Risk Evaluation of Chemical Clogging of Irrigation Emitters via Geostatistics and Multivariate Analysis in the Northern Region of Minas Gerais, Brazil
Abstract
:1. Introduction
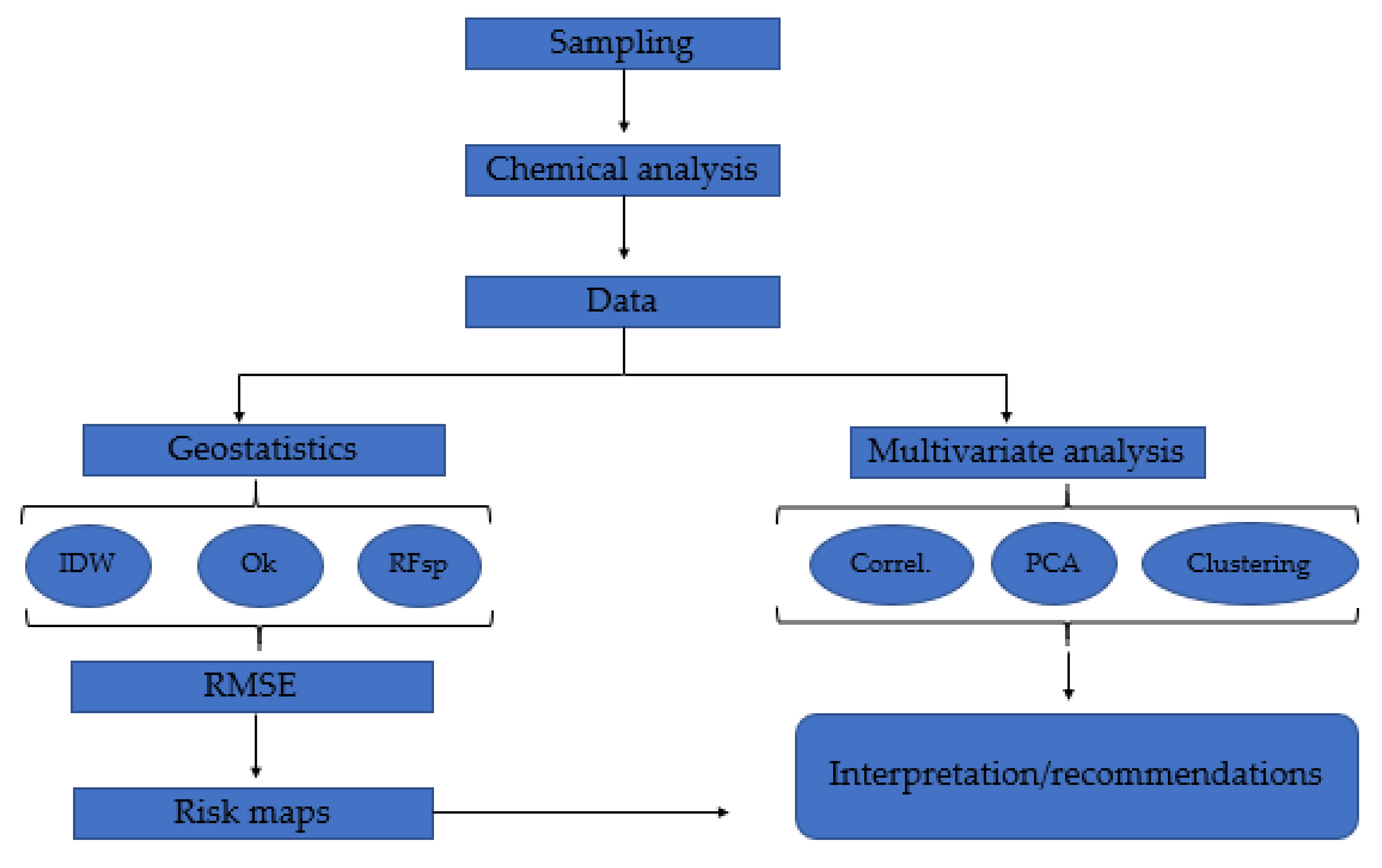
2. Materials and Methods
2.1. Study Area
2.2. Water Quality Data
2.3. Area Classification Limits
2.4. Geostatistics
2.5. Multivariate Analysis
3. Results and Discussion
3.1. Characterization of Samples
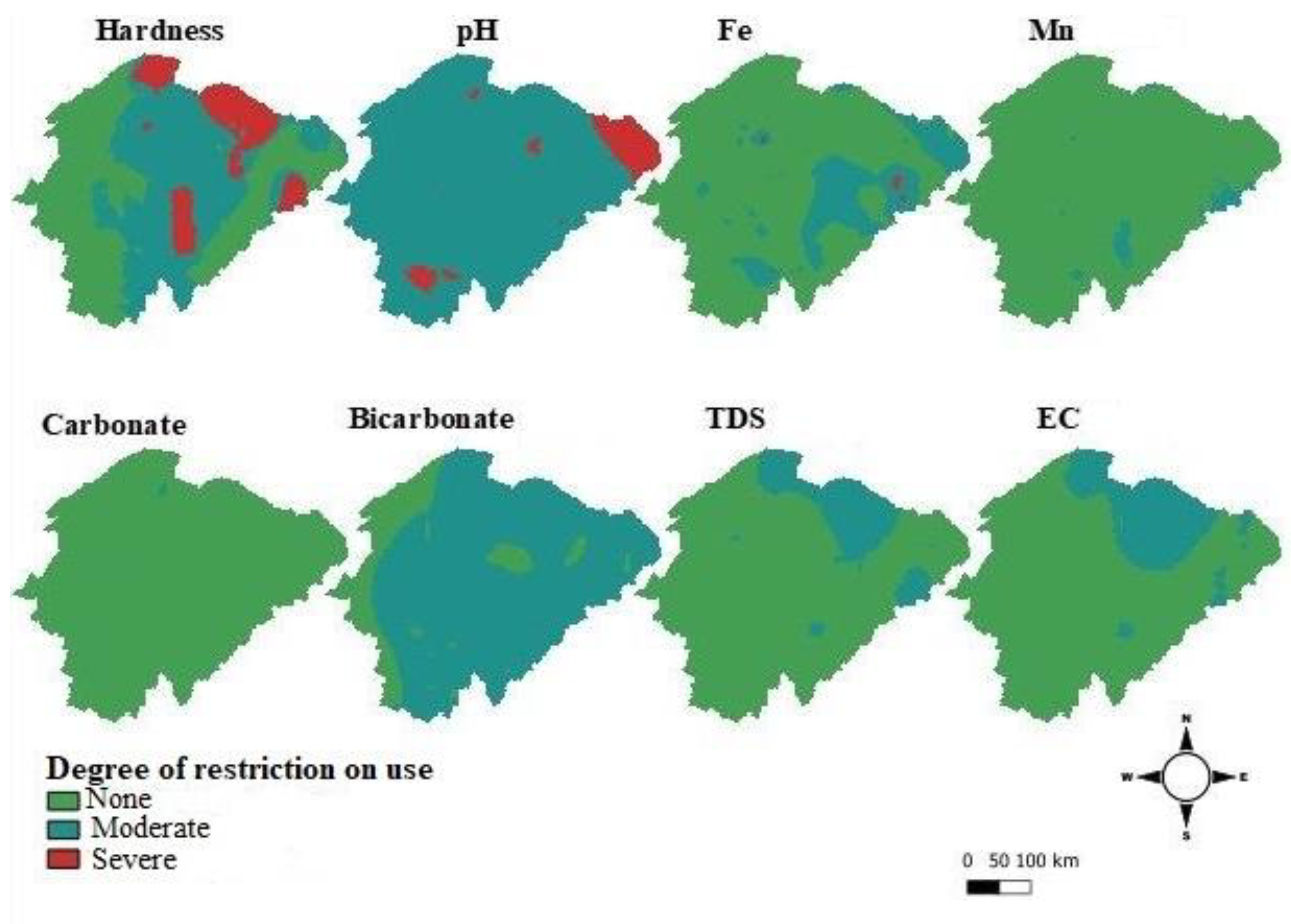
3.2. Characteristic Maps
3.2.1. Total Hardness
3.2.2. pH
3.2.3. Bicarbonates and Carbonates
3.2.4. Iron and Manganese
3.2.5. Total Dissolved Solids (TDS) and Electrical Conductivity (EC)
3.3. Multivariate Analysis
3.3.1. Correlation Analysis
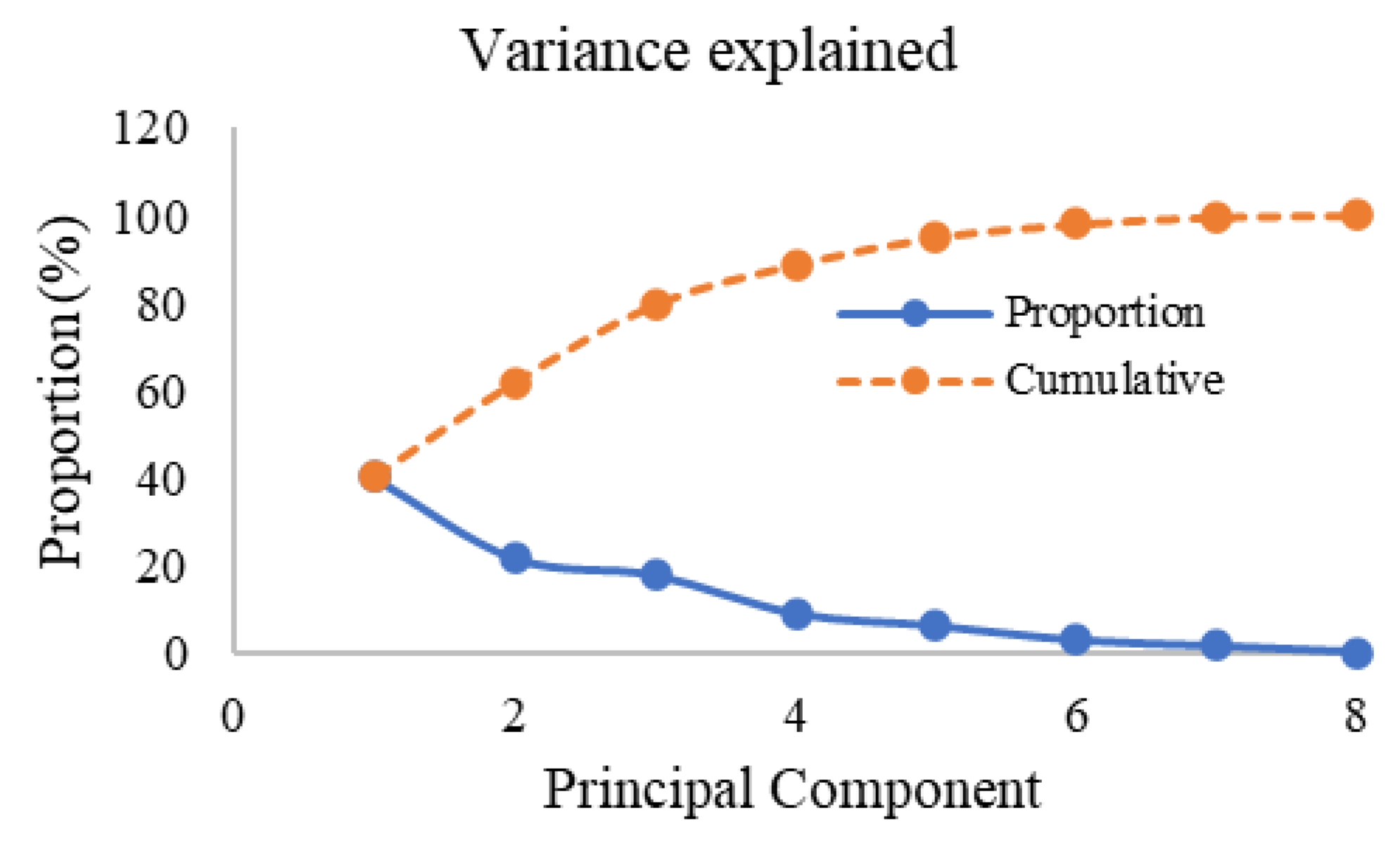
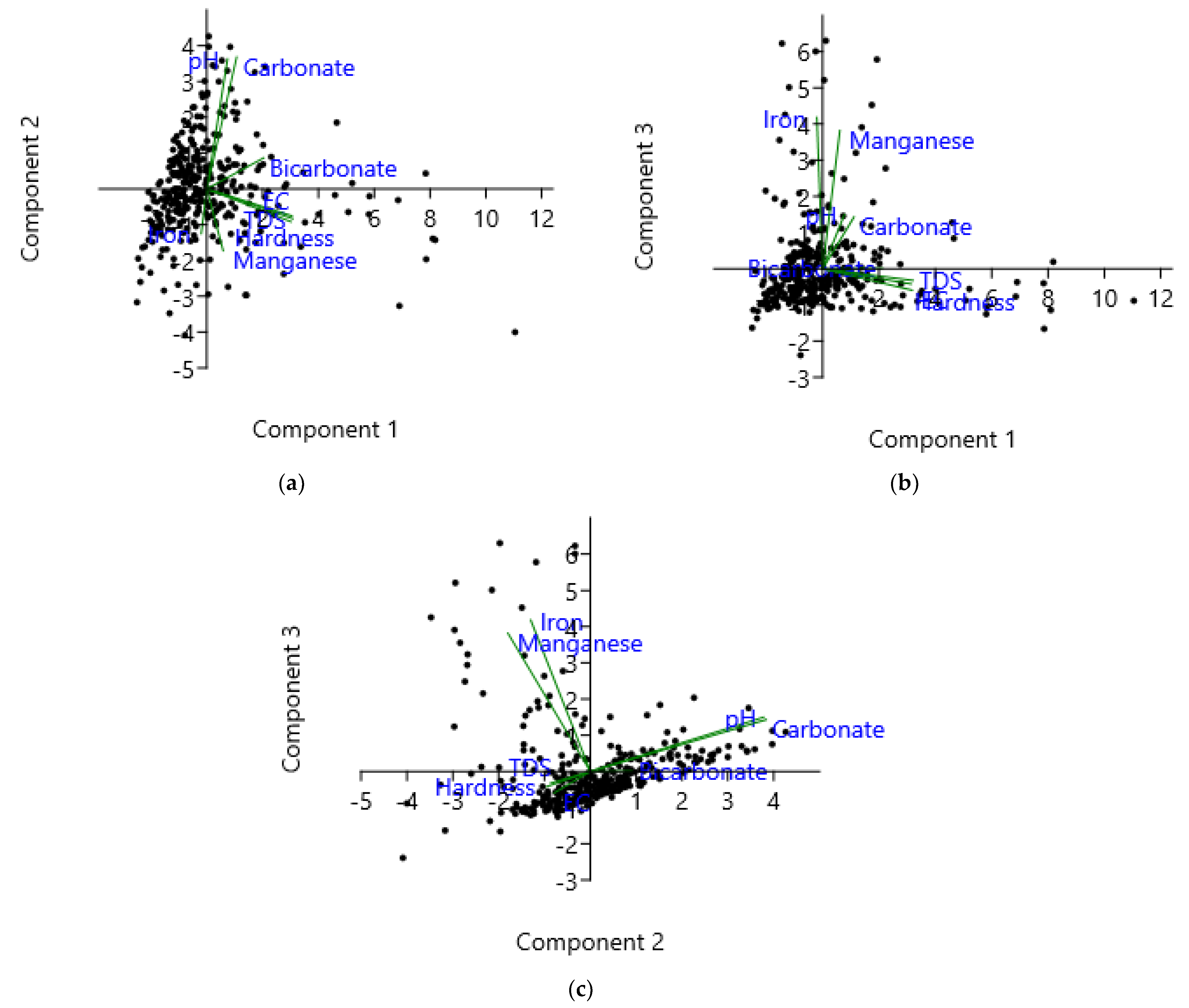
3.3.2. Principal Component Analysis (PCA)
3.3.3. Cluster Analysis
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Souza, M.; Oliveira, S.; Paixão, M.; Haussmann, M. Aspectos Hidrodinâmicos e Qualidade Das Águas Subterrâneas Do Aquífero Bambuí No Norte de Minas Gerais. Rev. Bras. Recur. Hídricos 2014, 19, 119–129. [Google Scholar] [CrossRef]
- Vieira, G.H.S.; Mantovani, E.C.; Sediyama, G.C.; Delazari, F.T. Produtividade e Rendimento Industrial Da Cana-de-Açúcar Em Função de Lâminas de Irrigação. Biosci. J. 2014, 30, 55–64. [Google Scholar]
- Emater Ações Da Emater-Mg Anater Beneficiam Projetos Jaíba e Gorutuba. Available online: http://www.emater.mg.gov.br/portal.do/site-noticias/acoes-da-emater-mg-e-anater-beneficiam-projetos-jaiba-e-gorutuba/?flagweb=novosite_pagina_interna&id=22576 (accessed on 22 June 2018).
- Reis, P.R.d.C.; Silveira, S.d.F.R.; Rodrigues, P.E.L. Impactos Da Política Nacional de Irrigação Sobre o Desenvolvimento Socioeconômico Da Região Norte de Minas Gerais: Uma Avaliação Do Projeto Gorutuba. Rev. Admin. Publ. 2012, 46, 1101–1130. [Google Scholar] [CrossRef] [Green Version]
- Dong, S.; Wan, S.; Kang, Y.; Li, X. Prospects of Using Drip Irrigation for Ecological Conservation and Reclaiming Highly Saline Soils at the Edge of Yinchuan Plain. Agric. Water Manag. 2020, 239, 106255. [Google Scholar] [CrossRef]
- Frizzone, J.A.; Freitas, P.S.L.; Rezende, R.; Faria, M.A. Microirrigação: Gotejamento e Microaspersão, 1st ed.; Eduem: Maringá, Brazil, 2012; ISBN 978-85-7628-460-4. [Google Scholar]
- Wang, Y.; Li, S.; Qin, S.; Guo, H.; Yang, D.; Lam, H.-M. How Can Drip Irrigation Save Water and Reduce Evapotranspiration Compared to Border Irrigation in Arid Regions in Northwest China. Agric. Water Manag. 2020, 239, 106256. [Google Scholar] [CrossRef]
- Yang, D.; Li, S.; Kang, S.; Du, T.; Guo, P.; Mao, X.; Tong, L.; Hao, X.; Ding, R.; Niu, J. Effect of Drip Irrigation on Wheat Evapotranspiration, Soil Evaporation and Transpiration in Northwest China. Agric. Water Manag. 2020, 232, 106001. [Google Scholar] [CrossRef]
- Oliveira, F.C.; Lavanholi, R.; Camargo, A.P.; Ait-Mouheb, N.; Frizzone, J.A.; Tomas, S.; Molle, B. Clogging of Drippers Caused by Suspensions of Kaolinite and Montmorillonite Clays. Irrig. Sci. 2020, 38, 65–75. [Google Scholar] [CrossRef]
- De Camargo, A.P.; Molle, B.; Tomas, S.; Frizzone, J.A. Assessment of Clogging Effects on Lateral Hydraulics: Proposing a Monitoring and Detection Protocol. Irrig. Sci. 2014, 32, 181–191. [Google Scholar] [CrossRef] [Green Version]
- Lavanholi, R.; Oliveira, F.C.; Camargo, A.P.; Frizzone, J.A.; Molle, B.; Ait-Mouheb, N.; Tomas, S. Methodology to Evaluate Dripper Sensitivity to Clogging Due to Solid Particles: An Assessment. Sci. World J. 2018, 2018, 7697458. [Google Scholar] [CrossRef] [Green Version]
- Amaral, M.A.C.M.; Coelho, R.D.; Oliveira Costa, J.; Pereira, D.J.S.; Camargo, A.P. Dripper Clogging by Soil Particles Entering Lateral Lines Directly during Irrigation Network Assembly in the Field. Agric. Water Manag. 2022, 273, 107884. [Google Scholar] [CrossRef]
- de Sousa Pereira, D.J.; Lavanholi, R.; de Araújo, A.C.S.; de Camargo, A.P.; Ait-Mouheb, N.; Frizzone, J.A.; Molle, B. Evaluating Sensitivity to Clogging by Solid Particles in Irrigation Emitters: Assessment of a Laboratory Protocol. J. Irrig. Drain. Eng. 2020, 146, 04020033. [Google Scholar] [CrossRef]
- Pinto, M.F.; Molle, B.; Alves, D.G.; Ait-Mouheb, N.; Camargo, A.P.; Frizzone, J.A. Flow Rate Dynamics of Pressure-Compensating Drippers under Clogging Effect. Rev. Bras. Eng. Agric. Ambient. 2017, 21, 304–309. [Google Scholar] [CrossRef] [Green Version]
- Rizk, N.; Ait-Mouheb, N.; Bourrié, G.; Molle, B.; Roche, N. Parameters Controlling Chemical Deposits in Micro-Irrigation with Treated Wastewater. J. Water Supply Res. Tec. 2017, 66, 587–597. [Google Scholar] [CrossRef] [Green Version]
- Song, P.; Li, Y.; Zhou, B.; Zhou, C.; Zhang, Z.; Li, J. Controlling Mechanism of Chlorination on Emitter Bio-clogging for Drip Irrigation Using Reclaimed Water. Agric. Water Manag. 2017, 184, 36–45. [Google Scholar] [CrossRef]
- Zhangzhong, L.; Yang, P.; Zhen, W.; Zhang, X.; Wang, C. A Kinetic Model for the Chemical Clogging of Drip Irrigation System Using Saline Water. Agric. Water Manag. 2019, 223, 105696. [Google Scholar] [CrossRef]
- Zhou, H.; Li, Y.; Wang, Y.; Zhou, B.; Bhattarai, R. Composite Fouling of Drip Emitters Applying Surface Water with High Sand Concentration: Dynamic Variation and Formation Mechanism. Agric. Water Manag. 2019, 215, 25–43. [Google Scholar] [CrossRef]
- Bucks, D.A.; Nakayama, F.S.; Gilbert, R.G. Trickle Irrigation Water Quality and Preventive Maintenance. Agric. Water Manag. 1979, 2, 149–162. [Google Scholar] [CrossRef]
- García-Nieto, P.J.; García-Gonzalo, E.; Puig-Bargués, J.; Duran-Ros, M.; Ramírez de Cartagena, F.; Arbat, G. Prediction of Outlet Dissolved Oxygen in Micro-Irrigation Sand Media Filters Using a Gaussian Process Regression. Biosyst. Eng. 2020, 195, 198–207. [Google Scholar] [CrossRef]
- Green, O.; Katz, S.; Tarchitzky, J.; Chen, Y. Formation and Prevention of Biofilm and Mineral Precipitate Clogging in Drip Irrigation Systems Applying Treated Wastewater. Irrig. Sci. 2018, 36, 257–270. [Google Scholar] [CrossRef]
- Xiao, Y.; Liu, Y.; Ma, C.; Muhammad, T.; Zhou, B.; Zhou, Y.; Song, P.; Li, Y. Using Electromagnetic Fields to Inhibit Biofouling and Scaling in Biogas Slurry Drip Irrigation Emitters. J. Hazard. Mater. 2021, 401, 123265. [Google Scholar] [CrossRef]
- Muniz, G.L.; Duarte, F.V.; Rakocevic, M. Assessment and Optimization of Carbonated Hard Water Softening with Moringa Oleifera Seeds. Desalin. Water Treat. 2020, 173, 156–165. [Google Scholar] [CrossRef]
- Muniz, G.L.; Camargo, A.P.; Signorelli, F.; Bertran, C.A.; Pereira, D.J.S.; Frizzone, J.A. Influence of Suspended Solid Particles on Calcium Carbonate Fouling in Dripper Labyrinths. Agric. Water Manag. 2022, 273, 107890. [Google Scholar] [CrossRef]
- Qian, M.; Zuo, Y.; Chen, Z.; Chen, Y.; Yin, X.; Liu, Y.; Yang, W. Crystallization of CaCO3 in Aqueous Solutions with Extremely High Concentrations of NaCl. Crystals 2019, 9, 647. [Google Scholar] [CrossRef] [Green Version]
- Sahin, U.; Tunc, T.; Eroğlu, S. Evaluation of CaCO3 Clogging in Emitters with Magnetized Saline Waters. Desalin. Water Treat. 2012, 40, 168–173. [Google Scholar] [CrossRef]
- Liu, F.; Zhao, Z.; Yang, L.; Ma, Y.; Xu, Y.; Gong, L.; Liu, H. Geochemical Characterization of Shallow Groundwater Using Multivariate Statistical Analysis and Geochemical Modeling in an Irrigated Region along the Upper Yellow River, Northwestern China. J. Geochem. Explor. 2020, 215, 106565. [Google Scholar] [CrossRef]
- Trabelsi, R.; Zouari, K. Coupled Geochemical Modeling and Multivariate Statistical Analysis Approach for the Assessment of Groundwater Quality in Irrigated Areas: A Study from North Eastern of Tunisia. Sustain. Dev. 2019, 8, 413–427. [Google Scholar] [CrossRef]
- Udeshani, W.A.C.; Dissanayake, H.M.K.P.; Gunatilake, S.K.; Chandrajith, R. Assessment of Groundwater Quality Using Water Quality Index (WQI): A Case Study of a Hard Rock Terrain in Sri Lanka. Sustain. Dev. 2020, 11, 100421. [Google Scholar] [CrossRef]
- Reboita, M.S.; Rodrigues, M.; Silva, L.F.; Alves, M.A.; Do Estado De Minas Gerais, A.C. Aspectos Climáticos do estado de Minas Gerais. Rev. Bras. Climatol. 2015, 17, 206–226. [Google Scholar] [CrossRef] [Green Version]
- IGAM-MG Bacia Hidrográfica Do Rio São Francisco. Available online: http://www.igam.mg.gov.br/index.php?option=com_content&task=view&id=154&Itemid=140 (accessed on 24 June 2020).
- APHA. Standard Methods for the Examination of Water and Wastewater, 23rd ed.; APHA: Washington, DC, USA, 2017. [Google Scholar]
- Marcal-Silva, H.; Duarte, F.V.; Oliveira, A.L.G. Avaliação Do Abrandamento de Água Calcária Utilizando Hidróxido de Cálcio e Carbonatação. Águas Subterrâneas 2017, 31, 310. [Google Scholar] [CrossRef] [Green Version]
- Ford, H.W.; Tucker, D.P.H. Blockage of Drip Irrigation Filters and Emitters by Iron-Sulfur-Bacterial Products1. Hortic. Sci. 1975, 10, 62–64. [Google Scholar] [CrossRef]
- Nakayama, F.S.; Bucks, D.A.; French, O.F. Reclaiming Partially Clogged Trickle Emitters. Trans. ASABE 1977, 20, 278–280. [Google Scholar] [CrossRef]
- Lamm, F.R.; Ayars, J.E.; Nakayama, F.S. Microirrigation for Crop Production: Design, Operation and Management; Elsevier: Amsterdam, The Netherlands, 2007; ISBN 9780444506078. [Google Scholar]
- Haman, D.Z. Causes and Prevention of Emitter Plugging in Microirrigation Systems; UF/IFAS Extension: Gainesville, FL, USA, 2011; Volume 11. [Google Scholar]
- Goyal, M.R.; Chavan, V.K.; Tripathi, V.K. Principles and Management of Clogging in Micro Irrigation; Apple Academic Press: Boca Raton, FL, USA, 2016; ISBN 9781771882781. [Google Scholar]
- Eroglu, S.; Sahin, U.; Tunc, T.; Sahin, F. Bacterial Application Increased the Flow Rate of CaCO3-Clogged Emitters of Drip Irrigation System. J. Environ. Manag. 2012, 98, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, R.G.; Nakayama, F.S.; Bucks, D.A.; French, O.F.; Adamson, K.C. Trickle Irrigation: Emitter Clogging and Other Flow Problems. Agric. Water Manag. 1981, 3, 159–178. [Google Scholar] [CrossRef]
- De Andrade Júnior, A.S.; de F. e Silva, Ê.F.; Bastos, E.A.; de B. Melo, F.; Leal, C.M. Uso e Qualidade Da Água Subterrânea Para Irrigação No Semi-Árido Piauiense. Rev. Bras. Eng. 2006, 10, 873–880. [Google Scholar] [CrossRef] [Green Version]
- Haldar, K.; Kujawa-Roeleveld, K.; Dey, P.; Bosu, S.; Datta, D.K.; Rijnaarts, H.H.M. Spatio-Temporal Variations in Chemical-Physical Water Quality Parameters Influencing Water Reuse for Irrigated Agriculture in Tropical Urbanized Deltas. Sci. Total Environ. 2020, 708, 134559. [Google Scholar] [CrossRef]
- Ayers, R.S.; Westcot, D.W. Water Quality for Agriculture—FAO29; Food and Agriculture Organization of the United Nations: Rome, Italy, 1985; ISBN 9251022631. [Google Scholar]
- Chen, F.-W.; Liu, C.-W. Estimation of the Spatial Rainfall Distribution Using Inverse Distance Weighting (IDW) in the Middle of Taiwan. Paddy Water Environ. 2012, 10, 209–222. [Google Scholar] [CrossRef]
- Bashir, N.; Saeed, R.; Afzaal, M.; Ahmad, A.; Muhammad, N.; Iqbal, J.; Khan, A.; Maqbool, Y.; Hameed, S. Water Quality Assessment of Lower Jhelum Canal in Pakistan by Using Geographic Information System (GIS). Sustain. Dev. 2020, 10, 100357. [Google Scholar] [CrossRef]
- Lima, I.Q.; Muñoz, M.O.; Ramos, O.E.R.; Bhattacharya, P.; Choque, R.Q.; Aguirre, J.Q.; Sracek, O. Hydrochemical Assessment with Respect to Arsenic and Other Trace Elements in the Lower Katari Basin, Bolivian Altiplano. Sustain. Dev. 2019, 8, 281–293. [Google Scholar] [CrossRef]
- Singh, P.; Verma, P. A Comparative Study of Spatial Interpolation Technique (IDW and Kriging) for Determining Groundwater Quality. In GIS and Geostatistical Techniques for Groundwater Science; Elsevier: Amsterdam, The Netherlands, 2019; pp. 43–56. [Google Scholar]
- Belkhiri, L.; Tiri, A.; Mouni, L. Spatial Distribution of the Groundwater Quality Using Kriging and Co-Kriging Interpolations. Sustain. Dev. 2020, 11, 100473. [Google Scholar] [CrossRef]
- De Oliveira, R.P.; Grego, C.R.; Brandão, Z.N. Geoestatística Aplicada Na Agricultura de Precisão Utilizando o Vesper; Embrapa: Brasilia, Brazil, 2015; ISBN 9788570355720. [Google Scholar]
- de Carvalho, P.R.M.; Rasera, L.G.; Costa, J.F.C.L.; Araújo, M.G.S.; Varella, L.E.S. Variogram Modeling of Broadband Artifacts of a Seafloor Map for Filtering with Factorial Kriging. J. Appl. Geophys. 2019, 161, 92–104. [Google Scholar] [CrossRef]
- Sun, X.L.; Wu, Y.J.; Zhang, C.; Wang, H.L. Performance of Median Kriging with Robust Estimators of the Variogram in Outlier Identification and Spatial Prediction for Soil Pollution at a Field Scale. Sci. Total Environ. 2019, 666, 902–914. [Google Scholar] [CrossRef] [PubMed]
- Houborg, R.; McCabe, M.F. A Hybrid Training Approach for Leaf Area Index Estimation via Cubist and Random Forests Machine-Learning. ISPRS J. Photogramm. 2018, 135, 173–188. [Google Scholar] [CrossRef]
- Ashrafzadeh, A.; Roshandel, F.; Khaledian, M.; Vazifedoust, M.; Rezaei, M. Assessment of Groundwater Salinity Risk Using Kriging Methods: A Case Study in Northern Iran. Agric. Water Manag. 2016, 178, 215–224. [Google Scholar] [CrossRef]
- Boufekane, A.; Saighi, O. Assessing Groundwater Quality for Irrigation Using Geostatistical Method—Case of Wadi Nil Plain (North-East Algeria). Sustain. Dev. 2019, 8, 179–186. [Google Scholar] [CrossRef]
- Gidey, A. Geospatial Distribution Modeling and Determining Suitability of Groundwater Quality for Irrigation Purpose Using Geospatial Methods and Water Quality Index (WQI) in Northern Ethiopia. Appl. Water Sci. 2018, 8, 82. [Google Scholar] [CrossRef] [Green Version]
- Singh, C.K.; Kumar, A.; Shashtri, S.; Kumar, A.; Kumar, P.; Mallick, J. Multivariate Statistical Analysis and Geochemical Modeling for Geochemical Assessment of Groundwater of Delhi, India. J. Geochem. Explor. 2017, 175, 59–71. [Google Scholar] [CrossRef]
- Sudhakaran, S.; Mahadevan, H.; Arun, V.; Krishnakumar, A.P.; Krishnan, K.A. A Multivariate Statistical Approach in Assessing the Quality of Potable and Irrigation Water Environs of the Netravati River Basin (India). Sustain. Dev. 2020, 11, 100462. [Google Scholar] [CrossRef]
- Kaiser, H.F. The Application of Electronic Computers to Factor Analysis. Educ. Psychol. Meas. 1960, 20, 141–151. [Google Scholar] [CrossRef]
- Liu, C.W.; Lin, K.H.; Kuo, Y.M. Application of Factor Analysis in the Assessment of Groundwater Quality in a Blackfoot Disease Area in Taiwan. Sci. Total Environ. 2003, 313, 77–89. [Google Scholar] [CrossRef]
- Badeenezhad, A.; Tabatabaee, H.R.; Nikbakht, H.-A.; Radfard, M.; Abbasnia, A.; Baghapour, M.A.; Alhamd, M. Estimation of the Groundwater Quality Index and Investigation of the Affecting Factors Their Changes in Shiraz Drinking Groundwater, Iran. Sustain. Dev. 2020, 11, 100435. [Google Scholar] [CrossRef]
- Linden, R. Técnicas de Agrupamento. Rev. Sist. Inf. FSMA 2009, 4, 18–36. [Google Scholar]
- De Freitas, M.N.; da Rosa, S.D.V.F.; Pereira, C.C.; Malta, M.R.; Dias, C.T.S. Identification of Physiological Analysis Parameters Associated with Coffee Beverage Quality. Ciênc. Agrotec 2020, 44, e031019. [Google Scholar] [CrossRef]
- De Amorim, J.R.A.; Cruz, M.A.S.; Resende, R.S. Qualidade Da Água Subterrânea Para Irrigação Na Bacia Hidrográfica Do Rio Piauí, Em Sergipe. Rev. Bras. Eng. Agric. Ambient. 2010, 14, 804–811. [Google Scholar] [CrossRef]
- da Silva Júnior, J.C.; Medeiros, V.; Garrozi, C.; Montenegro, A.; Gonçalves, G.E. Random Forest Techniques for Spatial Interpolation of Evapotranspiration Data from Brazilian’s Northeast. Comput. Electron. Agric. 2019, 166, 105017. [Google Scholar] [CrossRef]
- Machado, D.F.T.; Silva, S.H.G.; Curi, N.; de Menezes, M.D. Soil Type Spatial Prediction from Random Forest: Different Training Datasets, Transferability, Accuracy and Uncertainty Assessment. Sci. Agric. 2019, 76, 243–254. [Google Scholar] [CrossRef]
- Stumm, W.; Morgan, J.J. Aquatic Chemistry: Chemical Equilibria and Rates in Natural Waters, 3rd ed.; Wiley Interscience: New York, NY, USA, 1995; ISBN 0471511854. [Google Scholar]
- Li, Y.; Pan, J.; Chen, X.; Xue, S.; Feng, J.; Muhammad, T.; Zhou, B. Dynamic Effects of Chemical Precipitates on Drip Irrigation System Clogging Using Water with High Sediment and Salt Loads. Agric. Water Manag. 2019, 213, 833–842. [Google Scholar] [CrossRef]
- Lili, Z.; Yang, P.; Ren, S.; Li, Y.; Liu, Y.; Xia, Y. Chemical Clogging of Emitters and Evaluation of Their Suitability for Saline Water Drip Irrigation. Irrig. Drain. 2016, 65, 439–450. [Google Scholar] [CrossRef]
- Hills, D.J.; Nawar, F.M.; Waller, P.M. Effects of Chemical Clogging on Drip-Tape Irrigation Uniformity. Trans. ASABE 1989, 32, 1202–1206. [Google Scholar] [CrossRef]
- Langelier, W.F. The Analytical Control of Anti-Corrosion Water Treatment. Water Works Assoc. 1936, 28, 1500–1521. [Google Scholar] [CrossRef]
- Barik, R.; Pattanayak, S.K. Assessment of Groundwater Quality for Irrigation of Green Spaces in the Rourkela City of Odisha, India. Sustain. Dev. 2019, 8, 428–438. [Google Scholar] [CrossRef]
- Pinto, M.F.; Camargo, A.P.; Almeida, A.C.S.; Alves, D.G.; Botrel, T.A. Desenvolvimento de um Sistema para o Controle Do PH Da Água Para Microirrigação. Rev. Bras. Eng. Agric. Ambient. 2011, 15, 211–217. [Google Scholar] [CrossRef] [Green Version]
- Masoud, A.M.; Ali, M.H. Coupled Multivariate Statistical Analysis and WQI Approaches for Groundwater Quality Assessment in Wadi El-Assiuty Downstream Area, Eastern Desert, Egypt. J. Afr. Earth Sci. 2020, 172, 103982. [Google Scholar] [CrossRef]
- Maia, C.E.; de Morais, E.R.C.; de Oliveira, M. Estimativa de Carbonato de Cálcio Aplicado via Água de Irrigação Nas Regiões Da Chapada Do Apodi e Baixo Açu, RN. Rev. Bras. Eng. Agric. Ambient. 2001, 5, 71–75. [Google Scholar] [CrossRef]
- Ellis, D.; Bouchard, C.; Lantagne, G. Removal of Iron and Manganese from Groundwater by Oxidation and Microfiltration. Desalination 2000, 130, 255–264. [Google Scholar] [CrossRef]
- Gerke, T.L.; Little, B.J.; Barry Maynard, J. Manganese Deposition in Drinking Water Distribution Systems. Sci. Total Environ. 2016, 541, 184–193. [Google Scholar] [CrossRef] [Green Version]
- Ribeiro, T.A.P.; Paterniani, J.E.S.; Coletti, C. Chemical Treatment to Unclogg Dripper Irrigation Systems Due to Biological Problems. Sci. Agric. 2008, 65, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Nakayama, F.S.; Bucks, D.A. Trickle Irrigation for Crop Production: Design, Operation and Management; Elsevier Science: Amsterdam, The Netherlands, 1986; ISBN 9780444600967. [Google Scholar]
- Elumalai, V.; Nethononda, V.G.; Manivannan, V.; Rajmohan, N.; Li, P.; Elango, L. Groundwater Quality Assessment and Application of Multivariate Statistical Analysis in Luvuvhu Catchment, Limpopo, South Africa. J. Afr. Earth Sci. 2020, 171, 103967. [Google Scholar] [CrossRef]
- Ashraf, S.; Rizvi, N.B.; Rasool, A.; Mahmud, T.; Huang, G.G.; Zulfajri, M. Evaluation of Heavy Metal Ions in the Groundwater Samples from Selected Automobile Workshop Areas in Northern Pakistan. Sustain. Dev. 2020, 11, 100428. [Google Scholar] [CrossRef]
- Varol, M.; Şen, B. Assessment of Surface Water Quality Using Multivariate Statistical Techniques: A Case Study of Behrimaz Stream, Turkey. Environ. Monit. Assess. 2009, 159, 543–553. [Google Scholar] [CrossRef]
- Varol, M. Use of Water Quality Index and Multivariate Statistical Methods for the Evaluation of Water Quality of a Stream Affected by Multiple Stressors: A Case Study. Environ. Pollut. 2020, 266, 115417. [Google Scholar] [CrossRef]
- Liu, P.; Hoth, N.; Drebenstedt, C.; Sun, Y.; Xu, Z. Hydro-Geochemical Paths of Multi-Layer Groundwater System in Coal Mining Regions—Using Multivariate Statistics and Geochemical Modeling Approaches. Sci. Total Environ. 2017, 601–602, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Tay, C.K.; Hayford, E.K.; Hodgson, I.O.A. Application of Multivariate Statistical Technique for Hydrogeochemical Assessment of Groundwater within the Lower Pra Basin, Ghana. Appl. Water Sci. 2017, 7, 1131–1150. [Google Scholar] [CrossRef] [Green Version]
- Asgharinia, S.; Petroselli, A. A Comparison of Statistical Methods for Evaluating Missing Data of Monitoring Wells in the Kazeroun Plain, Fars Province, Iran. Sustain. Dev. 2020, 10, 100294. [Google Scholar] [CrossRef]




| Parameters | Degree of Restriction | Reference | ||
|---|---|---|---|---|
| None | Moderate | Severe | ||
| pH | <7.00 | 7.00–8.00 | >8 | [38] |
| Total hardness (mg L−1 as CaCO3) | <150 | 150–300 | >300 | [38] |
| Iron (mg L−1) | <0.2 | 0.2–1.5 | >1.5 | [38] |
| Manganese (mg L−1) | <0.2 | 0.2–1.5 | >1.5 | [38] |
| Bicarbonate (mg L−1) | <91.5 | 91.4–518.5 | >518.5 | [43] |
| Carbonate (mg L−1) | <6.0 | 6.0–12.0 | >12.0 | [41] |
| Total dissolved solids (mg L−1) | <500.0 | 500.0–2000.0 | >2000.0 | [38] |
| Electrical conductivity (dS m−1) | <0.7 | 0.7–3.0 | >0.3 | [43] |
| Parameters | Max | Min | x1 | Sx2 |
|---|---|---|---|---|
| pH | 8.90 | 4.52 | 7.76 | 0.52 |
| Total hardness (mg L−1 as CaCO3) | 2316.00 | 13.88 | 292.30 | 304.30 |
| Bicarbonate (mg L−1) | 917.00 | 11.0 | 216.0 | 114.0 |
| Carbonate (mg L−1) | 11.00 | <0.01 | 2.00 | 2.00 |
| Iron (mg L−1) | 22.60 | 0.04 | 0.49 | 1.67 |
| Manganese (mg L−1) | 3.13 | 0.02 | 0.23 | 0.36 |
| Total dissolved solids (mg L−1) | 5018.00 | 26.00 | 541.40 | 622.00 |
| Electrical conductivity (dS m−1) | 6.96 | 0.05 | 0.78 | 0.86 |
| Interpolation Method | pH | Hardness (mg L−1 as CaCO3) | Bicarbonate (mg L−1) | Carbonate (mg L−1) | Iron (mg L−1) | Manganese (mg L−1) | TDS (mg L−1) | EC (dS m−1) |
|---|---|---|---|---|---|---|---|---|
| RFsp | 0.52 | 164.38 | 90.58 | 2.24 | 0.50 | 0.46 | 551.20 | 0.73 |
| IDW | 0.51 | 174.84 | 99.99 | 2.19 | 0.80 | 0.35 | 492.77 | 0.69 |
| Ok | 0.52 | 211.78 | 107.88 | 2.14 | 0.60 | 0.35 | 390.60 | 0.68 |
| Parameter | pH | HCO3− | CO32− | Hardness | Fe | Mn | TDS | EC |
|---|---|---|---|---|---|---|---|---|
| pH | 1.00 | |||||||
| HCO3− | 0.117 * | 1.00 | ||||||
| CO32− | 0.714 * | 0.356 * | 1.00 | |||||
| Hardness | 0.098 | 0.497 * | 0.104 | 1.00 | ||||
| Fe | −0.027 | −0.050 | −0.033 | −0.054 | 1.00 | |||
| Mn | −0.046 | 0.031 | −0.043 | 0.167 * | 0.467 * | 1.00 | ||
| TDS | 0.091 | 0.407 * | 0.138 * | 0.896 * | −0.050 | 0.179 * | 1.00 | |
| EC | 0.087 | 0.417 * | 0.129 * | 0.871 * | −0.079 | 0.115 * | 0.972 * | 1.00 |
| Parameter | PC1 | PC2 | PC3 |
|---|---|---|---|
| pH | 0.127 | 0.621 | 0.246 |
| HCO3− | 0.349 | 0.148 | 0.002 |
| CO32− | 0.183 | 0.630 | 0.235 |
| Hardness | 0.516 | −0.157 | −0.069 |
| Fe | −0.034 | −0.214 | 0.687 |
| Mn | 0.100 | −0.296 | 0.628 |
| TDS | 0.528 | −0.142 | −0.053 |
| EC | 0.522 | −0.129 | −0.097 |
| Eigenvalue | 3.215 | 1.735 | 1.426 |
| Proportion | 0.402 | 0.217 | 0.178 |
| Group | Frequency | SQ | MD | MaxD | Distance between the Centroids of the Groups | |||
|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | |||||
| 1 | 23 | 248.868 | 2.976 | 7.402 | - | 5.35 | 6.63 | 6.02 |
| 2 | 116 | 393.526 | 1.713 | 3.678 | 5.35 | - | 4.34 | 2.19 |
| 3 | 26 | 282.060 | 3.042 | 7.149 | 6.63 | 4.34 | - | 4.05 |
| 4 | 185 | 451.594 | 1.390 | 5.950 | 6.02 | 2.19 | 4.05 | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Muniz, G.L.; Oliveira, A.L.G.; Benedito, M.G.; Cano, N.D.; Camargo, A.P.d.; Silva, A.J.d. Risk Evaluation of Chemical Clogging of Irrigation Emitters via Geostatistics and Multivariate Analysis in the Northern Region of Minas Gerais, Brazil. Water 2023, 15, 790. https://doi.org/10.3390/w15040790
Muniz GL, Oliveira ALG, Benedito MG, Cano ND, Camargo APd, Silva AJd. Risk Evaluation of Chemical Clogging of Irrigation Emitters via Geostatistics and Multivariate Analysis in the Northern Region of Minas Gerais, Brazil. Water. 2023; 15(4):790. https://doi.org/10.3390/w15040790
Chicago/Turabian StyleMuniz, Gustavo Lopes, Agda Loureiro Gonçalves Oliveira, Maria Geralda Benedito, Nicolás Duarte Cano, Antonio Pires de Camargo, and Ariovaldo José da Silva. 2023. "Risk Evaluation of Chemical Clogging of Irrigation Emitters via Geostatistics and Multivariate Analysis in the Northern Region of Minas Gerais, Brazil" Water 15, no. 4: 790. https://doi.org/10.3390/w15040790






