Hydrochemical Anomalies in the Vicinity of the Abandoned Molybdenum Ores Processing Tailings in a Permafrost Region (Shahtama, Transbaikal Region)
Abstract
1. Introduction
2. Materials and Methods
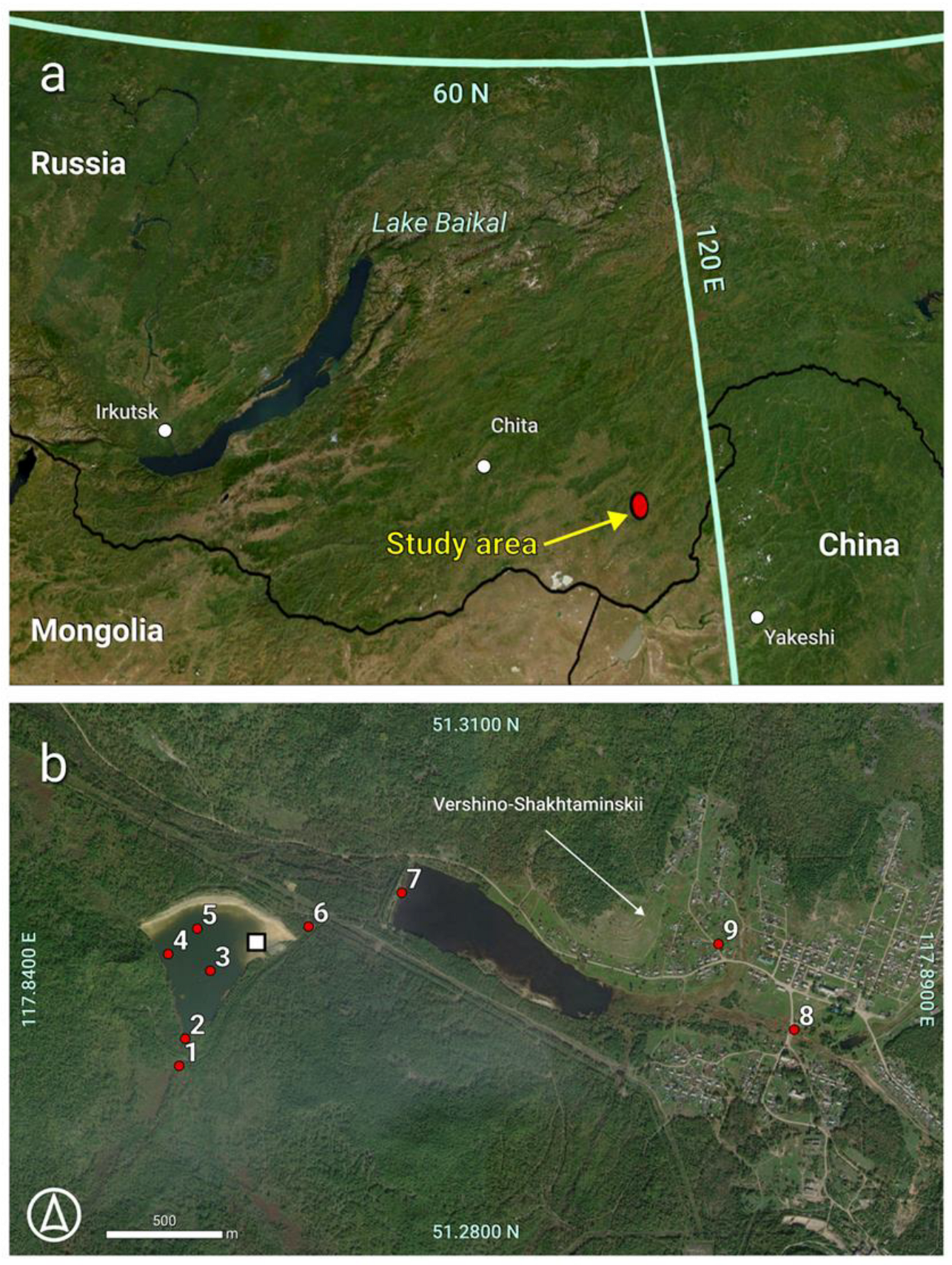
2.1. Object
2.2. Methods
2.2.1. Sampling and Field Study
2.2.2. Laboratory Analysis
3. Results
3.1. The Composition of the Solid Matter
3.2. The Composition of the Water Samples
4. Discussion
5. Conclusions
- The composition of molybdenum ore processing wastes contained in the abandoned Shakhtama storage was determined. The largest amounts are Ti, Mn, Ba, Pb, Mo, Cu, Rb, and Sr. The tailings under consideration contain precious metals: on average 0.51 g/t gold and 7.4 g/t silver. In addition, they contain extremely scarce components, indium (0.42 g/t) and germanium (2.4 g/t). The composition of the waste also includes elements of the first hazard class: Tl (2.7 g/t) and As (65 g/t).
- Waste has been shown to be a source of a wide range of chemical elements migrating with water flows. Elements pass into an aqueous solution due to the oxidation and dissolution of ore minerals that remain in the waste. This is also due to hypercryogenesis reactions: weathering of tailings material under the influence of seasonal temperature difference and when the alternation of water freezing and thawing leads to the material cracking and the intensification of its oxidation.
- In aqueous solutions in the vicinity of the tailings, among the elements whose concentrations exceed the clark values for river waters, there are Ca, Mg, K, Na, Al, Sr, Rb, Ba, Ni, Mn, Li, Zn, Sb, Se, Cu, Mo, La, Cs, Ga, Be, As, Sn, Cd, Tl, and Pb.
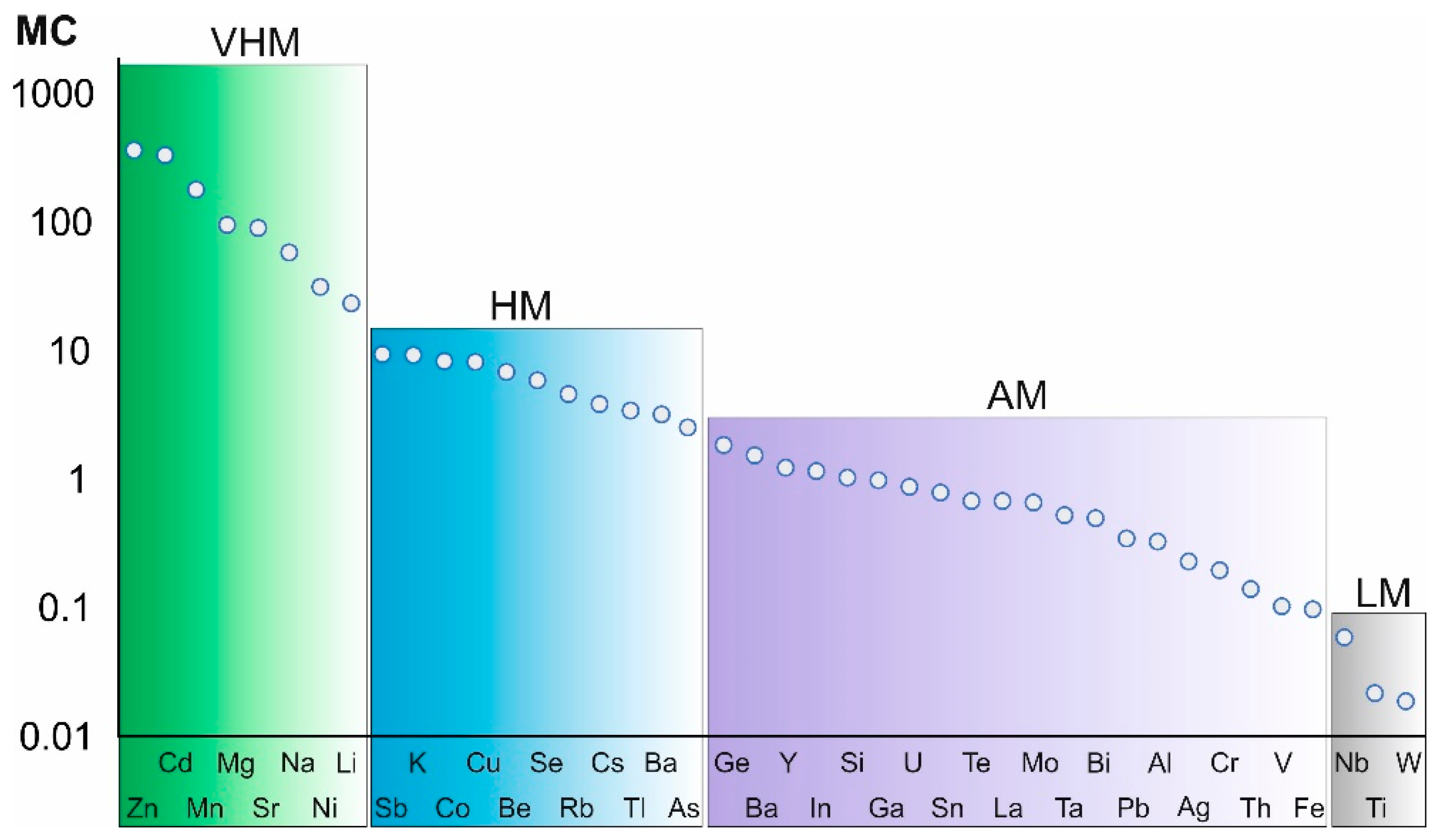
- Elements with very high mobility include alkaline and alkaline earth metals, i.e., the main rock-forming components (Na, Li, Mg, Sr), which enter the solution due to leaching from minerals of the host rocks, and metals (Zn, Cd, Mn, Ni), which enter the solution due to the dissolution of sulfides present in the composition of ores. Elements with high mobility include Sb, Co, Cu, Be, Se, and Tl, which enter the solution due to the dissolution of ore minerals and their impurities, as well as desorption. It should be noted that Shakhtama waste is a potential source of beryllium and thallium, elements of the first hazard class. Elements with medium mobility include As, an element of the first hazard class, as well as Mo, Fe, Pb. Elements Nb, Ti, and W are inactive.
- Hydrochemical anomalies of cadmium and lead in surface and underground waters in the vicinity of the tailings can be associated with the release of the studied waste from the substance, and the anomalies of molybdenum and arsenic are more likely due to the regional background.
- The main chemical forms due to the presence of elements in the solutions of ponds on the surface of the tailings are free-ion and sulfate complexes. In the samples of Shahtama River and ground waters, these include carbonate, hydrocarbonate, and hydroxide complexes.
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Angelakis, A.N.; Snyder, S.A. Wastewater Treatment and Reuse: Past, Present, and Future. Water 2015, 7, 4887–4895. [Google Scholar] [CrossRef]
- Wolkersdorfer, C.; Mugova, E. Effects of Mining on Surface Water. In Encyclopedia of Inland Waters, 2nd ed.; Elsevier: Amsterdam, The Netherlands, 2021. [Google Scholar]
- Motyka, J.; d’Obyrn, K. The radius of influence of mine drainage-definitions, methods of determination, and practical issues. J. Hydrol. 2022, 613, 128422. [Google Scholar] [CrossRef]
- Plante, B.; Bussière, B.; Benzaazoua, M. Lab to field scale effects on contaminated neutral drainage prediction from the Tio mine waste rocks. J. Geochem. Explor. 2014, 137, 37–47. [Google Scholar] [CrossRef]
- Tame, C.; Hudson-Edwards, K.; Potter, H. Weathering of Zinc-(Zn)-bearing Mine Wastes in a Neutral Mine Drainage Setting, Gunnerside Gill, Yorkshire. Procedia Earth Planet. Sci. 2017, 17, 284–287. [Google Scholar] [CrossRef]
- Barago, N.; Pavoni, E.; Floreani, F.; Crosera, M.; Adami, G.; Lenaz, D.; Covelli, S. Hydrogeochemistry of thallium and other potentially toxic elements in neutral mine drainage at the decommissioned Pb-Zn Raibl mine (Eastern Alps, Italy). J. Geochem. Explor. 2023, 245, 107129. [Google Scholar] [CrossRef]
- Majzlan, J.; Števko, M.; Chovan, M.; Luptáková, J.; Milovská, S.; Milovský, R.; Kupka, D. Mineralogy and geochemistry of the copper-dominated neutral mine drainage at the Cu deposit Ľubietová-Podlipa (Slovakia). Appl. Geochem. 2018, 92, 59–70. [Google Scholar] [CrossRef]
- Lefebvre, R.; Hockley, D.; Smolensky, J.; Gélinas, P. Multiphase transfer processes in waste rock piles producing acid mine drainage: 1: Conceptual model and system characterization. J. Contam. Hydrol. 2001, 52, 137–164. [Google Scholar] [CrossRef]
- Klassen, R.; Arenson, L.U.; Sego, D.C.; Biggar, K.W. Heat convection modeling in waste rock piles. In Proceedings of the Assessment and Remediation of Contaminated Sites in the Arctic and Cold Climates (ARCSACC), Edmonton, AB, Canada, 8–10 May 2005; pp. 247–255. [Google Scholar]
- Akhtar, S.; Hollaender, H.; Yuan, Q. Impact of heat and contaminants transfer from landfills to permafrost subgrade in arctic climate: A review. Cold Reg. Sci. Technol. 2022, 206, 103737. [Google Scholar] [CrossRef]
- Pham, H.N. Heat Transfer in Waste-Rock Piles Constructed in a Continuous Permafrost Region; University of Alberta: Edmonton, AB, Canada, 2013. [Google Scholar]
- Nordstrom, D.K. Baseline and premining geochemical characterization of mined sites. Appl. Geochem. 2015, 57, 17–34. [Google Scholar] [CrossRef]
- Muniruzzaman, M.; Kauppila, P.M.; Karlsson, T. Water quality prediction of mining waste facilities based on predictive models. Geol. Surv. Finl. Open File Res. Rep. 2018, 16, 67. [Google Scholar]
- Vriens, B.; Plante, B.; Seigneur, N.; Jamieson, H. Mine Waste Rock: Insights for Sustainable Hydrogeochemical Management. Minerals 2020, 10, 728. [Google Scholar] [CrossRef]
- Sui, C.; Fatichi, S.; Burlando, P.; Weber, E.; Battista, G. Modeling distributed metal pollution transport in a mine impacted catchment: Short and long-term effects. Sci. Total. Environ. 2022, 812, 151473. [Google Scholar] [CrossRef] [PubMed]
- Davé, N.; Blanchette, M. Role of cold climatic conditions in management of reactive mining waste in northern Canada. In Mining in the Arctic; CRC Press: Boca Raton, FL, USA, 2020; pp. 115–124. [Google Scholar] [CrossRef]
- Yi, X.; Su, D.; Seigneur, N.; Mayer, K.U. Modeling of Thermal-Hydrological-Chemical (THC) Processes during Waste Rock Weathering Under Permafrost Conditions. Front. Water 2021, 3, 645675. [Google Scholar] [CrossRef]
- Bao, Z.; Bain, J.; Holland, S.P.; Wilson, D.; Ptacek, C.J.; Blowes, D.W. Hydrogeochemical Response of a Variably Saturated Sulfide-Bearing Mine Waste-Rock Pile to Precipitation: A Field-Scale Study in the Discontinuous Permafrost Region of Northern Canada. Water Resour. Res. 2022, 58, e2021WR031082. [Google Scholar] [CrossRef]
- Schudel, G.; Plante, B.; Bussière, B.; Boulanger-Martel, V.; McBeth, J.M. Sulfide oxidation during the simulated weathering of pyrrhotite-rich tailings: Impacts of freeze-thaw cycles and chloride salinity. Cold Reg. Sci. Technol. 2023, 209, 103802. [Google Scholar] [CrossRef]
- Lu, J.; Walder, I.; Leiviskä, T. Impact of temperature on the leaching of sulphate, Co, Fe, Mn, Ni and Zn from the Ballangen tailings deposit, Norway: A laboratory column experiment. J. Water Clim. Change 2021, 12, 3558–3572. [Google Scholar] [CrossRef]
- Costis, S.; Coudert, L.; Mueller, K.K.; Cecchi, E.; Neculita, C.M.; Blais, J.-F. Assessment of the leaching potential of flotation tailings from rare earth mineral extraction in cold climates. Sci. Total. Environ. 2020, 732, 139225. [Google Scholar] [CrossRef]
- Yurkevich, N.; Olenchenko, V.; Bortnikova, S.; Saeva, O.; Korneeva, T. Cyanides, Arsenic, and Noble Metals in Abandoned Gold Ore Cyanidation Tailings and Surface Waters in a Permafrost Region (Transbaikal Territory, Russia). Mine Water Environ. 2021, 40, 943–955. [Google Scholar] [CrossRef]
- Nordstrom, D.K. Thermochemical redox equilibria of ZoBell’s solution. Geochim. Cosmochim. Acta 1977, 41, 1835–1841. [Google Scholar] [CrossRef]
- Sobek, A.A. Field and Laboratory Methods Applicable to Overburdens and Minesoils; Industrial Environmental Research Laboratory, Office of Research and Development, US Environmental Protection Agency: Washington, DC, USA, 1978. [Google Scholar]
- Gustafsson, J.P. Visual MINTEQ 3.1; KTH, Deptartment of Land and Water Resources Engineering: Stockholm, Sweden, 2014. [Google Scholar]
- Ball, J.W.; Nordstrom, D.K. WATEQ4F-User’s Manual with Revised Thermodynamic Data Base and Test Cases for Calculating Speciation of Major, Trace and Redox Elements in Natural Waters; USGS: Reston, VA, USA, 1991; pp. 91–183. [Google Scholar]
- Taylor, S.R.; McLennan, S.M. The Continental Crust: Its Composition and Evolution; OSTI: Oak Ridge, TN, USA, 1985. [Google Scholar]
- WHO. Guidelines for Drinking Water Quality; World Health Organization: Geneva, Switzerland, 2008. [Google Scholar]
- A General List. In General List of Fisheries Norms: Maximum Permissible Concentrations and Approximately Safe Levels of Harmful Substances in Water of Fishery Water Bodies; VNIRO: Moscow, Russia, 1999; p. 304.
- Parviainen, A.; Isoaari, P.; Loukola-Ruskeeniemi, K.; Nieto, J.M.; Gervilla, F. Occurrence and mobility of As in the Ylöjärbi Cu-W-As mine tailings. J. Geochem. Explor. 2012, 114, 36–45. [Google Scholar] [CrossRef]
- Wurl, J.; Mendez-Rodriguez, L.; Acosta-Vargas, B. Arsenic content in groundwater from the southern part of the San Antonio-El Triunfo mining district, Baja California Sur, Mexico. J. Hydrol. 2014, 518, 447–459. [Google Scholar] [CrossRef]
- Smuda, J.; Dold, B.; Spangenberg, J.E.; Friese, K.; Kobek, M.R.; Bustos, C.A.; Pfeifer, H.-R. Element cycling during the transition from alkaline to acidic environment in an active porphyry copper tailings impoundment, Chuquicamata, Chile. J. Geochem. Explor. 2014, 140, 23–40. [Google Scholar] [CrossRef]
- Aykol, A.; Budakoglu, M.; Kumral, M.; Gultekin, A.H.; Turhan, M.; Esenli, V.; Yavuz, F.; Orgun, Y. Heavy metal pollution and acid drainage from the abandoned Balya Pb-Zn sulfide Mine, NW Anatolia, Turkey. Environ. Geol. 2003, 45, 198–208. [Google Scholar] [CrossRef]
- Pavoni, E.; Covelli, S.; Adami, G.; Baracchini, E.; Cattelan, R.; Crosera, M.; Higueras, P.; Lenaz, D.; Petranich, E. Mobility and fate of Thallium and other potentially harmful elements in drainage waters from a decommissioned Zn-Pb mine (North-Eastern Italian Alps). J. Geochem. Explor. 2018, 188, 1–10. [Google Scholar] [CrossRef]
- Hällström, L.P. Mobility of Be, Bi, F, Ga, Ge and W in Surface Water and the Water Quality Impact on Epilithic Diatoms Downstream of the Historical Yxsjöberg Mine Site, Sweden. Mine Water Environ. 2022, 41, 731–747. [Google Scholar] [CrossRef]
- Batayneh, A.T. Toxic (aluminum, beryllium, boron, chromium and zinc) in groundwater: Health risk assessment. Int. J. Environ. Sci. Technol. 2012, 9, 153–162. [Google Scholar] [CrossRef]
- Liu, H.Y.; Probst, A.; Liao, B. Metal contamination of soils and crops affected by the Chenzhou lead/zinc mine spill (Hunan, China). Sci. Tot. Environ. 2005, 339, 153–166. [Google Scholar] [CrossRef]
- Linnik, P.N.; Nabivanets, B.I. Forms of Metals Migration in Fresh Surface Waters; Mir: Moscow, Russia, 1986; 271p. [Google Scholar]
- Florence, T.M. The speciation of trace elements in waters. Talanta 1982, 5, 345–364. [Google Scholar] [CrossRef]



| Element | Min | Max | Average |
|---|---|---|---|
| % | |||
| Si | 25 | 30 | 27 |
| Al | 5.4 | 8.9 | 7.3 |
| K | 3.5 | 5.1 | 4.2 |
| Fe | 2.3 | 4.5 | 3.3 |
| Na | 0.46 | 2.8 | 1.7 |
| Mg | 0.29 | 1.6 | 0.8 |
| g/t | |||
| Ti | 2000 | 5200 | 3400 |
| Mn | 210 | 3200 | 1500 |
| Ba | 740 | 2200 | 1400 |
| Pb | 62 | 4000 | 1100 |
| Zn | 130 | 1800 | 880 |
| Mo | 230 | 1000 | 630 |
| Cu | 100 | 1400 | 410 |
| Rb | 190 | 340 | 270 |
| Sr | 87 | 340 | 240 |
| Cr | 18 | 150 | 66 |
| As | 26 | 160 | 65 |
| V | 33 | 76 | 53 |
| Sb | 8.1 | 240 | 48 |
| Li | 20 | 55 | 40 |
| Ni | 21 | 41 | 31 |
| Ga | 14 | 25 | 20 |
| Nb | 12 | 24 | 17 |
| Y | 7.6 | 26 | 16 |
| Th | 6.0 | 22 | 13 |
| Cs | 6.6 | 18 | 11 |
| Sn | 3.1 | 15 | 10 |
| Ag | 0.48 | 24 | 7.4 |
| U | 1.5 | 17 | 6.6 |
| Co | 1.0 | 11 | 5.2 |
| Cd | 0.45 | 11 | 4.6 |
| Tl | 0.88 | 4.1 | 2.7 |
| Ge | 1.8 | 2.7 | 2.4 |
| Te | 1.1 | 3.5 | 1.9 |
| Au | 0.16 | 1.0 | 0.51 |
| In | 0.16 | 1.0 | 0.42 |
| Sample | pH | Eh, mV | Χ, µSm/cm | mg/L | ||
|---|---|---|---|---|---|---|
| SO42− | Cl− | HCO3− | ||||
| 1 | 6.93 | 498 | 72 | 15 | 0.07 | 40 |
| 2 | 6.80 | 506 | 102 | 22 | 0.08 | 55 |
| 3 | 7.09 | 507 | 74 | 20 | 0.05 | 34 |
| 4 | 6.76 | 512 | 97 | 35 | 0.06 | 23 |
| 5 | 5.66 | 560 | 320 | 150 | 0.05 | 12 |
| 6 | 6.80 | 523 | 65 | 15 | 0.09 | 37 |
| 7 | 7.93 | 489 | 280 | 66 | 1.5 | 92 |
| 8 | 7.42 | 482 | 390 | 110 | 1.7 | 120 |
| 9 | 8.06 | 468 | 320 | 60 | 1.2 | 130 |
| Element | Sample | Clark 1 | MPC WHO 2 | MPC RF 3 | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | ||||
| mg/L | ||||||||||||
| Ca | 5.7 | 9.0 | 5.5 | 7.6 | 33 | 4.7 | 28 | 43 | 36 | 12 | 75 | 180 |
| Mg | 1.9 | 3.0 | 1.9 | 2.5 | 7.3 | 1.7 | 7.5 | 12 | 8.9 | 2.9 | - | 40 |
| K | 1.0 | 1.1 | 1.0 | 1.2 | 4.0 | 1.1 | 2.2 | 2.8 | 2.1 | 2 | - | 50 |
| Na | 4.7 | 5.1 | 5.0 | 6.2 | 10 | 6 | 10 | 12 | 7.3 | 5 | 200 | 120 |
| Si | 4.5 | 4.4 | 2.7 | 3.2 | 2.9 | 4.4 | 0.2 | 4.2 | 8.1 | 6 | - | - |
| µg/L | ||||||||||||
| Al | 57 | 21 | 16 | 30 | 240 | 38 | 37 | 31 | 27 | 160 | 200 | 40 |
| Fe | 126 | 38 | 46 | 103 | 32 | 49 | 33 | 66 | 408 | 40 | 300 | 100 |
| Sr | 45 | 77 | 45 | 59 | 216 | 39 | 336 | 460 | 487 | 30 | - | 400 |
| Ba | 7.4 | 9.2 | 8.7 | 10 | 22 | 7.3 | 12 | 19 | 10 | 20 | 700 | 700 |
| Ni | 1.4 | 1.5 | 0.86 | 1.7 | 10 | 1.5 | 1.9 | 2.8 | 1.5 | 20 | 70 | 20 |
| Mn | 14 | 12 | 9.5 | 410 | 2800 | 9.2 | 20 | 29 | 160 | 3.0 | 400 | 100 |
| Te | 0.008 | 0.005 | 0.007 | 0.005 | 0.013 | 0.008 | 0.004 | 0.008 | 0.005 | 3.0 | - | 3 |
| Rb | 1.4 | 1.7 | 1.9 | 3.0 | 13 | 1.7 | 4.8 | 6.8 | 6.2 | 2.6 | - | 100 |
| Li | 1.6 | 2.3 | 2.0 | 2.9 | 9.5 | 1.5 | 3.1 | 4.3 | 6.6 | 2.5 | - | 80 |
| Y | 0.2 | 0.2 | 0.1 | 0.3 | 0.2 | 0.2 | 0.1 | 0.1 | 0.2 | 2.5 | - | - |
| Nd | 0.2 | 0.2 | 0.1 | 0.3 | 0.1 | 0.2 | 0.0 | 0.1 | 0.1 | 2.5 | - | - |
| W | 0.03 | 0.05 | 0.06 | 0.05 | 0.02 | 0.02 | 0.17 | 0.06 | 2.1 | 2.0 | - | 0.8 |
| Ti | 1.5 | 1.1 | 0.6 | 0.9 | 0.7 | 1.4 | 0.2 | 1.1 | 1.5 | 1.0 | - | 60 |
| Cr | 0.62 | 0.45 | 0.35 | 0.28 | 0.13 | 0.48 | 0.22 | 0.33 | 0.27 | 1.0 | 50 | 20 |
| Zn | 13 | 11 | 25 | 190 | 3300 | 11 | 7 | 24 | 8 | 1.0 | 3000 | 1000 |
| Sb | 0.40 | 0.97 | 1.3 | 2.2 | 4.6 | 0.41 | 5.9 | 4.4 | 1.1 | 1.0 | 20 | 5 |
| Ce | 0.37 | 0.22 | 0.07 | 0.50 | 0.27 | 0.26 | 0.11 | 0.13 | 0.17 | 1.0 | - | - |
| Lu | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 1.0 | - | - |
| Co | 0.10 | 0.09 | 0.07 | 0.12 | 0.44 | 0.08 | 0.16 | 0.21 | 0.27 | 0.7 | n.d. | 100 |
| Sc | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.30 | - | - |
| Se | 0.05 | 0.16 | 0.35 | 0.15 | 0.42 | 0.03 | 0.03 | 0.20 | 0.02 | 0.20 | 40 | 2 |
| Ag | 0.01 | 0.01 | 0.02 | 0.06 | 0.02 | 0.02 | 0.01 | 0.01 | 0.01 | 0.20 | 50 | 50 |
| In | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.20 | - | - |
| Th | 0.13 | 0.03 | 0.02 | 0.03 | 0.02 | 0.09 | 0.00 | 0.01 | 0.02 | 0.10 | - | - |
| U | 0.45 | 0.30 | 0.17 | 0.17 | 0.06 | 0.29 | 15 | 28 | 4.0 | 0.10 | - | - |
| Cu | 5.5 | 7.2 | 13 | 29 | 34 | 6.7 | 4.9 | 9.1 | 2.7 | 0.08 | 2000 | 1000 |
| Mo | 1.7 | 10 | 28 | 27 | 4.2 | 1.4 | 96 | 120 | 34 | 0.07 | 70 | 1 |
| La | 0.24 | 0.18 | 0.07 | 0.30 | 0.21 | 0.18 | 0.04 | 0.07 | 0.10 | 0.04 | - | - |
| V | 0.05 | 0.15 | 0.10 | 0.05 | 0.06 | 0.22 | 0.05 | 0.27 | 0.24 | 0.02 | - | 1 |
| Cs | 0.03 | 0.02 | 0.04 | 0.11 | 0.43 | 0.02 | 0.08 | 0.17 | 0.70 | 0.01 | - | - |
| Ga | 0.02 | 0.01 | 0.01 | 0.04 | 0.19 | 0.01 | 0.01 | 0.01 | 0.02 | 0.01 | - | - |
| Be | 0.02 | 0.01 | 0.01 | 0.01 | 0.17 | 0.01 | 0.01 | 0.01 | 0.02 | 0.01 | 12 | 0.2 |
| As | 1.21 | 1.07 | 1.02 | 1.43 | 1.37 | 0.99 | 2.00 | 0.96 | 2.98 | 0.004 | 10 | 10 |
| Sn | 0.12 | 0.10 | 0.06 | 0.10 | 0.08 | 0.05 | 0.05 | 0.08 | 0.06 | 0.004 | 1 | 0.12 |
| Cd | 0.15 | 0.15 | 0.73 | 0.92 | 16 | 0.19 | 0.29 | 0.82 | 0.14 | 0.002 | 3 | 1 |
| Tl | 0.01 | 0.01 | 0.01 | 0.04 | 0.10 | 0.01 | 0.01 | 0.02 | 0.01 | 0.001 | - | - |
| Pb | 4.0 | 2.5 | 1.8 | 10 | 4.0 | 2.0 | 1.8 | 3.7 | 1.7 | 0.001 | 10 | 10 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yurkevich, N.; Olenchenko, V.; Kartoziia, A.; Korneeva, T.; Bortnikova, S.; Saeva, O.; Tulisova, K.; Abrosimova, N. Hydrochemical Anomalies in the Vicinity of the Abandoned Molybdenum Ores Processing Tailings in a Permafrost Region (Shahtama, Transbaikal Region). Water 2023, 15, 1476. https://doi.org/10.3390/w15081476
Yurkevich N, Olenchenko V, Kartoziia A, Korneeva T, Bortnikova S, Saeva O, Tulisova K, Abrosimova N. Hydrochemical Anomalies in the Vicinity of the Abandoned Molybdenum Ores Processing Tailings in a Permafrost Region (Shahtama, Transbaikal Region). Water. 2023; 15(8):1476. https://doi.org/10.3390/w15081476
Chicago/Turabian StyleYurkevich, Nataliya, Vladimir Olenchenko, Andrei Kartoziia, Tatyana Korneeva, Svetlana Bortnikova, Olga Saeva, Kristina Tulisova, and Natalya Abrosimova. 2023. "Hydrochemical Anomalies in the Vicinity of the Abandoned Molybdenum Ores Processing Tailings in a Permafrost Region (Shahtama, Transbaikal Region)" Water 15, no. 8: 1476. https://doi.org/10.3390/w15081476
APA StyleYurkevich, N., Olenchenko, V., Kartoziia, A., Korneeva, T., Bortnikova, S., Saeva, O., Tulisova, K., & Abrosimova, N. (2023). Hydrochemical Anomalies in the Vicinity of the Abandoned Molybdenum Ores Processing Tailings in a Permafrost Region (Shahtama, Transbaikal Region). Water, 15(8), 1476. https://doi.org/10.3390/w15081476








