Production, Application, and Efficacy of Biodefoamers from Bacillus, Aeromonas, Klebsiella, Comamonas spp. Consortium for the Defoamation of Poultry Slaughterhouse Wastewater
Abstract
:1. Introduction
2. Materials and Methods
2.1. Isolation and Identification
2.2. Metagenomics Analysis of the MLSS Microbial Community
2.3. Biodefoamer Production and Extraction
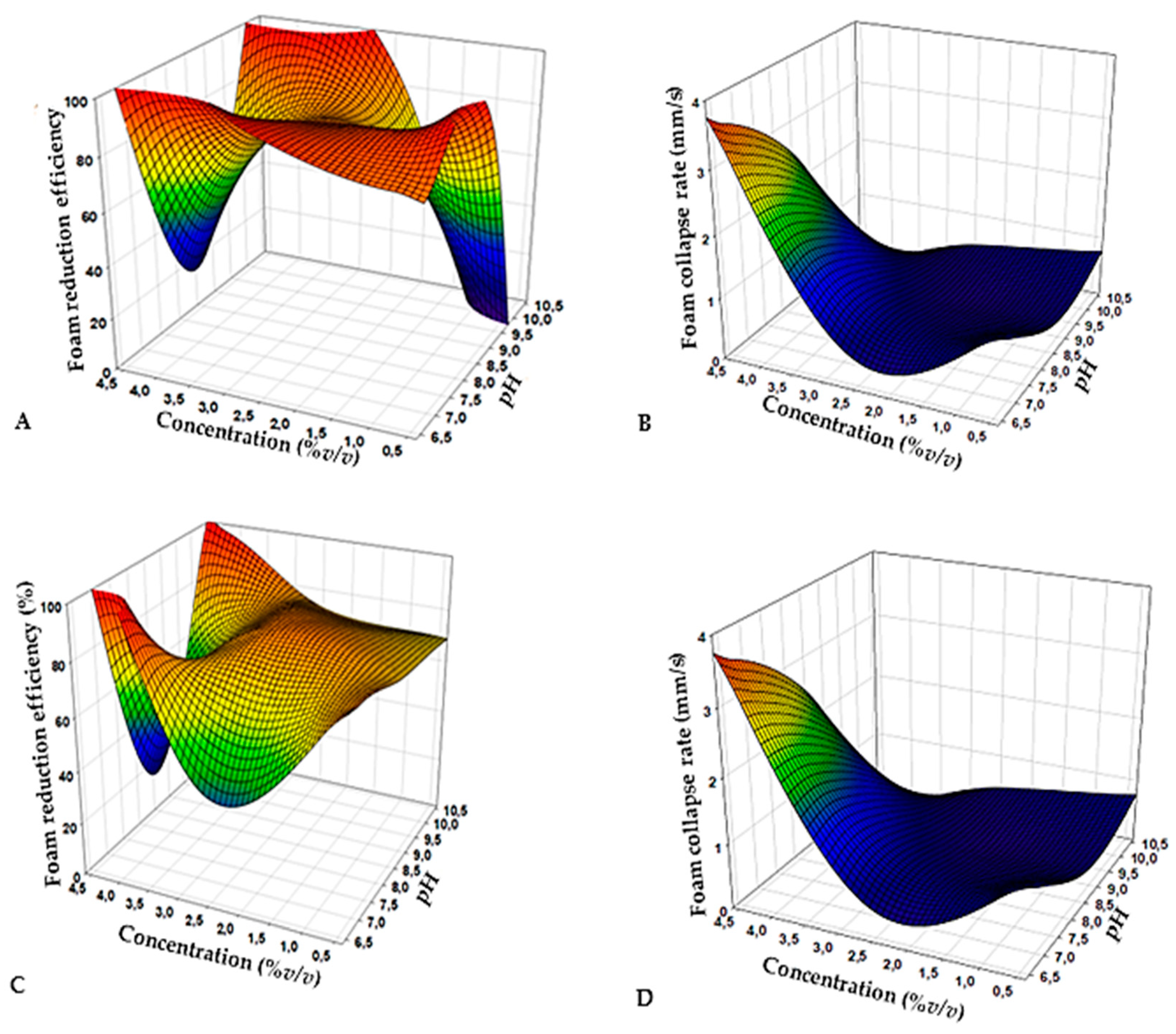
2.4. Response Surface Methodology
2.5. Foaming Behavior Tests
2.5.1. Foam Reduction Efficiency
2.5.2. Foam Collapse (Decay) Rate
2.5.3. Microscopic Analysis of Sludge Agglomeration in the Presence and Absence of Bio- and Synthetic Defoamers
2.6. Characterization of the Biodefoamers
3. Results and Discussion
3.1. Microbial Isolation and Identification of Biodefoamer-Producing Isolates
3.2. Mixed Liquor Suspended Solids (MLSS) Metagenomics Analysis
3.3. Biodefoamer Production, Reactor Conditions Optimization and Characterization
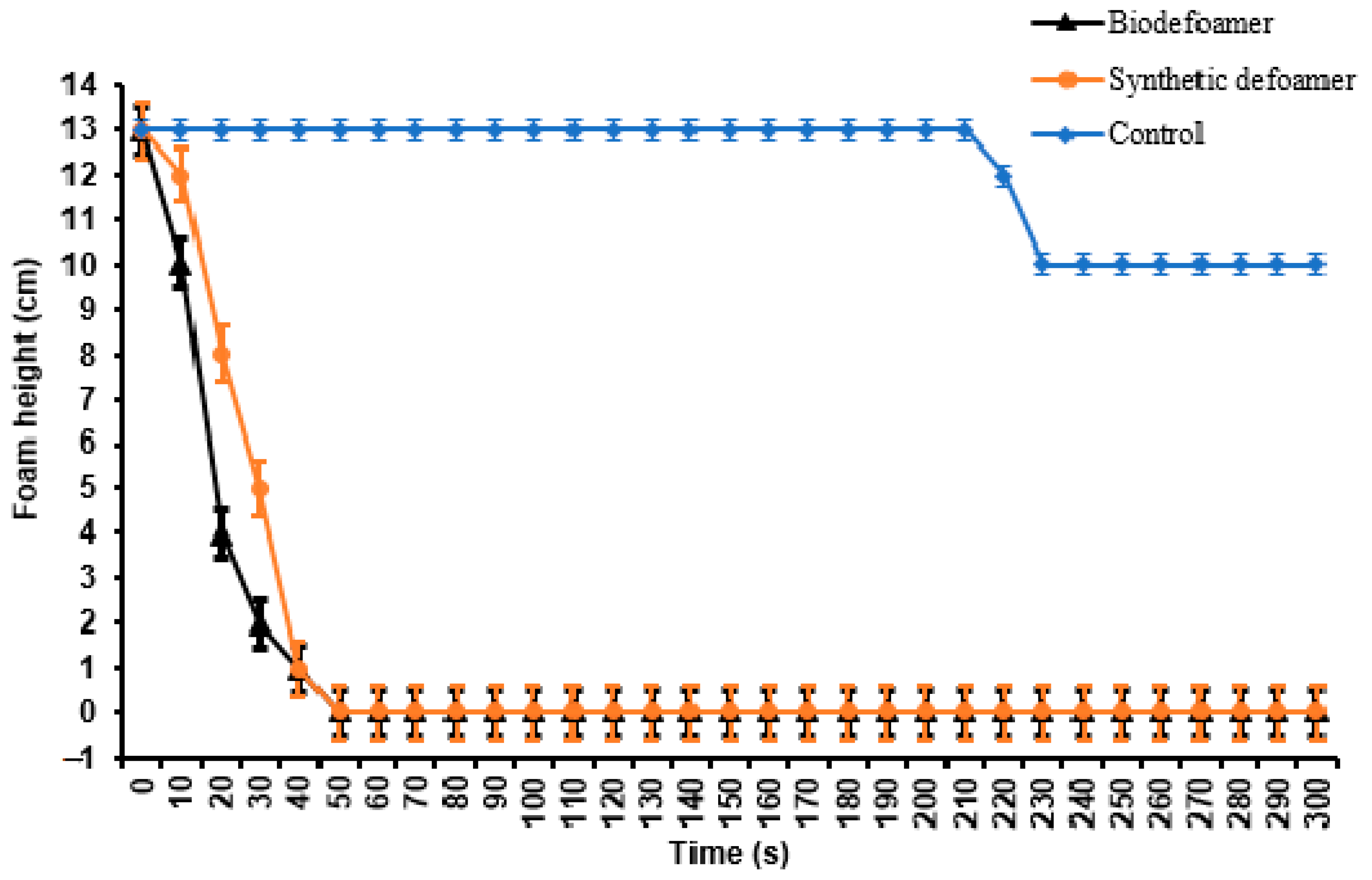
3.4. Dynamic Foam Decay Test
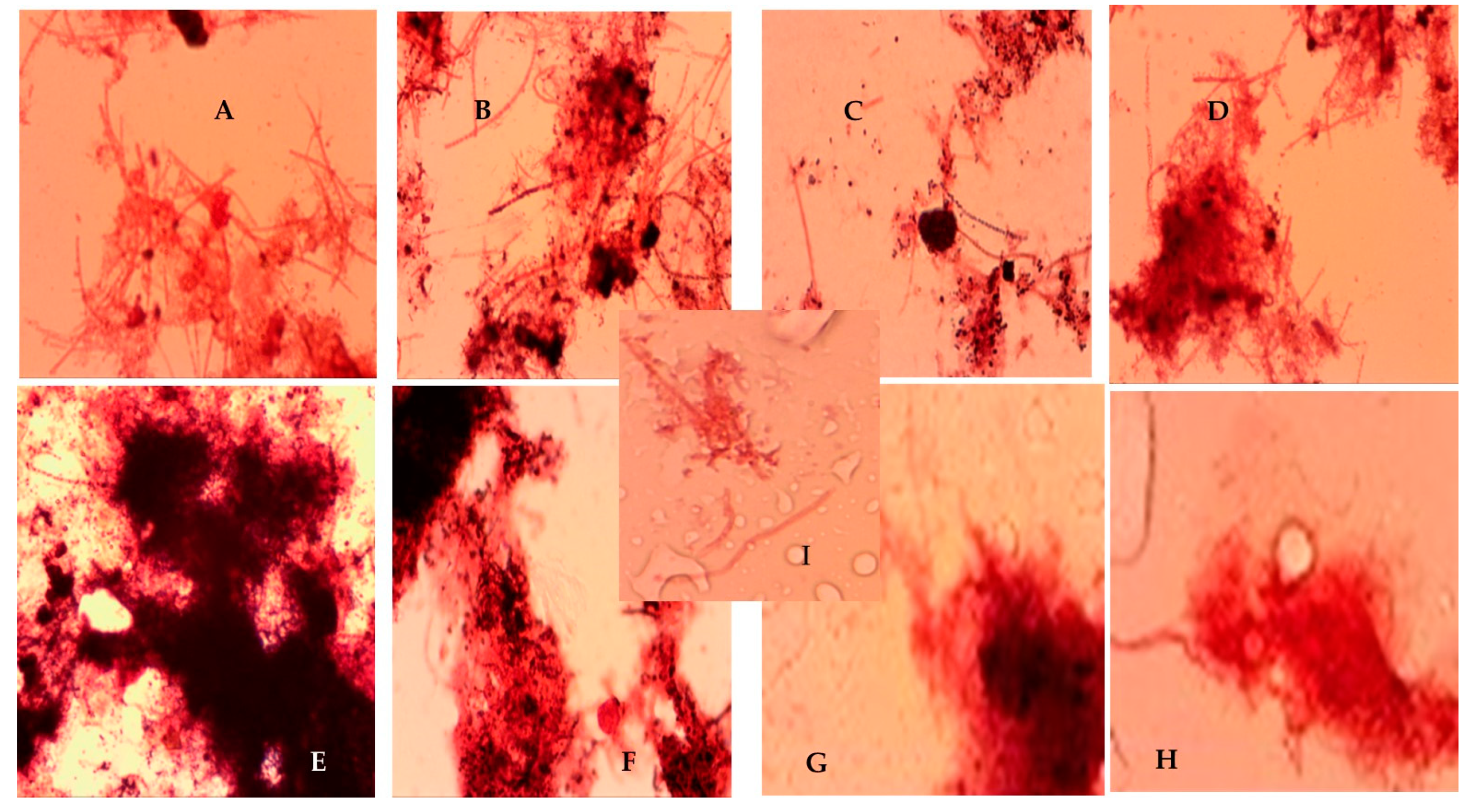
3.5. Microscopic Analysis of Recovered Activated Sludge in the Presence and Absence of Bio- and Synthetic Defoamers
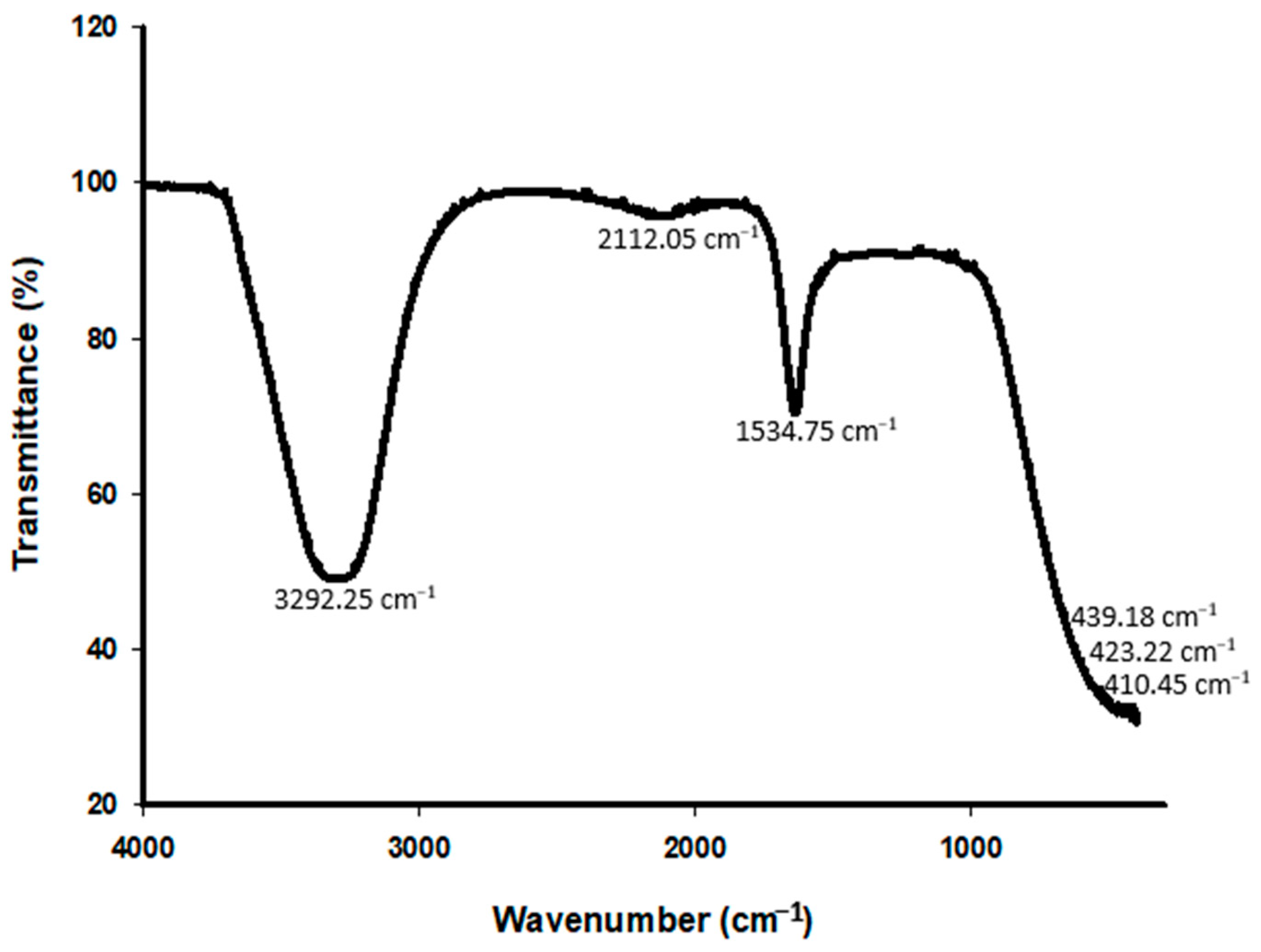
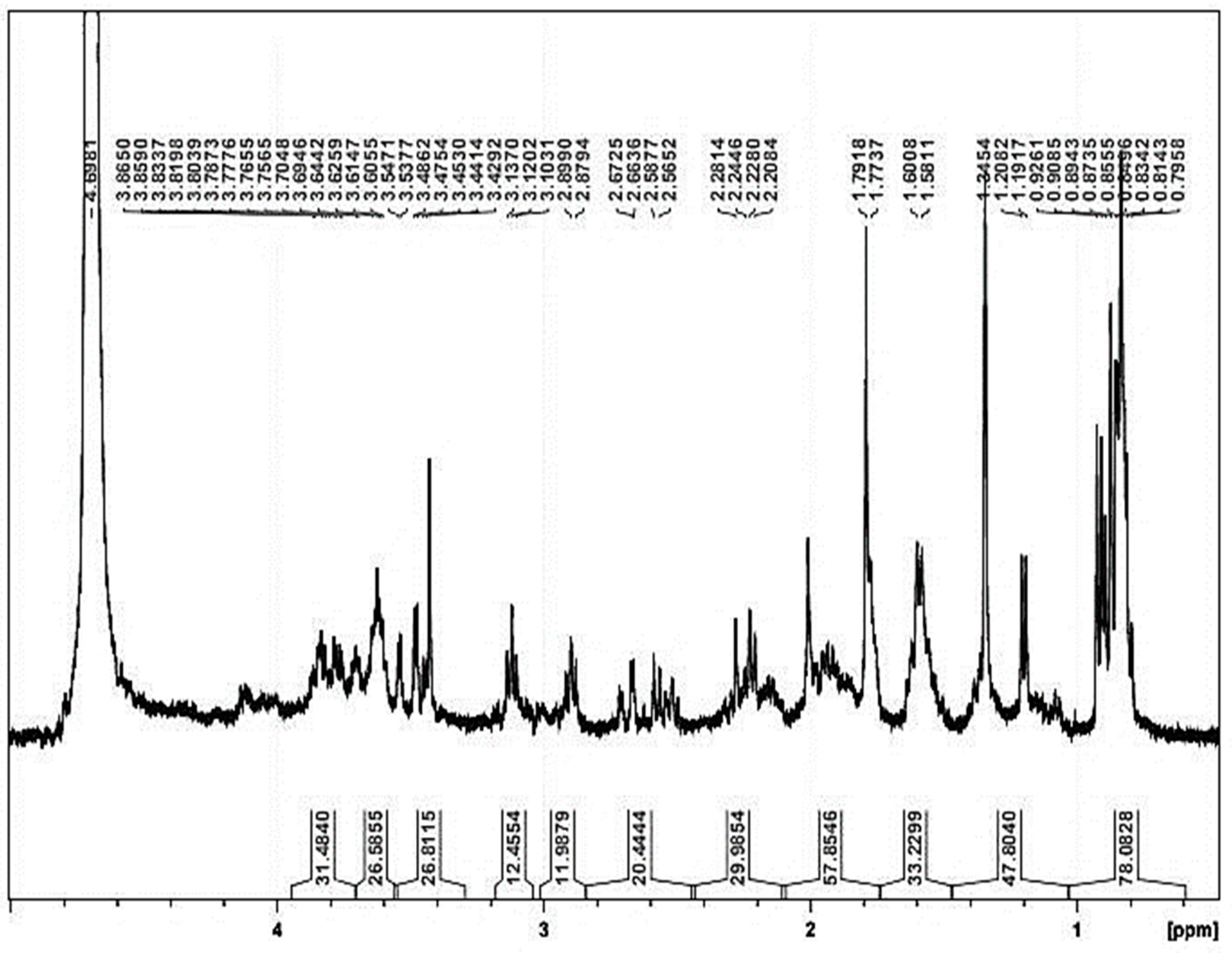
3.6. Biodefoamer FTIR and 1H NMR Characterization
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Burger, W.; Krysiak-Baltyn, K.; Scales, P.J.; Martin, G.J.O.; Stickland, A.D.; Gras, S.L. The influence of protruding filamentous bacteria on floc stability and solid-liquid separation in the activated sludge process. Water Res. 2017, 123, 578–585. [Google Scholar] [CrossRef]
- Zhou, S.; Liu, M.; Chen, B.; Sun, L.; Lu, H. Microbubble- and nanobubble-aeration for upgrading conventional activated sludge process: A review. Bioresour. Technol. 2022, 362, 127826. [Google Scholar] [CrossRef]
- Gemza, N.; Janiak, K.; Zięba, B.; Przyszlak, J.; Kuśnierz, M. Long-term effects of hydrocyclone operation on activated sludge morphology and full-scale secondary settling tank wet-weather operation in long sludge age WWTP. Sci. Total. Environ. 2022, 845, 157224. [Google Scholar] [CrossRef]
- Borzooei, S.; Miranda, G.H.B.; Abolfathi, S.; Scibilia, G.; Meucci, L.; Zanetti, M.C. Application of unsupervised learning and process simulation for energy optimization of a WWTP under various weather conditions. Water Sci. Technol. 2020, 81, 1541–1551. [Google Scholar] [CrossRef]
- Deepnarain, N.; Nasr, M.; Kumari, S.; Stenström, T.A.; Reddy, P.; Pillay, K.; Bux, F. Decision tree for identification and prediction of filamentous bulking at full-scale activated sludge wastewater treatment plant. Process. Saf. Environ. Prot. 2019, 126, 25–34. [Google Scholar] [CrossRef]
- Dlamini, N.G.; Basson, A.K.; Pullabhotla, R.V.S.R. Wastewater treatment by a polymeric bioflocculant and iron nanoparticles synthesized from a bioflocculant. Polymers 2020, 12, 1618. [Google Scholar] [CrossRef]
- Dlangamandla, C.; Basitere, M.; Okeleye, B.I.; Chidi, B.S.; Ntwampe, S.K.O. Biofoam formation and defoamation in global wastewater treatment systems. Water Pract. Technol. 2021, 16, 1–18. [Google Scholar] [CrossRef]
- Borzooei, S.; Amerlinck, Y.; Panepinto, D.; Abolfathi, S.; Nopens, I.; Scibilia, G.; Meucci, L.; Zanetti, M.C. Energy optimization of a wastewater treatment plant based on energy audit data: Small investment with high return. Environ. Sci. Pollut. Res. 2020, 27, 17972–17985. [Google Scholar] [CrossRef]
- Dehestaniathar, S.; Nesari, S.; Borzooei, S.; Abolfathi, S. Application of natural biodegradable fiber as biofilm medium and carbon source in DEnitrifying AMmonium OXidation (DEAMOX) process for nitrogen removal from wastewater. J. Taiwan Inst. Chem. Eng. 2021, 119, 108–114. [Google Scholar] [CrossRef]
- Guo, F.; Wang, Z.P.; Yu, K.; Zhang, T. Detailed investigation of the microbial community in foaming activated sludge reveals novel foam formers. Sci. Rep. 2015, 5, 7637. [Google Scholar] [CrossRef] [Green Version]
- Shi, Y.; Liu, Y. Evolution of extracellular polymeric substances (EPS) in aerobic sludge granulation: Composition, adherence and viscoelastic properties. Chemosphere 2021, 262, 128033. [Google Scholar] [CrossRef]
- Pajdak-Stós, A.; Kocerba-Soroka, W.; Fyda, J.; Sobczyk, M.; Fiałkowska, E. Foam-forming bacteria in activated sludge effectively reduced by rotifers in laboratory- and real-scale wastewater treatment plant experiments. Environ. Sci. Pollut. Res. 2017, 24, 13004–13011. [Google Scholar] [CrossRef] [PubMed]
- Deval, A.S.; Parikh, H.A.; Kadier, A.; Chandrasekhar, K.; Bhagwat, A.M.; Dikshit, A.K. Sequential microbial activities mediated bioelectricity production from distillery wastewater using bio-electrochemical system with simultaneous waste remediation. Int. J. Hydrogen Energy 2017, 42, 1130–1141. [Google Scholar] [CrossRef]
- Bukhari, N.A.; Loh, S.K.; Nasrin, A.B.; Jahim, J.M. Enzymatic Hydrolysate of Palm Oil Mill Effluent as Potential Substrate for Bioflocculant BM-8 Production. Waste Biomass Valorization 2020, 11, 17–29. [Google Scholar] [CrossRef]
- Kougias, P.G.; Boe, K.; Tsapekos, P.; Angelidaki, I. Foam suppression in overloaded manure-based biogas reactors using antifoaming agents. Bioresour. Technol. 2014, 153, 198–205. [Google Scholar] [CrossRef] [PubMed]
- Basitere, M.; Njoya, M.; Ntwampe, S.K.O.; Sheldon, M.S. Up-flow vs downflow anaerobic digester reactor configurations for treatment of fats-oil-grease laden poultry slaughterhouse wastewater: A review. Water Pract. Technol. 2020, 15, 248–260. [Google Scholar] [CrossRef]
- Dlangamandla, C.; Dyantyi, S.A.; Mpentshu, Y.P.; Ntwampe, S.K.O.; Basitere, M. Optimisation of bioflocculant production by a biofilm forming microorganism from poultry slaughterhouse wastewater for use in poultry wastewater treatment. Water Sci. Technol. 2016, 73, 1963–1968. [Google Scholar] [CrossRef]
- Bingo, M.N.; Njoya, M.; Basitere, M.; Ntwampe, S.K.O.; Kaskote, E. Performance evaluation of an integrated multi-stage poultry slaughterhouse wastewater treatment system. J. Water Process. Eng. 2021, 43, 102309. [Google Scholar] [CrossRef]
- Rinquest, Z.; Basitere, M.; Ntwampe, S.K.O.; Njoya, M. Poultry slaughterhouse wastewater treatment using a static granular bed reactor coupled with single stage nitrification-denitrification and ultrafiltration systems. J. Water Process. Eng. 2019, 29, 100778. [Google Scholar] [CrossRef]
- Mangundu, J. Optimisation of Defoamer in A Bio-Reactor. Master’s Thesis, University of the Witwatersrand, Johannesburg, South Africa, 2017. not published. [Google Scholar]
- Martins, A.M.P.; Pagilla, K.; Heijnen, J.J.; Van Loosdrecht, M.C.M. Filamentous bulking sludge-A critical review. Water Res. 2004, 38, 793–817. [Google Scholar] [CrossRef]
- Diao, H.; Li, L.; Liang, J.; Lu, C.; Ding, X.; Guo, W. Optimized extraction process and compositional analysis of bioflocculant produced by Klebsiella M1. BioResources 2019, 14, 3146–3167. [Google Scholar] [CrossRef]
- Garg, A.; Stensel, H.D.; Bucher, B.; Sukapanpotharam, P.; Winkler, M.K.H. Effect of waste activated sludge pretreatment methods to mitigate Gordonia foaming potential in anaerobic digestion. Water Environ. J. 2021, 35, 381–389. [Google Scholar] [CrossRef]
- Tsilo, P.H.; Basson, A.K.; Ntombela, Z.G.; Maliehe, T.S.; Pullabhotla, R.V.S.R. Isolation and Optimization of Culture Conditions of a Bioflocculant-Producing Fungi from Kombucha Tea SCOBY. Microbiol. Res. 2021, 12, 950–966. [Google Scholar] [CrossRef]
- Biswas, J.K.; Banerjee, A.; Sarkar, B.; Sarkar, D.; Sarkar, S.K.; Rai, M.; Vithanage, M. Exploration of an extracellular polymeric substance from earthworm gut bacterium (Bacillus licheniformis) for bioflocculation and heavy metal removal potential. Appl. Sci. 2020, 10, 349. [Google Scholar] [CrossRef]
- Lee, S.; Lee, J.; Hwang, N. Effect of the dispersion state in Y5O4F7 suspension on YOF coating deposited by suspension plasma spray. Coatings 2021, 11, 831. [Google Scholar] [CrossRef]
- Pauzi, S.M.; Halbert, D.A.; Azizi, S.; Ahmad, N.A.A.; Marpani, F. Effect of organic antifoam’s concentrations on filtration performance. J. Phys. Conf. Ser. 2019, 1349, 012141. [Google Scholar] [CrossRef]
- Mamais, D.; Kalaitzi, E.; Andreadakis, A. Foaming control in activated sludge treatment plants by coagulants addition. Glob. Nest J. 2011, 13, 237–245. [Google Scholar] [CrossRef]
- Hatta, N.S.M.; Lau, S.W.; Takeo, M.; Chua, H.B.; Baranwal, P.; Mubarak, N.M.; Khalid, M. Novel cationic chitosan-like bioflocculant from Citrobacter youngae GTC 01314 for the treatment of kaolin suspension and activated sludge. J. Environ. Chem. Eng. 2021, 9, 105297. [Google Scholar] [CrossRef]
- Routledge, S.J. Beyond de-foaming: The effects of antifoams on bioprocess productivity. Comput. Struct. Biotechnol. J. 2012, 3, e201210001. [Google Scholar] [CrossRef]
- Manyele, S. Foaming Characteristics of the Food-Industry Biological Wastewater Treatment Plant. Tanzan. J. Eng. Technol. 2007, 30, 17–28. [Google Scholar]
- Gao, N.; Xia, M.; Dai, J.; Yu, D.; An, W.; Li, S.; Liu, S.; He, P.; Zhang, L.; Wu, Z.; et al. Both widespread PEP-CTERM proteins and exopolysaccharides are required for floc formation of Zoogloea resiniphila and other activated sludge bacteria. Environ. Microbiol. 2018, 20, 1677–1692. [Google Scholar] [CrossRef]
- Wang, J.; Nguyen, A.V.; Farrokhpay, S. Foamability of sodium dodecyl sulfate solutions: Anomalous effect of dodecanol unexplained by conventional theories. Colloids Surf. A Physicochem. Eng. Asp. 2016, 495, 110–117. [Google Scholar] [CrossRef]
- Nilsson, F.; Davidsson, Å.; Falås, P.; Bengtsson, S.; Bester, K.; Jönsson, K. Impact of activated sludge ozonation on filamentous bacteria viability and possible added benefits. Environ. Technol. 2019, 40, 2601–2607. [Google Scholar] [CrossRef]
- Omar, M.M.; Mohamed, G.G.; Ibrahim, A.A. Spectroscopic characterization of metal complexes of novel Schiff base. Synthesis, thermal and biological activity studies. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2009, 73, 358–369. [Google Scholar] [CrossRef]
- Pal, P.; Khairnar, K.; Paunikar, W.N. Causes and remedies for filamentous foaming in activated sludge treatment plant. Glob. NEST J. 2014, 16, 762–772. [Google Scholar]
- Pedrazzani, R.; Menoni, L.; Nembrini, S.; Manili, L.; Bertanza, G. Suitability of Sludge Biotic Index (SBI), Sludge Index (SI) and filamentous bacteria analysis for assessing activated sludge process performance: The case of piggery slaughterhouse wastewater. J. Ind. Microbiol. Biotechnol. 2016, 43, 953–964. [Google Scholar] [CrossRef]
- Petrovski, S.; Dyson, Z.A.; Quill, E.S.; McIlroy, S.J.; Tillett, D.; Seviour, R.J. An examination of the mechanisms for stable foam formation in activated sludge systems. Water Res. 2011, 45, 2146–2154. [Google Scholar] [CrossRef] [PubMed]
- Simjoo, M.; Rezaei, T.; Andrianov, A.; Zitha, P.L.J. Foam stability in the presence of oil: Effect of surfactant concentration and oil type. Colloids Surf. A Physicochem. Eng. Asp. 2013, 438, 148–158. [Google Scholar] [CrossRef]
- Vanaja, M.; Paulkumar, K.; Baburaja, M.; Rajeshkumar, S.; Gnanajobitha, G.; Malarkodi, C.; Sivakavinesan, M.; Annadurai, G. Degradation of methylene blue using biologically synthesized silver nanoparticles. Bioinorg. Chem. Appl. 2014, 2014, 742346. [Google Scholar] [CrossRef] [PubMed]
- Subudhi, S.; Bisht, V.; Batta, N.; Pathak, M.; Devi, A.; Lal, B. Purification and characterization of exopolysaccharide bioflocculant produced by heavy metal resistant Achromobacter xylosoxidans. Carbohydr. Polym. 2016, 137, 441–451. [Google Scholar] [CrossRef]
- Katritzky, A.R.; Akhmedov, N.G.; Denisenko, S.N.; Denisko, O.V. 1H NMR spectroscopic characterization of solutions of Sepia melanin, Sepia melanin free acid and human hair melanin. Pigment Cell Res. 2002, 15, 93–97. [Google Scholar] [CrossRef] [PubMed]

| Variables | Code | Units | High Level (+) | Medium (0) | Low Levels (−) |
|---|---|---|---|---|---|
| pH | A | - | 10.60 | 8.50 | 6.30 |
| Concentration | B | % (v/v) | 4.60 | 2.50 | 1.00 |
| Parameters | Minimum (mg/L) | Maximum (mg/L) | Average (mg/L) | References | Current Study (mg/L) |
|---|---|---|---|---|---|
| FOG | 131 | 684 | 406 | [16] | 710–1178 |
| Proteins | 0 | 368 | 184 | [17] | 29–96 |
| TSS | 198 | 3100 | 1207 | [18] | 2000–10,000 |
| COD | 2517 | 12,490 | 5216 ± 2534 | [19] | 4510–8355 |
| Dimensions | Specifications |
|---|---|
| Cylinder | |
| Material | Polypropylene |
| Height | 24.5 cm |
| Diameter | 3 cm |
| Air flow rate | 80 mL/min |
| Diffusers | |
| Porous length | 2.32 cm |
| Porous diameter | 1.2 cm |
| Media grade | 40 |
| Porous material | 316LSS |
| Run | pH | Concentration (%v/v) | Foam Reduction Efficiency (%v/v) | Foam Collapse Rate (mm/s) | ||
|---|---|---|---|---|---|---|
| Synthetic Defoamer | Biodefoamer | Synthetic Defoamer | Biodefoamer | |||
| 1 | 8.5 | 0.38 | 93 | 73 | 0.83 | 0.5 |
| 2 | 10 | 1 | 2.9 | 73 | 0.55 | 0.55 |
| 3 | 8.5 | 2.5 | 83 | 82 | 0.42 | 0.5 |
| 4 | 8.5 | 2.5 | 83 | 82 | 0.42 | 0.5 |
| 5 | 8.5 | 4.6 | 15 | 15 | 0.5 | 0.5 |
| 6 | 6.38 | 2.5 | 96 | 42 | 1 | 0.5 |
| 7 | 8.5 | 2.5 | 83 | 82 | 0.42 | 0.5 |
| 8 | 8.5 | 2.5 | 83 | 82 | 0.42 | 0.5 |
| 9 | 10.6 | 2.5 | 87 | 73 | 0.45 | 0.45 |
| 10 | 8.5 | 2.5 | 83 | 82 | 0.42 | 0.5 |
| 11 | 7 | 1 | 90 | 73 | 1.25 | 0.83 |
| 12 | 7 | 4 | 96 | 96 | 1.7 | 2.5 |
| 13 | 10 | 4 | 95 | 78 | 0.42 | 0.42 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dlangamandla, C.; Ntwampe, S.K.O.; Basitere, M.; Chidi, B.S.; Okeleye, B.I.; Mukandi, M.R. Production, Application, and Efficacy of Biodefoamers from Bacillus, Aeromonas, Klebsiella, Comamonas spp. Consortium for the Defoamation of Poultry Slaughterhouse Wastewater. Water 2023, 15, 655. https://doi.org/10.3390/w15040655
Dlangamandla C, Ntwampe SKO, Basitere M, Chidi BS, Okeleye BI, Mukandi MR. Production, Application, and Efficacy of Biodefoamers from Bacillus, Aeromonas, Klebsiella, Comamonas spp. Consortium for the Defoamation of Poultry Slaughterhouse Wastewater. Water. 2023; 15(4):655. https://doi.org/10.3390/w15040655
Chicago/Turabian StyleDlangamandla, Cynthia, Seteno K. O. Ntwampe, Moses Basitere, Boredi S. Chidi, Benjamin I. Okeleye, and Melody R. Mukandi. 2023. "Production, Application, and Efficacy of Biodefoamers from Bacillus, Aeromonas, Klebsiella, Comamonas spp. Consortium for the Defoamation of Poultry Slaughterhouse Wastewater" Water 15, no. 4: 655. https://doi.org/10.3390/w15040655





