethoxazole in aqueous solution: An optimization and kinetic study. Arab. J. Chem. 2022, 15, 103900. [Google Scholar] [CrossRef]
- Fuart Gatnik, M.; Worth, A.P. Review of software tools for toxicity prediction. JRC Sci. Technol. Rep. 2010, 1–22. Available online: https://op.europa.eu/en/publication-detail/-/publication/fb675f8d-8758-4451-83c6-638a153e1635/language-en (accessed on 30 March 2023).
- Cao, J.; Nie, W.; Huang, L.; Ding, Y.; Lv, K.; Tang, H. Photocatalytic activation of sulfite by nitrogen vacancy modified graphitic carbon nitride for efficient degradation of carbamazepine. Appl. Catal. B Environ. 2019, 241, 18–27. [Google Scholar] [CrossRef]
- Sim, W.J.; Lee, J.W.; Lee, E.S.; Shin, S.K.; Hwang, S.R.; Oh, J.E. Occurrence and distribution of pharmaceuticals in wastewater from households, livestock farms, hospitals and pharmaceutical manufactures. Chemosphere 2011, 82, 179–186. [Google Scholar] [CrossRef]
- Kasprzyk-Hordern, B.; Dinsdale, R.M.; Guwy, A.J. The removal of pharmaceuticals, personal care products, endocrine disruptors and illicit drugs during wastewater treatment and its impact on the quality of receiving waters. Water Res. 2009, 43, 363–380. [Google Scholar] [CrossRef]
- Albani, F.; Riva, R.; Baruzzi, A. Carbamazepine clinical pharmacology: A review. Pharmacopsychiatry 1995, 28, 235–244. [Google Scholar] [CrossRef]
- Bizi, M. Activated Carbon and the Principal Mineral Constituents of a Natural Soil in the Presence of Carbamazepine. Water 2019, 11, 2290. [Google Scholar] [CrossRef]
- Clara, M.; Strenn, B.; Gans, O.; Martinez, E.; Kreuzinger, N.; Kroiss, H. Removal of selected pharmaceuticals, fragrances and endocrine disrupting compounds in a membrane bioreactor and conventional wastewater treatment plants. Water Res. 2005, 39, 4797–4807. [Google Scholar] [CrossRef]
- Haroune, L.; Salaun, M.; Ménard, A.; Legault, C.Y.; Bellenger, J.P. Photocatalytic degradation of carbamazepine and three derivatives using TiO2 and ZnO: Effect of pH, ionic strength, and natural organic matter. Sci. Total Environ. 2014, 475, 16–22. [Google Scholar] [CrossRef]
- Hu, Z.; Cai, X.; Wang, Z.; Li, S.; Wang, Z.; Xie, X. Construction of carbon-doped supramolecule-based g-C3N4/TiO2 composites for removal of diclofenac and carbamazepine: A comparative study of operating parameters, mechanisms, degradation pathways. J. Hazard Mater. 2019, 380, 120812. [Google Scholar] [CrossRef]
- Li, S.; Wang, Z.; Zhao, X.; Yang, X.; Liang, G.; Xie, X. Insight into enhanced carbamazepine photodegradation over biochar-based magnetic photocatalyst Fe3O4/BiOBr/BC under visible LED light irradiation. Chem. Eng. J. 2019, 360, 600–611. [Google Scholar] [CrossRef]
- Yang, L.; Jia, Y.; Peng, Y.; Zhou, P.; Yu, D.; Zhao, C.; He, J.; Zhan, C.; Lai, B. Visible-light induced activation of persulfate by self-assembled EHPDI/TiO2 photocatalyst toward efficient degradation of carbamazepine. Sci. Total Environ. 2021, 783, 146996. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Wang, Z.; Wang, C.; Li, X.; Wang, C.C. Photocatalytic degradation of DOM in urban stormwater runoff with TiO2 nanoparticles under UV light irradiation: EEM-PARAFAC analysis and influence of co-existing inorganic ions. Environ. Pollut. 2018, 243, 177–188. [Google Scholar] [CrossRef]
- Bouarioua, A.; Zerdaoui, M. Photocatalytic activities of TiO2 layers immobilized on glass substrates by dip-coating technique toward the decolorization of methyl orange as a model organic pollutant. J. Environ. Chem. Eng. 2017, 5, 1565–1574. [Google Scholar] [CrossRef]
- Sánchez-Rodríguez, D.; Méndez Medrano, M.G.; Remita, H.; Escobar-Barrios, V. Photocatalytic properties of BiOCl-TiO2 composites for phenol photodegradation. J. Environ. Chem. Eng. 2018, 6, 1601–1612. [Google Scholar] [CrossRef]
- Saleh, T.S.; Badawi, A.K.; Salama, R.S.; Mostafa, M.M.M. Design and Development of Novel Composites Containing Nickel Ferrites Supported on Activated Carbon Derived from Agricultural Wastes and Its Application in Water Remediation. Materials 2023, 16, 2170. [Google Scholar] [CrossRef]
- Wang, Y.; Li, Z.; Fu, W.; Sun, Y.; Dai, Y. Core–Sheath CeO2/SiO2 Nanofibers as Nanoreactors for Stabilizing Sinter-Resistant Pt, Enhanced Catalytic Oxidation and Water Remediation. Adv. Fiber Mater. 2022, 4, 1278–1289. [Google Scholar] [CrossRef]
- Alshorifi, F.T.; Ali, S.L.; Salama, R.S. Promotional Synergistic Effect of Cs–Au NPs on the Performance of Cs–Au/MgFe2O4 Catalysts in Catalysis 3,4-Dihydropyrimidin-2(1H)-Ones and Degradation of RhB Dye. J. Inorg. Organomet. Polym. Mater. 2022, 32, 3765–3776. [Google Scholar] [CrossRef]
- Qi, K.; Song, M.; Xie, X.; Wen, Y.; Wang, Z.; Wei, B.; Wang, Z. CQDs/biochar from reed straw modified Z-scheme MgIn2S4/BiOCl with enhanced visible-light photocatalytic performance for carbamazepine degradation in water. Chemosphere 2022, 287, 132192. [Google Scholar] [CrossRef]
- Sakkas, V.A.; Lambropoulou, D.A.; Albanis, T.A. Study of chlorothalonil photodegradation in natural waters and in the presence of humic substances. Chemosphere 2002, 48, 939–945. [Google Scholar] [CrossRef] [PubMed]
- Kato, S.; Hattori, K.; Tanaka, Y.; Miyazaki, Y.; Ishii, G.; Koura, S.; Negishi, N. Development of monolithic titanium dioxide ceramic photocatalysts with high physical rigidity and photocatalytic activity for underwater use. Ceram. Int. 2020, 46, 19285–19292. [Google Scholar] [CrossRef]
- Banerjee, P.; Eckert, A.O.; Schrey, A.K.; Preissner, R. ProTox-II: A webserver for the prediction of toxicity of chemicals. Nucleic Acids Res. 2018, 46, W257–W263. [Google Scholar] [CrossRef]
- Ali, A.; Shoeb, M.; Li, Y.; Li, B.; Khan, M.A. Enhanced photocatalytic degradation of antibiotic drug and dye pollutants by graphene-ordered mesoporous silica (SBA 15)/TiO2 nanocomposite under visible-light irradiation. J. Mol. Liquids 2021, 324, 114696. [Google Scholar] [CrossRef]
- Chen, H.; Wang, X.; Bi, W.; Wu, Y.; Dong, W. Photodegradation of carbamazepine with BiOCl/Fe3O4 catalyst under simulated solar light irradiation. J. Colloid Interface Sci. 2017, 502, 89–99. [Google Scholar] [CrossRef] [PubMed]
- Bui, V.H.; Vu, T.K.; To, H.T.; Negishi, N. Application of TiO2-ceramic/UVA photocatalyst for the photodegradation of sulfamethoxazole. Sustain. Chem. Pharm. 2022, 26, 100617. [Google Scholar] [CrossRef]
- Mao, X.; Li, M.; Li, M. Fabrication of Bi4O5Br2 photocatalyst for carbamazepine degradation under visible-light irradiation. Water Sci. Technol. 2021, 84, 77–88. [Google Scholar] [CrossRef]
- Lin, A.Y.-C.; Reinhard, M. Photodegradation of common environmental pharmaceuticals and estrogens in river water. Environ. Toxicol. Chem. 2005, 24, 1303. [Google Scholar] [CrossRef]
- Selli, E.; Baglio, D.; Montanarella, L.; Bidoglio, G. Role of humic acids in the TiO2-photocatalyzed degradation of tetrachloroethene in water. Water Res. 1999, 33, 1827–1836. [Google Scholar] [CrossRef]
- Ren, D.; Huang, B.; Bi, T.; Xiong, D.; Pan, X. Effects of pH and dissolved oxygen on the photodegradation of 17α-ethynylestradiol in dissolved humic acid solution. Environ. Sci. Process Impacts 2016, 18, 78–86. [Google Scholar] [CrossRef]
- Negishi, N.; Miyazaki, Y.; Kato, S.; Yang, Y. Effect of HCO3− concentration in groundwater on TiO2 photocatalytic water purification. Appl. Catal. B Environ. 2019, 242, 449–459. [Google Scholar] [CrossRef]
- Lyu, X.J.; Li, W.W.; Lam, P.K.S.; Yu, H.Q. Photodegradation of perfluorooctane sulfonate in environmental matrices. Sep. Purif. Technol. 2015, 151, 172–176. [Google Scholar] [CrossRef]
- Luna-Sanguino, G.; Ruíz-Delgado, A.; Duran-Valle, C.J.; Malato, S.; Faraldos, M.; Bahamonde, A. Impact of water matrix and oxidant agent on the solar assisted photodegradation of a complex mix of pesticides over titania-reduced graphene oxide nanocomposites. Catal. Today 2021, 380, 114–124. [Google Scholar] [CrossRef]
- Rioja, N.; Zorita, S.; Peñas, F.J. Effect of water matrix on photocatalytic degradation and general kinetic modeling. Appl. Catal. B Environ. 2016, 180, 330–335. [Google Scholar] [CrossRef]
- Sun, X.; Wang, W.; Li, C. Removal and mineralization of toluene under VUV/UV lamp irradiation in humid air: Effect of light wavelength, O2 and H2O. Arab. J. Chem. 2022, 15, 104108. [Google Scholar] [CrossRef]
- Irawaty, W.; Friedmann, D.; Scott, J.; Amal, R. Relationship between mineralization kinetics and mechanistic pathway during malic acid photodegradation. J. Mol. Catal. A Chem. 2011, 335, 151–157. [Google Scholar] [CrossRef]
- Dudziak, S.; Bielan, Z.; Kubica, P.; Zielinska-Jurek, A. Optimization of carbamazepine photodegradation on defective TiO2-based magnetic photocatalyst. J. Environ. Chem. Eng. 2021, 9, 105782. [Google Scholar] [CrossRef]
- Donner, E.; Kosjek, T.; Qualmann, S.; Kusk, K.O.; Heath, E.; Revitt, D.M.; Ledin, A.; Andersen, H.R. Ecotoxicity of carbamazepine and its UV photolysis transformation products. Sci. Total Environ. 2013, 443, 870–876. [Google Scholar] [CrossRef]
- Ferguson, L.R.; Denny, W.A. Genotoxicity of non-covalent interactions: DNA intercalators. Mutat. Res.—Fundam. Mol. Mech. Mutagen. 2007, 623, 14–23. [Google Scholar] [CrossRef]
- Han, Y.; Ma, M.; Li, N.; Hou, R.; Huang, C.; Oda, Y.; Wang, Z. Chlorination, chloramination and ozonation of carbamazepine enhance cytotoxicity and genotoxicity: Multi-endpoint evaluation and identification of its genotoxic transformation products. J. Hazard Mater. 2018, 342, 679–688. [Google Scholar] [CrossRef]
| Time (min) | Flow Rate (mL/min) | % UPW | % MeOH |
|---|---|---|---|
| 0 | 0.4 | 90 | 10 |
| 8 | 0.4 | 10 | 90 |
| 11 | 0.4 | 10 | 90 |
| 11.1 | 0.4 | 90 | 10 |
| 15 | 0.4 | 90 | 10 |
| Name | Formula | m/z | Structure |
|---|---|---|---|
| Acetic acid | C2H4O2 | 60 | 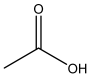 |
| Acridine | C13H9N | 180 | 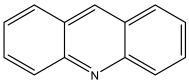 |
| Acridone | C13H9NO | 196 |  |
| Acridine-9-carboxylic acid | C14H9NO2 | 223 |  |
| Phenytoin | C15H12N2O2 | 253 | 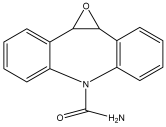 |
| 10,11-dihydro-10,11-dihydroxycarbamazepine | C15H14N2O3 | 270 | 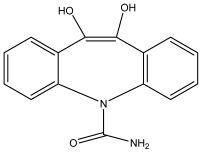 |
| Name | Predicted Toxicity Class |
|---|---|
| Carbamazepine | 4 |
| Acetic acid | 4 |
| Acridine | 1 |
| Acridone | 4 |
| Acridine-9-carboxylic acid | 5 |
| Phenytoin | 3 |
| 10,11-dihydro-10,11-dihydroxycarbamazepine | 4 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nghia, T.H.; Khanh, V.T.; Vu, C.T.; Oanh, N.T.K.; Van Anh, N.T.; Luyen, L.H.; Negishi, N.; Ouillon, S.; Van Hoi, B. Removal of Carbamazepine in Aqueous Solution by TiO2 Ceramic Photo-Catalyst under Simulated Solar Light: Kinetics, Effects of Environmental Factors and Degradation Pathways. Water 2023, 15, 1583. https://doi.org/10.3390/w15081583
Nghia TH, Khanh VT, Vu CT, Oanh NTK, Van Anh NT, Luyen LH, Negishi N, Ouillon S, Van Hoi B. Removal of Carbamazepine in Aqueous Solution by TiO2 Ceramic Photo-Catalyst under Simulated Solar Light: Kinetics, Effects of Environmental Factors and Degradation Pathways. Water. 2023; 15(8):1583. https://doi.org/10.3390/w15081583
Chicago/Turabian StyleNghia, Trinh Hoang, Vu Toan Khanh, Cam Tu Vu, Nguyen Thi Kieu Oanh, Nguyen Thi Van Anh, Le Hong Luyen, Nobuaki Negishi, Sylvain Ouillon, and Bui Van Hoi. 2023. "Removal of Carbamazepine in Aqueous Solution by TiO2 Ceramic Photo-Catalyst under Simulated Solar Light: Kinetics, Effects of Environmental Factors and Degradation Pathways" Water 15, no. 8: 1583. https://doi.org/10.3390/w15081583
APA StyleNghia, T. H., Khanh, V. T., Vu, C. T., Oanh, N. T. K., Van Anh, N. T., Luyen, L. H., Negishi, N., Ouillon, S., & Van Hoi, B. (2023). Removal of Carbamazepine in Aqueous Solution by TiO2 Ceramic Photo-Catalyst under Simulated Solar Light: Kinetics, Effects of Environmental Factors and Degradation Pathways. Water, 15(8), 1583. https://doi.org/10.3390/w15081583









