Hydrochemical Characterization and Quality Assessment of Groundwater in the Southern Plain of Hebei Province, China
Abstract
:1. Introduction
- (1)
- In this paper, ArcGIS spatial analysis is combined with the hydrochemical graphic method to systematically reveal the spatial distribution characteristics of groundwater hydrochemistry, which effectively makes up for the deficiency in the hydrochemical graphic method in studying spatial scale.
- (2)
- A clustering analysis was introduced to reveal the source similarity of ions and its influence relationship.
- (3)
- The correlation analysis and entropy weight water quality index were combined to identify and optimize the main impact indicators of water quality, and further reduce the cost of water quality monitoring.
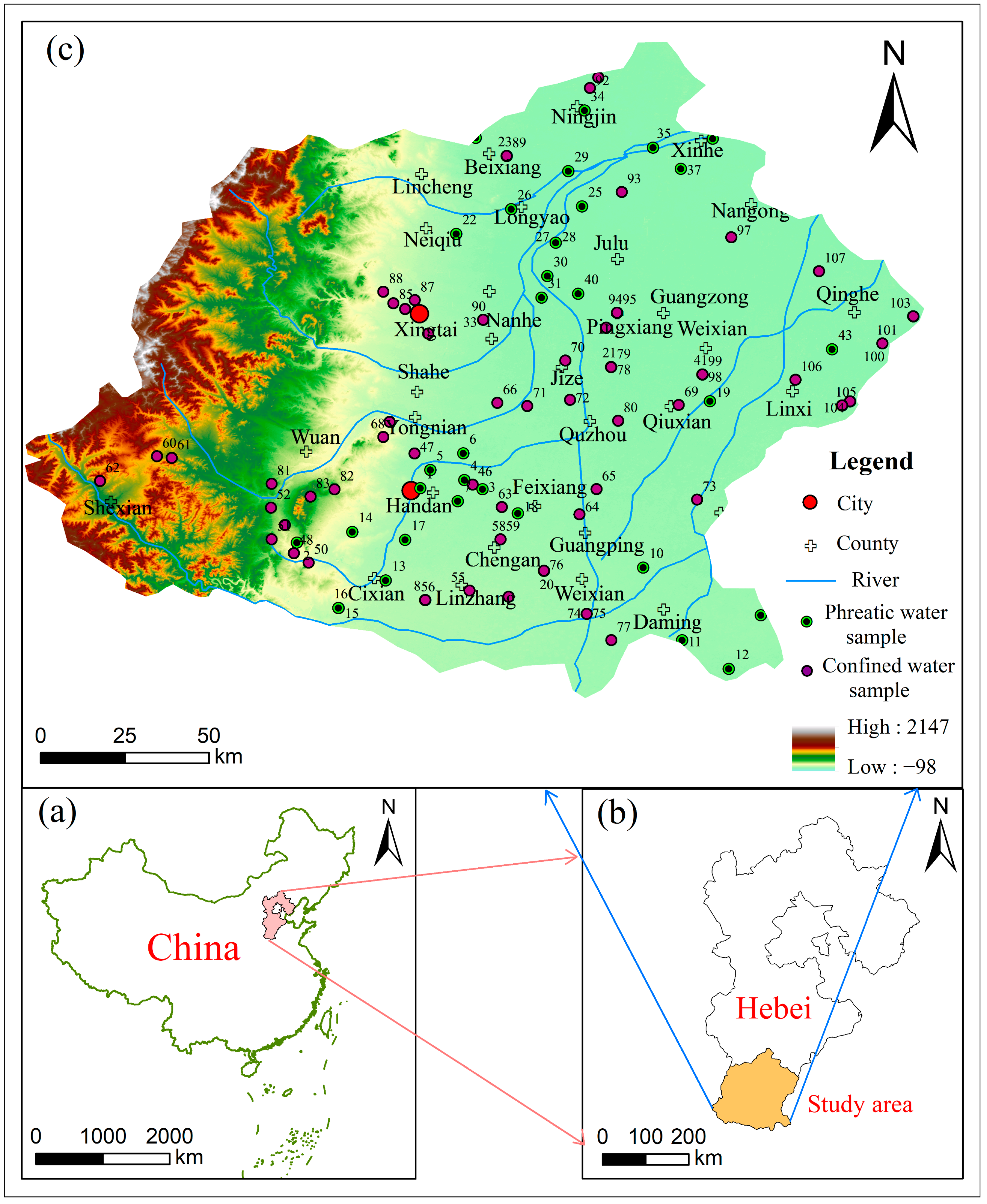
2. Study Area
3. Materials and Methods
3.1. Sample Collection and Analysis
3.2. Hydrogeochemical Modeling
3.3. Entropy Weighted Water Quality Index (EWQI)
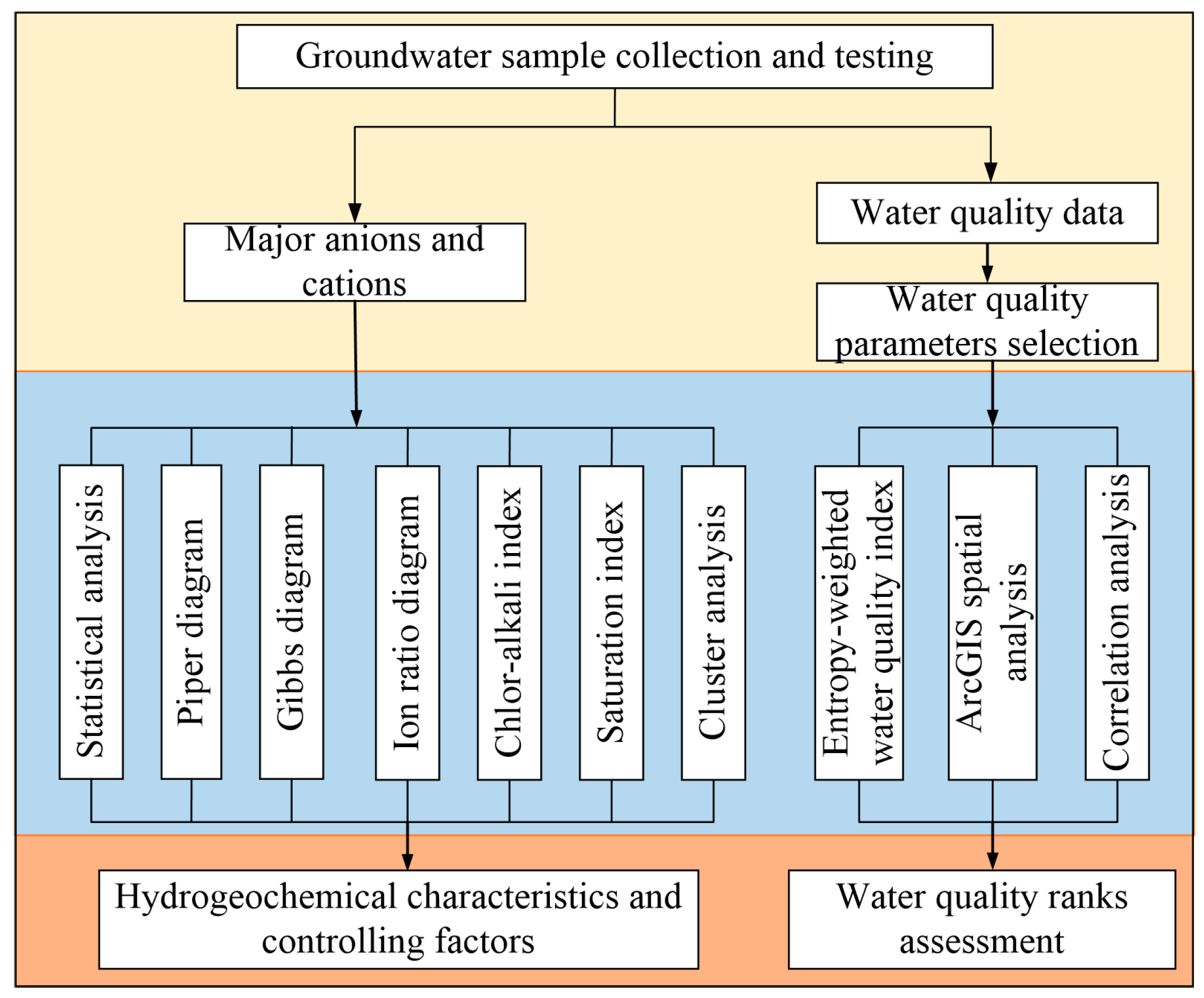
3.4. Research Content and Technical Route
4. Results and Discussion
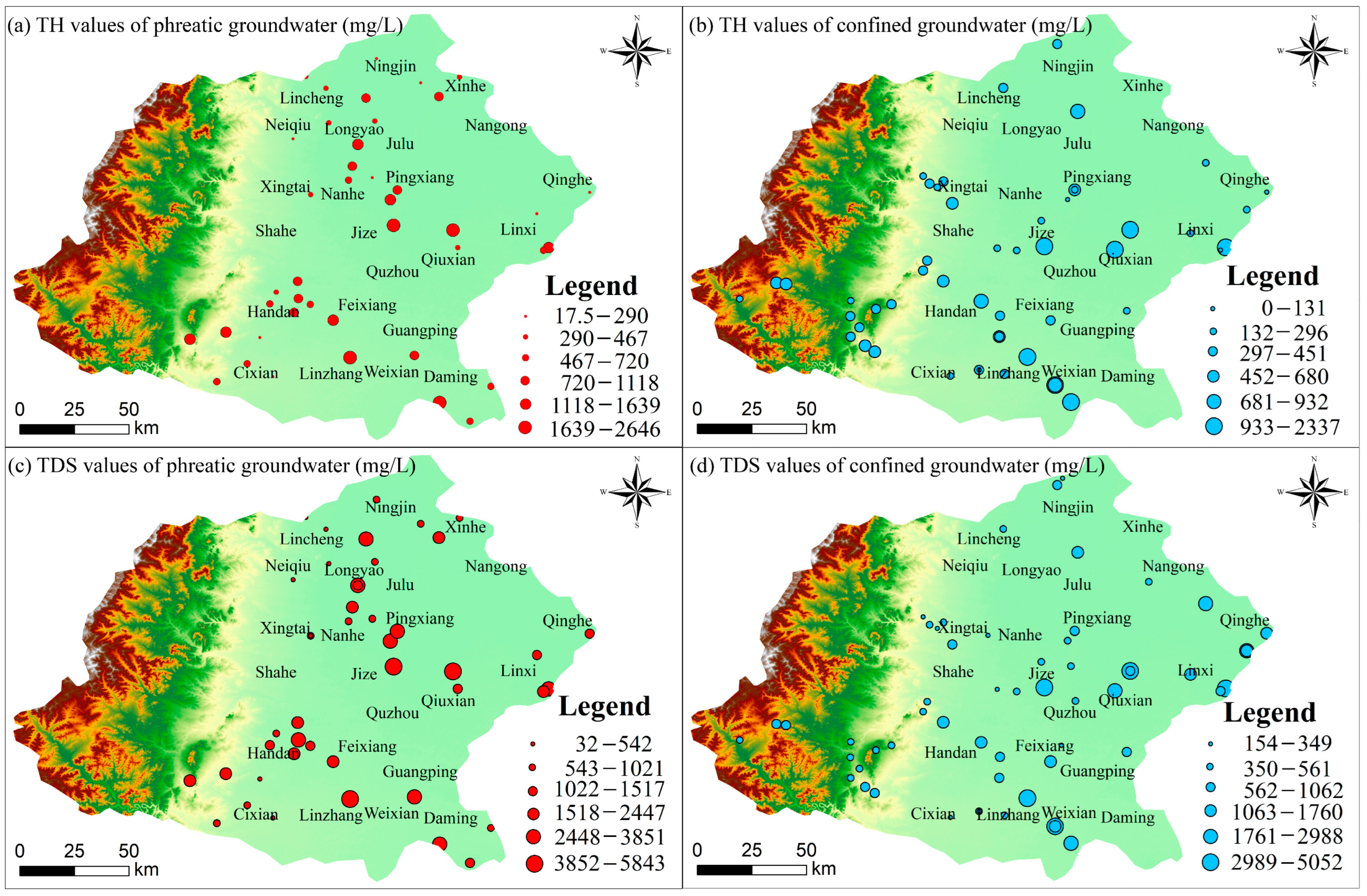
4.1. Hydrochemical Characteristics
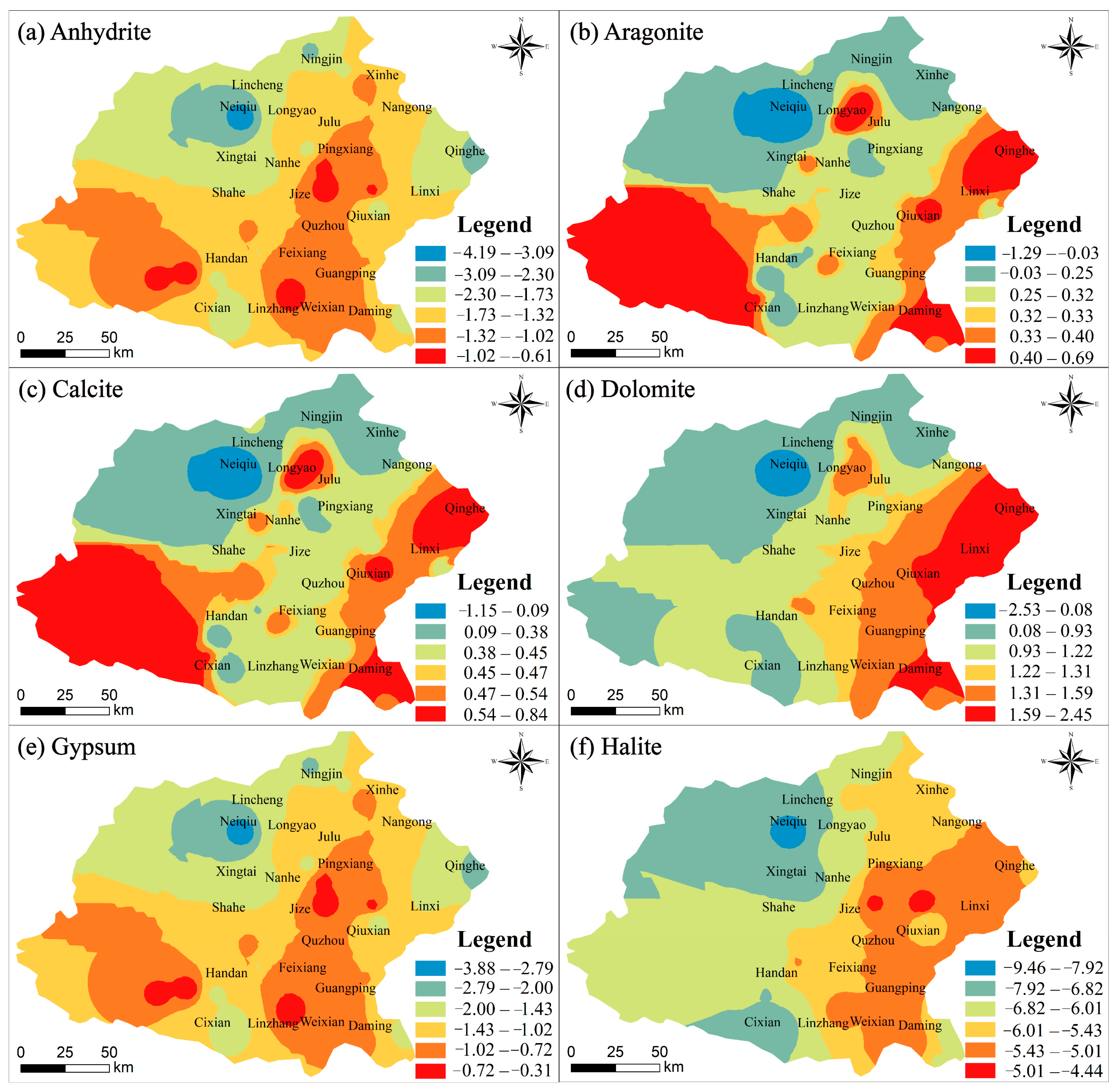
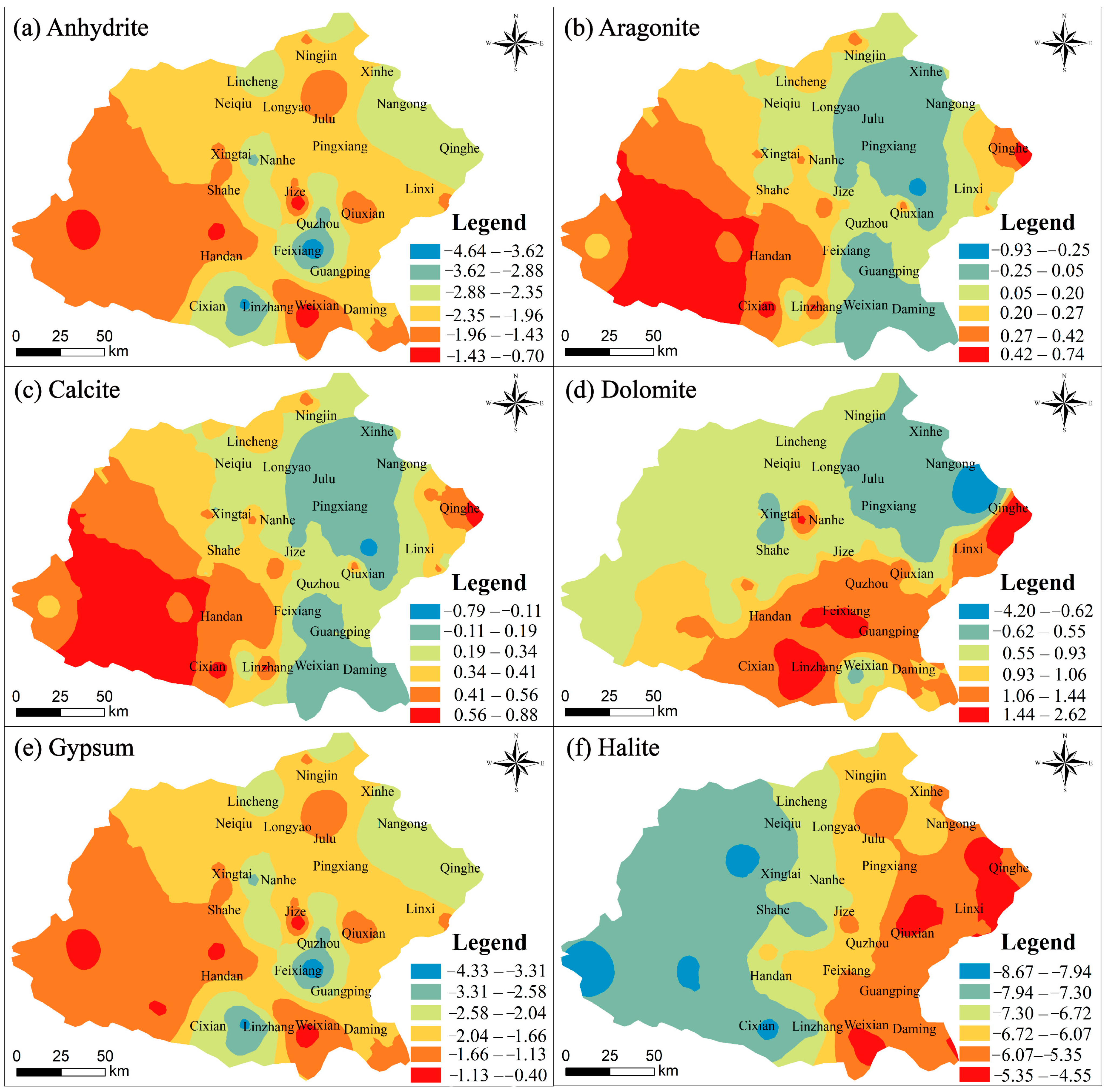
4.2. Mechanism Controlling Groundwater Hydrochemistry
4.2.1. Natural Factors Affecting Groundwater Hydrochemistry
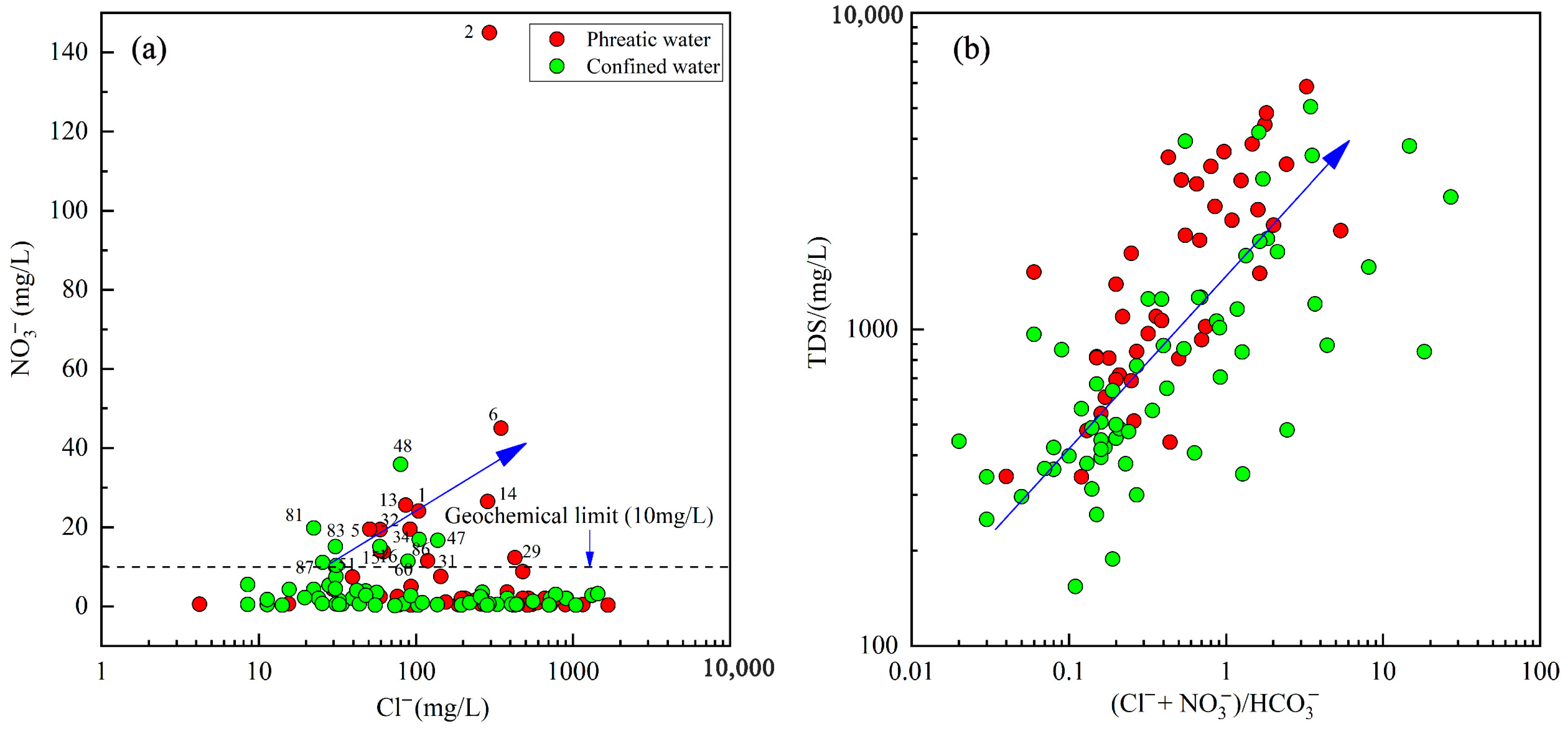
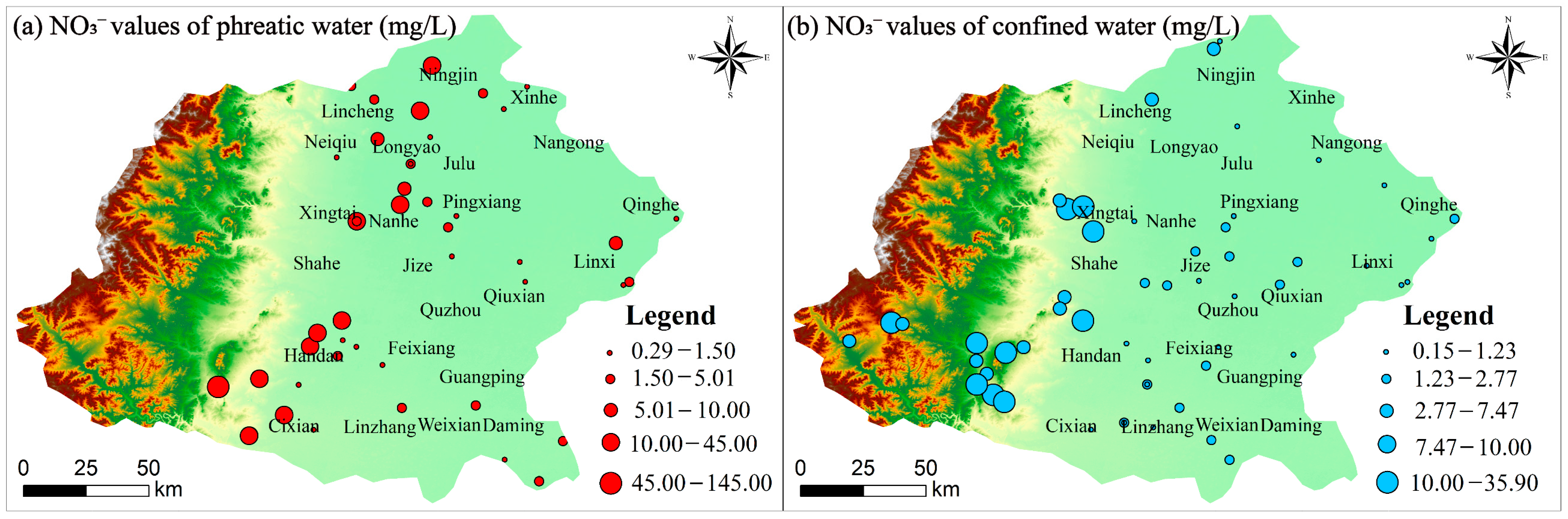
4.2.2. Anthropogenic Factors
4.3. Source Statistical Analysis
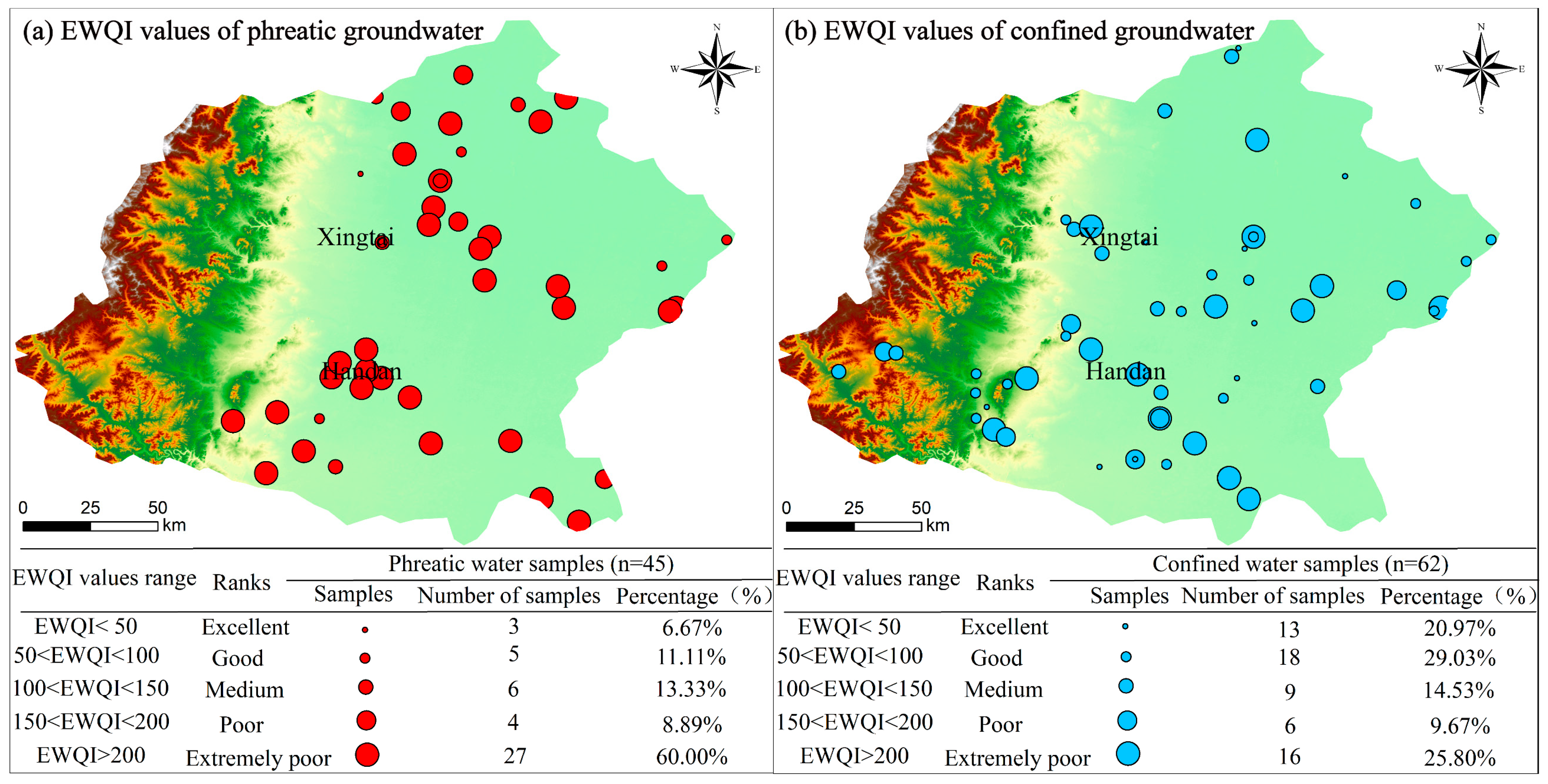
4.4. Groundwater Quality Evaluation
4.5. Corresponding Protection and Management Measures
- (1)
- Groundwater wells with EWQI values exceeding 100 should be stopped until remedial action is taken.
- (2)
- The use of nitrate-rich fertilizer and stockpiling of the waste ore and garbage should be reduced in the western piedmont plain, and appropriate ecological measures should be used to reduce nitrate concentrations.
- (3)
- Groundwater filtration equipment should be installed to reduce the concentrations of Mg2+, SO42−, Cl−, TH and TDS values in groundwater with high EWQI.
- (4)
- Real-time water quality monitoring systems and early warning platforms to enhance monitoring capabilities should be established.
- (5)
- A sound and scientific water resources management system, and rational plan for the development and utilization of groundwater should be established.
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Liu, Y.; Wang, P.; Ruan, H.; Wang, T.; Yu, J.; Cheng, Y.; Kulmatov, R. Sustainable Use of Groundwater Resources in the Transboundary Aquifers of the Five Central Asian Countries: Challenges and Perspectives. Water 2020, 12, 2101. [Google Scholar] [CrossRef]
- Gao, F.; Wang, H.; Liu, C. Long-term assessment of groundwater resources carrying capacity using GRACE data and Budyko model. J. Hydrol. 2020, 588, 125042. [Google Scholar] [CrossRef]
- Siebert, S.; Burke, J.; Faures, J.M.; Frenken, K.; Hoogeveen, J.; Döll, P.; Portmann, F.T. Groundwater use for irrigation—A global inventory. Hydrol. Earth Syst. Sci. 2010, 14, 1863–1880. [Google Scholar] [CrossRef]
- Gleeson, T.; Befus, K.M.; Jasechko, S.; Luijendijk, E.; Cardenas, M.B. The global volume and distribution of modern groundwater. Nat. Geosci. 2015, 9, 161–167. [Google Scholar] [CrossRef]
- Wang, H.; Jia, G. Satellite-based monitoring of decadal soil salinization and climate effects in a semi-arid region of China. Adv. Atmos. Sci. 2012, 29, 1089–1099. [Google Scholar] [CrossRef]
- Li, M.; Xie, Y.; Dong, Y.; Wang, L.; Zhang, Z. Review: Recent progress on groundwater recharge research in arid and semiarid areas of China. Hydrogeol. J. 2023, 278, 1–22. [Google Scholar] [CrossRef]
- Arabameri, A.; Rezaei, K.; Cerda, A.; Lombardo, L.; Rodrigo-Comino, J. GIS-based groundwater potential mapping in Shahroud plain, Iran. A comparison among statistical (bivariate and multivariate), data mining and MCDM approaches. Sci. Total Environ. 2019, 658, 160–177. [Google Scholar] [CrossRef]
- Valdes-Abellan, J.; Pardo, M.A.; Jodar-Abellan, A.; Pla, C.; Fernandez-Mejuto, M. Climate change impact on karstic aquifer hydrodynamics in southern Europe semi-arid region using the KAGIS model. Sci. Total Environ. 2020, 723, 138110. [Google Scholar] [CrossRef]
- Han, S.; Yang, Y.; Fan, T.; Xiao, D.; Moiwo, J.P. Precipitation-runoff processes in Shimen hillslope micro-catchment of Taihang Mountain, north China. Hydrol. Process. 2012, 26, 1332–1341. [Google Scholar] [CrossRef]
- Gastélum, J.R.; Cullom, C. Application of the Colorado River Simulation System Model to Evaluate Water Shortage Conditions in the Central Arizona Project. Water Resour. Manag. 2013, 27, 2369–2389. [Google Scholar] [CrossRef]
- Sun, Y.; Liu, N.; Shang, J.; Zhang, J. Sustainable utilization of water resources in China: A system dynamics model. J. Clean. Prod. 2017, 142, 613–625. [Google Scholar] [CrossRef]
- Chen, X.; Li, F.; Li, X.; Hu, Y.; Hu, P. Evaluating and mapping water supply and demand for sustainable urban ecosystem management in Shenzhen, China. J. Clean. Prod. 2020, 251, 119754. [Google Scholar] [CrossRef]
- Zhang, J.; Chen, L.; Hou, X.; Li, J.; Ren, X.; Lin, M.; Zhang, M.; Wang, Y.; Tian, Y. Effects of multi-factors on the spatiotemporal variations of deep confined groundwater in coal mining regions, North China. Sci. Total Environ. 2022, 823, 153741. [Google Scholar] [CrossRef] [PubMed]
- Bowell, R.J. Hydrogeochemistry of the Tsumeb Deposit: Implications for Arsenate Mineral Stability. Rev. Mineral. Geochem. 2014, 79, 589–627. [Google Scholar] [CrossRef]
- Yang, Q.; Li, Z.; Ma, H.; Wang, L.; Martin, J.D. Identification of the hydrogeochemical processes and assessment of groundwater quality using classic integrated geochemical methods in the Southeastern part of Ordos basin, China. Environ. Pollut. 2016, 218, 879–888. [Google Scholar] [CrossRef]
- Hoaghia, M.A.; Moldovan, A.; Kovacs, E.; Mirea, I.; Kenesz, M.; Brad, T.; Cadar, O.; Micle, V.; Levei, E.; Moldovan, O. Water Quality and Hydrogeochemical Characteristics of Some Karst Water Sources in Apuseni Mountains, Romania. Water 2021, 13, 857. [Google Scholar] [CrossRef]
- Chen, S.; Tang, Z.; Wang, J.; Wu, J.; Yang, C.; Kang, W.; Huang, X. Multivariate Analysis and Geochemical Signatures of Shallow Groundwater in the Main Urban Area of Chongqing, Southwestern China. Water 2020, 12, 2833. [Google Scholar] [CrossRef]
- Nobre, R.C.; Nobre, M.M. Natural attenuation of chlorinated organics in a shallow sand aquifer. J. Hazard. Mater. 2004, 110, 129–137. [Google Scholar] [CrossRef]
- Négrel, P.; Lemière, B.; Machard de Grammont, H.; Billaud, P.; Sengupta, B. Hydrogeochemical processes, mixing and isotope tracing in hard rock aquifers and surface waters from the Subarnarekha River Basin, (east Singhbhum District, Jharkhand State, India). Hydrogeol. J. 2007, 15, 1535–1552. [Google Scholar] [CrossRef]
- Liu, F.; Wang, S.; Wang, L.; Shi, L.; Song, X.; Zhen, P. Coupling hydrochemistry and stable isotopes to identify the major factors affecting groundwater geochemical evolution in the Heilongdong Spring Basin, North China. J. Geochem. Explor. 2019, 205, 106352. [Google Scholar] [CrossRef]
- Özdemir, Ö. Application of Multivariate Statistical Methods for Water Quality Assessment of Karasu-Sarmisakli Creeks and Kizilirmak River in Kayseri, Turkey. Pol. J. Environ. Stud. 2016, 25, 1149–1160. [Google Scholar] [CrossRef]
- Li, M.; Liu, Z.; Zhang, M.; Chen, Y. A workflow for spatio-seasonal hydro-chemical analysis using multivariate statistical techniques. Water Res. 2021, 188, 116550. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Dong, D.; Lv, S.; Ding, J.; Yan, M.; Han, G. Spatial evolution analysis of groundwater chemistry, quality, and fluoride health risk in southern Hebei Plain, China. Environ. Sci. Pollut. Res. 2023, 30, 61032–61051. [Google Scholar] [CrossRef]
- Zhang, H.; Singh, V.P.; Sun, D.; Yu, Q.; Cao, W. Has water-saving irrigation recovered groundwater in the Hebei Province plains of China? Int. J. Water Resour. Dev. 2016, 33, 534–552. [Google Scholar] [CrossRef]
- Yu, L.; Ling, M.; Chen, F.; Ding, Y.; Lv, C. Practices of groundwater over-exploitation control in Hebei Province. Water Policy 2020, 22, 591–601. [Google Scholar]
- Yuan, Z.; Shen, Y. Estimation of agricultural water consumption from meteorological and yield data: A case study of Hebei, North China. PLoS ONE 2013, 8, e58685. [Google Scholar] [CrossRef]
- Xu, Y.; Mo, X.; Cai, Y.; Li, X. Analysis on groundwater table drawdown by land use and the quest for sustainable water use in the Hebei Plain in China. Agric. Water Manag. 2005, 75, 38–53. [Google Scholar] [CrossRef]
- Hu, X.; Shi, L.; Zeng, J.; Yang, J.; Zha, Y.; Yao, Y.; Cao, G. Estimation of actual irrigation amount and its impact on groundwater depletion: A case study in the Hebei Plain, China. J. Hydrol. 2016, 543, 433–449. [Google Scholar] [CrossRef]
- Feng, W.; Zhong, M.; Lemoine, J.M.; Biancale, R.; Hsu, H.T.; Xia, J. Evaluation of groundwater depletion in North China using the Gravity Recovery and Climate Experiment (GRACE) data and ground-based measurements. Water Resour. Res. 2013, 49, 2110–2118. [Google Scholar] [CrossRef]
- Wang, Y.; Cheng, L.; Tian, H.; Liu, X. Water supply eco-economic benefit evaluation of middle route of south-to-north water diversion project in Hebei Water-recipient Area. IOP Conf. Ser. Earth Environ. Sci. 2018, 191, 012064. [Google Scholar] [CrossRef]
- Wang, M.; Liao, L.; Zhang, X.; Li, Z.; Xia, Z.; Cao, W. Adsorption of low-concentration ammonium onto vermiculite from Hebei Province, China. Clays Clay Miner. 2011, 59, 459–465. [Google Scholar] [CrossRef]
- Zhang, Q.; Han, X.; Li, J.; Zhao, J.; Zhou, W.; Gao, W. The Study on Environmental Evolved Characteristic of Sandy Coast. Appl. Mech. Mater. 2012, 226–228, 1170–1173. [Google Scholar] [CrossRef]
- Li, G.; Han, T.; Li, X. Groundwater environmental degradation mechanism and countermeasures in the plain area of Xingtai City. Groundwater 2011, 33, 63–66. [Google Scholar]
- Charlton, S.R.; Parkhurst, D.L. Modules based on the geochemical model PHREEQC for use in scripting and programming languages. Comput. Geosci. 2011, 37, 1653–1663. [Google Scholar] [CrossRef]
- Islam, A.R.M.T.; Ahmed, N.; Bodrud-Doza, M.; Chu, R. Characterizing groundwater quality ranks for drinking purposes in Sylhet district, Bangladesh, using entropy method, spatial autocorrelation index, and geostatistics. Environ. Sci. Pollut. Res. 2017, 24, 26350–26374. [Google Scholar] [CrossRef]
- Su, H.; Kang, W.; Xu, Y.; Wang, J. Assessing Groundwater Quality and Health Risks of Nitrogen Pollution in the Shenfu Mining Area of Shaanxi Province, Northwest China. Expo. Health 2017, 10, 77–97. [Google Scholar] [CrossRef]
- GB/T14848-2017; Standards for Groundwater Quality. Standards Press of China: Beijing, China, 2017.
- WHO. Guidelines for Drinking-Water Quality, 4th ed.; World Health Organ: Geneva, Switzerland, 2011. [Google Scholar]
- Carrera-Villacrés, D.; Hidalgo, A.; Guevara-García, P.; Vivero, M.T.; Delgado-Rodríguez, V. Hydrogeochemical analysis of volcanic and geothermal fluids in the Andes from Ecuador using hydrochemical plots (Stiff, Piper and Schoeller-Berkaloff diagrams). IOP Conf. Ser. Earth Environ. Sci. 2016, 39, 012062. [Google Scholar] [CrossRef]
- Gibbs, R.J. Mechanisms controlling world water chemistry. Science 1970, 170, 1088–1090. [Google Scholar] [CrossRef]
- Li, X.; Huang, X.; Zhang, Y. Spatio-temporal analysis of groundwater chemistry, quality and potential human health risks in the Pinggu basin of North China Plain: Evidence from high-resolution monitoring dataset of 2015–2017. Sci. Total Environ. 2021, 800, 149568. [Google Scholar] [CrossRef]
- Rajmohan, N.; Patel, N.; Singh, G.; Amarasinghe, U.A. Hydrochemical evaluation and identification of geochemical processes in the shallow and deep wells in the Ramganga Sub-Basin, India. Environ. Sci. Pollut. Res. Int. 2017, 24, 21459–21475. [Google Scholar] [CrossRef]
- Nematollahi, M.J.; Ebrahimi, P.; Razmara, M.; Ghasemi, A. Hydrogeochemical investigations and groundwater quality assessment of Torbat-Zaveh plain, Khorasan Razavi, Iran. Environ. Monit. Assess. 2015, 188, 2. [Google Scholar] [CrossRef] [PubMed]
- Wu, P.; Tang, C.; Zhu, L.; Liu, C.; Cha, X.; Tao, X. Hydrogeochemical characteristics of surface water and groundwater in the karst basin, southwest China. Hydrol. Process. 2009, 23, 2012–2022. [Google Scholar] [CrossRef]
- Nakhaei, M.; Dadgar, M.A.; Amiri, V. Geochemical processes analysis and evaluation of groundwater quality in Hamadan Province, Western Iran. Arab. J. Geosci. 2016, 9, 384. [Google Scholar] [CrossRef]
- Amiri, V.; Nakhaei, M.; Lak, R.; Kholghi, M. Investigating the salinization and freshening processes of coastal groundwater resources in Urmia aquifer, NW Iran. Environ. Monit. Assess. 2016, 188, 233. [Google Scholar] [CrossRef]
- Yu, Y.; Song, X.; Zhang, Y.; Zheng, F.; Liang, J.; Liu, L. Identifying spatio-temporal variation and controlling factors of chemistry in groundwater and river water recharged by reclaimed water at Huai River, North China. Stoch. Environ. Res. Risk Assess. 2013, 28, 1135–1145. [Google Scholar] [CrossRef]
- Li, P.; Zhang, Y.; Yang, N.; Jing, L.; Yu, P. Major Ion Chemistry and Quality Assessment of Groundwater in and around a Mountainous Tourist Town of China. Expo. Health 2016, 8, 239–252. [Google Scholar] [CrossRef]
- Abdesselem, K.; Azedine, H.; Lynda, C. Groundwater hydrochemistry and effects of anthropogenic pollution in Béchar city (SW Algeria). Desalination Water Treat. 2015, 57, 14034–14043. [Google Scholar] [CrossRef]
- Wu, J.; Li, P.; Qian, H.; Duan, Z.; Zhang, X. Using correlation and multivariate statistical analysis to identify hydrogeochemical processes affecting the major ion chemistry of waters: A case study in Laoheba phosphorite mine in Sichuan, China. Arab. J. Geosci. 2013, 7, 3973–3982. [Google Scholar] [CrossRef]
- Singh, A.K.; Mondal, G.C.; Singh, T.B.; Singh, S.; Tewary, B.K.; Sinha, A. Hydrogeochemical processes and quality assessment of groundwater in Dumka and Jamtara districts, Jharkhand, India. Environ. Earth Sci. 2012, 67, 2175–2191. [Google Scholar] [CrossRef]
- Li, P.; Tian, R.; Liu, R. Solute Geochemistry and Multivariate Analysis of Water Quality in the Guohua Phosphorite Mine, Guizhou Province, China. Expo. Health 2018, 11, 81–94. [Google Scholar] [CrossRef]
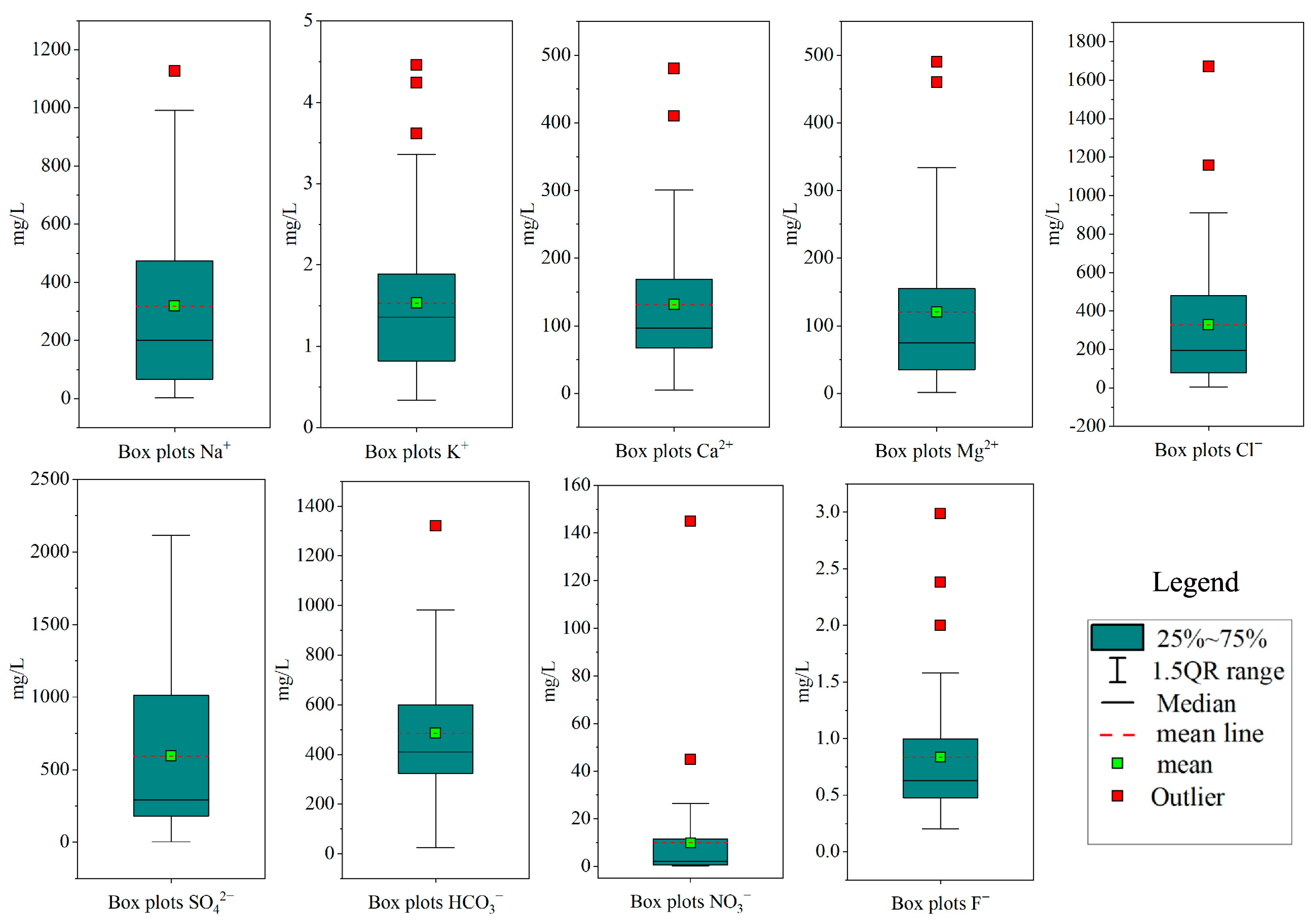
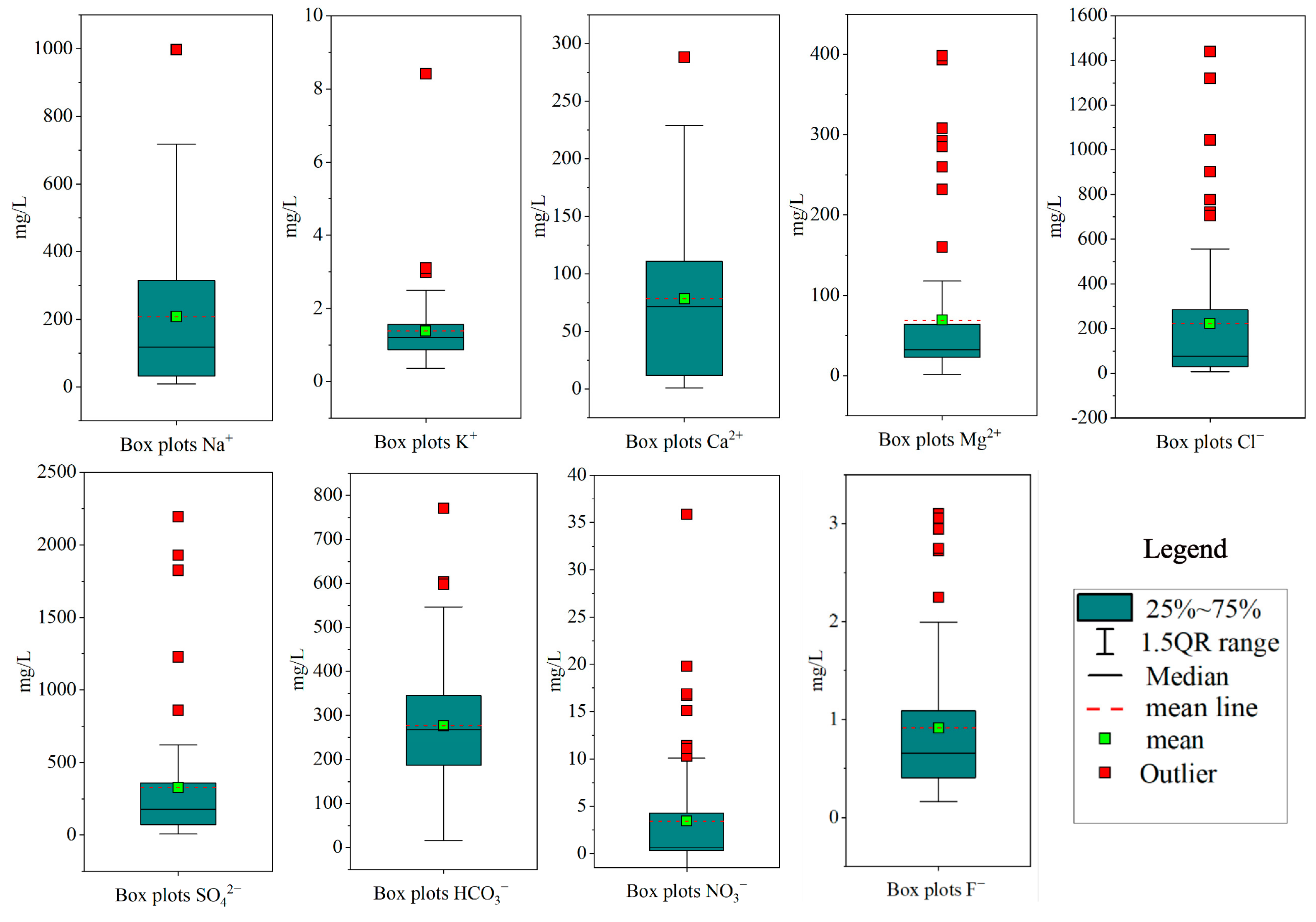
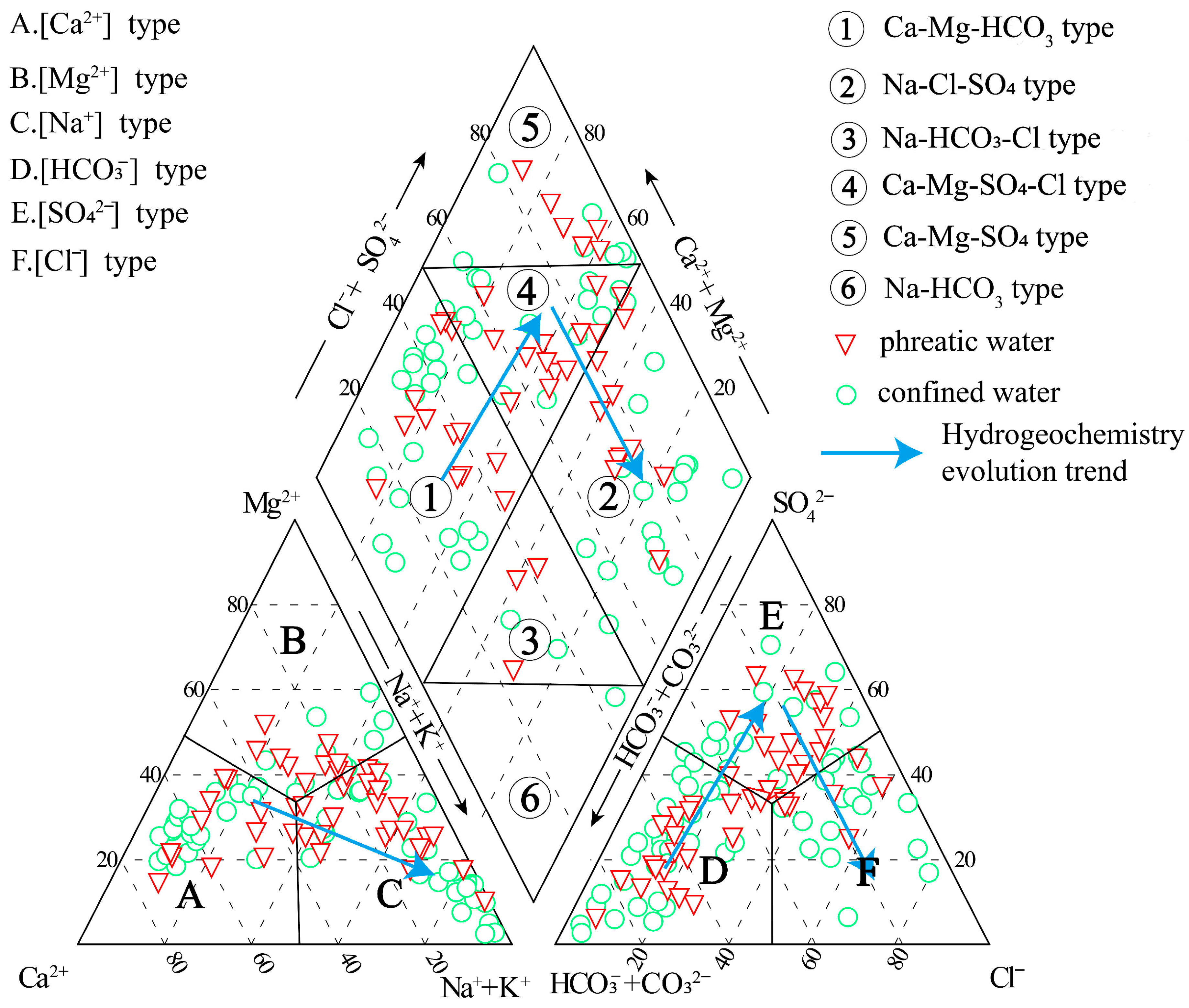
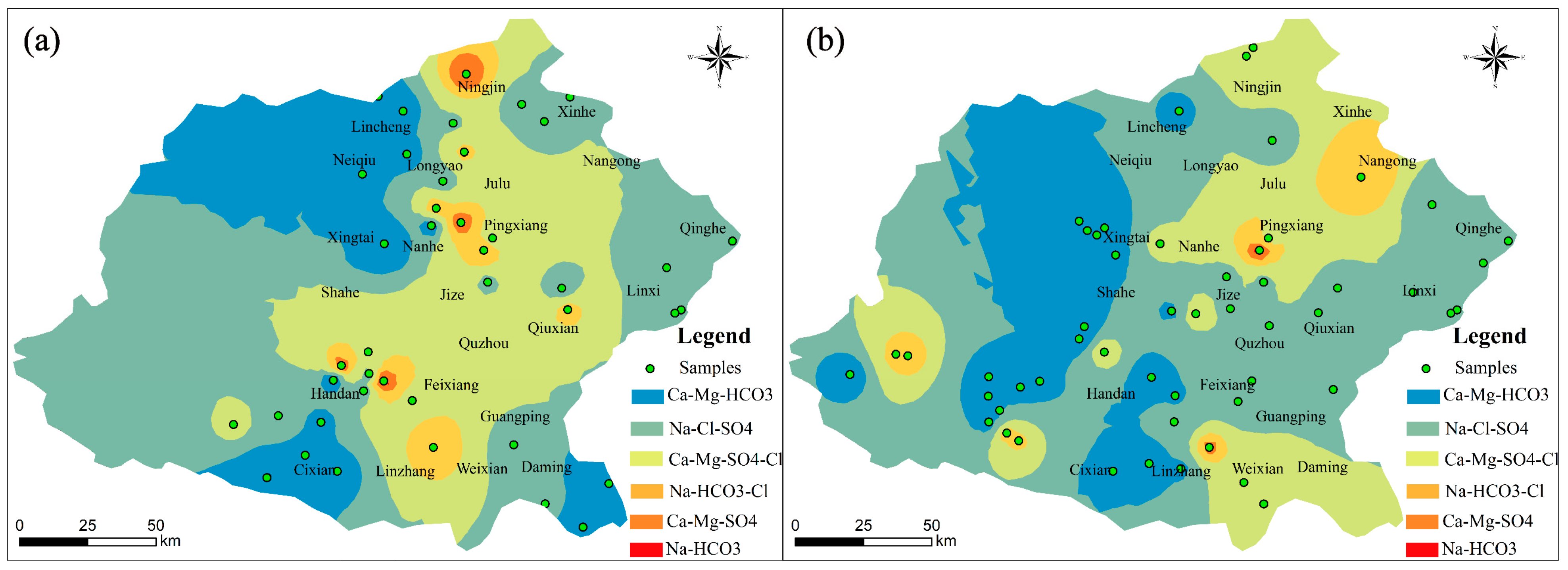
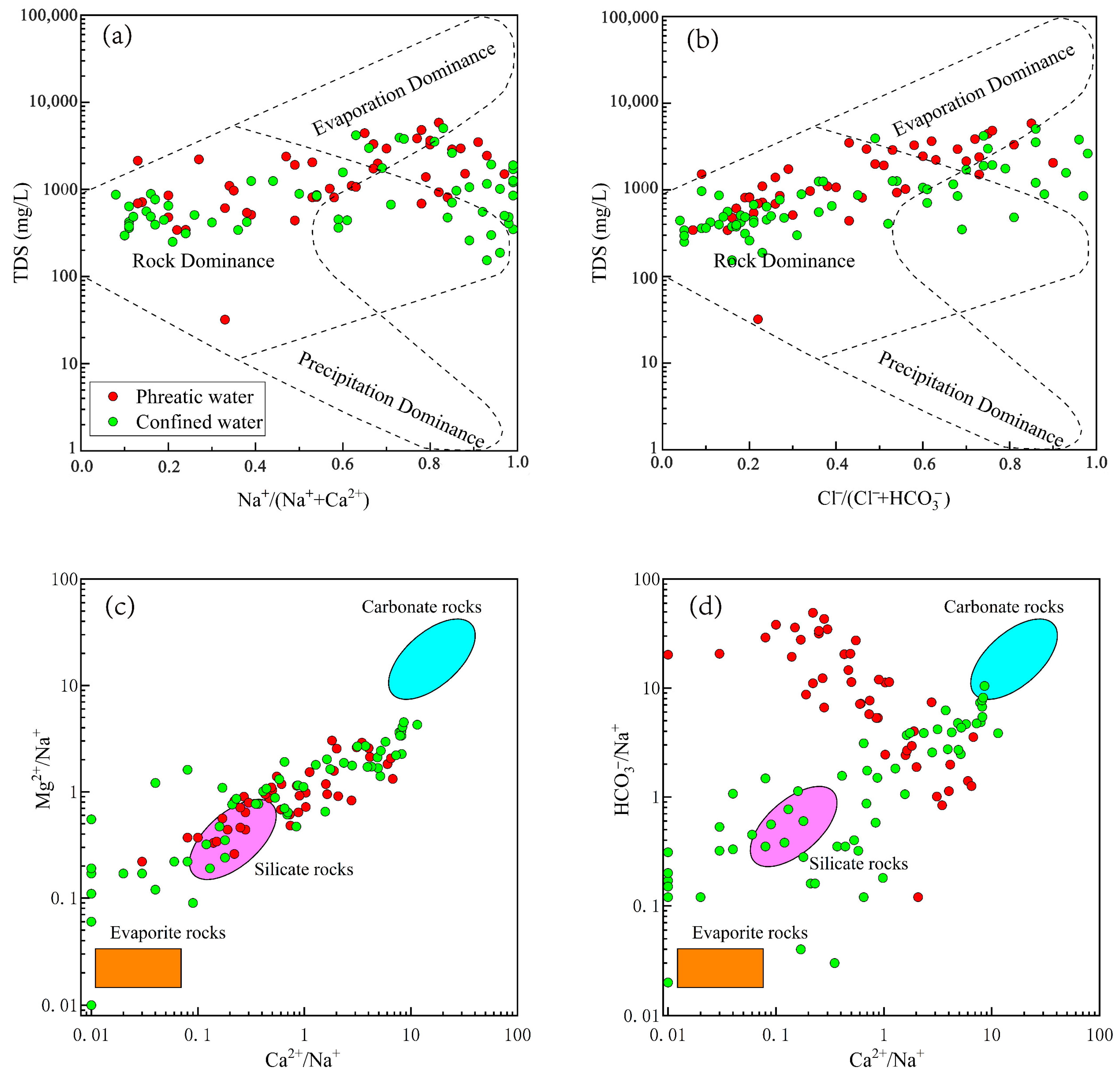




| Parameters | Guideline | Phreatic Groundwater | Confined Groundwater | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Min. | Max. | Ave. | SD | CV (%) | Min. | Max. | Ave. | SD | CV (%) | ||
| pH | 6.5–8.5 * | 7.02 | 9.08 | 7.45 | 0.39 | 5.31 | 7.00 | 10.60 | 8.02 | 0.87 | 10.87 |
| TH | 450 * | 268 | 2646.00 | 825.10 | 624.72 | 75.71 | 11.60 | 2337.00 | 478.20 | 509.26 | 106.49 |
| TDS | 1000 * | 342 | 5843.00 | 1772.22 | 1354.46 | 76.42 | 154.00 | 5052.00 | 1062.35 | 1080.78 | 101.73 |
| Na+ | 200 * | 2.69 | 1127.00 | 318.72 | 302.14 | 94.79 | 9.35 | 996.00 | 207.97 | 233.43 | 101.73 |
| K+ | 0.34 | 4.46 | 1.53 | 0.97 | 63.69 | 0.36 | 8.41 | 1.38 | 1.06 | 77.40 | |
| Ca2+ | 75 ** | 4.87 | 480.00 | 131.39 | 98.58 | 75.03 | 0.98 | 288.00 | 78.33 | 70.02 | 89.39 |
| Mg2+ | 50 ** | 1.29 | 490.00 | 120.71 | 115.30 | 95.51 | 1.90 | 398.00 | 68.97 | 92.92 | 134.73 |
| Cl− | 250 * | 4.20 | 1672.00 | 328.06 | 355.92 | 105.98 | 8.50 | 1439.00 | 223.30 | 318.83 | 142.78 |
| SO42− | 250 * | 3.66 | 2114.00 | 594.26 | 588.77 | 129.07 | 8.65 | 2192.00 | 329.48 | 480.54 | 145.84 |
| HCO3− | 25.00 | 1321.00 | 485.51 | 248.94 | 51.27 | 16.00 | 771.00 | 276.77 | 150.17 | 54.25 | |
| NO3− | 50 ** | 0.29 | 145.00 | 9.85 | 22.72 | 230.65 | 0.15 | 35.9 | 4.08 | 6.30 | 194.39 |
| F− | 1.0 * | 0.20 | 2.99 | 0.83 | 0.58 | 70.44 | 0.16 | 3.10 | 0.91 | 0.75 | 113.40 |
| TH | TDS | pH | Na+ | K+ | Ca2+ | Mg2+ | Cl− | SO42− | HCO3− | NO3− | F− | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Phreatic groundwater samples (n = 45) | ||||||||||||
| TH | 1 | |||||||||||
| TDS | 0.91 ** | 1 | ||||||||||
| pH | −0.47 ** | −0.31 * | 1 | |||||||||
| Na+ | 0.67 ** | 0.91 ** | −0.11 | 1 | ||||||||
| K+ | 0.17 | 0.10 | 1 | |||||||||
| Ca2+ | 0.73 ** | 0.50 ** | −0.45 ** | 0.16 | 0.28 | 1 | ||||||
| Mg2+ | 0.93 ** | 0.93 ** | −0.38 ** | 0.80 ** | 0.44 ** | 1 | ||||||
| Cl− | 0.87 ** | 0.93 ** | −0.21 | 0.84 ** | 0.17 | 0.43 ** | 0.91 ** | 1 | ||||
| SO42− | 0.89 ** | 0.97 ** | −0.30 * | 0.86 ** | 0.50 ** | 0.91 ** | 0.86 ** | 1 | ||||
| HCO3− | 0.20 | 0.37 * | −0.37 * | 0.52 ** | −0.19 | −0.11 | 0.32 * | 0.20 | 0.26 | 1 | ||
| NO3− | −0.24 | 0.45 ** | 0.59 ** | −0.19 | −0.12 | −0.12 | −0.18 | 1 | ||||
| F− | 0.11 | 0.49 ** | 0.28 | −0.19 | −0.33 * | 0.26 | −0.24 | 1 | ||||
| Confined groundwater samples (n = 62) | ||||||||||||
| TH | 1 | |||||||||||
| TDS | 0.90 ** | 1 | ||||||||||
| pH | −0.51 ** | −0.22 | 1 | |||||||||
| Na+ | 0.61 ** | 0.89 ** | 0.15 | 1 | ||||||||
| K+ | 1 | |||||||||||
| Ca2+ | 0.82 ** | 0.58 ** | −0.74 ** | 0.18 | 0.21 | 1 | ||||||
| Mg2+ | 0.97 ** | 0.94 ** | −0.35 ** | 0.73 ** | 0.64 ** | 1 | ||||||
| Cl− | 0.78 ** | 0.88 ** | 0.83 ** | 0.39 ** | 0.86 ** | 1 | ||||||
| SO42− | 0.88 ** | 0.95 ** | −0.26 * | 0.79 ** | 0.63 ** | 0.89 ** | 0.70 ** | 1 | ||||
| HCO3− | 0.28 * | 0.24 | −0.46 ** | 0.15 | 0.28 * | 0.24 | 0.23 | 1 | ||||
| NO3− | −0.19 | −0.34 ** | −0.37 ** | 0.28 | 0.38 ** | −0.19 | −0.25 * | −0.15 | 1 | |||
| F− | 0.26 * | 0.29 * | 0.55 ** | −0.14 | −0.29 * | 0.29 * | 0.15 | 0.20 | −0.29 * | 1 | ||
| Index | Category | TH | TDS | pH | Na+ | K+ | Ca2+ | Mg2+ | Cl− | SO42− | HCO3− | NO3− | F− |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Information entropy (ej) | Phreatic groundwater | 0.929 | 0.927 | 0.918 | 0.886 | 0.918 | 0.928 | 0.899 | 0.874 | 0.881 | 0.963 | 0.676 | 0.909 |
| Confined groundwater | 0.891 | 0.874 | 0.921 | 0.849 | 0.920 | 0.895 | 0.847 | 0.804 | 0.824 | 0.959 | 0.796 | 0.898 | |
| Entropy weight (wj) | Phreatic groundwater | 0.055 | 0.057 | 0.063 | 0.088 | 0.064 | 0.056 | 0.078 | 0.097 | 0.092 | 0.029 | 0.250 | 0.070 |
| Confined groundwater | 0.071 | 0.083 | 0.052 | 0.099 | 0.053 | 0.069 | 0.101 | 0.129 | 0.115 | 0.027 | 0.134 | 0.067 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, L.; Dong, D.; Lv, S.; Zhang, J.; Yan, M.; Han, G.; Li, H. Hydrochemical Characterization and Quality Assessment of Groundwater in the Southern Plain of Hebei Province, China. Water 2023, 15, 3791. https://doi.org/10.3390/w15213791
Zhang L, Dong D, Lv S, Zhang J, Yan M, Han G, Li H. Hydrochemical Characterization and Quality Assessment of Groundwater in the Southern Plain of Hebei Province, China. Water. 2023; 15(21):3791. https://doi.org/10.3390/w15213791
Chicago/Turabian StyleZhang, Longqiang, Donglin Dong, Situ Lv, Jialun Zhang, Maohua Yan, Guilei Han, and Huizhe Li. 2023. "Hydrochemical Characterization and Quality Assessment of Groundwater in the Southern Plain of Hebei Province, China" Water 15, no. 21: 3791. https://doi.org/10.3390/w15213791
APA StyleZhang, L., Dong, D., Lv, S., Zhang, J., Yan, M., Han, G., & Li, H. (2023). Hydrochemical Characterization and Quality Assessment of Groundwater in the Southern Plain of Hebei Province, China. Water, 15(21), 3791. https://doi.org/10.3390/w15213791







