Distribution of Silver (Ag) and Silver Nanoparticles (AgNPs) in Aquatic Environment
Abstract
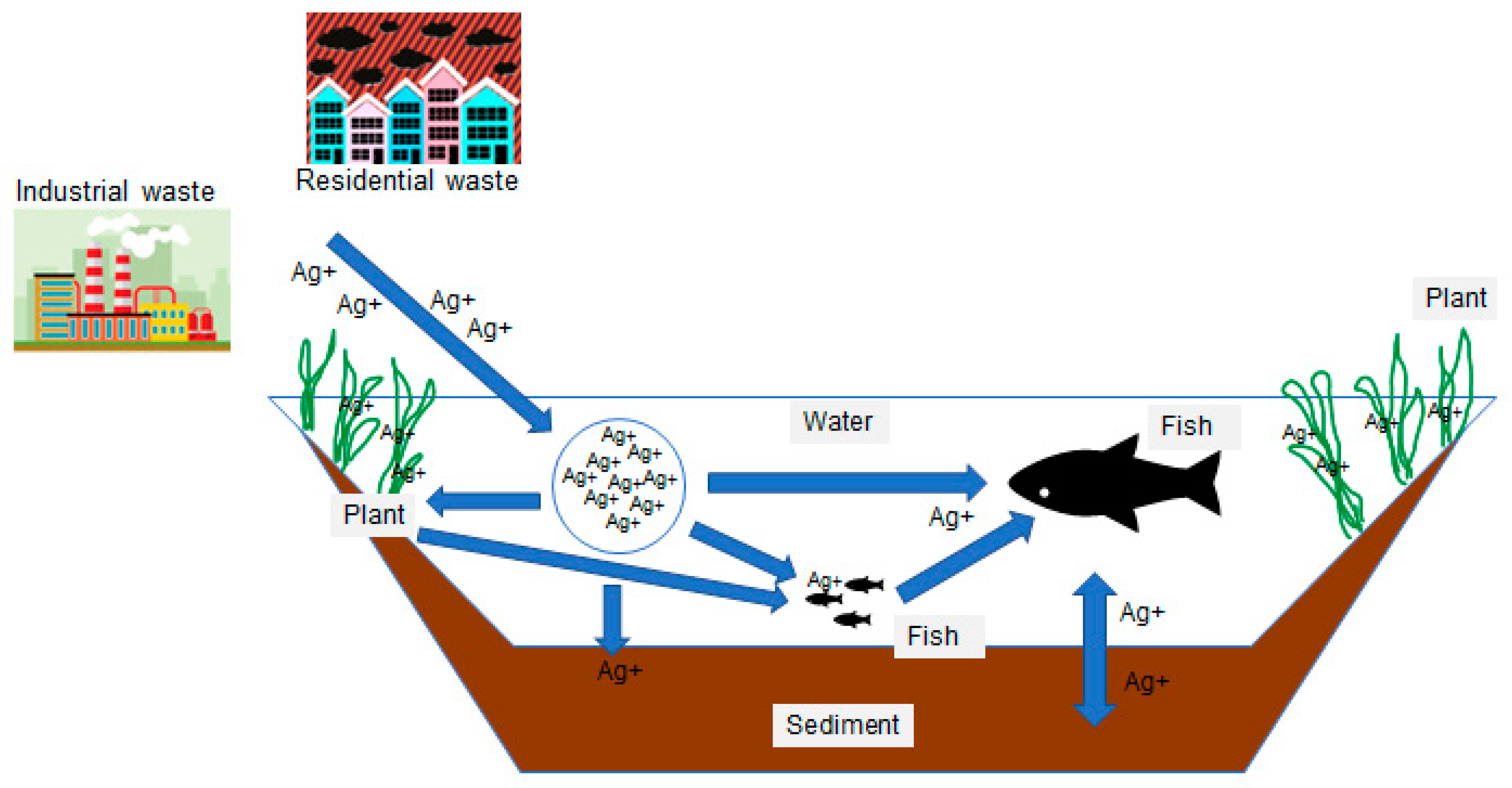
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Sample Collection
2.3. Preparation of Samples
2.4. Analytical Method
2.4.1. Inductively Coupled Plasma Mass Spectrometer (ICP-MS)
2.4.2. High-Resolution Transmission Electron Microscope (HR-TEM 200KV) with Energy-Dispersive X-ray Spectrometer (EDX)
2.5. Data Analysis
3. Results
3.1. Concentration of Ag in the Aquatic Environment
3.2. Analysis of Silver Nanoparticles in the Aquatic Environment
3.3. Accumulation of Ag in Sediment Samples
3.4. Accumulation of Ag in Plant Samples
3.5. Accumulation of Ag in Fish Organ
3.6. Values for BAF and BSAF
4. Discussion
4.1. The Presence of Ag in the Aquatic Environment
4.2. Nano-Size of Ag in the Aquatic Environment
4.3. Bioaccumulation of Ag in the Aquatic Ecosystem of Skudai River
4.4. Trophic Transfer of Ag through the Aquatic Food Chain
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gomathi, M.; Rajkumar, P.V.; Prakasam, A.; Ravichandran, K. Green synthesis of silver nanoparticles using Datura stramonium leaf extract and assessment of their antibacterial activity. Resour. Effic. Technol. 2017, 3, 280–284. [Google Scholar] [CrossRef]
- Al-Nasir, F.; Hijazin, T.J.; Al-Alawi, M.M.; Jiries, A.; Al-Madanat, O.Y.; Mayyas, A.; A. Al-Dalain, S.; Al-Dmour, R.; Alahmad, A.; Batarseh, M.I. Accumulation, Source Identification, and Cancer Risk Assessment of Polycyclic Aromatic Hydrocarbons (PAHs) in Different Jordanian Vegetables. Toxics 2022, 10, 643. [Google Scholar] [CrossRef]
- Jiries, A.; Al-Nasir, F.; Hijazin, T.J.; Al-Alawi, M.; El Fels, L.; Mayyas, A.; Al-Dmour, R.; Al-Madanat, O.Y. Polycyclic aromatic hydrocarbons in citrus fruit irrigated with fresh water under arid conditions: Concentrations, sources, and risk assessment. Arab. J. Chem. 2022, 15, 104027. [Google Scholar] [CrossRef]
- Gomes, H.I.O.; Martins, C.S.M.; Prior, J.A.V. Silver Nanoparticles as Carriers of Anticancer Drugs for Efficient Target Treatment of Cancer Cells. Nanomaterials 2021, 11, 964. [Google Scholar] [CrossRef]
- Alahmad, A.; Al-Zereini, W.A.; Hijazin, T.J.; Al-Madanat, O.Y.; Alghoraibi, I.; Al-Qaralleh, O.; Al-Qaraleh, S.; Feldhoff, A.; Walter, J.G.; Scheper, T. Green Synthesis of Silver Nanoparticles Using Hypericum perforatum L. Aqueous Extract with the Evaluation of Its Antibacterial Activity against Clinical and Food Pathogens. Pharmaceutics 2022, 14, 1104. [Google Scholar] [CrossRef]
- Kanwal, Z.; Raza, M.A.; Riaz, S.; Manzoor, S.; Tayyeb, A.; Sajid, I.; Naseem, S. Synthesis and characterization of silver nanoparticle-decorated cobalt nanocomposites (Co@AgNPs) and their density-dependent antibacterial activity. R. Soc. Open Sci. 2019, 6, 182135. [Google Scholar] [CrossRef] [Green Version]
- Salas-Papayanopolos, H.; Morales-Cepeda, A.B.; Sanchez, S.; Lafleur, P.G.; Gomez, I. Synergistic effect of silver nanoparticle content on the optical and thermo mechanical properties of poly (l-lactic acid)/glycerol triacetate blends. Polym. Bull. 2017, 74, 4799–4814. [Google Scholar] [CrossRef]
- Yaqoob, A.A.; Umar, K.; Ibrahim, M.N.M. Silver nanoparticles: Various methods of synthesis, size affecting factors and their potential applications—A review. Appl. Nanosci. 2020, 10, 1369–1378. [Google Scholar] [CrossRef]
- Salih, A.E.; Shanti, A.; Elsherif, M.; Alam, F.; Lee, S.; Polychronopoulou, K.; Almaskari, F.; AlSafar, H.; Yetisen, A.K.; Butt, H. Silver Nanoparticle-Loaded Contact Lenses for Blue-Yellow Color Vision Deficiency. Phys. Status Solidi (A) 2021, 219, 2100294. [Google Scholar] [CrossRef]
- McGillicuddy, E.; Murray, I.; Kavanagh, S.; Morrison, L.; Fogarty, A.; Cormican, M.; Dockery, P.; Prendergast, M.; Rowan, N.; Morris, D. Silver nanoparticles in the environment: Sources, detection and ecotoxicology. Sci. Total Environ. 2017, 575, 231–246. [Google Scholar] [CrossRef]
- Cascio, C.; Geiss, O.; Franchini, F.; Ojea-Jimenez, I.; Rossi, F.; Gilliland, D.; Calzolai, L. Detection, quantification and derivation of number size distribution of silver nanoparticles in antimicrobial consumer products. J. Anal. At. Spectrom. 2015, 30, 1255–1265. [Google Scholar] [CrossRef] [Green Version]
- Quadros, M.E.; Marr, L.C. Silver nanoparticles and total aerosols emitted by nanotechnology-related consumer spray products. Environ. Sci. Technol. 2011, 45, 10713–10719. [Google Scholar] [CrossRef]
- Rogers, K.R.; Navratilova, J.; Stefaniak, A.; Bowers, L.; Knepp, A.K.; Al-Abed, S.R.; Potter, P.; Gitipour, A.; Radwan, I.; Nelson, C.; et al. Characterization of engineered nanoparticles in commercially available spray disinfectant products advertised to contain colloidal silver. Sci. Total Environ. 2018, 619–620, 1375–1384. [Google Scholar] [CrossRef]
- Wasukan, N.; Srisung, S.; Kulthong, K.; Boonrungsiman, S.; Maniratanachote, R. Determination of silver in personal care nanoproducts and effects on dermal exposure. J. Nanopart. Res. 2015, 17, 425. [Google Scholar] [CrossRef]
- McGee, C.F. The effects of silver nanoparticles on the microbial nitrogen cycle: A review of the known risks. Environ. Sci. Pollut. Res. 2020, 27, 31061–31073. [Google Scholar] [CrossRef]
- Salieri, B.; Turner, D.A.; Nowack, B.; Hischier, R. Life cycle assessment of manufactured nanomaterials: Where are we? NanoImpact 2018, 10, 108–120. [Google Scholar] [CrossRef]
- Good, K.D.; Bergman, L.E.; Klara, S.S.; Leitch, M.E.; VanBriesen, J.M. Implications of engineered nanomaterials in drinking water sources. J. Am. Water Works Assoc. 2016, 108, E1–E17. [Google Scholar] [CrossRef]
- An, H.J.; Sarkheil, M.; Park, H.S.; Yu, I.J.; Johari, S.A. Comparative toxicity of silver nanoparticles (AgNPs) and silver nanowires (AgNWs) on saltwater microcrustacean, Artemia salina. Comp. Biochem. Physiol. Part-C Toxicol. Pharmacol. 2019, 218, 62–69. [Google Scholar] [CrossRef]
- Kabir, E.; Kumar, V.; Kim, K.H.; Yip, A.C.; Sohn, J.R. Environmental impacts of nanomaterials. J. Environ. Manag. 2018, 225, 261–271. [Google Scholar] [CrossRef]
- Garner, K.L.; Suh, S.; Lenihan, H.S.; Keller, A.A. Species sensitivity distributions for engineered nanomaterials. Environ. Sci. Technol. 2015, 49, 5753–5759. [Google Scholar] [CrossRef] [Green Version]
- Peters, R.J.B.; van Bemmel, G.; Milani, N.B.L.; den Hertog, G.C.T.; Undas, A.K.; van der Lee, M.; Bouwmeester, H. Detection of nanoparticles in Dutch surface waters. Sci. Total Environ. 2018, 621, 210–218. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Stoiber, M.; Wimmer, A.; Xu, Z.; Lindenblatt, C.; Helmreich, B.; Schuster, M. To What Extent Can Full-Scale Wastewater Treatment Plant Effluent Influence the Occurrence of Silver-Based Nanoparticles in Surface Waters? Environ. Sci. Technol. 2016, 50, 6327–6333. [Google Scholar] [CrossRef] [PubMed]
- Gottschalk, F.; Lassen, C.; Kjoelholt, J.; Christensen, F.; Nowack, B. Modeling flows and concentrations of nine engineered nanomaterials in the Danish environment. Int. J. Environ. Res. Public Health 2015, 12, 5581–5602. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sanchís, J.; Jiménez-Lamana, J.; Abad, E.; Szpunar, J.; Farré, M. Occurrence of Cerium-, Titanium-, and Silver-Bearing Nanoparticles in the Besòs and Ebro Rivers. Environ. Sci. Technol. 2020, 54, 3969–3978. [Google Scholar] [CrossRef] [PubMed]
- Dang, F.; Huang, Y.; Wang, Y.; Zhou, D.; Xing, B. Transfer and toxicity of silver nanoparticles in the food chain. Environ. Sci. Nano 2021, 8, 1519–1535. [Google Scholar] [CrossRef]
- Khoshnamvand, M.; Hao, Z.; Fadare, O.O.; Hanachi, P.; Chen, Y.; Liu, J. Toxicity of biosynthesized silver nanoparticles to aquatic organisms of different trophic levels. Chemosphere 2020, 258, 127346. [Google Scholar] [CrossRef]
- Marambio-Jones, C.; Hoek, E.V. A review of the antibacterial effects of silver nanomaterials and potential implications for human health and the environment. J. Nanopart. Res. 2010, 12, 1531–1551. [Google Scholar] [CrossRef]
- Fabrega, J.; Luoma, S.N.; Tyler, C.R.; Galloway, T.S.; Lead, J.R. Silver nanoparticles: Behaviour and effects in the aquatic environment. Environ. Int. 2011, 37, 517–531. [Google Scholar] [CrossRef]
- Sharifi, S.; Behzadi, S.; Laurent, S.; Laird Forrest, M.; Stroeve, P.; Mahmoudi, M. Toxicity of nanomaterials. Chem. Soc. Rev. 2012, 41, 2323–2343. [Google Scholar] [CrossRef] [Green Version]
- Bondarenko, O.; Juganson, K.; Ivask, A.; Kasemets, K.; Mortimer, M.; Kahru, A. Toxicity of Ag, CuO and ZnO nanoparticles to selected environmentally relevant test organisms and mammalian cells in vitro: A critical review. Arch. Toxicol. 2013, 87, 1181–1200. [Google Scholar] [CrossRef] [Green Version]
- Le Ouay, B.; Stellacci, F. Antibacterial activity of silver nanoparticles: A surface science insight. Nano Today 2015, 10, 339–354. [Google Scholar] [CrossRef] [Green Version]
- Soenen, S.J.; Parak, W.J.; Rejman, J.; Manshian, B. (Intra) Cellular stability of inorganic nanoparticles: Effects on cytotoxicity, particle functionality, and biomedical applications. Chem. Rev. 2015, 115, 2109–2135. [Google Scholar] [CrossRef] [PubMed]
- Kuehr, S.; Kaegi, R.; Maletzki, D.; Schlechtriem, C. Testing the bioaccumulation potential of manufactured nanomaterials in the freshwater amphipod Hyalella azteca. Chemosphere 2021, 263, 127961. [Google Scholar] [CrossRef] [PubMed]
- Xiao, B.; Zhang, Y.; Wang, X.; Chen, M.; Sun, B.; Zhang, T.; Zhu, L. Occurrence and trophic transfer of nanoparticulate Ag and Ti in the natural aquatic food web of Taihu Lake, China. Environ. Sci. Nano 2019, 6, 3431–3441. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, J.; Gao, Y.; Xiong, H.; Zhang, W.; Yan, B. The ZrO2 NPs enhanced the risk of arsenate by promoting its accumulation and reducing its detoxification during food chain transfer from Daphnia magna to zebrafish. J. Hazard. Mater. 2022, 424, 127338. [Google Scholar] [CrossRef] [PubMed]
- Griboff, J.; Horacek, M.; Wunderlin, D.A.; Monferran, M.V. Bioaccumulation and trophic transfer of metals, As and Se through a freshwater food web affected by antrophic pollution in Córdoba, Argentina. Ecotoxicol. Environ. Saf. 2018, 148, 275–284. [Google Scholar] [CrossRef]
- Goswami, L.; Kim, K.H.; Deep, A.; Das, P.; Bhattacharya, S.S.; Kumar, S.; Adelodun, A.A. Engineered nano particles: Nature, behavior, and effect on the environment. J. Environ. Manag. 2017, 196, 297–315. [Google Scholar] [CrossRef]
- Fang, T.; Lu, W.; Cui, K.; Li, J.; Yang, K.; Zhao, X.; Liang, Y.; Li, H. Distribution, bioaccumulation and trophic transfer of trace metals in the food web of Chaohu Lake, Anhui, China. Chemosphere 2019, 218, 1122–1130. [Google Scholar] [CrossRef]
- Ziyaadini, M.; Yousefiyanpour, Z.; Ghasemzadeh, J.; Zahedi, M.M. Biota-sediment accumulation factor and concentration of heavy metals (Hg, Cd, As, Ni, Pb and Cu) in sediments and tissues of Chiton lamyi (Mollusca: Polyplacophora: Chitonidae) in Chabahar Bay, Iran. Iran. J. Fish. Sci. 2017, 16, 1123–1134. [Google Scholar]
- Nourozifard, P.; Mortazavi, S.; Asad, S.; Hassanzadeh, N. Using Saccostrea cucullata as a biomonitor of heavy metals (Cu, Pb, Zn, Cd, Ni, and Cr) in water and sediments of Qeshm Island, Persian Gulf. Ecopersia 2020, 8, 181–190. [Google Scholar]
- Shirneshan, G.; Riyahi Bakhtiari, A. Accumulation and distribution of Cd, Cu, Pb and Zn in the soft tissue and shell of oysters collected from the northern coast of Qeshm Island, Persian Gulf, Iran. Chem. Speciat. Bioavailab. 2012, 24, 129–138. [Google Scholar] [CrossRef]
- Mao, L.; Liu, X.; Wang, Z.; Wang, B.; Lin, C.; Xin, M.; Zhang, B.T.; Wu, T.; He, M.; Ouyang, W. Trophic transfer and dietary exposure risk of mercury in aquatic organisms from urbanized coastal ecosystems. Chemosphere 2021, 281, 130836. [Google Scholar] [CrossRef]
- Wang, X.; Zhong, W.; Xiao, B.; Liu, Q.; Yang, L.; Covaci, A.; Zhu, L. Bioavailability and biomagnification of organophosphate esters in the food web of Taihu Lake, China: Impacts of chemical properties and metabolism. Environ. Int. 2019, 125, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Department of Environment, Malaysia. Malaysia Environmental Quality Report 2006. Available online: https://enviro2.doe.gov.my/ekmc/wp-content/uploads/2016/08/1403318138-Environmental_Quality_Report_(EQR)_2006.pdf (accessed on 22 July 2022).
- Gagnon, C.; Turcotte, P.; Gagné, F.; Smyth, S.A. Occurrence and size distribution of silver nanoparticles in wastewater effluents from various treatment processes in Canada. Environ. Sci. Pollut. Res. 2021, 28, 65952–65959. [Google Scholar] [CrossRef]
- Deycard, V.N.; Schafer, J.; Petit, J.C.J.; Coynel, A.; Lanceleur, L.; Dutruch, L.; Bossy, C.; Ventura, A.; Blanc, G. Inputs, dynamics and potential impacts of silver (Ag) from urban wastewater to a highly turbid estuary (SW France). Chemosphere 2017, 167, 501–511. [Google Scholar] [CrossRef] [PubMed]
- Syafiuddin, A.; Salmiati, S.; Hadibarata, T.; Kueh, A.B.H.; Salim, M.R.; Zaini, M.A.A. Silver Nanoparticles in the Water Environment in Malaysia: Inspection, characterization, removal, modeling, and future perspective. Sci. Rep. 2018, 8, 986. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Klaine, S.J.; Alvarez, P.J.; Batley, G.E.; Fernandes, T.F.; Handy, R.D.; Lyon, D.Y.; Mahendra, S.; McLaughlin, M.J.; Lead, J.R. Nanomaterials in the environment: Behavior, fate, bioavailability, and effects. Environ. Toxicol. Chem. 2008, 27, 1825–1851. [Google Scholar] [CrossRef]
- Benn, T.M.; Westerhoff, P. Nanoparticle Silver Released into Water from Commercially Available Sock Fabrics. Environ. Sci. Technol. 2008, 42, 4133–4139. [Google Scholar] [CrossRef]
- Ribeiro, F.; Gallego-Urrea, J.A.; Jurkschat, K.; Crossley, A.; Hassellov, M.; Taylor, C.; Soares, A.M.; Loureiro, S. Silver nanoparticles and silver nitrate induce high toxicity to Pseudokirchneriella subcapitata, Daphnia magna and Danio rerio. Sci. Total Environ. 2014, 466–467, 232–241. [Google Scholar] [CrossRef]
- Purcell, T.W.; Peters, J.J. Sources of silver in the environment. Environ. Toxicol. Chem. 1998, 17, 539–546. [Google Scholar] [CrossRef]
- Wimmer, A.; Markus, A.A.; Schuster, M. Silver Nanoparticle Levels in River Water: Real Environmental Measurements and Modeling Approaches—A Comparative Study [Rapid-communication]. Environ. Sci. Technol. Lett. 2019, 6, 353–358. [Google Scholar] [CrossRef]
- Jeon, C. Adsorption behavior of silver ions from industrial wastewater onto immobilized crab shell beads. J. Ind. Eng. Chem. 2015, 32, 195–200. [Google Scholar] [CrossRef]
- Blaser, S.A.; Scheringer, M.; MacLeod, M.; Hungerbuhler, K. Estimation of cumulative aquatic exposure and risk due to silver: Contribution of nano-functionalized plastics and textiles. Sci. Total Environ. 2008, 390, 396–409. [Google Scholar] [CrossRef] [PubMed]
- Doolette, C.L.; McLaughlin, M.J.; Kirby, J.K.; Navarro, D.A. Bioavailability of silver and silver sulfide nanoparticles to lettuce (Lactuca sativa): Effect of agricultural amendments on plant uptake. J. Hazard. Mater. 2015, 300, 788–795. [Google Scholar] [CrossRef]
- Navarro, E.; Piccapietra, F.; Wagner, B.; Marconi, F.; Kaegi, R.; Odzak, N.; Sigg, L.; Behra, R. Toxicity of silver nanoparticles to Chlamydomonas reinhardtii. Environ. Sci. Technol. 2008, 42, 8959–8964. [Google Scholar] [CrossRef]
- Park, H.G.; Kim, J.I.; Chang, K.H.; Lee, B.C.; Eom, I.C.; Kim, P.; Nam, D.H.; Yeo, M.K. Trophic transfer of citrate, PVP coated silver nanomaterials, and silver ions in a paddy microcosm. Environ. Pollut. 2018, 235, 435–445. [Google Scholar] [CrossRef]
- Zhao, J.; Wang, X.; Hoang, S.A.; Bolan, N.S.; Kirkham, M.B.; Liu, J.; Xia, X.; Li, Y. Silver nanoparticles in aquatic sediments: Occurrence, chemical transformations, toxicity, and analytical methods. J. Hazard. Mater. 2021, 418, 126368. [Google Scholar] [CrossRef]
- Caceres-Velez, P.R.; Fascineli, M.L.; Sousa, M.H.; Grisolia, C.K.; Yate, L.; de Souza, P.E.N.; Estrela-Lopis, I.; Moya, S.; Azevedo, R.B. Humic acid attenuation of silver nanoparticle toxicity by ion complexation and the formation of a Ag3+ coating. J. Hazard. Mater. 2018, 353, 173–181. [Google Scholar] [CrossRef] [PubMed]
- Tourinho, P.S.; Van Gestel, C.A.; Lofts, S.; Svendsen, C.; Soares, A.M.; Loureiro, S. Metal-based nanoparticles in soil: Fate, behavior, and effects on soil invertebrates. Environ. Toxicol. Chem. 2012, 31, 1679–1692. [Google Scholar] [CrossRef]
- Cunningham, S.; Joshi, L. Assessment of Exposure of Marine and Freshwater Model Organisms to Metallic Nanoparticles; EPA Research Report 150; Environmental Protection Agency: Johnstown Castle, Ireland, 2015; Available online: https://www.epa.ie/publications/research/water/Research-Report-150.pdf (accessed on 16 June 2022).
- Luoma, S.N. Silver Nanotechnologies and the Environment: Old Problems or New Challenges; Project on Emerging Nanotechnologies Publication 15; Woodrow Wilson International Center for Scholars and PEW Charitable Trusts: Washington, DC, USA, 2008. [Google Scholar]
- European Commission. Directorate-General for Health and Consumers, Opininon on Nanosilver: Safety, Health and Environmental Effects and Role in Antimicrobial Resistance, European Commission. 2014. Available online: https://ec.europa.eu/health/scientific_committees/emerging/docs/scenihr_o_039.pdf (accessed on 16 June 2022).
- Conde-González, J.; Peña-Méndez, E.; Rybáková, S.; Pasán, J.; Ruiz-Pérez, C.; Havel, J. Adsorption of silver nanoparticles from aqueous solution on copper-based metal organic frameworks (HKUST1). Chemosphere 2016, 150, 659–666. [Google Scholar] [CrossRef] [PubMed]
- Duran, N.; Silveira, C.P.; Durán, M.; Martinez, D.S.T. Silver nanoparticle protein corona and toxicity: A mini-review. J. Nanobiotechnol. 2015, 13, 55. [Google Scholar] [CrossRef] [Green Version]
- Lowry, G.V.; Gregory, K.B.; Apte, S.C.; Lead, J.R. Transformations of Nanomaterials in the Environment. Environ. Sci. Technol. 2012, 46, 6893–6899. [Google Scholar] [CrossRef] [PubMed]
- Button, M.; Auvinen, H.; Van Koetsem, F.; Hosseinkhani, B.; Rousseau, D.; Weber, K.P.; Du Laing, G. Susceptibility of constructed wetland microbial communities to silver nanoparticles: A microcosm study. Ecol. Eng. 2016, 97, 476–485. [Google Scholar] [CrossRef]
- Ratte, H.T. Bioaccumulation and Toxicity of Silver Compounds: A Review. Environ. Toxicol. Chem. 1999, 18, 89–108. [Google Scholar] [CrossRef]
- Ma, X.; Geiser-Lee, J.; Deng, Y.; Kolmakov, A. Interactions between engineered nanoparticles (ENPs) and plants: Phytotoxicity, uptake and accumulation. Sci. Total Environ. 2010, 408, 3053–3061. [Google Scholar] [CrossRef] [PubMed]
- Geisler-Lee, J.; Wang, Q.; Yao, Y.; Zhang, W.; Geisler, M.; Li, K.; Huang, Y.; Chen, Y.; Kolmakov, A.; Ma, X. Phytotoxicity, accumulation and transport of silver nanoparticles by Arabidopsis thaliana. Nanotoxicology 2013, 7, 323–337. [Google Scholar] [CrossRef]
- Carpita, N.C.; Gibeaut, D.M. Structural models of primary cell walls in flowering plants: Consistency of molecular structure with the physical properties of the walls during growth. Plant J. 1993, 3, 1–30. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, D.K.; Tripathi, A.; Singh, S.; Singh, Y.; Vishwakarma, K.; Yadav, G.; Sharma, S.; Singh, V.K.; Mishra, R.K.; Upadhyay, R.G. Uptake, accumulation and toxicity of silver nanoparticle in autotrophic plants, and heterotrophic microbes: A concentric review. Front. Microbiol. 2017, 8, 7. [Google Scholar] [CrossRef] [Green Version]
- Larue, C.; Castillo-Michel, H.; Sobanska, S.; Cécillon, L.; Bureau, S.; Barthès, V.; Ouerdane, L.; Carrière, M.; Sarret, G. Foliar exposure of the crop Lactuca sativa to silver nanoparticles: Evidence for internalization and changes in Ag speciation. J. Hazard. Mater. 2014, 264, 98–106. [Google Scholar] [CrossRef]
- Geisler-Lee, J.; Brooks, M.; Gerfen, J.R.; Wang, Q.; Fotis, C.; Sparer, A.; Ma, X.; Berg, R.H.; Geisler, M. Reproductive toxicity and life history study of silver nanoparticle effect, uptake and transport in Arabidopsis thaliana. Nanomaterials 2014, 4, 301–318. [Google Scholar] [CrossRef] [Green Version]
- Li, L.; Hartmann, G.; Döblinger, M. Schuster Quantification of nanoscale silver particles removal and release from municipal wastewater treatment plants in Germany. Environ. Sci. Technol. 2013, 47, 7317–7323. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Lombi, E.; Sun, S.; Scheckel, K.G.; Malysheva, A.; McKenna, B.A.; Menzies, N.W.; Zhao, F.J.; Kopittke, P.M. Characterizing the uptake, accumulation and toxicity of silver sulfide nanoparticles in plants. Environ. Sci. Nano 2017, 4, 448–460. [Google Scholar] [CrossRef] [PubMed]
- Yan, A.; Chen, Z. Impacts of Silver Nanoparticles on Plants: A Focus on the Phytotoxicity and Underlying Mechanism. Int. J. Mol. Sci. 2019, 20, 1003. [Google Scholar] [CrossRef] [PubMed]
- Schafer, S.; Buchmeier, G.; Claus, E.; Duester, L.; Heininger, P.; Körner, A.; Mayer, P.; Paschke, A.; Rauert, C.; Reifferscheid, G.; et al. Bioaccumulation in aquatic systems: Methodological approaches, monitoring and assessment. Environ. Sci. Eur. 2015, 27, 5. [Google Scholar] [CrossRef]
- Bawuro, A.A.; Voegborlo, R.B.; Adimado, A.A. Bioaccumulation of heavy metals in some tissues of fish in Lake Geriyo, Adamawa State, Nigeria. J. Environ. Public Health 2018, 2018, 1854892. [Google Scholar] [CrossRef] [Green Version]
- Jezierska, B.; Witeska, M. The metal uptake and accumulation in fish living in polluted waters. In Soil and Water Pollution Monitoring, Protection and Remediation; Twardowska, I., Allen, H.E., Häggblom, M.M., Stefaniak, S., Eds.; NATO Science Series; Springer: Dordrecht, The Netherlands, 2006; Volume 69. [Google Scholar]
- Yamazaki, M.; Tanizaki, Y.; Shimokawa, T. Silver and other trace elements in a freshwater fish, Carasius auratus langsdorfii, from the Asakawa River in Tokyo, Japan. Environ. Pollut. 1996, 94, 83–90. [Google Scholar] [CrossRef]
- Afifi, M.; Saddick, S.; Zinada, O.A.A. Toxicity of silver nanoparticles on the brain of Oreochromis niloticus and Tilapia zillii. Saudi J. Biol. Sci. 2016, 23, 754–760. [Google Scholar] [CrossRef] [Green Version]
- Yin, Y.; Liu, J.; Jiang, G. Sunlight-induced reduction of ionic Ag and Au to metallic nanoparticles by dissolved organic matter. ACS Nano 2012, 6, 7910–7919. [Google Scholar] [CrossRef]
- Zou, X.; Shi, J.; Zhang, H. Morphological evolution and reconstruction of silver nanoparticles in aquatic environments: The roles of natural organic matter and light irradiation. J. Hazard. Mater. 2015, 292, 61–69. [Google Scholar] [CrossRef]
- Geranio, L.; Heuberger, M.; Nowack, B. The behavior of silver nanotextiles during washing. Environ. Sci. Technol. 2009, 43, 8113–8118. [Google Scholar] [CrossRef] [Green Version]
- Kaegi, R.; Sinnet, B.; Zuleeg, S.; Hagendorfer, H.; Mueller, E.; Vonbank, R.; Boller, M.; Burkhardt, M. Release of silver nanoparticles from outdoor facades. Environ. Pollut. 2010, 158, 2900–2905. [Google Scholar] [CrossRef] [PubMed]
- Unrine, J.M.; Colman, B.P.; Bone, A.J.; Gondikas, A.P.; Matson, C.W. Biotic and abiotic interactions in aquatic microcosms determine fate and toxicity of Ag nanoparticles. Part 1. Aggregation and dissolution. Environ. Sci. Technol. 2012, 46, 6915–6924. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Albee, B.; Alemayehu, M.; Diaz, R.; Ingahm, L.; Kamal, S.; Rodriguez, M.; Bishnoi, S.W. Comparative toxicity study of Ag, Au, and Ag- Au bimetallic nanoparticles on Daphnia magna. Anal Bioanal. Chem. 2010, 398, 689–700. [Google Scholar] [CrossRef]
- Tejamaya, M.; Römer, I.; Merrifield, R.C.; Lead, J.R. Stability of citrate, PVP, and PEG coated silver nanoparticles in ecotoxicology media. Environ. Sci. Technol. 2012, 46, 7011–7017. [Google Scholar] [CrossRef]
- Cheng, Y.; Yin, L.; Lin, S.; Wiesner, M.; Bernhardt, E.; Liu, J. Toxicity reduction of polymer-stabilized silver nanoparticles by sunlight. J. Phys. Chem. C 2011, 115, 4425–4432. [Google Scholar] [CrossRef]
- Ozdak, N.; Kistler, D.; Behra, R.; Sigg, L. Dissolution of metal and metal oxide nanoparticles in aqueous media. Environ. Poll. 2014, 191, 132–138. [Google Scholar]
- Yin, Y.; Shen, M.; Zhou, X.; Yu, S.; Chao, J.; Liu, J.; Jiang, G. Photoreduction and stabilization capability of molecular weight fractionated natural organic matter in transformation of silver ion to metallic nanoparticle. Environ. Sci. Technol. 2014, 48, 9366–9373. [Google Scholar] [CrossRef]
- Adegboyega, N.F.; Sharma, V.K.; Siskova, K.; Zboril, R.; Sohn, M.; Schultz, B.J.; Banerjee, S. Interactions of aqueous Agþ with fulvic acids: Mechanisms of silver nanoparticle formation and investigation of stability. Environ. Sci. Technol. 2013, 47, 757–764. [Google Scholar] [CrossRef]
- Yu, S.; Yin, Y.; Zhou, X.; Dong, L.; Liu., J. Transformation kinetics of silver nanoparticles and silver ions in aquatic environments revealed by double stable isotope labeling 75,76. Environ. Sci. Nano 2016, 3, 883–893. [Google Scholar] [CrossRef]
- Zou, X.; Li, P.; Lou, J.; Fu, X.; Zhang, H. Stability of single dispersed silver nanoparticles in natural and synthetic freshwaters: Effects of dissolved oxygen. Environ. Pollut. 2017, 230, 674–682. [Google Scholar] [CrossRef]
- Li, M.; Wang, P.; Dang, F.; Zhou, D.M. The transformation and fate of silver nanoparticles in paddy soil: Effects of soil organic matter and redox conditions. Environ. Sci. Nano 2017, 4, 919–928. [Google Scholar] [CrossRef]
- Nowack, B.; Bucheli, T.D. Occurrence, behavior and effects of nanoparticles in the environment. Environ. Pollut. 2007, 150, 5–22. [Google Scholar] [CrossRef]
- Tangaa, S.R.; Selck, H.; Winther-Nielsen, M.; Khan, F.R. Trophic transfer of metal-based nanoparticles in aquatic environments: A review and recommendations for future research focus. Environ. Sci. Nano 2016, 3, 966–981. [Google Scholar] [CrossRef] [Green Version]
- Bouldin, J.L.; Ingle, T.M.; Sengupta, A.; Alexander, R.; Hannigan, R.E.; Buchanan, R.A. Aqueous toxicity and food chain transfer of quantum dots™ in freshwater algae and Ceriodaphnia dubia. Environ. Toxicol. Chem. 2008, 27, 1958–1963. [Google Scholar] [CrossRef] [PubMed]
- Saha, N.; Gupta, S.D. Low-dose toxicity of biogenic silver nanoparticles fabricated by Swertia chirata on root tips and flower buds of Allium cepa. J. Hazard Mater. 2017, 330, 18–28. [Google Scholar] [CrossRef] [PubMed]
- Lemon, G.D.; Posluszny, U.; Husband, B.C. Potential and realized rates of vegetative reproduction in Spirodelapolyrhiza, Lemna minor, and Wolffia borealis. Aquat Bot. 2001, 70, 79–87. [Google Scholar] [CrossRef]
- Pokhrel, L.R.; Dubey, B.; Scheuerman, P.R. Impacts of select organic ligands on the colloidal stability, dissolution dynamics, and toxicity of silver nanoparticles. Environ. Sci. Technol. 2013, 47, 12877–12885. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Huang, J.; Liang, L.; Yao, L.; Fang, T. Dual impact of dissolved organic matter on cytotoxicity of PVP-Ag NPs to Escherichia coli: Mitigation and intensification. Chemosphere 2019, 214, 754–763. [Google Scholar] [CrossRef] [PubMed]
- Holbrook, R.D.; Murphy, K.E.; Morrow, J.B.; Cole, K.D. Trophic transfer of nanoparticles in a simplified invertebrate food web. Nat. Nanotechnol. 2008, 3, 352–355. [Google Scholar] [CrossRef]
- Hedayati, S.A.; Farsani, H.G.; Naserabad, S.S.; Hoseinifar, S.H.; Van Doan, H. Protective effect of dietary vitamin E on immunological and biochemical induction through silver nanoparticles (AgNPs) inclusion in diet and silver salt (AgNO3) exposure on Zebrafish (Danio rerio). Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2019, 222, 100–107. [Google Scholar] [CrossRef]
- Ellis, L.J.A.; Baalousha, M.; Valsami-Jones, E.; Lead, J.R. Seasonal variability of natural water chemistry affects the fate and behaviour of silver nanoparticles. Chemosphere 2018, 191, 616–625. [Google Scholar] [CrossRef] [PubMed]
- Favari, L.; Lopez, E.; Martinez-Tabche, L.; DıazPardo, E. Effect of insecticides on plankton and fish of Ignacio Ramirez reservoir (Mexico): A biochemical and biomagnifications study. Ecotoxicol. Environ. Saf. 2002, 51, 177–186. [Google Scholar] [CrossRef] [PubMed]







| Sampling Station | Scientific Name | English Name |
|---|---|---|
| Bandar Putra | Anabas testudineus | Climbing Perch |
| Oreochromis niloticus | Nile Tilapia | |
| Lee Rubber | Clarias macrocephalus | Catfish |
| Oreochromis niloticus | Nile Tilapia | |
| Pangasianodon hypophthalmus | Silver Catfish | |
| Channa striata | Snakehead Murrel | |
| Barbus schwanenfeldii | Tinfoil Barb | |
| Impian Emas | Clarias macrocephalus | Catfish |
| Pangasianodon hypophthalmus | Silver Catfish | |
| Pengkalan Rinting | Clarias macrocephalus | Catfish |
| Oreochromis mossambicus | Mozambique Tilapia | |
| Pangasianodon hypophthalmus | Silver Catfish |
| Sample | Sample Weight (g) | Acids Used |
|---|---|---|
| Water | 5 | 7 mL HNO3 65% 1 mL H2O2 30% |
| Sediment | 0.25 | 6 mL HClO4 65% 6 mL HNO3 65% 1 mL H2O2 30% |
| Plants | 0.5 | 7 mL HNO3 65% 1 mL H2O2 30% |
| Fish | 0.5 | 7 mL HNO3 65% 1 mL H2O2 30% |
| Step | Time (min) | Temp. (T1) | Temp (T2) |
|---|---|---|---|
| 1 | 15 | 200 °C | 110 °C |
| 2 | 15 | 200 °C | 100 °C |
| Sampling Station | BAF | BSAF | |
|---|---|---|---|
| Average Ag Concentration in Sampled Surface Water 1 | Relative Limit of Ag Concentration 2 | ||
| Bandar Putra | 7570.95 | 1,476,334.75 | 6.03 |
| Lee Rubber | 3733.11 | 1,530,575.59 | 10.09 |
| Impian Emas | 703.49 | 140,698.66 | 1.01 |
| Pengkalan Rinting | 2385.52 | 131,203.74 | 2.50 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mat Lazim, Z.; Salmiati, S.; Marpongahtun, M.; Arman, N.Z.; Mohd Haniffah, M.R.; Azman, S.; Yong, E.L.; Salim, M.R. Distribution of Silver (Ag) and Silver Nanoparticles (AgNPs) in Aquatic Environment. Water 2023, 15, 1349. https://doi.org/10.3390/w15071349
Mat Lazim Z, Salmiati S, Marpongahtun M, Arman NZ, Mohd Haniffah MR, Azman S, Yong EL, Salim MR. Distribution of Silver (Ag) and Silver Nanoparticles (AgNPs) in Aquatic Environment. Water. 2023; 15(7):1349. https://doi.org/10.3390/w15071349
Chicago/Turabian StyleMat Lazim, Zainab, Salmiati Salmiati, Marpongahtun Marpongahtun, Nor Zaiha Arman, Mohd Ridza Mohd Haniffah, Shamila Azman, Ee Ling Yong, and Mohd Razman Salim. 2023. "Distribution of Silver (Ag) and Silver Nanoparticles (AgNPs) in Aquatic Environment" Water 15, no. 7: 1349. https://doi.org/10.3390/w15071349






