Impact of Antibiotic Pollution on the Bacterial Population within Surface Water with Special Focus on Mountain Rivers
Abstract
1. Introduction
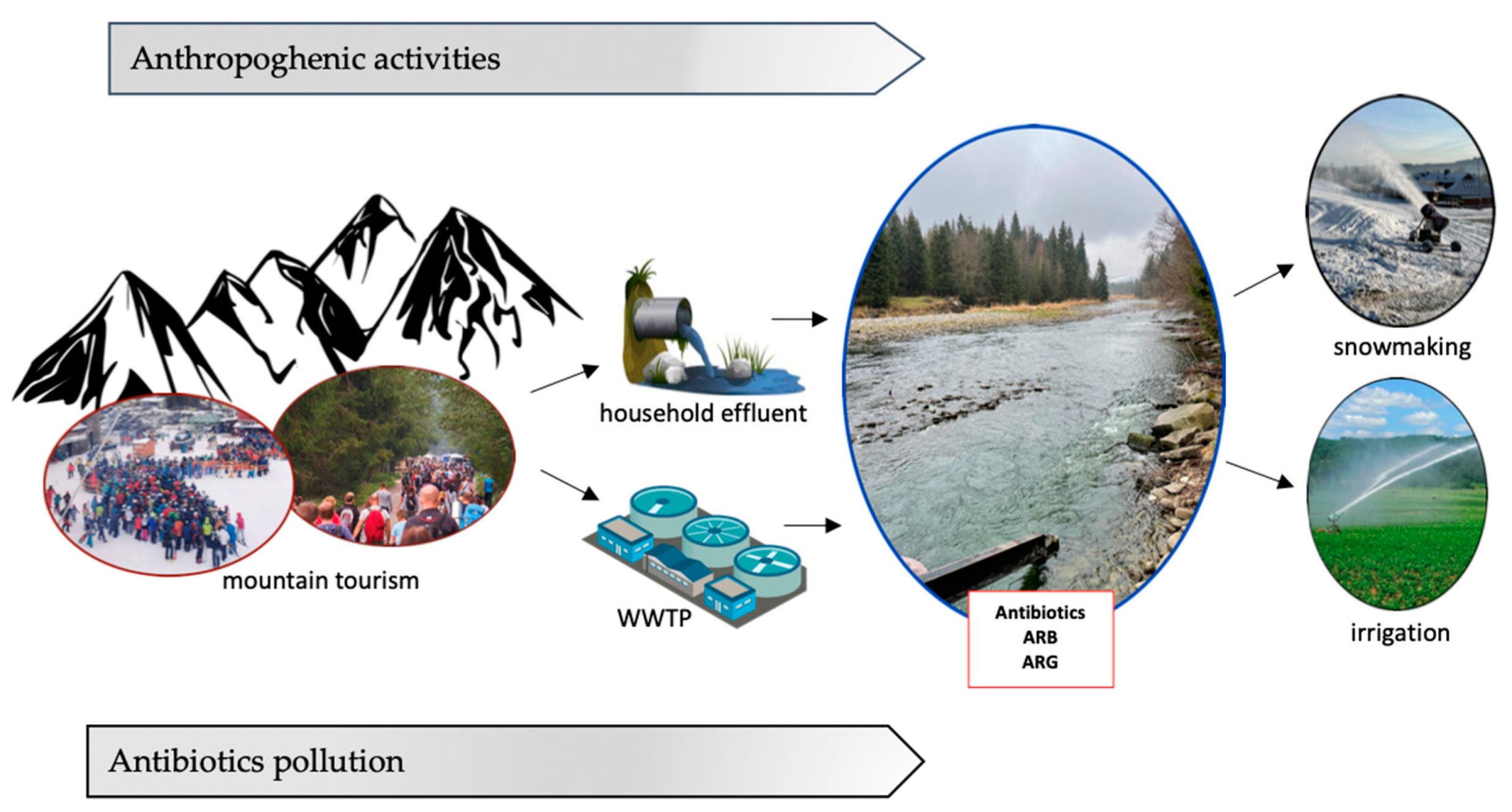
2. Sources of Antibiotics in Mountain Rivers
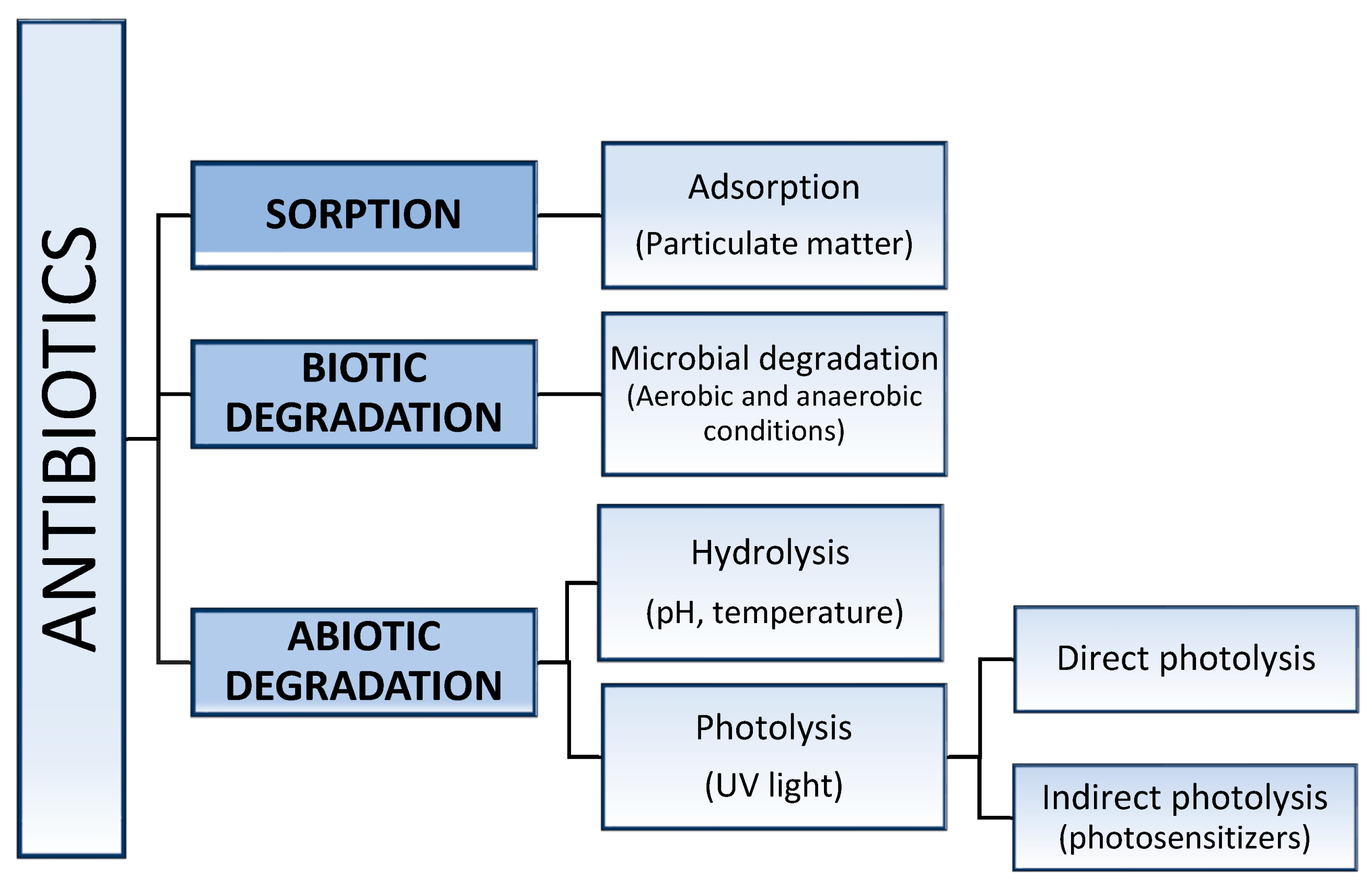
3. Stability of Antimicrobial Agents in Water Environments
4. Effect of Sub-Inhibitory Concentrations of Antibiotics on Bacterial Populations
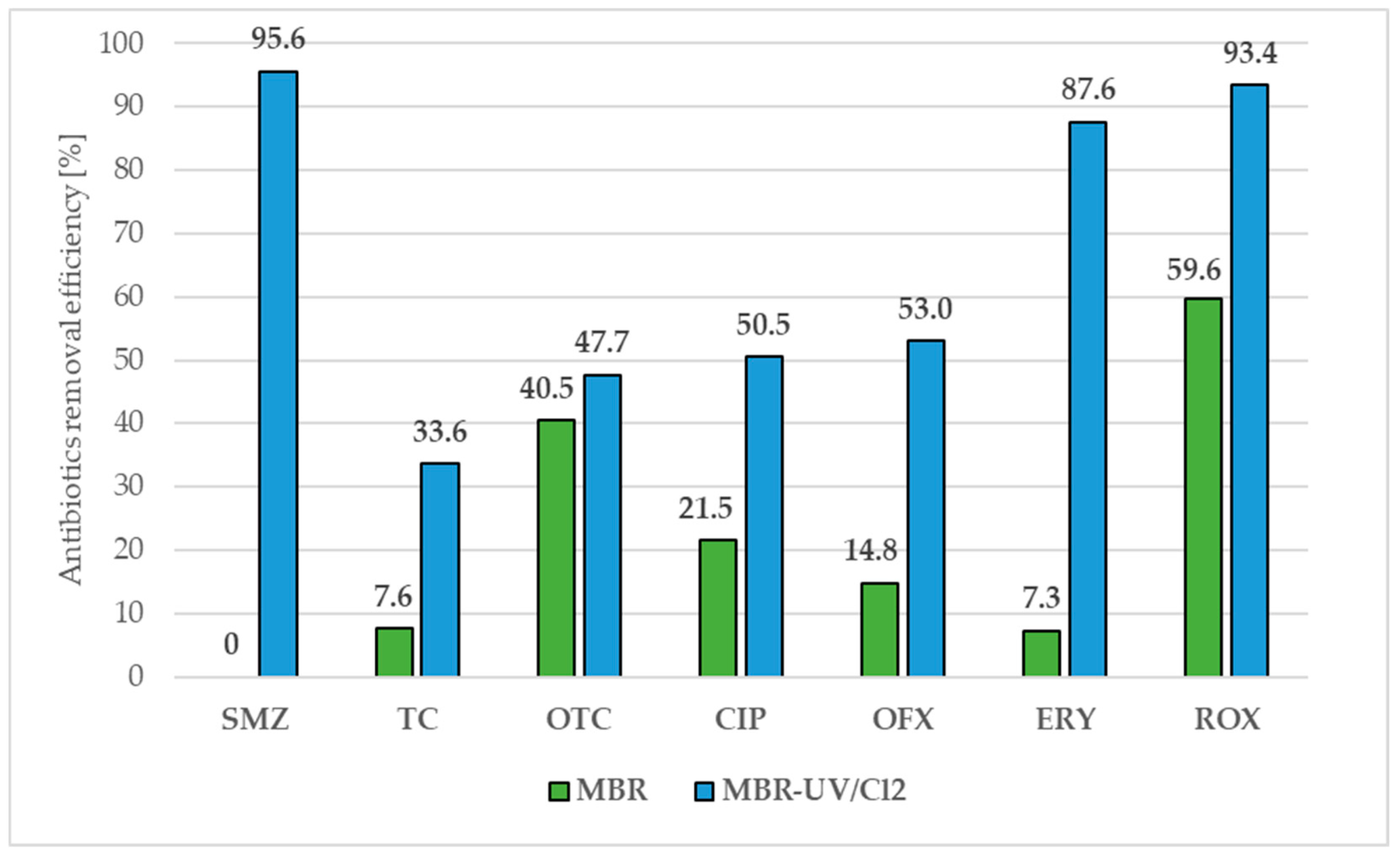
5. Antibiotic Removal Processes from Water and Wastewater
6. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Meek, R.W.; Vyas, H.; Piddock, L.J.V. Nonmedical Uses of Antibiotics: Time to Restrict Their Use? PLOS Biol. 2015, 13, e1002266. [Google Scholar] [CrossRef] [PubMed]
- Van, T.T.H.; Yidana, Z.; Smooker, P.M.; Coloe, P.J. Antibiotic Use in Food Animals Worldwide, with a Focus on Africa: Pluses and Minuses. J. Glob. Antimicrob. Resist. 2020, 20, 170–177. [Google Scholar] [CrossRef] [PubMed]
- Du, L.; Liu, W. Occurrence, Fate, and Ecotoxicity of Antibiotics in Agro-Ecosystems. A Review. Agron. Sustain. Dev. 2012, 32, 309–327. [Google Scholar] [CrossRef]
- Polianciuc, S.I.; Gurzău, A.E.; Kiss, B.; Ștefan, M.G.; Loghin, F. Antibiotics in the Environment: Causes and Consequences. Med. Pharm. Rep. 2020, 93, 231. [Google Scholar] [CrossRef] [PubMed]
- Chow, L.K.M.; Ghaly, T.M.; Gillings, M.R. A Survey of Sub-Inhibitory Concentrations of Antibiotics in the Environment. J. Environ. Sci. 2021, 99, 21–27. [Google Scholar] [CrossRef]
- Wang, M.; Shen, W.; Yan, L.; Wang, X.-H.; Xu, H. Stepwise Impact of Urban Wastewater Treatment on the Bacterial Community Structure, Antibiotic Contents, and Prevalence of Antimicrobial Resistance. Environ. Pollut. 2017, 231, 1578–1585. [Google Scholar] [CrossRef]
- Ching, C.; Orubu, E.S.F.; Sutradhar, I.; Wirtz, V.J.; Boucher, H.W.; Zaman, M.H. Bacterial Antibiotic Resistance Development and Mutagenesis Following Exposure to Subinhibitory Concentrations of Fluoroquinolones in Vitro: A Systematic Review of the Literature. JAC-Antimicrob. Resist. 2020, 2, dlaa068. [Google Scholar] [CrossRef]
- Atterby, C.; Nykvist, M.; Lustig, U.; Andersson, D.I.; Järhult, J.D.; Sandegren, L. Selection of Resistant Bacteria in Mallards Exposed to Subinhibitory Concentrations of Ciprofloxacin in Their Water Environment. Antimicrob. Agents Chemother. 2021, 65, e01858-20. [Google Scholar] [CrossRef]
- Manaia, C.M. Assessing the Risk of Antibiotic Resistance Transmission from the Environment to Humans: Non-Direct Proportionality between Abundance and Risk. Trends Microbiol. 2017, 25, 173–181. [Google Scholar] [CrossRef]
- Michael-Kordatou, I.; Karaolia, P.; Fatta-Kassinos, D. The Role of Operating Parameters and Oxidative Damage Mechanisms of Advanced Chemical Oxidation Processes in the Combat against Antibiotic-Resistant Bacteria and Resistance Genes Present in Urban Wastewater. Water Res. 2018, 129, 208–230. [Google Scholar] [CrossRef]
- Hao, H.; Shi, D.; Yang, D.; Yang, Z.; Qiu, Z.; Liu, W.; Shen, Z.; Yin, J.; Wang, H.; Li, J.; et al. Profiling of Intracellular and Extracellular Antibiotic Resistance Genes in Tap Water. J. Hazard. Mater. 2019, 365, 340–345. [Google Scholar] [CrossRef]
- Zarei-Baygi, A.; Smith, A.L. Intracellular versus Extracellular Antibiotic Resistance Genes in the Environment: Prevalence, Horizontal Transfer, and Mitigation Strategies. Bioresour. Technol. 2021, 319, 124181. [Google Scholar] [CrossRef]
- von Wintersdorff, C.J.H.; Penders, J.; van Niekerk, J.M.; Mills, N.D.; Majumder, S.; van Alphen, L.B.; Savelkoul, P.H.M.; Wolffs, P.F.G. Dissemination of Antimicrobial Resistance in Microbial Ecosystems through Horizontal Gene Transfer. Front. Microbiol. 2016, 7, 173. [Google Scholar] [CrossRef]
- Huijbers, P.M.C.; Blaak, H.; de Jong, M.C.M.; Graat, E.A.M.; Vandenbroucke-Grauls, C.M.J.E.; de Roda Husman, A.M. Role of the Environment in the Transmission of Antimicrobial Resistance to Humans: A Review. Environ. Sci. Technol. 2015, 49, 11993–12004. [Google Scholar] [CrossRef]
- Anh, H.Q.; Le, T.P.Q.; Da Le, N.; Lu, X.X.; Duong, T.T.; Garnier, J.; Rochelle-Newall, E.; Zhang, S.; Oh, N.-H.; Oeurng, C.; et al. Antibiotics in Surface Water of East and Southeast Asian Countries: A Focused Review on Contamination Status, Pollution Sources, Potential Risks, and Future Perspectives. Sci. Total Environ. 2021, 764, 142865. [Google Scholar] [CrossRef]
- Sta Ana, K.M.; Madriaga, J.; Espino, M.P. β-Lactam Antibiotics and Antibiotic Resistance in Asian Lakes and Rivers: An Overview of Contamination, Sources and Detection Methods. Environ. Pollut. 2021, 275, 116624. [Google Scholar] [CrossRef]
- Mittal, P.; Prasoodanan PK, V.; Dhakan, D.B.; Kumar, S.; Sharma, V.K. Metagenome of a Polluted River Reveals a Reservoir of Metabolic and Antibiotic Resistance Genes. Environ. Microbiome 2019, 14, 5. [Google Scholar] [CrossRef]
- Koniuszewska, I.; Korzeniewska, E.; Harnisz, M.; Kiedrzyńska, E.; Kiedrzyński, M.; Czatzkowska, M.; Jarosiewicz, P.; Zalewski, M. The Occurrence of Antibiotic-Resistance Genes in the Pilica River, Poland. Ecohydrol. Hydrobiol. 2020, 20, 1–11. [Google Scholar] [CrossRef]
- Harnisz, M.; Gołaś, I.; Pietruk, M. Tetracycline-Resistant Bacteria as Indicators of Antimicrobial Resistance in Protected Waters—The Example of the Drwęca River Nature Reserve (Poland). Ecol. Indic. 2011, 11, 663–668. [Google Scholar] [CrossRef]
- Lenart-Boroń, A.M.; Boroń, P.M.; Prajsnar, J.A.; Guzik, M.W.; Żelazny, M.S.; Pufelska, M.D.; Chmiel, M.J. COVID-19 Lockdown Shows How Much Natural Mountain Regions Are Affected by Heavy Tourism. Sci. Total Environ. 2022, 806, 151355. [Google Scholar] [CrossRef]
- Tan, B.; Ng, C.; Nshimyimana, J.P.; Loh, L.L.; Gin, K.Y.-H.; Thompson, J.R. Next-Generation Sequencing (NGS) for Assessment of Microbial Water Quality: Current Progress, Challenges, and Future Opportunities. Front. Microbiol. 2015, 6, 1027. [Google Scholar] [CrossRef] [PubMed]
- Yergeau, E.; Lawrence, J.R.; Sanschagrin, S.; Waiser, M.J.; Korber, D.R.; Greer, C.W. Next-Generation Sequencing of Microbial Communities in the Athabasca River and Its Tributaries in Relation to Oil Sands Mining Activities. Appl. Environ. Microbiol. 2012, 78, 7626–7637. [Google Scholar] [CrossRef] [PubMed]
- Nimnoi, P.; Pongsilp, N. Marine Bacterial Communities in the Upper Gulf of Thailand Assessed by Illumina Next-Generation Sequencing Platform. BMC Microbiol. 2020, 20, 19. [Google Scholar] [CrossRef] [PubMed]
- Viviroli, D.; Weingartner, R. The Hydrological Significance of Mountains: From Regional to Global Scale. Hydrol. Earth Syst. Sci. 2004, 8, 1017–1030. [Google Scholar] [CrossRef]
- Lenart-Boroń, A.; Prajsnar, J.; Guzik, M.; Boroń, P.; Chmiel, M. How Much of Antibiotics Can Enter Surface Water with Treated Wastewater and How It Affects the Resistance of Waterborne Bacteria: A Case Study of the Białka River Sewage Treatment Plant. Environ. Res. 2020, 191, 110037. [Google Scholar] [CrossRef]
- Lenart-Boroń, A.; Wolanin, A.; Jelonkiewicz, Ł.; Chmielewska-Błotnicka, D.; Żelazny, M. Spatiotemporal Variability in Microbiological Water Quality of the Białka River and Its Relation to the Selected Physicochemical Parameters of Water. Water. Air. Soil Pollut. 2016, 227, 22. [Google Scholar] [CrossRef]
- Mokhtar, M.B.; Aris, A.Z.; Abdullah, M.H.; Yusoff, M.K.; Abdullah, M.P.; Idris, A.R.; Raja Uzir, R.I. A Pristine Environment and Water Quality in Perspective: Maliau Basin, Borneo’s Mysterious World. Water Environ. J. 2009, 23, 219–228. [Google Scholar] [CrossRef]
- Bojarczuk, A.; Jelonkiewicz, Ł.; Lenart-Boroń, A. The Effect of Anthropogenic and Natural Factors on the Prevalence of Physicochemical Parameters of Water and Bacterial Water Quality Indicators along the River Białka, Southern Poland. Environ. Sci. Pollut. Res. 2018, 25, 10102–10114. [Google Scholar] [CrossRef]
- Vanat, L. International Report on Snow & Mountain Tourism—Overview of the Key Industry Figures for Ski Resorts. 2020. Available online: https://www.vanat.ch/RM-world-report-2020.pdf (accessed on 10 December 2022).
- Krzesiwo, K. Ocena wielkości ruchu turystycznego w ośrodku narciarskim kotelnica białczańska w sezonie zimowym 2014/2015. Pr. Geogr. 2016, 24, 47–70. [Google Scholar] [CrossRef]
- Krzesiwo, K. Ocena sytuacji rozwojowej i funkcjonalnej stacji narciarskich—Przykład polskich Karpat. Stud. Ind. Geogr. Comm. Pol. Geogr. Soc. 2021, 35, 259–276. [Google Scholar] [CrossRef]
- Pickering, C.M.; Harrington, J.; Worboys, G. Environmental Impacts of Tourism on the Australian Alps Protected Areas. Mt. Res. Dev. 2003, 23, 9. [Google Scholar] [CrossRef]
- Sanchez-Cid, C.; Keuschnig, C.; Torzewski, K.; Stachnik, Ł.; Kępski, D.; Luks, B.; Nawrot, A.; Niedzielski, P.; Vogel, T.M.; Larose, C. Environmental and Anthropogenic Factors Shape the Snow Microbiome and Antibiotic Resistome. Front. Microbiol. 2022, 13, 918622. [Google Scholar] [CrossRef]
- Krzesiwo, K.; Ziółkowska-Weiss, K.; Żemła, M. The Attractiveness of Selected Central European Countries for Winter Sports and Mountain Hiking. Turyzm/Tourism 2018, 28, 35–40. [Google Scholar] [CrossRef]
- Opermanis, O.; MacSharry, B.; Aunins, A.; Sipkova, Z. Connectedness and Connectivity of the Natura 2000 Network of Protected Areas across Country Borders in the European Union. Biol. Conserv. 2012, 153, 227–238. [Google Scholar] [CrossRef]
- Pietrzyk-Kaszyńska, A.; Cent, J.; Grodzińska-Jurczak, M.; Szymańska, M. Factors Influencing Perception of Protected Areas—The Case of Natura 2000 in Polish Carpathian Communities. J. Nat. Conserv. 2012, 20, 284–292. [Google Scholar] [CrossRef]
- Holden, A. Winter Tourism and the Environment in Conflict: The Case of Cairngorm, Scotland. Int. J. Tour. Res. 2000, 2, 247–260. [Google Scholar] [CrossRef]
- Tatra National Park, Poland Sale of Admission Tickets to the Tatra National Park—Statistics. 2022. Available online: https://tpn.pl/zwiedzaj/turystyka/statystyka (accessed on 10 December 2022).
- Now the Environment The Tatra National Park Wants to Channel All Shelters-an Interview. 2020. Available online: https://www.teraz-srodowisko.pl/aktualnosci/tatrzanski-park-narodowy-chce-skanalizowac-wszystkie-schroniska-8205.html (accessed on 10 December 2022).
- Lenart-Boroń, A.; Prajsnar, J.; Guzik, M.; Boroń, P.; Grad, B.; Żelazny, M. Antibiotics in Groundwater and River Water of Białka—A Pristine Mountain River. Appl. Sci. 2022, 12, 12743. [Google Scholar] [CrossRef]
- Kangas, K.; Vuori, K.-M.; Määttä-Juntunen, H.; Siikamäki, P. Impacts of Ski Resorts on Water Quality of Boreal Lakes: A Case Study in Northern Finland. Boreal Env. Res. 2012, 17, 313–325. [Google Scholar]
- Senetra, A.; Dynowski, P.; Cieślak, I.; Źróbek-Sokolnik, A. An Evaluation of the Impact of Hiking Tourism on the Ecological Status of Alpine Lakes—A Case Study of the Valley of Dolina Pięciu Stawów Polskich in the Tatra Mountains. Sustainability 2020, 12, 2963. [Google Scholar] [CrossRef]
- Kovalakova, P.; Cizmas, L.; McDonald, T.J.; Marsalek, B.; Feng, M.; Sharma, V.K. Occurrence and Toxicity of Antibiotics in the Aquatic Environment: A Review. Chemosphere 2020, 251, 126351. [Google Scholar] [CrossRef]
- Boxall, A.B.A.; Blackwell, P.; Cavallo, R.; Kay, P.; Tolls, J. The Sorption and Transport of a Sulphonamide Antibiotic in Soil Systems. Toxicol. Lett. 2002, 131, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Medication Datasheet: Levofloxacin 500 Mg Aurovitas Pharma Poland; Aurovitas Pharma: Warsaw, Poland, 2022.
- Varoquaux, O.; Lajoie, D.; Gobert, C.; Cordonnier, P.; Ducreuzet, C.; Pays, M.; Advenier, C. Pharmacokinetics of the Trimethoprim-Sulphamethoxazole Combination in the Elderly. Br. J. Clin. Pharmacol. 1985, 20, 575–581. [Google Scholar] [CrossRef] [PubMed]
- Medication Datasheet: METRONIDAZOL I.V. BRAUN 5 Mg/Ml. Regist. Med. Prod. B. Braun Melsungen AG, Germany. Available online: https://www.bbraun.co.id/en/products/b/metronidazole-b-braun.html (accessed on 15 December 2022).
- Leung, H.W.; Minh, T.B.; Murphy, M.B.; Lam, J.C.W.; So, M.K.; Martin, M.; Lam, P.K.S.; Richardson, B.J. Distribution, Fate and Risk Assessment of Antibiotics in Sewage Treatment Plants in Hong Kong, South China. Environ. Int. 2012, 42, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Regitano, J.B.; Leal, R.M.P. Comportamento e impacto ambiental de antibióticos usados na produção animal brasileira. Rev. Bras. Ciênc. Solo 2010, 34, 601–616. [Google Scholar] [CrossRef]
- Kolz, A.C.; Moorman, T.B.; Ong, S.K.; Scoggin, K.D.; Douglass, E.A. Degradation and Metabolite Production of Tylosin in Anaerobic and Aerobic Swine-Manure Lagoons. Water Environ. Res. 2005, 77, 49–56. [Google Scholar] [CrossRef]
- Montforts, M.H.M.M.; Kalf, D.F.; van Vlaardingen, P.L.A.; Linders, J.B.H.J. The Exposure Assessment for Veterinary Medicinal Products. Sci. Total Environ. 1999, 225, 119–133. [Google Scholar] [CrossRef]
- Carvalho, I.T.; Santos, L. Antibiotics in the Aquatic Environments: A Review of the European Scenario. Environ. Int. 2016, 94, 736–757. [Google Scholar] [CrossRef]
- Pulicharla, R.; Brar, S.K.; Drogui, P.; Verma, M.; Surampalli, R.Y. Removal Processes of Antibiotics in Waters and Wastewaters: Crucial Link to Physical-Chemical Properties and Degradation. J. Hazard. Toxic Radioact. Waste 2015, 19, 04015008. [Google Scholar] [CrossRef]
- Blair, B.; Nikolaus, A.; Hedman, C.; Klaper, R.; Grundl, T. Evaluating the Degradation, Sorption, and Negative Mass Balances of Pharmaceuticals and Personal Care Products during Wastewater Treatment. Chemosphere 2015, 134, 395–401. [Google Scholar] [CrossRef]
- Burch, K.D.; Han, B.; Pichtel, J.; Zubkov, T. Removal Efficiency of Commonly Prescribed Antibiotics via Tertiary Wastewater Treatment. Environ. Sci. Pollut. Res. 2019, 26, 6301–6310. [Google Scholar] [CrossRef]
- Wang, J.; Wang, S. Removal of Pharmaceuticals and Personal Care Products (PPCPs) from Wastewater: A Review. J. Environ. Manag. 2016, 182, 620–640. [Google Scholar] [CrossRef]
- Verlicchi, P.; Al Aukidy, M.; Zambello, E. Occurrence of Pharmaceutical Compounds in Urban Wastewater: Removal, Mass Load and Environmental Risk after a Secondary Treatment—A Review. Sci. Total Environ. 2012, 429, 123–155. [Google Scholar] [CrossRef]
- Buta, M.; Hubeny, J.; Zieliński, W.; Harnisz, M.; Korzeniewska, E. Sewage Sludge in Agriculture—The Effects of Selected Chemical Pollutants and Emerging Genetic Resistance Determinants on the Quality of Soil and Crops—A Review. Ecotoxicol. Environ. Saf. 2021, 214, 112070. [Google Scholar] [CrossRef]
- McClellan, K.; Halden, R.U. Pharmaceuticals and Personal Care Products in Archived U.S. Biosolids from the 2001 EPA National Sewage Sludge Survey. Water Res. 2010, 44, 658–668. [Google Scholar] [CrossRef]
- An, J.; Chen, H.; Wei, S.; Gu, J. Antibiotic Contamination in Animal Manure, Soil, and Sewage Sludge in Shenyang, Northeast China. Environ. Earth Sci. 2015, 74, 5077–5086. [Google Scholar] [CrossRef]
- Cheng, M.; Wu, L.; Huang, Y.; Luo, Y.; Christie, P. Total Concentrations of Heavy Metals and Occurrence of Antibiotics in Sewage Sludges from Cities throughout China. J. Soils Sediments 2014, 14, 1123–1135. [Google Scholar] [CrossRef]
- Hörsing, M.; Ledin, A.; Grabic, R.; Fick, J.; Tysklind, M.; la Jansen, J.C.; Andersen, H.R. Determination of Sorption of Seventy-Five Pharmaceuticals in Sewage Sludge. Water Res. 2011, 45, 4470–4482. [Google Scholar] [CrossRef]
- Economou, V.; Gousia, P. Agriculture and Food Animals as a Source of Antimicrobial-Resistant Bacteria. Infect. Drug Resist. 2015, 8, 49–91. [Google Scholar] [CrossRef]
- Baptiste, K.E. Do Antimicrobial Mass Medications Work? A Systematic Review and Meta-Analysis of Randomized Clinical Trials Investigating Antimicrobial Prophylaxis or Metaphylaxis against Naturally Occurring Bovine Respiratory Disease. Pathog. Dis. 2017, 29, ftx083. [Google Scholar] [CrossRef]
- Hosain, M.Z.; Kabir, S.M.L.; Kamal, M.M. Antimicrobial Uses for Livestock Production in Developing Countries. Vet. World 2021, 14, 210–221. [Google Scholar] [CrossRef]
- Spielmeyer, A. Occurrence and Fate of Antibiotics in Manure during Manure Treatments: A Short Review. Sustain. Chem. Pharm. 2018, 9, 76–86. [Google Scholar] [CrossRef]
- Zhang, X.; Li, Y.; Liu, B.; Wang, J.; Feng, C.; Gao, M.; Wang, L. Prevalence of Veterinary Antibiotics and Antibiotic-Resistant Escherichia Coli in the Surface Water of a Livestock Production Region in Northern China. PLoS ONE 2014, 9, e111026. [Google Scholar] [CrossRef] [PubMed]
- Slana, M.; Pahor, V.; Cvitkovič Maričič, L.; Sollner-Dolenc, M. Excretion Pattern of Enrofloxacin after Oral Treatment of Chicken Broilers. J. Vet. Pharmacol. Ther. 2014, 37, 611–614. [Google Scholar] [CrossRef] [PubMed]
- Ince, B.; Coban, H.; Turker, G.; Ertekin, E.; Ince, O. Effect of Oxytetracycline on Biogas Production and Active Microbial Populations during Batch Anaerobic Digestion of Cow Manure. Bioprocess Biosyst. Eng. 2013, 36, 541–546. [Google Scholar] [CrossRef]
- Qiu, J.; Zhao, T.; Liu, Q.; He, J.; He, D.; Wu, G.; Li, Y.; Jiang, C.; Xu, Z. Residual Veterinary Antibiotics in Pig Excreta after Oral Administration of Sulfonamides. Environ. Geochem. Health 2016, 38, 549–556. [Google Scholar] [CrossRef]
- Wei, R.; He, T.; Zhang, S.; Zhu, L.; Shang, B.; Li, Z.; Wang, R. Occurrence of Seventeen Veterinary Antibiotics and Resistant Bacterias in Manure-Fertilized Vegetable Farm Soil in Four Provinces of China. Chemosphere 2019, 215, 234–240. [Google Scholar] [CrossRef]
- Kumar, K.; Gupta, S.C.; Baidoo, S.K.; Chander, Y.; Rosen, C.J. Antibiotic Uptake by Plants from Soil Fertilized with Animal Manure. J. Environ. Qual. 2005, 34, 2082–2085. [Google Scholar] [CrossRef]
- Laconi, A.; Mughini-Gras, L.; Tolosi, R.; Grilli, G.; Trocino, A.; Carraro, L.; Di Cesare, F.; Cagnardi, P.; Piccirillo, A. Microbial Community Composition and Antimicrobial Resistance in Agricultural Soils Fertilized with Livestock Manure from Conventional Farming in Northern Italy. Sci. Total Environ. 2021, 760, 143404. [Google Scholar] [CrossRef]
- Lenart-Boroń, A.; Prajsnar, J.; Boroń, P. Survival and Antibiotic Resistance of Bacteria in Artificial Snow Produced from Contaminated Water. Water Environ. Res. 2017, 89, 2059–2069. [Google Scholar] [CrossRef]
- Segawa, T.; Takeuchi, N.; Rivera, A.; Yamada, A.; Yoshimura, Y.; Barcaza, G.; Shinbori, K.; Motoyama, H.; Kohshima, S.; Ushida, K. Distribution of Antibiotic Resistance Genes in Glacier Environments: Antibiotic Resistance Genes in Snow and Ice. Environ. Microbiol. Rep. 2013, 5, 127–134. [Google Scholar] [CrossRef]
- Yang, S.; Carlson, K. Evolution of Antibiotic Occurrence in a River through Pristine, Urban and Agricultural Landscapes. Water Res. 2003, 37, 4645–4656. [Google Scholar] [CrossRef]
- Jalloul, G.; Keniar, I.; Tehrani, A.; Boyadjian, C. Antibiotics Contaminated Irrigation Water: An Overview on Its Impact on Edible Crops and Visible Light Active Titania as Potential Photocatalysts for Irrigation Water Treatment. Front. Environ. Sci. 2021, 9, 767963. [Google Scholar] [CrossRef]
- Pan, M.; Chu, L.M. Fate of Antibiotics in Soil and Their Uptake by Edible Crops. Sci. Total Environ. 2017, 599–600, 500–512. [Google Scholar] [CrossRef]
- Yang, Q.; Gao, Y.; Ke, J.; Show, P.L.; Ge, Y.; Liu, Y.; Guo, R.; Chen, J. Antibiotics: An Overview on the Environmental Occurrence, Toxicity, Degradation, and Removal Methods. Bioengineered 2021, 12, 7376–7416. [Google Scholar] [CrossRef]
- Mitchell, S.M. PH and Temperature Effects on the Hydrolysis of Three β-Lactam Antibiotics: Ampicillin, Cefalotin and Cefoxitin. Sci. Total Environ. 2014, 9, 547–555. [Google Scholar] [CrossRef]
- Xuan, R.; Arisi, L.; Wang, Q.; Yates, S.R.; Biswas, K.C. Hydrolysis and Photolysis of Oxytetracycline in Aqueous Solution. J. Environ. Sci. Health Part B 2009, 45, 73–81. [Google Scholar] [CrossRef]
- Fang, L.; Zhou, Y.; Huang, Z.; Yang, G.; Li, T.; Song, C.; Chen, J. Dynamic Elimination of Enrofloxacin Under Varying Temperature and PH in Aquaculture Water: An Orthogonal Study. Bull. Environ. Contam. Toxicol. 2021, 106, 866–872. [Google Scholar] [CrossRef]
- Jiang, M.; Wang, L.; Ji, R. Biotic and Abiotic Degradation of Four Cephalosporin Antibiotics in a Lake Surface Water and Sediment. Chemosphere 2010, 80, 1399–1405. [Google Scholar] [CrossRef]
- Reis, A.C.; Kolvenbach, B.A.; Nunes, O.C.; Corvini, P.F.X. Biodegradation of Antibiotics: The New Resistance Determinants—Part I. New Biotechnol. 2020, 54, 34–51. [Google Scholar] [CrossRef]
- Timm, A.; Borowska, E.; Majewsky, M.; Merel, S.; Zwiener, C.; Bräse, S.; Horn, H. Photolysis of Four Β-lactam Antibiotics under Simulated Environmental Conditions: Degradation, Transformation Products and Antibacterial Activity. Sci. Total Environ. 2019, 651, 1605–1612. [Google Scholar] [CrossRef]
- Liu, X.; Lv, K.; Deng, C.; Yu, Z.; Shi, J.; Johnson, A.C. Persistence and Migration of Tetracycline, Sulfonamide, Fluoroquinolone, and Macrolide Antibiotics in Streams Using a Simulated Hydrodynamic System. Environ. Pollut. 2019, 252, 1532–1538. [Google Scholar] [CrossRef] [PubMed]
- Wei, C.; Li, X.; Xie, Y.; Wang, X. Direct Photo Transformation of Tetracycline and Sulfanomide Group Antibiotics in Surface Water: Kinetics, Toxicity and Site Modeling. Sci. Total Environ. 2019, 686, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Andreozzi, R.; Raffaele, M.; Nicklas, P. Pharmaceuticals in STP Effluents and Their Solar Photodegradation in Aquatic Environment. Chemosphere 2003, 50, 1319–1330. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.-C.; Hsiao, K.-W.; Lin, A.Y.-C. Photolytic Degradation of Ciprofloxacin in Solid and Aqueous Environments: Kinetics, Phototransformation Pathways, and Byproducts. Environ. Sci. Pollut. Res. 2018, 25, 2303–2312. [Google Scholar] [CrossRef]
- Kwon, J.-W. Environmental Impact Assessment of Veterinary Drug on Fish Aquaculture for Food Safety: Environmental Impact Assessment on Veterinary Drugs for Aquaculture. Drug Test. Anal. 2016, 8, 556–564. [Google Scholar] [CrossRef]
- Han, S.; Li, X.; Huang, H.; Wang, T.; Wang, Z.; Fu, X.; Zhou, Z.; Du, P.; Li, X. Simultaneous Determination of Seven Antibiotics and Five of Their Metabolites in Municipal Wastewater and Evaluation of Their Stability under Laboratory Conditions. Int. J. Env. Res. Public Health 2021, 14, 10640. [Google Scholar] [CrossRef]
- Martín, B.S.; Cornejo, J.; Iragüen, D.; Hidalgo, H.; Anadón, A. Depletion Study of Enrofloxacin and Its Metabolite Ciprofloxacin in Edible Tissues and Feathers of White Leghorn Hens by Liquid Chromatography Coupled with Tandem Mass Spectrometry. J. Food Prot. 2007, 70, 1952–1957. [Google Scholar] [CrossRef]
- Jiao, S.; Zheng, S.; Yin, D.; Wang, L.; Chen, L. Aqueous Photolysis of Tetracycline and Toxicity of Photolytic Products to Luminescent Bacteria. Chemosphere 2008, 73, 377–382. [Google Scholar] [CrossRef]
- He, X.; Mezyk, S.P.; Michael, I.; Fatta-Kassinos, D.; Dionysiou, D.D. Degradation Kinetics and Mechanism of β-Lactam Antibiotics by the Activation of H2O2 and Na2S2O8 under UV-254 nm Irradiation. J. Hazard. Mater. 2014, 279, 375–383. [Google Scholar] [CrossRef]
- Pils, J.R.V.; Laird, D.A. Sorption of Tetracycline and Chlortetracycline on K- and Ca-Saturated Soil Clays, Humic Substances, and Clay−Humic Complexes. Environ. Sci. Technol. 2007, 41, 1928–1933. [Google Scholar] [CrossRef]
- Li, J.; Zhang, K.; Zhang, H. Adsorption of Antibiotics on Microplastics. Environ. Pollut. 2018, 237, 460–467. [Google Scholar] [CrossRef]
- Wang, S.; Xue, N.; Li, W.; Zhang, D.; Pan, X.; Luo, Y. Selectively Enrichment of Antibiotics and ARGs by Microplastics in River, Estuary and Marine Waters. Sci. Total Environ. 2020, 708, 134594. [Google Scholar] [CrossRef]
- McCormick, A.R.; Hoellein, T.J.; London, M.G.; Hittie, J.; Scott, J.W.; Kelly, J.J. Microplastic in Surface Waters of Urban Rivers: Concentration, Sources, and Associated Bacterial Assemblages. Ecosphere 2016, 7, e01556. [Google Scholar] [CrossRef]
- Woappi, Y.; Gabani, P.; Singh, A.; Singh, O.V. Antibiotrophs: The Complexity of Antibiotic-Subsisting and Antibiotic-Resistant Microorganisms. Crit. Rev. Microbiol. 2016, 42, 17–30. [Google Scholar] [CrossRef]
- Cha, J.; Carlson, K.H. Biodegradation of Veterinary Antibiotics in Lagoon Waters. Process Saf. Environ. Prot. 2019, 127, 306–313. [Google Scholar] [CrossRef]
- Yang, C.-W.; Liu, C.; Chang, B.-V. Biodegradation of Amoxicillin, Tetracyclines and Sulfonamides in Wastewater Sludge. Water 2020, 12, 2147. [Google Scholar] [CrossRef]
- Wang, J.; Wang, S. Microbial Degradation of Sulfamethoxazole in the Environment. Appl. Microbiol. Biotechnol. 2018, 102, 3573–3582. [Google Scholar] [CrossRef]
- Ding, C.; He, J. Effect of Antibiotics in the Environment on Microbial Populations. Appl. Microbiol. Biotechnol. 2010, 87, 925–941. [Google Scholar] [CrossRef]
- Klaver, A.L.; Matthews, R.A. Effects of Oxytetracycline on Nitrification in a Model Aquatic System. Aquaculture 1994, 123, 237–247. [Google Scholar] [CrossRef]
- Halling-Sørensen, B. Inhibition of Aerobic Growth and Nitrification of Bacteria in Sewage Sludge by Antibacterial Agents. Arch. Environ. Contam. Toxicol. 2001, 40, 451–460. [Google Scholar] [CrossRef]
- Fountoulakis, M.; Drillia, P.; Stamatelatou, K.; Lyberatos, G. Toxic Effect of Pharmaceuticals on Methanogenesis. Water Sci. Technol. 2004, 50, 335–340. [Google Scholar] [CrossRef] [PubMed]
- Córdova-Kreylos, A.L.; Scow, K.M. Effects of Ciprofloxacin on Salt Marsh Sediment Microbial Communities. ISME J. 2007, 1, 585–595. [Google Scholar] [CrossRef] [PubMed]
- Özel Duygan, B.D.; Gaille, C.; Fenner, K.; van der Meer, J.R. Assessing Antibiotics Biodegradation and Effects at Sub-Inhibitory Concentrations by Quantitative Microbial Community Deconvolution. Front. Environ. Sci. 2021, 9, 737247. [Google Scholar] [CrossRef]
- Kümmerer, K. Antibiotics in the Aquatic Environment—A Review—Part II. Chemosphere 2009, 75, 435–441. [Google Scholar] [CrossRef]
- Baquero, F.; Martínez, J.-L.; Cantón, R. Antibiotics and Antibiotic Resistance in Water Environments. Curr. Opin. Biotechnol. 2008, 19, 260–265. [Google Scholar] [CrossRef]
- Khan, G.A.; Berglund, B.; Khan, K.M.; Lindgren, P.-E.; Fick, J. Occurrence and Abundance of Antibiotics and Resistance Genes in Rivers, Canal and near Drug Formulation Facilities—A Study in Pakistan. PLoS ONE 2013, 8, e62712. [Google Scholar] [CrossRef]
- Fram, M.S.; Belitz, K. Occurrence and Concentrations of Pharmaceutical Compounds in Groundwater Used for Public Drinking-Water Supply in California. Sci. Total Environ. 2011, 409, 3409–3417. [Google Scholar] [CrossRef]
- Jiang, L.; Hu, X.; Xu, T.; Zhang, H.; Sheng, D.; Yin, D. Prevalence of Antibiotic Resistance Genes and Their Relationship with Antibiotics in the Huangpu River and the Drinking Water Sources, Shanghai, China. Sci. Total Environ. 2013, 458–460, 267–272. [Google Scholar] [CrossRef]
- Gullberg, E.; Cao, S.; Berg, O.G.; Ilbäck, C.; Sandegren, L.; Hughes, D.; Andersson, D.I. Selection of Resistant Bacteria at Very Low Antibiotic Concentrations. PLoS Pathog. 2011, 7, e1002158. [Google Scholar] [CrossRef]
- Aminov, R.I. The Role of Antibiotics and Antibiotic Resistance in Nature. Environ. Microbiol. 2009, 11, 2970–2988. [Google Scholar] [CrossRef]
- Andersson, D.I.; Hughes, D. Microbiological Effects of Sublethal Levels of Antibiotics. Nat. Rev. Microbiol. 2014, 12, 465–478. [Google Scholar] [CrossRef]
- Davies, J.; Spiegelman, G.B.; Yim, G. The World of Subinhibitory Antibiotic Concentrations. Curr. Opin. Microbiol. 2006, 9, 445–453. [Google Scholar] [CrossRef]
- Kümmerer, K. Antibiotics in the Aquatic Environment—A Review—Part I. Chemosphere 2009, 75, 417–434. [Google Scholar] [CrossRef]
- Sharma, D.; Misba, L.; Khan, A.U. Antibiotics versus Biofilm: An Emerging Battleground in Microbial Communities. Antimicrob. Resist. Infect. Control 2019, 8, 76. [Google Scholar] [CrossRef]
- Bagge, N.; Schuster, M.; Hentzer, M.; Ciofu, O.; Givskov, M.; Greenberg, E.P.; Høiby, N. Pseudomonas Aeruginosa Biofilms Exposed to Imipenem Exhibit Changes in Global Gene Expression and β-Lactamase and Alginate Production. Antimicrob. Agents Chemother. 2004, 48, 1175–1187. [Google Scholar] [CrossRef]
- Hoffman, L.R.; D’Argenio, D.A.; MacCoss, M.J.; Zhang, Z.; Jones, R.A.; Miller, S.I. Aminoglycoside Antibiotics Induce Bacterial Biofilm Formation. Nature 2005, 436, 1171–1175. [Google Scholar] [CrossRef]
- Linares, J.F.; Gustafsson, I.; Baquero, F.; Martinez, J.L. Antibiotics as Intermicrobial Signaling Agents Instead of Weapons. Proc. Natl. Acad. Sci. USA 2006, 103, 19484–19489. [Google Scholar] [CrossRef]
- Sailer, F.C.; Meberg, B.M.; Young, K.D. Î2-Lactam Induction of Colanic Acid Gene Expression in Escherichia Coli. FEMS Microbiol. Lett. 2003, 226, 245–249. [Google Scholar] [CrossRef]
- Crane, J.K.; Alvarado, C.L.; Sutton, M.D. Role of the SOS Response in the Generation of Antibiotic Resistance In Vivo. Antimicrob. Agents Chemother. 2021, 65, e00013-21. [Google Scholar] [CrossRef]
- Qin, T.-T.; Kang, H.-Q.; Ma, P.; Li, P.-P.; Huang, L.-Y.; Gu, B. SOS Response and Its Regulation on the Fluoroquinolone Resistance. Ann. Transl. Med. 2015, 3, 17. [Google Scholar]
- Beaber, J.W.; Hochhut, B.; Waldor, M.K. SOS Response Promotes Horizontal Dissemination of Antibiotic Resistance Genes. Nature 2004, 427, 72–74. [Google Scholar] [CrossRef] [PubMed]
- Crofts, T.S.; Gasparrini, A.J.; Dantas, G. Next-Generation Approaches to Understand and Combat the Antibiotic Resistome. Nat. Rev. Microbiol. 2017, 15, 422–434. [Google Scholar] [CrossRef] [PubMed]
- Berglund, B. Environmental Dissemination of Antibiotic Resistance Genes and Correlation to Anthropogenic Contamination with Antibiotics. Infect. Ecol. Epidemiol. 2015, 5, 28564. [Google Scholar] [CrossRef] [PubMed]
- Wellington, E.M.; Boxall, A.B.; Cross, P.; Feil, E.J.; Gaze, W.H.; Hawkey, P.M.; Johnson-Rollings, A.S.; Jones, D.L.; Lee, N.M.; Otten, W.; et al. The Role of the Natural Environment in the Emergence of Antibiotic Resistance in Gram-Negative Bacteria. Lancet Infect. Dis. 2013, 13, 155–165. [Google Scholar] [CrossRef]
- Grenni, P.; Ancona, V.; Barra Caracciolo, A. Ecological Effects of Antibiotics on Natural Ecosystems: A Review. Microchem. J. 2018, 136, 25–39. [Google Scholar] [CrossRef]
- Felis, E.; Kalka, J.; Sochacki, A.; Kowalska, K.; Bajkacz, S.; Harnisz, M.; Korzeniewska, E. Antimicrobial Pharmaceuticals in the Aquatic Environment—Occurrence and Environmental Implications. Eur. J. Pharmacol. 2020, 866, 172813. [Google Scholar] [CrossRef]
- Cairns, J.; Ruokolainen, L.; Hultman, J.; Tamminen, M.; Virta, M.; Hiltunen, T. Ecology Determines How Low Antibiotic Concentration Impacts Community Composition and Horizontal Transfer of Resistance Genes. Commun. Biol. 2018, 1, 35. [Google Scholar] [CrossRef]
- Zhou, Z.; Zhang, Z.; Feng, L.; Zhang, J.; Li, Y.; Lu, T.; Qian, H. Adverse Effects of Levofloxacin and Oxytetracycline on Aquatic Microbial Communities. Sci. Total Environ. 2020, 734, 139499. [Google Scholar] [CrossRef]
- Waiser, M.J.; Swerhone, G.D.W.; Roy, J.; Tumber, V.; Lawrence, J.R. Effects of Erythromycin, Trimethoprim and Clindamycin on Attached Microbial Communities from an Effluent Dominated Prairie Stream. Ecotoxicol. Environ. Saf. 2016, 132, 31–39. [Google Scholar] [CrossRef]
- Ghoshdastidar, A.J.; Fox, S.; Tong, A.Z. The Presence of the Top Prescribed Pharmaceuticals in Treated Sewage Effluents and Receiving Waters in Southwest Nova Scotia, Canada. Environ. Sci. Pollut. Res. 2015, 22, 689–700. [Google Scholar] [CrossRef]
- Ni, B.-J.; Zeng, S.; Wei, W.; Dai, X.; Sun, J. Impact of Roxithromycin on Waste Activated Sludge Anaerobic Digestion: Methane Production, Carbon Transformation and Antibiotic Resistance Genes. Sci. Total Environ. 2020, 703, 134899. [Google Scholar] [CrossRef]
- Michael, I.; Rizzo, L.; McArdell, C.S.; Manaia, C.M.; Merlin, C.; Schwartz, T.; Dagot, C.; Fatta-Kassinos, D. Urban Wastewater Treatment Plants as Hotspots for the Release of Antibiotics in the Environment: A Review. Water Res. 2013, 47, 957–995. [Google Scholar] [CrossRef]
- Le-Clech, P. Membrane Bioreactors and Their Uses in Wastewater Treatments. Appl. Microbiol. Biotechnol. 2010, 88, 1253–1260. [Google Scholar] [CrossRef]
- Yang, W.; Cicek, N. Treatment of Swine Wastewater by Submerged Membrane Bioreactors with Consideration of Estrogenic Activity Removal. Desalination 2008, 231, 200–208. [Google Scholar] [CrossRef]
- Marti, E.; Monclús, H.; Jofre, J.; Rodriguez-Roda, I.; Comas, J.; Balcázar, J.L. Removal of Microbial Indicators from Municipal Wastewater by a Membrane Bioreactor (MBR). Bioresour. Technol. 2011, 102, 5004–5009. [Google Scholar] [CrossRef]
- Kovalova, L.; Siegrist, H.; Singer, H.; Wittmer, A.; McArdell, C.S. Hospital Wastewater Treatment by Membrane Bioreactor: Performance and Efficiency for Organic Micropollutant Elimination. Environ. Sci. Technol. 2012, 46, 1536–1545. [Google Scholar] [CrossRef]
- Xiao, Y.; Yaohari, H.; De Araujo, C.; Sze, C.C.; Stuckey, D.C. Removal of Selected Pharmaceuticals in an Anaerobic Membrane Bioreactor (AnMBR) with/without Powdered Activated Carbon (PAC). Chem. Eng. J. 2017, 321, 335–345. [Google Scholar] [CrossRef]
- Xia, S.; Jia, R.; Feng, F.; Xie, K.; Li, H.; Jing, D.; Xu, X. Effect of Solids Retention Time on Antibiotics Removal Performance and Microbial Communities in an A/O-MBR Process. Bioresour. Technol. 2012, 106, 36–43. [Google Scholar] [CrossRef]
- Li, B.; Zhang, T. Mass Flows and Removal of Antibiotics in Two Municipal Wastewater Treatment Plants. Chemosphere 2011, 83, 1284–1289. [Google Scholar] [CrossRef]
- Liu, D.; Song, K.; Xie, G.; Li, L. MBR-UV/Cl2 System in Treating Polluted Surface Water with Typical PPCP Contamination. Sci. Rep. 2020, 10, 8835. [Google Scholar] [CrossRef]
- Alacabey, İ. Antibiotic Removal from the Aquatic Environment with Activated Carbon Produced from Pumpkin Seeds. Molecules 2022, 27, 1380. [Google Scholar] [CrossRef] [PubMed]
- Yu, F.; Li, Y.; Han, S.; Ma, J. Adsorptive Removal of Antibiotics from Aqueous Solution Using Carbon Materials. Chemosphere 2016, 153, 365–385. [Google Scholar] [CrossRef] [PubMed]
- Tambosi, J.L.; de Sena, R.F.; Gebhardt, W.; Moreira, R.F.P.M.; José, H.J.; Schröder, H.F. Physicochemical and Advanced Oxidation Processes—A Comparison of Elimination Results of Antibiotic Compounds Following an MBR Treatment. Ozone Sci. Eng. 2009, 31, 428–435. [Google Scholar] [CrossRef]
- Watkinson, A.J.; Murby, E.J.; Kolpin, D.W.; Costanzo, S.D. The Occurrence of Antibiotics in an Urban Watershed: From Wastewater to Drinking Water. Sci. Total Environ. 2009, 407, 2711–2723. [Google Scholar] [CrossRef]
- Dolar, D.; Gros, M.; Rodriguez-Mozaz, S.; Moreno, J.; Comas, J.; Rodriguez-Roda, I.; Barceló, D. Removal of Emerging Contaminants from Municipal Wastewater with an Integrated Membrane System, MBR–RO. J. Hazard. Mater. 2012, 239–240, 64–69. [Google Scholar] [CrossRef]
- Dong, X.; Li, Y.; Li, D.; Liao, D.; Quin, T.; Prakash, O.; Kumar, A.; Liu, J. A New 3D 8-Connected Cd(Ii) MOF as a Potent Photocatalyst for Oxytetracycline Antibiotic Degradation. CrystEngComm 2022, 24, 6933–6943. [Google Scholar] [CrossRef]
- Naseer, M.N.; Jaafar, J.; Junoh, H.; Zaidi, A.A.; Kumar, M.; Alqahtany, A.; Jamil, R.; Alyami, S.H.; Aldossary, N.A. Metal-Organic Frameworks for Wastewater Decontamination: Discovering Intellectual Structure and Research Trends. Materials 2022, 15, 5053. [Google Scholar] [CrossRef]
- Saddique, Z.; Imran, M.; Javaid, A.; Rizvi, N.B.; Akhtar, M.N.; Iqbal, H.M.N.; Bilal, M. Enzyme-Linked Metal Organic Frameworks for Biocatalytic Degradation of Antibiotics. Catal. Lett. 2023. [Google Scholar] [CrossRef]
- Nosakhare Amenaghawon, A.; Lewis Anyalewechi, C.; Uyi Osazuwa, O.; Agbovhimen Elimian, E.; Oshiokhai Eshiemogie, S.; Kayode Oyefolu, P.; Septya Kusuma, H. A Comprehensive Review of Recent Advances in the Synthesis and Application of Metal-Organic Frameworks (MOFs) for the Adsorptive Sequestration of Pollutants from Wastewater. Sep. Purif. Technol. 2023, 311, 123246. [Google Scholar] [CrossRef]
- Liu, X.; Shan, Y.; Zhang, S.; Kong, Q.; Pang, H. Application of Metal Organic Framework in Wastewater Treatment. Green Energy Environ. 2022, in press. [Google Scholar] [CrossRef]
- Li, J.; Wang, H.; Yuan, X.; Zhang, J.; Chew, J.W. Metal-Organic Framework Membranes for Wastewater Treatment and Water Regeneration. Coord. Chem. Rev. 2020, 404, 213116. [Google Scholar] [CrossRef]
- Zhou, Y.; Yang, Q.; Zhang, D.; Gan, N.; Li, Q.; Cuan, J. Detection and Removal of Antibiotic Tetracycline in Water with a Highly Stable Luminescent MOF. Sens. Actuators B Chem. 2018, 262, 137–143. [Google Scholar] [CrossRef]
- Grela, A.; Kuc, J.; Bajda, T. A Review on the Application of Zeolites and Mesoporous Silica Materials in the Removal of Non-Steroidal Anti-Inflammatory Drugs and Antibiotics from Water. Materials 2021, 14, 4994. [Google Scholar] [CrossRef]
- Blasioli, S.; Martucci, A.; Paul, G.; Gigli, L.; Cossi, M.; Johnston, C.T.; Marchese, L.; Braschi, I. Removal of Sulfamethoxazole Sulfonamide Antibiotic from Water by High Silica Zeolites: A Study of the Involved Host–Guest Interactions by a Combined Structural, Spectroscopic, and Computational Approach. J. Colloid Interface Sci. 2014, 419, 148–159. [Google Scholar] [CrossRef]
- Zuo, X.; Qian, C.; Ma, S.; Xiong, J.; He, J.; Chen, Z. Removal of Sulfonamide Antibiotics from Water by High-Silica ZSM-5. Water Sci. Technol. 2019, 80, 507–516. [Google Scholar] [CrossRef]
- Hacıosmanoğlu, G.G.; Mejías, C.; Bueno, J.M.; Santos, J.L.; Aparicio, I.; Alonso, E. Antibiotic Adsorption by Natural and Modified Clay Minerals as Designer Adsorbents for Wastewater Treatment: A Comprehensive Review. J. Environ. Manag. 2022, 317, 115397. [Google Scholar] [CrossRef]
- Hacıosmanoğlu, G.G.; Arenas, M.; Mejías, C.; Martín, J.; Santos, J.L.; Aparicio, I.; Alonso, E. Adsorption of Fluoroquinolone Antibiotics from Water and Wastewater by Colemanite. Int. J. Environ. Res. Public. Health 2023, 20, 2646. [Google Scholar] [CrossRef]
- Iorhemen, O.; Hamza, R.; Tay, J. Membrane Bioreactor (MBR) Technology for Wastewater Treatment and Reclamation: Membrane Fouling. Membranes 2016, 6, 33. [Google Scholar] [CrossRef]

| Antibiotic | Class | Excretion Level | Reference |
|---|---|---|---|
| Levofloxacin | Fluoroquinolones | 85% (h) | [45] |
| Sulfamethoxazole | Sulfonamides | 12% (h) | [46] |
| Trimethoprim | Nitroimidazoles | 60% (h) | [46] |
| Metronidazole | Nitroimidazoles | 60–80% (h) | [47] |
| Erythromycin | Macrolides | 5% (h) | [48] |
| Ofloxacin | Fluoroquinolones | 80% (h) | [48] |
| Tetracycline | Tetracyclines | 80% (h) | [49] |
| Tylosin | Macrolides | 40% (a) | [50] |
| Oxytetracycline | Tetracyclines | 21% (a) | [51] |
| Chlortetracycline | Tetracyclines | 17–75% (a) | [51] |
| Chemical Groups | Compound | Sample Type | Temp. [°C] | T1/2 | Reference |
|---|---|---|---|---|---|
| Cephalosporin | Cefradine 1st Cefuroxime 2nd Ceftriaxone 3rd Cefepime 4th | lake water | 25 ± 3 | 6.3 d 3.1 d 18.7 d 2.7 d | [83] |
| (Amino)penicillin | Amoxicillin Ampicillin Penicillin V Piperacillin | ultrapure water | 19 ± 0.5 | 3.32 ± 0.61 h 3.89 ± 0.43 h 4.37 ± 0.22 h 6.99 ± 0.45 h | [85] |
| Tetracycline | Tetracycline Oxytetracycline Chlortetracycline | river water river water surface water | 25 ± 1 | 4.15 d 1.82 d 3.35 h | [86] [87] |
| Sulfonamide | Sulfamethoxazole | surface water STP effluents river water | 25 ± 1 Winter 25 ± 1 | 14.22 h 2.4 d 17.8 d | [87] [88] [86] |
| Sulfamethazine | surface water river water | 25 ± 1 | 1.3 d 17.3 d | [87] [86] | |
| Fluoroquinolones | Enrofloxacin | surface water river water | 25 ± 1 | 3.34–6.75 d 8.78 d | [82] [86] |
| Ciprofloxacin | deionized water kaolinite suspension river water | 19± 1 19± 1 25 ± 1 | 0.33 h 1.2 h 5.33 d | [89] [89] [86] | |
| Ofloxacin | STP effluents river water | winter 25 ± 1 | 10.6 d 11.1 d | [88] [86] | |
| Norfloxacin | river water | 25 ± 1 | 5.64 d | [86] | |
| Macrolides | Erythromycin | sea water river water | 18± 2 25 ± 1 | 11.11 d 4.22 d | [90] [86] |
| Roxithromycin | wastewater river water | 4 25 ± 1 | 2.9 d 2.76 d | [91] [86] | |
| Clarithromycin Azithromycin | wastewater | 4 | 2.9 d 4.8 d | [91] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kulik, K.; Lenart-Boroń, A.; Wyrzykowska, K. Impact of Antibiotic Pollution on the Bacterial Population within Surface Water with Special Focus on Mountain Rivers. Water 2023, 15, 975. https://doi.org/10.3390/w15050975
Kulik K, Lenart-Boroń A, Wyrzykowska K. Impact of Antibiotic Pollution on the Bacterial Population within Surface Water with Special Focus on Mountain Rivers. Water. 2023; 15(5):975. https://doi.org/10.3390/w15050975
Chicago/Turabian StyleKulik, Klaudia, Anna Lenart-Boroń, and Kinga Wyrzykowska. 2023. "Impact of Antibiotic Pollution on the Bacterial Population within Surface Water with Special Focus on Mountain Rivers" Water 15, no. 5: 975. https://doi.org/10.3390/w15050975
APA StyleKulik, K., Lenart-Boroń, A., & Wyrzykowska, K. (2023). Impact of Antibiotic Pollution on the Bacterial Population within Surface Water with Special Focus on Mountain Rivers. Water, 15(5), 975. https://doi.org/10.3390/w15050975







