Facile Green Synthesis of ZnO NPs and Plasmonic Ag-Supported ZnO Nanocomposite for Photocatalytic Degradation of Methylene Blue
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Preparation of ZnO and Plasmonic Ag-Supported ZnO Nanocomposite
2.3. Characterization
2.4. Photocatalytic Experiments
3. Results and Discussion
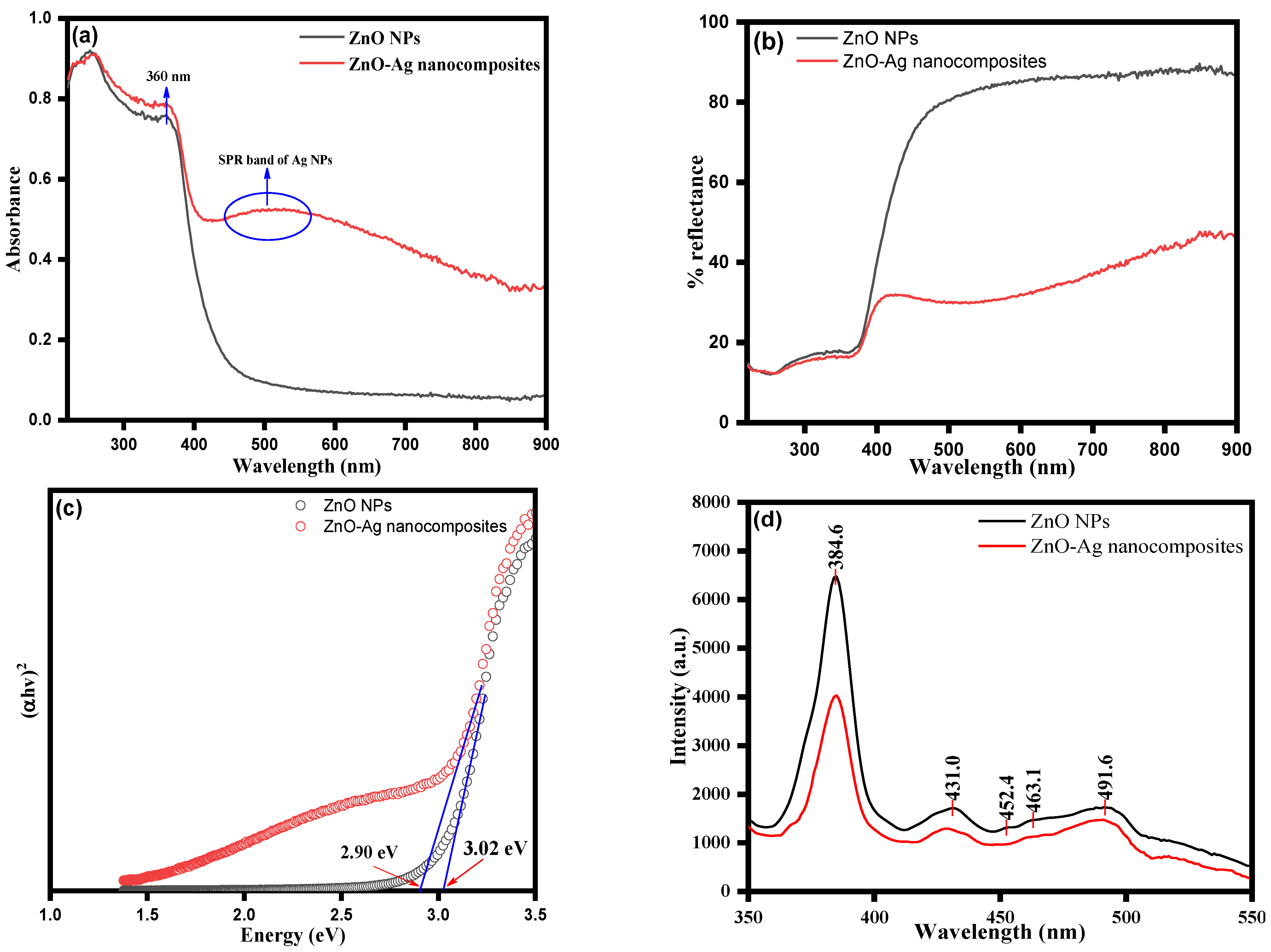
3.1. Spectroscopic Analysis
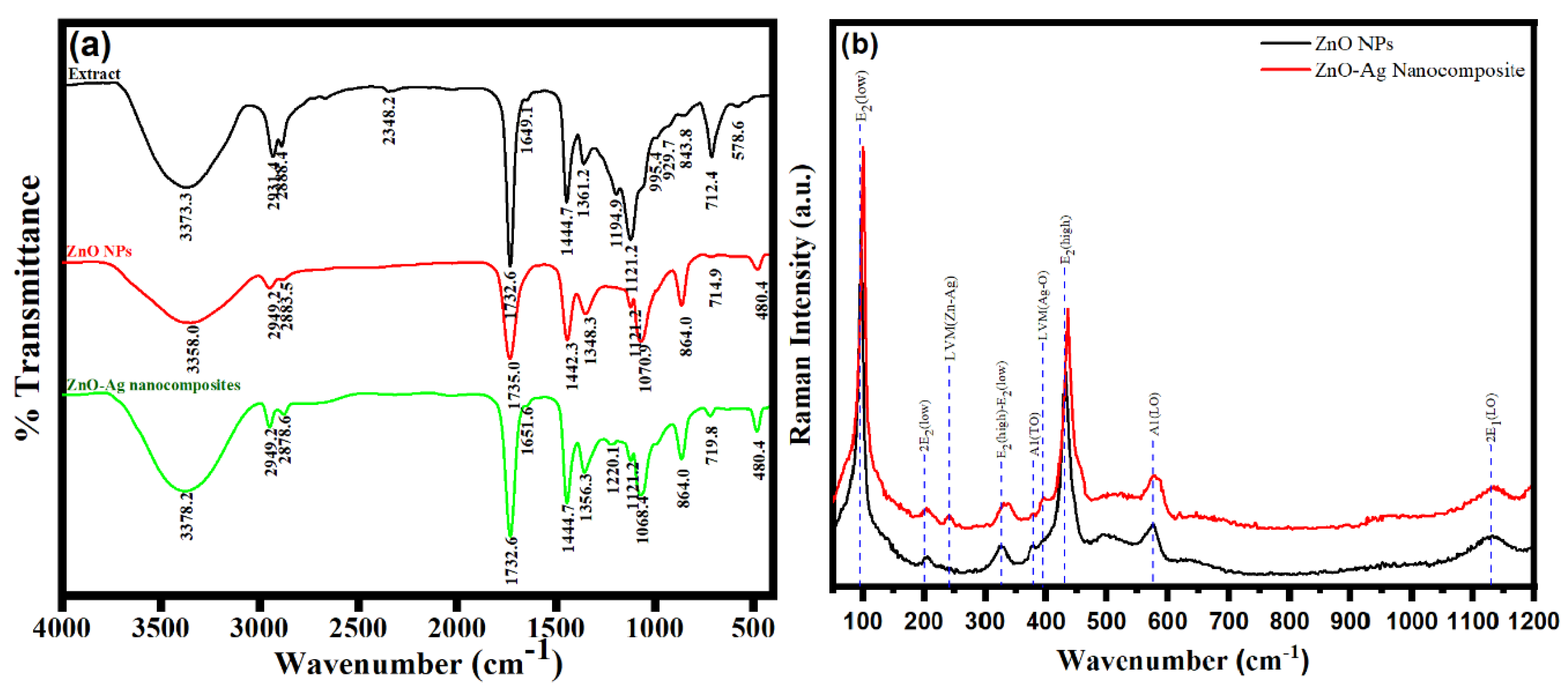
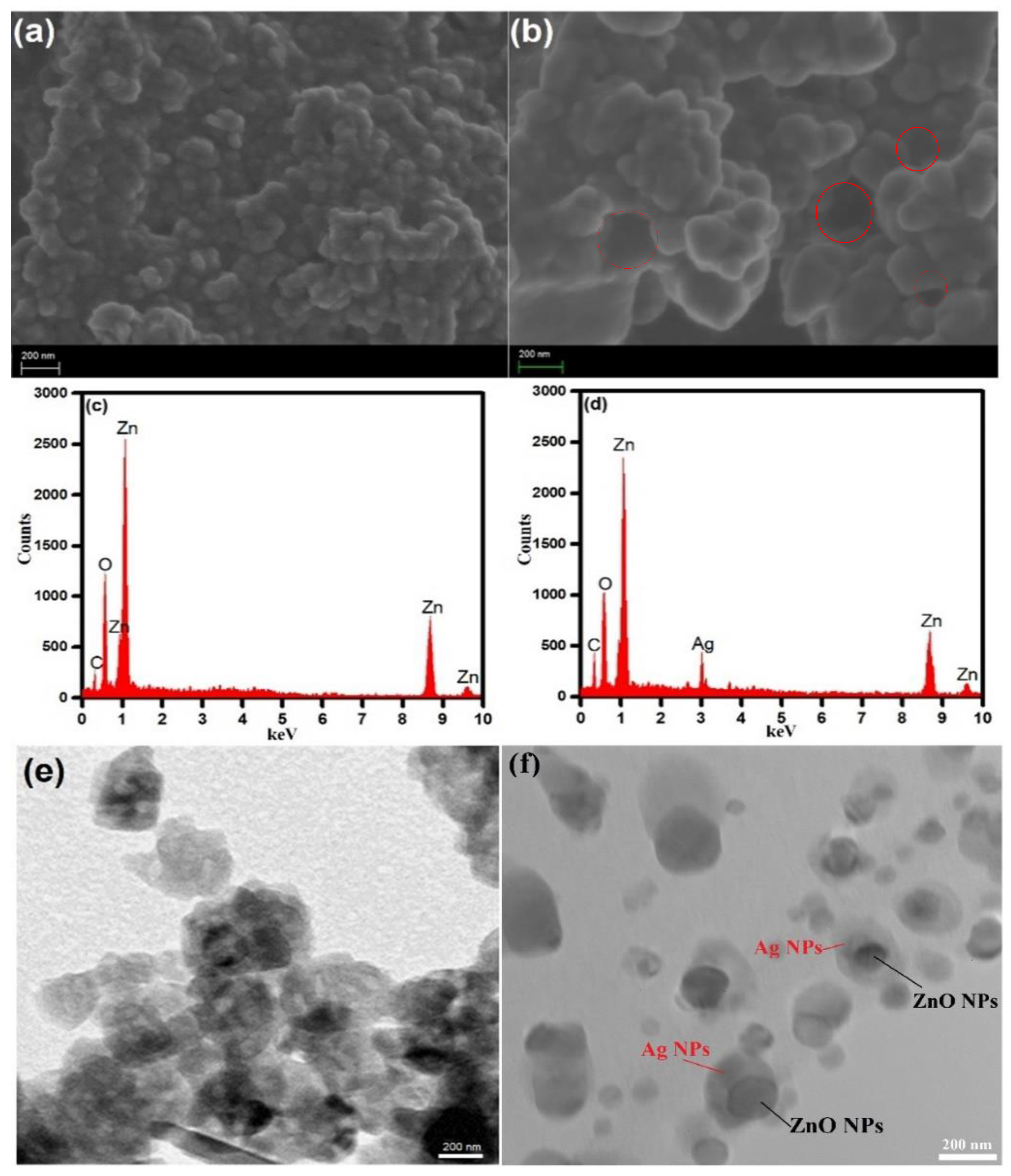
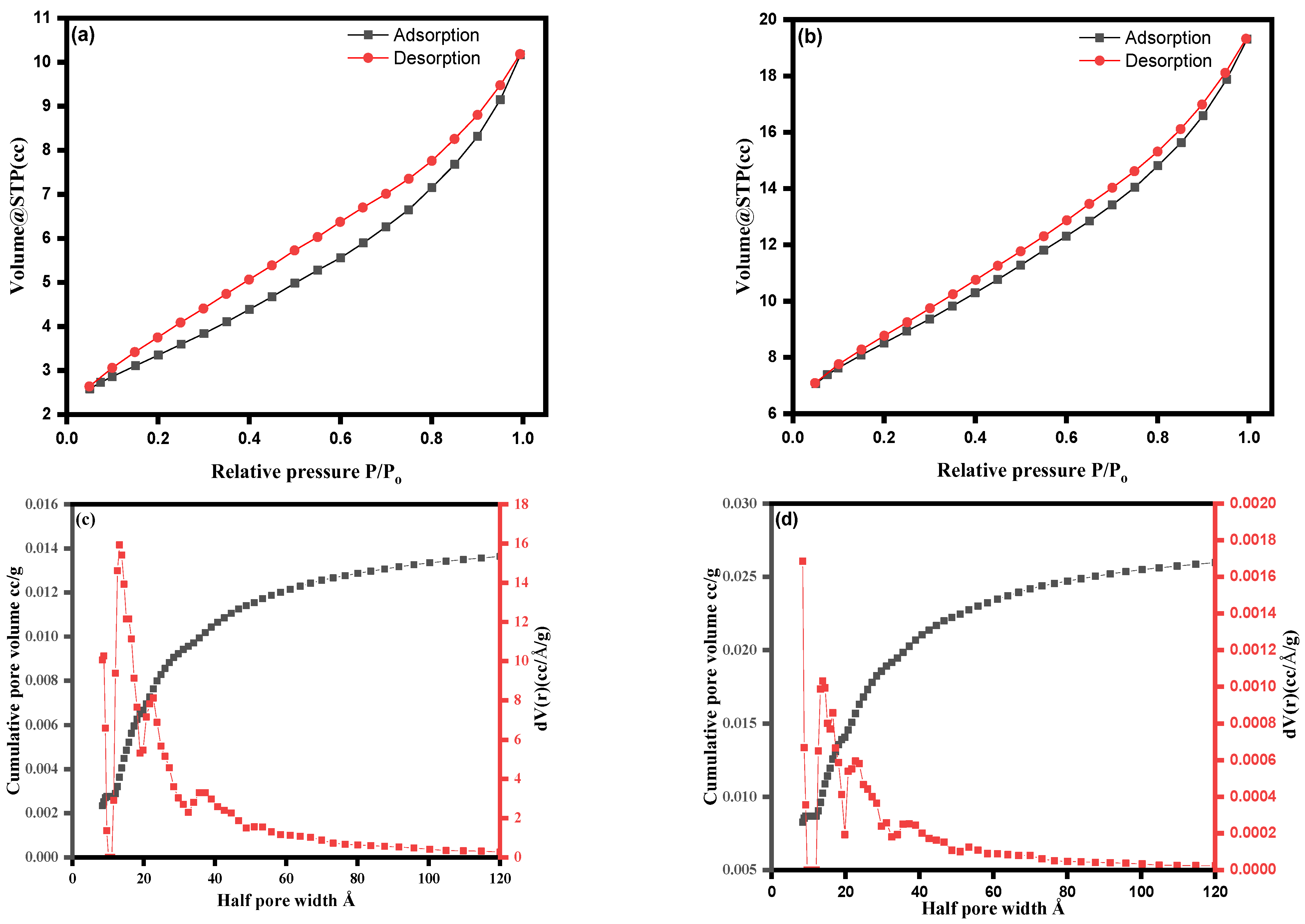
3.2. Characterization of ZnO NPs and ZnO–Ag Nanocomposite
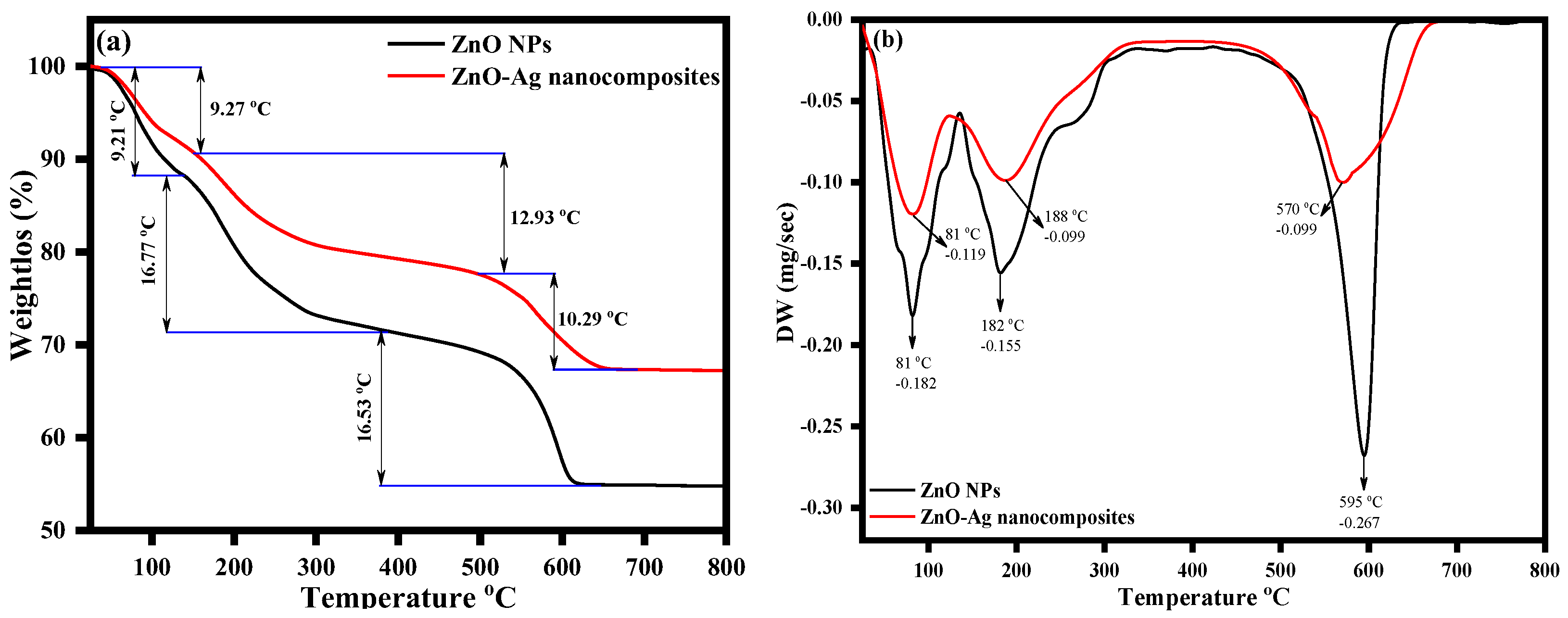
3.3. Thermogravimetric Analysis
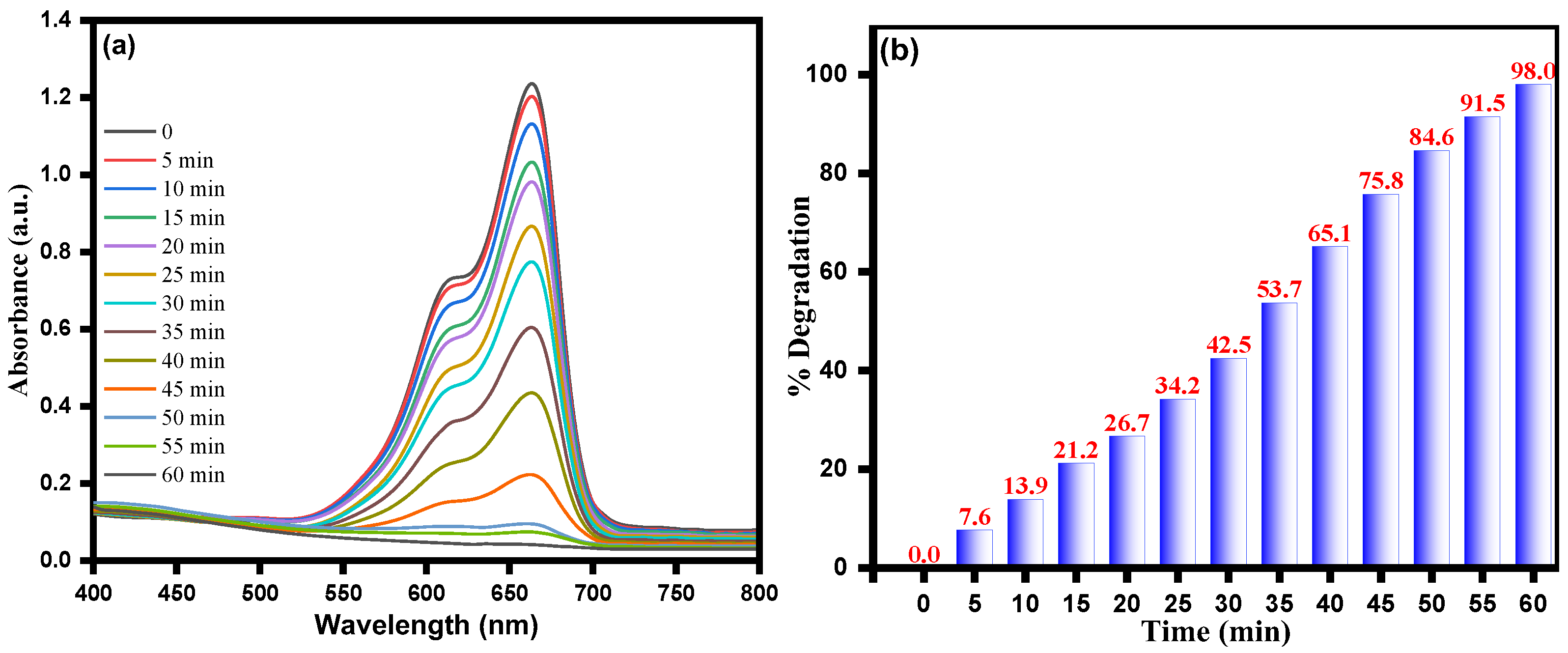
3.4. Photocatalytic Activity of ZnO–Ag Nanocomposite for MB Dye Degradation
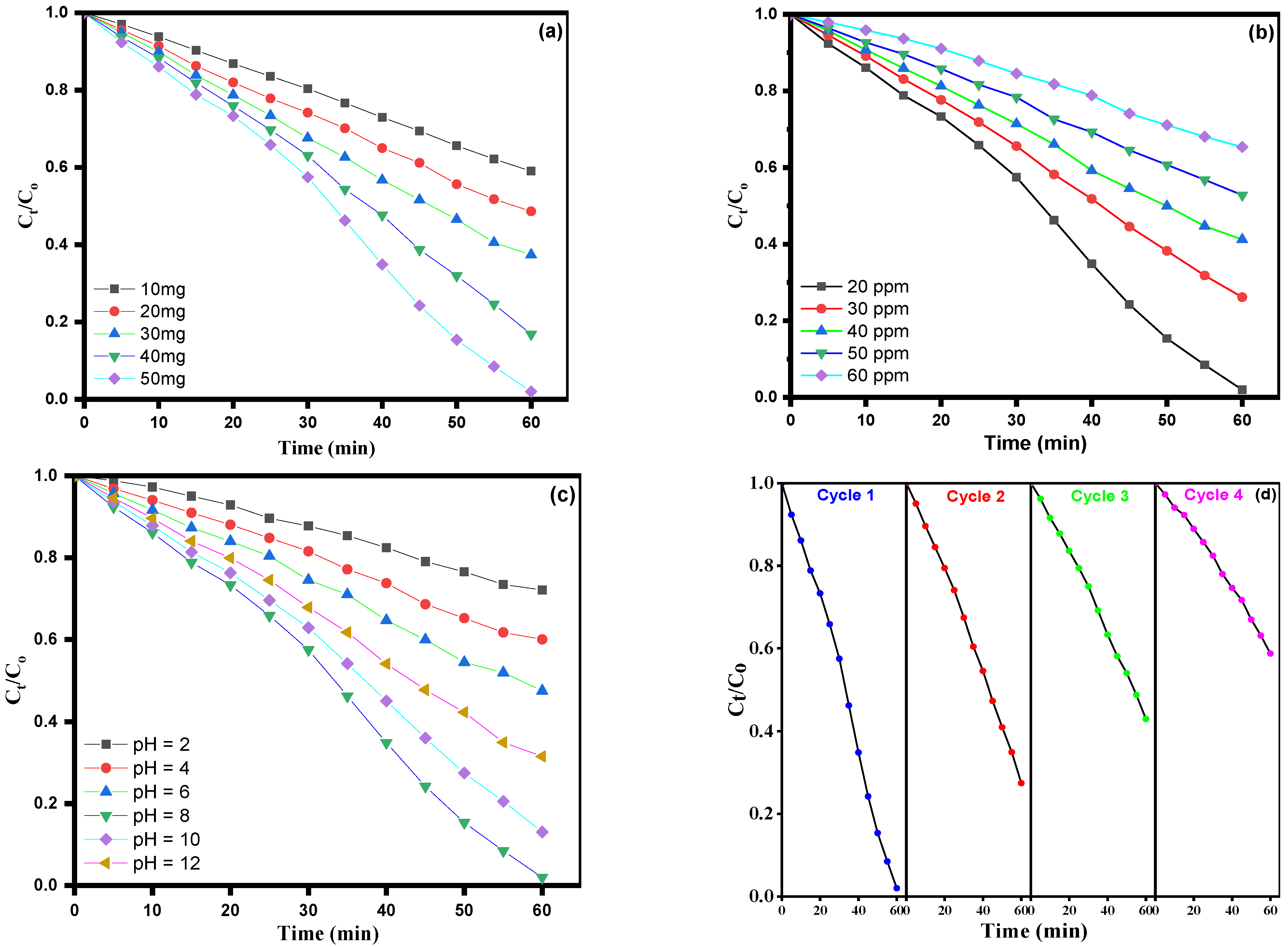
3.5. Effect of Catalyst Lodging
3.6. Effect of Dye Concentration
3.7. Effect of pH Dependency
3.8. The Recyclability Tests
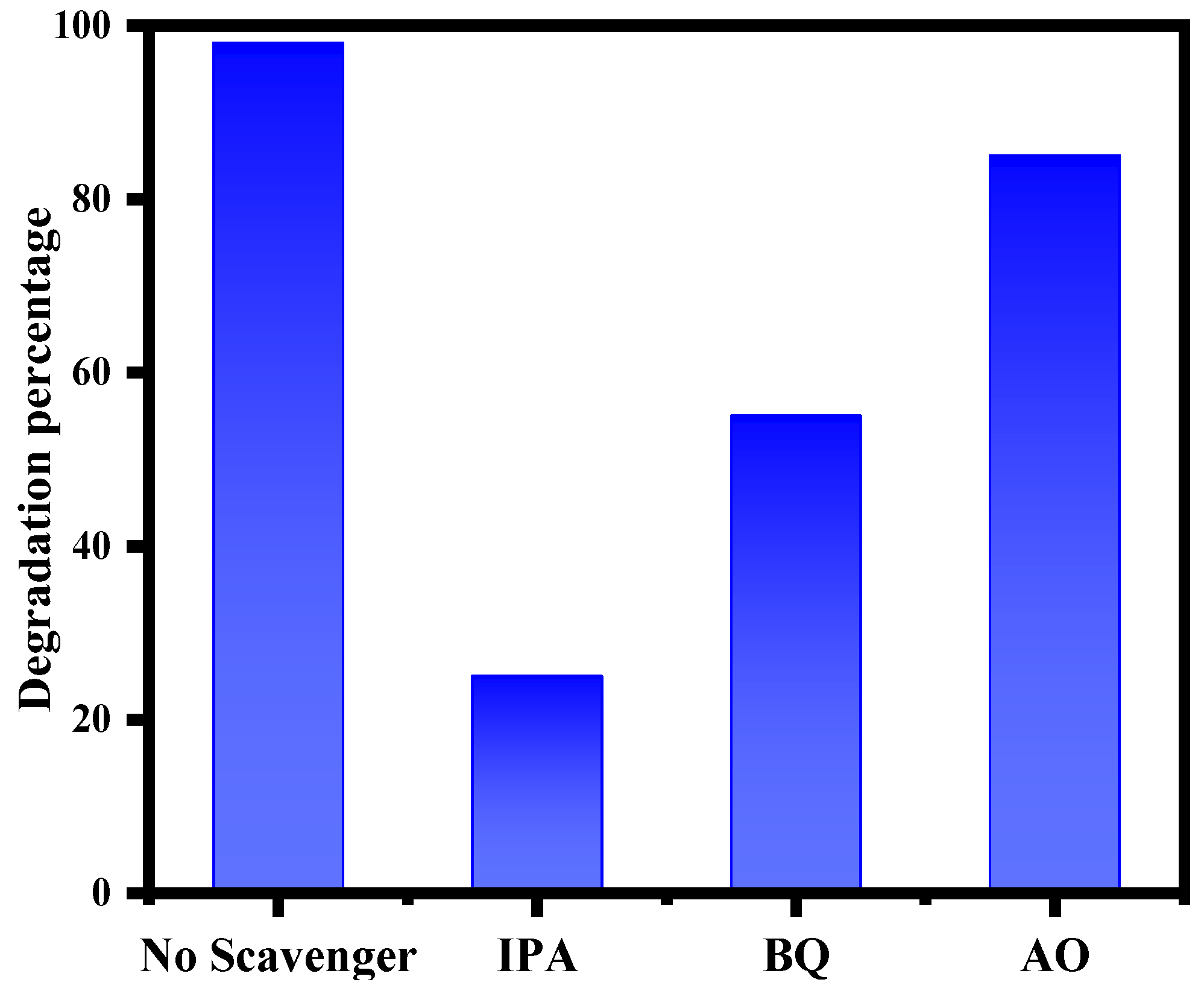
3.9. Possible Mechanism of Photodegradation of MB by ZnO–Ag Nanocomposite
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mohapatra, S.; Nguyen, T.A.; Nguyen-Tri, P. Noble metal-metal oxide hybrid nanoparticles: Fundamentals and applications; Elsevier: Amsterdam, The Netherlands, 2018. [Google Scholar]
- Zhou, M.; Li, C.; Fang, J. Noble-metal based random alloy and intermetallic nanocrystals: Syntheses and applications. Chem. Rev. 2020, 121, 736–795. [Google Scholar] [CrossRef] [PubMed]
- Karki, S.; Gohain, M.B.; Yadav, D.; Ingole, P.G. Nanocomposite and bio-nanocomposite polymeric materials/membranes development in energy and medical sector: A review. Int. J. Biol. Macromol. 2021, 193, 2121–2139. [Google Scholar] [CrossRef] [PubMed]
- Shabatina, T.; Vernaya, O.; Shumilkin, A.; Semenov, A.; Melnikov, M. Nanoparticles of Bioactive Metals/Metal Oxides and Their Nanocomposites with Antibacterial Drugs for Biomedical Applications. Materials 2022, 15, 3602. [Google Scholar] [CrossRef] [PubMed]
- Chue-Gonçalves, M.; Pereira, G.N.; Faccin-Galhardi, L.C.; Kobayashi, R.K.; Nakazato, G. Metal nanoparticles against viruses: Possibilities to fight SARS-CoV-2. Nanomaterials 2021, 11, 3118. [Google Scholar] [CrossRef]
- Saleh, R.; Djaja, N.F. UV light photocatalytic degradation of organic dyes with Fe-doped ZnO nanoparticles. Superlattices Microstruct. 2014, 74, 217–233. [Google Scholar] [CrossRef]
- Vijayakumari, G.; Selvakumar, N.; Jeyasubramanian, K.; Mala, R. Investigation on the electrical properties of polymer metal nanocomposites for physiological sensing applications. Phys. Procedia 2013, 49, 67–78. [Google Scholar] [CrossRef]
- Zare, Y.; Shabani, I. Polymer/metal nanocomposites for biomedical applications. Mater. Sci. Eng. C 2016, 60, 195–203. [Google Scholar] [CrossRef]
- Hussein, A.K. Applications of nanotechnology in renewable energies—A comprehensive overview and understanding. Renew. Sustain. Energy Rev. 2015, 42, 460–476. [Google Scholar] [CrossRef]
- Mamiyev, Z.; Balayeva, N.O. Metal Sulfide Photocatalysts for Hydrogen Generation: A Review of Recent Advances. Catalysts 2022, 12, 1316. [Google Scholar] [CrossRef]
- Bwatanglang, I.B.; Obulapuram, P.K.; Mohammad, F.; Albalawi, A.N.; Chavali, M.; Al-Lohedan, H.A.; Ibrahim, T. Metal oxide-involved photocatalytic technology in cosmetics and beauty products. In Metal Oxides for Optoelectronics and Optics-Based Medical Applications; Elsevier: Amsterdam, The Netherlands, 2022; pp. 301–337. [Google Scholar]
- Niska, K.; Zielinska, E.; Radomski, M.W.; Inkielewicz-Stepniak, I. Metal nanoparticles in dermatology and cosmetology: Interactions with human skin cells. Chem.-Biol. Interact. 2018, 295, 38–51. [Google Scholar] [CrossRef]
- Zare, E.N.; Jamaledin, R.; Naserzadeh, P.; Afjeh-Dana, E.; Ashtari, B.; Hosseinzadeh, M.; Vecchione, R.; Wu, A.; Tay, F.R.; Borzacchiello, A. Metal-based nanostructures/PLGA nanocomposites: Antimicrobial activity, cytotoxicity, and their biomedical applications. ACS Appl. Mater. Interfaces 2019, 12, 3279–3300. [Google Scholar] [CrossRef]
- Khan, M.E. State-of-the-art developments in carbon-based metal nanocomposites as a catalyst: Photocatalysis. Nanoscale Adv. 2021, 3, 1887–1900. [Google Scholar] [CrossRef] [PubMed]
- Shifrina, Z.B.; Matveeva, V.G.; Bronstein, L.M. Role of polymer structures in catalysis by transition metal and metal oxide nanoparticle composites. Chem. Rev. 2019, 120, 1350–1396. [Google Scholar] [CrossRef] [PubMed]
- Liang, S.; Zhang, M.; Biesold, G.M.; Choi, W.; He, Y.; Li, Z.; Shen, D.; Lin, Z. Recent advances in synthesis, properties, and applications of metal halide perovskite nanocrystals/polymer nanocomposites. Adv. Mater. 2021, 33, 2005888. [Google Scholar] [CrossRef] [PubMed]
- Zada, A.; Muhammad, P.; Ahmad, W.; Hussain, Z.; Ali, S.; Khan, M.; Khan, Q.; Maqbool, M. Surface plasmonic-assisted photocatalysis and optoelectronic devices with noble metal nanocrystals: Design, synthesis, and applications. Adv. Funct. Mater. 2020, 30, 1906744. [Google Scholar] [CrossRef]
- Jin, S.-E.; Jin, H.-E. Antimicrobial activity of zinc oxide nano/microparticles and their combinations against pathogenic microorganisms for biomedical applications: From physicochemical characteristics to pharmacological aspects. Nanomaterials 2021, 11, 263. [Google Scholar] [CrossRef]
- Zeng, T.; Zhang, X.; Guo, Y.; Niu, H.; Cai, Y. Enhanced catalytic application of Au@ polyphenol-metal nanocomposites synthesized by a facile and green method. J. Mater. Chem. A 2014, 2, 14807–14811. [Google Scholar] [CrossRef]
- Dobrucka, R. Selected applications of metal nanoparticles in medicine and pharmacology. LogForum 2019, 15, 449–457. [Google Scholar] [CrossRef]
- Hussain, M.Z.; Yang, Z.; Huang, Z.; Jia, Q.; Zhu, Y.; Xia, Y. Recent advances in metal–organic frameworks derived nanocomposites for photocatalytic applications in energy and environment. Adv. Sci. 2021, 8, 2100625. [Google Scholar] [CrossRef]
- Kura, A.U.; Hussein, M.Z.; Fakurazi, S.; Arulselvan, P. Layered double hydroxide nanocomposite for drug delivery systems; bio-distribution, toxicity and drug activity enhancement. Chem. Cent. J. 2014, 8, 1–8. [Google Scholar] [CrossRef]
- Naseem, T.; Durrani, T. The role of some important metal oxide nanoparticles for wastewater and antibacterial applications: A review. Environ. Chem. Ecotoxicol. 2021, 3, 59–75. [Google Scholar] [CrossRef]
- Smith, S.C.; Rodrigues, D.F. Carbon-based nanomaterials for removal of chemical and biological contaminants from water: A review of mechanisms and applications. Carbon 2015, 91, 122–143. [Google Scholar] [CrossRef]
- Dutta, V.; Singh, P.; Shandilya, P.; Sharma, S.; Raizada, P.; Saini, A.K.; Gupta, V.K.; Hosseini-Bandegharaei, A.; Agarwal, S.; Rahmani-Sani, A. Review on advances in photocatalytic water disinfection utilizing graphene and graphene derivatives-based nanocomposites. J. Environ. Chem. Eng. 2019, 7, 103132. [Google Scholar] [CrossRef]
- Kyomuhimbo, H.D.; Michira, I.N.; Mwaura, F.B.; Derese, S.; Feleni, U.; Iwuoha, E.I. Silver–zinc oxide nanocomposite antiseptic from the extract of bidens pilosa. SN Appl. Sci. 2019, 1, 1–17. [Google Scholar] [CrossRef]
- Liu, H.; Shao, G.; Zhao, J.; Zhang, Z.; Zhang, Y.; Liang, J.; Liu, X.; Jia, H.; Xu, B. Worm-like Ag/ZnO core–shell heterostructural composites: Fabrication, characterization, and photocatalysis. J. Phys. Chem. C 2012, 116, 16182–16190. [Google Scholar] [CrossRef]
- Choudhary, M.K.; Kataria, J.; Bhardwaj, V.K.; Sharma, S. Green biomimetic preparation of efficient Ag–ZnO heterojunctions with excellent photocatalytic performance under solar light irradiation: A novel biogenic-deposition-precipitation approach. Nanoscale Adv. 2019, 1, 1035–1044. [Google Scholar] [CrossRef] [PubMed]
- Aguirre, M.E.; Rodríguez, H.B.; San Román, E.; Feldhoff, A.; Grela, M.A. Ag@ ZnO core–shell nanoparticles formed by the timely reduction of Ag+ ions and zinc acetate hydrolysis in N, N-dimethylformamide: Mechanism of growth and photocatalytic properties. J. Phys. Chem. C 2011, 115, 24967–24974. [Google Scholar] [CrossRef]
- Van Hengel, I.; Putra, N.; Tierolf, M.; Minneboo, M.; Fluit, A.; Fratila-Apachitei, L.; Apachitei, I.; Zadpoor, A. Biofunctionalization of selective laser melted porous titanium using silver and zinc nanoparticles to prevent infections by antibiotic-resistant bacteria. Acta Biomater. 2020, 107, 325–337. [Google Scholar] [CrossRef]
- Kokate, M.; Garadkar, K.; Gole, A. Zinc-oxide-silica-silver nanocomposite: Unique one-pot synthesis and enhanced catalytic and anti-bacterial performance. J. Colloid Interface Sci. 2016, 483, 249–260. [Google Scholar] [CrossRef]
- Chan, Y.Y.; Pang, Y.L.; Lim, S.; Chong, W.C. Facile green synthesis of ZnO nanoparticles using natural-based materials: Properties, mechanism, surface modification and application. J. Environ. Chem. Eng. 2021, 9, 105417. [Google Scholar] [CrossRef]
- Elgohary, E.A.; Mohamed, Y.M.A.; El Nazer, H.A.; Baaloudj, O.; Alyami, M.S.; El Jery, A.; Assadi, A.A.; Amrane, A. A Review of the Use of Semiconductors as Catalysts in the Photocatalytic Inactivation of Microorganisms. Catalysts 2021, 11, 1498. [Google Scholar] [CrossRef]
- Gea, S.; Situmorang, S.A.; Pasaribu, N.; Piliang, A.F.; Attaurrazaq, B.; Sari, R.M.; Pasaribu, K.M.; Goutianos, S. Facile Synthesis of ZnO-Ag Nanocomposite Supported by Graphene Oxide with Stabilised Band-Gap and Wider Visible-Light Region for Photocatalyst Application. J. Mater. Res. Technol. 2022, 19, 2730–2741. [Google Scholar] [CrossRef]
- Imtiaz, F.; Rashid, J.; Xu, M. Semiconductor nanocomposites for visible light photocatalysis of water pollutants. In Concepts of Semiconductor Photocatalysis; IntechOpen: London, UK, 2019. [Google Scholar]
- Lwin, H.M.; Zhan, W.; Jia, F.; Song, S. Microwave-assisted hydrothermal synthesis of MoS2–Ag3PO4 nanocomposites as visible light photocatalyst for the degradation of tetracycline hydrochloride. Environ. Technol. 2022, 43, 149–162. [Google Scholar] [CrossRef] [PubMed]
- Sahu, P.; Das, D. Two-Step Visible Light Photocatalytic Dye Degradation Phenomena in Ag2O-Impregnated ZnO Nanorods via Growth of Metallic Ag and Formation of ZnO/Ag0/Ag2O Heterojunction Structures. Langmuir 2022, 38, 4503–4520. [Google Scholar] [CrossRef] [PubMed]
- Kamani, H.; Nasseri, S.; Khoobi, M.; Nabizadeh Nodehi, R.; Mahvi, A.H. Sonocatalytic degradation of humic acid by N-doped TiO2 nano-particle in aqueous solution. J. Environ. Health Sci. Eng. 2016, 14, 1–9. [Google Scholar] [CrossRef]
- Oliveira, A.G.; de Lara Andrade, J.; Montanha, M.C.; Lima, S.M.; da Cunha Andrade, L.H.; Hechenleitner, A.A.W.; Pineda, E.A.G.; de Oliveira, D.M.F. Decontamination and disinfection of wastewater by photocatalysis under UV/visible light using nano-catalysts based on Ca-doped ZnO. J. Environ. Manag. 2019, 240, 485–493. [Google Scholar] [CrossRef]
- Ra’ouf Ahmed, H.A. Preparation and Characterization of Copper-Doped and Silver-Doped Titanium Dioxide Nano-Catalysts for Photocatalytic Applications. Master’s Thesis, United Arab Emirates University, Al Ain, United Arab Emirates, 2015. [Google Scholar]
- Chekuri, R.D.; Tirukkovalluri, S.R. Synthesis of cobalt doped titania nano material assisted by gemini surfactant: Characterization and application in degradation of Acid Red under visible light irradiation. South Afr. J. Chem. Eng. 2017, 24, 183–195. [Google Scholar] [CrossRef]
- Fageria, P.; Gangopadhyay, S.; Pande, S. Synthesis of ZnO/Au and ZnO/Ag nanoparticles and their photocatalytic application using UV and visible light. Rsc Adv. 2014, 4, 24962–24972. [Google Scholar] [CrossRef]
- Rajbongshi, B.M.; Ramchiary, A.; Samdarshi, S. Influence of N-doping on photocatalytic activity of ZnO nanoparticles under visible light irradiation. Mater. Lett. 2014, 134, 111–114. [Google Scholar] [CrossRef]
- Zeng, H.; Cai, W.; Liu, P.; Xu, X.; Zhou, H.; Klingshirn, C.; Kalt, H. ZnO-based hollow nanoparticles by selective etching: Elimination and reconstruction of metal− semiconductor interface, improvement of blue emission and photocatalysis. ACS Nano 2008, 2, 1661–1670. [Google Scholar] [CrossRef]
- Lam, S.-M.; Quek, J.-A.; Sin, J.-C. Mechanistic investigation of visible light responsive Ag/ZnO micro/nanoflowers for enhanced photocatalytic performance and antibacterial activity. J. Photochem. Photobiol. A Chem. 2018, 353, 171–184. [Google Scholar] [CrossRef]
- Zhang, X.; Chen, Y.L.; Liu, R.-S.; Tsai, D.P. Plasmonic photocatalysis. Rep. Prog. Phys. 2013, 76, 046401. [Google Scholar] [CrossRef] [PubMed]
- Awazu, K.; Fujimaki, M.; Rockstuhl, C.; Tominaga, J.; Murakami, H.; Ohki, Y.; Yoshida, N.; Watanabe, T. A plasmonic photocatalyst consisting of silver nanoparticles embedded in titanium dioxide. J. Am. Chem. Soc. 2008, 130, 1676–1680. [Google Scholar] [CrossRef] [PubMed]
- Dao, T.D.; Han, G.; Arai, N.; Nabatame, T.; Wada, Y.; Hoang, C.V.; Aono, M.; Nagao, T. Plasmon-mediated photocatalytic activity of wet-chemically prepared ZnO nanowire arrays. Phys. Chem. Chem. Phys. 2015, 17, 7395–7403. [Google Scholar] [CrossRef] [PubMed]
- Barabadi, H.; Mojab, F.; Vahidi, H.; Marashi, B.; Talank, N.; Hosseini, O.; Saravanan, M. Green synthesis, characterization, antibacterial and biofilm inhibitory activity of silver nanoparticles compared to commercial silver nanoparticles. Inorg. Chem. Commun. 2021, 129, 108647. [Google Scholar] [CrossRef]
- Majhi, R.K.; Mohanty, S.; Khan, M.I.; Mishra, A.; Brauner, A. Ag@ ZnO nanoparticles induce antimicrobial peptides and promote migration and antibacterial activity of keratinocytes. ACS Infect. Dis. 2021, 7, 2068–2072. [Google Scholar] [CrossRef]
- Schiffmann, A.; Jauk, T.; Knez, D.; Fitzek, H.; Hofer, F.; Lackner, F.; Ernst, W.E. Helium droplet assisted synthesis of plasmonic Ag@ ZnO core@ shell nanoparticles. Nano Res. 2020, 13, 2979–2986. [Google Scholar] [CrossRef]
- Rafaie, H.; Nor, R.; Azmina, M.; Ramli, N.; Mohamed, R. Decoration of ZnO microstructures with Ag nanoparticles enhanced the catalytic photodegradation of methylene blue dye. J. Environ. Chem. Eng. 2017, 5, 3963–3972. [Google Scholar] [CrossRef]
- Singh, T.A.; Sharma, A.; Tejwan, N.; Ghosh, N.; Das, J.; Sil, P.C. A state of the art review on the synthesis, antibacterial, antioxidant, antidiabetic and tissue regeneration activities of zinc oxide nanoparticles. Adv. Colloid Interface Sci. 2021, 295, 102495. [Google Scholar] [CrossRef]
- Zare, M.; Namratha, K.; Alghamdi, S.; Mohammad, Y.H.E.; Hezam, A.; Zare, M.; Drmosh, Q.A.; Byrappa, K.; Chandrashekar, B.N.; Ramakrishna, S. Novel green biomimetic approach for synthesis of ZnO-Ag nanocomposite; antimicrobial activity against food-borne pathogen, biocompatibility and solar photocatalysis. Sci. Rep. 2019, 9, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Dang, G.H.; Le, T.T.; Ta, A.K.; Ho, T.N.; Pham, T.V.; Doan, T.V.; Luong, T.H. Removal of Congo red and malachite green from aqueous solution using heterogeneous Ag/ZnCo-ZIF catalyst in the presence of hydrogen peroxide. Green Process. Synth. 2020, 9, 567–577. [Google Scholar] [CrossRef]
- Liu, Y.; Wei, S.; Gao, W. Ag/ZnO heterostructures and their photocatalytic activity under visible light: Effect of reducing medium. J. Hazard. Mater. 2015, 287, 59–68. [Google Scholar] [CrossRef] [PubMed]
- Mohammadzadeh, S.; Olya, M.; Arabi, A.; Shariati, A.; Nikou, M.K. Synthesis, characterization and application of ZnO-Ag as a nanophotocatalyst for organic compounds degradation, mechanism and economic study. J. Environ. Sci. 2015, 35, 194–207. [Google Scholar] [CrossRef] [PubMed]
- Ishak, N.M.; Kamarudin, S.; Timmiati, S. Green synthesis of metal and metal oxide nanoparticles via plant extracts: An overview. Mater. Res. Express 2019, 6, 112004. [Google Scholar] [CrossRef]
- Feng, T.; Feng, G.S.; Yan, L.; Pan, J.H. One-dimensional nanostructured TiO2 for photocatalytic degradation of organic pollutants in wastewater. Int. J. Photoenergy 2014, 2014. [Google Scholar] [CrossRef]
- Zare, E.N.; Iftekhar, S.; Park, Y.; Joseph, J.; Srivastava, V.; Khan, M.A.; Makvandi, P.; Sillanpaa, M.; Varma, R.S. An overview on non-spherical semiconductors for heterogeneous photocatalytic degradation of organic water contaminants. Chemosphere 2021, 280, 130907. [Google Scholar] [CrossRef]
- Ambat, I.; Srivastava, V.; Iftekhar, S.; Haapaniemi, E.; Sillanpää, M. Dual application of divalent ion-anchored catalyst: Biodiesel synthesis and photocatalytic degradation of carbamazepine. Catal. Green Chem. Eng. 2019, 2, 25–42. [Google Scholar] [CrossRef]
- Hammouda, S.B.; Salazar, C.; Zhao, F.; Ramasamy, D.L.; Laklova, E.; Iftekhar, S.; Babu, I.; Sillanpää, M. Efficient heterogeneous electro-Fenton incineration of a contaminant of emergent concern-cotinine-in aqueous medium using the magnetic double perovskite oxide Sr2FeCuO6 as a highly stable catalayst: Degradation kinetics and oxidation products. Appl. Catal. B Environ. 2019, 240, 201–214. [Google Scholar] [CrossRef]
- Hossain, S.S.; Tarek, M.; Munusamy, T.D.; Karim, K.M.R.; Roopan, S.M.; Sarkar, S.M.; Cheng, C.K.; Khan, M.M.R. Facile synthesis of CuO/CdS heterostructure photocatalyst for the effective degradation of dye under visible light. Environ. Res. 2020, 188, 109803. [Google Scholar] [CrossRef]
- Aadil, M.; Rahman, A.; Zulfiqar, S.; Alsafari, I.A.; Shahid, M.; Shakir, I.; Agboola, P.O.; Haider, S.; Warsi, M.F. Facile synthesis of binary metal substituted copper oxide as a solar light driven photocatalyst and antibacterial substitute. Adv. Powder Technol. 2021, 32, 940–950. [Google Scholar] [CrossRef]
- Mukhtar, F.; Munawar, T.; Nadeem, M.S.; Batool, S.; Hasan, M.; Riaz, M.; Iqbal, F. Highly efficient tri-phase TiO2–Y2O3–V2O5 nanocomposite: Structural, optical, photocatalyst, and antibacterial studies. J. Nanostructure Chem. 2022, 12, 547–564. [Google Scholar] [CrossRef]
- Begum, G.; Manna, J.; Rana, R.K. Controlled orientation in a bio-inspired assembly of Ag/AgCl/ZnO nanostructures enables enhancement in visible-light-induced photocatalytic performance. Chem.–A Eur. J. 2012, 18, 6847–6853. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Janbandhu, S.Y.; Sukhadeve, G.K.; Gedam, R.S. Visible-light assisted surface plasmon resonance triggered Ag/ZnO nanocomposites: Synthesis and performance towards degradation of indigo carmine dye. 2022. Environ. Sci. Pollut. Res. 2022. [Google Scholar] [CrossRef]
- Lu, W.; Huang, Q.; Zhang, Y.; Yao, K.; Wang, J. Plasmon-enhanced photodegradation of ionic liquids with Ag nanocubes/ZnO microsphere composites. Ind. Eng. Chem. Res. 2018, 57, 15597–15605. [Google Scholar] [CrossRef]
- Thatikayala, D.; Banothu, V.; Kim, J.; Shin, D.S.; Vijayalakshmi, S.; Park, J. Enhanced photocatalytic and antibacterial activity of ZnO/Ag nanostructure synthesized by Tamarindus indica pulp extract. J. Mater. Sci. Mater. Electron. 2020, 31, 5324–5335. [Google Scholar] [CrossRef]
- Trang, T.N.Q.; Phan, T.B.; Nam, N.D.; Thu, V.T.H. In situ charge transfer at the Ag@ ZnO photoelectrochemical interface toward the high photocatalytic performance of H2 evolution and RhB degradation. ACS Appl. Mater. Interfaces 2020, 12, 12195–12206. [Google Scholar] [CrossRef]
- Zhou, X.; Tang, L.; Xu, Y.; Zhou, G.; Wang, Z. Towards a better understanding of medicinal uses of Carthamus tinctorius L. in traditional Chinese medicine: A phytochemical and pharmacological review. J. Ethnopharmacol. 2014, 151, 27–43. [Google Scholar] [CrossRef]
- Mirzajani, F.; Bernard, F.; Zeinali, S.M.; Goodarzi, R. Identification of hydroxy-safflor yellow A, safflor yellow B, and precarthaminin safflower using LC/ESI–MSMS. J. Food Meas. Charact. 2015, 9, 332–336. [Google Scholar] [CrossRef]
- Clementi, C.; Basconi, G.; Pellegrino, R.; Romani, A. Carthamus tinctorius L.: A photophysical study of the main coloured species for artwork diagnostic purposes. Dye. Pigment. 2014, 103, 127–137. [Google Scholar] [CrossRef]
- Kazuma, K.; Takahashi, T.; Sato, K.; Takeuchi, H.; Matsumoto, T.; Okuno, T. Quinochalcones and flavonoids from fresh florets in different cultivars of Carthamus tinctorius L. Biosci. Biotechnol. Biochem. 2000, 64, 1588–1599. [Google Scholar] [CrossRef]
- Navas, D.; Ibañez, A.; González, I.; Palma, J.L.; Dreyse, P. Controlled dispersion of ZnO nanoparticles produced by basic precipitation in solvothermal processes. Heliyon 2020, 6, e05821. [Google Scholar] [CrossRef]
- Kyomuhimbo, H.D.; Michira, I.N.; Iwuoha, E.I.; Feleni, U. Electroconductive Polyaniline–Ag-ZnO Green Nanocomposite Material. Processes 2022, 10, 457. [Google Scholar] [CrossRef]
- Nagaraju, G.; Prashanth, S.; Shastri, M.; Yathish, K.; Anupama, C.; Rangappa, D. Electrochemical heavy metal detection, photocatalytic, photoluminescence, biodiesel production and antibacterial activities of Ag–ZnO nanomaterial. Mater. Res. Bull. 2017, 94, 54–63. [Google Scholar] [CrossRef]
- Saravanadevi, K.; Kavitha, M.; Karpagavinayagam, P.; Saminathan, K.; Vedhi, C. Biosynthesis of ZnO and Ag doped ZnO nanoparticles from Vitis vinifera leaf for antibacterial, photocatalytic application. Mater. Today: Proc. 2020, 48, 352–356. [Google Scholar] [CrossRef]
- Kareem, M.; Bello, I.; Shittu, H.; Sivaprakash, P.; Adedokun, O.; Arumugam, S. Synthesis, characterization, and photocatalytic application of silver doped zinc oxide nanoparticles. Clean. Mater. 2022, 3, 100041. [Google Scholar] [CrossRef]
- Guidelli, E.; Baffa, O.; Clarke, D. Enhanced UV emission from silver/ZnO and gold/ZnO core-shell nanoparticles: Photoluminescence, radioluminescence, and optically stimulated luminescence. Sci. Rep. 2015, 5, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Dinesh, V.; Biji, P.; Ashok, A.; Dhara, S.; Kamruddin, M.; Tyagi, A.; Raj, B. Photocatalytic degradation of industrial textile dyes using hybrid ZnO@ Ag core–shell nanorods. RSC Adv. 2014, 4, 58930–58940. [Google Scholar] [CrossRef]
- Alharthi, F.A.; Alghamdi, A.A.; Al-Zaqri, N.; Alanazi, H.S.; Alsyahi, A.A.; Marghany, A.E.; Ahmad, N. Facile one-pot green synthesis of Ag–ZnO Nanocomposites using potato peeland their Ag concentration dependent photocatalytic properties. Sci. Rep. 2020, 10, 1–14. [Google Scholar] [CrossRef]
- Mukhtar, F.; Munawar, T.; Nadeem, M.S.; ur Rehman, M.N.; Khan, S.A.; Koc, M.; Batool, S.; Hasan, M.; Iqbal, F. Dual Z-scheme core-shell PANI-CeO2-Fe2O3-NiO heterostructured nanocomposite for dyes remediation under sunlight and bacterial disinfection. Environ. Res. 2022, 215, 114140. [Google Scholar] [CrossRef]
- Chanu, L.A.; Singh, W.J.; Singh, K.J.; Devi, K.N. Effect of operational parameters on the photocatalytic degradation of Methylene blue dye solution using manganese doped ZnO nanoparticles. Results Phys. 2019, 12, 1230–1237. [Google Scholar] [CrossRef]
- Kaur, J.; Singhal, S. Facile synthesis of ZnO and transition metal doped ZnO nanoparticles for the photocatalytic degradation of Methyl Orange. Ceram. Int. 2014, 40, 7417–7424. [Google Scholar] [CrossRef]
- Nenavathu, B.P.; Rao, A.K.; Goyal, A.; Kapoor, A.; Dutta, R.K. Synthesis, characterization and enhanced photocatalytic degradation efficiency of Se doped ZnO nanoparticles using trypan blue as a model dye. Appl. Catal. A Gen. 2013, 459, 106–113. [Google Scholar] [CrossRef]
- Wani, A.A.; Khan, A.M.; Manea, Y.K.; Salem, M.A. Enhanced photocatalytic degradation of organic dyes from aqueous environment using neodymium-doped mesoporous layered double hydroxide. J. Rare Earths 2022, 40, 1554–1563. [Google Scholar] [CrossRef]
- Balayeva, N.O.; Fleisch, M.; Bahnemann, D.W. Surface-grafted WO3/TiO2 photocatalysts: Enhanced visible-light activity towards indoor air purification. Catal. Today 2018, 313, 63–71. [Google Scholar] [CrossRef]
- Khanal, V.; Balayeva, N.O.; Günnemann, C.; Mamiyev, Z.; Dillert, R.; Bahnemann, D.W.; Subramanian, V.R. Photocatalytic NOx removal using tantalum oxide nanoparticles: A benign pathway. Appl. Catal. B Environ. 2021, 291, 119974. [Google Scholar] [CrossRef]
- Dutta, S.; Banerjee, P.; Das, P.; Mukhopadhyay, A. Phytogenic synthesis of nanoparticles and their application in photo catalysis of dye rich effluents. Photocatalytic Degrad. Dye. 2021, 647–694. [Google Scholar]
- Height, M.J.; Pratsinis, S.E.; Mekasuwandumrong, O.; Praserthdam, P. Ag-ZnO catalysts for UV-photodegradation of methylene blue. Appl. Catal. B Environ. 2006, 63, 305–312. [Google Scholar] [CrossRef]
- Ibhadon, A.O.; Fitzpatrick, P. Heterogeneous photocatalysis: Recent advances and applications. Catalysts 2013, 3, 189–218. [Google Scholar] [CrossRef]
- Zheng, Y.; Chen, C.; Zhan, Y.; Lin, X.; Zheng, Q.; Wei, K.; Zhu, J. Photocatalytic activity of Ag/ZnO heterostructure nanocatalyst: Correlation between structure and property. J. Phys. Chem. C 2008, 112, 10773–10777. [Google Scholar] [CrossRef]
- Jaafar, N.; Najman, A.; Marfur, A.; Jusoh, N. Strategies for the formation of oxygen vacancies in zinc oxide nanoparticles used for photocatalytic degradation of phenol under visible light irradiation. J. Photochem. Photobiol. A Chem. 2020, 388, 112202. [Google Scholar] [CrossRef]
- Abdullah, F.; Bakar, N.A.; Bakar, M.A. Low temperature biosynthesis of crystalline zinc oxide nanoparticles from Musa acuminata peel extract for visible-light degradation of methylene blue. Optik 2020, 206, 164279. [Google Scholar] [CrossRef]
- Mohammed, M.K. Carbon nanotubes loaded ZnO/Ag ternary nanohybrid with improved visible light photocatalytic activity and stability. Optik 2020, 217, 164867. [Google Scholar] [CrossRef]
- Wang, C.; Wang, D.; Pang, C.; Wang, X.; Yin, J. The fabrication and UV photosensitive characteristics of Al/ZnO/Ag Schottky barrier diode. In Proceedings of the 2012 International Conference on Measurement, Information and Control, Harbin, China, 18–20 May 2012; pp. 659–662. [Google Scholar]
- Kalpana, K.; Ravichandran, K.; Sindhuja, E.; Seelan, K.S.; Jothivenkatachalam, K.; Sriram, S.; Dhanraj, C. Enhancement of photocatalytic dye degradation efficiency of ZnO/Ag film deposited on flexible stainless steel meshes through g-C3N4 addition. Mater. Res. Express 2018, 6, 016422. [Google Scholar] [CrossRef]
- Ullattil, S.G.; Jabeen Fatima, M.; Abdel-Wahab, A. Defect minimized Ag-ZnO microneedles for photocatalysis. Environ. Sci. Pollut. Res. 2020, 27, 37036–37043. [Google Scholar] [CrossRef] [PubMed]
- Yan, F.; Wang, Y.; Zhang, J.; Lin, Z.; Zheng, J.; Huang, F. Schottky or ohmic metal–semiconductor contact: Influence on photocatalytic efficiency of Ag/ZnO and Pt/ZnO model systems. ChemSusChem 2014, 7, 101–104. [Google Scholar] [CrossRef]
- Xie, W.; Li, Y.; Sun, W.; Huang, J.; Xie, H.; Zhao, X. Surface modification of ZnO with Ag improves its photocatalytic efficiency and photostability. J. Photochem. Photobiol. A Chem. 2010, 216, 149–155. [Google Scholar] [CrossRef]
- Akman, B.; Aras, O. Usability, durability and regeneration of Ag/ZnO coated microreactor for photocatalytic degradation of methylene blue. J. Mol. Struct. 2022, 1251, 132003. [Google Scholar] [CrossRef]
- Stanley, R.; Jebasingh, J.A.; Stanley, P.K.; Ponmani, P.; Shekinah, M.; Vasanthi, J. Excellent Photocatalytic degradation of Methylene Blue, Rhodamine B and Methyl Orange dyes by Ag-ZnO nanocomposite under natural sunlight irradiation. Optik 2021, 231, 166518. [Google Scholar]
- Khan, I.; Saeed, K.; Zekker, I.; Zhang, B.; Hendi, A.H.; Ahmad, A.; Ahmad, S.; Zada, N.; Ahmad, H.; Shah, L.A. Review on methylene blue: Its properties, uses, toxicity and photodegradation. Water 2022, 14, 242. [Google Scholar] [CrossRef]
- Tayade, R.J.; Natarajan, T.S.; Bajaj, H.C. Photocatalytic degradation of methylene blue dye using ultraviolet light emitting diodes. Ind. Eng. Chem. Res. 2009, 48, 10262–10267. [Google Scholar] [CrossRef]
- Ravichandran, K.; Uma, R.; Sriram, S.; Balamurgan, D. Fabrication of ZnO: Ag/GO composite thin films for enhanced photocatalytic activity. Ceram. Int. 2017, 43, 10041–10051. [Google Scholar] [CrossRef]
- Siddiqui, V.U.; Ansari, A.; Ansari, M.T.; Akram, M.K.; Siddiqi, W.A. Fabrication of a zinc oxide/alginate (ZnO/Alg) bionanocomposite for enhanced dye degradation and its optimization study. RSC Adv. 2022, 12, 7210–7228. [Google Scholar] [CrossRef] [PubMed]
- Ichipi, E.O.; Tichapondwa, S.M.; Chirwa, E.M. Plasmonic effect and bandgap tailoring of Ag/Ag2S doped on ZnO nanocomposites for enhanced visible-light photocatalysis. Adv. Powder Technol. 2022, 33, 103596. [Google Scholar] [CrossRef]
- Ma, L.; Ai, X.; Jiang, W.; Liu, P.; Chen, Y.; Lu, K.; Song, X.; Wu, X. Zn/Ce metal-organic framework-derived ZnO@ CeO2 nano-heterojunction for enhanced photocatalytic activity. Colloid Interface Sci. Commun. 2022, 49, 100636. [Google Scholar] [CrossRef]
- Loka, C.; Lee, K.-S. Enhanced Visible-Light-Driven Photocatalysis of Ag/Ag2O/ZnO Nanocomposite Heterostructures. Nanomaterials 2022, 12, 2528. [Google Scholar] [CrossRef]
- Tran, M.L.; Nguyen, C.H.; Fu, C.-C.; Juang, R.-S. Hybridizing Ag-Doped ZnO nanoparticles with graphite as potential photocatalysts for enhanced removal of metronidazole antibiotic from water. J. Environ. Manag. 2019, 252, 109611. [Google Scholar] [CrossRef]
- Wang, Z.; Ye, X.; Chen, L.; Huang, P.; Wang, Q.; Ma, L.; Hua, N.; Liu, X.; Xiao, X.; Chen, S. Silver nanoparticles decorated grassy ZnO coating for photocatalytic activity enhancement. Mater. Sci. Semicond. Process. 2021, 121, 105354. [Google Scholar] [CrossRef]
- Peng, J.; Lu, T.; Ming, H.; Ding, Z.; Yu, Z.; Zhang, J.; Hou, Y. Enhanced photocatalytic ozonation of phenol by Ag/ZnO nanocomposites. Catalysts 2019, 9, 1006. [Google Scholar] [CrossRef]
- Zhu, X.; Wang, J.; Yang, D.; Liu, J.; He, L.; Tang, M.; Feng, W.; Wu, X. Fabrication, characterization and high photocatalytic activity of Ag–ZnO heterojunctions under UV-visible light. RSC Adv. 2021, 11, 27257–27266. [Google Scholar] [CrossRef]
- Alshamsi, H.A.H.; Hussein, B.S. Hydrothermal preparation of silver doping zinc oxide nanoparticles: Study the characterization and photocatalytic activity. Orient. J. Chem. 2018, 34, 1898. [Google Scholar] [CrossRef]
- Alshehri, A.A.; Malik, M.A. Biogenic fabrication of ZnO nanoparticles using Trigonella foenum-graecum (Fenugreek) for proficient photocatalytic degradation of methylene blue under UV irradiation. J. Mater. Sci. Mater. Electron. 2019, 30, 16156–16173. [Google Scholar] [CrossRef]
- Sharwani, A.A.; Narayanan, K.B.; Khan, M.E.; Han, S.S. Photocatalytic degradation activity of goji berry extract synthesized silver-loaded mesoporous zinc oxide (Ag@ ZnO) nanocomposites under simulated solar light irradiation. Sci. Rep. 2022, 12, 1–18. [Google Scholar]
- Balayeva, N.O.; Mamiyev, Z. Integrated processes involving adsorption, photolysis, and photocatalysis. In Hybrid and Combined Processes for Air Pollution Control; Elsevier: Amsterdam, The Netherlands, 2022; pp. 117–153. [Google Scholar]
- Anisuzzaman, S.; Joseph, C.G.; Pang, C.K.; Affandi, N.A.; Maruja, S.N.; Vijayan, V. Current Trends in the Utilization of Photolysis and Photocatalysis Treatment Processes for the Remediation of Dye Wastewater: A Short Review. ChemEngineering 2022, 6, 58. [Google Scholar] [CrossRef]


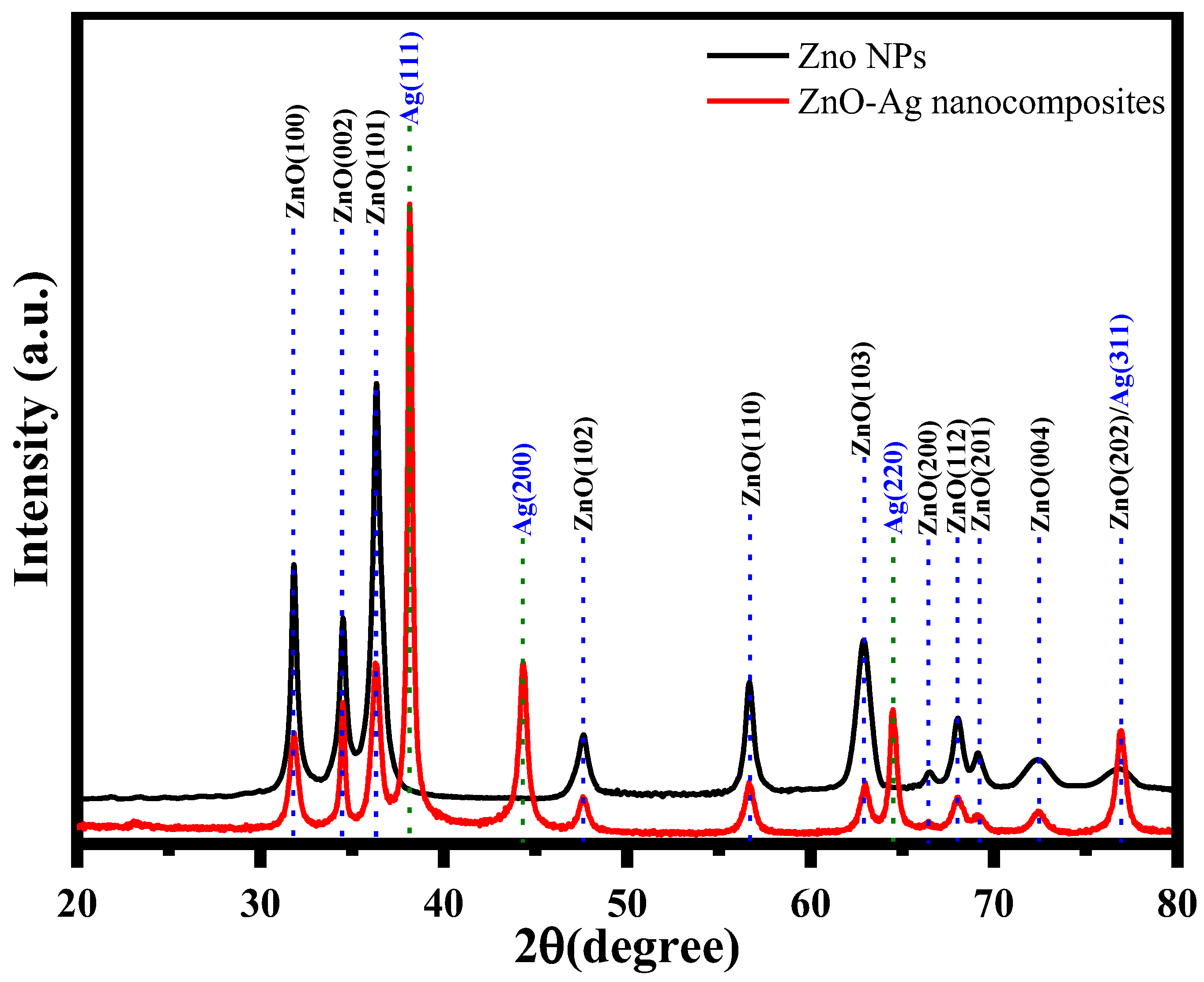

| ZnO NPs | ZnO–Ag Nanocomposite | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| hkl | 2θ | d-Spacing | FWHM | d(nm) | hkl | 2θ | d-Spacing | FWHM | d(nm) |
| 100 | 31.83 | 2.81 | 0.42 | 19.88 | 100 | 31.82 | 2.81 | 0.53 | 15.61 |
| 002 | 34.50 | 2.60 | 0.49 | 16.92 | 002 | 34.49 | 2.60 | 0.28 | 30.21 |
| 101 | 36.35 | 2.47 | 0.57 | 14.69 | 101 | 36.31 | 2.47 | 0.53 | 15.88 |
| 102 | 47.60 | 1.91 | 0.69 | 12.63 | 111(Ag) | 38.16 | 2.36 | 0.32 | 26.62 |
| 110 | 56.68 | 1.62 | 0.56 | 15.98 | 200(Ag) | 44.33 | 2.04 | 0.54 | 15.90 |
| 103 | 62.89 | 1.48 | 0.83 | 11.28 | 102 | 47.61 | 1.91 | 0.50 | 17.41 |
| 200 | 66.47 | 1.41 | 0.76 | 12.49 | 110 | 56.67 | 1.62 | 0.48 | 18.94 |
| 112 | 68.03 | 1.38 | 0.64 | 15.04 | 103 | 62.97 | 1.47 | 0.49 | 19.13 |
| 201 | 69.17 | 1.36 | 0.65 | 14.94 | 220(Ag) | 64.51 | 1.44 | 0.42 | 22.52 |
| 004 | 72.36 | 1.30 | 1.66 | 5.93 | 200 | 66.42 | 1.41 | 0.27 | 34.91 |
| 202 | 76.38 | 1.25 | 2.41 | 4.19 | 112 | 68.05 | 1.38 | 0.64 | 14.89 |
| Average crystallite size | ZnO NPs | 16.0 nm | 201 | 69.15 | 1.36 | 0.49 | 19.61 | ||
| ZnO–Ag nanocomposite | 20.84 nm | 004 | 72.42 | 1.30 | 0.57 | 17.30 | |||
| Ag NPs-Support | 22.71 nm | 202 | 76.47 | 1.24 | 0.52 | 19.31 | |||
| Catalyst | SBET (m²/g) | Pore Diameter (nm) | Pore Volume (cc/g) |
|---|---|---|---|
| ZnO NPs | 11.847 | 2.76 | 0.014 |
| ZnO–Ag | 28.193 | 1.68 | 0.027 |
| Catalyst | Dye/Pharmaceuticals | Degradation Time (Min) | Degradation (%) | Ref. |
|---|---|---|---|---|
| ZnO-CeO2 | Rhodamine B (RhB) | 35 | 92.8 | [109] |
| Ag-Ag2O-ZnO | Methylene blue (MB), methyl orange (MO), and rhodamine B (Rh B) | 60 | 97.3, 91.1 and 94.8, respectively | [110] |
| Ag–ZnO/GP | Metronidazole antibiotic | 180 | 87.1 | [111] |
| Ag–ZnO | Rhodamine B (RhB) | 150 | 95.0 | [112] |
| Ag–ZnO | Phenol | 60 | 100 | [113] |
| Ag–ZnO | Methylene Blue (MB) | 15 | 97.1 | [114] |
| Ag–ZnO | Cibacron brilliant yellow 3G-B | 120 | 65.0 | [115] |
| ZnO | Methylene blue (MB) | 90 | 88.37 | [116] |
| Ag–ZnO | Methylene blue (MB) and congo red (CR) | 90 and 55 | 99.3 and 98.5, respectively | [117] |
| Ag–ZnO | Phenol | 120 | 97.2 | [54] |
| Ag–ZnO | Rhodamine B (RhB) | 250 | 98.0 | [63] |
| Ag–ZnO | Methylene blue (MB) | 80 | 96.0 | [82] |
| ZnO–Ag | Methylene blue (MB) | 60 | 98 | Present Work |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alzahrani, E.A.; Nabi, A.; Kamli, M.R.; Albukhari, S.M.; Althabaiti, S.A.; Al-Harbi, S.A.; Khan, I.; Malik, M.A. Facile Green Synthesis of ZnO NPs and Plasmonic Ag-Supported ZnO Nanocomposite for Photocatalytic Degradation of Methylene Blue. Water 2023, 15, 384. https://doi.org/10.3390/w15030384
Alzahrani EA, Nabi A, Kamli MR, Albukhari SM, Althabaiti SA, Al-Harbi SA, Khan I, Malik MA. Facile Green Synthesis of ZnO NPs and Plasmonic Ag-Supported ZnO Nanocomposite for Photocatalytic Degradation of Methylene Blue. Water. 2023; 15(3):384. https://doi.org/10.3390/w15030384
Chicago/Turabian StyleAlzahrani, Elham A., Arshid Nabi, Majid Rasool Kamli, Soha M. Albukhari, Shaeel Ahmed Althabaiti, Sami A. Al-Harbi, Imran Khan, and Maqsood Ahmad Malik. 2023. "Facile Green Synthesis of ZnO NPs and Plasmonic Ag-Supported ZnO Nanocomposite for Photocatalytic Degradation of Methylene Blue" Water 15, no. 3: 384. https://doi.org/10.3390/w15030384
APA StyleAlzahrani, E. A., Nabi, A., Kamli, M. R., Albukhari, S. M., Althabaiti, S. A., Al-Harbi, S. A., Khan, I., & Malik, M. A. (2023). Facile Green Synthesis of ZnO NPs and Plasmonic Ag-Supported ZnO Nanocomposite for Photocatalytic Degradation of Methylene Blue. Water, 15(3), 384. https://doi.org/10.3390/w15030384






