Use of Sawdust (Aspidosperma polyneuron) in the Preparation of a Biocarbon-Type Adsorbent Material for Its Potential Use in the Elimination of Cationic Contaminants in Wastewater
Abstract
:1. Introduction
2. Materials and Methods
2.1. Aspidosperma Polyneuron Sawdust Pre-Treatment
2.2. Characterization of Biomaterial-Type Carbon
2.3. Adsorption Tests
3. Results
3.1. Biomaterial Synthesis
3.2. Characterization of Bioadsorbents
3.3. Zero Loading Point pHpzc
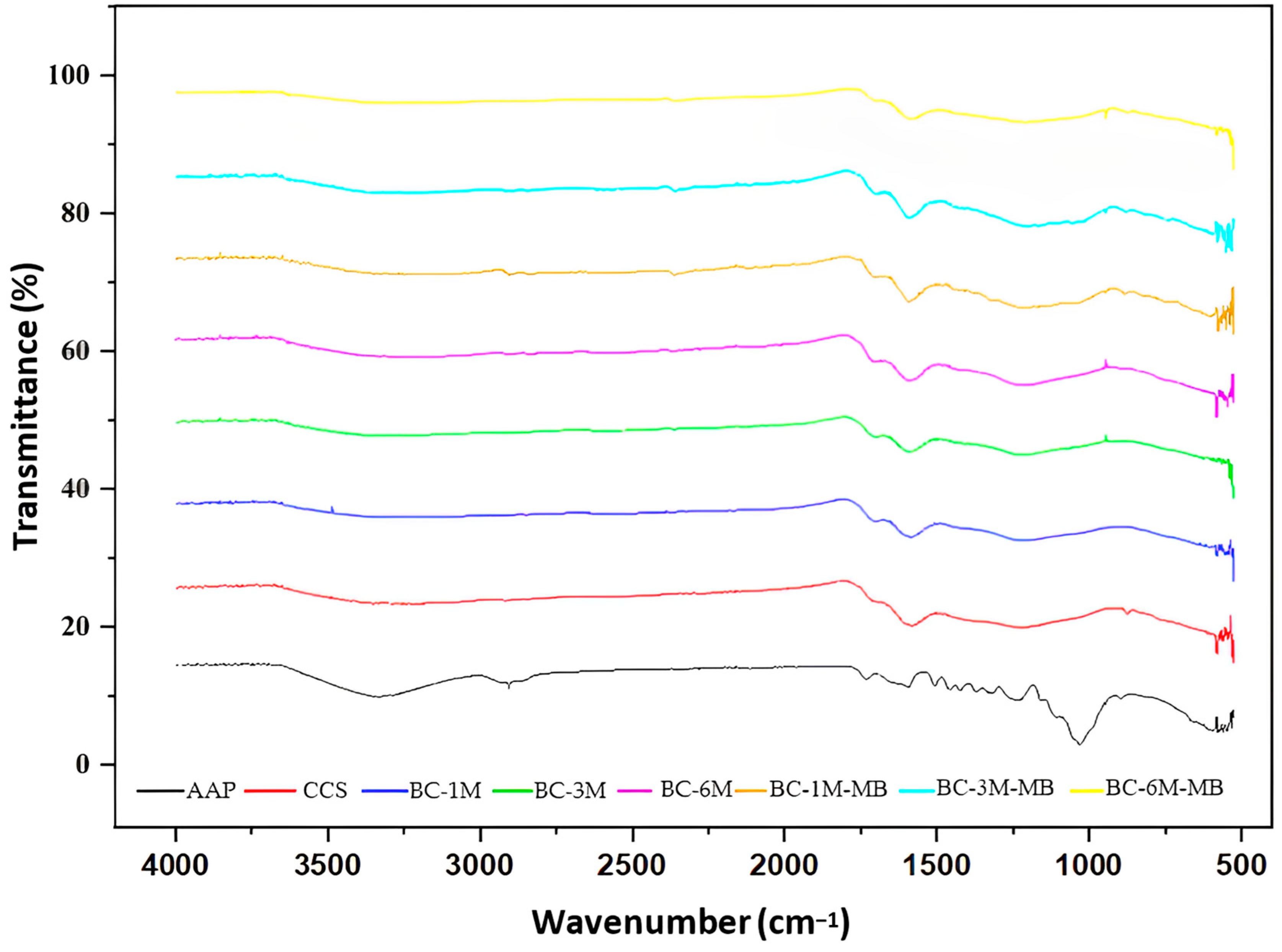
3.4. FTIR Analysis
3.5. Scanning Electron Microscopy
3.6. Methylene Blue Adsorption Tests
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Sample | Weight (g) | Volume (cm3) | Apparent Density (g/cm3) |
|---|---|---|---|
| AAP | 1.080 | 3 | 0.360 |
| CCS | 0.840 | 3 | 0.280 |
| BC-1M | 0.930 | 3 | 0.310 |
| BC-3M | 0.908 | 3 | 0.303 |
| BC-6M | 0.930 | 3 | 0.310 |
| Sample | Sample Weight (g) | Sample Weight + Water (g) | Real Density (g/cm3) | Porosity % |
|---|---|---|---|---|
| AAP | 1.26 | 10.87 | 2.455 | 85.3 |
| CCS | 0.55 | 8.8 | 0.296 | 0.5 |
| BC-1M | 0.49 | 9.1 | 0.327 | 5.1 |
| BC-3M | 0.38 | 9.26 | 0.308 | 1.8 |
| BC-6M | 0.75 | 9.91 | 0.783 | 60.4 |
| Passing Fraction | Particle Number | ΔØ/Dpi | Ni*Dpi | ΔØ*Dpi |
|---|---|---|---|---|
| 1 | - | - | - | - |
| 0.939 | 0.002 | 0.041 | 0.004 | 0.091 |
| 0.618 | 0.101 | 0.428 | 0.075 | 0.241 |
| 0.482 | 0.229 | 0.317 | 0.098 | 0.058 |
| 0.282 | 1.158 | 0.705 | 0.328 | 0.057 |
| 0.174 | 2.409 | 0.598 | 0.436 | 0.020 |
| 0.080 | 5.952 | 0.739 | 0.762 | 0.012 |
| NT | ||||
| 9.851 | 2.827 | 1.703 | 0.479 | |
| Shape factor (a) | 0.524 | |||
| Particle density (Kg/cm3) | 2455 | |||
| S-V diameter (mm) | 0.354 | |||
| Arithmetic mean diameter (mm) | 0.173 | |||
| Mass median diameter (mm) | 0.479 | |||
References
- Sahina, S.C.; Aksub, S. Adsorption of dyes from aqueous textile byproducts on activated carbon from scenedesmus obliquus. Anal. Lett. 2017, 50, 1812–1830. [Google Scholar] [CrossRef]
- Zhang, L.; Lei, M.; Feng, T.; Chang, W.; Ye, A.; Yi, H.; Yi, C. A case study of the wastewater treatment system modification in denim textile industry. J. Text. Inst. 2021, 112, 1666–1670. [Google Scholar] [CrossRef]
- Vyavahare, G.D.; Gurav, R.G.; Jadhav, P.P.; Patil, R.R.; Aware, C.B.; Jadhav, J.P. Response surface methodology optimization for sorption of malachite green dye on sugarcane bagasse biochar and evaluating the residual dye for phyto and cytogenotoxicity. Chemosphere 2018, 194, 306–315. [Google Scholar] [CrossRef] [PubMed]
- Chung, K.T. Azo dyes and human health: A review. J. Environ. Sci. Health Part C 2016, 34, 233–261. [Google Scholar] [CrossRef]
- Shaheen, S.M.; Niazi, N.K.; Hassan, N.E.E.; Bibi, I.; Wang, H.; Tsang, D.C.W.; Ok, Y.S.; Bolan, N.; Rinklebe, J. Wood-based biochar for the removal of potentially toxic elements in water and wastewater: A critical review. Int. Mater. Rev. 2019, 64, 216–247. [Google Scholar] [CrossRef]
- Kuang, Y.; Zhang, X.; Zhou, S. Adsorption of Methylene Blue in Water onto Activated Carbon by Surfactant Modification. Water 2020, 12, 587. [Google Scholar] [CrossRef]
- Zbair, M.; Anfar, Z.; Khallok, H.; Ahsaine, H.A.; Ezahri, M.; Elalem, N. Adsorption kinetics and surface modeling of aqueous methylene blue onto activated carbonaceous wood sawdust. Fuller. Nanotub. Carbon Nanostructures 2018, 26, 433–442. [Google Scholar] [CrossRef]
- Bernal, L.A.; Hernández, M.K.; Berber, M.S.; Martínez, M.E.; Delgado, R.D.; Espinosa, M.Á. Removal of compound dye remazol orange from wastewater generated in the textile industry. Av. Cienc. Ing. 2017, 8, 51–57. [Google Scholar]
- Ardila-Ramírez, C.; Palacio-Londoño, Á.; Barrera-Zapata, R. Pinneapple peel as adsorbent of typical textile industry dyes. Cienc. Desarro. 2018, 9, 161–168. [Google Scholar]
- Li, L.; Yang, M.; Lu, Q.; Zhu, W.; Ma, H.; Dai, L. Oxygen-rich biochar from torrefaction: A versatile adsorbent for water pollution control. Bioresour. Technol. 2019, 294, 122142. [Google Scholar] [CrossRef]
- Alshammari, M.; Essawy, A.A.; El-Nggar, A.M.; Sayyah, S.M. Ultrasonic-Assisted Synthesis and Characterization of Chitosan-Graft-Substituted Polyanilines: Promise Bio-Based Nanoparticles for Dye Removal and Bacterial Disinfection. J. Chem. 2020, 2020, 3297184. [Google Scholar] [CrossRef]
- Bortoluz, J.; Ferrarini, F.; Bonetto, L.R.; da Silva Crespo, J.; Giovanela, M. Use of low-cost natural waste from the furniture industry for the removal of methylene blue by adsorption: Isotherms, kinetics and thermodynamics. Cellulose 2020, 27, 6445–6466. [Google Scholar] [CrossRef]
- Bhowmik, S.; Chakraborty, V.; Das, P. Batch adsorption of indigo carmine on activated carbon prepared from sawdust: A comparative study and optimization of operating conditions using Response Surface Methodology. Results Surf. Interfaces 2021, 3, 100011. [Google Scholar] [CrossRef]
- Sun, L.; Chen, D.; Wan, S.; Yu, Z. Performance, kinetics, and equilibrium of methylene blue adsorption on biochar derived from eucalyptus saw dust modified with citric, tartaric, and acetic acids. Bioresour. Technol. 2015, 198, 300–308. [Google Scholar] [CrossRef] [PubMed]
- Oyewo, O.A.; Adeniyi, A.; Sithole, B.B.; Onyango, M.S. Sawdust-Based Cellulose Nanocrystals Incorporated with ZnO Nanoparticles as Efficient Adsorption Media in the Removal of Methylene Blue Dye. ACS Omega 2020, 5, 18798–18807. [Google Scholar] [CrossRef]
- Sajjadi, B.; Broome, J.W.; Chen, W.Y.; Mattern, D.L.; Egiebor, N.O.; Hammer, N.; Smith, C.L. Urea functionalization of ultrasound-treated biochar: A feasible strategy for enhancing heavy metal adsorption capacity. Ultrason. Sonochem. 2019, 51, 20–30. [Google Scholar] [CrossRef]
- Kasera, N.; Hall, S.; Kolar, P. Effect of surface modification by nitrogen-containing chemicals on morphology and surface characteristics of N-doped pine bark biochars. J. Environ. Chem. Eng. 2021, 9, 105161. [Google Scholar] [CrossRef]
- Kalak, T.; Kaczmarek, M.; Nowicki, P.; Pietrzak, R.; Tachibana, Y.; Cierpiszewski, R. Preparation of nitrogen-enriched pine sawdust-based activated carbons and their application for copper removal from the aquatic environment. Wood Sci. Technol. 2022, 56, 1721–1742. [Google Scholar] [CrossRef]
- Chen, X.; Li, H.; Liu, W.; Meng, Z.; Wu, Z.; Wang, G.; Liang, Y.; Bi, S. Low-temperature constructing N-doped graphite-like mesoporous structure biochar from furfural residue with urea for removal of chlortetracycline from wastewater and hydrothermal catalytic degradation mechanism. Colloids Surfaces A Physicochem. Eng. Asp. 2020, 600, 124873. [Google Scholar] [CrossRef]
- ASTM C136/C136M-19; Standard Test Method for Sieve Analysis of Fine and Coarse Aggregates. ASTM: West Conshohocken, PA, USA, 2020.
- Lin, Y.Q.; Tsai, W.T. Liquid-Phase Removal of Methylene Blue as Organic Pollutant by Mesoporous Activated Carbon Prepared from Water Caltrop Husk Using Carbon Dioxide Activation. Processes 2021, 9, 238. [Google Scholar] [CrossRef]
- Ovhal, S.D.; Rodrigues, C.S.D.; Madeira, L.M. Photocatalytic wet peroxide assisted degradation of Orange II dye by reduced graphene oxide and zeolites. J. Chem. Technol. Biotechnol. 2021, 96, 349–359. [Google Scholar] [CrossRef]
- ASTM D 3860-98; Standard Practice for Determination of Adsorptive Capacity of Activated Carbon by Aqueous Phase Isotherm Technique. ASTM: West Conshohocken, PA, USA, 2014.
- Ramakrishnan, R.K.; Padil, V.V.T.; Wacławek, S.; Černík, M.; Varma, R.S. Eco-Friendly and Economic, Adsorptive Removal of Cationic and Anionic Dyes by Bio-Based Karaya Gum—Chitosan Sponge. Polymers 2021, 13, 251. [Google Scholar] [CrossRef] [PubMed]
- Hou, Y.; Yan, S.; Huang, G.; Yang, Q.; Huang, S.; Cai, J. Fabrication of N-doped carbons from waste bamboo shoot shell with high removal efficiency of organic dyes from water. Bioresour. Technol. 2020, 303, 122939. [Google Scholar] [CrossRef]
- Paolella, M.S. Linear Models and Time-Series Analysis: Regression, ANOVA, ARMA and GARCH; John Wiley & Sons: Hoboken, NJ, USA, 2018. [Google Scholar]
- Tejada-Tovar, C.; Villabona-Ortíz, A.; Ortega-Toro, R. Determination of Kinetic Parameters in the Biosorption of Chromium (VI) in Aqueous Solution. Ing. Cienc. 2020, 16, 129–143. [Google Scholar] [CrossRef]
- Krysanova, K.; Krylova, A.; Kulikova, M.; Kulikov, A.; Rusakova, O. Biochar characteristics produced via hydrothermal carbonization and torrefaction of peat and sawdust. Fuel 2022, 328, 125220. [Google Scholar] [CrossRef]
- Qin, F.; Zhang, C.; Zeng, G.; Huang, D.; Tan, X.; Duan, A. Lignocellulosic biomass carbonization for biochar production and characterization of biochar reactivity. Renew. Sustain. Energy Rev. 2022, 157, 112056. [Google Scholar] [CrossRef]
- Salehi, E.; Askari, M.; Velashjerdi, M.; Arab, B. Phosphoric acid-treated Spent Tea Residue Biochar for Wastewater Decoloring: Batch Adsorption Study and Process Intensification using Multivariate Data-based Optimization. Chem. Eng. Process.-Process Intensif. 2020, 158, 108170. [Google Scholar] [CrossRef]
- Nath, H.; Saikia, A.; Goutam, P.J.; Saikia, B.K.; Saikia, N. Removal of methylene blue from water using okra (Abelmoschus esculentus L.) mucilage modified biochar. Bioresour. Technol. Reports 2021, 14, 100689. [Google Scholar] [CrossRef]
- Prías, J.J.; Rojas, C.A.; Echeverry, N.A.; Fonthal, G.; Ariza, H. Identification of optimal variables from the precursor Guadua angustifolia Kunth. Rev. Acad. Colomb. Cienc. Exactas Físicas Nat. 2011, 35, 157–166. [Google Scholar]
- Saletnik, B.; Saletnik, A.; Zaguła, G.; Bajcar, M.; Puchalski, C. The Use of Wood Pellets in the Production of High Quality Biocarbon Materials. Materials 2022, 15, 4404. [Google Scholar] [CrossRef]
- Prieto García, J.O.; Pérez Leiva, A.; Curbelo Sánchez, A.E.; Enríquez García, M.; Mollineda Trujillo, Á. Adsorption of lead (II) ions in isothermal conditions using activated carbon from sugarcane bagasse, marabou and bamboo. Cent. Azúcar 2021, 48, 21–28. [Google Scholar]
- Velázquez-Maldonado, J.; Juárez-López, P.; Anzaldo-Hernández, J.; Alejo-Santiago, G.; Valdez-Aguilar, L.A.; Alia-Tejacal, I.; López-Martínez, V.; Guillén-Sánchez, G.A.; Dagoberto, P.-A. Nutrient concentration of rice husk biochar. Rev. Fitotécnica Mex. 2019, 42, 129–136. [Google Scholar]
- Yang, C.D.; Lu, S.G. Effects of five different biochars on aggregation, water retention and mechanical properties of paddy soil: A field experiment of three-season crops. Soil Tillage Res. 2021, 205, 104798. [Google Scholar] [CrossRef]
- Anggraeni, S.; Chelvina, G.; Girsang, S.; Bayu, A.; Nandiyanto, D.; Roild Bilad, M. Effects of particle size and composition of sawdust/carbon from rice husk on the briquette performance. J. Eng. Sci. Technol. 2021, 16, 2298–2311. [Google Scholar]
- Serret-Guasch, N.; Giralt-Ortega, G.; Quintero-Ríos, M. Characterization of Sawdust of different Woods. Tecnol. Química 2016, 36, 395–405. [Google Scholar]
- Mengist, M.; Woldeyes, B.; Gabbiye, N. Production and Characterization of Sawdust Briquettes for Firewood Substitution. In International Conference on Advances of Science and Technology; Springer: Cham, Switzerland, 2021; Volume 384, pp. 3–26. [Google Scholar]
- Prieto García, J.O.; Gehan Geulamussein, N.; Pérez Leiva, A.; Martínez Albelo, E.; Enríquez García, M. Adsorption of Cadmium from an Aqueous Solution on Activated Coal from Bagasse of Sugarcane Roxa Variety. Rev. Cent. Azúcar 2020, 47, 1. Available online: http://centroazucar.uclv.edu.cu/index.php/centro_azucar/article/view/172 (accessed on 27 August 2023).
- Daza Barranco, L.M.; Orrego, M.J.F.; Fernández Hincapie, J.J. Preparation and characterization of activated carbons from coal of Cesar’s basin (Colombia). Rev. Politécnica 2018, 14, 75–88. [Google Scholar] [CrossRef]
- Chen, Y.D.; Lin, Y.C.; Ho, S.H.; Zhou, Y.; Ren, N.Q. Highly efficient adsorption of dyes by biochar derived from pigments-extracted macroalgae pyrolyzed at different temperature. Bioresour. Technol. 2018, 259, 104–110. [Google Scholar] [CrossRef]
- Mu, Y.; Ma, H. NaOH-modified mesoporous biochar derived from tea residue for methylene Blue and Orange II removal. Chem. Eng. Res. Des. 2021, 167, 129–140. [Google Scholar] [CrossRef]
- Ahmed, M.J.; Okoye, P.U.; Hummadi, E.H.; Hameed, B.H. High-performance porous biochar from the pyrolysis of natural and renewable seaweed (Gelidiella acerosa) and its application for the adsorption of methylene blue. Bioresour. Technol. 2019, 278, 159–164. [Google Scholar] [CrossRef]
- Zuo, H.; Qin, X.; Liu, Z.; Fu, Y. Preparation and characterization of modified corn stalk biochar. BioResources 2021, 16, 7428–7443. [Google Scholar] [CrossRef]
- Liu, X.J.; Li, M.F.; Singh, S.K. Manganese-modified lignin biochar as adsorbent for removal of methylene blue. J. Mater. Res. Technol. 2021, 12, 1434–1445. [Google Scholar] [CrossRef]
- Liu, S.; Li, J.; Xu, S.; Wang, M.; Zhang, Y.; Xue, X. A modified method for enhancing adsorption capability of banana pseudostem biochar towards methylene blue at low temperature. Bioresour. Technol. 2019, 282, 48–55. [Google Scholar] [CrossRef] [PubMed]
- Ying, Z.; Chen, X.; Li, H.; Liu, X.; Zhang, C.; Zhang, J.; Yi, G. Efficient Adsorption of Methylene Blue by Porous Biochar Derived from Soybean Dreg Using a One-Pot Synthesis Method. Molecules 2021, 26, 661. [Google Scholar] [CrossRef] [PubMed]
- Llenque-Díaz, L.A.; Quintana, A.; Torres, L.; Segura, R. Bioethanol production from organic plant waste. Rebiol 2020, 40, 21–29. [Google Scholar] [CrossRef]
- Jayakrishnan, U.; Deka, D.; Das, G. Waste as feedstock for polyhydroxyalkanoate production from activated sludge: Implications of aerobic dynamic feeding and acidogenic fermentation. J. Environ. Chem. Eng. 2021, 9, 105550. [Google Scholar] [CrossRef]
- Haile, A.; Gelebo, G.G.; Tesfaye, T.; Mengie, W.; Mebrate, M.A.; Abuhay, A.; Limeneh, D.Y. Pulp and paper mill wastes: Utilizations and prospects for high value-added biomaterials. Bioresour. Bioprocess. 2021, 8, 35. [Google Scholar] [CrossRef]
- Le Pera, A.; Sellaro, M.; Bencivenni, E.; D’Amico, F. Environmental sustainability of an integrate anaerobic digestion-composting treatment of food waste: Analysis of an Italian plant in the circular bioeconomy strategy. Waste Manag. 2022, 139, 341–351. [Google Scholar] [CrossRef]
- Khasri, A.; Bello, O.S.; Ahmad, M.A. Mesoporous activated carbon from Pentace species sawdust via microwave-induced KOH activation: Optimization and methylene blue adsorption. Res. Chem. Intermed. 2018, 44, 5737–5757. [Google Scholar] [CrossRef]
- Zhang, Z.B.; Liu, X.Y.; Li, D.W.; Gao, T.T.; Lei, Y.Q.; Wu, B.G.; Zhao, J.W.; Wang, Y.K.; Wei, L. Effects of the ultrasound-assisted H3PO4 impregnation of sawdust on the properties of activated carbons produced from it. New Carbon Mater. 2018, 33, 409–416. [Google Scholar] [CrossRef]
- Jock, A.A.; Joel, A.S.; Olubajo, O.O.; Zang, C.U.; Ayuba, M.S.; Wakili, P.T. Development of activated carbon from sawdust by pyrolysis and methylene blue adsorption. Int. J. Chem. React. Eng. 2021, 19, 473–481. [Google Scholar] [CrossRef]
- Hoslett, J.; Ghazal, H.; Mohamad, N.; Jouhara, H. Removal of methylene blue from aqueous solutions by biochar prepared from the pyrolysis of mixed municipal discarded material. Sci. Total Environ. 2020, 714, 136832. [Google Scholar] [CrossRef] [PubMed]
- Zeng, H.; Qi, W.; Zhai, L.; Wang, F.; Zhang, J.; Li, D. Magnetic biochar synthesized with waterworks sludge and sewage sludge and its potential for methylene blue removal. J. Environ. Chem. Eng. 2021, 9, 105951. [Google Scholar] [CrossRef]




| Carbonization Temperature (°C) | Dwell Time (h) | Initial Mass (g) | Final Mass (g) | Yield (%) | Ash in the Final Mass |
|---|---|---|---|---|---|
| 500 | 3 | 50 | 0.8 | 1.6 | Yes |
| 500 | 2 | 50 | 0.8 | 1.6 | Yes |
| 250 | 1 | 50 | 4.2 | 8.4 | No |
| 250 | ½ | 50 | 6.3 | 12.6 | No |
| Sample | Volume (cm3) | Apparent Density (g/cm3) | Real Density (kg/cm3) | Porosity % |
|---|---|---|---|---|
| AAP | 3 | 0.360 | 2.455 | 85.3 |
| CCS | 3 | 0.280 | 0.296 | 0.5 |
| BC-1M | 3 | 0.310 | 0.327 | 5.1 |
| BC-3M | 3 | 0.303 | 0.308 | 1.8 |
| BC-6M | 3 | 0.310 | 0.783 | 60.4 |
| Mass (g) | Mesh | Aperture (mm) | Dpi (mm) | (ΔØ) | (ØR) | |
|---|---|---|---|---|---|---|
| - | - | 10 | 2 | - | - | - |
| m1 | 10.7 | 18 | 1 | 1.5 | 0.061 | 0.061 |
| m2 | 56.4 | 35 | 0.5 | 0.75 | 0.321 | 0.382 |
| m3 | 23.8 | 45 | 0.355 | 0.428 | 0.136 | 0.518 |
| m4 | 35.1 | 70 | 0.212 | 0.284 | 0.200 | 0.718 |
| m5 | 19.0 | 100 | 0.15 | 0.181 | 0.108 | 0.826 |
| m6 | 16.6 | 140 | 0.106 | 0.128 | 0.095 | 0.920 |
| m7 | 14 | Bottom | Bottom | - | 0.080 | 1.000 |
| MT | 175.6 | - | - | - | 1 | - |
| Adsorbent | Source of Variation | Sum of Squares | Degrees of Freedom | F0 | p Value |
|---|---|---|---|---|---|
| CCS | Treatments | 0.0014 | 2 | 3.92 | 0.1456 |
| Error | 0.0005 | 3 | |||
| Total | 0.0019 | 5 | |||
| BC-1M | Treatments | 0.4562 | 2 | 0.63 | 0.5915 |
| Error | 1.0885 | 3 | |||
| Total | 1.5447 | 5 | |||
| BC-3M | Treatments | 0.0009 | 2 | 1.47 | 0.3584 |
| Error | 0.0009 | 3 | |||
| Total | 0.0018 | 5 | |||
| BC-6M | Treatments | 2.4637 | 2 | 1.80 | 0.3063 |
| Error | 2.0515 | 3 | |||
| Total | 4.5152 | 5 |
| Biomass | Synthesis Variables | Characterizations | References | |||
|---|---|---|---|---|---|---|
| Methylene Blue (MB) Test | Techniques | |||||
| Sawdust mix | T (°C) | 450 | qe (mg/g) | 32 | SEM: Identification of the polymeric layer on the surface of the biochar. FTIR: Amide I and Amide II. Amino acids and proteins (1635 and 1565 cm−1). Carboxylate ion (1395 cm−1) and pectin pyranoid ring (1130 cm−1). Phenolic O-H (1260 cm−1) Organic phosphorus P–O and/or sugar C–O bond present in the biopolymer. pHpzx = 7.95 | [31] |
| t (h) | 4 | Adsorbent dose (g/L) | 1 | |||
| Free of O2 | No | |||||
| Modification | Biopolymer layer (Okra mucilage). Activation with NaOH | MB concentration (ppm) | 25 | |||
| pH | 7 | |||||
| Eucalyptus wood | T (°C) | 700 | qe (mg/g) | 112 | SEM: Presence of channels in the form of columns and slits. FTIR: Peaks at 3000–3500 cm−1 for –OH groups; at 1799 cm−1 for C=O from carbonyl and carboxyl groups; 1579 cm−1 for C=C bonds; 1400 cm−1 for COO carboxylic groups; 1122 cm−1 for C-O bonds; 588, 622, 696, and 877cm−1 for FeO bonds. SBET = 645.23 m2/g. Average pore diameter: 2.71 nm. Pore volume: 0.44 cm3/g. | [42] |
| t (h) | 1 h, 15 min | Adsorbent dose (g/L) | 1 | |||
| Free of O2 | No | |||||
| Modification | Activation with FeCl3 | MB concentration (ppm) | 50 | |||
| T (°C) | 25 | |||||
| Sawdust (PSAC) from the Pentace species | T (°C) | 700 | qe (mg/g) | 42.58 | SEM: Rough and uneven surface textures. Pores of different sizes and shapes were visualized. The chemical activation process produced pores on the carbon’s surface and obtained carbon with a large surface area and a porous structure. FTIR: Peaks at 3611–3951 cm−1 for OH vibrations; 2720 cm−1 for C–H groups; 2335 cm−1 for C-C bonds; 1689 and 1520 cm−1 for C = O bonds and aromatic rings C = C, respectively; and 1269 cm−1 related to the C-O stretch of the phenolic group. SBET: 914.15 m2/g, Pore volume: 0.52 cm3/g. Average pore diameter: 3.19 nm. | [53] |
| t (h) | 2 | Adsorbent dose (g/L) | 1 | |||
| Free of O2 | Yes | |||||
| Modification | Activation with KOH | MB concentration (ppm) | 50 | |||
| pH | 7 | |||||
| Sawdust | T (°C) | 425 | qe (mg/g) | 210 | SEM: The surface textures of the precursors were rough and uneven. Pores of different sizes and shapes were evident. FTIR: Peaks at 3420 cm−1 for OH groups; 2926 and 2853 cm−1 for CH groups; 1458 and 1377 cm−1 for C=O bonds; 1704 cm−1 for COO- groups; and P-containing groups at 1167 cm−1 for C-O-P formed by the reaction of phosphorus oxides. SBET: 1504 m2/g. Pore volume: 0.78 cm3/g. | [54] |
| t (h) | 2 | Adsorbent dose (g/L) | - | |||
| Free of O2 | Yes | |||||
| Modification | Activation with H3PO4 assisted with ultrasound for 5 min | MB concentration (ppm) | - | |||
| pH | - | |||||
| Sugarcane bagasse | T (°C) | 400 | qe (mg/g) | 15 | Pycnometric density: 1.09 g/mL. Bulk density: 0.28 g/mL. Total porosity: 74%. Fixed Coal: 12%. Bulk density: 0.33 g/mL. Average pore radius: 535 nm. | [34] |
| t (h) | 1 | Adsorbent dose (g/L) | - | |||
| Free of O2 | No | |||||
| Modification | Activation first with H3PO4 followed of activation with HNO3 | MB concentration (ppm) | - | |||
| pH | - | |||||
| Sugarcane bagasse | T (°C) | 600 | qe (mg/g) | 15 | Pycnometric density: 1.09 g/mL. Bulk density: 0.28 g/mL. Total porosity: 74%. Fixed Coal: 12%. Apparent density: 0.28 g/mL. SBET: 305 m2/g. pHpzx: 6.1. | [40] |
| t (h) | 2 | Adsorbent dose (g/L) | - | |||
| Free of O2 | No | |||||
| Modification | Activation first with H3PO4 followed of activation with HNO3 | MB concentration (ppm) | - | |||
| pH | - | |||||
| Tea residue | T (°C) | 700 | qe (mg/g) | 48.39 | SEM: Porous channel structure rich and well developed, better than unmodified biochar. Elemental analysis by EDS: Detection of C, N, O, Na, and K atoms with a concentration of 81.29%, 8.79%, 6.19%, 1.59%, and 0.03%, respectively. SBET: 178 m2/g, Pore volume: 0.164 cm3/g. | [43] |
| t (h) | 4 | Adsorbent dose (g/L) | 1 | |||
| Free of O2 | No | |||||
| Modification | Activation with NaOH at 10% w/w | MB concentration (ppm) | 50 | |||
| pH | 10 | |||||
| Tea residue | T (°C) | 300 | qe (mg/g) | 31.46 | SEM: Semi-finished porous structure was observed. FTIR: Peaks at 3413 cm−1 for OH groups; 2921 cm−1 and 2851 cm−1 for -CH bonds; 1613 cm−1 for secondary amine; 1440 cm−1 for C-C bonds in aromatic rings; 1000–1163 cm−1 for C-O groups; and bands at 960–970 cm−1 for C–H bonds in aromatic rings. SBET: 1.9 m2/g. Pore volume: 0.0048 cm3/g. | [30] |
| t (h) | - | Adsorbent dose (g/L) | 1.3 | |||
| Free of O2 | Yes | |||||
| Modification | Activation with H3PO4 | MB concentration (ppm) | 50 | |||
| pH | 9.65 | |||||
| Lignin | T (°C) | 400 | qe (mg/g) | 162.93 | SEM: Biochar samples have micropore structure. Unmodified and modified biochar samples had similar pore diameter distributions. FTIR: Peaks at 3442 cm-1 for OH groups; 1620 cm−1 for C-H groups in aromatic ring; 1384 cm-1 for -CH2 and -CH3 groups; 1262 cm−1 for ether bond; and 1082 cm-1 for C-O bond. SBET: 349.646 m2/g. Average pore diameter: 2.184 nm. Pore width: 1.178 nm. Pore volume: 0.029 cm3/g. | [46] |
| t (h) | 1 | Adsorbent dose (g/L) | - | |||
| Free of O2 | No | |||||
| Modification | Modification with MnO2 | MB concentration (ppm) | 50 | |||
| pH | 12 | |||||
| Banana pseudostem | T (°C) | 200 | qe (mg/g) | 81.54 | SEM: It has a mesopore structure. The modified biochar showed smaller pore size, which could be caused by Mo filling in the pores or pore collapse. FTIR: Peaks at 3440 cm−1 for OH groups; 2927 cm−1 for C-H groups; 1595 and 1642 cm−1 attributed to C-O and C-C groups, respectively; 1434 and 1449 cm−1 for C-C groups in aromatics; 1040 and 1097 cm−1 for C-O bonds in aliphatic and aromatic structures; 951 and 799 cm−1 for Mo-O bonds. Average pore diameter: 43.283 μm. Pore diameter: 14.33 nm, Pore volume: 0.01234 cm3/g. SBET: 3.741 m2/g. | [47] |
| t (h) | 1 | Adsorbent dose (g/L) | 0.5 | |||
| Free of O2 | Yes | |||||
| Modification | Phosphomolybdic acid | MB concentration (ppm) | 50 | |||
| pH | 7 | |||||
| Seaweed (Gelidiella acerosa) | T (°C) | 800 | qe (mg/g) | 96 | SEM/EDS: Pores and cavities are observed on the surface/C (72.96%), O (15.03%), N (5.78%) and S (6.22%). FTIR: Peaks at 3440 cm-1 for O–H group; 1625 cm−1 for primary amine N–H group; 1389 cm-1 for N–O group; 1168 and 1127 cm−1 for C–O group. SBET: 926.39 m2/g. Mesopore volume and size: 0.57 cm3/g and 2.45 nm, respectively. | [44] |
| t (h) | 3 | Adsorbent dose (g/L) | 1 | |||
| Free of O2 | Yes | |||||
| Modification | The biomass was washed with HCl and distilled water at pH 6.5 | MB concentration (ppm) | 100 | |||
| pH | 7 | |||||
| Fir sawdust | T (°C) | 400 | qe (mg/g) | 0.38 | SEM: Irregular sheet-like structure, sparse texture, and presence of few small pores on the carbon surface. FTIR: Peaks at 3700 cm−1 for O-H group; 2840 and 2349 cm−1 for C–H and O=C=O groups, respectively; 1650 cm−1 for C–H bond; 1050 cm-1 for C-O group; and 875 for C-Cl bond. Elemental analysis by EDS: Detection of C, Ca, Si, Al, K, and S atoms with a concentration of 78.03%, 18.64%, 1.469%, 0.562%, 0.103%, and 0.139%, respectively. | [55] |
| t (h) | - | Adsorbent dose (g/L) | 30 | |||
| Free of O2 | Yes | |||||
| Modification | No | MB concentration (ppm) | 60 | |||
| pH | 6.5 | |||||
| Water caltrop shell | T (°C) | 750 | qe (mg/g) | 125 | SEM: Small pores were observed in addition to a rigid frame on the surface of the resulting carbon. SBET: 810.5 ± 25.7 m2/g. Pore volume: 0.441 ± 0.024 cm3/g. Average pore diameter: 21.7 ± 0.8 Å. Real density: 1.787 g/cm3. Particle density: 0.999 g/cm3. Porosity: 0.441. | [21] |
| t (h) | 1.5 | Adsorbent dose (g/L) | 0.15 | |||
| Free of O2 | Yes | |||||
| Modification | No | MB concentration (ppm) | 20 | |||
| pH | 7 | |||||
| Soybean dross | T (°C) | 800 | qe (mg/g) | 996.37 | FTIR: Peaks at 3411 cm−1 for O-H group; 2933 cm−1 for C-H bond; 1710 and 1587 cm−1 for C = O and C = C bonds; 1095 cm−1 for C-OH bond. SEM: Random pore structures were evident on the surface after high-temperature activation with KHCO3. SBET: 1620 m2/g. Elemental analysis by EDS: Detection of C, O, H, N, and S atoms with a concentration of 66.28%, 28.11%, 1.67%, 0.542%, and 0.383%, respectively. Pore volume: 0.7509 cm3/g. Average pore diameter: 1.859 nm. Yield: 15.61%. | [48] |
| t (h) | - | Adsorbent dose (g/L) | 1 | |||
| Free of O2 | Yes | |||||
| Modification | Activated with KHCO3 | MB concentration (ppm) | 1000 | |||
| pH | ||||||
| Corn stalk | T (°C) | 300 | qe (mg/g) | 16.41 | SEM: Increased pores were observed on the surface after biomass modification. FTIR: Peaks at 3410 cm−1 for O–H group; 2925 and 2842 cm−1 for -CH3 and -CH2 groups; 1680 and 1430 cm−1 for vibration of the benzene ring; 1109 cm−1 for C-O bond from phenols and oxyhydrogen groups; 873 cm−1 due to weaker aromatization. SBET: 2.56 m2/g. Pore volume: 0.00864 cm3/g Average pore diameter: 13.5 nm. | [45] |
| t (h) | 1 | Adsorbent dose (g/L) | 2 | |||
| Free of O2 | Yes | |||||
| Modification | Modification with MgCl2 | MB concentration (ppm) | 45 | |||
| pH | ||||||
| Mixture of food, garden, paper, and cardboard waste | T (°C) | 300 | qe (mg/g) | 5.018 | SEM: It was evaluated two samples of BC from paper/cardboard and food/garden, showing differences in the structure of the surface, demonstrating the variability of the structure of carbons from this type of waste. FTIR: Peaks at 1416, 873, and 712 cm−1 for CO3 presence; peaks between 3500 and 3000 cm−1 for OH groups; and peaks between 1550 and 1350 cm−1 for calcite. | [56] |
| t (h) | 12 | Adsorbent dose (g/L) | 5 | |||
| Free of O2 | Yes | |||||
| Modification | No | MB concentration (ppm) | 75 | |||
| pH | 5 | |||||
| Sewage sludge | T (°C) | 180 °C in drying oven | qe (mg/g) | 15.77 | FTIR: Peaks at 3420 cm−1 for OH group; 2920 and 2853 cm−1 for methylene; 1630 and 1030 cm−1 for carbonyl; and 575 cm−1 attributed to Fe-O. | [57] |
| t (h) | 12 h | Adsorbent dose (g/L) | 0.5 | |||
| Free of O2 | No (hydro thermal process) | |||||
| Modification | Mixing and carbonization with Fe-rich sludge (by-product of groundwater purification). | MB concentration (ppm) | 10 | |||
| pH | ||||||
| Sawdust from Aspidosperma polyneuron | T (°C) | 250 | qe (mg/g) | 12.45 | FTIR: Peak at 3183 cm−1 for OH group; 1586 cm−1 for C = C bond from the aromatic ring; 1215 cm−1 for carboxyl group; peaks from 581 to 533 cm−1 for hydrocarbon groups of single, double, and triple bonds. SEM/EDS: Porous surface was observed; however, the pores decreased in size with respect to the urea concentration. Elemental analysis by EDS: Detection of C and O atoms with a concentration of 68.45% and 31.55%, respectively. Actual density: 0.783 g/cm3. Porosity: 0.604. pHpzc: 5.6 | Present study |
| t (h) | 0.5 | Adsorbent dose (g/L) | 3.5 | |||
| Free of O2 | No | |||||
| Modification | Activation with H3PO4 and functionalization with urea (6M) | MB concentration (ppm) | 60 | |||
| pH | 7 | |||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ortega-Toro, R.; Villabona-Ortíz, Á.; Tejada-Tovar, C.; Herrera-Barros, A.; Cabrales-Sanjuan, D. Use of Sawdust (Aspidosperma polyneuron) in the Preparation of a Biocarbon-Type Adsorbent Material for Its Potential Use in the Elimination of Cationic Contaminants in Wastewater. Water 2023, 15, 3868. https://doi.org/10.3390/w15213868
Ortega-Toro R, Villabona-Ortíz Á, Tejada-Tovar C, Herrera-Barros A, Cabrales-Sanjuan D. Use of Sawdust (Aspidosperma polyneuron) in the Preparation of a Biocarbon-Type Adsorbent Material for Its Potential Use in the Elimination of Cationic Contaminants in Wastewater. Water. 2023; 15(21):3868. https://doi.org/10.3390/w15213868
Chicago/Turabian StyleOrtega-Toro, Rodrigo, Ángel Villabona-Ortíz, Candelaria Tejada-Tovar, Adriana Herrera-Barros, and Daniela Cabrales-Sanjuan. 2023. "Use of Sawdust (Aspidosperma polyneuron) in the Preparation of a Biocarbon-Type Adsorbent Material for Its Potential Use in the Elimination of Cationic Contaminants in Wastewater" Water 15, no. 21: 3868. https://doi.org/10.3390/w15213868









