Reduction of Cr(VI) by Synergistic Effects of Iron-Rich Biochar and Pseudomonas aeruginosa
Abstract
:1. Introduction
2. Materials and Methods
2.1. Iron-Rich Biochar Preparation
2.2. The Cultivation of PA
2.3. Experimental Procedures for the Reduction of Cr(VI) to Cr(III)
2.4. The Different Processes of the Potential Synergistic Reduction of Cr(VI) by Iron-Rich Biochar and PA
2.4.1. Preparations of Cell Components and Chromium Reductase Activity Assays
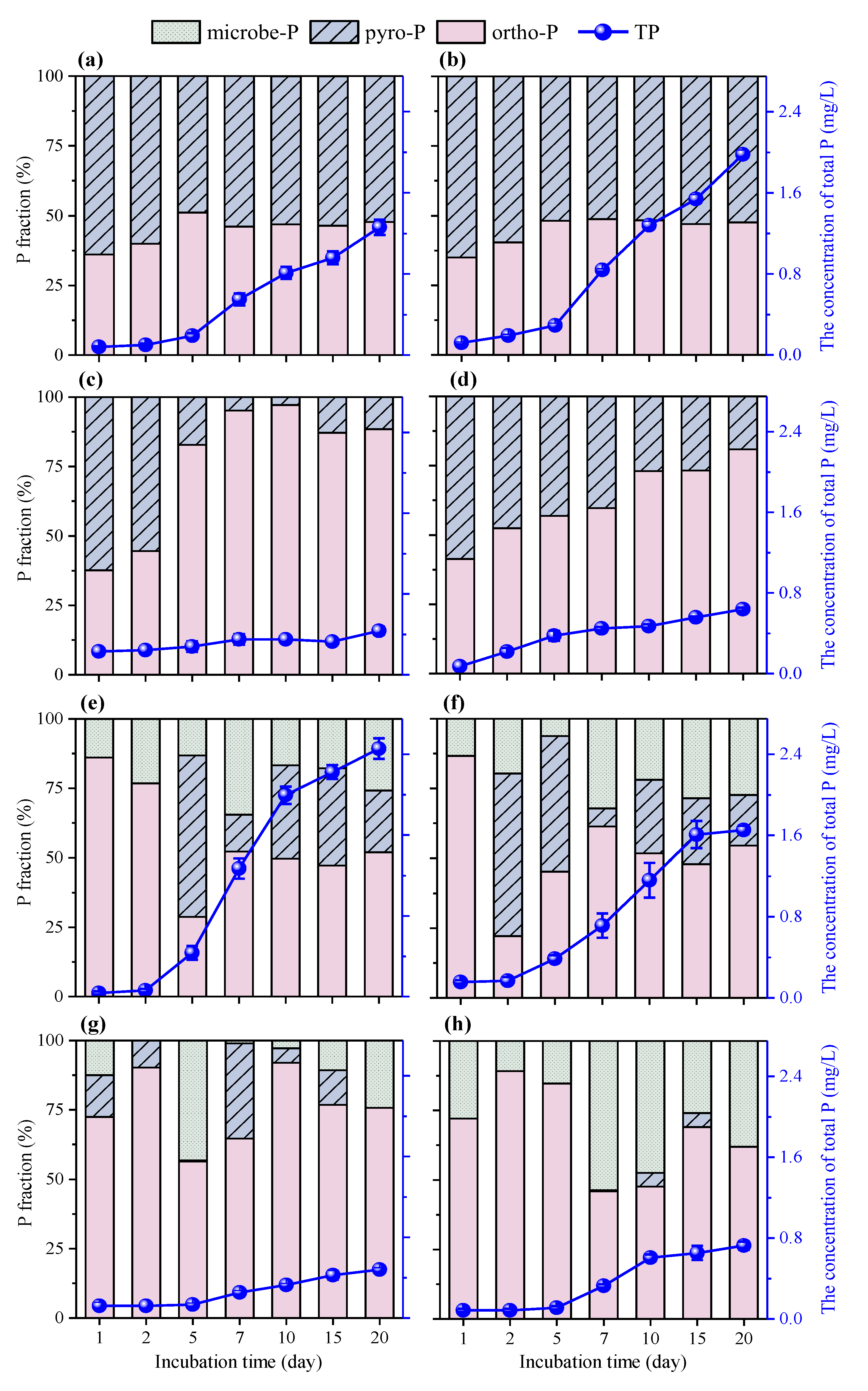
2.4.2. The Changes of Phosphorus Concentration and Species
2.4.3. The Impacts of Different Iron Species on the Reduction of Cr(VI) to Cr(III)
2.5. Analytical Methods
2.6. Statistical Analysis
3. Results and Discussion
3.1. Reduction of Cr (VI) to Cr(III) by Iron-Rich Biochar and PA
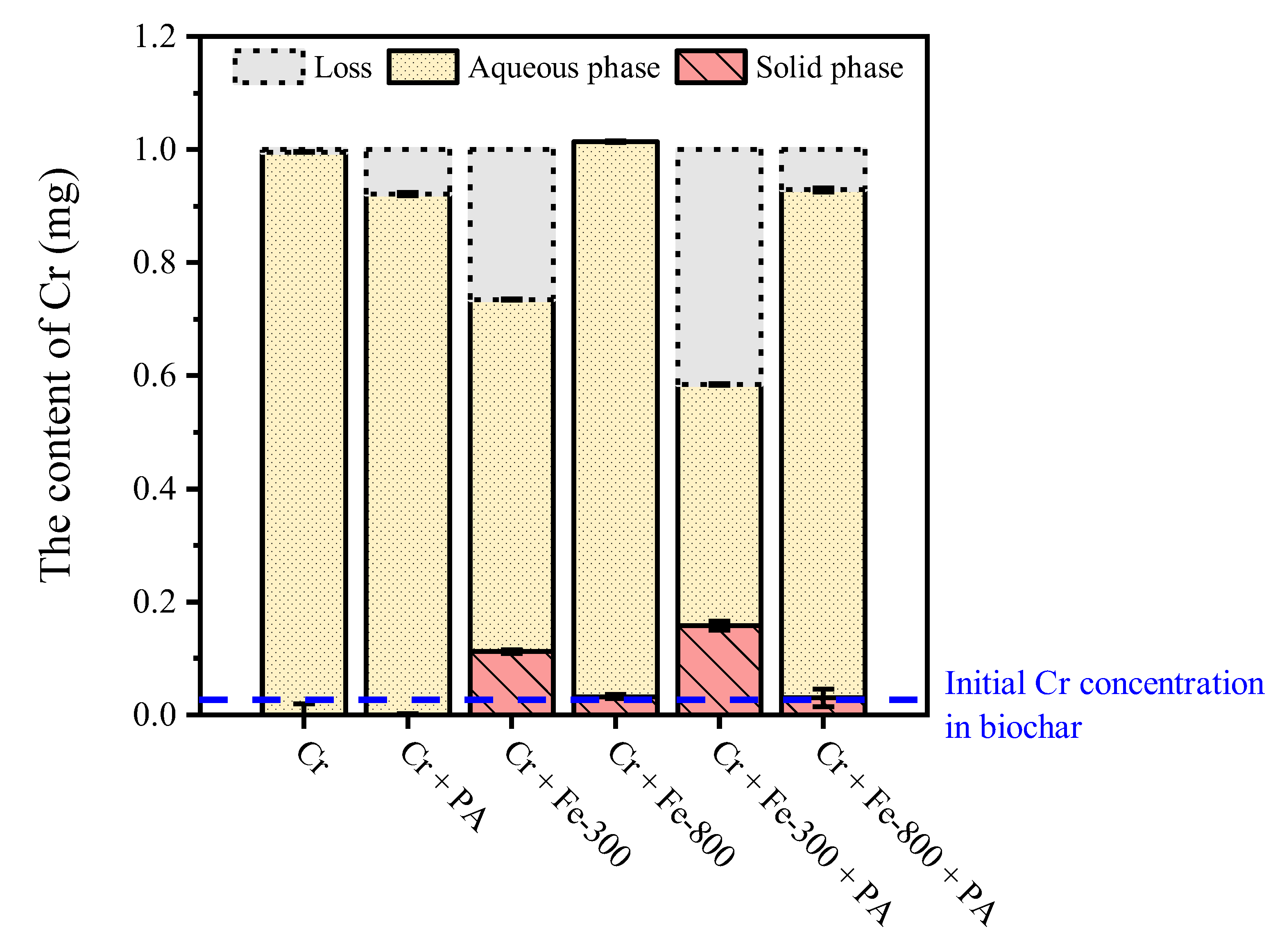
3.1.1. Changes of Total Chromium Concentration
3.1.2. The Changes of Cr(VI) and Cr(III)
3.2. Different Processes of Iron-Rich Biochar and PA Synergistic Reduction of Cr(VI)
3.2.1. The Cr(VI) Reduction Effect of Protein Extracts from Different Cellular Components
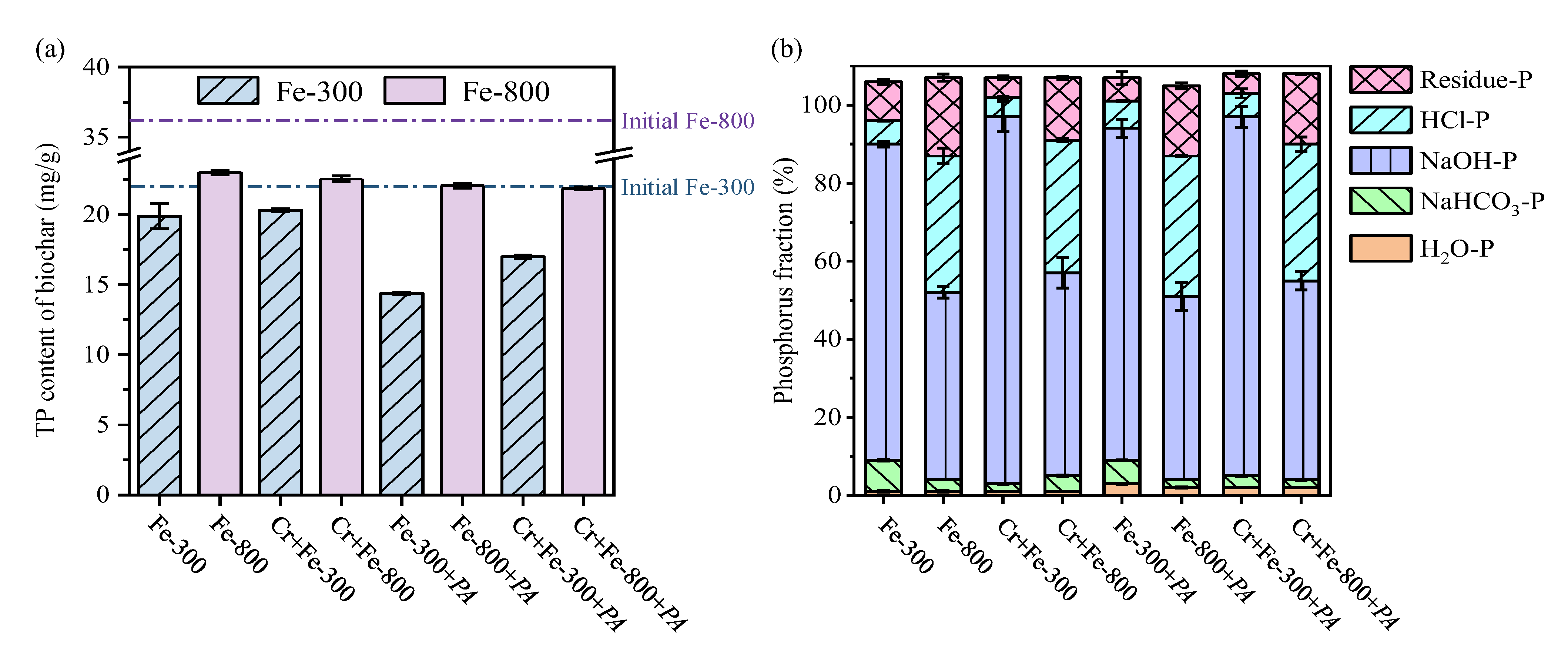
3.2.2. Phosphorus Release from Biochar by PA
3.2.3. The Impacts of Different Iron Species on the Reduction of Cr(VI) to Cr(III)
3.2.4. Morphology Characteristics of Biochar and PA

4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Prasad, S.; Yadav, K.K.; Kumar, S.; Gupta, N.; Cabral-Pinto, M.M.S.; Rezania, S.; Radwan, N.; Alam, J. Chromium contamination and effect on environmental health and its remediation: A sustainable approaches. J. Environ. Manag. 2021, 285, 112174. [Google Scholar] [CrossRef] [PubMed]
- Ukhurebor, K.E.; Aigbe, U.O.; Onyancha, R.B.; Nwankwo, W.; Osibote, O.A.; Paumo, H.K.; Ama, O.M.; Adetunji, C.O.; Siloko, I.U. Effect of hexavalent chromium on the environment and removal techniques: A review. J. Environ. Manag. 2021, 280, 111809. [Google Scholar] [CrossRef] [PubMed]
- Jin, W.; Du, H.; Zheng, S.; Zhang, Y. Electrochemical processes for the environmental remediation of toxic Cr(VI): A review. Electrochim. Acta 2016, 191, 1044–1055. [Google Scholar] [CrossRef]
- Sinha, V.; Pakshirajan, K.; Chaturvedi, R. Chromium tolerance, bioaccumulation and localization in plants: An overview. J. Environ. Manag. 2018, 206, 715–730. [Google Scholar] [CrossRef] [PubMed]
- Haney, J.T.; Erraguntla, N.; Sielken, R.L.; Valdez-Flores, C. Development of an inhalation unit risk factor for hexavalent chromium. Regul. Toxicol. Pharmacol. 2014, 68, 201–211. [Google Scholar] [CrossRef] [Green Version]
- Chen, J.; Tian, Y. Hexavalent chromium reducing bacteria: Mechanism of reduction and characteristics. Environ. Sci. Pollut. Res. 2021, 28, 20981–20997. [Google Scholar] [CrossRef]
- Uddin, M.J.; Jeong, Y.; Lee, W. Microbial fuel cells for bioelectricity generation through reduction of hexavalent chromium in wastewater: A review. Int. J. Hydrogen. Energ. 2021, 46, 11458–11481. [Google Scholar] [CrossRef]
- Veer, S.; Vishal, M. Sustainable reduction of Cr (VI) and its elemental mapping on chitosan coated citrus limetta peels biomass in synthetic wastewater. Sep. Sci. Technol. 2022, 57, 1609–1626. [Google Scholar]
- Jiang, B.; Niu, Q.; Li, C.; Oturan, N.; Oturan, M.A. Outstanding performance of electro-Fenton process for efficient decontamination of Cr(III) complexes via alkaline precipitation with no accumulation of Cr(VI): Important roles of iron species. Appl. Catal. B Environ. 2020, 272, 119002. [Google Scholar] [CrossRef]
- Religa, P.; Kowalik-Klimczak, A.; Gierycz, P. Study on the behavior of nanofiltration membranes using for chromium(III) recovery from salt mixture solution. Desalination 2013, 315, 115–123. [Google Scholar] [CrossRef]
- Sun, P.; Wang, Z.; An, S.; Zhao, J.; Yan, Y.; Zhang, D.; Wu, Z.; Shen, B.; Lyu, H. Biochar-supported nZVI for the removal of Cr(VI) from soil and water: Advances in experimental research and engineering applications. J. Environ. Manag. 2022, 316, 115211. [Google Scholar] [CrossRef]
- Xing, X.; Alharbi, N.S.; Ren, X.; Chen, C. A comprehensive review on emerging natural and tailored materials for chromium contaminated water treatment and environmental remediation. J. Environ. Chem. Eng. 2022, 10, 107325. [Google Scholar] [CrossRef]
- Gopinath, A.; Divyapriya, G.; Srivastava, V.; Laiju, A.R.; Nidheesh, P.V.; Kumar, M.S. Conversion of sewage sludge into biochar: A potential resource in water and wastewater treatment. Environ. Res. 2021, 194, 110656. [Google Scholar] [CrossRef]
- Qiu, Y.; Zhang, Q.; Gao, B.; Li, M.; Fan, Z.; Sang, W.; Hao, H.; Wei, X. Removal mechanisms of Cr(VI) and Cr(III) by biochar supported nanosized zero-valent iron: Synergy of adsorption, reduction and transformation. Environ. Pollut. 2020, 265, 115018. [Google Scholar] [CrossRef]
- Chen, T.; Zhou, Z.; Xu, S.; Wang, H.; Lu, W. Adsorption behavior comparison of trivalent and hexavalent chromium on biochar derived from municipal sludge. Bioresour. Technol. 2015, 190, 388–394. [Google Scholar] [CrossRef]
- Diao, Z.; Du, J.; Jiang, D.; Kong, L.; Huo, W.; Liu, C.; Wu, Q.; Xu, X. Insights into the simultaneous removal of Cr6+ and Pb2+ by a novel sewage sludge-derived biochar immobilized nanoscale zero valent iron: Coexistence effect and mechanism. Sci. Total Environ. 2018, 642, 505–515. [Google Scholar] [CrossRef]
- Wang, H.; Xiao, K.; Yang, J.; Yu, Z.; Yu, W.; Xu, Q.; Wu, Q.; Liang, S.; Hu, J.; Hou, H.; et al. Phosphorus recovery from the liquid phase of anaerobic digestate using biochar derived from iron-rich sludge: A potential phosphorus fertilizer. Water Res. 2020, 174, 115629. [Google Scholar] [CrossRef]
- Costa, R.C.C.; Moura, F.C.C.; Oliveira, P.E.F.; Magalhães, F.; Ardisson, J.D.; Lago, R.M. Controlled reduction of red mud waste to produce active systems for environmental applications: Heterogeneous Fenton reaction and reduction of Cr(VI). Chemosphere 2010, 78, 1116–1120. [Google Scholar] [CrossRef]
- Wang, X.; Gu, L.; Zhou, P.; Zhu, N.; Li, C.; Tao, H.; Wen, H.; Zhang, D. Pyrolytic temperature dependent conversion of sewage sludge to carbon catalyst and their performance in persulfate degradation of 2-Naphthol. Chem. Eng. J. 2017, 324, 203–215. [Google Scholar] [CrossRef]
- Qin, J.; Li, Q.; Liu, Y.; Niu, A.; Lin, C. Biochar-driven reduction of As(V) and Cr(VI): Effects of pyrolysis temperature and low-molecular-weight organic acids. Ecotoxicol. Environ. Saf. 2020, 201, 110873. [Google Scholar] [CrossRef]
- Hashem, M.A.; Payel, S.; Mim, S.S.; Hasan, M.A.; Nur-A-Tomal, M.S.; Rahman, M.A.; Sarker, M.I. Chromium adsorption on surface activated biochar made from tannery liming sludge: A waste-to-wealth approach. Water Sci. Eng. 2022, 15, 328–336. [Google Scholar] [CrossRef]
- Xu, Z.; Xu, X.; Zhang, Y.; Yu, Y.; Cao, X. Pyrolysis-Temperature depended electron donating and mediating mechanisms of biochar for Cr(VI) reduction. J. Hazard. Mater. 2020, 388, 121794. [Google Scholar] [CrossRef] [PubMed]
- Priester, J.H.; Olson, S.G.; Webb, S.M.; Neu, M.P.; Hersman, L.E.; Holden, P.A. Enhanced exopolymer production and chromium stabilization in Pseudomonas putida unsaturated biofilms. Appl. Environ. Microb. 2006, 72, 1988–1996. [Google Scholar] [CrossRef] [Green Version]
- Wani, P.A.; Wani, J.A.; Wahid, S. Recent advances in the mechanism of detoxification of genotoxic and cytotoxic Cr (VI) by microbes. J. Environ. Chem. Eng. 2018, 6, 3798–3807. [Google Scholar] [CrossRef]
- Sathishkumar, K.; Murugan, K.; Benelli, G.; Higuchi, A.; Rajasekar, A. Bioreduction of hexavalent chromium by Pseudomonas stutzeri L1 and Acinetobacter baumannii L2. Ann. Microbiol. 2017, 67, 91–98. [Google Scholar] [CrossRef]
- Song, H.; Liu, Y.; Xu, W.; Zeng, G.; Aibibu, N.; Xu, L.; Chen, B. Simultaneous Cr(VI) reduction and phenol degradation in pure cultures of Pseudomonas aeruginosa CCTCC AB91095. Bioresour. Technol. 2009, 100, 5079–5084. [Google Scholar] [CrossRef]
- Kang, C.; Wu, P.; Li, L.; Yu, L.; Ruan, B.; Gong, B.; Zhu, N. Cr(VI) reduction and Cr(III) immobilization by resting cells of Pseudomonas aeruginosa CCTCC AB93066: Spectroscopic, microscopic, and mass balance analysis. Environ. Sci. Pollut. Res. 2017, 24, 5949–5963. [Google Scholar] [CrossRef]
- Ackerley, D.F.; Gonzalez, C.F.; Park, C.H.; Blake, R.; Keyhan, M.; Matin, A. Chromate-reducing properties of soluble flavoproteins from Pseudomonas putida and Escherichia coli. Appl. Environ. Microbiol. 2004, 70, 873–882. [Google Scholar] [CrossRef] [Green Version]
- Yu, Z.; Sun, M.; Xiao, K.; Ou, B.; Liang, S.; Hou, H.; Yang, J. Changes of phosphorus species during (hydro) thermal treatments of iron-rich sludge and their solubilization mediated by a phosphate solubilizing microorganism. Sci. Total Environ. 2022, 838, 156612. [Google Scholar] [CrossRef]
- Xiao, K.; Yu, Z.; Wang, H.; Yang, J.; Liang, S.; Hu, J.; Hou, H.; Liu, B. Investigation on emission control of NOx precursors and phosphorus reclamation during pyrolysis of ferric sludge. Sci. Total Environ. 2019, 670, 932–940. [Google Scholar] [CrossRef]
- Qian, T.; Yang, Q.; Jun, D.; Dong, F.; Zhou, Y. Transformation of phosphorus in sewage sludge biochar mediated by a phosphate-solubilizing microorganism. Chem. Eng. J. 2019, 359, 1573–1580. [Google Scholar] [CrossRef]
- Zhang, H.; Lu, H.; Wang, J.; Zhou, J.; Sui, M. Cr(VI) reduction and Cr(III) immobilization by Acinetobacter sp. HK-1 with the assistance of a novel quinone/graphene oxide composite. Environ. Sci. Technol. 2014, 48, 12876–12885. [Google Scholar] [CrossRef] [PubMed]
- He, D.; Zheng, M.; Ma, T.; Li, C.; Ni, J. Interaction of Cr(VI) reduction and denitrification by strain Pseudomonas aeruginosa PCN-2 under aerobic conditions. Bioresour. Technol. 2015, 185, 346–352. [Google Scholar] [CrossRef] [PubMed]
- Puzon, G.J.; Roberts, A.G.; Kramer, D.M.; Xun, L. Formation of soluble organo-chromium(III) complexes after chromate reduction in the presence of cellular organics. Environ. Sci. Technol. 2005, 39, 2811–2817. [Google Scholar] [CrossRef] [PubMed]
- Rajapaksha, A.U.; Alam, M.S.; Chen, N.; Alessi, D.S.; Igalavithana, A.D.; Tsang, D.C.W.; Ok, Y.S. Removal of hexavalent chromium in aqueous solutions using biochar: Chemical and spectroscopic investigations. Sci. Total Environ. 2018, 625, 1567–1573. [Google Scholar] [CrossRef]
- Zhou, B.; Chen, T. Biodegradation of phenol with chromium (VI) reduction by the Pseudomonas sp. strain JF122. Desalination Water Treat. 2016, 57, 3544–3551. [Google Scholar] [CrossRef]
- Li, Y.; Wang, H.; Wu, P.; Yu, L.; Rehman, S.; Wang, J.; Yang, S.; Zhu, N. Bioreduction of hexavalent chromium on goethite in the presence of Pseudomonas aeruginosa. Environ. Pollut. 2020, 265, 114765. [Google Scholar] [CrossRef]
- Zhang, B.; Jiao, W. Biochar facilitated bacterial reduction of Cr(VI) by Shewanella Putrefaciens CN32: Pathways and surface characteristics. Environ. Res. 2022, 214, 113971. [Google Scholar] [CrossRef]
- Xu, X.; Huang, H.; Zhang, Y.; Xu, Z.; Cao, X. Biochar as both electron donor and electron shuttle for the reduction transformation of Cr(VI) during its sorption. Environ. Pollut. 2019, 244, 423–430. [Google Scholar] [CrossRef]
- Viti, C.; Marchi, E.; Decorosi, F.; Giovannetti, L. Molecular mechanisms of Cr(VI) resistance in bacteria and fungi. FEMS Microbiol. Rev. 2014, 38, 633–659. [Google Scholar] [CrossRef] [Green Version]
- Hale, S.E.; Alling, V.; Martinsen, V.; Mulder, J.; Breedveld, G.D.; Cornelissen, G. The sorption and desorption of phosphate-P, ammonium-N and nitrate-N in cacao shell and corn cob biochars. Chemosphere 2013, 91, 1612–1619. [Google Scholar] [CrossRef]
- Yu, L.; Huang, H.; Wang, X.; Li, S.; Feng, N.; Zhao, H.; Huang, X.; Li, Y.; Li, H.; Cai, Q.; et al. Novel phosphate-solubilising bacteria isolated from sewage sludge and the mechanism of phosphate solubilisation. Sci. Total Environ. 2019, 658, 474–484. [Google Scholar] [CrossRef]
- Acevedo, E.; Galindo-Castañeda, T.; Prada, F.; Navia, M.; Romero, H.M. Phosphate-solubilizing microorganisms associated with the rhizosphere of oil palm (Elaeis guineensis Jacq.) in Colombia. Appl. Soil Ecol. 2014, 80, 26–33. [Google Scholar] [CrossRef]
- Yang, S.; Wen, Q.; Chen, Z. Effect of KH2PO4-modified biochar on immobilization of Cr, Cu, Pb, Zn and as during anaerobic digestion of swine manure. Bioresour. Technol. 2021, 339, 125570. [Google Scholar] [CrossRef]
- Liang, J.; Huang, X.; Yan, J.; Li, Y.; Zhao, Z.; Liu, Y.; Ye, J.; Wei, Y. A review of the formation of Cr(VI) via Cr(III) oxidation in soils and groundwater. Sci. Total Environ. 2021, 774, 145762. [Google Scholar] [CrossRef]
- Su, Y.; Yuan, S.; Guo, Y.; Tan, Y.; Mao, H.; Cao, Y.; Chen, Y. Highly efficient and sustainable removal of Cr(VI) in aqueous solutions by photosynthetic bacteria supplemented with phosphor salts. Chemosphere 2021, 283, 131031. [Google Scholar] [CrossRef]
- Adhikari, S.; Gascó, G.; Méndez, A.; Surapaneni, A.; Jegatheesan, V.; Shah, K.; Paz-Ferreiro, J. Influence of pyrolysis parameters on phosphorus fractions of biosolids derived biochar. Sci. Total Environ. 2019, 695, 133846. [Google Scholar] [CrossRef]
- Figueiredo, C.C.d.; Reis, A.d.S.P.J.; Araujo, A.S.d.; Blum, L.E.B.; Shah, K.; Paz-Ferreiro, J. Assessing the potential of sewage sludge-derived biochar as a novel phosphorus fertilizer: Influence of extractant solutions and pyrolysis temperatures. Waste Manag. 2021, 124, 144–153. [Google Scholar] [CrossRef]
- Wang, Z.; Chen, H.; Zhu, Z.; Xing, S.; Wang, S.; Chen, B. Low-Temperature straw biochar: Sustainable approach for sustaining higher survival of B. megaterium and managing phosphorus deficiency in the soil. Sci. Total Environ. 2022, 830, 154790. [Google Scholar] [CrossRef]
- Mortazavian, S.; An, H.; Chun, D.; Moon, J. Activated carbon impregnated by zero-valent iron nanoparticles (AC/nZVI) optimized for simultaneous adsorption and reduction of aqueous hexavalent chromium: Material characterizations and kinetic studies. Chem. Eng. J. 2018, 353, 781–795. [Google Scholar] [CrossRef]
- Zhang, S.; Lyu, H.; Tang, J.; Song, B.; Zhen, M.; Liu, X. A novel biochar supported CMC stabilized nano zero-valent iron composite for hexavalent chromium removal from water. Chemosphere 2019, 217, 686–694. [Google Scholar] [CrossRef] [PubMed]
- Pan, J.; Jiang, J.; Xu, R. Removal of Cr(VI) from aqueous solutions by Na2SO3/FeSO4 combined with peanut straw biochar. Chemosphere 2014, 101, 71–76. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Ye, X.; Chen, D.; Xiao, W.; Zhao, S.; Li, J.; Li, H. Chromium(VI) removal from synthetic solution using novel zero-valent iron biochar composites derived from iron-rich sludge via one-pot synthesis. J. Water Process 2022, 47, 102720. [Google Scholar] [CrossRef]
- Zheng, X.; Xu, W.; Dong, J.; Yang, T.; Shangguan, Z.; Qu, J.; Li, X.; Tan, X. The effects of biochar and its applications in the microbial remediation of contaminated soil: A review. J. Hazard. Mater. 2022, 438, 129557. [Google Scholar] [CrossRef]
- Li, Y.; Chen, X.; Liu, L.; Liu, P.; Zhou, Z.; Hu, H.; Wu, Y.; Lei, T. Characteristics and adsorption of Cr(VI) of biochar pyrolyzed from landfill leachate sludge. J. Anal. Appl. Pyrol. 2022, 162, 105449. [Google Scholar] [CrossRef]
- Ai, D.; Tang, Y.; Yang, R.; Meng, Y.; Wei, T.; Wang, B. Hexavalent chromium (Cr(VI)) removal by ball-milled iron-sulfur @biochar based on P-recovery: Enhancement effect and synergy mechanism. Bioresour. Technol. 2023, 371, 128598. [Google Scholar] [CrossRef]
- Wang, M.; Zhao, Z.; Zhang, Y. Magnetite-Contained biochar derived from Fenton sludge modulated electron transfer of microorganisms in anaerobic digestion. J. Hazard. Mater. 2021, 403, 123972. [Google Scholar] [CrossRef]
- Su, C.; Wang, S.; Zhou, Z.; Wang, H.; Xie, X.; Yang, Y.; Feng, Y.; Liu, W.; Liu, P. Chemical processes of Cr(VI) removal by Fe-modified biochar under aerobic and anaerobic conditions and mechanism characterization under aerobic conditions using synchrotron-related techniques. Sci. Total Environ. 2021, 768, 144604. [Google Scholar] [CrossRef]
- Lan, G.; Fan, Q.; Liu, Y.; Chen, C.; Li, G.; Liu, Y.; Yin, X. Rhamnolipid production from waste cooking oil using Pseudomonas SWP-4. Biochem. Eng. J. 2015, 101, 44–54. [Google Scholar] [CrossRef] [Green Version]
- Kang, S.; Lee, J.; Kim, K. Biosorption of Cr(III) and Cr(VI) onto the cell surface of Pseudomonas aeruginosa. Biochem. Eng. J. 2007, 36, 54–58. [Google Scholar] [CrossRef]
- Yu, W.; Yang, J.; Tao, S.; Shi, Y.; Yu, J.; Lv, Y.; Liang, S.; Xiao, K.; Liu, B.; Hou, H.; et al. A comparatively optimization of dosages of oxidation agents based on volatile solids and dry solids content in dewatering of sewage sludge. Water Res. 2017, 126, 342–350. [Google Scholar] [CrossRef]
- Kim, S.O.; Moon, S.H.; Kim, K.W.; Yun, S.T. Pilot scale study on the ex-situ electrokinetic removal of heavy metals from municipal wastewater sludges. Water Res. 2002, 36, 4765–4774. [Google Scholar] [CrossRef]
- USEPA. Method 7196A: Chromium, Hexavalent (Colorimetric). 1992. Available online: https://www.google.com.hk/search?q=Method+7196A%3A+Chromium%2C+Hexavalent+%28Colorimetric&ei=fNQSZJ20Asyy2roP6eazuA4&ved=0ahUKEwid4oaWheD9AhVMmVYBHWnzDOcQ4dUDCA4&uact=5&oq=Method+7196A%3A+Chromium%2C+Hexavalent+%28Colorimetric&gs_lcp=Cgxnd3Mtd2l6LXNlcnAQAzIKCAAQRxDWBBCwAzIKCAAQRxDWBBCwAzIKCAAQRxDWBBCwAzIKCAAQRxDWBBCwAzIKCAAQRxDWBBCwAzIKCAAQRxDWBBCwAzIKCAAQRxDWBBCwAzIKCAAQRxDWBBCwA0oECEEYAFAAWABgsQ5oAnABeACAAQCIAQCSAQCYAQDIAQjAAQE&sclient=gws-wiz-serp (accessed on 12 February 2023).
- Yu, Y.; An, Q.; Zhou, Y.; Deng, S.; Miao, Y.; Zhao, B.; Yang, L. Highly synergistic effects on ammonium removal by the co-system of Pseudomonas stutzeri XL-2 and modified walnut shell biochar. Bioresour. Technol. 2019, 280, 239–246. [Google Scholar] [CrossRef]
- Hedley, M.; Stewart, J.; Chauhan, B. Changes in inorganic and organic soil phosphorus fractions induced by cultivation practices and by laboratory incubations. Soil Sci. Soc. Am. J. 1982, 46, 970–976. [Google Scholar] [CrossRef]
- Schnell, S.; Ratering, S.; Jansen, K. Simultaneous determination of iron(III), iron(II), and manganese(II) in environmental samples by ion chromatography. Environ. Sci. Technol. 1998, 32, 1530–1537. [Google Scholar] [CrossRef]
- Xu, H.; Sun, Y.; Li, J.; Li, F.; Guan, X. Aging of zerovalent iron in synthetic groundwater: X-ray photoelectron spectroscopy depth profiling characterization and cepassivation with uniform magnetic field. Environ. Sci. Technol. 2016, 50, 8214–8222. [Google Scholar] [CrossRef]
- Ri, C.; Tang, J.; Liu, F.; Lyu, H.; Li, F. Enhanced microbial reduction of aqueous hexavalent chromium by Shewanella oneidensis MR-1 with biochar as electron shuttle. J. Environ. Sci. 2022, 113, 12–25. [Google Scholar] [CrossRef]
- Ma, R.; Yan, X.; Pu, X.; Fu, X. An exploratory study on the aqueous Cr(VI) removal by the sulfate reducing sludge-based biochar. Sep. Purif. Technol. 2021, 276, 119314. [Google Scholar] [CrossRef]
- Zhao, X.; Feng, H.; Jia, P.; An, Q.; Ma, M. Removal of Cr(VI) from aqueous solution by a novel ZnO-sludge biochar composite. Environ. Sci. Pollut. Res. Int. 2022, 29, 83045–83059. [Google Scholar] [CrossRef]
- Chen, X.; Fan, G.; Li, H.; Li, Y.; Zhang, R.; Huang, Y.; Xu, X. Nanoscale zero-valent iron particles supported on sludge-based biochar for the removal of chromium (VI) from aqueous system. Environ. Sci. Pollut. Res. Int. 2022, 29, 3853–3863. [Google Scholar] [CrossRef]
- Fei, Y.; Li, M.; Ye, Z.; Guan, J.; Huang, Z.; Xiao, T.; Zhang, P. The pH-sensitive sorption governed reduction of Cr(VI) by sludge derived biochar and the accelerating effect of organic acids. J. Hazard. Mater. 2022, 423, 127205. [Google Scholar] [CrossRef] [PubMed]

| Experimental Objectives | Test Groups | Cr(VI) (mg/L) | Fe-300 (g/L) | Fe-800 (g/L) | Fe(II) (g/L) | Fe(0) (g/L) | PA (%, v/v) | The Determination Parameters |
|---|---|---|---|---|---|---|---|---|
| Section 2.3. Reduction of Cr(VI) to Cr(III) by Iron-Rich Biochar and PA | Cr (control group 1) | 10 | - | - | - | - | - | Cr |
| Cr + PA | 10 | - | - | - | - | 2 | ||
| Cr + Fe-300 | 10 | 1 | - | - | - | - | ||
| Cr + Fe-800 | 10 | - | 1 | - | - | - | ||
| Cr + Fe-300 + PA | 10 | 1 | - | - | - | 2 | ||
| Cr + Fe-800 + PA | 10 | - | 1 | - | - | 2 | ||
| Section 2.4.2. Phosphorus Release from Biochar by PA | Fe-300 (control group 2) | - | 1 | - | - | - | - | P |
| Fe-800 (control group 3) | - | - | 1 | - | - | - | ||
| Cr + Fe-300 | 10 | 1 | - | - | - | - | ||
| Cr + Fe-800 | 10 | - | 1 | - | - | - | ||
| Fe-300 + PA | - | 1 | - | - | - | 2 | ||
| Fe-800 + PA | - | - | 1 | - | - | 2 | ||
| Cr + Fe-300 + PA | 10 | 1 | - | - | - | 2 | ||
| Cr + Fe-800 + PA | 10 | - | 1 | - | - | 2 | ||
| Section 2.4.3. The Impacts of Different Iron Species on the Reduction of Cr(VI) to Cr(III) | Cr + Fe-300 | 10 | 1 | - | - | - | - | Fe |
| Cr + Fe(II) | 10 | - | - | 1 | - | - | ||
| Cr + Fe-800 | 10 | - | 1 | - | - | - | ||
| Cr + Fe(0) | 10 | - | - | - | 1 | - | ||
| Cr + Fe-300 + PA | 10 | 1 | - | - | - | 2 | ||
| Cr + Fe(II) + PA | 10 | - | - | 1 | - | 2 | ||
| Cr + Fe-800 + PA | 10 | - | 1 | - | - | 2 | ||
| Cr + Fe(0) + PA | 10 | - | - | - | 1 | 2 |
| Test Groups | Fe(II) | Fe(III) | Total Fe |
|---|---|---|---|
| Cr + Fe-300 | 0.69 | 0.12 | 0.81 |
| Cr + Fe(II) | 302.61 | 4.81 | 307.42 |
| Cr + Fe-800 | 0.21 | 0.49 | 0.70 |
| Cr + Fe(0) | 1.65 | 0.29 | 1.94 |
| Cr + Fe-300 + PA | 0.45 | 0.15 | 0.60 |
| Cr + Fe(II) + PA | 268.30 | 12.75 | 281.05 |
| Cr + Fe-800 + PA | 0.41 | 0.04 | 0.45 |
| Cr + Fe(0) + PA | 0.52 | 0.37 | 0.89 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ou, B.; Wang, H.; Xiao, K.; Zhu, Y.; Liu, Y.; Liang, S.; Hou, H.; Yu, W.; Hu, J.; Yang, J. Reduction of Cr(VI) by Synergistic Effects of Iron-Rich Biochar and Pseudomonas aeruginosa. Water 2023, 15, 1159. https://doi.org/10.3390/w15061159
Ou B, Wang H, Xiao K, Zhu Y, Liu Y, Liang S, Hou H, Yu W, Hu J, Yang J. Reduction of Cr(VI) by Synergistic Effects of Iron-Rich Biochar and Pseudomonas aeruginosa. Water. 2023; 15(6):1159. https://doi.org/10.3390/w15061159
Chicago/Turabian StyleOu, Bei, Hui Wang, Keke Xiao, Yuwei Zhu, Yuan Liu, Sha Liang, Huijie Hou, Wenbo Yu, Jingping Hu, and Jiakuan Yang. 2023. "Reduction of Cr(VI) by Synergistic Effects of Iron-Rich Biochar and Pseudomonas aeruginosa" Water 15, no. 6: 1159. https://doi.org/10.3390/w15061159





