Comparative Study of Two Organic Wastes as Adsorbents in the Treatment of Water Rich in Nitrogen Compounds
Abstract
:1. Introduction
2. Materials and Methods
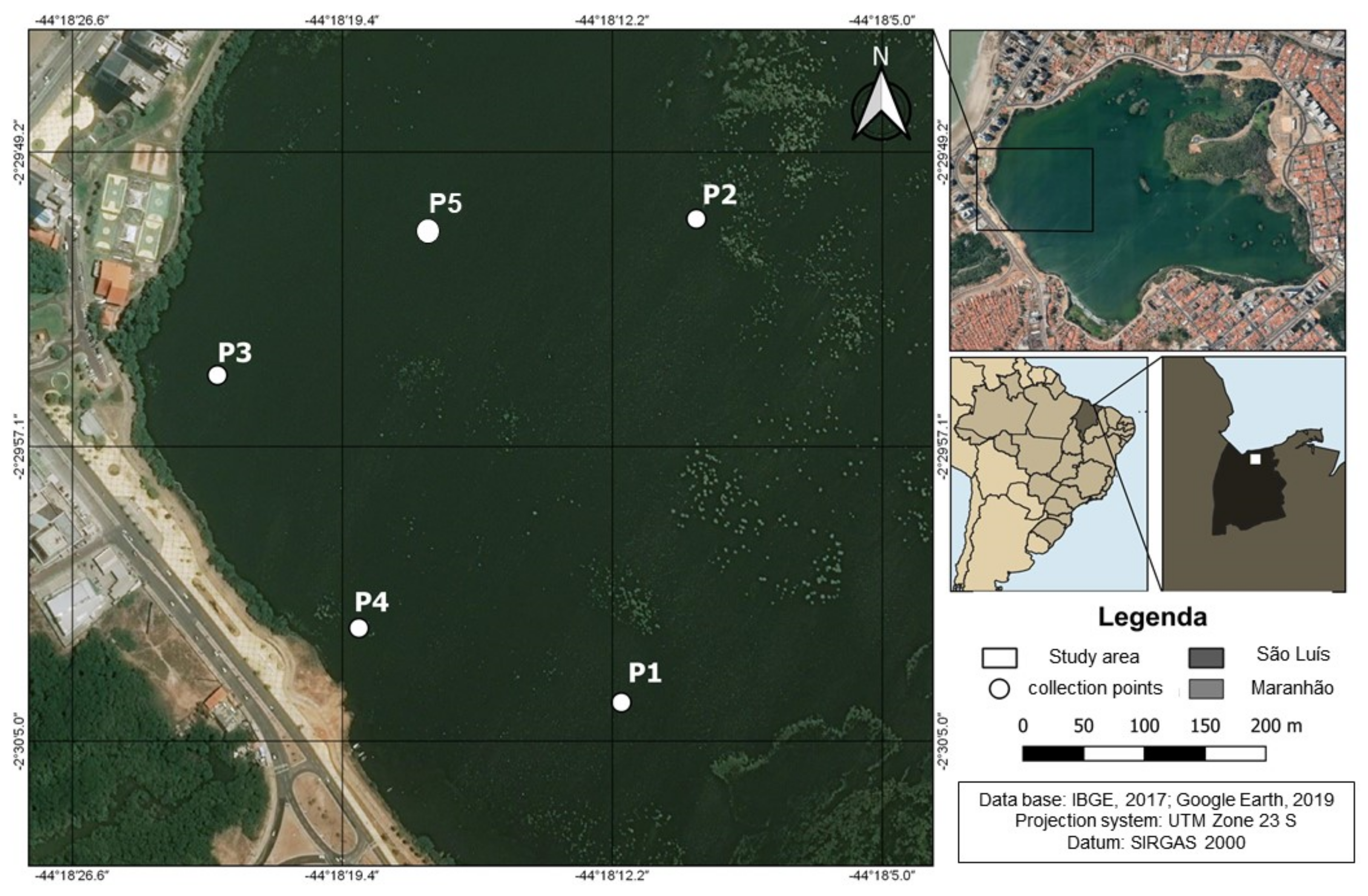
2.1. Study Area
2.2. Physicochemical Characterization of Lagoon Water (Adsorbate)
2.3. Preparation of Adsorptive Material
2.3.1. Pineapple Crown
2.3.2. Chitin and Chitosan Preparation
2.4. Characterization of Adsorbent Support
2.4.1. Morphological Characterization
2.4.2. Structural Characterization
2.5. Treatment Conditions
2.6. Toxicity Assessment
2.7. Statistical Analysis
3. Results
3.1. Physicochemical Characterization of Crude Adsorbate
3.2. Characterization of Adsorptive Supports
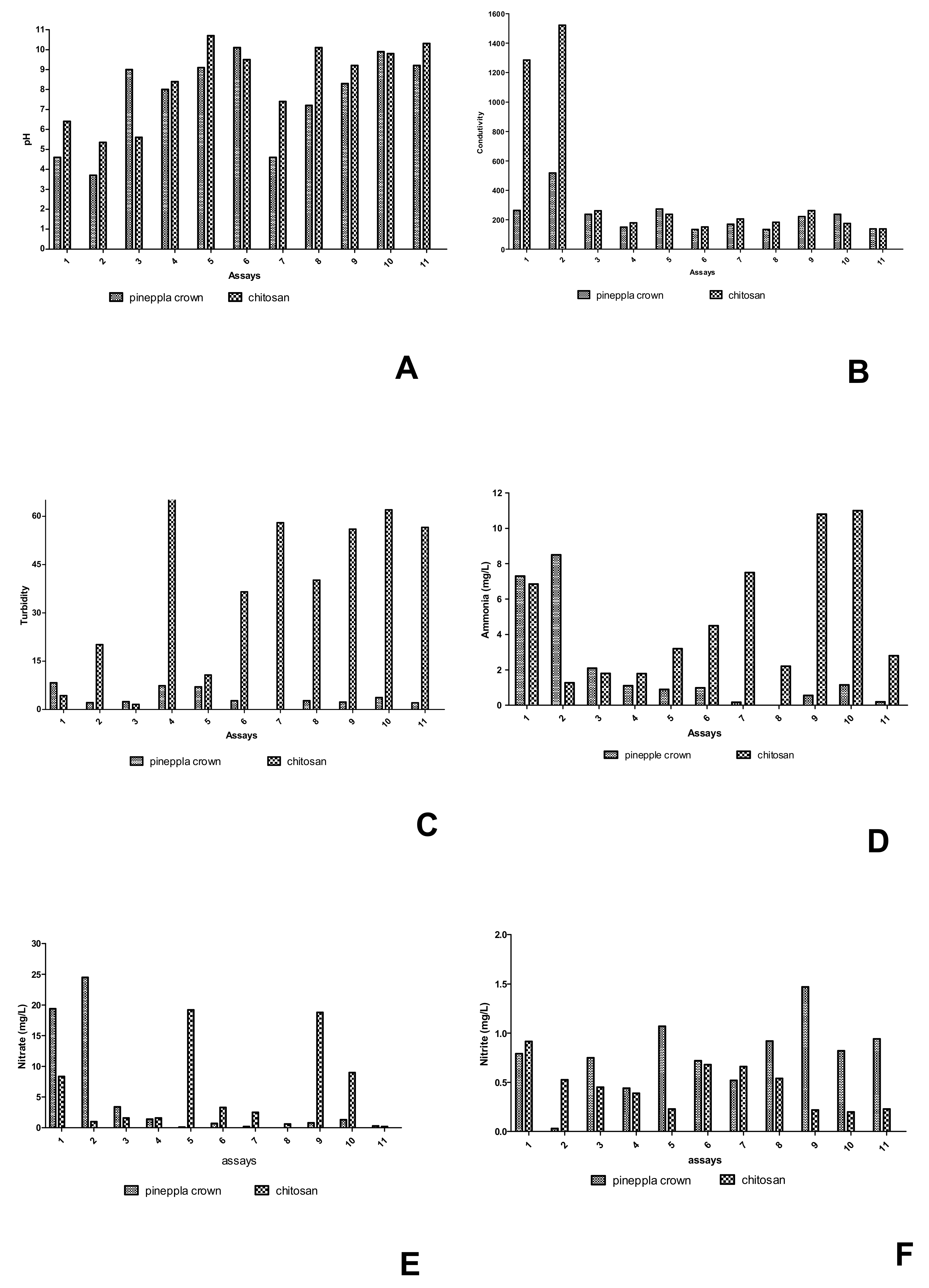
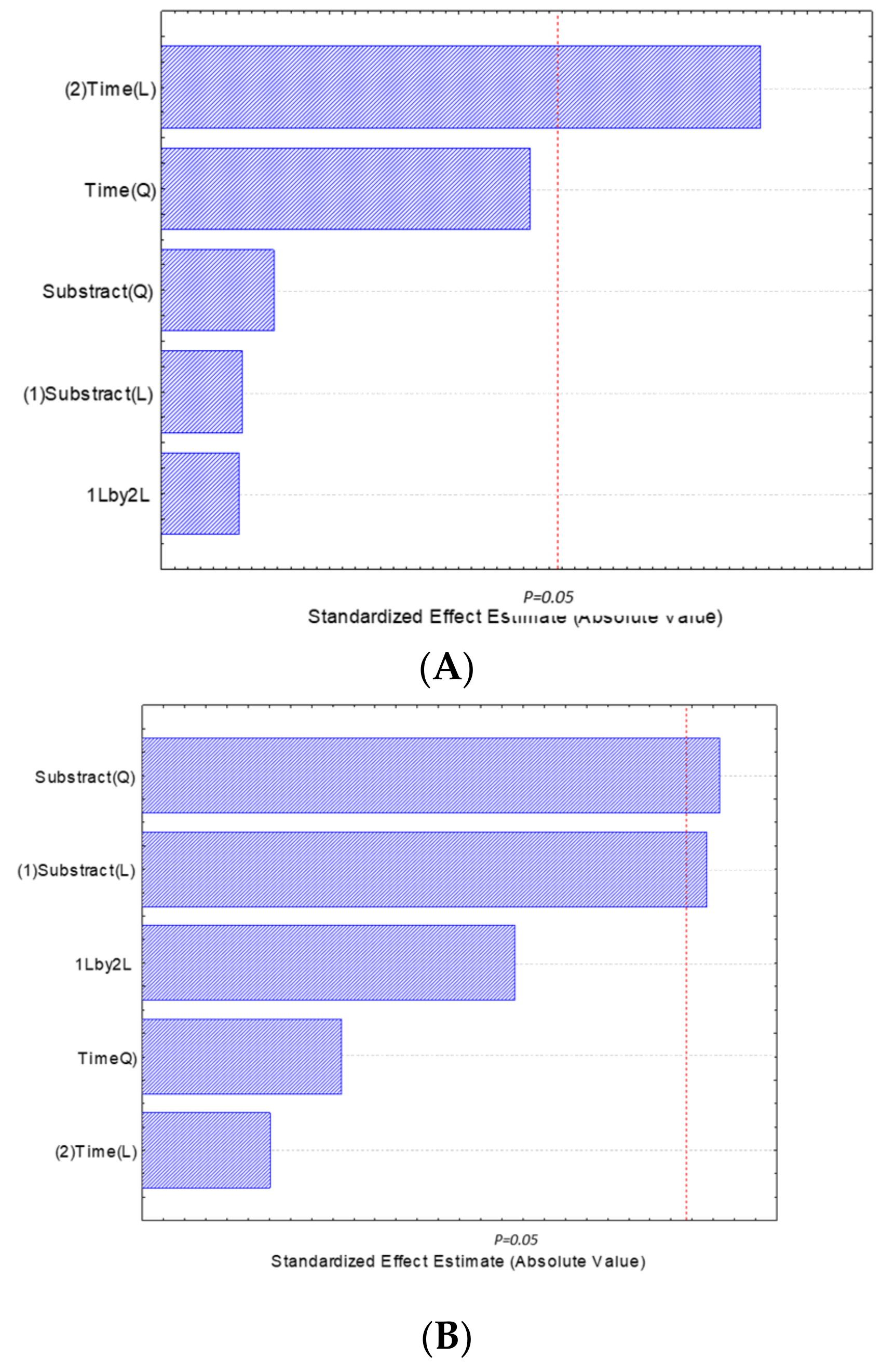
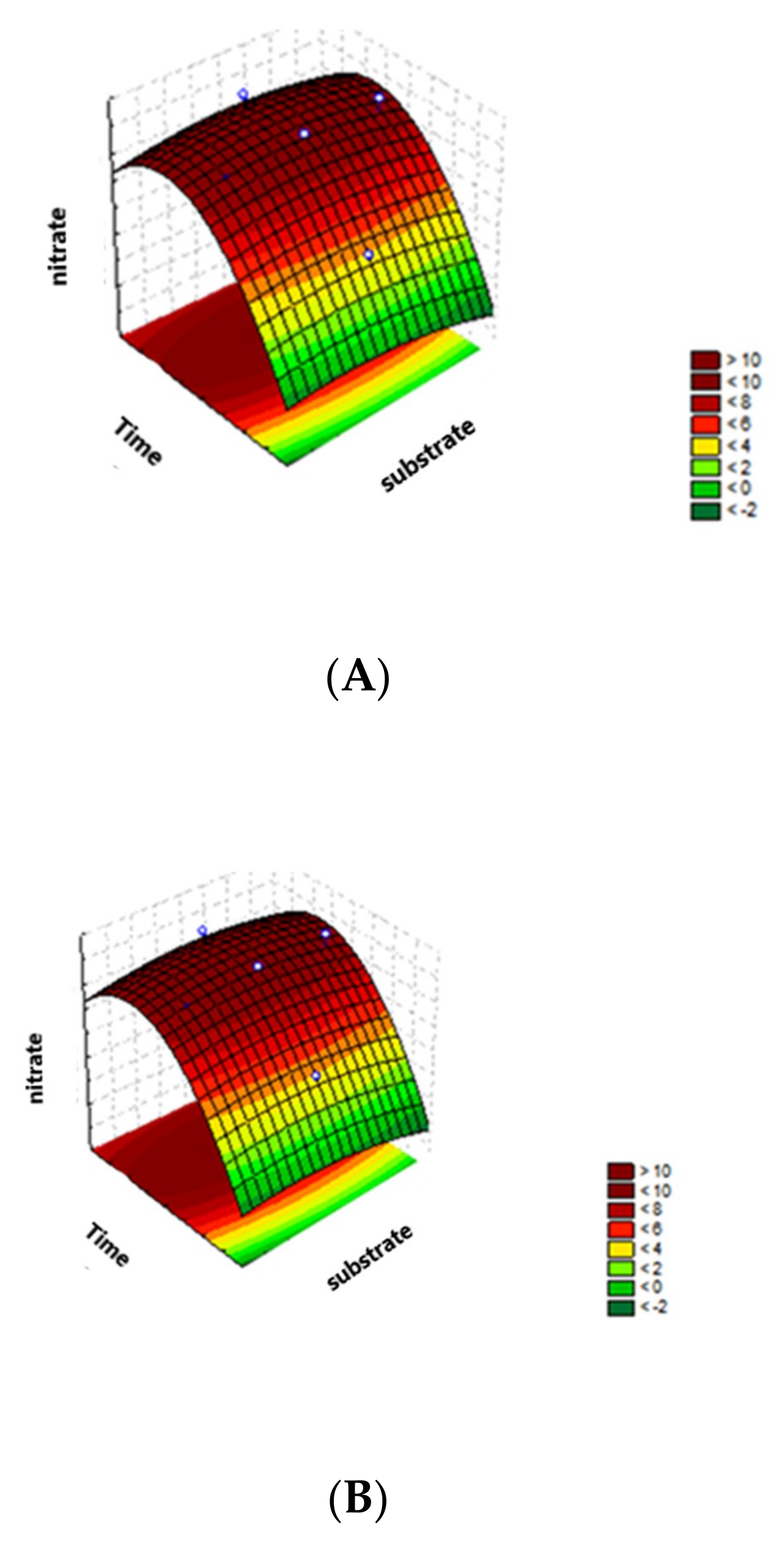
3.3. Lagoon Water Treatment Conditions
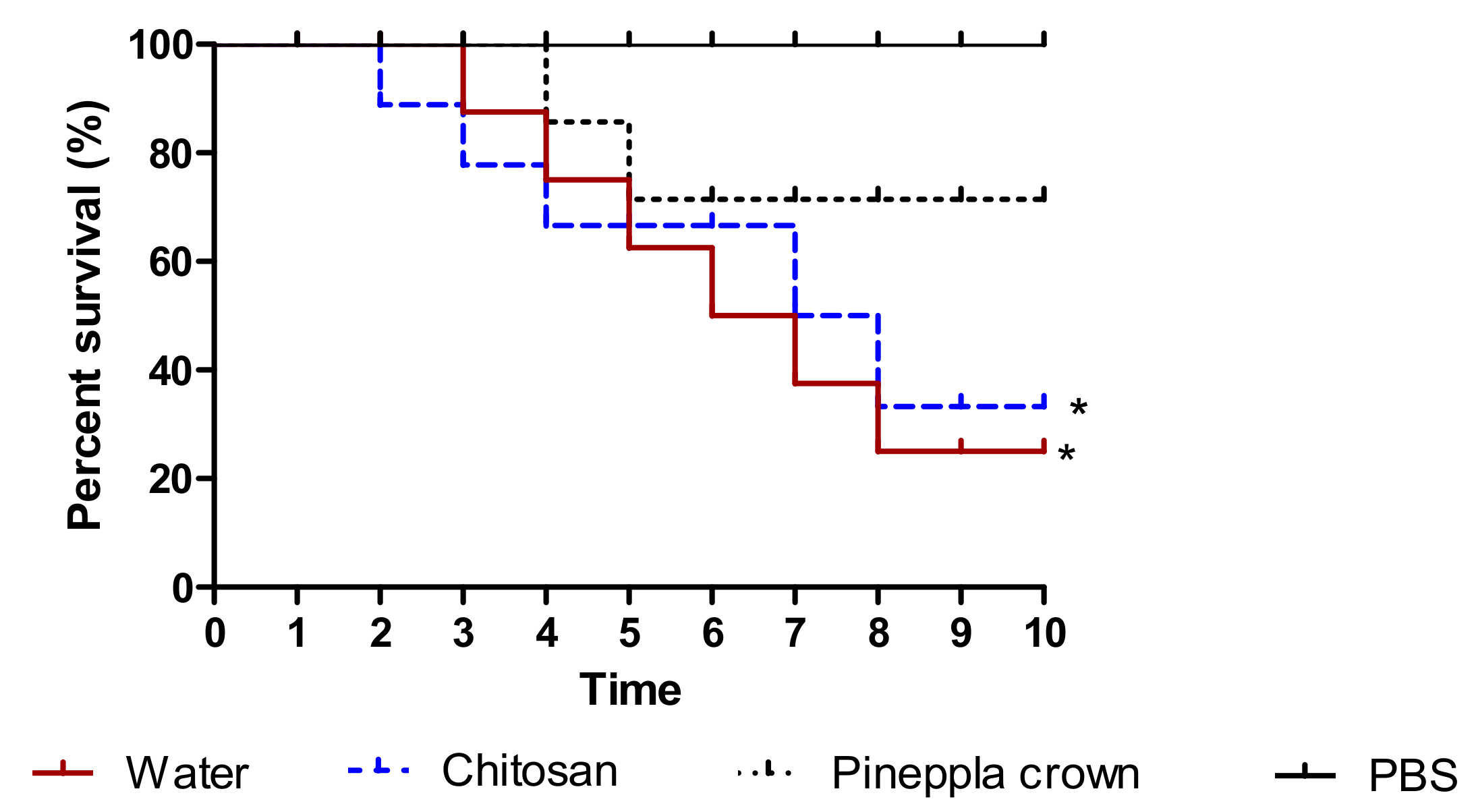
3.4. Toxicity Assessment
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Haughton, G.E.; Hunter, C. Sustainable Development and Geographical Equity. In Proceedings of the Annual Conference of the Association of American Geographers, Chicago, IL, USA, 24–28 February 1994. [Google Scholar]
- Harphan, T.; Vaughan, P.; Lusty, L. In the Shadow of the City: Community Health and the Urban Poor; Oxford University Press: Oxford, UK, 1998. [Google Scholar]
- Tucci, C.E. Urbanização e Recursos Hídricos; Análise Estratégica; Instituto de Pesquisas Hidráulicas da Universidade Federal do Rio Grande do Sul: Águas do Brasil, São Paulo, Brazil, 2007. [Google Scholar]
- SNIS; Sistema Nacional de Informações Sobre Saneamento [SNS]; Secretaria Nacional de Saneamento. Informações e Indicadores Municipais Consolidados. 2016. Available online: http://app4.cidades.gov.br/serieHistorica/# (accessed on 13 August 2019).
- Silva, V.E.C.; Franco, D.; Fonseca, A.L.; Fontes, M.L.; Donnangelo, A.R. Space-time evolution 59 of the trophic state of a subtropical lagoon: 60 Lagoa da Conceição, Florianópolis Island of 61 Santa Catarina, Brazil. Scielo Anal.-62 Rev. Bras. De Recur. Hídricos 2017, 22, 1–6317. [Google Scholar]
- Rojas, M.O.I.; Costa Neto, J.d.J.; Barbiere, R.; Siqueira, L.F.S. Avaliação físico-química da água da Laguna da Jansen. Acta Tecnol. 2013, 8, 19–24. [Google Scholar] [CrossRef]
- Silva, K.; Guimarães, T.; Wasserman, J.C. Distribuição espacial da concentração de nutrientes em sedimentos na Laguna hipersalina de Araruama—RJ, Brasil. Geochim. Bras. 2019, 33, 16–27. [Google Scholar] [CrossRef]
- McLannes, A.S.; Quigg, A. Near-annual fish kills in small embayments: Casual vs. causal factors. J. Coast. Res. 2010, 26, 957–966. [Google Scholar] [CrossRef]
- Souza, R.S.; Lima, L.M.R.; Silva, V.L.M.M. Adsorção de óleo diesel em sistema de leito diferencial com biomassa bagaço de cana-de-açúcar. Rev. Eletrônica Mater. E Process. 2011, 6, 123–126. [Google Scholar]
- Machado, C.R.; Saggioro, E.M.; Silva, Y.G.L.; Pereira, L.P.d.S.; Campos, J.C. Avaliação da adsorção de fenol e bisfenol A em carvões ativados comerciais de diferentes matrizes carbonáceas. Rev. Ambient. Água Taubaté 2015, 10, 915–927. [Google Scholar]
- IBGE. Levantamento Sistemático da Produção Agrícola (LSPA). 2010. Available online: https://biblioteca.ibge.gov.br/index.php/biblioteca-catalogo?view=detalhes&id=76 (accessed on 12 September 2021).
- Baidhe, E.; Kigozi, J.; Mukisa, I.; Muyanja, C.; Namubiru, L.; Kitarikawe, B. Unearthing the potential of solid waste generated along the pineapple drying process line in Uganda: A review. Environ. Chall. 2020, 2, 100012. [Google Scholar] [CrossRef]
- IBGE. Culturas Temporárias e Permanentes; Instituto Brasileiro de Geografia e Estatística: Rio de Janeiro, Brazil, 2015. [Google Scholar]
- Gildberg, A.; Stenberg, E. A new process for advanced utilization of shrimp waste. Process Biochem. 2001, 36, 809–812. [Google Scholar] [CrossRef]
- APHA (American Public Health Association); AWWA (American Water Works Association); WPCF (Water Pollution Control Federation). Standard Methods for the Examination of Water and Wastewater, 21st ed.; American Public Health Association: Washington, WA, USA, 2012. [Google Scholar]
- Miranda, R.C.M.; Neta, J.L.V.; Ferreira, L.F.R.; Gomes, W.A.; do Nascimento, C.S.; Gomes, E.B.; Mattedi, S.; Soares, C.M.F. Pineapple crown delignification using low-cost ionic liquid based on ethanolamine and organic acids. Carbohydr. Polym. 2018, 206, 302–308. [Google Scholar] [CrossRef]
- Moura, C.; Muszinski, P.; Schmidt, C.; Almeida, J.; de Pinto, L.A.A. Quitina Quitosana produzidas a partir de resíduos de camarão e siri: Avaliação do processo em escala piloto. Rev. Ciências Exatas Eng. 2006, 16, 37–45. [Google Scholar]
- Soares, N.M. Obtenção e purificação da quitosana a partir de resíduos de camarão em escala piloto. In Proceedings of the Encontro de Iniciação Científica Latino-Americano, São José dos Campos, Brazil, 15–18 October 2002. [Google Scholar]
- UFRGS. Espectroscopia No Infravermelho Com Transformada de Fourier (FTIR). 2019. Available online: http://www.ufrgs.br/lapol/ftir.htm (accessed on 16 September 2019).
- Brasil, C.D.M. Resolução CONAMA n. 357, de 17 de março de 2005; Diário Oficial da União: Brasília, Brazil, 2005; pp. 58–63. [Google Scholar]
- Silverstein, R.M.; Bassler, G.C. Identificação Espectrométrica de Compostos Orgânicos, 5th ed.; LTC: Guanabara, Brazil, 2006. [Google Scholar]
- Pimentel, P.M.; Silva, C.N., Jr.; Melo, D.M.A.; Maldonado, G.; Henrique, D.M. Caracterização e uso de xisto para adsorção de chumbo (II) em solução. Cerâmica 2006, 52, 194–199. [Google Scholar] [CrossRef] [Green Version]
- Vedula, S.S.; Yadav, G.D. Wastewater treatment containing methylene blue dye as pollutant using adsorption by chitosan lignin membrane: Development of membrane, characterization and kinetics of adsorption. J. Indian Chem. Soc. 2021, 99, 100263. [Google Scholar] [CrossRef]
- Ahmed, H.B.; Helal, M.H.; Abdo, M.H.; Fekry, M.M.; Abdelhamid, A.E. Disarmament of micropollutants from wastewater using nylon waste/chitosan blended with algal biomass as recoverable membrane. Polym. Test. 2021, 104, 107381. [Google Scholar] [CrossRef]
- Zhang, J.; Wang, Y.; Zhang, L.; Zhang, R.; Liu, G.; Cheng, G. Understanding changes in cellulose crystalline structure of lignocellulosic biomass during ionic liquid pretreatment by XRD. Bioresour. Technol. 2014, 151, 402–405. [Google Scholar] [CrossRef] [PubMed]
- Piratoba, A.R.A.; Ribeiro, H.M.C.; Morales, G.P.; Gonçalves, W.G. Caracterização de parâmetros de qualidade da água na área portuária de Barbacena, PR. Rev. Ambiente Água 2017, 12, 435–456. [Google Scholar] [CrossRef] [Green Version]
- Wilson, P.L. Water Quality Nots: Water Clarity (Turbidity, Suspended Solids, and Color); University of Florida: Gainesville, FL, USA, 2010. [Google Scholar]
- Kou, S.; Peters, L.; Mucalo, M. Chitosan: A review of molecular structure, bioactivities and interactions with the human body and micro-organisms. Carbohydr. Polym. 2022, 282, 119132. [Google Scholar] [CrossRef]
- Musarurwa, H.; Tavengwa, N.T. Advances in the application of chitosan-based metal organic frameworks as adsorbents for environmental remediation. Carbohydr. Polym. 2022, 283, 119153. [Google Scholar] [CrossRef]
- Cavalcanti, K.G.S.; da Silva, D.F.; Lima, N.S.; Miranda, R.C.M.; Neto, W.R.N.; Silva, M.R.C. Quantitative vulnerability assessment of Corda river water basin: Impacts of seasonality on water quality in the State of Maranhao. Ciência E Nat. 2020, 42, e2. [Google Scholar] [CrossRef]
- Cavalcante, G.H.; Vieira, F.; Abouleish, M.; Atabay, S.; Campos, E.; Bento, R. Environmental aspects of semi-closed lagoons in the Sharjah coastline during spring/neap tides, southern Arabian/Persian Gulf coast. Reg. Stud. Mar. Sci. 2021, 46, 101896. [Google Scholar] [CrossRef]
- Couson, D.O.; Richardson, J.F. Chemical Engineering. Particle Technology and Separation Process; Butteworth-Heinemann: New York, NY, USA, 2002; Volume 2. [Google Scholar]
- Meurer, M.C.; Silva, F.V. Uso de Erva-mate Como Biossorvente de Contaminantes no Tratamento da Água. Rev. Tecnol. Tendências 2018, 1, 35–60. [Google Scholar] [CrossRef]
- Sun, L.; Li, C.; Xue, Z.; Simmons, B.A.; Singh, S. Unveiling high-resolution, tissue specific dynamic changes in corn stover during ionic liquid pretreatment. RSC Adv. 2012, 3, 2017–2027. [Google Scholar] [CrossRef]
- Vaz Junior, S. Towards a Renewable Brazilian Chemistry from Vegetal Biomass. Rev. Virtual Química 2017, 9, 238–247. [Google Scholar] [CrossRef]
- Albina, P.; Durban, N.; Bertron, A.; Albrecht, A.; Robinet, J.-C.; Erable, B. Nitrate and nitrite bacterial reduction at alkaline pH and high nitrate concentrations, comparison of acetate versus dihydrogen as electron donors. J. Environ. Manag. 2020, 280, 111859. [Google Scholar] [CrossRef]
- Másmela-Mendoza, J.E.; Lizarazo-Forero, L.M.; Aranguren Riaño, N.J. Bacterias nitrificantes cultivables de la zona limnética del lago de Tota, Boyacá. Rev. UDCA Actual. Divulg. Científica 2019, 22, 1–10. [Google Scholar]
- Costa, D.D.; Kempka, A.P.; Skoronski, E. A contaminação de mananciais de abastecimento pelo nitrato: O panorama do problema no Brasil, suas consequências e as soluções potenciais. REDE Rev. Eletrôn. Prodema 2017, 10, 49–61. [Google Scholar] [CrossRef]
- Peng, X.; Hu, X.; Fu, D.; Lam, F.L. Adsorption removal of acid black 1 from aqueous solution using ordered mesoporous carbon. Appl. Surf. Sci. 2014, 294, 71–80. [Google Scholar] [CrossRef]
- Ojha, A.K.; Bulasara, V.K. Adsorption characteristics of jackfruit leaf powder for the removal of Amido black 10B dye. Environ. Prog. Sustain. Energy 2014, 34, 461–470. [Google Scholar] [CrossRef]
- Mittal, A.; Thakur, V.; Gajbe, V. Adsorptive removal of toxic azo dye Amido Black 10B by hen feather. Environ. Sci. Pollut. Res. 2012, 20, 260–269. [Google Scholar] [CrossRef]
- Annadurai, G.; Juang, R.-S.; Lee, D.-J. Use of cellulose-based wastes for adsorption of dyes from aqueous solutions. J. Hazard. Mater. 2002, 92, 263–274. [Google Scholar] [CrossRef]
- Hoseinzadeh, E.; Samarghandi, M.-R.; McKay, G.; Rahimi, N.; Jafari, J. Removal of acid dyes from aqueous solution using potato peel waste biomass: A kinetic and equilibrium study. Desalination Water Treat. 2013, 52, 4999–5006. [Google Scholar] [CrossRef]
- Çelekli, A.; Küçükgüner, B.; Bozkurt, H. Diazo dye sorption by Ni-modified pumpkin husk. Desalinat. Water Treat. 2016, 57, 25242–25255. [Google Scholar] [CrossRef]
- Dalhatou, S.; Laminsi, S.; Pétrier, C.; Baup, S. Competition in sonochemical degradation of Naphthol Blue Black: Presence of an organic (nonylphenol) and a mineral (bicarbonate ions) matrix. J. Environ. Chem. Eng. 2018, 7, 102819. [Google Scholar] [CrossRef]
- Shirani, Z.; Santhosh, C.; Iqbal, J.; Bhatnagar, A. Waste Moringa oleifera seed pods as green sorbent for efficient removal of toxic aquatic pollutants. J. Environ. Manag. 2018, 227, 95–106. [Google Scholar] [CrossRef] [PubMed]
- Daneshvar, E.; Kousha, M.; Jokar, M.; Koutahzadeh, N.; Guibal, E. Acidic dye biosorption onto marine brown macroalgae: Isotherms, kinetic and thermodynamic studies. Chem. Eng. J. 2012, 204, 225–234. [Google Scholar] [CrossRef]
- Wang, W.; Huang, G.; An, C.; Xin, X.; Zhang, Y.; Liu, X. Transport behaviors of anionic azo dyes at interface between sur-factant-modified flax shives and aqueous solution: Synchrotron infrared and adsorption studies. Appl. Surf. Sci. 2017, 405, 119. [Google Scholar] [CrossRef]
- Ahmad, A.; Khan, N.; Giri, B.S.; Chowdhary, P.; Chaturvedi, P. Removal of methylene blue dye using rice husk, cow dung and sludge biochar: Characterization, application, and kinetic studies. Bioresour. Technol. 2020, 306, 123202. [Google Scholar] [CrossRef] [PubMed]
- Yao, X.; Ji, L.; Guo, J.; Ge, S.; Lu, W.; Cai, L.; Wang, Y.; Song, W.; Zhang, H. Magnetic activated bioc nanocomposites derived from wakame and its application in methylene blue adsorption. Bioresourc. Technol. 2020, 302, 122842. [Google Scholar] [CrossRef]
- Oveisi, M.; Asli, M.A.; Mahmoodi, N.M. MIL-Ti metal-organic frameworks (MOFs) nanomaterials as superior adsorbents: Synthesis and ultrasound-aided dye adsorption from multicomponent wastewater systems. J. Hazard. Mater. 2018, 347, 123–140. [Google Scholar] [CrossRef]
- Methneni, N.; GonzÃlez, J.A.M.; Van Loco, J.; Anthonissen, R.; de Maele, J.V.; Verschaeve, L.; Fernandez-Serrano, M.; Mansour, H.B. Ecotoxicity profile of heavily contaminated surface water of two rivers in Tunisia. Environ. Toxicol. Pharmacol. 2021, 82, 103550. [Google Scholar] [CrossRef]


| Runs | Independent Paramenters | |||
|---|---|---|---|---|
| Codes Values | Reals Values | |||
| Substrate (g) | Time (d) | Substrate (g) | Time (d) | |
| 1 | −1 | −1 | 8 | 4 |
| 2 | +1 | −1 | 22 | 4 |
| 3 | −1 | +1 | 8 | 26 |
| 4 | +1 | +1 | 22 | 26 |
| 5 | −1.41 | 0 | 5 | 15 |
| 6 | +1.41 | 0 | 25 | 15 |
| 7 | 0 | −1.41 | 15 | 0 |
| 8 | 0 | +1.41 | 15 | 30 |
| 9 | 0 | 0 | 15 | 15 |
| 10 | 0 | 0 | 15 | 15 |
| 11 | 0 | 0 | 15 | 15 |
| Physicochemical Parameters | Unit | CONAMA 357/2005 | Gross Adsorbate |
|---|---|---|---|
| pH | 6.5–8.5 | 6.65 ± 0.89 | |
| Conductivity | (µS/cm) | *** | 845.5 ± 2.61 |
| Turbidity | (UNT) | 40 | 153 ± 2.24 ** |
| Ammonia | (mg/NL−1) | 5 | 13.57 ± 1.86 |
| Nitrate | (mg/L) | 0.4 | 4.95 ± 0.15 ** |
| Nitrite | (mg/L) | 0.07 | 0.23 ± 0.00 ** |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Neulls, T.F.; Gouveia, P.L.S.; da Silva Pereira, C.D.; Souza, C.S.D.; Chaves, F.N.S.; Souza, I.N.; Tavarez, R.R.d.J.; Aliança, A.S.d.S.; Gonçalves, M.C.; Gomes, W.C.; et al. Comparative Study of Two Organic Wastes as Adsorbents in the Treatment of Water Rich in Nitrogen Compounds. Water 2023, 15, 876. https://doi.org/10.3390/w15050876
Neulls TF, Gouveia PLS, da Silva Pereira CD, Souza CSD, Chaves FNS, Souza IN, Tavarez RRdJ, Aliança ASdS, Gonçalves MC, Gomes WC, et al. Comparative Study of Two Organic Wastes as Adsorbents in the Treatment of Water Rich in Nitrogen Compounds. Water. 2023; 15(5):876. https://doi.org/10.3390/w15050876
Chicago/Turabian StyleNeulls, Thais Fernandes, Paulo Leonardo Santos Gouveia, Carlos Drielson da Silva Pereira, Camilla Stephane Dias Souza, Fernanda Nayr Sena Chaves, Isabela Nascimento Souza, Rudys Rodolfo de Jesus Tavarez, Amanda Silva dos Santos Aliança, Maria Cláudia Gonçalves, Wolia Costa Gomes, and et al. 2023. "Comparative Study of Two Organic Wastes as Adsorbents in the Treatment of Water Rich in Nitrogen Compounds" Water 15, no. 5: 876. https://doi.org/10.3390/w15050876
APA StyleNeulls, T. F., Gouveia, P. L. S., da Silva Pereira, C. D., Souza, C. S. D., Chaves, F. N. S., Souza, I. N., Tavarez, R. R. d. J., Aliança, A. S. d. S., Gonçalves, M. C., Gomes, W. C., da Silva, D. F., Silva, M. R. C., Silva, F. B., Lima, Á. S., Gomes, E. d. B., & Miranda, R. d. C. M. d. (2023). Comparative Study of Two Organic Wastes as Adsorbents in the Treatment of Water Rich in Nitrogen Compounds. Water, 15(5), 876. https://doi.org/10.3390/w15050876










