Application Progress of New Adsorption Materials for Removing Fluorine from Water
Abstract
:1. Introduction
2. New Defluorination Adsorbent and Its Mechanism
2.1. Metal−Based Adsorbents
2.1.1. Aluminum−Based Adsorbent
2.1.2. Iron−Based Adsorbent
2.1.3. Calcium−Based Adsorbent
2.2. Natural Adsorbent
2.2.1. Zeolite
2.2.2. Natural Minerals
2.2.3. Other Modified Natural Adsorbents
2.3. Nanomaterial Adsorbents
2.4. Carbon−Based Adsorbents
2.5. Composite Metallization
2.6. Layered Double Hydroxide
2.7. Agricultural and Forestry Waste
2.8. Comparison of Different Adsorbents
3. Defluorination by Adsorption Method Coupled with Other Processes
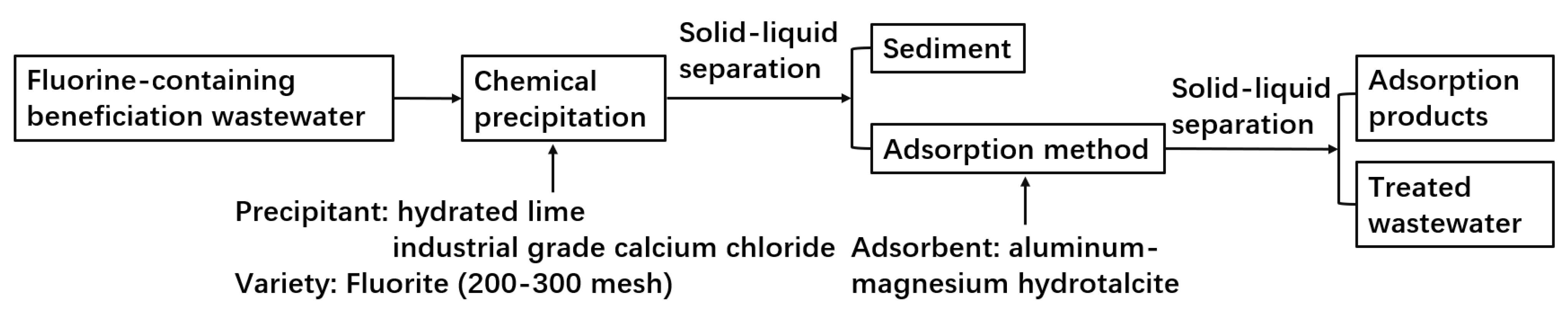
3.1. Chemical Precipitation−Adsorption Method
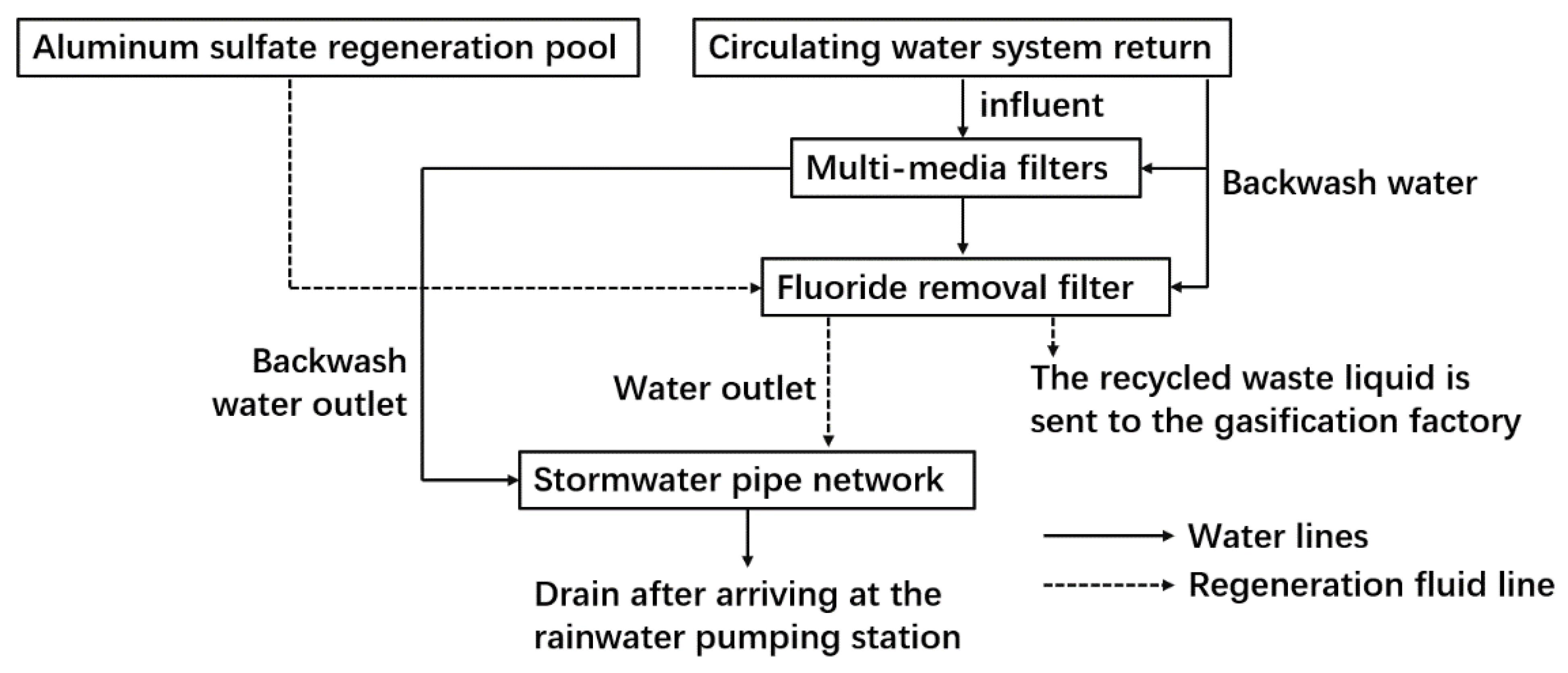
3.2. Filtration−Adsorption Method
3.3. Super−Magnetic Separation Technology−Adsorption Method
4. Conclusions
- (a)
- Recyclability. In the current research, there are only a few works of literature on the cyclic regeneration of adsorbents. More detailed regeneration studies should be conducted on adsorbents with lower regeneration efficiency to improve their fluorine absorption efficiency and regeneration efficiency while reducing the cost and waste generated by the adsorption process, thereby improving the economic viability of the process.
- (b)
- Suitability. A new type of adsorbent may show a high fluorine removal rate in laboratory studies, but it will be affected by many factors in practical application, which may significantly reduce its fluorine removal efficiency. Practical research on adsorbents, such as enhancing hydraulic conductivity, should be strengthened to enhance the practical value of these adsorbents on a commercial scale.
- (c)
- Environmental Safety. Research on the environmental safety of adsorbents should be strengthened because both of emerging nano−adsorbent materials and traditional F adsorbents may cause secondary pollution of environment.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Paudyal, H.; Pangeni, B.; Inoue, K.; Kawakita, H.; Ohto, K.; Harada, H.; Alam, S. Adsorptive removal of fluoride from aqueous solution using orange waste loaded with multi−valent metal ions. J. Hazard. Mater. 2011, 192, 676–682. [Google Scholar] [CrossRef]
- Kawamura, K.; Kunimatsu, Y.; Nakano, T.; Hasegawa, H.; Arakawa, H.; Mukai, Y. Anti−demineralization effect of desensitizer containing copolymer and sodium fluoride on root dentin—A transverse microradiographic study. Acta Biomater. Odontol. Scand. 2019, 5, 38–43. [Google Scholar] [CrossRef]
- Nakata, T.; Kitasako, Y.; Sadr, A.; Nakashima, S.; Tagami, J. Effect of a calcium phosphate and fluoride paste on prevention of enamel demineralization. Dent. Mater. J. 2018, 37, 65–70. [Google Scholar] [CrossRef] [PubMed]
- Schlueter, N.; Ganss, C.; Mueller, U.; Klimek, J. Effect of titanium tetrafluoride and sodium fluoride on erosion progression in enamel and dentine in vitro. Caries Res. 2007, 41, 141–145. [Google Scholar] [CrossRef]
- Nozari, A.; Ajami, S.; Rafiei, A.; Niazi, E. Impact of nano hydroxyapatite, nano silver fluoride and sodium fluoride varnish on primary teeth enamel remineralization: An In vitro study. J. Clin. Diagn. Res. 2017, 11, 97–100. [Google Scholar]
- Owusu−Agyeman, I.; Shen, J.; Schäfer, A.I. Renewable energy powered membrane technology: Impact of pH and ionic strength on fluoride and natural organic matter removal. Sci. Total. Environ. 2018, 621, 138–147. [Google Scholar] [CrossRef]
- Ruiz−Payan, A.; Ortiz, M.; Duarte−Gardea, M. Determination of fluoride in drinking water and in urine of adolescents living in three counties in Northern Chihuahua Mexico using a fluoride ion selective electrode. Microchem. J. 2005, 81, 19–22. [Google Scholar] [CrossRef]
- Rocha, R.A.; Calatayud, M.; Devesa, V.; Vélez, D. Evaluation of exposure to fluoride in child population of North Argentina. Environ. Sci. Pollut. Res. Int. 2017, 24, 22040–22047. [Google Scholar] [CrossRef] [PubMed]
- Ozsvath, D.L. Fluoride and environmental health: A review. Rev. Environ. Sci. Bio/Technol. 2008, 8, 59–79. [Google Scholar] [CrossRef]
- Dec, K.; Łukomska, A.; Maciejewska, D.; Jakubczyk, K.; Baranowska−Bosiacka, I.; Chlubek, D.; Wąsik, A.; Gutowska, I. The Influence of fluorine on the disturbances of homeostasis in the central nervous system. Biol. Trace Elem. Res. 2017, 177, 224–234. [Google Scholar] [CrossRef]
- Jha, S.K.; Singh, R.K.; Damodaran, T.; Mishra, V.K.; Sharma, D.K.; Rai, D. Fluoride in groundwater: Toxicological exposure and remedies. J. Toxicol. Environ. Health Part B 2013, 16, 52–66. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Chen, C.; Chen, Y. China’s drinking water hygiene standards. Chin. J. Public Health 2007, 23, 1281–1282. [Google Scholar]
- Dong, J. Treatment of highly concentrated fluoride−containing wastewater by preliminary sedimentation−coagulation precipitation−adsorption process. Ind. Water Treat. 2014, 34, 82–84. [Google Scholar]
- Cheng, H. Study on Treatment Methods of High Fluorine Wastewater; Lanzhou Jiaotong University: Lanzhou, China, 2019. [Google Scholar]
- Song, Y.; Zhou, L.; Hao, W.; Song, W.; Li, Y. Pollution status and related research progress of perfluorinated compounds. Environ. Eng. 2017, 35, 82–86. [Google Scholar]
- Banks, D.; Reimann, C.; Røyset, O.; Skarphagen, H.; Sæther, O.M. Natural concentrations of major and trace elements in some Norwegian bedrock groundwaters. Appl. Geochem. 1995, 10, 1–16. [Google Scholar] [CrossRef]
- Lü, J.; Qiu, H.; Lin, H.; Yuan, Y.; Chen, Z.; Zhao, R. Source apportionment of fluorine pollution in regional shallow groundwater at You’xi County southeast China. Chemosphere 2016, 158, 50–55. [Google Scholar] [CrossRef]
- Narsimha, A.; Rajitha, S. Spatial distribution and seasonal variation in fluoride enrichment in groundwater and its associated human health risk assessment in Telangana State, South India. Hum. Ecol. Risk Assess. Int. J. 2018, 24, 2119–2132. [Google Scholar] [CrossRef]
- Peng, S.; Qing, T.; Yu, X.; Yu, L.; Hui, X. Effect of humic acid in water on Al13 coagulation defluorination. Chin. J. Environ. Eng. 2022, 16, 143–153. [Google Scholar]
- Gan, Y.; Wang, X.; Zhang, L.; Wu, B.; Zhang, G.; Zhang, S. Coagulation removal of fluoride by zirconium tetrachloride: Performance evaluation and mechanism analysis. Chemosphere 2019, 218, 860–868. [Google Scholar] [CrossRef]
- Choong, C.E.; Wong, K.T.; Jang, S.B.; Nah, I.W.; Choi, J.; Ibrahim, S.; Yoon, Y.; Jang, M. Fluoride removal by palm shell waste based powdered activated carbon vs. functionalized carbon with magnesium silicate: Implications for their application in water treatment. Chemosphere 2019, 239, 124765. [Google Scholar] [CrossRef]
- Paudyal, H.; Inoue, K.; Kawakita, H.; Ohto, K.; Kamata, H.; Alam, S. Removal of fluoride by effectively using spent cation exchange resin. J. Mater. Cycles Waste Manag. 2017, 20, 975–984. [Google Scholar] [CrossRef]
- Gmar, S.; Sayadi, I.B.S.; Helali, N.; Tlili, M.; Ben Amor, M. Desalination and Defluoridation of Tap Water by Electrodialysis. Environ. Process. 2015, 2, 209–222. [Google Scholar] [CrossRef]
- Sharma, P.P.; Yadav, V.; Maru, P.D.; Makwana, B.S.; Sharma, S.; Kulshrestha, V. Mitigation of Fluoride from Brackish Water via Electrodialysis: An Environmentally Friendly Process. Chemistryselect 2018, 3, 779–784. [Google Scholar] [CrossRef]
- Zhang, J.; Brutus, T.E.; Cheng, J.; Meng, X. Fluoride removal by Al, Ti, and Fe hydroxides and coexisting ion effect. J. Environ. Sci. 2017, 57, 190–195. [Google Scholar] [CrossRef]
- Sehn, P. Fluoride removal with extra low energy reverse osmosis membranes: Three years of large scale field experience in Finland. Desalination 2008, 223, 73–84. [Google Scholar] [CrossRef]
- Delgadillo−Velasco, L.; Hernández−Montoya, V.; Cervantes, F.J.; Montes−Morán, M.A.; Lira−Berlanga, D. Bone char with antibacterial properties for fluoride removal: Preparation, characterization and water treatment. J. Environ. Manag. 2017, 201, 277–285. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Yu, Y.; Ding, Z.; Zuo, M.; Jing, C. Competitive adsorption of arsenic and fluoride on {201} TiO2. Appl. Surf. Sci. 2019, 466, 425–432. [Google Scholar] [CrossRef]
- Nazir, M.A.; Najam, T.; Shahzad, K.; Wattoo, M.A.; Hussain, T.; Tufail, M.K.; Shah, S.S.A.; Rehman, A.U. Heterointerface engineering of water stable ZIF−8@ZIF−67: Adsorption of rhodamine B from water. Surfaces Interfaces 2022, 34, 102324. [Google Scholar] [CrossRef]
- Nazir, M.A.; Najam, T.; Jabeen, S.; Wattoo, M.A.; Bashir, M.S.; Shah, S.S.A.; Rehman, A. Facile synthesis of Tri−metallic layered double hydroxides (NiZnAl−LDHs): Adsorption of Rhodamine−B and methyl orange from water. Inorg. Chem. Commun. 2022, 145, 110008. [Google Scholar] [CrossRef]
- Shahzad, K.; Nazir, M.A.; Jamshaid, M.; Kumar, O.P.; Najam, T.; Shah, S.S.A.; Rehman, A.U. Synthesis of nanoadsorbent entailed mesoporous organosilica for decontamination of methylene blue and methyl orange from water. Int. J. Environ. Anal. Chem. 2021, 1–14. [Google Scholar] [CrossRef]
- Hunge, Y.; Yadav, A.; Kang, S.−W.; Kim, H.; Fujishima, A.; Terashima, C. Nanoflakes−like nickel cobaltite as active electrode material for 4−nitrophenol reduction and supercapacitor applications. J. Hazard. Mater. 2021, 419, 126453. [Google Scholar] [CrossRef]
- Hunge, Y.; Yadav, A.; Kang, S.−W.; Kim, H. Facile synthesis of multitasking composite of Silver nanoparticle with Zinc oxide for 4−nitrophenol reduction, photocatalytic hydrogen production, and 4−chlorophenol degradation. J. Alloys Compd. 2022, 928, 167133. [Google Scholar] [CrossRef]
- Hunge, Y.M.; Yadav, A.; Kang, S. −W. Photocatalytic Degradation of Eriochrome Black−T Using BaWO4/MoS2 Composite. Catalysts 2022, 12, 1290. [Google Scholar] [CrossRef]
- Bhatnagar, A.; Kumar, E.; Sillanpää, M. Fluoride removal from water by adsorption—A review. Chem. Eng. J. 2011, 171, 811–840. [Google Scholar] [CrossRef]
- Yadav, K.K.; Gupta, N.; Kumar, V.; Khan, S.A.; Kumar, A. A review of emerging adsorbents and current demand for defluoridation of water: Bright future in water sustainability. Environ. Int. 2018, 111, 80–108. [Google Scholar] [CrossRef]
- Mohapatra, M.; Anand, S.; Mishra, B.; Giles, D.E.; Singh, P. Review of fluoride removal from drinking water. J. Environ. Manag. 2009, 91, 67–77. [Google Scholar] [CrossRef]
- Belekar, R.; Dhoble, S. Activated Alumina Granules with nanoscale porosity for water defluoridation. Nano−Struct. Nano−Objects 2018, 16, 322–328. [Google Scholar] [CrossRef]
- Xu, L.; Ma, P.; Ding, W. Defluorination performance of activated alumina modified by ferric sulfate. J. Beijing Univ. Chem. Technol. 2017, 44, 18–24. [Google Scholar]
- Kumari, U.; Siddiqi, H.; Bal, M.; Meikap, B. Calcium and zirconium modified acid activated alumina for adsorptive removal of fluoride: Performance evaluation, kinetics, isotherm, characterization and industrial wastewater treatment. Adv. Powder Technol. 2020, 31, 2045–2060. [Google Scholar] [CrossRef]
- Li, J. Preparation of Novel Nano Materials for Fluoride Adsorption from Water; Shihezi University: Shihezi, China, 2019. [Google Scholar]
- Cao, Z.; Xu, J.; Li, H.; Ma, T.; Lou, L.; Henkelman, G.; Xu, X. Dechlorination and defluorination capability of sulfidized nanoscale zerovalent iron with sup−pressed water reactivity. Chem. Eng. J. 2020, 400, 125–900. [Google Scholar] [CrossRef]
- Wu, C.; Chen, C.; Liang, Z.; Huang, S. Adsorption of Fluoride from Groundwater by Nanoscale Fe3O4. J. Green Sci. Technol. 2019, 2019, 79–82. [Google Scholar] [CrossRef]
- Xiang, W.; Zhu, Z.; Wei, S. Preparation, characterization and fluoride adsorption characteristics of three hematite samples. Chem. Res. Appl. 2018, 30, 290–296. [Google Scholar]
- Yang, C.; Guan, L.; Wang, J.; Yang, X.; Lin, M.; You, G.; Tan, S.; Yu, X.; Ge, M. Enhanced fluoride removal behaviour and mechanism by dicalcium phosphate from aqueous solution. Environ. Technol. 2018, 40, 3668–3677. [Google Scholar] [CrossRef]
- Arcibar−Orozco, J.A.; Flores−Rojas, A.I.; Rangel−Mendez, J.R.; Díaz−Flores, P.E. Synergistic effect of zeolite/chitosan in the removal of fluoride from aqueous solution. Environ. Technol. 2020, 41, 1554–1567. [Google Scholar] [CrossRef]
- Rahmani, A.; Nouri, J.; Ghadiri, S.K.; Mahvi, A.; ZareM, R. Adsorption of fluoride from water by Al3+ and Fe3+ pretreated natural Iranian zeolites. Int. J. Environ. 2010, 4, 607–614. [Google Scholar] [CrossRef]
- Nabbou, N.; Belhachemi, M.; Boumelik, M.; Merzougui, T.; Lahcene, D.; Harek, Y.; Zorpas, A.A.; Jeguirim, M. Removal of fluoride from groundwater using natural clay (kaolinite): Optimization of ad−sorption conditions. Comptes Rendus Chim. 2019, 22, 105–112. [Google Scholar] [CrossRef]
- Lu, C.; Gou, X.; Han, H.; Sun, W. Adsorption of fluoride by natural aluminosilicate minerals. Conserv. Util. Miner. Resour. 2020, 40, 28–36. [Google Scholar]
- Wang, E.; Guo, Z.; Lei, S.; Zhang, S.; Zhong, L. Research on adsorption and purification technology for fluorinion from mineral processing wastewater. Min. Res. Dev. 2015, 35, 71–74. [Google Scholar]
- Malakootian, M.; Moosazadeh, M.; Yousefi, N.; Fatehizadeh, A. Fluoride removal from aqueous solution by pumice: Case study on Kuhbonan water. Afr. J. Environ. Sci. Technol. 2011, 5, 299–306. [Google Scholar]
- Sengupta, P.; Saha, S.; Banerjee, S.; Dey, A.; Sarkar, P. Removal of fluoride ion from drinking water by a new Fe(OH)3/nano CaO impregnated chitosan composite adsorbent. Polym. Plast. Technol. Mater. 2020, 59, 1191–1203. [Google Scholar] [CrossRef]
- Premathilaka, R.W.; Liyanagedera, N.D. Fluoride in Drinking Water and Nanotechnological Approaches for Eliminating Excess Fluoride. J. Nanotechnol. 2019, 2019, 2192383. [Google Scholar] [CrossRef]
- Zari, F.; Nasr, I.; Mahmood, T. Removal of fluoride ion from aqueous solutions by titania−grafted β−cyclodextrin nanocomposite. Environ. Sci. Pollut. Res. Int. 2020, 27, 3281–3294. [Google Scholar]
- Zaidi, R.; Khan, S.U.; Farooqi, I.H.; Azam, A. Investigation of kinetics and adsorption isotherm for fluoride removal from aqueous solutions using mesoporous cerium−aluminum binary oxide nanomaterials. RSC Adv. 2021, 11, 28744–28760. [Google Scholar] [CrossRef]
- Bakhta, S.; Sadaoui, Z.; Lassi, U.; Romar, H.; Kupila, R.; Vieillard, J. Performances of metals modified activated carbons for fluoride removal from aqueous solutions. Chem. Phys. Lett. 2020, 754, 137705. [Google Scholar] [CrossRef]
- Thakur, R.S.; Katoch, S.S.; Modi, A. Assessment of pine cone derived activated carbon as an adsorbent in defluoridation. SN Appl. Sci. 2020, 2, 1–12. [Google Scholar] [CrossRef]
- Pang, T.; Chan, T.S.A.; Jande, Y.A.C.; Shen, J. Removal of fluoride from water using activated carbon fibres modified with zirconium by a drop−coating method. Chemosphere 2020, 255, 126950. [Google Scholar] [CrossRef]
- Chaudhary, M.; Rawat, S.; Jain, N.; Bhatnagar, A.; Maiti, A. Chitosan−Fe−Al−Mn metal oxyhydroxides composite as highly efficient fluoride scavenger for aqueous medium. Carbohydr. Polym. 2019, 216, 140–148. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Wu, L.; Li, J.; Tang, D.; Zhang, G. Simultaneous and efficient removal of fluoride and phosphate by Fe−La composite: Adsorption kinetics and mechanism. J. Alloys Compd. 2018, 753, 422–432. [Google Scholar] [CrossRef]
- Liu, J.; Xie, L.; Yue, X.; Xu, C.; Lu, X. Removal of fluoride and hardness by layered double hydroxides: Property and mechanism. Int. J. Environ. Sci. Technol. 2019, 17, 673–682. [Google Scholar] [CrossRef]
- Li, F.; Jin, J.; Shen, Z.; Ji, H.; Yang, M.; Yin, Y. Removal and recovery of phosphate and fluoride from water with reusable mesoporous Fe3O4@mSiO2@mLDH composites as sorbents. J. Hazard. Mater. 2019, 388, 121734. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, M.; Siddique, A.; Verma, L.; Singh, J.; Koduru, J.R. Adsorptive removal of fluoride from aqueous solution by biogenic iron permeated activated carbon derived from sweet lime waste. Acta Chim. Slov. 2019, 66, 123–136. [Google Scholar] [CrossRef] [PubMed]
- Tao, W.; Zhong, H.; Pan, X.; Wang, P.; Wang, H.; Huang, L. Removal of fluoride from wastewater solution using Ce−AlOOH with oxalic acid as modification. J. Hazard. Mater. 2020, 384, 121373. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Meng, F.; Yang, M. Preparation of magnetic nanomaterial Fe3O4@Ce(OH)3 and its fluoride removal performance. Environ. Prot. Technol. 2019, 25, 5–10+15. [Google Scholar]
- Huiling, W.; Yanyan, Z.; Wei, X.; Rimao, H.; Xuede, L. Fluorine removing performance and mechanism of modified activated magnesium oxide. Chin. J. Environ. Eng. 2015, 9, 2125–2130. [Google Scholar]
- Pillai, P.; Dharaskar, S.; Shah, M.; Sultania, R. Determination of fluoride removal using silica nano adsorbent modified by rice husk from water. Groundw. Sustain. Dev. 2020, 11, 100423. [Google Scholar] [CrossRef]
- Rashid, U.S.; Bezbaruah, A.N. Citric acid modified granular activated carbon for enhanced defluoridation. Chemosphere 2020, 252, 126639. [Google Scholar] [CrossRef]
- Mei, L.; Qiao, H.; Ke, F.; Peng, C.; Hou, R.; Wan, X.; Cai, H. One−step synthesis of zirconium dioxide−biochar derived from Camellia oleifera seed shell with enhanced removal capacity for fluoride from water. Appl. Surf. Sci. 2019, 509, 144685. [Google Scholar] [CrossRef]
- Kim, W.; Singh, R.; Smith, J.A. Modified crushed oyster shells for fluoride removal from water. Sci. Rep. 2020, 10, 57–59. [Google Scholar] [CrossRef]
- Huang, S.; Hu, M.; Li, D.; Wang, L.; Zhang, C.; Li, K.; He, Q. Fluoride sorption from aqueous solution using Al(OH)3−modified hydroxyapatite nanosheet. Fuel 2020, 279, 118486. [Google Scholar] [CrossRef]
- Vilakati, B.R.; Sivasankar, V.; Nxumalo, E.N.; Mamba, B.B.; Omine, K.; Msagati, T.A. Fluoride removal studies using virgin and Ti (IV)−modified Musa paradisiaca (plantain pseudo−stem) carbons. Environ. Sci. Pollut. Res. Int. 2019, 26, 11565–11578. [Google Scholar] [CrossRef]
- Mupa, M.; Gwaku, F.; Gwizangwe, I. Pediastrum boryanum Immobilized on Rice Husk Ash Silica as Bio−sorbent for Fluoride Removal from Drinking Water. Indian J. Sci. Technol. 2016, 9, 48. [Google Scholar] [CrossRef]
- Zhang, Y.; Huang, K. Grape pomace as a biosorbent for fluoride removal from groundwater. RSC Adv. 2019, 9, 7767–7776. [Google Scholar] [CrossRef]
- Siddique, A.; Nayak, A.; Singh, J. Synthesis of FeCl3 −activated carbon derived from waste Citrus limetta peels for removal of fluoride: An eco−friendly approach for the treatment of groundwater and bio−waste collectively. Groundw. Sustain. Dev. 2020, 10, 100339. [Google Scholar] [CrossRef]
- Teng, S.X.; Wang, S.G.; Gong, W.X.; Liu, X.W.; Gao, B.Y. Removal of Fluoride in Alkaline Solution Using Fine Alumina with High Specific Surface Area. Mater. Rep. 2020, 34, 4020–4024+4055. [Google Scholar]
- Li, X.; Liu, S.; Liu, Y.; Shang, F.; Lv, Y.; Zhang, Z. Experimental study on adsorption of fluoride ion in froundwater by serpentineclay composite. Bull. Chin. Ceram. Soc. 2018, 37, 3343–3348+3354. [Google Scholar]
- Ding, C.; Xu, F.; Liu, F.; Wang, H.; Jiang, L.; Sun, B.; Zhu, S. The preparation of iron−aluminum double hydroxide and study on fluoride removal in water. Environ. Dev. 2019, 31, 90–93+95. [Google Scholar]
- Tang, D.; Zhang, G. Efficient removal of fluoride by hierarchical Ce−Fe bimetal oxides adsorbent: Thermo−dynamics, kinetics and mechanism. Chem. Eng. J. 2016, 283, 721–729. [Google Scholar] [CrossRef]
- Jiang, Y. Study on Treatment of Fluoride−Containing Beneficiatic on Wastewater by Chemical Precipitation and Adsorption Composite Technology; Southwest University of Science and Technology: Mianyang, China, 2020. [Google Scholar]
- Xue, Z. Application of activated alumina in fluoride removal from industrial circulation. Chem. Eng. Des. Commun. 2020, 46, 258+275. [Google Scholar]
- He, S.; Ji, J.; Huang, H.; Xiao, B.; Yi, Y.; Yang, J. Experimental study on removal of high concentration fluoride wastewater by ultra−magnetic separator and adsorption method. Environ. Eng. 2019, 37, 20–23. [Google Scholar]
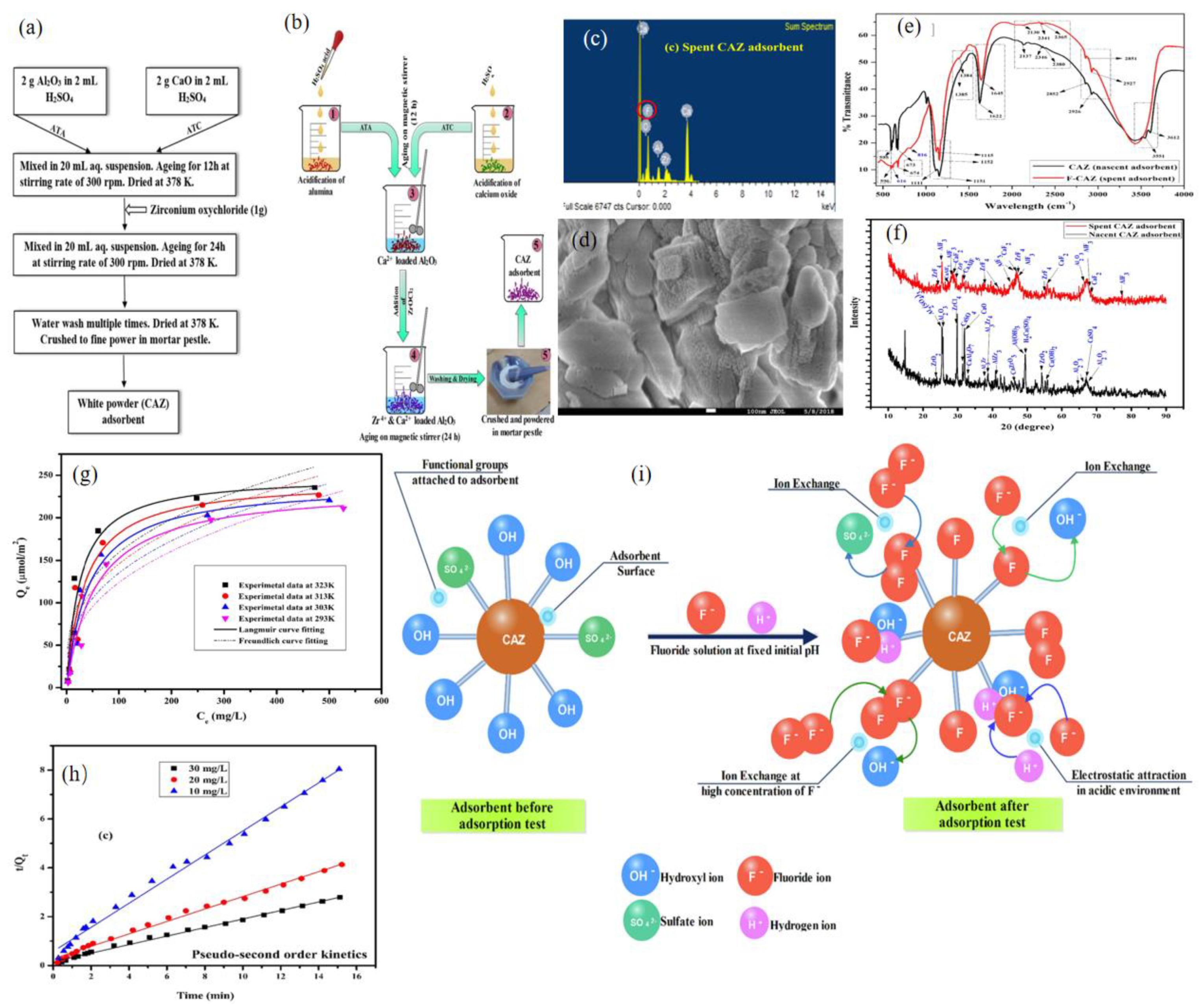
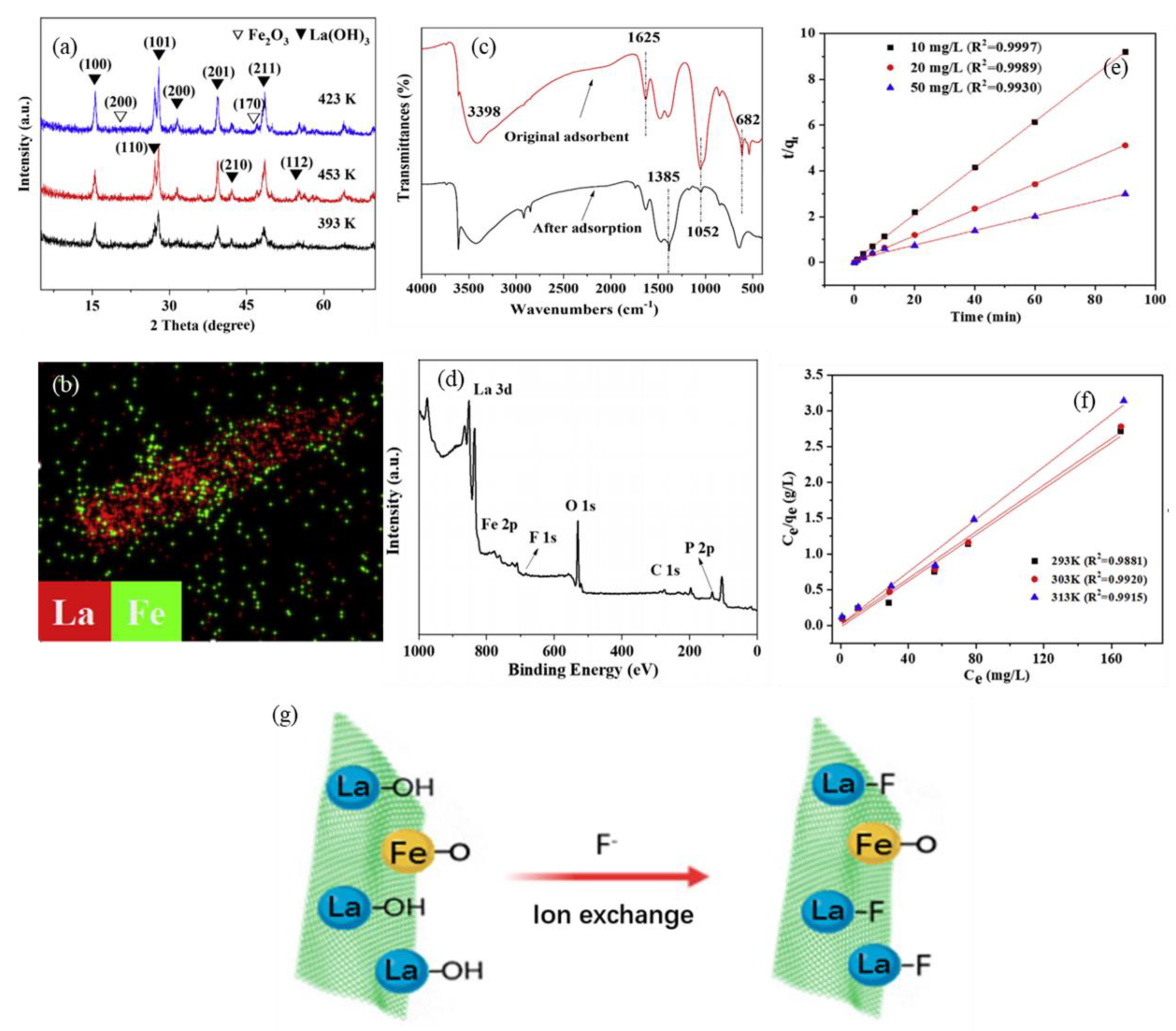


| Adsorbent | Adsorption Capacity (mg g−1) | Concentration Range (mg L−1) | Adsorption Time (h) | pH | Adsorption Isotherms | Adsorption Kinetic | References |
|---|---|---|---|---|---|---|---|
| Ferric sulfate solution modified activated alumina | 16.78 | 20 | 12 | 5 ± 0.2 | Langmuir | pseudo−secondorder kinetics | [39] |
| Calcium zirconium modified acid activated alumina | 216 | 30 | 3 | 6 | Langmuir | pseudo−secondorder kinetics | [40] |
| Dicalcium phosphate | 66.72 | 50 | 4 | 3~10 | Langmuir | pseudo−secondorder kinetics | [45] |
| Nanoscale iron oxide | 2.1834 | 5 | 2.5 | 3 | Freundlich | pseudo−secondorder kinetics | [43] |
| Non−calcined synthetic hydroxyapatite | 6.30 | 10 | 24 | 5 | Langmuir | − | [46] |
| Kaolin | 0.556 | 5 | 2 | 4~6 | Freundlich | pseudosecond−order | [48] |
| Modified citrus biochar | 12.6 | 5~30 | 3 | 4 | Langmuir | pseudo−secondorder kinetics | [63] |
| Palm shell activated carbon powder | 116 | 5~125 | 2 | 5 | Langmuir | pseudo−secondorder kinetics | [21] |
| Magnesium silicate−modified palm shell−activated carbon powder | 150 | 5~125 | 2 | 5 | Freundlich | pseudosecond−order | [21] |
| Pine cone activated carbon | 1.12 | 6 | 1 | 6.5~7.5 | Langmuir | pseudo−secondorder kinetics | [57] |
| Zirconium modified activated carbon fiber | 20 | 28.50 | 6 | 5 | Langmuir | pseudo−secondorder kinetics | [58] |
| Mg Al−LDH composite material | 28.51 | 10 | 24 | 3 | Langmuir | − | [62] |
| Chitosan−Fe−Al−Mn composite adsorbent | 40 ± 0.5 | 10 | 0.75 | 3.5~8.5 | Freundlich | pseudosecond−order | [59] |
| Fe−La composites | 27.42 | 10 | − | 4~6 | Freundlich | pseudosecond−order | [60] |
| Ce−AlOOH | 88 | 100 | 2 | 1~3 | Freundlich | pseudosecond−order | [64] |
| Fe3O4 @ Ce(OH)3 nanomaterials | 59.52 | 100 | 0.33 | 3.5 | Langmuir | pseudo−secondorder kinetics | [65] |
| Modified activated magnesium oxide | 88.50 | 5~10 | 5 | 4~9.5 | Langmuir | pseudo−secondorder kinetics | [66] |
| Rice husk modified silica nanoparticles | 28.50 | 10 | 1 | 8 | Freundlich | pseudosecond−order | [67] |
| Citric acid modified activated carbon | 1.65 | 5 | 4 | 2 | D−R | pseudosecond−order | [68] |
| Camellia seed shell biochar | 11.04 | 10 | 3 | 3~9 | Langmuir | pseudo−secondorder kinetics | [69] |
| Modified crushed oyster shell biochar | 10 | 10 | − | 4~10 | Freundlich | − | [70] |
| Al(OH)3−hydroxyapatite nanosheets | 194.2 | 200 | 2 | 3~9 | Freundlich | pseudosecond−order | [71] |
| Ti(IV) modified plantain | 17.2 | 2 | 1 | 2 | Langmuir | pseudo−secondorder kinetics | [72] |
| Rice husk ash silica | 25.641 | − | 1.5 | 5 | Langmuir | pseudo−secondorder kinetics | [73] |
| Zr−modified grape pomace | 7.54 | 19.91 | 1 | 3 | Langmuir | − | [74] |
| Aluminum−modified jujube mud biochar | 13.03 | 10 | 1 | 5~10 | Langmuir | − | [56] |
| Modified FeCl3−lemon sheet−activated carbon | 4.926~9.709 | 1000 | 4 | 6.6 | Langmuir | pseudo−secondorder kinetics | [75] |
| Alumina powder | 9.28 | 50~500 | 20 | 8. 1 | Langmuir | pseudo−secondorder kinetics | [76] |
| Serpentine−clay composite particles | − | 5~15 | 2 | 6.5~7.0 | Langmuir | pseudo−secondorder kinetics | [77] |
| Iron−aluminum layered double hydroxide | 87.09 | − | 2 | 3~10 | Freundlich | pseudosecond−order | [78] |
| Cerium−iron bimetallic oxide adsorbent | 60.97 | 10 | 0.67 | 2.9~10.1 | Langmuir | pseudo−secondorder kinetics | [79] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, M.; Wang, Q.; Krua, L.S.N.; Yi, R.; Zou, R.; Li, X.; Huang, P. Application Progress of New Adsorption Materials for Removing Fluorine from Water. Water 2023, 15, 646. https://doi.org/10.3390/w15040646
Zhao M, Wang Q, Krua LSN, Yi R, Zou R, Li X, Huang P. Application Progress of New Adsorption Materials for Removing Fluorine from Water. Water. 2023; 15(4):646. https://doi.org/10.3390/w15040646
Chicago/Turabian StyleZhao, Ming−Ming, Qiang Wang, Luke Saye Nenwon Krua, Rong−Nan Yi, Run−Jun Zou, Xin−Yuan Li, and Peng Huang. 2023. "Application Progress of New Adsorption Materials for Removing Fluorine from Water" Water 15, no. 4: 646. https://doi.org/10.3390/w15040646





