Phytoremediation Potential of the Coastal Plain Willow Salix caroliniana for Heavy Metals with Emphasis on Mercury
Abstract
:1. Introduction
2. Materials and Methods
2.1. Tree Sampling
2.2. Sample Preparation Methods
2.3. Analysis Using the ICP-MS
3. Results
3.1. Trunk Heavy Metals Data
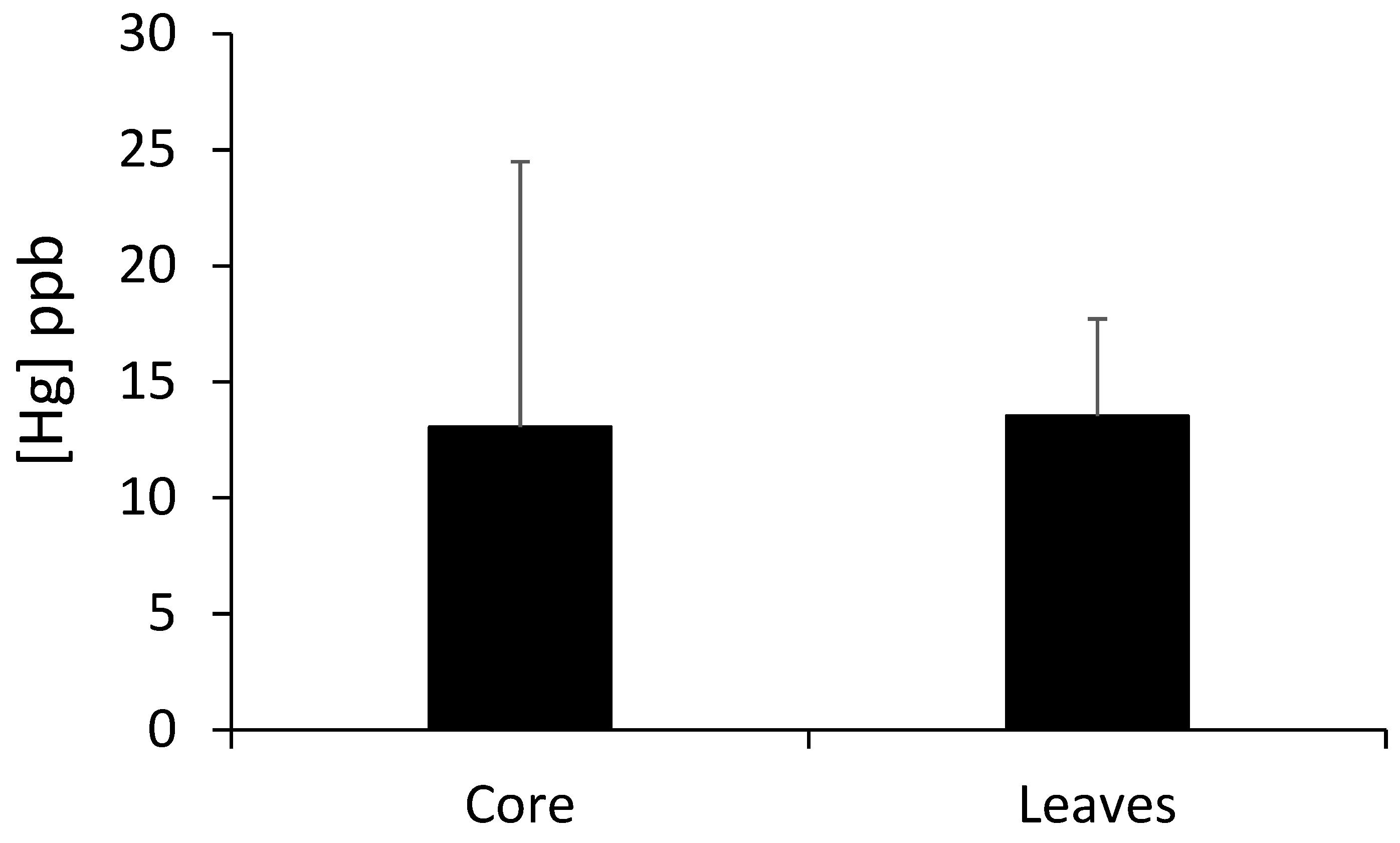
3.2. Quantitative Comparison of the Mercury Concentrations in the Wood and Leaves
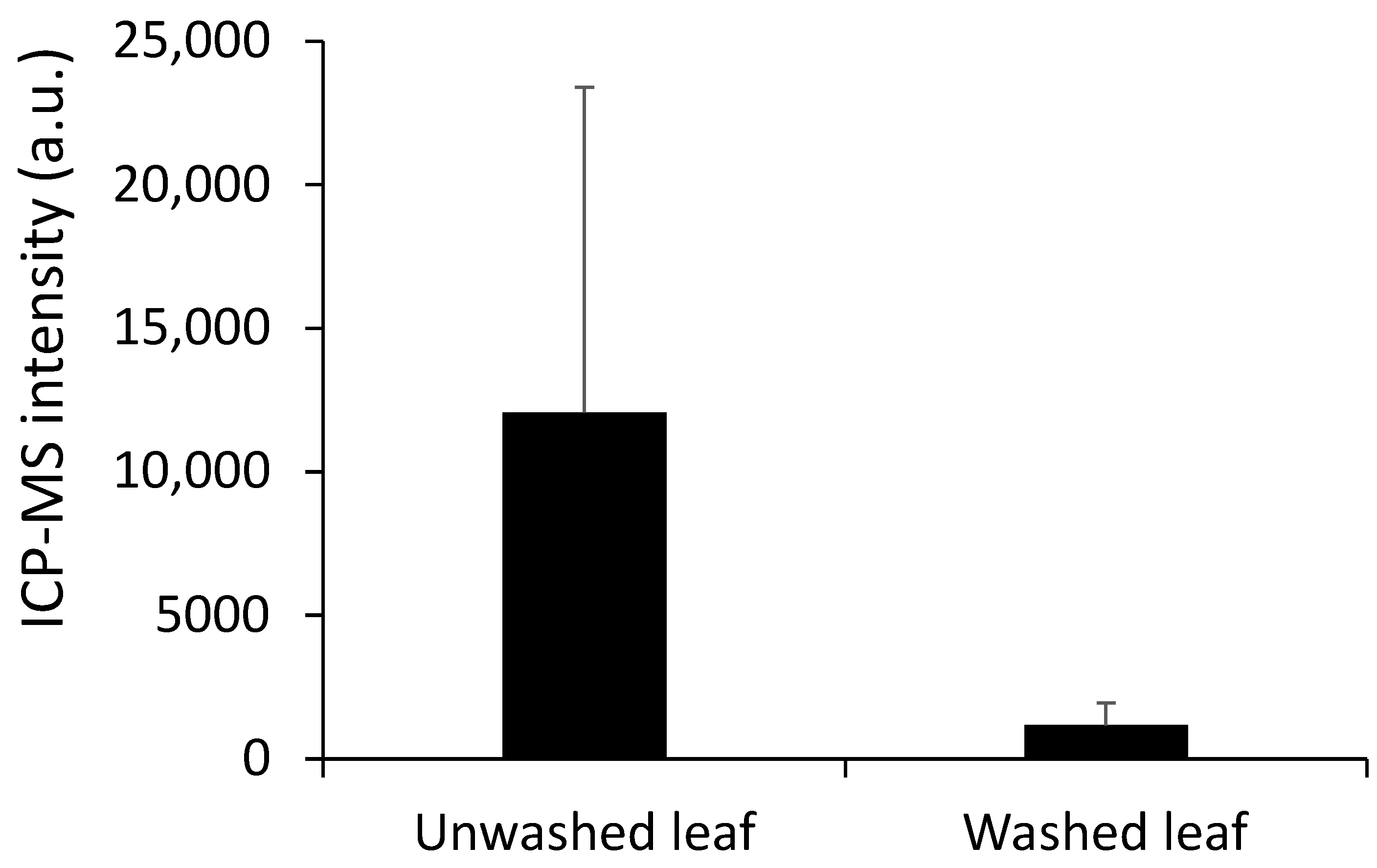
3.3. Difference in Measured Mercury between Unwashed and Washed Leaf Material
3.4. Statistical Analyses for Comparison of the Data
4. Discussion
4.1. Possible Mercury Transfer within S. caroliniana from the Soil and Groundwater
4.2. Mercury in the Leaves of S. caroliniana
4.3. Possible Sources of Mercury for Leaf Surface Deposition
4.4. Usefulness of Willow (Salix) for Mercury Contamination Phytoremediation
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wang, Q.; Kim, D.; Dionysiou, D.D.; Sorial, G.A.; Timberlake, D. Sources and remediation for mercury contamination in aquatic systems—A literature review. Environ. Pollut. 2004, 131, 323–336. [Google Scholar] [CrossRef] [PubMed]
- Agency for Toxic Substances and Disease Registry (ASTDR). Toxicological Profile for Mercury (Draft for Public Comment); U.S. Department of Health and Human Services, Public Health Service: Atlanta, GA, USA, 2022. Available online: https://wwwn.cdc.gov/TSP/ToxProfiles/ToxProfiles.aspx?id=115&tid=24 (accessed on 12 March 2022).
- Kumar, V.; Sharma, A.; Cerdal, A. Heavy Metals in the Environment: Impacts, Assessment and Remediation; Elsevier: Amsterdam, The Netherlands, 2021. [Google Scholar]
- Gworek, B.; Dmechowski, W.; Baczewska-Dabrowska, A.H. Mercury in the terrestrial environment: A review. Environ. Sci. Eur. 2020, 32, 128. [Google Scholar] [CrossRef]
- Li, Y.; Cai, Y. Progress in the study of mercury methylation and demethylation in aquatic environments. Chin. Sci. Bull. 2012, 58, 177–185. [Google Scholar] [CrossRef]
- Kim, K.-H.; Kabirb, E.; Ara Jahanc, S. A review on the distribution of Hg in the environment and its human health impacts. J. Hazard Mat. 2016, 306, 376–385. [Google Scholar] [CrossRef]
- Bank, M.S. The mercury science policy interface history, evolution and progress of the Minamata Convention. Sci. Total Environ. 2000, 722, 137832. [Google Scholar] [CrossRef] [PubMed]
- Mousavi, A.; Chávez, R.D.; Ali, A.-M.S.; Cabaniss, S.E. Mercury in natural waters: A mini-review. Environ. Forensics 2011, 12, 14–18. [Google Scholar] [CrossRef]
- Coufalik, P.; Krmiček, L.; Zvĕřina, O.; Meszarosova, N.; Hladil, J.; Komárek, J. Model of mercury flux associated with volcanic activity. Bull. Environ. Contam. Toxicol. 2018, 101, 549–553. [Google Scholar] [CrossRef]
- Guentzel, J.L.; Landing, W.M.; Gill, G.A.; Pollman, C.D. Processes influencing rainfall deposition of mercury in Florida. Environ. Sci. Tech. 2001, 35, 863–873. [Google Scholar] [CrossRef]
- Sillman, S.; Marsik, F.; Dvonch, J.T.; Keeler, G.J. Atmospheric deposition of mercury in Florida, USA: Local versus global sources and models versus measurements. E3S Web Conf. 2013, 1, 07708. [Google Scholar] [CrossRef]
- Friedli, H.R.; Arellano, A.F.; Cinnirella, S.; Pirrone, N. Mercury emissions from global biomass burning: Spatial and temporal distribution. In Mercury Fate and Transport in the Global Atmosphere: Emissions, Measurements and Models; Pirrone, N., Mahaffey, K.E., Eds.; Springer Science and Business Media: Berlin, Germany, 2009; Chapter 8, pp. 193–220. [Google Scholar]
- Chen, J.; Yang, Z.M. Mercury toxicity, molecular response and tolerance in higher plants. BioMetals 2012, 25, 847–857. [Google Scholar] [CrossRef]
- Velásquez-Riaño, M.; Benavides-Otaya, H.D. Bioremediation techniques applied to aqueous media contaminated with mercury. Crit. Rev. Biotechnol. 2015, 36, 1124–1130. [Google Scholar] [CrossRef] [PubMed]
- Sundseth, K.; Pacyna, J.; Pacyna, E.; Pirrone, N.; Thorne, R. Global sources and pathways of mercury in the context of human health. Intern. J. Environ. Res. Public Health 2017, 14, 105. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Kang, S.; Yin, R.; Ram, K.; Liu, X.; Lu, H.; Guo, J.; Chen, S.; Tripathee, L. Desert dust as a significant carrier of atmospheric mercury. Environ. Pollut. 2020, 267, 115442. [Google Scholar] [CrossRef]
- Leclerc, M.; Harrison, M.C.; Storck, V.; Planas, D.; Amyot, M.; Walsh, D.A. Microbial diversity and mercury methylation activity in periphytic biofilms at a run-od-river hydroelectric dam and constructed wetlands. mSphere 2021, 6, e00021-21. [Google Scholar] [CrossRef]
- Boening, D.W. Ecological effects, transport, and fate of mercury: A general review. Chemosphere 2000, 40, 1335–1351. [Google Scholar] [CrossRef] [PubMed]
- Smieja-Król, B.; Fiałkiewicz-Kozieł, B.; Sikorski, J.; Palowsky, B. Heavy metal behavior in peat—A mineralogical perspective. Sci. Total Environ. 2010, 408, 5924–5931. [Google Scholar] [CrossRef]
- Liu, L.; Li, W.; Song, W.; Guo, M. Remediation techniques for heavy metal-contaminated soils: Principles and applicability. Sci. Total Environ. 2018, 633, 206–219. [Google Scholar] [CrossRef]
- Labrecque, M.; Hu, Y.; Vincent, G.; Shang, K. The use of willow microcuttings for phytoremediation in a copper, zinc and lead contaminated field trial in Shanghai, China. Intern. J. Phytoremediation 2020, 22, 1331–1337. [Google Scholar] [CrossRef]
- Kuzovkina, Y.A.; Volk, T.A. The characterization of willow (Salix L.) varieties for use in ecological engineering applications: Co-ordination of structure, function and autecology. Ecol. Engineer. 2009, 35, 1178–1189. [Google Scholar] [CrossRef]
- Pulford, I.D.; Watson, C. Phytoremediation of heavy metal-contaminated land by trees—A review. Environ. Int. 2003, 29, 529–540. [Google Scholar] [CrossRef]
- Abdelsalam, I.M.; Elshobary, M.E.; Eladawy, M.M.; Nagah, M. Utilization of multi-tasking non-edible plants for phytoremediation and bioenergy source-A review. Phyton 2019, 88, 69–90. [Google Scholar] [CrossRef]
- Liu, Z.; Chen, B.; Wang, L.; Urbanovich, O.; Nagorskaya, L.; Li, X.; Tang, L. A review on phytoremediation of mercury contaminated soils. J. Hazard. Mater. 2020, 400, 123138. [Google Scholar] [CrossRef] [PubMed]
- Patra, M.; Sharma, A. Mercury toxicity in plants. Bot. Rev. 2000, 66, 379–422. [Google Scholar] [CrossRef]
- Algreen, M.; Trapp, S.; Rein, A. Phytoscreening and phytoextraction of heavy metals at Danish polluted sites using willow and popular trees. Environ. Sci. Pollut. Res. Intern. 2013, 21, 8992–9001. [Google Scholar] [CrossRef] [PubMed]
- Kacalkova, L.; Tlustos, P.; Szakpova, J. Phytoextraction of risk elements by willow and poplar trees. Intern. J. Phytoremediation 2015, 17, 414–421. [Google Scholar] [CrossRef]
- Argus, G.W. The Gus Salix (Salicaceae) in the Southern United States; Systematic Botany Monographs; American Society of Plant Taxonomists: St. Louis, MO, USA, 1986; pp. 1–170. [Google Scholar]
- Mabberley, D.J. The Plant Book; Cambridge University Press: Cambridge, UK, 1997. [Google Scholar]
- Dickman, D.I.; Huzovkina, J. Populars and willows of the world, with emphasis on silviculturally important species. In Poplars and Willows: Trees for Society and the Environment; Isebrands, J.G., Richardson, J., Eds.; CAB International and Food and Agriculture Organization of the United Nations: Rome, Italy, 2014; pp. 8–91. [Google Scholar] [CrossRef]
- Wagner, N.; Li, H.; Hörandi, E. The evolutionary history, diversity, and ecology of willows (Salix L.) in the European Alps. Diversity 2021, 13, 146. [Google Scholar] [CrossRef]
- Zacchini, M.; Pietrini, F.; Mugnozza, S.M.; Iori, V.; Pietrosanti, L.; Massacci, A. Cadnium accumulation and tolerance in Populus nigra and Salix alba. Water Air Soil Pollut. 2009, 55, 383–386. [Google Scholar] [CrossRef]
- Wang, S.; Shi, X.; Sun, H.; Chen, Y.; Pan, H.; Yang, X.; Rafig, T. Variations in metal tolerance and accumulation in three hydroponically cultivated varieties of Salix intergra treated with lead. PLos ONE 2014, 9, e108568. [Google Scholar] [CrossRef]
- Ruiz, O.N.; Daniell, H. Genetic engineering to enhance mercury phytoremediation. Curr. Opin. Biotechnol. 2009, 20, 213–219. [Google Scholar] [CrossRef]
- Pulford, I.D.; Riddell-Black, D.; Stewart, C. Heavy metal uptake by willow clones from sewage sludge-treated soil. Intern. J. Phytoremediation 2002, 4, 59–72. [Google Scholar] [CrossRef]
- Dickinson, N.M.; Pulford, I.D. Cadmium phytoextraction using short-rotation coppice Salix: The evidence trail. Environ. Int. 2005, 31, 609–613. [Google Scholar] [CrossRef] [PubMed]
- Maxted, A.P.; Black, C.R.; West, H.M.; Crout, N.M.J.; McGrath, S.P.; Young, S.D. Phytoextraction of cadmium and zinc by Salix from soil historically amended with sewage sludge. Plant Soil 2007, 290, 157–172. [Google Scholar] [CrossRef]
- Meers, E.; Vandecasteele, B.; Ruttens, A.; Vangronsveld, J.; Tack, F.M.G. Potential of five willow species (Salix sp) for phytoextraction of heavy metals. Environ. Environ. Bot. 2007, 60, 57–68. [Google Scholar] [CrossRef]
- Jensen, J.K.; Holm, P.E.; Nejrup, J.; Larsen, M.B.; Borggaard, O.K. The potential of willow for remediation of heavy metals polluted alcareous urban soils. Environ. Pollut. 2009, 157, 931–937. [Google Scholar] [CrossRef]
- Konlechner, C.; Türktas, M.; Langer, I.; Vaculík, M.; Wenzel, W.W.; Ouschenreiter, M.; Hauser, M.T. Expression of zinc and cadmium responsive genes in leaves of willow (Salix caprea L.) genotypes with different accumulations characteristics. Environ. Pollut. 2013, 178, 121–127. [Google Scholar] [CrossRef]
- Workman, R.W. Growing Native; Banyan Books; The Sanibel-Captiva Conservation Foundation: Sanibel, FL, USA, 1981. [Google Scholar]
- Ewel, K.C. Swamps. In Ecosystems of Florida, 2nd ed.; Myers, R.L., Ewel, J.J., Eds.; University of Central Florida Press, University Presses of Florida: Gainesville, FL, USA, 1991; pp. 281–323. [Google Scholar]
- Marmiroli, M.; Pietrini, F.; Maestri, E.; Zacchini, M.; Marmiroli, N.; Massacci, A. Growth, physiological and molecular traits in Salicaceae trees investigated for phytoremediation of heavy metals and organics. Tree Physiol. 2011, 31, 1319–1334. [Google Scholar] [CrossRef]
- Sas-Nowosielska, A.; Galimska-Stypa, R.; Kucharski, R.; Zielonka, U.; Małkowski, E.; Gray, L. Remediation aspect of microbial changes of plant rhizosphere in mercury contaminated soil. Environ. Monit. Assess. 2007, 137, 101–109. [Google Scholar] [CrossRef]
- Niu, Z.; Zhang, X.; Wang, S.; Ci, Z.; Kong, X.; Wang, Z. The linear accumulation of atmospheric mercury by vegetable and grass leaves: Potential biomonitors for atmospheric mercury pollution. Environ. Sci. Pollution Res. 2013, 20, 6337–6343. [Google Scholar] [CrossRef]
- Siwik, E.I.H.; Campbell, L.M.; Mierle, G. Distribution and trends of mercury in deciduous tree cores. Environ. Pollut. 2010, 158, 2067–2073. [Google Scholar] [CrossRef]
- Schmidt, U. Enhancing phytoextraction. J. Environ. Qual. 2003, 32, 1939. [Google Scholar] [CrossRef]
- Gill, R.; Karlen, M.; Weber, M.; Rüegg, J.; Barmettler, K.; Bieter, H.; Boiven, P.; Kretzchmar, R. Speciation and mobility of mercury in soils contaminated by legacy emissions from a chemical factory in the Rhône valley in canton of Valais, Switzerland. Soil Syst. 2018, 2, 44. [Google Scholar] [CrossRef]
- Meng, B.; Feng, X.; Qiu, G.; Wang, D.; Li, P.; Shang, L.; Sommar, J. Distribution patterns on inorganic mercury and methylmercury in tissues of rice (Oryza sativa) plants and possible bioaccumulation pathways. Agr. Food Chem. 2010, 58, 4951–4958. [Google Scholar] [CrossRef] [PubMed]
- Gustin, M.S.; Ericksen, J.A.; Schorran, D.E.; Johnson, D.W.; Lindberg, S.E.; Coleman, J.S. Application of controlled mesocosms for understanding mercury air-soil-plant exchange. Environ. Sci. Technol. 2004, 38, 6044–6050. [Google Scholar] [CrossRef] [PubMed]
- Fantozzi, L.; Ferrara, R.; Dini, F.; Tamburello, L.; Pirrone, H.; Sprovien, F. Study on the atmospheric reduction of mercury emissions from mine waste enriched soils through native grass cover in the Mt. Amiata region of Italy. Environ. Res. 2013, 125, 69–74. [Google Scholar] [CrossRef] [PubMed]
- Manzur, M.; Mitchell, C.P.J.; Eckley, C.S.; Eggert, S.L.; Kolka, P.K.; Sebestyen, S.D.; Swain, E.B. Gaseous mercury fluxes from forest soils in response to forest harvesting intensity: A field manipulation experiment. Sci. Total Environ. 2014, 496, 678–687. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Greger, M. Clonal differences in mercury tolerance, accumulation, and distribution in willow. J. Environ. Qual. 2004, 33, 1779–1785. [Google Scholar] [CrossRef]
- Frescholtz, T.F.; Gustin, M.S.; Schorran, D.E.; Fernandez, G.C. Assessing the source of mercury in follar tissue of quaking aspen. Environ. Toxicol. Chem. 2003, 22, 2114–2119. [Google Scholar] [CrossRef]
- Gustin, M.S.; Lindberg, S.E. Terrestrial Hg fluxes: Is the next exchange up, down or neither? In Dynamic of Mercury Pollution on Regional and Global Scales; Pirrone, N., Mahaffey, K.E., Eds.; Springer: Boston, MA, USA, 2005; pp. 241–259. [Google Scholar] [CrossRef]
- Assad, M.; Parelle, J.; Cazaux, D.; Gimbert, F.; Chalot, M.; Tatin-Froux, F. Mercury uptake in popular leaves. Chemosphere 2016, 146, 1–7. [Google Scholar] [CrossRef]
- Arnold, J.; Gustin, M.S.; Weisberg, P.J. Evidence for nonstromatal uptake of Hg by aspen and translocation of Hg from foliage to tree rings in Austrian pine. Environ. Sci. Technol. 2018, 52, 1174–1182. [Google Scholar] [CrossRef]
- Laacouri, A.; Nater, E.A.; Kolka, P.K. Distribution and uptake dynamics of mercury leaves of common deciduous tree species in Minnesota. Environ. Sci. Technol. 2013, 47, 10462–10470. [Google Scholar] [CrossRef]
- Pleijel, H.; Klingberg, J.; Nerentorp, M.; Broberg, M.C.; Nyirambagutse, B.; Munthe, J.; Wallin, G. Mercury accumulation in leaves of different plant types—The significance of tissue age and leaf area. Biogeoscience 2021, 18, 6313–6328. [Google Scholar] [CrossRef]
- Rood, B.E.; Gottgens, J.F.; Delfino, J.J.; Earle, C.D.; Crisman, T.L. Mercury accumulation trends in Florida Everglades and Savannas marsh flooded soils. Water Air Soil Pollut. 1995, 80, 981–990. [Google Scholar] [CrossRef]
- Tsiros, I.X.; Ambrose, R.B. Environmental screening modeling of mercury in the upper Everglades of South Florida. J. Environ Sci. Health Part A 1998, 33, 497–525. [Google Scholar] [CrossRef]
- Cohen, M.J.; Lamal, S.; Osborne, T.Z.; Bonzongo, J.C.J.; Newman, S.; Reddy, K.R. Soil total mercury concentrations across the Greater Everglades. Soil Sci. Am. J. 2009, 73, 675–685. [Google Scholar] [CrossRef]
- Guentzel, J.L.; Landing, W.M.; Gill, G.A.; Pollman, C.D. Atmospheric deposition of mercury in Florida: The FAMAS Project (199-1994). Water Air Soil Pollut. 1995, 80, 393–402. [Google Scholar] [CrossRef]
- Landing, W.M.; Perry, J.J., Jr.; Guentzel, J.L.; Gill, G.A.; Pollman, C.D. Relationships between the atmospheric deposition of trace elements, major ions, and mercury in Florida: The FMAS Project (1992–1993). Water Air Soil Pollut. 1995, 80, 343–352. [Google Scholar] [CrossRef]
- Pollman, C.D.; Gill, G.A.; Landing, W.M.; Guentzel, J.L.; Bare, D.; Porcella, D.; Zillioux, E.; Atkeson, T. Overview of the Florida atmospheric mercury study (FAMS). Water Air Soil Pollut. 1995, 80, 285–290. [Google Scholar] [CrossRef]
- Gustin, M.S. Are mercury emissions from geologic sources significant? A status report. Sci. Total Environ. 2003, 304, 153–167. [Google Scholar] [CrossRef]
- Stmenkovic, J.; Gustin, M.S. Evaluation of use of ExoCell technology for quantifying total gaseous mercury fluxes over background substrates. Atmos. Environ. 2007, 41, 3702–3712. [Google Scholar] [CrossRef]

| Trunk Sample Number | Mercury Concentration (ppb) | Lead Concentration (ppb) | Arsenic Concentration (ppb) | Cadmium Concentration (ppb) |
|---|---|---|---|---|
| 1 | 0.18 | 23.06 | 2.51 | 22.02 |
| 2 | 0.15 | 90.06 | BDL | 55.69 |
| 3 | 0.32 | 478 | 7.05 | 10.05 |
| 4 | 0.63 | 14.22 | 2.48 | 25.92 |
| 5 | 0.30 | 1095 | 13.38 | 15.16 |
| 6 | 0.50 | 251.5 | 5.29 | 18.61 |
| 7 | 1.40 | 173.4 | 6.01 | 31.19 |
| 8 | 0.37 | 844.2 | 2.28 | 6.55 |
| 9 | 0.35 | 54.49 | 3.49 | 3.12 |
| 10 | 0.30 | 29.49 | 4.22 | 2.45 |
| 11 | 0.76 | 91.22 | 21.68 | 2.11 |
| 12 | 0.74 | 400.5 | 7.45 | 44.16 |
| 13 | 0.12 | 184.9 | 2.62 | 1.96 |
| 14 | 0.79 | 33.87 | 6.77 | 7.96 |
| Mean | 0.49 | 268.9 | 6.09 | 17.64 |
| Standard Deviation | 0.33 | 320.7 | 5.34 | 16.11 |
| Variance | 0.11 | 102,825 | 28.55 | 259.4 |
| Tree Leaf Sample Number | Mercury Concentration (ppb) | Lead Concentration (ppb) | Arsenic Concentration (ppb) | Cadmium Concentration (ppb) |
|---|---|---|---|---|
| 1 | 1.25 | 3.88 | 14.28 | 15.85 |
| 2 | 0.19 | 4.68 | 6.22 | 9.74 |
| 3 | 1.36 | 4.10 | 4.62 | 2.52 |
| 4 | 0.84 | 1.12 | 1.82 | 18.83 |
| 5 | 2.14 | 0.34 | 5.66 | 8.95 |
| 6 | 0.69 | 0.09 | 2.59 | 4.47 |
| 7 | 1.46 | 2.27 | 3.56 | 11.37 |
| 8 | 1.36 | 1.91 | 6.68 | 2.40 |
| 9 | 1.78 | 7.14 | 8.52 | 2.70 |
| 10 | 1.40 | 2.25 | 6.97 | 2.17 |
| 11 | 0.96 | 2.13 | 12.74 | 3.44 |
| 12 | 1.81 | 0.81 | 6.45 | 6.28 |
| 13 | 1.53 | 7.19 | 5.71 | 0.37 |
| 14 | 1.24 | 1.58 | 3.69 | 4.18 |
| Mean | 1.28 | 2.80 | 6.34 | 6.66 |
| Standard Deviation | 0.48 | 2.20 | 3.41 | 5.36 |
| Variance | 0.23 | 4.86 | 11.26 | 28.69 |
| Sample Number | Trunk Mercury Concentration (ppb) | Leaf Mercury Concentration (ppb) |
|---|---|---|
| 1 | 47.09 | 20.18 |
| 2 | 17.85 | 3.97 |
| 3 | 5.91 | 15.25 |
| 4 | 4.95 | 11.25 |
| 5 | 1.69 | 8.86 |
| 6 | 9.75 | 14.91 |
| 7 | 17.76 | 10.03 |
| 8 | 14.60 | 12.70 |
| 9 | 4.37 | 16.93 |
| 10 | 17.97 | 14.03 |
| 11 | 5.58 | 17.63 |
| 12 | 8.92 | 11.79 |
| 13 | 19.02 | 19.75 |
| 14 | 7.23 | 1.86 |
| Mean | 11.91 | 13.51 |
| Standard Deviation | 11.30 | 4.26 |
| Variance | 127.80 | 18.11 |
| Leaf Sample Number | Unwashed Mercury Intensity | Washed Leaf Mercury Intensity |
|---|---|---|
| 1 | 41,011 | 2676 |
| 2 | 30,059 | 2447 |
| 3 | 17,994 | 1389 |
| 4 | 1664 | 1544 |
| 5 | 10,676 | 1594 |
| 6 | 13,025 | 1358 |
| 7 | 9302 | 1606 |
| 8 | 6673 | 351 |
| 9 | 3248 | 723 |
| 10 | 2762 | 935 |
| 11 | 4956 | 568 |
| 12 | 6006 | 975 |
| 13 | 3416 | 51 |
| 14 | 3230 | 78 |
| Mean | 12,071 | 1164 |
| Standard Deviation | 10,895 | 768 |
| Variance | 118,694,129 | 590,157 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Greenplate, R.; Thomas, S.; Danley-Thomson, A.; Missimer, T.M. Phytoremediation Potential of the Coastal Plain Willow Salix caroliniana for Heavy Metals with Emphasis on Mercury. Water 2023, 15, 3628. https://doi.org/10.3390/w15203628
Greenplate R, Thomas S, Danley-Thomson A, Missimer TM. Phytoremediation Potential of the Coastal Plain Willow Salix caroliniana for Heavy Metals with Emphasis on Mercury. Water. 2023; 15(20):3628. https://doi.org/10.3390/w15203628
Chicago/Turabian StyleGreenplate, Ryan, Serge Thomas, Ashley Danley-Thomson, and Thomas M. Missimer. 2023. "Phytoremediation Potential of the Coastal Plain Willow Salix caroliniana for Heavy Metals with Emphasis on Mercury" Water 15, no. 20: 3628. https://doi.org/10.3390/w15203628
APA StyleGreenplate, R., Thomas, S., Danley-Thomson, A., & Missimer, T. M. (2023). Phytoremediation Potential of the Coastal Plain Willow Salix caroliniana for Heavy Metals with Emphasis on Mercury. Water, 15(20), 3628. https://doi.org/10.3390/w15203628









