The Effect of Peatland Restoration on Ciliate Communities: Long-Term Analyses
Abstract
:1. Introduction
- Restoration causes temporary destabilization of environmental parameters.
- Ciliates can be an excellent group of indicator species useful in monitoring the effects of peatland restoration.
2. Materials and Methods
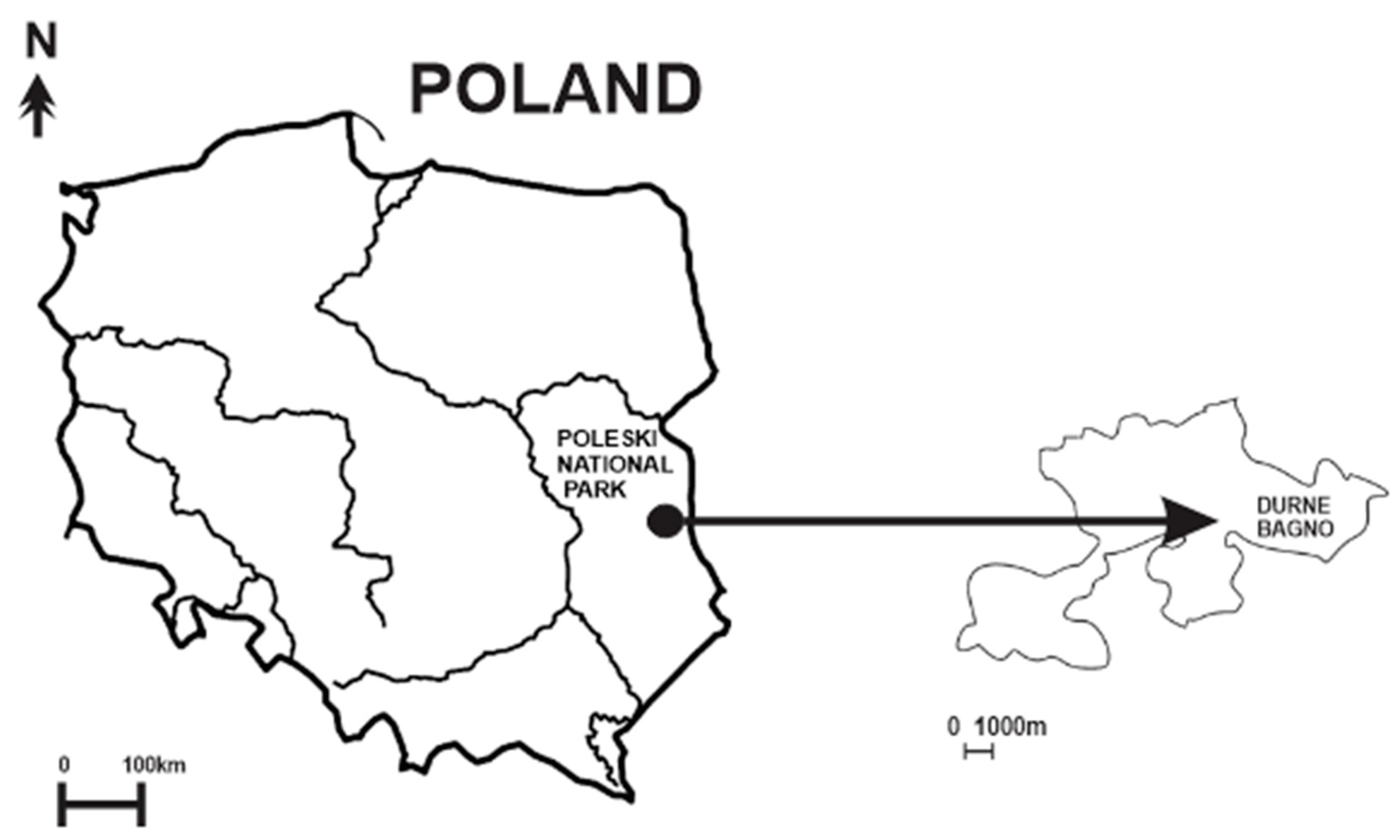
2.1. Study Site
2.2. Abiotic Variables
2.3. Ciliata Communities
2.4. Data Analyses
3. Results
3.1. Abiotic Variables
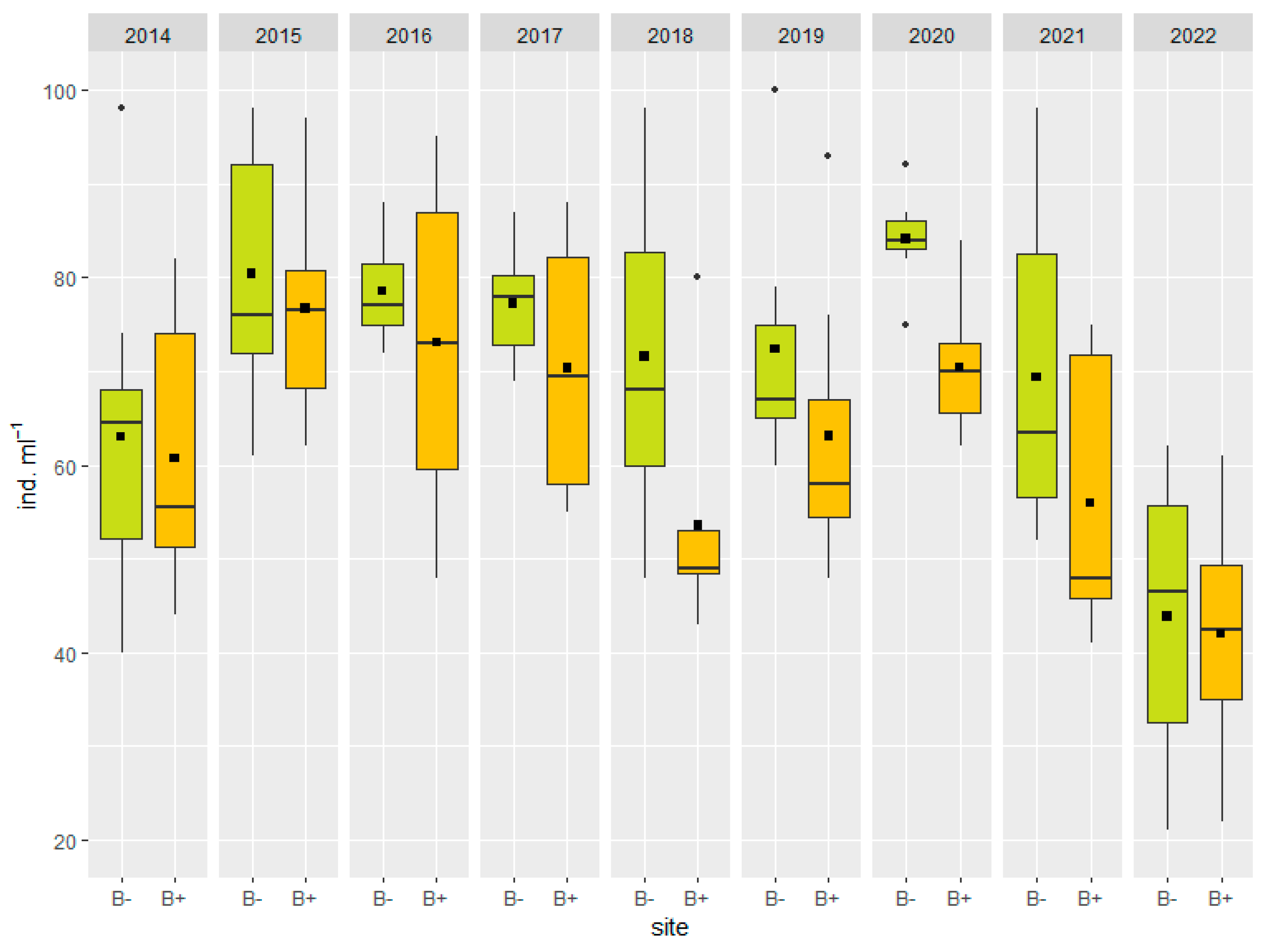
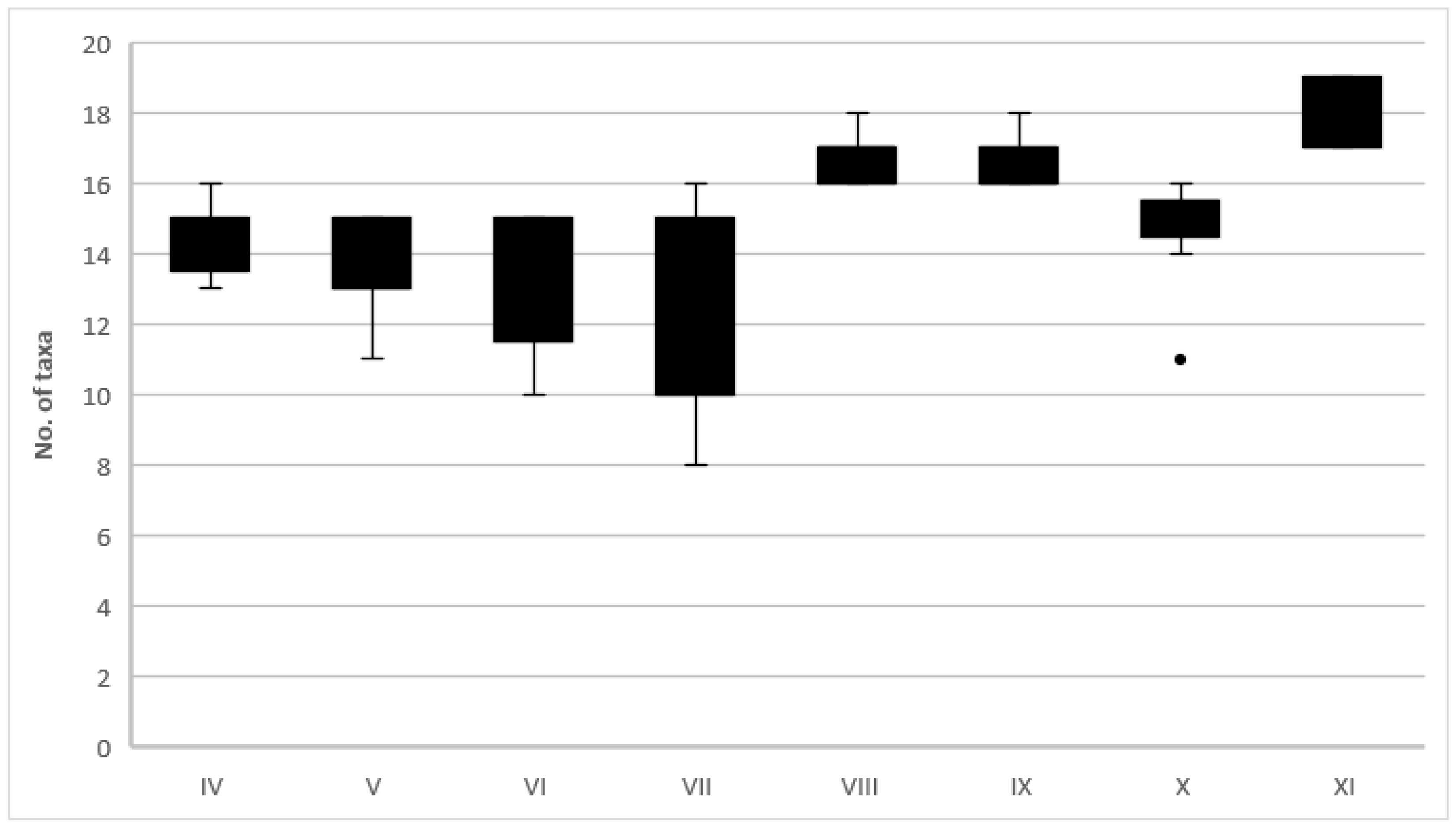
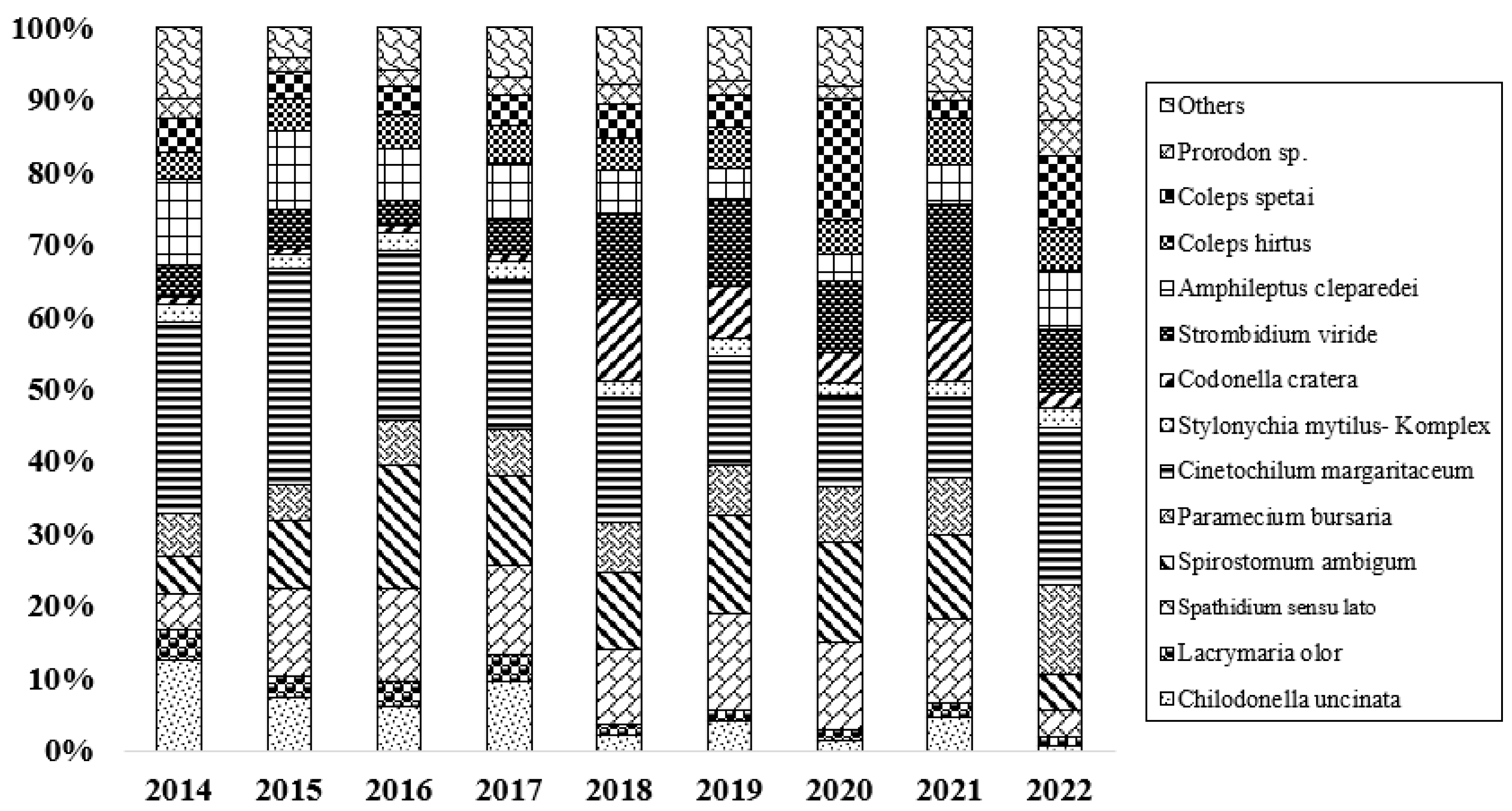
3.2. Microbial Communities
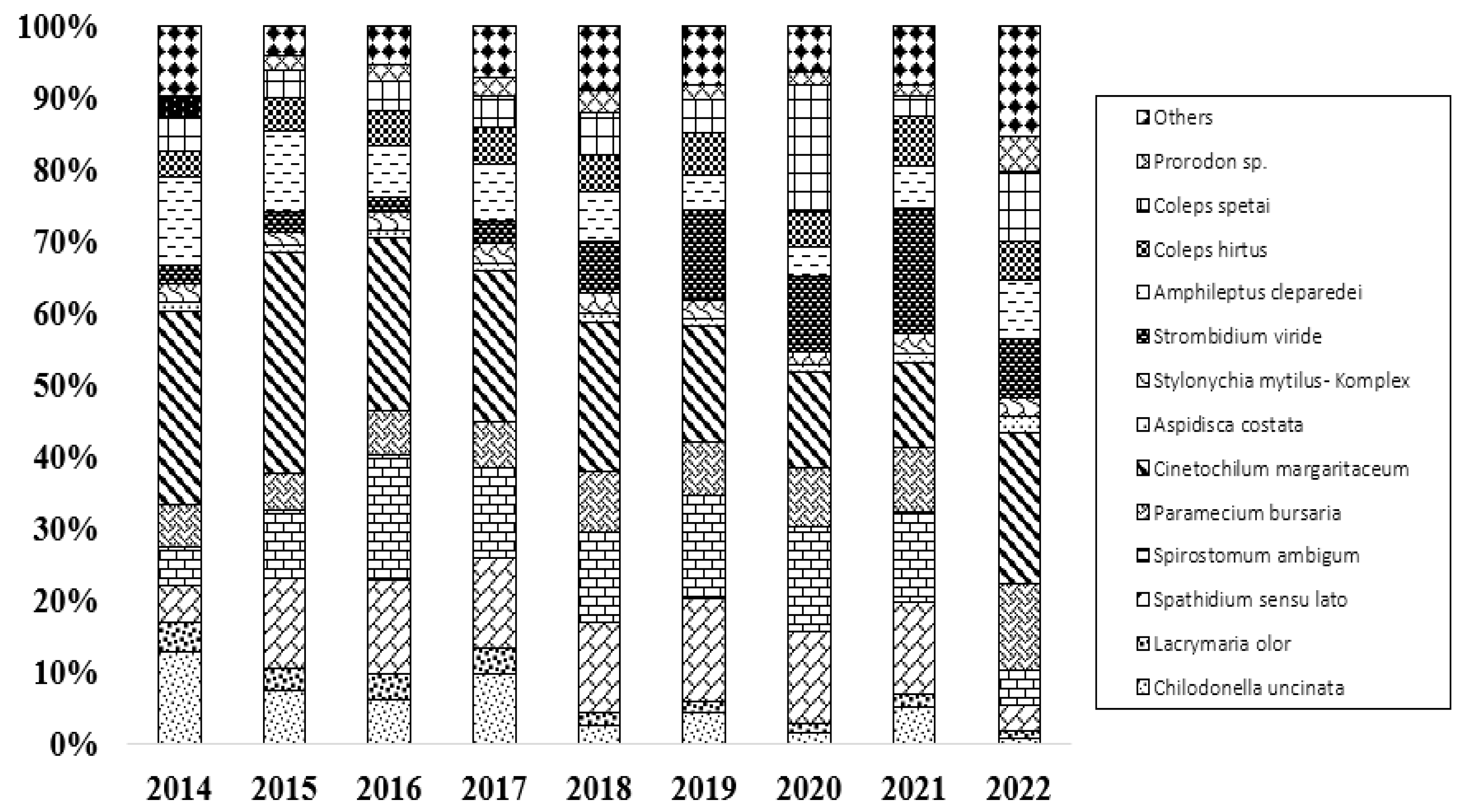
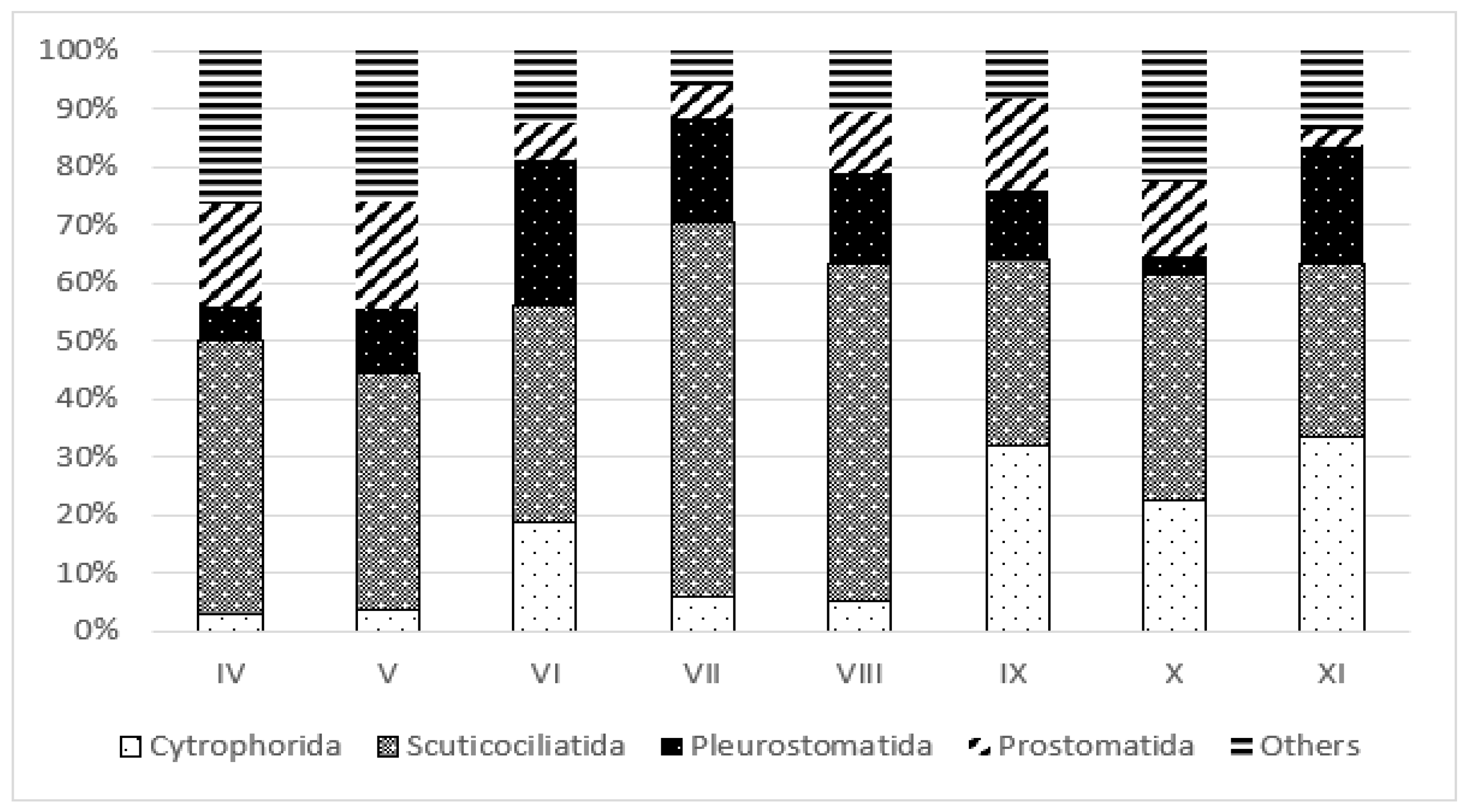
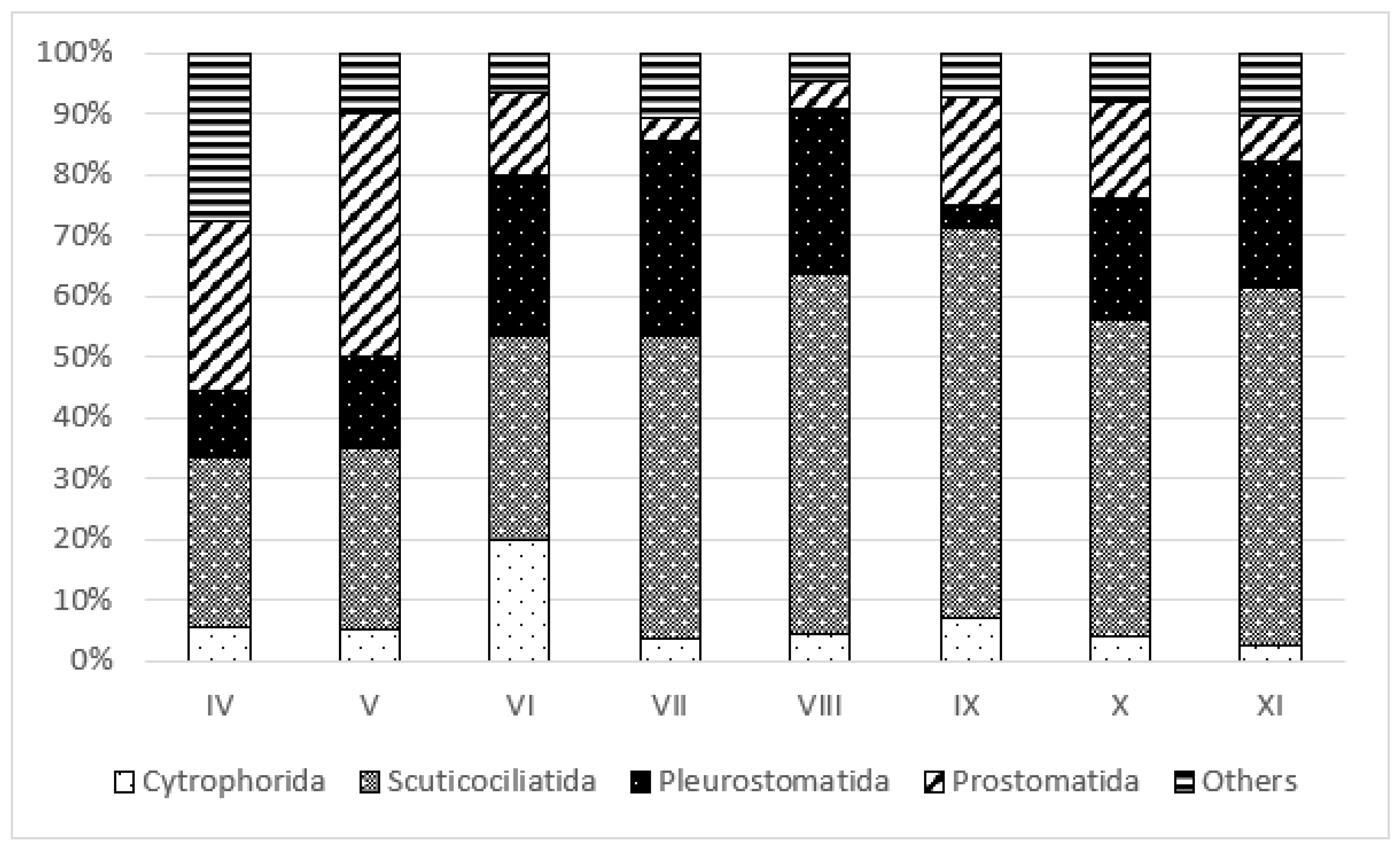
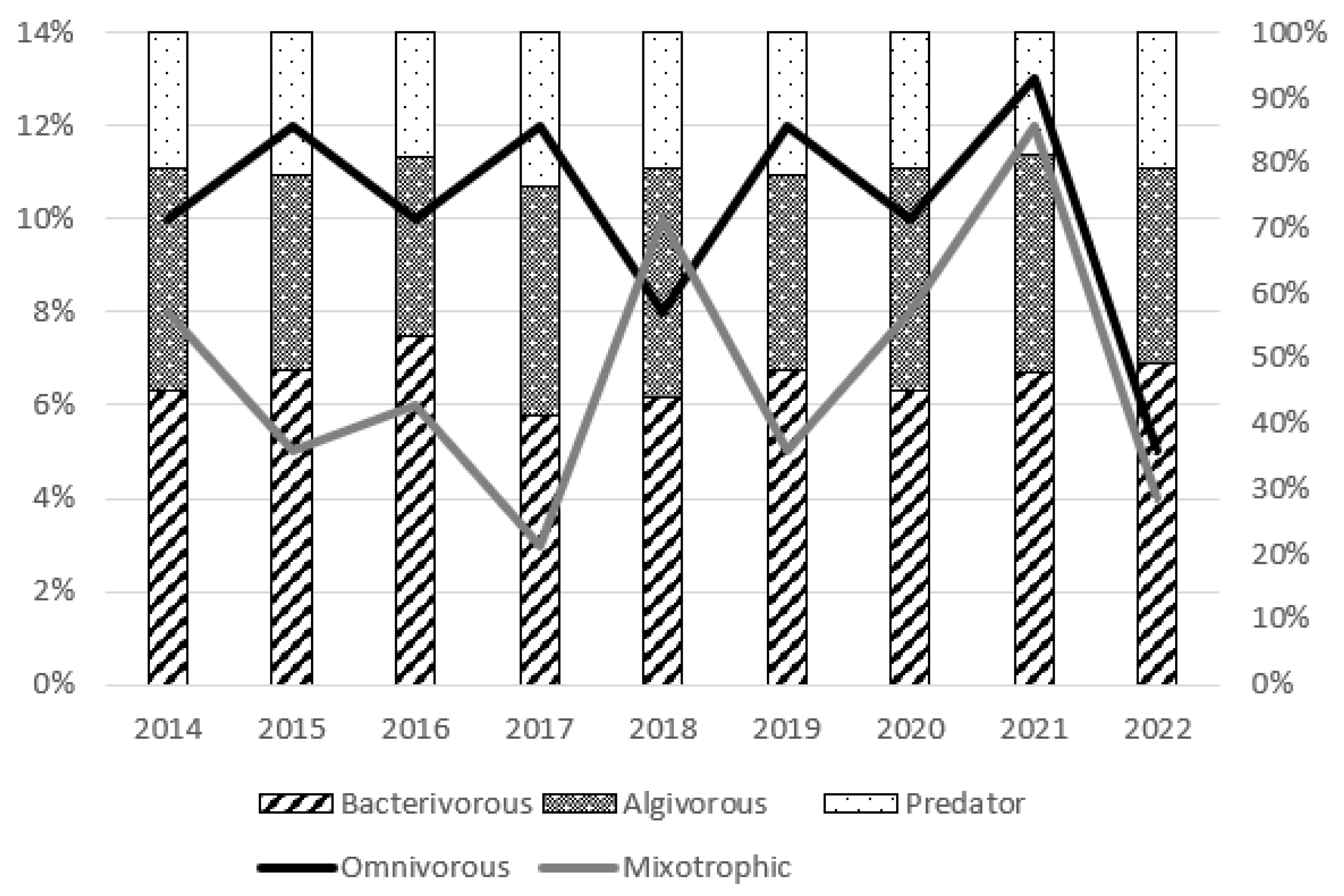
3.3. Trophic Structure
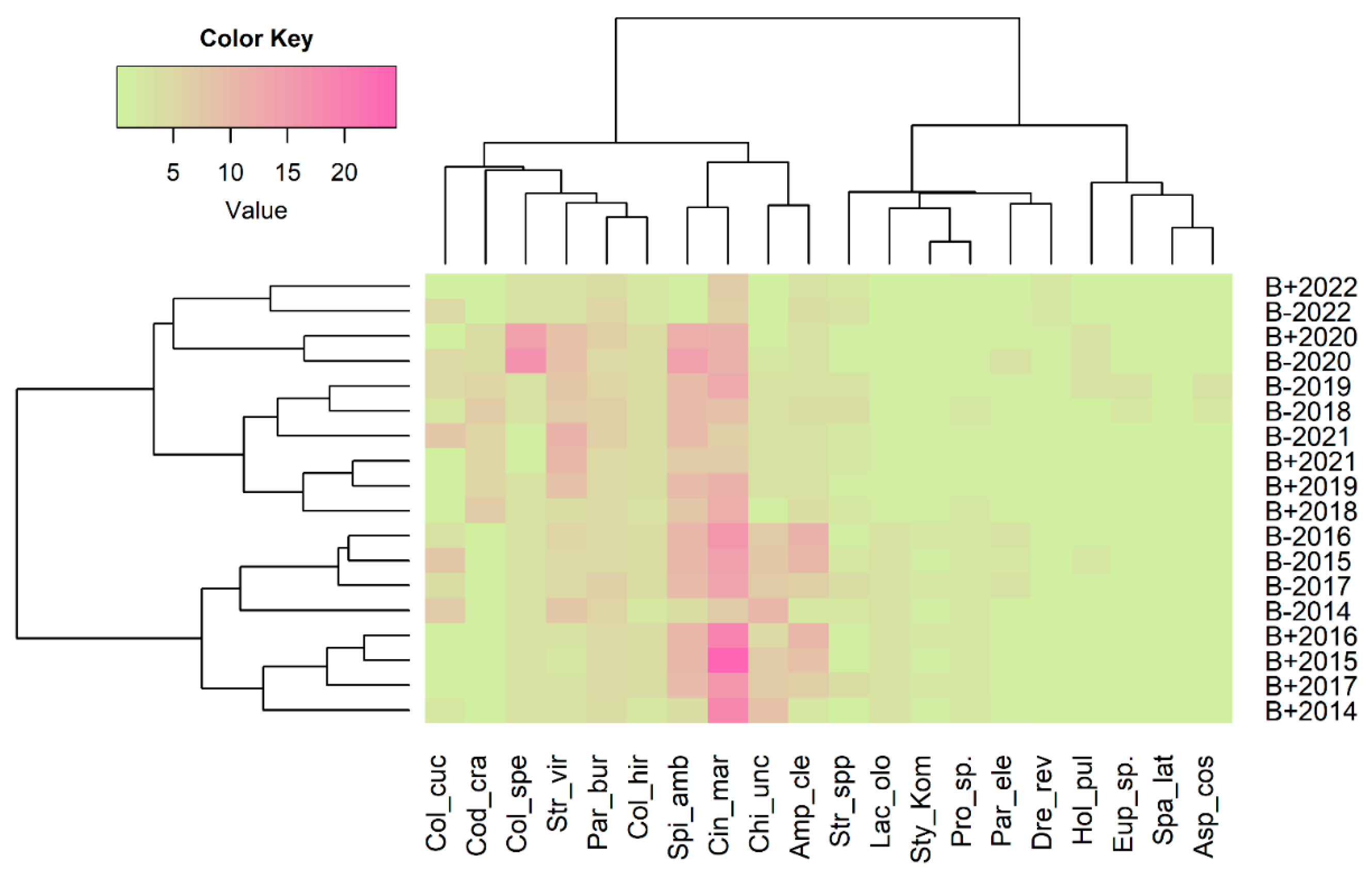
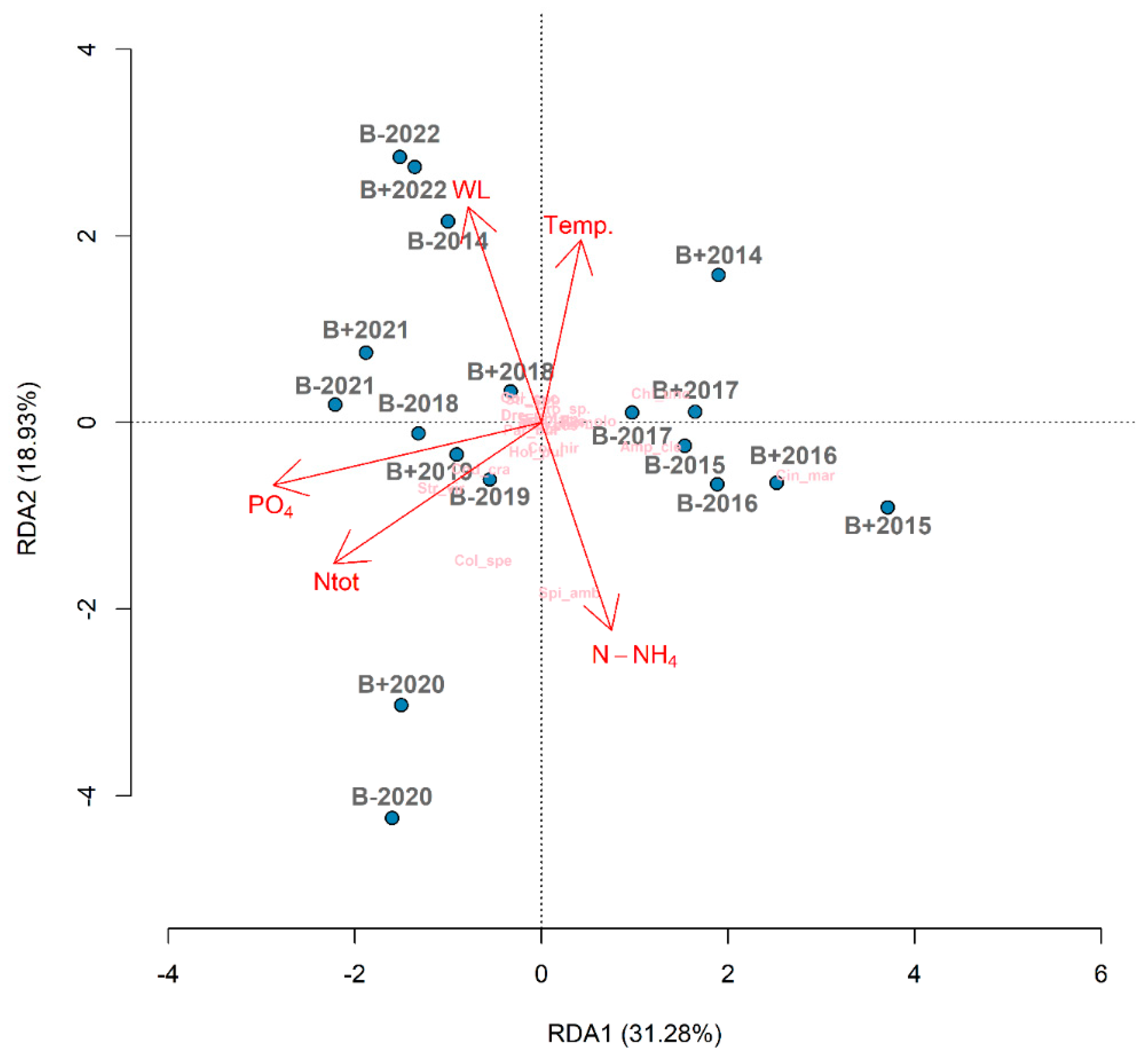
3.4. Redundancy Analysis (RDA) and Correlations
4. Discussion
4.1. Effect of Restoration on Environmental Factors
4.2. Effect of Restoration on the Composition and Abundance of Ciliates
4.3. Effect of Restoration on Functional Groups of Ciliates
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Yu, Z.; Beilman, D.W.; Jones, M.C. Sensitivity of northernpeatland carbon dynamics to Holocene climate change, in CarbonCycling in Northern Peatlands. Geophys. Monogr. Ser. 2009, 184, 55–69. [Google Scholar]
- Oleszczuk, R. Wielkość Emisji Gazów Cieplarnianych i Sposoby Jej Ograniczania z Torfowisk Użytkowanych Rolniczo. Wybrane Problemy Ochrony Mokradeł. Współczesne Problemy Kształtowania i Ochrony Środowiska, Monografie 3. 2012. Available online: https://www.uwm.edu.pl/environ/vol03p/vol_03p_rozdzial04.pdf (accessed on 2 September 2023).
- Bardgett, R.D.; Wardle, D.A. Aboveground-Belowground Linkages. Biotic Interactions, Ecosystem Processes, and Global Change; Oxford University Press: England, UK, 2010; pp. 5–310. [Google Scholar]
- Headley, A.D.; Wheeler, B.D.; Baker, A.J.M. The impact of man on vegetation of Crymlyn bog. Impact Assess. 1992, 11, 257–261. [Google Scholar]
- Dorrepaal, E.; Aerts, R.; Cornelissen, J.H.C.; Van Logtestijn, R.S.P.; Callaghan, T.V. Sphagnum modifies climate-change impacts on subarctic vascular bog plants. Funct. Ecol. 2016, 20, 31–41. [Google Scholar] [CrossRef]
- Willis, K.J.; Whittaker, R.J. Species Diversity Scale Matters. Science 2002, 295, 1245–1248. [Google Scholar] [CrossRef] [PubMed]
- Pawlaczyk, P. Akumulacja i Emisja Węgla Przez Torfowiska, w Tym Przez Torfowiska Alkaliczne; Klub Przyrodników: Świebodzin, Poland, 2014; pp. 3–33. [Google Scholar]
- Kruk, M. Biogeochemical multifunctionality of wetland ecotones in Lakeland agricultural landscape. Pol. J. Ecol. 2003, 2, 247–254. [Google Scholar]
- Lamentowicz, M.; Mueller, M.; Gałka, M.; Barabach, J.; Milecka, K.; Goslar, T.; Binkowski, M. Reconstructing human impact on peatland development during the past 200 years in CE Europe through biotic proxies and X-ray tomography. Quat. Int. 2015, 357, 282–294. [Google Scholar] [CrossRef]
- Golterman, H.L. Methods for Chemical Analysis of Freshwaters; Blackwell Scientific Publications: Oxford, UK; Edinburgh, Scotland, 1969; pp. 1–172. [Google Scholar]
- Basińska, M.; Reczuga, M.K.; Gąbka, M.; Stróżecki, M.; Luców, D.; Samson, M.; Urbaniak, M.; Leśny, J.; Chojnicki, B.H.; Gilbert, D.; et al. Experimental warming, and precipitation reduction affect the biomass of microbial communities in Sphagnum peatland. Ecol. Indic. 2020, 112, 106059. [Google Scholar] [CrossRef]
- Lamentowicz, M.; Gałka, M.; Milecka, K.; Tobolski, K.; Lamentowicz, T.; Fiałkiewicz-Kozieł, B.; Blaauw, M. A 1300 years multi-proxy, high-resolution record from a rich fen in northern Poland: Reconstructing hydrology, land-use and climate change. J. Quarternary Sci. 2013, 28, 582–594. [Google Scholar] [CrossRef]
- Poulin, M.; Andersen, R.; Rochefort, L. New Approach for Tracking Vegetation Change after Restoration: A Case Study with Peatlands. Ecol. Restor. 2012, 3, 363–371. [Google Scholar] [CrossRef]
- De Jong, R.; Blaauw, M.; Chambers, F.M.; Christensen, T.R.; de Vleeschouwer, F.; Finsinger, W.; Fronzek, S.; Johansson, M.; Kokfelt, U.; Lamentowicz, M.; et al. Climate and peatlands. In Changing Climates, Earth Systems and Society; Springer: Dordrecht, The Netherlands, 2010; pp. 85–121. [Google Scholar]
- Bayley, S.; Thormann, M.N.; Szumigalski, A.R. Nitrogen mineralization and decomposition in western boreal bog and fen peat. Ecoscience 2005, 12, 455–465. [Google Scholar] [CrossRef]
- Mitchell, E.A.D.; Gilbert, D.; Amblard, C.; Grosverinier, P.; Gobat, J.M. Structure of microbial communities in Sphagnum peatlands and effect of atmospheric carbon dioxide enrichment. Microb. Ecol. 2003, 46, 187–199. [Google Scholar] [CrossRef] [PubMed]
- Kexin, Z.; Muqi, X.; Biao, L.; Hong, C. Characteristics of microfauna and their relationships with the performance of an activated sludge plant in China. J. Environ. Sci. 2007, 20, 482–486. [Google Scholar]
- Nguyen-Viet, H.; Gilbert, D.; Mitchell, E.A.D.; Badot, P.M.; Bernard, N. Effects of experimental lead pollution on the microbial com-munities associated with Sphagnum fallax (Bryophyta). Microb. Ecol. 2007, 54, 232–241. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, D.M.; Mitchell, E.A.D. Testate amoebae and nutrientcycling with particular reference to soils. Geomicrobiol. J. 2010, 27, 520–533. [Google Scholar] [CrossRef]
- Mieczan, T.; Tarkowska-Kukuryk, M.; Bielańska-Grajner, I. Hydrochemical and microbiological distinction and function of ombrotrophic peatland lagg as ecotone between Sphagnum peatland and forest catchmetn (Poleski National Park, eastern Poland). Ann. Limnol. Int. J. Limnol. 2021, 48, 323–336. [Google Scholar] [CrossRef]
- Payne, R.J. Can testate amoeba-based palaeohydrology be extended to fens? J. Quat. Sci. 2011, 26, 15–27. [Google Scholar] [CrossRef]
- Lamentowicz, M.; Mitchell, E.A. The ecology of Testate amoebae (Protists) in Sphagnum in north-western Poland in relation to peatland ecology. Microb. Ecol. 2005, 50, 48–63. [Google Scholar] [CrossRef]
- Mieczan, T. Species diversity of protozoa (Rhizopoda, Ciliata) on mosses of Sphagnum genus in restoration area of the Poleski National Park. Acta Agroph. 2006, 7, 453–459. [Google Scholar]
- Mieczan, T. Ciliates in Sphagnum peatlands:vertical micro-distribution, and relationships of species assemblages with environmental parameters. Zool. Stud. 2009, 48, 33–48. [Google Scholar]
- Mieczan, T.; Adamczuk, M.; Pogorzelec, M. Ciliates as restoration indicators in peatbogs—10 years of study. Europ. J. Protis. 2018, 62, 11–23. [Google Scholar] [CrossRef]
- Mieczan, T.; Tarkowska-Kukuryk, M. Microbial Communities as Environmental Indicators of Ecological Disturbance in Restored Carbonate Fen-Results of 10 Years of Studies. Microb. Ecol. 2017, 74, 384–401. [Google Scholar] [CrossRef] [PubMed]
- Utermöhl, H. Zur vervollkommung der quantative phytoplankton methodic. Mitt. Internat Ver. Limnol 1985, 9, 1–38. [Google Scholar]
- Foissner, W.; Berger, H. A user-friendly guide to the ciliates (Protozoa, Ciliophora) commonly used by hydrobiologists as bioindicators in rivers, lakes and waste waters, with notes on their ecology. Freshw. Biol. 1996, 35, 375–470. [Google Scholar] [CrossRef]
- Foissner, W.; Berger, H.; Schaumburg, J. Identification and Ecology of Limnetic Plankton Ciliates; Informationsberichte des Bayer. Landesamtes für Wasserwirtschaft: München, Germany, 1999; pp. 1–793. [Google Scholar]
- Kotlarz, J.; Kacprzak, M. Species diversity of forest stands estimation algorithm using RGB images of the tree crowns. Meas. Robot. Autom. 2017, 223, 59–66. [Google Scholar] [CrossRef]
- Ter Braak, C.J.F. (1988–1992) CANOCO–FORTRAN Program for Canonical Community Ordination, Version 2.1; Microcomputer Power: Ithaca, NY, USA, 1987. [Google Scholar]
- Andersen, R.; Wells, C.; Macrae, M.; Price, J. Nutrient mineralisation and microbial functional diversity in a restored bog approach natural conditions 10 years post restoration. Soil Biol. Biochem. 2013, 64, 37–47. [Google Scholar] [CrossRef]
- Glińska-Lewczuk, K.; Burandt, P.; Łaźniewska, I.; Łaźniewski, J.; Menderski, S.; Pisarek, W. Ochrona I Renaturyzacja Torfowisk Wysokich w Rezerwatach Gązwa, Zielony Mechacz i Sołtysek w Północno-Wschodniej Polsce; Wydawnictwo Polskiego Towarzystwa Ochrony Ptaków: Białowieża, Poland, 2014; pp. 1–122. [Google Scholar]
- Grzywna, A. Peatland surface subsidence in drainage grassland Polesie Lubelskie. Acta Sci. Pol. 2016, 15, 81–89. [Google Scholar]
- Słowińska, S.; Słowiński, M. Torfowisk mszarne strefy północnej a zmiana klimatu. Kosmos 2021, 70, 569–578. [Google Scholar] [CrossRef] [PubMed]
- Degroot, D.; Anchukaitis, K.; Bauch, M.; Burnham, J.; Carnegy, F.; Cui, J.; de Luna, K.; Guzowski, P.; Hambrecht, G.; Huhtamaa, H.; et al. Towards a rigorous understanding of societal responses to climate change. Nature 2021, 591, 539–550. [Google Scholar] [CrossRef]
- Gilbert, D.; Amblard, C.; Bourdier, G.; Francez, A.J. The microbial loop at the surface of a peatland: Structure, functioning and impact of nutrients inputs. Microb. Ecol. 1998, 35, 89–93. [Google Scholar] [CrossRef]
- Scheffer, M. Ecology of Shallow Lakes; Kulwer Academic Publishers: Dordrecht, The Netherlands, 2004. [Google Scholar]
- Wetzel, R.G. Limnology: Lake & River Ecosystems; Academic Press: Cambridge, MA, USA, 2001; p. 429. [Google Scholar]
- Ricklefs, R.E.; Miller, G.L. Ecology, 4th ed.; W.H. Freeman and Company: New York, NY, USA, 1999. [Google Scholar]
- Burkholder, J.M. Eutrophication and Oligotrophication. In Encyclopedia of Biodiversity; Levin, S.A., Ed.; Academic Press: San Diego, CA, USA, 2001; pp. 649–670. [Google Scholar]
- Wassen, M.J.; Venterink, H.O.; Lapshina, E.D.; Tanneberger, F. Endangered plants persist under phosphorus limitation. Nature 2005, 437, 547–550. [Google Scholar] [CrossRef]
- Bauer, D.C.; Gómez, N.; Hualde, P.D. Biofilms coating Schoenoplectus californicus as indicators of water quality in the Río de la Plata Estuary (Argentina). Environ. Monit. Assess. 2007, 133, 309–320. [Google Scholar] [CrossRef] [PubMed]
- Samuelsson, J.; Berglund, A.; Anderson, A. Factors structuring the heterotrophic flagellate and ciliate community along a brackish water primary production gradient. J. Plankton Res. 2006, 28, 345–359. [Google Scholar] [CrossRef]
- Gong, J.; Song, W.; Warren, A. Periphytic ciliate colonization: Annual cycle and responses to environmental conditions. Aquatic Microb. Ecol. 2005, 39, 159–170. [Google Scholar] [CrossRef]
- Jassey, V.E.J.; Chiapusio, G.; Mitchell, E.A.D.; Binet, P.; Toussaint, M.-L.; Gilbert, D. Fine-scale horizontal and vertical micro-distribution patterns of testate amoebae along a narrow fen/bog gradient. Microb. Ecol. 2011, 61, 374–385. [Google Scholar] [CrossRef] [PubMed]
- Kuczyńska-Kippen, N. On the body size and habitat selection of rotifers in the macrophyte-dominated lake Budzyńskie in Poland. Aquatic Ecol. 2005, 39, 447–454. [Google Scholar]
- Weisse, T.; Montagnes, D.J.S. Ecology of planktonic ciliates in a changing world: Concepts, methods, and challenges. J. Eukaryot. Microbiol. 2022, 69, 1–37. [Google Scholar] [CrossRef] [PubMed]
- Ju¨rgens, K.; Pernthaler, J.; Schalla, J.; Amann, R. Morphological and Compositional Changes in a Planktonic Bacterial Community in Response to Enhanced Protozoan Grazing. Appl. Environ. Microbiol. 1999, 65, 241–1250. [Google Scholar]
- Mieczan, T.; Tarkowska-Kukuryk, M.; Adamczuk, M.; Pęczuła, W.; Demetraki-Paleolog, A.; Niedźwiecki, M. Research of different types of peatbogs: Relationschips of biocenosis structures and physico-chemical parameters. Pol. J. Environ. Stud. 2015, 1, 191–198. [Google Scholar]
- Pereira, D.G.; da Silva, B.S.T.; Camargo, J.C.; Velho, L.F.M.; Pauleto, G.M.; Lansac-Tôha, F.A. Effects of eutrophication on flagellates associated with Eichhornia crassipes: An experimental approach. Int. Rev. Hydrobiol. 2010, 95, 72–85. [Google Scholar] [CrossRef]
- Buosi, P.R.B.; Pauleto, G.M.; Lansac-Toha, F.A.; Velho, L.F.M. Ciliate community associated with aquatic macrophyte roots: Effects of nutrient enrichment on the community composition and species richness. Eur. J. Protisol. 2011, 47, 86–102. [Google Scholar] [CrossRef]
- Finlay, B.J. Effects of Seasonal Anoxia on the Community of Benthic Ciliated Protozoa in a Productive Lake. Arch. Protistenkd. 1982, 125, 215–222. [Google Scholar] [CrossRef]
- Mieczan, T. Vertical micro-zonation of testate amoebae and ciliates in peatland waters in relation to potential food resources and grazing pressure. Int. Rev. Hydrobiol. 2010, 95, 86–102. [Google Scholar] [CrossRef]
- Mieczan, T. Seasonal patterns of testate amoebae and ciliates in three peatbogs:relationship to bacteria and flagellates (Poleski National Park, Eastern Poland). Ecohydrol. Hydrobiol. 2007, 7, 79–88. [Google Scholar] [CrossRef]
- Ju, L.; Yang, J.; Lui, L.; Wilkinson, D.M. Diversity and distribu-tion of freshwater testate amoebae (Protozoa) among latitudinal andtrophic gradients in China. Microb. Ecol. 2004, 4, 657–670. [Google Scholar]
- Chróst, J.R.; Siuda, I. Microbial production, utilization, and enzymatic degradation of organic matter in the upper trophogenic layer in the pelagial zone of lakes along a eutrophication gradient. Limnol. Oceanogr. 2006, 51, 749–762. [Google Scholar] [CrossRef]
- Lukić, D.; Limberger, R.; Agatha, S.; Montagnes, D.J.; Weisse, T. LETTER Thermal performance of planktonic ciliates differs between marine and freshwaters: A case study providing guidance for climate change studies Scientific Significance Statement. Limnol. Oceanogr. 2022, 7, 520–526. [Google Scholar] [CrossRef]
- Boechat, I.G.; Adrian, R. Biochemical composition of algivorous freshwater ciliates: You are not what you eat. Microb. Ecol. 2005, 53, 393–400. [Google Scholar] [CrossRef] [PubMed]
- Weisse, T.; Anderson, R.; Arndt, H.; Calbet, A.; Hansen, P.J.; Montagnes, D.J.S. Functional ecology of aquatic phagotrophic protists—Concepts, limitations, and perspectives. Eur. J. Antistol. 2016, 55, 50–74. [Google Scholar] [CrossRef]
- Weisse, T. Freshwater ciliates as ecophysiological model organisms—Lessons from Daphnia, major achievements, and future perspectives. Arch. Hydrobiol. 2006, 167, 371–402. [Google Scholar] [CrossRef]
- Posch, T.; Eugster, B.; Pomati, F.; Pernthaler, J.; Pitsch, G.; Eckert, E.M. Network of interactions between ciliates and phytoplankton during spring. Front. Microbiol. 2015, 6, 1289. [Google Scholar] [CrossRef]
- Mitra, A.; Flynn, K.J.; Tillmann, U.; Raven, J.A.; Caron, D.; Stoecker, D.K.; Not, F.; Hansen, P.J.; Hallegraeff, G.; Sanders, R.; et al. Defining planktonic protist functional groups on mechanisms for energy and nutrient acquisition: Incorporation of diverse mixotrophic strategies. Protist 2016, 167, 106–120. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Yang, M.; Qi, R.; An, W.; Zhou, J. Comparative study of protozoan communities in full-scale MWTPs in Beijing related to treatment processes. Wat. Res. 2008, 42, 1907–1918. [Google Scholar] [CrossRef] [PubMed]
- Madoni, P. Protozoa in wastewater treatment processes: A minireview Ital. J. Zool. 2011, 78, 3–11. [Google Scholar] [CrossRef]
- Wu, S.; Carvalho, P.N.; Müller, J.A.; Manoj, V.R.; Dong, R. Sanitation in constructed wetlands: A review on the removal of human pathogens and fecal indicators. Sci. Total Environ. 2016, 541, 8–22. [Google Scholar] [CrossRef] [PubMed]
- Amblard, C.; Carrias, J.; Bourdier, F.; Maurin, N. The microbialloop in a humic lake: Seasonal and vertical variations in the structureof different communities. Hydrobiol 1995, 300, 71–84. [Google Scholar] [CrossRef]
- Gaiser, E.E.; Texler, J.C.; Richards, J.H.; Childers, D.L.; Lee, D.; Edwards, A.L.; Scinto, L.J.; Jayachandaran, K.; Noe, G.B.; Jones, R.D. Cascading ecological effects of low-level phosphorus enrichment in the Florida everglades. J. Environ. Qual. 2005, 34, 717–723. [Google Scholar] [CrossRef] [PubMed]
- Johansson, M.; Gorokhova, E.; Larsson, U. Annual variability in ciliate community structure, potential prey and predators in the open northern Baltic Sea proper. J. Plankton Res. 2004, 26, 67–80. [Google Scholar] [CrossRef]
- Stoecker, D.K.; Cucci, T.L.; Hulburt, E.M.; Yentsch, C.M. Selective feeding by Balanion sp. (Ciliata: Balanionidae) on phytoplankton that best support its growth. J. Exp. Mar. Biol. Ecol. 1986, 95, 113–130. [Google Scholar] [CrossRef]
- Ju, R.T.; Wang, F.; Li, B. Effects of temperature on the development and population growth of the sycamore lace bug, Corythucha ciliata. J. Insect Sci. 2011, 11, 16. [Google Scholar] [CrossRef]
- Keeling, P.J. The endosymbiotic origin, diversification and fate of plastids. Philos. Trans. R. Soc. B Biol. Sci. 2010, 365, 729–748. [Google Scholar] [CrossRef]
- Mondragón-Camarillo, L.; Zaragoza, S.R. Food Webs. In Plankton Communities; BoD-Books on Demand: Norderstedt, Germany, 2022; Volume 1, pp. 1–24. [Google Scholar]
- Stoecker, D.K.; Michaels, A.E. Respiration, photosynthesis and carbon metabolism in plank-tonic ciliates. Mar. Biol. 1991, 108, 44–447. [Google Scholar] [CrossRef]
- Genoveva, F.; Fenchel, T.; Finlay, J. Mixotrophy in Ciliates. Protist 2010, 161, 621–641. [Google Scholar]
- Mieczan, T. Kształtowanie się struktury jakościowej i ilościowej orzęsków peryfitonowych w zbiornikach zapadliskowych na Polesiu Lubelskim. Acta Agrophysica Część II 2002, 67, 181–188. [Google Scholar]
- Stoecker, W.F. Design of Thermal Systems, 3rd ed.; McGraw-Hill: New York, NY, USA, 1989; pp. 1–561. [Google Scholar]
- Dolan, J.R. Doing a Protist Book. Protist 2013, 164, 154–159. [Google Scholar] [CrossRef]



| Size | B− | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Parameters/years | 2014 | 2015 | 2016 | 2017 | 2018 | 2019 | 2020 | 2021 | 2022 |
| Water level (cm) | 4–25 | 11–24 | 11–23 | 14–27 | 14–27 | 14–27 | 2–23 | 2–15 | 34–56 |
| Temp. °C | 4–22 | 2–21 | 2–17 | 4–16 | 3–19 | 4–16 | 3–23 | 3–20 | 6–21 |
| pH | 7–9.6 | 7–9.8 | 8–9.8 | 8–9.8 | 7–9.8 | 7–9 | 6–9.8 | 6–9.2 | 7–9.2 |
| Cond. (μS cm−1) | 180–321 | 78–278 | 78–278 | 98–278 | 98–278 | 98–232 | 98–340 | 320–498 | 202–343 |
| O2 (mg L−1) | 5–9 | 5–9 | 7–9 | 8–11 | 7–11 | 7–11 | 7–11 | 8–11 | 3.4–9.1 |
| NH4+ (mg L−1) | 0.124–0.970 | 0.124–0.970 | 0.124–0.890 | 0.124–0.970 | 0.124–0.970 | 0.124–0.894 | 0.124–0.970 | 0.124–0.970 | 0.022–0.343 |
| NO3− (mg L−1) | 0.112–0.470 | 0.112–0.470 | 0.114–0.431 | 0.157–0.431 | 0.14–0.431 | 0.157–0.431 | 0.114–0.431 | 0.114–0.431 | 0.023–0.343 |
| Ntot (mg L−1) | 1.456–1.852 | 1.456–1.952 | 1.456–1.884 | 1.456–1.884 | 1.456–1.884 | 1.466–1.884 | 1.456–3.456 | 1.466–3.456 | 1.267–1.884 |
| PO43− (mg L−1) | 0.021–0.112 | 0.021–0.132 | 0.011–0.169 | 0.011–0.169 | 0.011–0.169 | 0.011–0.169 | 0.011–0.169 | 0.011–0.169 | 0–0.102 |
| Ptot (mg L−1) | 0.136–0.636 | 0.082–0.636 | 0.023–0.346 | 0.023–0.346 | 0.023–0.3460 | 0.023–0.346 | 0.090–0.346 | 0.090–0.346 | 0.029–1.087 |
| Chlorophyll a (mg L−1) | 9–27 | 5–37 | 2–19.04 | 2–19.04 | 2–19.04 | 2–19.04 | 2–23.04 | 2–23.04 | 5.93–110.23 |
| TOC (mg L−1) | 15.9–37 | 15.9–37 | 18.5–34 | 18.3–34 | 18.3–34 | 18.5–34 | 18.3–34 | 18.5–36.2 | 17.8–27.5 |
| Site | B+ | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Parameters/years | 2014 | 2015 | 2016 | 2017 | 2018 | 2019 | 2020 | 2021 | 2022 |
| Water level (cm) | 6–13 | 8–23 | 11–23 | 14–27 | 14–27 | 23–43 | 4–45 | 12–19 | 34–46 |
| Temp. °C | 3–23 | 2–21 | 2–16 | 4–16 | 3–14 | 4–16 | 3–24 | 3–24 | 6–20 |
| pH | 6–9 | 7–9 | 8–9 | 8–9.8 | 7–9.8 | 8–14 | 8–9.8 | 6–9.2 | 3–9 |
| Cond. (μS cm−1) | 180–321 | 180–321 | 78–278 | 98–278 | 98–278 | 98–230 | 98–332 | 280–390 | 225–314 |
| O2 (mg L−1) | 5–8 | 4–12 | 7–9 | 8–11 | 7–11 | 7–11 | 7–11 | 8–11 | 4.3–7.9 |
| NH4+ (mg L−1) | 0.124–0.894 | 0.124–0.894 | 0.124–0.923 | 0.124–0.970 | 0.124–0.970 | 0.124–0.894 | 0.124–0.970 | 0.124–0.970 | 0.011–0.778 |
| NO3− (mg L−1) | 0.112–0.470 | 0.028–0.314 | 0.015–0.161 | 0.112–0.431 | 0.112–0.431 | 0.157–0.431 | 0.114–0.431 | 0.114–0.431 | 0.018–0.300 |
| Ntot (mg L−1) | 1.466–1.884 | 1.466–1.884 | 1.466–1.884 | 1.456–1.884 | 1.456–1.884 | 1.466–1.884 | 1.466–3.456 | 1.466–3.456 | 1.531–2.456 |
| PO43− (mg L−1) | 0.021–0.112 | 0.001–0.033 | 0.003–0.029 | 0.011–0.169 | 0.011–0.169 | 0.011–0.169 | 0.011–0.169 | 0.011–0.169 | 0.01–0.112 |
| Ptot (mg L−1) | 0.136–0.636 | 0.005–0.211 | 0011–0.522 | 0.023–0.346 | 0.023–0.346 | 0.023–0.346 | 0.090–0.346 | 0.090–0.346 | 0.010–0.260 |
| Chlorophyll a (mg L−1) | 9–27 | 9–27 | 2.3–8 | 2–16.04 | 2–19.04 | 2–19.04 | 2–23.04 | 2–23.04 | 4.76–23.59 |
| TOC (mg L−1) | 15.9–37 | 13.2–24 | 13.2–21.9 | 18.3–34 | 18.3–224.0 | 18.3–34 | 18.3–34 | 18.5–36.5 | 15.6–26.2 |
| Kruskal–Wallis H | Statistical Significance (p) | |||
|---|---|---|---|---|
| B− | B+ | B− | B+ | |
| WL | 7.95 | 4.82 | 0.33 | 0.68 |
| pH | 14.08 | 40.81 | 0 | 0 |
| N-NH4 | 41.26 | 40.81 | 0 | 0 |
| N-NO3 | 36.41 | 19.62 | 0 | 0 |
| Ntot | 44.16 | 36.33 | 0 | 0 |
| P-PO4 | 26.26 | 23.5 | 0 | 0.01 |
| Ptot | 41.83 | 18.76 | 0 | 0 |
| Chl a | 2.2 | 21.17 | 0.01 | 0 |
| TOC | 26.94 | 18.42 | 0 | 0.01 |
| Cond. | 20.32 | 8.47 | 0 | 0.29 |
| T | 47.52 | 43.47 | 0 | 0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mieczan, T.; Bartkowska, A.; Bronowicka-Mielniczuk, U.; Rudyk-Leuska, N. The Effect of Peatland Restoration on Ciliate Communities: Long-Term Analyses. Water 2023, 15, 3793. https://doi.org/10.3390/w15213793
Mieczan T, Bartkowska A, Bronowicka-Mielniczuk U, Rudyk-Leuska N. The Effect of Peatland Restoration on Ciliate Communities: Long-Term Analyses. Water. 2023; 15(21):3793. https://doi.org/10.3390/w15213793
Chicago/Turabian StyleMieczan, Tomasz, Aleksandra Bartkowska, Urszula Bronowicka-Mielniczuk, and Natalia Rudyk-Leuska. 2023. "The Effect of Peatland Restoration on Ciliate Communities: Long-Term Analyses" Water 15, no. 21: 3793. https://doi.org/10.3390/w15213793





