Three New Nitzschia (Bacillariophyceae) Species from Highly Acidic Artificial Lakes in Çanakkale, Türkiye
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Sampling
2.3. Sample Preparation
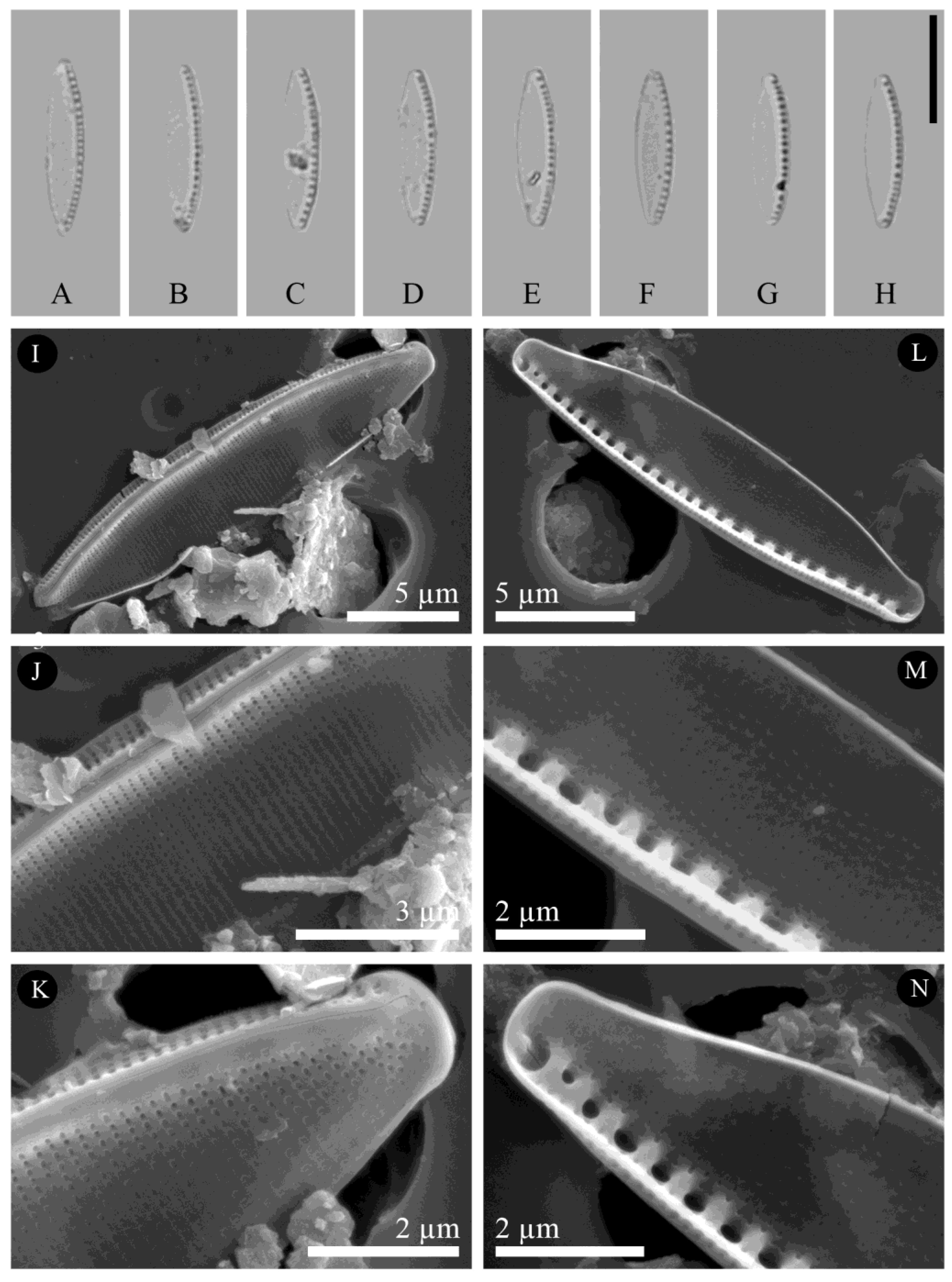
3. Results
| Valve Length (µm) | Valve Width (µm) | Striae (in 10 µm) | Fibulae (in 10 µm) | Valve Shape | Apex Shape | Keel Shape | Copulae | Reference | |
|---|---|---|---|---|---|---|---|---|---|
| N. arslaniana | 12.0–14.0 | 2.0–2.5 | 27–29 | 13–15 | linear | broadly rounded, cuneate | elevated with conopeum, recessed at center | open bands, 4+, 2 rows of large pores | this study |
| N. szaboiana | 13.0–17.0 | 3.0–4.0 | 47–50 | 14–16 | linear-elliptic | broadly rounded, cuneate | round, elevated, terminates at end on valve face | -- | this study |
| N. efeiana | 20.0–43.0 | 3.5–4.0 | 23–27 | 6–10 | linear, linear-lanceolate | capitate | flat, marginal, terminates at apex | Open bands, 4+, one row of small round pores | this study |
| N. acidoclinata | 8.0–45.0 | 2.5–3.0 | 27–34 | 10–16 | linear-lanceolate to narrowly linear | shortly subrostrate | -- | -- | [25] |
| N. anatoliana | 7.8–16.1 | 2.7–3.7 | 48–52 | 20–23 | linear-elliptic | broadly rounded | marginal | -- | [26] |
| N. bremensis | 60.0–90.0 | 6.0–9.0 | 26–32 | 5–9 | linear | rounded to flat truncated | Submarginal, broad, on valve face, elevated, with conopeum? | - | [27] |
| N. capitellata | 20.0–70.0 | 3.5–6.5 | 23–40 | 10–18 | linear, linear-lanceolate | rounded, wedge shaped | marginal, small | -- | [27] |
| N. homburgiensis | 32.0–52.0 | 5.0–6.0 | 34–40 | 9–15 | linear, strongly concave in the middle | rounded, wedge shaped | submarginal | -- | [27] |
| N. hybrida | 34–125 | 5–9 | 22–26 | 9–12 | linear, strongly concave in the middle | wedge shaped | marginal, round, elevated, recessed | -- | [26] |
| N. inconspicua | 6.0–11.5 | 2.6–3.1 | 24–29 | 11–17 | linear to lanceolate, elliptic-lanceolate | slightly protracted, narrow | marginal, round, not elevated, interrupted at center | copulae 4+, open bands, one row of pores | [24] |
| N. frustulum | 10.8–34 | 3.0–3.9 | 27–30 | 13–15 | linear-lanceolate to lanceolate, elliptic-lanceolate | slightly protracted, narrow | marginal, round, not elevated, terminating along margin at apex | open copulae, one row of pores | [24] |
| N. palustris | 35.0–60.0 | 4.0–7.0 | 22–28 | 6–10 | linear | rounded to flat truncated | marginal, interrupted at center | -- | [28] |
| N. pellucida | -- | -- | 30–40 | 12–18 | linear-lanceolate | boat-shaped | submarginal, round, elevated, terminating on valve face at end | -- | [27,29] |
| N. perminuta | 8.0–45.0 | 2.5–3.0 | 26–36 | 10–16 | linear-lanceolate to narrowly linear | subrostrate to subcapitate | marginal | -- | [25] |
| N. perspicua | 8.0–55.0 | 1.5–4.0 | 44–45 | 14–19 | rhombic-lanceolate to narrow linear-lanceolate | gradually narrowing rounded | marginal | -- | [27] |
| N. pusilla | 8.0–33.0 | 2.5–5.0 | 43–55 | 14–20 | linear-lanceolate to linear | broadly rounded, slightly protruding in larger forms | marginal | -- | [27] |
4. Discussion
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Jonson, D.B.; Hallberg, K.B. The microbiology of acidic mine waters. Res. Mıcrobiol. 2003, 154, 466–473. [Google Scholar] [CrossRef] [PubMed]
- Noerdstrom, D.K.; Alpers, C.N. Geochemistry of acid mine waters. In The Geochemistry of Mineral Deposits; Plemlee, G.S., Logsdon, M.J., Eds.; Society for Economic Geologists: Littleton, CO, USA, 1999; Volume 6, pp. 133–160. [Google Scholar]
- Büyüksaraç, A.; Eyisüren, O.; Bektaş, Ö.; Karaca, Ö. Bedrock depth calculation of Çanakkale (Turkey) basin using Rayleigh ellipticity and microgravity survey. Geofis. Int. 2023, 62, 387–401. [Google Scholar] [CrossRef]
- DeNicola, D.M. A review of diatoms found in highly acidic environments. Hydrobiologia 2000, 433, 111–122. [Google Scholar] [CrossRef]
- Mann, D.G.; Trobajo, R.; Sato, S.; Li, C.; Witkowski, A.; Rimet, F.; Ashworth, M.P.; Hollands, M.R.; Theriot, E.C. Ripe for reassessment: A synthesis of available molecular data for the speciose diatom family Bacillariaceae. Mol. Phylogenetics Evol. 2021, 158, 106985. [Google Scholar] [CrossRef] [PubMed]
- Luis, A.T.; Córdoba, F.; Antunes, C.; Loayza-Muno, R.; Grande, J.A.; Silve, B.; Daiz-Curiel, J.; da Silva, E.F. Extremely Acidic Eukaryotic (Micro) Organisms: Life in Acid Mine Drainage Polluted Environments—Mini-Review. Int. J. Environ. Res. Public Health 2022, 19, 376. [Google Scholar] [CrossRef]
- Hustedt, F. Bacillaiophyta (Diatomeae) zweite auflage. In Die Süsswasser-Flora Mitteleuropas; Pascher, A., Ed.; Fischer: Jena, Germany, 1930; pp. 1–466. [Google Scholar]
- Rabenhorst, L. Die algen sachsens. Resp. Mittel-Europa’s gesammelt und herausgegeben von Dr. L. Rabenhorst, 1848–1860. Dec. 1–100. No. 1–1000. National University of Ireland: Galway, Ireland.
- Grunow, A. Die österreichischen diatomeen nebst Anschluss einiger neuen arten von andern lokalitäten und arten. Verhandlungen Der Kais. Königlichen Zool. Bot. Gessellschaft Wien 1862, 12, 315–472, 545–588. [Google Scholar]
- Rabenhorst, L. Erklärung der Tafel VI. Hedwigia 1860, pp. 2–40.
- Rabenhorst, L. Die Algen Europas, Fortsetzung der Algen Sachsens, Resp. Mittel-Europas. 1861-1882, Decades I-CIX. pp. 1–1600.
- Hustedt, F. Die Diatomeenflora des Küstengebietes der Nordsee vom Dollart bis zur Elbemündung. I. Die Diatomeenflora in den Sedimenten der Unteren Ems Sowie auf den Watten in der Leybucht, des Memmert und bei der Insel Juist. Adhandlungen des Naturwissenschaftlichen Verein zu Bremen, 1861–1882. 31(2/3); Naturwissenschaftlichen Verein zuBremen: Bremen, Germany, 1939; pp. 571–677. [Google Scholar]
- Brake, S.S.; Hasiotis, S.T.; Dannelly, H.K. Diatoms in acid mine drainage and their role in the formation of iron-rich stromatolites. Geomicrobiol. J. 2004, 21, 331–340. [Google Scholar] [CrossRef]
- Taylor, J.C.; Harding, W.R.; Archibald, C.G.M. A Methods Manuel for the Collection, Preparation and Analysis of Diatom Samples; DH Environmental Consulting: Seattle, DC, USA, 2006. [Google Scholar]
- Turland, N.J.; Wiersema, J.H.; Barrie, F.R.; Greuter, W.; Hawksworth, D.L.; Herendeen, P.; Knapp, S.S.; Kusber, W.-H.; Li, D.-Z.K.; Marhold, K.; et al. International Code of Nomenclature for algae, fungi, and plants (Shenzhen Code). Regnum Veg. 2018, 159, 254. [Google Scholar]
- Swift, E. Cleaning diatom frustules with ultraviolet radiation and peroxide 1. Phycologia 1967, 6, 161–163. [Google Scholar] [CrossRef]
- Round, F.E.; Crawford, R.M.; Mann, D.G. The Diatoms: Biology and Morphology of the Genera; Cambridge University Press: Cambridge, UK, 1990; p. 747. [Google Scholar]
- Hustedt, F. Die diatomeenflora des fluß-systems der weser im gebiet der Hansestadt Bremen. Abh. Naturwissenchaftlichen Ver. Brem. 1957, 34, 181–440. [Google Scholar]
- Lange-Bertalot, H. Zur systematik, taxonomie und ökologie des abwasserspezifisch wichtigen formenkreises um “Nitzschia thermalis”. Nova Hedwig. 1978, 30, 635–652. [Google Scholar] [CrossRef]
- Cleve, P.T.; Grunow, A. Beiträge zur kenntniss der arctischen diatomeen. Kongliga Sven. Vetensk. Akad. Handl. 1880, 17, 1–121. [Google Scholar]
- Hustedt, F. Die diatomeenflora von Poggenpohls moor bei dötlingen in Oldenburg. Abh. Vorträgen Bremer Wissenchaftlichen Gessellschaft 1934, 8/9, 362–403. [Google Scholar]
- Solak, C.N.; Gastineau, R.; Lemieux, C.; Turmel, M.; Gorecka, E.; Trobajo, R.; Rybak, M.; Yılmaz, E.; Witkowski, A. Nitzschia anatoliensis sp. nov., a cryptic diatom species from the highly alkaline Van Lake (Turkey). PeerJ 2021, 9, e12220. [Google Scholar] [CrossRef]
- Cholnoky, B.J. Beiträge zur kenntnis der ökologie der diatomeen in den Swartkops-Bachne nahe Port Elizabeth (Südost-Kaapland). Osterr. Bot. Z. Hydrobiol. 1960, 16, 229–287. [Google Scholar]
- Trobajo, R.; Rovira, L.; Ector, L.; Wetzel, C.E.; Kelly, M.; Mann, D.G. Morphology and identification of some small ecollogically important small Nitzschia species. Diatom Res. 2013, 28, 37–59. [Google Scholar] [CrossRef]
- Krammer, K. The genus Pinnularia. In Diatoms of Europe; Lange-Bertalot, H., Ed.; A.R.G. Gantner Verlag: Frankfurt, Germany, 2000; Volume 3, pp. 1–703. [Google Scholar]
- van Dam, H.; Mertens, D.; Sinkeldam, J. A coded checklist and ecological indicator values of freshwater diatoms from The Netherlands. Neth. J. Aquat. Ecol. 1994, 28, 117–133. [Google Scholar]
- Maidana, N.I.; Romero, O.E. Diatoms from the hypersaline “La Amarga” lake (La Pampa, Argentina). Cryptogam. Algol. 1995, 16, 173–188. [Google Scholar]
- Krammer, K.; Lange-Bertalot, H. Bacillariophyceae, 2. Teil: Bacillariaceae, Epithemiaceae, Surirellaceae. Süßwasserflora Mitteleur. 1988, 2, 1–596. [Google Scholar]
- Poulin, M.; Bérard-Therriault, L.; Cardinal, A.; Hamilton, P.B. Les diatomées (Bacillariophyta) benthique de substrats durs des eaux marines et saumâtres du Québec. 9. Bacillariaceae. Nat. Can. 1990, 117, 73–101. [Google Scholar]
- Antoniades, D.; Hamilton, P.B.; Douglas, M.S.V.; Smol, J.P. Diatoms of North America: The Freshwater Floras of Prince Patrick, Elef Ringnes and Northern Ellesmere Islands from the Canadian Arctic Archipelago, 17th ed.; Iconographi Diatomologica; A.R.G. Gantner: Frankfurt, Germany, 2008; pp. 1–649. [Google Scholar]
- Kützing, F.T. Die Kieselschaligen Bacillarien Oder Diatomeen; Taylor & Francis: Abingdon, UK, 1844; pp. 1–152. [Google Scholar]
- Van Heurck, H. Synopsis der Diatomées de Belgigue Atlas; Ducaju & Cie, Anvers: Meulebeke, Belgium, 1881; pp. 31–77. [Google Scholar]
- Lange-Bertalot, H. Eine revision zur taxonomie der Nitzschia lanceolatae Grunow. Die “klassischen” bis 1930 beschribenen Sisswasserarten Europas. Nova Hedwig. 1976, 28, 253–307. [Google Scholar]
- Patrick, R. Ecology of freshwater diatoms and diatom communities. In The Biology of Diatoms; Werner, D., Ed.; University of California Press: Berkeley, CA, USA, 1977; pp. 284–332. [Google Scholar]
- Lange-Bertalot, H.; Bąk, M.; Witkowski, A.; Tagliaventi, N. Eunotia and some related genera. In Diatoms of Europe; Lange-Bertalot, H., Ed.; A.R.G. Gantner Verlag: Frankfurt, Germany, 2011; Volume 6, pp. 1–747. [Google Scholar]
- Lessmann, D.; Fyson, A.; Nixdorf, B. Phytoplankton of the extremely acidic mining lakes of Lusatia (Germany) with pH < 3. Hydrobiologia 2000, 433, 123–128. [Google Scholar]
- Whitton, B.A.; Diaz, B.M. Influence of environmental factors on photosynthetic species composition in highly acidic waters. Verh. Int. Ver. Limnol. 1981, 21, 1459–1465. [Google Scholar] [CrossRef]





Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sevindik, T.O.; Hamilton, P.B.; Solak, C.N.; Yilmaz, E.; Güzel, U. Three New Nitzschia (Bacillariophyceae) Species from Highly Acidic Artificial Lakes in Çanakkale, Türkiye. Water 2023, 15, 3784. https://doi.org/10.3390/w15213784
Sevindik TO, Hamilton PB, Solak CN, Yilmaz E, Güzel U. Three New Nitzschia (Bacillariophyceae) Species from Highly Acidic Artificial Lakes in Çanakkale, Türkiye. Water. 2023; 15(21):3784. https://doi.org/10.3390/w15213784
Chicago/Turabian StyleSevindik, Tuğba Ongun, Paul Brian Hamilton, Cüneyt Nadir Solak, Elif Yilmaz, and Uğur Güzel. 2023. "Three New Nitzschia (Bacillariophyceae) Species from Highly Acidic Artificial Lakes in Çanakkale, Türkiye" Water 15, no. 21: 3784. https://doi.org/10.3390/w15213784
APA StyleSevindik, T. O., Hamilton, P. B., Solak, C. N., Yilmaz, E., & Güzel, U. (2023). Three New Nitzschia (Bacillariophyceae) Species from Highly Acidic Artificial Lakes in Çanakkale, Türkiye. Water, 15(21), 3784. https://doi.org/10.3390/w15213784







