Abstract
Lakes in cold and arid regions are extremely vulnerable to global climate change, and the study of seasonal spatial and temporal fluctuations of lake-groundwater chemistry is of major significance for water resource management and environmental preservation. In this study, we combined hydrogeochemical, multivariate statistical, and spatial interpolation methods to assess spatial and temporal variations of lake and groundwater chemistry in Hulun Lake during the frozen and non-frozen periods. The results show that sodium (Na+) is the most abundant cation in the Hulun Lake area. Bicarbonate (HCO3−) and sulfate (SO42−) are the most predominant anions in the lake, river, and ground water during both seasons. The higher Na+ + K+ concentrations in the frozen season were related to longer circulation time and lower renewable rate. The water chemistry of the lake was of the HCO3-SO4-Cl-Na type and that of groundwater in the east and west regions was of the SO4-Cl-Na and HCO3-Na types, respectively. The chemical compositions of groundwater in the non-frozen season were mainly affected by evaporation and concentration, while rock weathering, evaporation, and human activities jointly controlled groundwater chemical component in the frozen period. Based on hierarchical cluster analysis (HCA) and principal component analysis (PCA) methods, Ca2+, NO3−, and SO42− were identified as the main controlling indicators of the chemical characteristics of groundwater and lake water. The increase of Ca2+ concentration in the center of the lake was related to groundwater discharge along the marginal tectonic fracture zone along the lake shores, which was the potential groundwater discharge area. The unconsolidated aquifer provides recharge channels for groundwater on the eastern side, which has a certain influence on the increase of nutrient concentration (NO3−) in the lake on the eastern shore. This research adds to our rough understanding of the lake-groundwater interaction in Hulun Lake, and provides a scientific foundation for the sustainable use of water resources, as well as the eco-logical integrity preservation in cold and arid regions.
1. Introduction
Lakes play a crucial role in the natural water cycle and serve as an indicator for global climate change [1,2]. According to their geographic location and climatic characteristics, lakes can be categorized as cold and arid lakes or as humid lakes. The former are highly sensitive to climate change and are a key driver of the evolution of the ecogeography of the area around them [3]. Lake shrinkage, declining water quality, and ecological degradation of the watershed are all current issues affected by global warming and human activities [4,5,6]. They pose serious threats to water resource management and protection of the aquatic environment. In cold and arid regions, groundwater recharge is an assignable source of both water and pollutants for lakes, and cause ecological problems, such as lake eutrophication and cyanobacterial outbreaks [7,8]. Due to global warming and permafrost degradation, groundwater circulation and lake-groundwater interaction may have significant differences in frozen and non-frozen seasons. In cold and arid region, especially during the icebound season, groundwater may be the dominant recharge source for the lake [9,10]. Thus, a spatial-temporal characteristics analysis and influencing factors of lake water and groundwater chemistry are critical for understanding water circulation mechanisms and lake-groundwater interaction in cold and arid regions.
As the largest freshwater lake in Northeast China, Hulun Lake is important for water resource sustainability, maintaining biodiversity, and climate control [11]. The water level of Hulun Lake has been decreasing in recent years, and the water quality is deteriorating as a result of global warming and human activities [12,13,14]. Previous studies reported that the primary factors affecting the water balance in Hulun Lake are precipitation and evaporation, although some researchers believe that river runoff is the major influencing factor [15]. The weathering and dissolution of carbonate and evaporite rocks, as well as groundwater discharge are dominant factors controlling the water quantity and quality of Hulun Lake [16]. The high chloride concentration in Hulun Lake might be caused by the discharge of highly chlorinated groundwater with isotope tracer method [17]. However, most previous studies took the groundwater and the lake as two independent research individuals. Few studies integrated surface water with groundwater and analyzed the spatial-temporal differentiation of the hydrochemical characteristics for lake water and groundwater in Hulun Lake. Therefore, it is crucial to characterize the spatial and temporal distribution of the chemical components of lake water and groundwater, as well as the influencing factors in order to reveal the lake-groundwater interaction in Hulun Lake area.
To figure out the physicochemical factors involved in lake-groundwater interactions, researchers use geochemistry [18,19], isotope and temperature tracing [20,21], hydrologic analysis [22], or a building model [23,24,25]. Additionally, the main determinants impacting water quality can be extracted using multivariate statistical approaches. In this study, we combined hydrogeochemistry and multivariate statistical methods to characterize the hydrochemical properties and water-rock interactions of lake water and groundwater during the frozen and non-frozen seasons in Hulun Lake. The primary factors controlling water chemistry were selected to analyze the spatial and temporal variation patterns of lake water and groundwater. The results provide a solid scientific foundation for ecological rehabilitation and sustainable water resource development in the Hulun Lake area.
2. Materials and Methods
2.1. Study Area
Hulun Lake, also referred to as Dalai Nuoer, is the fourth largest freshwater lake in China. It has a medium-temperature semi-arid continental climate because it is situated between the Mongolian Plateau and the Daxinganling Mountains, where it receives little of the humid summer wind from the Pacific Ocean. The average annual temperature in the lake area is −0.2 °C, with maximum and minimum temperatures of 20.8 °C and −23.3 °C, respectively [26]. For up to 180 days a year, the lake is frozen. Average yearly evaporation is 1470 mm, which is significantly greater than the average annual rainfall of 235 mm. Hulun Lake is divided into a steep basin in the west and a gentle basin in the east [27]. The lithology on the western side is mainly composed of medium acidic volcanic rocks and the eastern region is characterized by fine-grained fluvial delta sediments [28]. The aquifers can be classified into two categories: fissured bedrock confined aquifers in the western region. The quaternary unconsolidated unconfined aquifer is distributed in the eastern and southern parts of Hulun Lake. Both types of aquifers have various degrees of impact on the variations in the water level of Hulun Lake [29]. The Kherlan, Urson, and Hailar Rivers are the primary inflow rivers, and the Xinkai River is an outflow river. Every year, nearly 2.83 billion cubic meters of river water replenish the Hulun lake. Surface runoff and groundwater are the main sources of the lake’s replenishment.
2.2. Collection and Analysis of the Samples
During the non-frozen period in mid-July 2021 and the frozen season in early January 2022, we conducted a survey and field sampling around Hulun Lake. We collected 21 sets of groundwater samples from around the lake, 18 sets of lake water samples, and 8 sets of river water samples at both time points. Figure 1 shows the sampling sites.
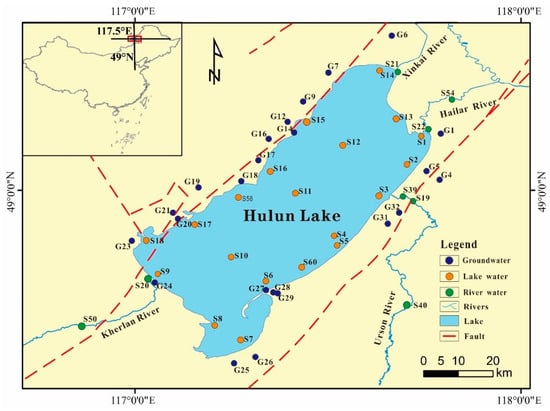
Figure 1.
Plot of the Hulun Lake area and distribution of the sampling points.
We used a 5 L Plexiglas water collector to collect lake and river water samples during the non-frozen season. A spiral ice breaker was utilized to break through the ice during the frozen season to collect water samples from under the ice surface. Groundwater is pumped for about 3 min before sampling in both frozen and non-frozen periods. The water samples were kept in 1 L brown polyethylene plastic bottles that were sealed and placed in a 4 °C refrigerator. We used a simple multi-parameter meter to measure the physical and chemical parameters in the field, including temperature, pH, electrical conductivity, total dissolved solid (TDS) content, dissolved oxygen (DO) content, and salinity (Multi 3630 IDS, WTW, Oberbayern, Germany). The samples were filtered using a 0.45 μm pore size filter membrane. Carbonate (CO32−) and bicarbonate (HCO3−) contents of the samples were obtained by double indicator titration and averaged after three parallel titrations. The trace fractions (Na+, K+, Ca2+, Mg2+, Cl−, SO42−, NO3−) were measured using inductively coupled plasma mass spectrometry (ICS-940, Metrohm, Herisau, Switzerland). The differences between the cation–anion balances of all samples were <5%.
2.3. Research Methodology
2.3.1. Hydrogeochemical Analysis
The hydrogeochemical method [30,31] is used to study the interaction between water and rocks, gases, and organic substances; the nature of their interaction, evolution, and endogenous and exogenous sources; the composition of the underground hydrosphere, and the contribution of the hydrosphere to the evolution of the Earth. Hydrogeochemical research methods can be separated into geological, isotopic, and thermodynamic techniques. In this study, we used water geochemistry methods, a multivariate statistical analysis, and ion scale factor analysis to investigate the reactions between lake water and groundwater and the primary factors that control the ion concentrations in the water.
2.3.2. Multivariate Statistical Methods
The multivariate statistical method of the correlation analysis examines two or more relevant variables and then measures the degree of correlation between them. The correlation coefficient, often symbolized by the symbol r, measures the degree of correlation. It can be used to quantitatively investigate the connection between various groundwater chemical components [32]. According to the degree of correlation, the variables are classified as perfectly correlated, incompletely correlated, or uncorrelated; positively or negatively correlated according to the direction of the correlation; linearly or nonlinearly correlated based on the nature of the correlation equation; and single or multiple correlated according to the number of influencing factors. We conducted Pearson’s correlation analysis of the major anions and cations in groundwater and lake water using SPSS software (version 22.0, IBM, Chicago, IL, USA) to explore the major variables that affect the water chemistry features of the research region. The calculation formulae were as follows [33]:
where r is the correlation coefficient, n is the number of sample variables; xi, yi are the two variables to be analyzed; are the average values of x, y; and σx, σy are the standard deviations of x, y.
2.3.3. Geostatistical Interpolation Methods
The distribution maps of the entire region were deduced from the limited discrete water sample data to transform groundwater quality evaluation from point sampling analysis to regional spatial and temporal evolution analysis and enhancing the effectiveness of the groundwater evaluation [34]. Using the existing data, the primary ions of Lake Hulun groundwater during the frozen and non-frozen periods were spatially interpolated by ArcGIS software (version 10.5, Esri, Redlands, CA, USA) using the inverse distance weighting method, and the formula was as follows [35]:
where ci is the ion concentration at the i point to be calculated, cj is the known ion concentration at the j point to be inserted in the area, n is the number of points in the area, and μj is the weight of the j point in the area:
where dij is the distance from the center of the i point to the j point and is the weight power coefficient.
2.3.4. Hierarchical Cluster Analysis
Cluster analysis is a multivariate statistical method that classifies variables or samples by their own similarities (measured in sample spacing). Rapid clustering and hierarchical clustering are the two fundamental clustering techniques used in the cluster analysis. Hierarchical clustering can be subdivided into single linkage, complete linkage, median linkage, prime linkage, mean linkage, and Ward ‘s minimum variance [36]. In this study, average within-groups linkage was chosen to further understand the groundwater chemical characteristics and contaminant sources in the study area.
2.3.5. Principal Component Analysis Methods
The principal component analysis method is one of the most widely used algorithms for data dimensionality reduction, which linearly converts the original variables into a new set of uncorrelated variables that are sorted in descending order of variance. The variable with the greatest variation is referred to as the first principle component, the variable with the next greatest variance is referred to as the second principal component, and so on. The basic principle is as follows [37]:
With p water samples, each with q indicator variables, a matrix of order p*q is formed.
Using q original variables converted to a small number of new composite variables, the new variables are linear combinations of the original variables. The original variables are x1, x2, …, xq, and the new composite variable indications are y1, y2, …, ym (m ≤ q), then:
The coefficient aij in Equation (5) is dependent on:
yi and yj are independent of one another (i ≠ j, i j = 1, 2, …, q); where y1 (first principal component) is the largest variance among all linear combinations in x1, x2, …, xq; y2 (second principal component) is the largest variance among all linear combinations of x1, x2, …, xq, that are not correlated with y1; ym (mth principal component) is the largest variance among all linear combinations of x1, x2, …, xq, that are not correlated with y1, y2, …, ym−1.
3. Results and Discussion
3.1. Chemical Characteristics of the Various Water Bodies in the Hulun Lake Basin
Table 1 lists the TDS and anion and cation concentrations of lake water, river water, and groundwater from the Hulun Lake Basin during the frozen and non-frozen seasons. The lake water in the basin was weakly alkaline and the groundwater was neutral-alkaline in both frozen and non-frozen seasons. The overall pH trend was lake water > river water > groundwater. The TDS content of lake water varied from 467.0 to 1904.0 mg/L during the frozen period and from 830.0 to 1518.0 mg/L during the non-frozen period; and the TDS content of groundwater varied from 346.0 to 10,740.0 mg/L during the frozen period and from 558.0 to 10,760.0 mg/L during the non-frozen period. The TDS content of river water varied within a smaller range. The variances in TDS content across various water bodies as well as the huge standard deviation of groundwater TDS value and each ion concentration indicates that groundwater chemical components differ significantly. Because the salinity in the lake water was diluted due to sufficient rainfall in summer and the TDS moved from the ice to the water under the ice during the frozen period in winter [38], the TDS concentration in the lake water in the frozen season was higher than that in the non-frozen season, while the TDS concentration in the river water and groundwater was higher in the non-frozen season than that in the frozen season.

Table 1.
Results of water chemistry analysis during the frozen and non-frozen seasons in the Hulun Lake Basin.
DO is the primary sign of a water body’s ability to repair itself. It is also a redox indicator of groundwater and is closely related to oxygen partial pressure in air, water temperature, and salinity. Because the oxygen concentration of the air changes less in its natural condition, the key factor influencing DO content is water temperature; thus, the lower the water temperature, the higher the DO content of the water [39]. The mean values of DO in lake water, river water, and groundwater during the non-frozen period were 9.3, 9.1, and 4.8 mg/L, respectively, and the mean values during the frozen period remained high at 13.9, 13.4, and 5.9 mg/L, respectively. The DO changes in the distinct water bodies in the Hulun Lake Basin showed a high degree of consistency over time, with all values higher during the frozen season compared to the non-frozen season, likely due to the lower water temperature during the frozen period leading to an increase in DO content in the water body.
The concentrations of anions and cations in lake water varied between the frozen and non-frozen periods, but the dominant anions and cations were Na+ and HCO3−. The rainfall increased during the non-frozen season, the mean concentration of SO42− in the river water was lower than that in the frozen period due to dilution, and the mean concentration and relative abundance of major cations differed slightly. The trend of cationic concentrations in groundwater during the frozen and non-frozen seasons was the same (Na+ > Mg2+ > Ca2+ > K+), and the dominant anion during the frozen period was SO42−. Overall, Na+ dominated the cations of both groundwater and surface water, and HCO3− was the major anion in surface water. Due to the interaction between atmospheric precipitation and lake sediments, CO2 is produced by microorganisms when degrading soil organic matter. Additionally, the depth of the groundwater table and strong evaporation cause SO42− enrichment and the decomposition of organic matter input by human activities, resulting in higher SO42− concentrations in groundwater compared to surface water, and SO42− [40,41] and HCO3− dominate groundwater anions. The main cations in lake water and groundwater were the same, reflecting the tight link between the two and the potential recharge and discharge relationship. Li Shi [42] used multi-source data to study the dynamic changes of surface water and groundwater in the Hulun Lake Basin, which verified the presence of water exchange between them. The concentration of NO3− in each water body was lower in the frozen season than in the non-frozen period, which is strongly related to the rejection of each pollutant during the ice formation process [38].
3.2. Water Chemistry Characteristics of Each Water Body in the Hulun Lake Basin
Piper diagrams were generated and other analyses of the water chemistry of the surface and groundwater in the Hulun Lake Basin were conducted at different time points (Figure 2). The water samples, with the exception of the river water during the frozen period, component point of the cation diagrams biased toward the Na+ + K+ endmember; the river water samples during the frozen period were close to the Ca2+ endmember. Most of the water samples fell towards the HCO3− + CO32− extremes of the anion composition spectrum, which illustrates the impact of carbonate rocks on the water chemistry of the basin. The anion and cation compositions were less varied during the non-frozen period than during the frozen period with the similar trends.
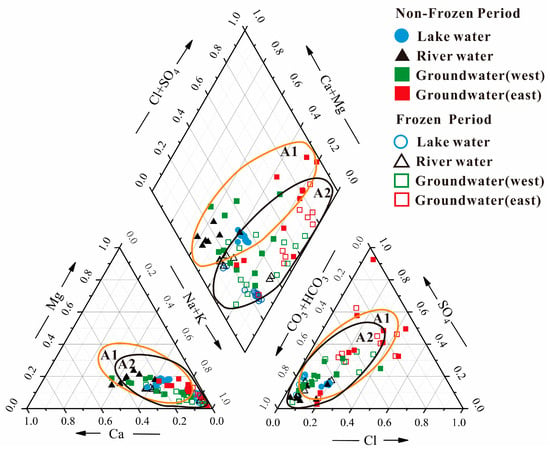
Figure 2.
Piper diagrams of groundwater chemical concentrations in different seasons in each water body in the study area. A1: non-frozen period; A2: frozen period.
The major cation in the lake water during the frozen season was of the HCO3-SO4-Cl-Na type, with Na+ making up 77% of all cations and Ca2+ and Mg2+ present in relatively low amounts; HCO3− was the dominant anion. The lake’s water chemistry was primarily of the HCO3-Na-Ca type. With HCO3-SO4-Cl-Na type water in the lake and HCO3-Na-Ca-Mg as the primary water chemistry of the rivers entering the lake, the water chemistry of the lake water during the non-frozen period differed considerably from that during the frozen season. The water chemistry types during the frozen season were primarily HCO3-Na and SO4-Cl-Na, while that of some groundwater in the western portion of the lake was HCO3-SO4-Cl-Na. The dominant cation of the groundwater in the western and eastern regions of the lake was Na+. The groundwater in the eastern part of the lake was primarily of the SO4-Cl-Na type. During the non-frozen period, the groundwater in the western part of the lake was characterized by seven types of water chemistry, primarily the HCO3-Na-Ca-Mg type and the HCO3-Na type. The groundwater level kept rising due to global warming. Thus, the quaternary unconsolidated aquifer in the eastern region was more liable to be affected by the leaching of surface contaminants, and led to the difference of water types for groundwater in two sides of the lake [43].
In Figure 2, A1 and A2 are the primary distribution zones of each body of water during the non-frozen and frozen periods, respectively. The distribution of the sample sites on the Piper diagram is separated into these two regions. The weakened dynamics of the water column beneath the ice cover, which resulted in less water-rock interaction, caused a significant difference in the ion concentrations at the sampling locations: the cation composition did not differ significantly from one period to the next, but the anions were closer to the HCO3− + CO32− endmember during the frozen period. The groundwater on the western shore of the lake was closely distributed with the lake and river water in the different seasons, and the water chemistry types were similar. This phenomenon suggests that the interactions between the three were somewhat similar. This is primarily due to the strong evaporation in the summer and the influence of wind direction and sediment input from rivers, resulting in lower concentrations of HCO3− and higher concentrations of Cl− during the non-frozen period. The higher Na+ + K+ concentrations in the frozen season were related to longer circulation time, and lower renewable rate.
3.3. Water-Rock Reactions in the Hulun Lake Basin
One of the most crucial techniques for qualitatively identifying the source of a water body’s chemistry is the Gibbs diagram. In our study, the groundwater in the eastern part of Hulun Lake mainly fell outside of the model, indicating that it is more influenced by human activities, whereas the sampling points in each water body in the study area were distributed in the middle and upper parts of the Gibbs diagram, and most of them fell inside the model (Figure 3).
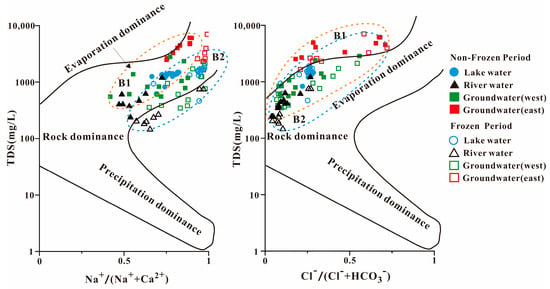
Figure 3.
Gibbs diagram of the main water chemical ions in different seasons in each water body in the study area. B1: non-frozen period; B2: frozen period.
The Cl−/(Cl− + HCO3−) ratio was typically between 0 and 0.5 and was distributed in the area jointly controlled by rock weathering and evaporation concentration, exhibiting characteristics similar to arid zone lakes. In Lake Hulun, the mean values of Na+/(Na+ + Ca2+) were 0.79 and 0.91 in the non-frozen and frozen periods, respectively, and were near the evaporation concentration area. Evapotranspiration and concentration have the biggest effects on the ionic composition of the water in Hulun Lake, with the weathering of rocks playing a supporting role. The same elements that determine the ionic composition of lake water also affect river water. On the west bank of Hulun Lake, rock weathering and evapotranspiration work together to control the ion composition, while evapotranspiration and concentration dominate the water chemistry on the east side. None of the water bodies were plotted in the atmospheric precipitation area, demonstrating that the ionic composition of the water bodies in the Hulun Lake Basin is not controlled by atmospheric precipitation. This is primarily because the basin is located in the Inner Mongolia grasslands, far from the ocean, and the influence of the eastern Daxinganling Mountains reduces the amount of moist air brought by the Pacific summer winds.
As shown in Figure 3, the distribution of the water samples is divided into two sections. B1 and B2 are the major distribution regions for the non-frozen and frozen samples, respectively, which is in accordance with the distribution of each water body sample on the Gibbs plot. The anion Cl−/(Cl− + HCO3−) ratio tended towards the rock weathering area and was less affected by evaporation and concentration, which is consistent with the water chemistry results. The difference in cations between the frozen and non-frozen seasons was small. Some of the locations in the Gibbs plot are outside the model, where they may be altered by ion exchange or human activity [44].
3.4. Multivariate Statistical Analysis of Different Water Bodies in the Hulun Lake Basin
3.4.1. Correlation Analysis
Figure 4 demonstrates the relationship between TDS content and standing ions in lake water and groundwater in the Hulun Lake Basin. The correlation between TDS content and ions in groundwater during the non-frozen period was strong. The strongest correlation was between Cl− and Na+ and Mg2+, which indicates that these ions may be the same component source, implying that weathering and leaching of rocks affect the chemical characteristics of groundwater during the non-frozen period. Additionally, the significant relationship between Ca2+ and SO42− indicates that Ca2+ and SO42− are co-sourced and that the SO42− migration from river water to lake water was due to water-rock interaction.
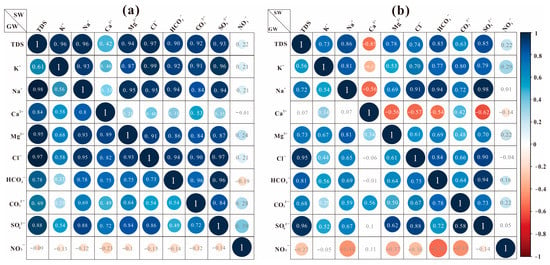
Figure 4.
Correlation of the major ions between lake water and groundwater during the different seasons: (a) non-frozen season; (b) frozen season.
During the frozen season, the association between groundwater TDS content and SO42− was highest, followed by Cl−. When combined with the Gibbs plot, it is clear that the groundwater sample sites are mostly concentrated in the areas of rock weathering and evaporation concentration and that these processes are important controlling factors in groundwater chemical composition. During the frozen period, the TDS content of the lake water was negatively correlated with Ca2+ and NO3−, and positively correlated with other ions. HCO3− contributed the most to TDS content, showing that it was the major source of TDS [41].
Overall, there was a good linear relationship between the TDS ions, and groundwater TDS content was correlated with Cl− and Na+ in both the frozen and non-frozen periods. However, the correlation with Na+ was weaker in the frozen period, indicating that evaporation concentration played an auxiliary role in groundwater chemistry during this season. In contrast, the non-frozen period was primarily controlled by the combination of evaporation concentrators. Cl− was not strongly correlated with other ions throughout the frozen period, indicating that the source of Cl− is complicated. It may include processes, such as dissolution of rock salt. NO3−, mainly influenced by human activities, has a weak correlation with other ions in the lake water and negatively related with major components in groundwater.
3.4.2. Hierarchical Cluster Analysis (HCA)
Ten chemical indicators, Na+, K+, Ca2+, Mg2+, Cl−, SO42−, CO32−, HCO3−, NO3−, and TDS, were selected for cluster analysis. The lake waters in non-frozen season were clustered into two categories and the average distance between groups was 10 (Figure 5a). NO3− were clustered into one category and TDS was clustered into another with other indicators. The results supported the correlation analysis results in Section 3.4.1, which shown that TDS was negatively correlated with NO3− and positively connected with other ions. Cluster 2 might be defined as the impact of human activity, including sewage and livestock production. Contrarily, Cluster 1 was associated with natural factors, including dissolution and evaporation. At an average distance of 10, it is possible to divide the lake water during the frozen period into four clusters (Figure 5b). When the average distance was less than two, Na+, K+, and Cl− were grouped into one category, indicating the similar sources, such as dissolution and cation exchange. Sulfate ions in lake water were divided into different categories in frozen and non-frozen seasons. This is mainly due to the thermal power industry and SO42− were more influenced by human activities in winter. The groundwater in the non-frozen season (Figure 5c) can be divided into two categories at the average distance >18. Indicators in Cluster 1 and Cluster 2 were mainly dominated by the natural factors and human activities, respectively. Four groups of groundwater may be distinguished in the frozen period at an average distance of 15 (Figure 5d). TDS, Cl−, and SO42− were in Cluster 1, which indicated that Cl− and SO42− are the dominant anions in groundwater during the frozen period. CO32− and HCO3− had a high correlation (0.78) according to the correlation analysis results and were classified in the same group.
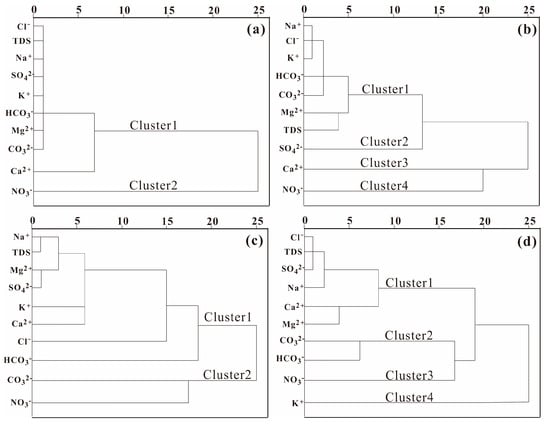
Figure 5.
Hierarchical cluster analysis of the water bodies in Hulun Lake Basin in different seasons: (a) lake water in the non-frozen season; (b) lake water in the frozen season; (c) groundwater in the non-frozen season; (d) groundwater in the frozen season.
3.4.3. Principal Component Analysis (PCA)
PCA results (Figure 6) revealed that the indicators with a high correlation with PC1 included TDS, Na+, Mg2+, CO32−, HCO3−, and SO42−, whereas NO3− had a higher correlation with PC2. HCA analysis results showed that the eight principal ions are mostly related to the dissolution and NO3− is mostly produced by human activity. Thus, the eight main ions and NO3− are therefore somewhat suggestive of natural and anthropogenic influences on the chemical composition of the water. PC1 showed positive loading values for all water chemistry indicators in the non-frozen groundwater, including SO42−, Na+, Mg2+, Ca2+, K+, and TDS ions, and PC2 showed higher loading values for NO3−. PC1 had positive loading values for all water chemistry indicators in the lake water during the non-frozen season. Most of the major chemical ions in lake water and groundwater fell into the fourth quadrant, indicating that groundwater is influenced more by natural factors and less by anthropogenic factors.
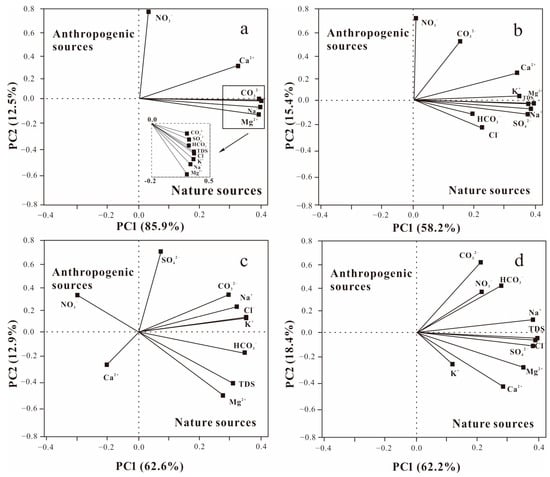
Figure 6.
Principal component analysis of the major ions in water bodies in the Hulun Lake Basin in different seasons: (a) lake water in the non-frozen season; (b) groundwater in the non-frozen season; (c) lake water in the frozen season; (d) groundwater in the frozen season.
The ions in groundwater during the frozen period were primarily distributed in quadrants 1 and 4, with TDS, Cl−, SO42−, K+, Ca2+, and Mg2+ having high positive loading values on PC1 and the loading values of water chemical indexes on PC2 showing some divergence. This shows that the groundwater during the frozen period is primarily influenced by natural factors [45], with anthropogenic factors being auxiliary. The majority of characteristic lake water ions were distributed in the positive half axes of PC1 and PC2, indicating that the lake water was influenced by water-rock interactions and human activities during the frozen period. Overall, the axes of PC1 show that natural factors, such as groundwater dissolution and filtration and salt rock dissolution affect the water chemistry of the water bodies, whereas the axes of PC2 show that anthropogenic factors affect the water chemistry of water bodies. Therefore, the contribution of natural and anthropogenic factors to the variability of water chemistry data can be identified through PC1 and PC2 [46]. Based on the multivariate statistical method [47], we identified three primary ions (Ca2+, NO3−, and SO42−) as the key control variables of natural and anthropogenic influences on the water chemistry features of groundwater.
3.5. Spatial-Temporal Differentiation of Water Chemistry in the Hulun Lake Basin
Figure 7 shows that the differences in the spatial distribution characteristics of the normative ion concentrations in the water bodies of Hulun Lake between the frozen and non-frozen periods were large. Moreover, the spatial patterns of lake water and groundwater may indicate the potential path by which groundwater flows into the lake.
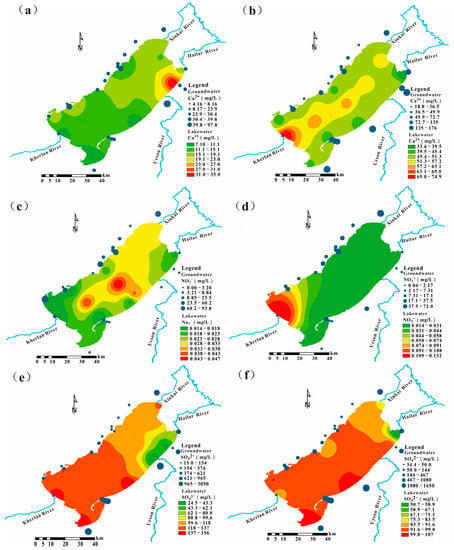
Figure 7.
Spatial interpolation map of the main hydrochemical ions in the Hulun Lake Basin: (a) Ca2+ concentration during the frozen season; (b) Ca2+ concentration during the non-frozen season; (c) NO3− concentration during the frozen season; (d) NO3− concentration during the non-frozen season; (e) SO42− concentration during the frozen season; (f) season SO42− concentration during the non-frozen.
Ca2+ showed a decreasing trend from the north to the south during the frozen period, probably due to the decrease of lake water recharge and mineral input to the lake during the frozen period (Figure 7a,b). At this time of year, the groundwater chemical fraction is the main factor controlling the lake chemistry. In contrast, during the non-frozen season, river runoff increases and the mineral-rich and easily weathered areas on both sides of the Kherlan River lead to increased Ca2+ content. Combined with the regional geological conditions, the marginal fracture ice structure of Hulun Lake during the frozen period is distributed around the lake shore, and the fracture ice body in the center of the lake is developed along the long axis [48]. Therefore, the increase of Ca2+ concentration in the center of the lake was mainly related to groundwater discharge along the marginal tectonic fracture zone from the east and west shores of the lake to the lateral fracture in the center of the lake, which was the potential groundwater discharge area.
As shown in Figure 7c,d, the spatial distribution of NO3− during the frozen period was characterized by a gradual decrease in concentration from the center of the lake to the surrounding area [49]. The NO3− concentration of nutrient salts in the ice body was higher at −25 °C [50]. The aquifer on the eastern shore of the lake is sandy and gravelly and has a fast recharge rate of groundwater. The unconsolidated aquifer also provides a recharge channel for groundwater on the eastern side, which has a certain influence on the increase of nutrient salt concentration in the lake on the eastern shore. The nitrogen pollution in the Kherlan River is serious, which is higher during the non-frozen period compared to the frozen period.
Except for Urson River (which was not frozen when sampled), the concentration of sulfate ions at the inlet of other rivers was higher than the average concentration of sulfate ions in the lake. It may be related to groundwater discharge, which leads to a higher concentration of ions at the mouth of the river. However, the SO42− concentration at the Hailar inlet was low due to the dilution effect of the river recharge (Figure 7e,f).
Our combined data indicate that groundwater from the east and west shores of the lake is recharged to the central part of the lake through the fault, resulting in increased ion concentration in the lake. The sandy aquifers and fault structures on the eastern side are the main recharge channels for groundwater during the non-frozen period, and the shallow aquifers are affected by freezing during the frozen season. Overall, groundwater in Hulun Lake is mainly recharged through subsurface runoff, fracture structures, and fractured aquifer structures during the frozen period; during the non-frozen period, it is mainly recharged through subsurface runoff, fracture structures, and sandy aquifers.
4. Conclusions
The major cation in the lake water during the frozen season was of the HCO3-SO4-Cl-Na type. HCO3-SO4-Cl-Na type and HCO3-Na-Ca-Mg were the primary water chemistry types of the lake during the frozen season. The surface contaminants more easily affected the groundwater in the eastern part of the lake through leaching and this resulted in two different hydrochemical types of SO4-Cl-Na and HCO3-Na in the eastern and western parts of the lake, respectively. During the non-frozen period, evaporation and concentration influenced the chemical composition of groundwater, whereas in the frozen period, rock weathering, evaporation, and concentration dominated the groundwater chemistry.
TDS was negatively correlated with NO3− and positively connected with other ions. Three primary ions (Ca2+, NO3−, and SO42−) were identified as the key control variables for natural and anthropogenic influences on the water chemistry of groundwater and lake water. The groundwater discharge into the center of the lake through the tectonic fault zone may lead to high concentrations of certain ions. In the non-frozen season, groundwater discharges into the lake through the sandy aquifer and tectonic fault around the lake shore. While in the frozen period, deep groundwater might infiltrate into the lake mainly through fissured bedrock aquifer.
To explore the geographical and temporal patterns of water chemistry and the factors that influence lake water and groundwater in the Hulun Lake area, we combined hydrogeochemical, multivariate statistical, and spatial interpolation methods. However, water circulation mechanism still needs advanced research. Thus, long-term groundwater monitoring and isotopic methods should be combined in future research to precisely characterize the relationship between lakes and groundwater in cold and arid regions.
Author Contributions
Formal analysis, B.L. and Y.H.; Investigation, M.X., C.Y. and Y.J.; Methodology, W.L.; Project administration, Y.T.; Supervision, X.Z.; Writing—original draft, W.W. and M.X.; Writing—review & editing, X.G. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by Science and Technology Cooperation Project with Inner Mongolia (2021CG0037), Science and Technology Project of Hulunbuir City (HZ2020003), National Training Program of Innovation for Undergraduates, China (202210304055Z), First Phase of the Project of Strengthening the Scientific and Technological Research Capacity of Hulun Lake Nature Reserve (HSZCS-C-F-210094), and Ecological Security Investigation and Assessment Project of Hulun Lake (HSZCS-G-F-210059), Budget project of the Ministry of Ecology and Environment of the People’s Republic of China: Supervision and Management of Surface Water Ecological Environment, and Construction, operation and maintenance of supporting facilities of Hulun Lake Wetland field observation station (ZX2022QT040).
Institutional Review Board Statement
Not applicable.
Data Availability Statement
Restrictions apply to the availability of these data. Data was obtained from Hulunbuir Academy of Inland Lakes in Northern Cold and Arid Areas and are available from Xiaomin Gu with the permission of Hulunbuir Academy of Inland Lakes in Northern Cold and Arid Areas.
Acknowledgments
We would like to thank the assistance and contribution of Hulunbuir Academy of Inland Lakes in Northern Cold and Arid Areas during field samplings.
Conflicts of Interest
The authors declare that they have no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
References
- JingBo, Z.; YanDong, M.; XiaoQing, L.; DaPeng, Y.; TianJie, S.; ZhiBao, D. The discovery of surface runoff in the megadunes of Badain Jaran Desert, China, and its significance. Sci. China (Earth Sci.) 2017, 60, 707–719. [Google Scholar]
- Vystavna, Y.; Harjung, A.; Monteiro, L.R.; Matiatos, I.; Wassenaar, L.I. Stable isotopes in global lakes integrate catchment and climatic controls on evaporation. Nat. Commun. 2021, 12, 7224. [Google Scholar] [CrossRef] [PubMed]
- Toney, J.L.; García-Alix, A.; Jiménez-Moreno, G.; Anderson, R.S.; Moossen, H.; Seki, O. New insights into Holocene hydrology and temperature from lipid biomarkers in western Mediterranean alpine wetlands. Quat. Sci. Rev. 2020, 240, 106395. [Google Scholar] [CrossRef]
- Riebe, K.; Dressel, A. The impact on food security of a shrinking Lake Chad. J. Arid Environ. 2021, 189, 104486. [Google Scholar] [CrossRef]
- Wei, W.; Gao, Y.; Huang, J.; Gao, J. Exploring the effect of basin land degradation on lake and reservoir water quality in China. J. Clean. Prod. 2020, 268, 122249. [Google Scholar] [CrossRef]
- Woolway, R.I.; Kraemer, B.M.; Lenters, J.D.; Merchant, C.J.; O’Reilly, C.M.; Sharma, S. Global lake responses to climate change. Nat. Rev. Earth Environ. 2020, 1, 388–403. [Google Scholar] [CrossRef]
- Sun, X.; Du, Y.; Deng, Y.; Fan, H.; Tao, Y.; Ma, T. Contribution of groundwater discharge and associated contaminants input to Dongting Lake, Central China, using multiple tracers ((222)Rn, (18)O, Cl(-)). Environ. Geochem. Health 2021, 43, 1239–1255. [Google Scholar] [CrossRef]
- Yu, L.; Rozemeijer, J.C.; Broers, H.P.; van Breukelen, B.M.; Middelburg, J.J.; Ouboter, M.R.L.; van der Velde, Y. Drivers of nitrogen and phosphorus dynamics in a groundwater-fed urban catchment revealed by high-frequency monitoring. Hydrol. Earth Syst. Sci. 2020, 25, 69–87. [Google Scholar] [CrossRef]
- Boreux, M.; Lamoureux, S.; Cumming, B. Use of water isotopes and chemistry to infer the type and degree of exchange between groundwater and lakes in an esker complex of northeastern Ontario, Canada. Hydrol. Earth Syst. Sci. 2021, 25, 6309–6332. [Google Scholar] [CrossRef]
- Wallace, H.; Wexler, E.J.; Malott, S.; Robinson, C.E. Evaluating lacustrine groundwater discharge to a large glacial lake using regional scale radon-222 surveys and groundwater modelling. Hydrol. Process. 2021, 35, e14165. [Google Scholar] [CrossRef]
- Shang, Y.; Wu, X.; Wang, X.; Wei, Q.; Ma, S.; Sun, G.; Zhang, H.; Wang, L.; Dou, H.; Zhang, H. Factors affecting seasonal variation of microbial community structure in Hulun Lake, China. Sci. Total Environ. 2022, 805, 150294. [Google Scholar] [CrossRef]
- Fu, C.; Wu, H.; Zhu, Z.; Song, C.; Xue, B.; Wu, H.; Ji, Z.; Dong, L. Exploring the potential factors on the striking water level variation of the two largest semi-arid-region lakes in northeastern Asia. Catena 2021, 198, 105037. [Google Scholar] [CrossRef]
- Zhao, C.; Zhang, Y.; Guo, W.; Fahad Baqa, M. Dynamics and Drivers of Water Clarity Derived from Landsat and In-Situ Measurement Data in Hulun Lake from 2010 to 2020. Water 2022, 14, 1189. [Google Scholar] [CrossRef]
- Fang, C.; Song, K.; Shang, Y.; Ma, J.; Wen, Z.; Du, J. Remote Sensing of Harmful Algal Blooms Variability for Lake Hulun Using Adjusted FAI (AFAI) Algorithm. J. Environ. Inform. 2018, 34, 108–122. [Google Scholar] [CrossRef]
- Guo, J.; Zhang, Y.; Shi, X.-h.; Sun, B.; Wu, L.; Wang, W. Driving Mechanisms of the Evolution and Ecological Water Demand of Hulun Lake in Inner Mongolia. Water 2022, 14, 3415. [Google Scholar] [CrossRef]
- Han, Z.; Shi, X.; Jia, K.; Sun, B.; Zhao, S.; Fu, C. Determining the Discharge and Recharge Relationships between Lake and Groundwater in Lake Hulun Using Hydrogen and Oxygen Isotopes and Chloride Ions. Water 2019, 11, 264. [Google Scholar] [CrossRef]
- Gao, H.; Ryan, M.C.; Li, C.; Sun, B. Understanding the Role of Groundwater in a Remote Transboundary Lake (Hulun Lake, China). Water 2017, 9, 363. [Google Scholar] [CrossRef]
- Li, J.; Wang, W.; Wang, D.; Li, J.; Dong, J. Hydrochemical and Stable Isotope Characteristics of Lake Water and Groundwater in the Beiluhe Basin, Qinghai–Tibet Plateau. Water 2020, 12, 2269. [Google Scholar] [CrossRef]
- Jia, S.; Dai, Z.; Du, X.; Meng, B.; Yang, Z.; Lan, T. Quantitative evaluation of groundwater and surface water interaction characteristics during a dry season. Water Environ. J. 2021, 35, 1348–1361. [Google Scholar] [CrossRef]
- Saleem, M.; Jeelani, G. Geochemical, isotopic and hydrological mass balance approaches to constrain the lake water–groundwater interaction in Dal Lake, Kashmir Valley. Environ. Earth Sci. 2017, 76, 533. [Google Scholar] [CrossRef]
- Su, X.; Cui, G.; Du, S.; Yuan, W.; Wang, H. Using multiple environmental methods to estimate groundwater discharge into an arid lake (Dakebo Lake, Inner Mongolia, China). Hydrogeol. J. 2016, 24, 1707–1722. [Google Scholar] [CrossRef]
- Ferraz, G.d.F.; Krámer, T. Surface Water–Groundwater Interactions and Bank Storage during Flooding: A Review. Period. Polytech. Civ. Eng. 2021, 66, 149–163. [Google Scholar] [CrossRef]
- Xu, S.; Frey, S.K.; Erler, A.R.; Khader, O.B.; Berg, S.J.; Hwang, H.T.; Callaghan, M.V.; Davison, J.; Sudicky, E.A. Investigating groundwater-lake interactions in the Laurentian Great Lakes with a fully-integrated surface water-groundwater model. J. Hydrol. 2021, 594, 125911. [Google Scholar] [CrossRef]
- Liao, F.; Wang, G.; Shi, Z.; Cheng, G.; Kong, Q.; Mu, W.; Guo, L. Estimation of groundwater discharge and associated chemical fluxes into Poyang Lake, China: Approaches using stable isotopes (δD and δ18O) and radon. Hydrogeol. J. 2018, 26, 1625–1638. [Google Scholar] [CrossRef]
- Yang, X.; Hu, J.; Lu, Z. Integrated Hydrologic Modelling of Groundwater-Surface Water Interactions in Cold Regions. Front. Earth Sci. 2020, 9, 721009. [Google Scholar] [CrossRef]
- Wang, W.; Zhao, L.; Li, W.; Chen, J.; Wang, S. Response mechanism of sediment organic matter of plateau lakes in cold and arid regions to climate change: A case study of Hulun Lake, China. Environ. Sci. Pollut. Res. Int. 2022. [Google Scholar] [CrossRef]
- Xue, B.; Qu, W.; Wang, S.-m.; Ma, Y.; Dickman, M.D. Lake level changes documented by sediment properties and diatom of Hulun Lake, China since the late Glacial. Hydrobiologia 2003, 498, 133–141. [Google Scholar] [CrossRef]
- Xia, S.; Liu, Z.; Liu, J.; Chang, Y.; Li, P.; Gao, N.; Ye, D.; Wu, G.; Yu, L.; Qu, L.; et al. The controlling factors of modern facies distributions in a half-graben lacustrine rift basin: A case study from Hulun Lake, Northeastern China. Geol. J. 2018, 53, 977–991. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, S.; Xu, W.; Zhang, B.-K.; Yi, L.; Lu, X. Geochemical Characteristics and Their Environmental Implications for the Water Regime of Hulun Lake, Inner Mongolia, China. Water 2022, 14, 3696. [Google Scholar] [CrossRef]
- Jeevanandam, M.; Kannan, R.; Srinivasalu, S.; Rammohan, V. Hydrogeochemistry and groundwater quality assessment of lower part of the Ponnaiyar River Basin, Cuddalore district, South India. Environ. Monit. Assess. 2007, 132, 263–274. [Google Scholar] [CrossRef]
- Narsimha, A.; Sudarshan, V. Hydrogeochemistry of groundwater in Basara area, Adilabad District, Andhra Pradesh, India. J. Appl. Geochem. 2013, 15, 224–237. [Google Scholar]
- Loáiciga, H.A.; Charbeneau, R.J.; Everett, L.G.; Fogg, G.E.; Hobbs, B.F.; Rouhani, S. Review of Ground-Water Quality Monitoring Network Design. J. Hydraul. Eng. 1993, 118, 11–37. [Google Scholar] [CrossRef]
- Zhang, B.; Song, X.; Zhang, Y.; Han, D.; Tang, C.; Yu, Y.; Ma, Y. Hydrochemical characteristics and water quality assessment of surface water and groundwater in Songnen plain, Northeast China. Water Res. 2012, 46, 2737–2748. [Google Scholar] [CrossRef]
- Murphy, R.R.; Curriero, F.C.; Ball, W.P. Comparison of spatial interpolation methods for water quality evaluation in the Chesapeake Bay. J. Environ. Eng. 2010, 136, 160–171. [Google Scholar] [CrossRef]
- Shukla, K.K.; Kumar, P.; Mann, G.S.; Khare, M. Mapping spatial distribution of particulate matter using Kriging and Inverse Distance Weighting at supersites of megacity Delhi. Sustain. Cities Soc. 2020, 54, 101997. [Google Scholar] [CrossRef]
- Bu, J.; Liu, W.; Pan, Z.; Ling, K. Comparative Study of Hydrochemical Classification Based on Different Hierarchical Cluster Analysis Methods. Int. J. Environ. Res. Public Health 2020, 17, 9515. [Google Scholar] [CrossRef]
- Yao, R.S.; Yan, Y.; Wei, C.; Luo, M.; Xiao, Y.; Zhang, Y. Hydrochemical Characteristics and Groundwater Quality Assessment Using an Integrated Approach of the PCA, SOM, and Fuzzy c-Means Clustering: A Case Study in the Northern Sichuan Basin. Front. Environ. Sci. 2022, 10, 907872. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, C.; Shi, X.; Li, C. The migration of total dissolved solids during natural freezing process in Ulansuhai Lake. J. Arid Land 2012, 4, 85–94. [Google Scholar] [CrossRef]
- Garizi, A.Z.; Sheikh, V.; Sadoddin, A. Assessment of seasonal variations of chemical characteristics in surface water using multivariate statistical methods. Int. J. Environ. Sci. Technol. 2011, 8, 581–592. [Google Scholar] [CrossRef]
- Luo, A.; Wang, G.; Dong, S.; Wang, H.; Shi, Z.; Ji, Z.; Xue, J. Effect of Large-Scale Mining Drainage on Groundwater Hydrogeochemical Evolution in Semi-Arid and Arid Regions. Front. Environ. Sci. 2022, 10, 926866. [Google Scholar] [CrossRef]
- Gao, Z.-j.; Liu, J.-t.; Feng, J.; Wang, M.; Wu, G. Hydrogeochemical Characteristics and the Suitability of Groundwater in the Alluvial-Diluvial Plain of Southwest Shandong Province, China. Water 2019, 11, 1577. [Google Scholar] [CrossRef]
- Li, S.; Chen, J.; Xiang, J.; Pan, Y.; Huang, Z.; Wu, Y. Water level changes of Hulun Lake in Inner Mongolia derived from Jason satellite data. J. Vis. Commun. Image Represent. 2019, 58, 565–575. [Google Scholar] [CrossRef]
- Donohue, S.; McCarthy, V.; Rafferty, P.; Orr, A.; Flynn, R. Geophysical and hydrogeological characterisation of the impacts of on-site wastewater treatment discharge to groundwater in a poorly productive bedrock aquifer. Sci. Total. Environ. 2015, 523, 109–119. [Google Scholar] [CrossRef] [PubMed]
- Panneerselvam, B.; Paramasivam, S.K.; Karuppannan, S.; Ravichandran, N.; Selvaraj, P. A GIS-based evaluation of hydrochemical characterisation of groundwater in hard rock region, South Tamil Nadu, India. Arab. J. Geosci. 2020, 13, 837. [Google Scholar] [CrossRef]
- Selvakumar, S.; Chandrasekar, N.; Kumar, G. Hydrogeochemical characteristics and groundwater contamination in the rapid urban development areas of Coimbatore, India. Water Resour. Ind. 2017, 17, 26–33. [Google Scholar] [CrossRef]
- Ramalingam, S.; Panneerselvam, B.; Kaliappan, S.P. Effect of high nitrate contamination of groundwater on human health and water quality index in semi-arid region, South India. Arab. J. Geosci. 2022, 15, 242. [Google Scholar] [CrossRef]
- Gu, X.; Xiao, Y.; Yin, S.; Pan, X.; Niu, Y.; Shao, J.; Cui, Y.; Zhang, Q.; Hao, Q. Natural and anthropogenic factors affecting the shallow groundwater quality in a typical irrigation area with reclaimed water, North China Plain. Environ. Monit. Assess. 2017, 189, 514. [Google Scholar] [CrossRef]
- Cai, Z.; Jin, T.; Li, C.; Ofterdinger, U.S.; Zhang, S.; Ding, A.; Li, J. Is China’s fifth-largest inland lake to dry-up? Incorporated hydrological and satellite-based methods for forecasting Hulun lake water levels. Adv. Water Resour. 2016, 94, 185–199. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhao, C.; Yu, A.; Zhao, W.; Ren, F.; Liu, Y. The Migration Pattern of Atrazine during the Processes of Water Freezing and Thawing. Toxics 2022, 10, 603. [Google Scholar] [CrossRef]
- Blackwell, M.S.A.; Brookes, P.C.; Fuente-Martinez, N.d.l.; Gordon, H.; Murray, P.J.; Snars, K.E.; Williams, J.K.; Bol, R.; Haygarth, P.M. Phosphorus solubilization and potential transfer to surface waters from the soil microbial biomass following drying-rewetting and freezing-thawing. Adv. Agron. 2010, 106, 1–35. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).