Spatial Distribution of Pine Pollen Grains Concentrations as a Source of Biologically Active Substances in Surface Waters of the Southern Baltic Sea
Abstract
:1. Introduction
2. Materials and Methods
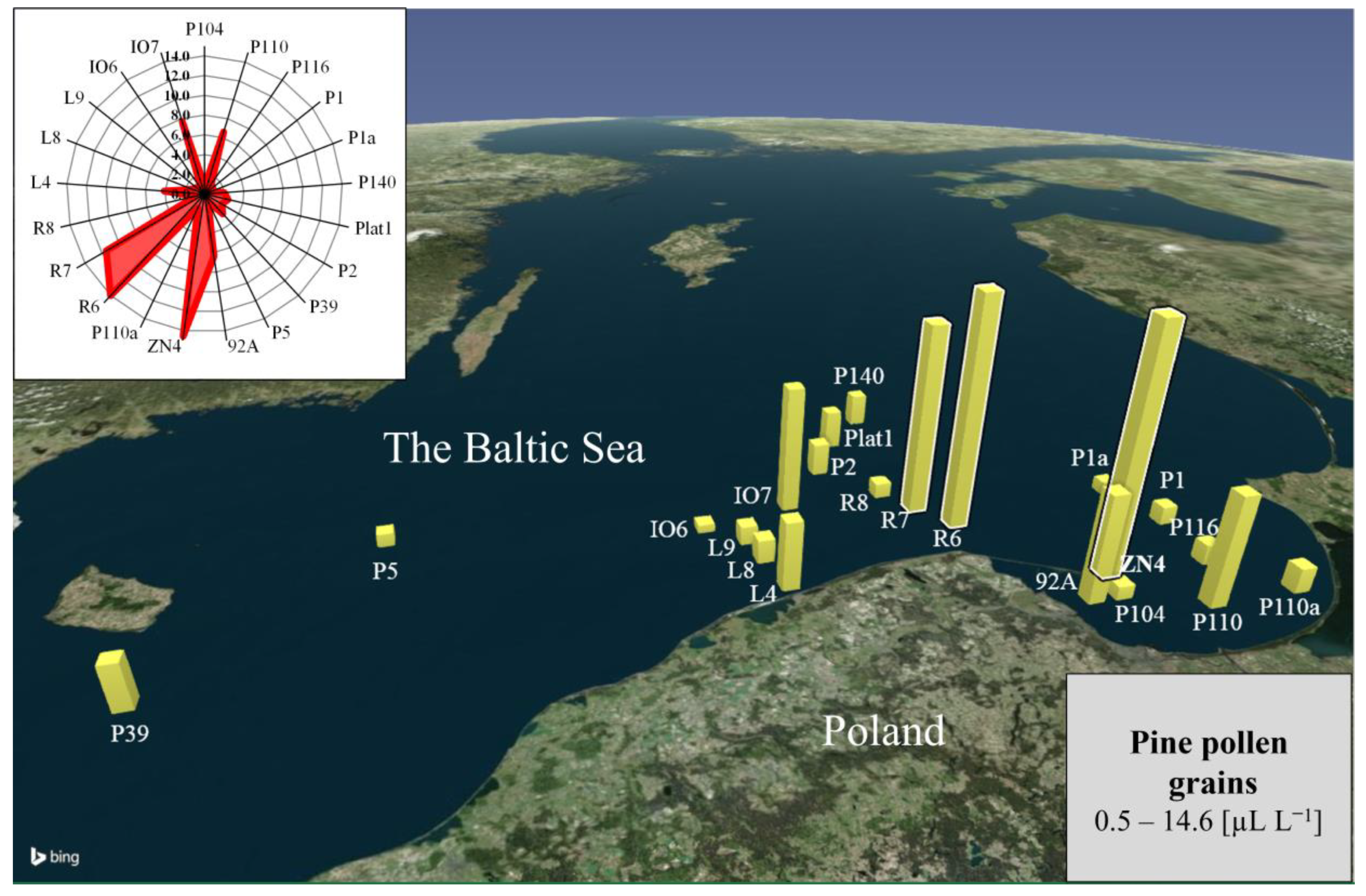
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Stedmon, C.A.; Markager, S.; Tranvik, L.J.; Kronberg, L.; Slätis, T.; Martinsen, W. Photochemical production of ammonium and transformation of dissolved organic matter in the Baltic Sea. Mar. Chem. 2007, 104, 227–240. [Google Scholar] [CrossRef]
- Schneider, B.; Dellwig, O.; Kuliński, K.; Omstedt, A.; Pollehne, F.; Rehder, G.; Savchuk, O. Biogeochemical cycles. In Biological Oceanography of the Baltic Sea; Snoeijs-Leijonmalm, P., Schubert, H., Radziejewska, T., Eds.; Springer: Dordrecht, The Netherlands, 2017; pp. 87–122. [Google Scholar] [CrossRef]
- Gustafsson, E.; Savchuk, O.P.; Gustafsson, B.G.; Muller-Karulis, B. Key processes in the coupled carbon, nitrogen, and phosphorus cycling of the Baltic Sea. Biogeochemistry 2017, 134, 301–317. [Google Scholar] [CrossRef] [Green Version]
- Kuliński, K.; Rehder, G.; Asmala, E.; Bartosova, A.; Carstensen, J.; Gustafsson, B.; Hall, P.O.J.; Humborg, C.; Jilbert, T.; Jürgens, K.; et al. Biogeochemical functioning of the Baltic Sea. Earth Syst. Dynam. 2022, 13, 633–685. [Google Scholar] [CrossRef]
- Savchuk, O. Studies of the assimilation capacity and effects of nutrient load reductions in the eastern Gulf of Finland with a biogeochemical model. Boreal Environ. Res. 2000, 5, 147–163. [Google Scholar]
- Neumann, T. Towards a 3D-ecosystem model of the Baltic Sea. J. Mar. Syst. 2000, 25, 405–419. [Google Scholar] [CrossRef]
- Eilola, K.; Almroth-Rosell, E.; Dieterich, C.; Fransner, F.; Höglund, A.; Meier, H.E.M. Modeling nutrient transports and exchanges of nutrients between shallow regions and the open Baltic Sea in present and future climate. Ambio 2012, 41, 586–599. [Google Scholar] [CrossRef] [Green Version]
- Friedland, R.; Neumann, T.; Schernewski, G. Climate change and the Baltic Sea action plan: Model simulations on the future of the western Baltic Sea. J. Mar. Syst. 2012, 105–108, 175–186. [Google Scholar] [CrossRef]
- Meier, H.E.M.; Müller-Karulis, B.; Andersson, H.C.; Dieterich, C.; Eilola, K.; Gustafsson; Höglund, A.; Hordoir, R.; Kuznetsov, I.; Neumann, T.; et al. Impact of climate change on ecological quality indicators and biogeochemical fluxes in the Baltic Sea: A multi-model ensemble study. Ambio 2012, 41, 558–573. [Google Scholar] [CrossRef]
- Meier, H.E.M.; Müller-Karulis, B.; Andersson, H.C.; Dieterich, C.; Eilola, K.; Gustafsson, B.G.; Höglund, A.; Hordoir, R.; Kuznetsov, I.; Neumann, T.; et al. Ensemble modeling of the Baltic Sea ecosystem to provide scenarios for management. Ambio 2014, 43, 37–48. [Google Scholar] [CrossRef] [Green Version]
- Richardson, D.M.; Rundel, P.W. Ecology and Biogeography of Pinus: An introduction. In Ecology and Biogeography of Pinus; Richardson, D.M., Ed.; Cambridge University Press: Cambridge, UK, 2000; Part 1: 3–46; ISBN 9780521789103. [Google Scholar]
- EUFORGEN–The European Forest Genetic Resources Programme. Distribution Map of Scots Pine (Pinus sylvestris). 2009. Available online: https://www.euforgen.org/ (accessed on 1 February 2023).
- Nobisa, M.P.; Traiserb, C.; Roth-Nebelsickc, A. Latitudinal variation in morphological traits of the genus Pinus and its relation to environmental and phylogenetic signals. Plant Ecol. Divers. 2012, 5, 1–11. [Google Scholar] [CrossRef]
- Houston Durrant, T.; de Rigo, D.; Caudullo, G. Pinus sylvestris in Europe: Distribution, habitat, usage and threats. In European Atlas of Forest Tree Species; San-Miguel-Ayanz, J., de Rigo, D., Caudullo, G., Houston Durrant, T., Mauri, A., Eds.; Publication Office of the European Union: Luxembourg, 2016; ISBN 978-92-79-52833-0. [Google Scholar] [CrossRef]
- Erdtman, G. Pollen grains recovered from the atmosphere over the Atlantic. Acta Horti Gotob. 1938, 12, 185–196. [Google Scholar]
- Dyakowska, J. Textbook of Palynology. Methods and Problems; Geological Publications: Warsaw, Poland, 1959. [Google Scholar]
- Środoń, A. Pollen spectra from Spitsbergen. Folia Quaternaria 1960, 3, 1–17. [Google Scholar]
- Benkman, C.W. Wind dispersal capacity of pine seeds and the evolution of different seed dispersal modes in pines. Oikos 1995, 73, 221–224. [Google Scholar] [CrossRef] [Green Version]
- Harmata, K.; Olech, M. Transect for aerobiological studies from Antarctica to Poland. Grana 1991, 30, 458–463. [Google Scholar] [CrossRef] [Green Version]
- Proctor, M.; Yeo, P.; Lack, A. The Natural History of Pollination; Harper Collins New Naturalist: London, UK, 1996. [Google Scholar]
- Rousseau, D.-D.; Schevin, P.; Ferrier, J.; Jolly, D.; Andreasen, T.; Ascanius, S.E.; Hendriksen, S.-E.; Poulsen, U. Long-distance pollen transport from North America to Greenland in spring. J. Geophys. Res. 2008, 113, G02013. [Google Scholar] [CrossRef]
- Robledo-Arnuncio, J.J. Wind pollination over mesoscale distances: An investigation with Scots pine. N. Phytol. 2011, 190, 222–233. [Google Scholar] [CrossRef]
- Sitters, J.; Atkinson, C.L.; Guelzow, N.; Kelly, P.; Sullivan, L.L. Spatial stoichiometry: Cross-ecosystem material flows and their impact on recipient ecosystems and organisms. Oikos 2015, 124, 920–930. [Google Scholar] [CrossRef]
- Hesselman, H. Uber die Verbeitungsfahigheit des Waldbaumpollens; Medol: Skagsforsohs, 1919; p. 16. [Google Scholar]
- Szczepanek, K.; Myszkowska, D.; Worobiec, E.; Piotrowicz, K.; Ziemianin, M.; Bielec-Bąkowska, Z. The long-range transport of Pinaceae pollen: An example in Kraków (southern Poland). Aerobiologia 2017, 33, 109–125. [Google Scholar] [CrossRef] [Green Version]
- Dyakowska, J. The pollen rain on the sea and on the coast of Greenland. Bull. L’academie Pol. Sci. Lett. Ser. B 1948, 1, 25–33. [Google Scholar]
- Campbell, I.D.; Mc Donald, K.; Flannigan, M.D.; Kringayark, J. Long-distance transport of pollen in to the Arctic. Nature 1999, 399, 29–30. [Google Scholar] [CrossRef]
- Hu, C.; Qi, L.; English, D.C.; Wang, M.; Mikelsons, K.; Barnes, B.B.; Pawlik, M.M.; Ficek, D. Pollen in the Baltic Sea as viewed from space. Remote Sens. Environ. 2023, 284, 113337. [Google Scholar] [CrossRef]
- Pawlik, M.; Ficek, D. Pine pollen grains in coastal waters of the Baltic Sea. Int. J. Oceanol. Hydrobiol. Stud. 2016, 45, 35–41. [Google Scholar] [CrossRef]
- Pawlik, M.; Ficek, D. Validation of measurements of pine pollen grain concentrations in Baltic Sea waters. Oceanologia 2022, 64, 233–243. [Google Scholar] [CrossRef]
- Lienart, C.; Cirtwill, A.R.; Hedgespeth, M.L.; Bradshaw, C. A sprinkling of gold dust: Pine pollen as a carbon source in Baltic Sea coastal food webs. Limnol. Oceanogr. 2022, 67, 53–65. [Google Scholar] [CrossRef]
- Doskey, P.V.; Ugoagwu, B.J. Atmospheric deposition of macronutrients by pollen at a semi-remote site in northern Wisconsin. Atmos. Environ. 1989, 23, 2761–2766. [Google Scholar] [CrossRef]
- Richerson, P.J.; Moshiri, G.A.; Godshalk, G.L. Certain ecological aspects of pollen dispersion in Lake Tahoe (California–Nevada). Limnol. Oceanogr. 1970, 15, 149–153. [Google Scholar] [CrossRef]
- Cole, J.J.; Caraco, N.F.; Likens, G.E. Short-range atmospheric transport: A significant source of phosphorus to an oligotrophic lake. Limnol. Oceanogr. 1990, 35, 1230–1237. [Google Scholar] [CrossRef]
- Banks, H.H.; Nighswander, J.E. Relative contribution of hemlock pollen to the phosphorus loading of the Clear Lake Ecosystem near Minden, Ontario. In Proceedings of the Symposium on Sustainable Management of Hemlock Ecosystems in Eastern North America, Durham, NH, USA, 22–24 June 1999; Gen. Tech. Rep. NE-267. Department of Agriculture, Forest Service, Northeastern Forest Experiment Station: Newtown Square, PA, USA, 2000; pp. 168–174. [Google Scholar]
- Graham, M.D.; Vinebrooke, R.D.; Turner, M. Coupling of boreal forests and lakes: Effects of conifer pollen on littoral communities. Limnol. Oceanogr. 2006, 51, 1524–1529. [Google Scholar] [CrossRef]
- Rösel, S.; Rychła, A.; Wurzbacher, C.; Grossart, H.-P. Effects of pollen leaching and microbial degradation on organic carbon and nutrient availability in lake water. Aquat. Sci. 2012, 74, 87–99. [Google Scholar] [CrossRef]
- Keller, M.D.; Matrai, P.A. Optical Characteristics of Pollen Grains in Coastal Waters of the Gulf of Maine; Report Documentation; Bigelow Laboratory for Ocean Sciences: East Boothbay, ME, USA, 1998; pp. 1–6. [Google Scholar]
- Page, K.A.; Flannery, M.K. Chytrid fungi associated with pollen decomposition in crater lake, Oregon. Appl. Environ. Microbiol. 2018, 4, 83–100. [Google Scholar] [CrossRef]
- Siska, P.; Bryant, V.M., Jr.; Hung, I.-K. Geospatial analysis of southern pine biome and pollen distribution patterns in Southeastern United States. Geogr. Časopis 2006, 58, 4. [Google Scholar]
- Stavn, R.H.; Rick, H.J.; Falster, A.V. Correcting the errors from variable sea salt retention and water of hydration in loss on ignition analysis: Implications for studies of estuarine and coastal waters. Estuar. Coast. Shelf Sci. 2009, 81, 575–582. [Google Scholar] [CrossRef] [Green Version]
- Woźniak, S.B.; Meler, J.; Lednicka, B.; Zdun, A.; Stoń-Egiert, J. Inherent optical properties of suspended particulate matter in the southern Baltic Sea. Oceanologia 2011, 53, 691–729. [Google Scholar]
- Jeffrey, S.W.; Humphrey, G.F. New spectrophotometric equation for determining chlorophyll a, b, c1 and c2. Biochem. Physiol. Pflanz. 1975, 167, 194–204. [Google Scholar] [CrossRef]
- Barbarino, E.; Lourenço, S.O. Comparison of CHN Analysis and Hach Acid Digestion to Quantify Total Nitrogen in Marine Organisms. Limnology and Oceanography: Methods 2009, 7, 751–760. [Google Scholar] [CrossRef]
- Greenfield, L.G. Weight loss and release of mineral nitrogen from decomposing pollen. Soil Biol. Biochem. 1999, 31, 353–361. [Google Scholar] [CrossRef]
- Lee, E.J.; Booth, T. Macronutrient input from pollen in two regenerating pine stands in southeast Korea. Ecol. Res. 2003, 18, 423–430. [Google Scholar] [CrossRef]
- Filipiak, M. Pollen Stoichiometry May Influence Detrital Terrestrial and Aquatic Food Webs. Front. Ecol. Evol. 2016, 4, 138. [Google Scholar] [CrossRef] [Green Version]
- Kwiatkowski, A.; Lubliner-Mianowska, K. Badania składu chemicznego pyłku. II. Badania błon pyłkowych. Acta Soc. Botan. 1957, 26, 3. [Google Scholar]
- Zetsche, F.; Vicari, H. Uterssuchungen über die Membran der Sporen und Pollen III. 2. Picea orientalis, Pinus silvestris L., Corylus Avellana L. Acta Chim. Helv. 1931, 14, 62–67. [Google Scholar] [CrossRef]
- Lee, E.J.; Kenkel, N.; Booth, T. Atmospheric deposition of macronutrients by pollen in the boreal forest. Ecoscience 1996, 3, 304–309. [Google Scholar] [CrossRef]
- Stark, N. Nutrient cycling pathways and litter fungi. Bioscience 1972, 22, 355–360. [Google Scholar] [CrossRef]
- Shaw, G. The chemistry of sporopollenin. In Sporopollenin; Brooks, J., Grant, P.R., Muir, M., van Gijzel, P., Shaw, G., Eds.; Academic Press: New York, NY, USA, 1971; pp. 305–348. [Google Scholar]
- Wiermann, R.; Gubatz, S. Pollen wall and sporopollenin. In Sexual Reproduction in Flowering Plants; Russell, S.D., Dumas, C., Eds.; Academic Press: New York, NY, USA, 1992; pp. 35–72. [Google Scholar]
- Scott, R.J. Pollen exine: The sporopollenin enigma and the physics of pattern. In Molecular and Cellular Aspects of Plant Reproduction; Scott, R.J., Stead, A.D., Eds.; University Press: Cambridge, UK, 1994; pp. 49–81. [Google Scholar]
- Goldstein, S. Degradation of pollen by phycomycetes. Ecology 1960, 41, 543–545. [Google Scholar] [CrossRef]
- Hutchison, L.J.; Barron, G.L. Parasitism of pollen as a nutritional source for lignicolous Basidiomycota and other fungi. Mycol. Res. 1997, 101, 191–194. [Google Scholar] [CrossRef]
- Czeczuga, B.; Muszyńska, E. Zoosporic fungi growing on gymnosperm pollen in water of varied trophic state. Pol. J. Environ. Stud. 2001, 10, 89–94. [Google Scholar]
- Brooks, J.; Grant, P.R.; Muir, M. Sporopollenin. In Proceedings of the Symposium Held at the Geology Department, Imperial College, London, UK, 23–25 September 1970. [Google Scholar]
- Wurzbacher, C.M.; Baerlocher, F.; Grossart, H.-P. Fungi in lake ecosystems. Aquat. Microb. Ecol. 2010, 59, 125–149. [Google Scholar] [CrossRef] [Green Version]
- Grossart, H.-P.; Simon, M. Limnetic macroscopic organic aggregates (lake snow): Occurrence, characteristics, and microbial dynamics in Lake Constance. Limnol. Oceanogr. 1993, 38, 532–546. [Google Scholar] [CrossRef]
- Grossart, H.-P.; Kiorboe, T.; Tang, K.W.; Ploug, H. Bacterial colonization of particles: Growth and interactions. Appl. Environ. Microbiol. 2003, 69, 3500–3509. [Google Scholar] [CrossRef] [Green Version]
- Grossart, H.-P.; Kiorboe, T.; Tang, K.W.; Allgaier, M.; Yam, E.M.; Ploug, H. Interactions between marine snow and heterotrophic bacteria: Aggregate formation and microbial dynamics. Aquat. Microb. Ecol. 2006, 42, 19–26. [Google Scholar] [CrossRef] [Green Version]
- Masclaux, H.; Bec, A.; Kagami, M.; Perga, M.-E.; Sime-Ngando, T.; Desvilettes, C.; Bourdier, G. Food quality of anemophilous plant pollen for zooplankton. Limnol. Oceanogr. 2011, 56, 939–946. [Google Scholar] [CrossRef]
- Masclaux, H.; Perga, M.E.; Kagami, M.; Desvilettes, C.; Bourdier, G.; Bec, A. How pollen organic matter enters fresh water food webs. Limnol. Oceanogr. 2013, 58, 1185–1195. [Google Scholar] [CrossRef]
- Wurzbacher, C.; Rösel, S.; Rychła, A.; Grossart, H.-P. Importance of Saprotrophic fresh water fungi for pollen degradation. PLoS ONE 2014, 9, e94643. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Del Giorgio, P.A.; Cole, J.J. Bacterial growth efficiency in natural aquatic systems. Annu. Rev. Ecol. Syst. 1998, 29, 503–541. [Google Scholar] [CrossRef] [Green Version]
- Kominkova, D.; Kuehn, K.A.; Büsing, N.; Steiner, D.; Gessner, M.O. Microbial biomass, growth, and respiration associated with submerged litter of Phragmites australis decomposing in a littoral reed stand of a large lake. Aquat. Microb. Ecol. 2000, 22, 271–282. [Google Scholar] [CrossRef]




| Data | Station | Geographic Coordinates | Secchi Depth (m) | SPM(g m−3) | Chlorophyll a (mg m−3) | |
|---|---|---|---|---|---|---|
| Latitude (°N) | Longitude (°E) | |||||
| 14 May 2018 | P104 | 54.58 | 18.79 | 6.0 | 2.41 | 2.40 |
| 14 May 2018 | P110 | 54.50 | 19.11 | 5.0 | 1.99 | 2.20 |
| 15 May 2018 | P104c | 54.58 | 19.00 | 5.0 | 8.11 | 4.00 |
| 15 May 2018 | P116 | 54.58 | 19.00 | 5.5 | 2.91 | 3.10 |
| 15 May 2018 | P1 | 54.65 | 19.29 | 6.5 | 2.31 | 2.30 |
| 15 May 2018 | P1a | 54.83 | 19.32 | 5.5 | 1.63 | 3.50 |
| 16 May 2018 | P140 | 55.00 | 19.23 | 4.0 | 4.04 | 8.40 |
| 16 May 2018 | Plat1 | 55.55 | 18.39 | 5.0 | 4.11 | 4.30 |
| 16 May 2018 | P2 | 55.43 | 18.16 | 7.5 | 1.52 | 3.00 |
| 16 May 2018 | P39 | 55.29 | 18.00 | 8.5 | 1.90 | 1.10 |
| 17 May 2018 | P5 | 54.74 | 15.11 | 7.5 | 2.11 | 2.70 |
| 17 May 2018 | NP | 54.45 | 18.67 | 4.0 | 4.01 | 4.70 |
| 18 May 2018 | P115d | 54.44 | 18.75 | 4.0 | 2.69 | 2.03 |
| 18 May 2018 | P101 | 54.53 | 18.61 | 4.0 | 1.86 | 1.14 |
| 18 May 2018 | Rewa1 | 54.63 | 18.55 | 4.5 | 1.54 | 1.14 |
| 18 May 2018 | 92A | 55.21 | 15.98 | 6.0 | 2.07 | 3.20 |
| 20 May 2018 | ZN4 | 54.45 | 18.66 | 5.5 | 2.15 | 3.70 |
| 20 May 2018 | P110d | 54.50 | 18.79 | 7.0 | 2.08 | 2.50 |
| 20 May 2018 | ZN2 | 54.38 | 18.96 | 1.5 | 10.32 | 39.30 |
| 20 May 2018 | ZN2r | 54.33 | 18.94 | bd | 15.42 | 34.29 |
| 21 May 2018 | P114 | 54.40 | 19.00 | 2.0 | 5.89 | 6.68 |
| 21 May 2018 | ZN2b | 54.38 | 19.08 | 3.0 | 3.81 | 6.68 |
| 21 May 2018 | ZN2a | 54.45 | 18.92 | 6.5 | 3.86 | 3.34 |
| 21 May 2018 | P110a | 54.58 | 18.66 | 3.5 | 3.85 | 6.10 |
| 21 May 2018 | P104a | 54.58 | 19.42 | 7.0 | 1.70 | 1.40 |
| 21 May 2018 | P110b | 54.50 | 19.33 | 5.0 | 1.33 | 1.62 |
| 22 May 2018 | R4 | 54.88 | 18.37 | 14.0 | 0.94 | 0.50 |
| 22 May 2018 | R6 | 54.66 | 18.83 | 6.5 | 1.62 | 1.70 |
| 22 May 2018 | R7 | 54.50 | 18.79 | 5.5 | 2.16 | 3.00 |
| 22 May 2018 | R8 | 54.38 | 18.95 | 5.5 | 2.29 | 2.50 |
| 23 May 2018 | L4 | 54.50 | 19.51 | 14.0 | 0.73 | 0.60 |
| 23 May 2018 | L7 | 54.83 | 17.53 | 9.0 | 0.85 | 0.90 |
| 23 May 2018 | L8 | 54.58 | 19.41 | 7.0 | 0.70 | 0.60 |
| 23 May 2018 | L9 | 54.88 | 18.36 | 8.0 | 0.51 | 0.47 |
| 23 May 2018 | IO6 | 54.95 | 18.41 | 11.0 | 0.51 | 0.35 |
| 23 May 2018 | IO7 | 55.04 | 18.30 | 6.5 | 1.00 | 1.07 |
| Component | Content % of a Component/an Element in Dry Mass | References |
|---|---|---|
| cellulose | 3.60% | [48] |
| 2.00% | [49] | |
| sporopollenins | 19.60% | [48] |
| 21.90% | [49] | |
| C | 47.66% | own research |
| 50.79% | [47] | |
| 49.50% | [40] | |
| 50.00% | [32] | |
| P | 0.32% | own research |
| 0.34% | [47] | |
| 0.30% | [47] | |
| 0.30% | [46] | |
| N | 2.50% | own research |
| 2.24% | [47] | |
| 2.00–2.40% | [45] | |
| S | 0.26% | [47] |
| K | 0.94% | [47] |
| Mg | 0.15% | [47] |
| Ca | 0.05% | [47] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pawlik, M.M.; Ficek, D. Spatial Distribution of Pine Pollen Grains Concentrations as a Source of Biologically Active Substances in Surface Waters of the Southern Baltic Sea. Water 2023, 15, 978. https://doi.org/10.3390/w15050978
Pawlik MM, Ficek D. Spatial Distribution of Pine Pollen Grains Concentrations as a Source of Biologically Active Substances in Surface Waters of the Southern Baltic Sea. Water. 2023; 15(5):978. https://doi.org/10.3390/w15050978
Chicago/Turabian StylePawlik, Magdalena Maria, and Dariusz Ficek. 2023. "Spatial Distribution of Pine Pollen Grains Concentrations as a Source of Biologically Active Substances in Surface Waters of the Southern Baltic Sea" Water 15, no. 5: 978. https://doi.org/10.3390/w15050978





