Toxic Algae in Inland Waters of the Conterminous United States—A Review and Synthesis
Abstract
1. Introduction
2. Ecology
2.1. Biology and Biogeography
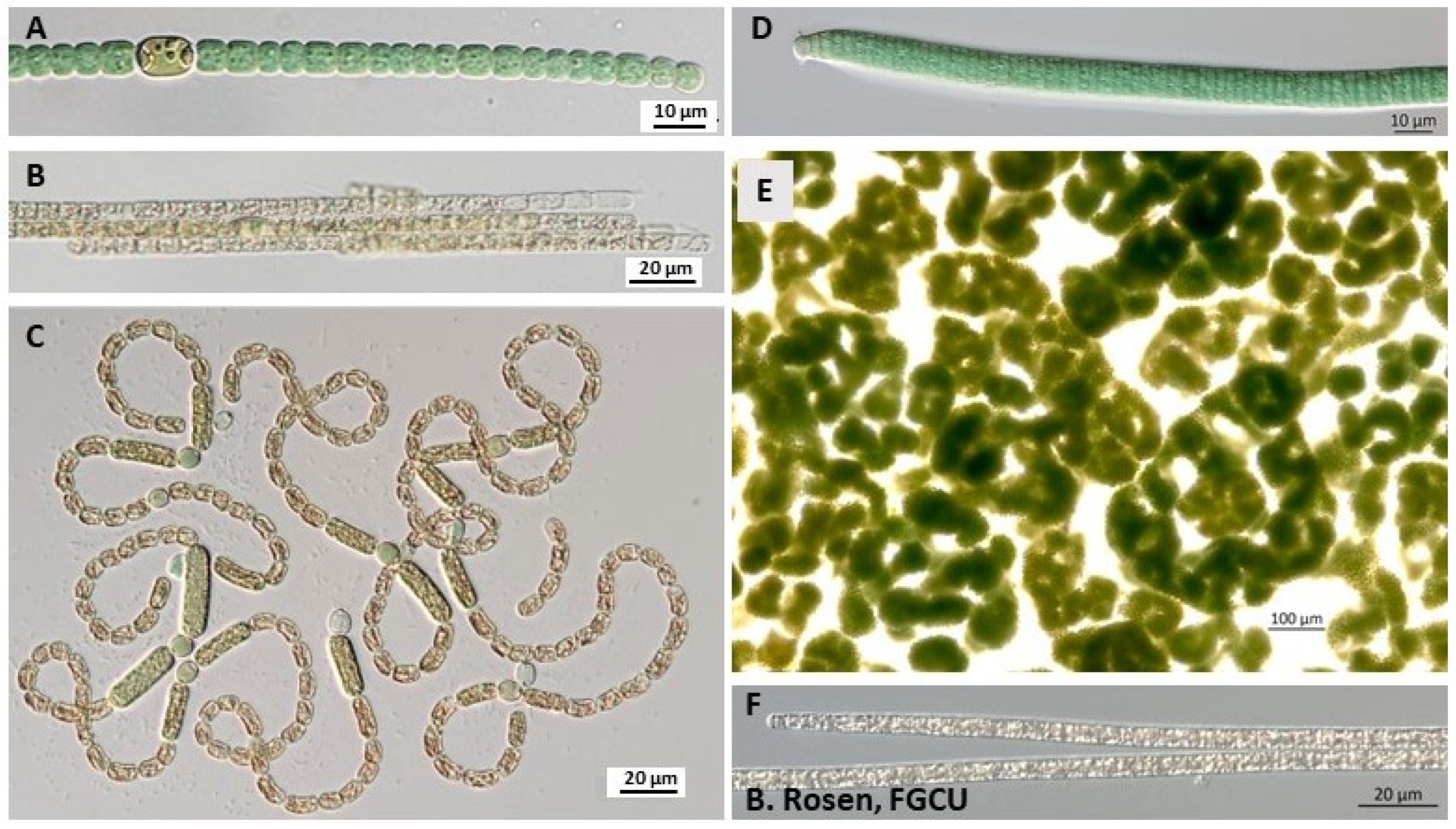
2.1.1. Cyanobacteria
2.1.2. Haptophytes
2.1.3. Euglenoids
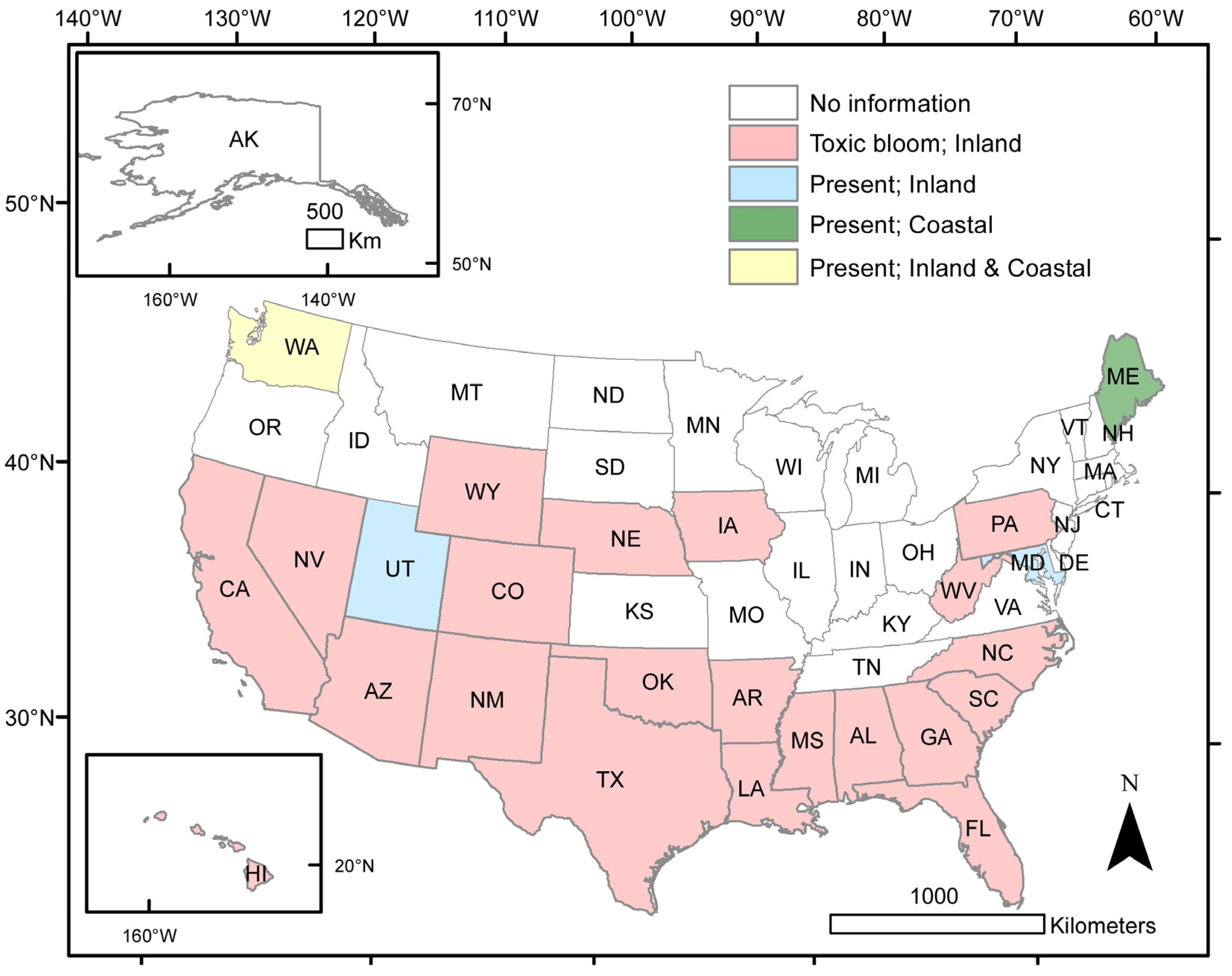
2.2. Toxic Bloom Habitats
2.2.1. Cyanobacteria
2.2.2. Haptophytes
2.2.3. Euglenoids
2.3. Influence of Land Cover and Use
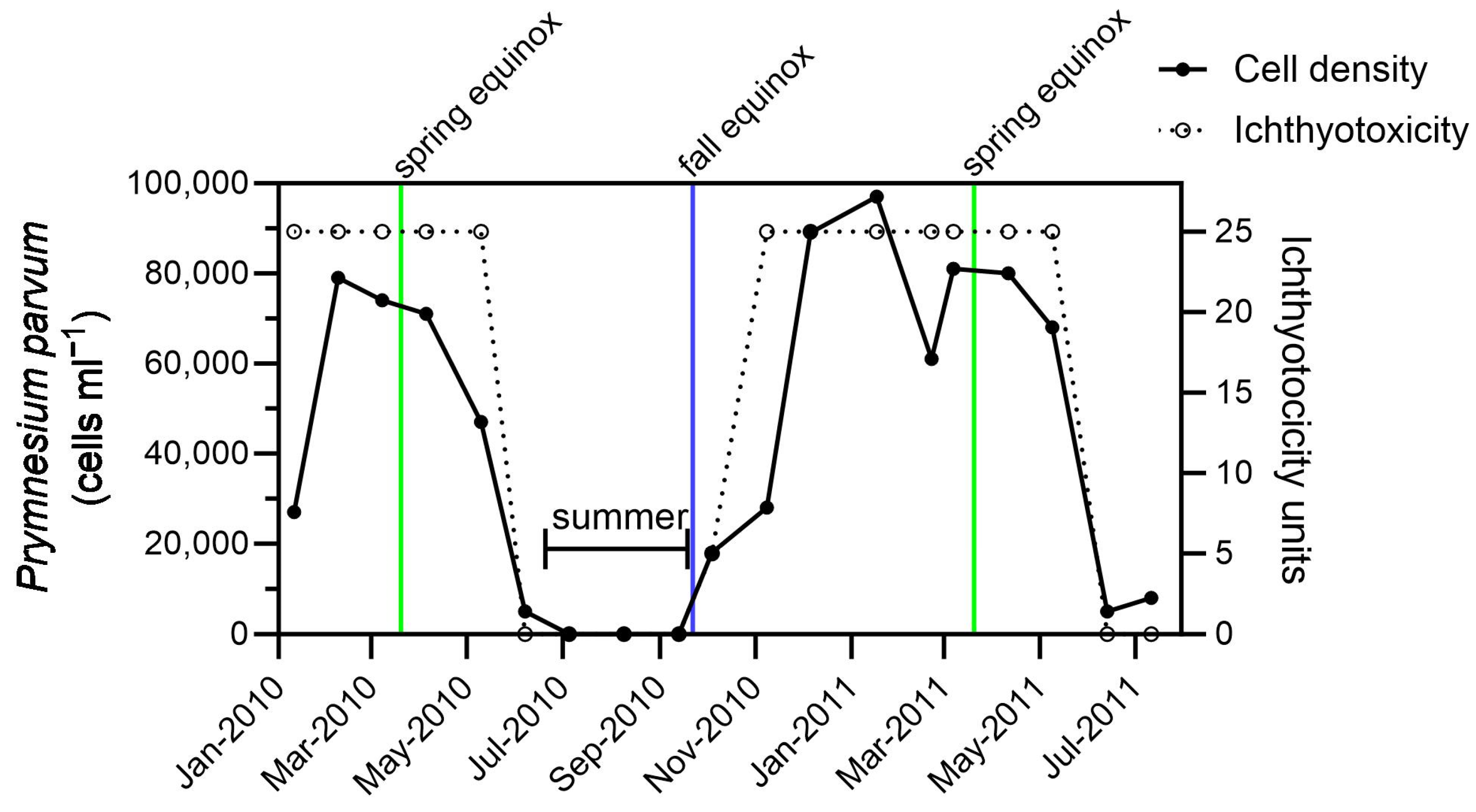
2.4. Seasonal Bloom Dynamics
3. Toxins
3.1. Anatoxins
3.2. Cylindrospermopsin
3.3. Euglenophycin
3.4. Microcystins
3.5. Prymnesins
3.6. Saxitoxins
3.7. Lesser Studied Toxins and Toxin Co-Occurrence
4. Potential for Future Expansion of Toxic Blooms in the USA
4.1. Cyanobacteria
4.2. Haptophytes
4.3. Euglenoids
5. Summary and Conclusions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Codd, G.A.; Pliński, M.; Surosz, W.; Hutson, J.; Fallowfield, H.J. Publication in 1672 of Animal Deaths at the Tuchomskie Lake, Northern Poland and a Likely Role of Cyanobacterial Blooms. Toxicon 2015, 108, 285–286. [Google Scholar] [CrossRef] [PubMed]
- Lopez, C.B.; Jewett, E.B.; Dortch, Q.; Walton, B.T.; Hudnell, H.K. Scientific Assessment of Freshwater Harmful Algal Blooms; Interagency Working Group on Harmful Algal Blooms, Hypoxia, and Human Health of the Joint Subcommittee on Ocean Science and Technology: Washington, DC, USA, 2008. [Google Scholar]
- Roelke, D.L.; Barkoh, A.; Brooks, B.W.; Grover, J.P.; Hambright, K.D.; Laclaire, J.W.; Moeller, P.D.R.; Patino, R. A Chronicle of a Killer Alga in the West: Ecology, Assessment, and Management of Prymnesium parvum Blooms. Hydrobiologia 2016, 764, 29–50. [Google Scholar] [CrossRef]
- Trainer, V.L. GlobalHAB: Evaluating, Reducing, and Mitigating the Cost of Harmful Algal Blooms: A Compendium of Case Studies. PICES Sci. Rep. 2020, 59, 107. [Google Scholar]
- Pindilli, E.J.; Loftin, K. What ’s It Worth? Estimating the Potential Value of Early Warnings of Cyanobacterial Harmful Algal Blooms for Managing Freshwater Reservoirs in Kansas, United States. Front. Environ. Sci. 2022, 10, 805165. [Google Scholar] [CrossRef]
- Nabout, J.C.; da Silva Rocha, B.; Carneiro, F.M.; Sant’Anna, C.L. How Many Species of Cyanobacteria Are There? Using a Discovery Curve to Predict the Species Number. Biodivers. Conserv. 2013, 22, 2907–2918. [Google Scholar] [CrossRef]
- Bernard, C.; Ballot, A.; Thomazeau, S.; Maloufi, S.; Furey, A.; Mankiewicz-Boczek, J.; Pawlik-Skowrońska, B.; Capelli, C.; Salmaso, N. Appendix 2: Cyanobacteria Associated with the Production of Cyanotoxins. In Handbook of Cyanobacterial Monitoring and Cyanotoxin Analysis; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2017; pp. 501–525. [Google Scholar] [CrossRef]
- Yoo, R.S.; Carmichael, W.W.; Hoehn, R.C.; Hrudey, S.E. Cyanobacterial (Blue-Green Algal) Toxins: A Resource Guide; AWWA Foundation: Denver, CO, USA, 1995. [Google Scholar]
- Hudnell, H.K. Cyanobacterial Harmful Algal Blooms: State of the Science and Research Needs; Springer: Berlin/Heidelberg, Germany, 2008; ISBN 9780387758640. [Google Scholar]
- Graham, J.L.; Foster, G.M.; Williams, T.J.; Kramer, A.R.; Harris, T.D. Occurrence of Cyanobacteria, Microcystin, and Taste-and-Odor Compounds in Cheney Reservoir, Kansas, 2001–2016; US Geological Survey: Lawrence, KS, USA, 2017. [Google Scholar]
- Anderson, D.M.; Fensin, E.; Gobler, C.J.; Hoeglund, A.E.; Hubbard, K.A.; Kulis, D.M.; Landsberg, J.H.; Lefebvre, K.A.; Provoost, P.; Richlen, M.L.; et al. Marine Harmful Algal Blooms (HABs) in the United States: History, Current Status and Future Trends. Harmful Algae 2021, 102, 101975. [Google Scholar] [CrossRef] [PubMed]
- Tisdale, E.S. Epidemic of Intestinal Disorders in Charlston, W. VA., Occuring Simulataneously with Unprecedented Water Supply Conditions. Public Health Eng. 1931, 21, 198–200. [Google Scholar] [CrossRef]
- Lippy, E.C.; Erb, J. Gastrointestinal Illness at Sewickley, Pa. AWWA J. 1976, 68, 606–610. [Google Scholar] [CrossRef]
- Hilborn, E.D.; Roberts, V.A.; Backer, L.; DeConno, E.; Egan, J.S.; Hyde, J.B.; Nicholas, D.C.; Wiegert, E.J.; Billing, L.M.; DiOrio, M.; et al. Algal Bloom-Associated Disease Outbreaks among Users of Freshwater Lakes—United States, 2009–2010. MMWR Morb. Mortal. Wkly. Rep. 2014, 63, 11–15. [Google Scholar]
- Trevino-Garrison, I.; Dement, J.; Ahmed, F.S.; Haines-Lieber, P.; Langer, T.; Ménager, H.; Neff, J.; Van Der Merwe, D.; Carney, E. Human Illnesses and Animal Deaths Associated with Freshwater Harmful Algal Blooms—Kansas. Toxins 2015, 7, 353–366. [Google Scholar] [CrossRef]
- Figgat, M.; Muscatiello, N.; Wilson, L.; Dziewulski, D. Harmful Algal Bloom–Associated Illness Surveillance: Lessons from Reported Hospital Visits in New York, 2008–2014. Am. J. Public Health 2016, 106, 440–442. [Google Scholar] [CrossRef] [PubMed]
- U.S. Centers for Disease Control and Prevention OHHAB Data. Available online: https://www.cdc.gov/habs/data/index.html (accessed on 19 April 2023).
- Chorus, I.; Bartram, J. Toxic Cyanobacteria in Water: A Guide to Their Public Health Consequences, Monitoring and Management; World Health Organization Report; World Health Organization: London, UK, 1999. [Google Scholar]
- Chorus, I.; Welker, M. Toxic Cyanobacteria in Water, 2nd ed.; Chorus, I., Welker, M., Eds.; CRC Press on Behalf of the World Health Organization: Boca Raton, FL, USA, 2021; ISBN 9781626239777. [Google Scholar]
- U.S. Center for Disease Control and Prevention One Health Harmful Algal Bloom System (OHHABs). Available online: https://www.cdc.gov/habs/ohhabs.html (accessed on 19 April 2023).
- Rose, E.T. Toxic Algae in Iowa Lakes. Proc. Iowa Acad. Sci. 1953, 60, 738–745. [Google Scholar]
- Carmichael, W.W. The Toxins of Cyanobacteria. Sci. Am. 1994, 270, 78–86. [Google Scholar] [CrossRef] [PubMed]
- Svirčev, Z.; Lalić, D.; Bojadžija Savić, G.; Tokodi, N.; Drobac Backović, D.; Chen, L.; Meriluoto, J.; Codd, G.A. Global Geographical and Historical Overview of Cyanotoxin Distribution and Cyanobacterial Poisonings. Arch. Toxicol. 2019, 93, 2429–2481. [Google Scholar] [CrossRef]
- Wacklin, P.; Hoffmann, L.; Komárek, J. Nomenclatural Validation of the Genetically Revised Cyanobacterial Genus Dolichospermum (Ralfs Ex Bornet et Flahault) Comb, Nova. Fottea 2009, 9, 59–64. [Google Scholar] [CrossRef]
- Komárek, J. Modern Taxonomic Revision of Planktic Nostocacean Cyanobacteria: A Short Review of Genera. Hydrobiologia 2010, 639, 231–243. [Google Scholar] [CrossRef]
- Suda, S.; Watanabe, M.M.; Otsuka, S.; Mahakahant, A.; Yongmanitchai, W.; Nopartnaraporn, N.; Liu, Y.; Day, J.G. Taxonomic Revision of Water-Bloom-Forming Species of Oscillatorioid Cyanobacteria. Int. J. Syst. Evol. Microbiol. 2002, 52, 1577–1595. [Google Scholar] [CrossRef]
- Hardy, F.J.; Preece, E.; Backer, L. Status of State CyanoHAB Outreach and Monitoring Efforts, United States. Lake Reserv. Manag. 2021, 37, 246–260. [Google Scholar] [CrossRef]
- U.S. Environmental Protection Agency. National Lakes Assessment: A Collaborative Survey of the Nation’s Lakes; U.S. Environmental Protection Agency: Washington, DC, USA, 2009. Available online: https://www.epa.gov/national-aquatic-resource-surveys/nla (accessed on 15 August 2022).
- Loftin, K.A.; Graham, J.L.; Hilborn, E.D.; Lehmann, S.C.; Meyer, M.T.; Dietze, J.E.; Griffith, C.B. Cyanotoxins in Inland Lakes of the United States: Occurrence and Potential Recreational Health Risks in the EPA National Lakes Assessment 2007. Harmful Algae 2016, 56, 77–90. [Google Scholar] [CrossRef]
- U.S. Environmental Protection Agency. National Lakes Assessment: A Collaborative Survey of the Nation’s Lakes; U.S. Environmental Protection Agency: Washington, DC, USA, 2017. Available online: https://www.epa.gov/national-aquatic-resource-surveys/nla (accessed on 15 August 2022).
- Graham, L.E.; Graham, J.M.; Wilcox, L.W.; Cook, M.E. Algae, 3rd ed.; LJLM Press: Upper Saddle River, NJ, USA, 2016; ISBN 9780986393532. [Google Scholar]
- Nicholls, H.H. Haptophyte Algae. In Freshwater Algae of North America; Wehr, J.D., Sheath, R.G., Kociolek, J.P., Eds.; Academic Press: San Diego, CA, USA, 2015; pp. 587–605. [Google Scholar]
- Reifel, K.M.; McCoy, M.P.; Tiffany, M.A.; Rocke, T.E.; Trees, C.C.; Barlow, S.B.; Faulkner, D.J.; Hurlbert, S.H. Pleurochrysis pseudoroscoffensis (Prymnesiophyceae) Blooms on the Surface of the Salton Sea, California. Hydrobiologia 2001, 466, 177–185. [Google Scholar] [CrossRef]
- Nicholls, K.H.; Beaver, J.L.; Estabrook, R.H. Lakewide Odours in Ontario and New Hampshire Caused by Chrysochromulina breviturrita Nich. (Prymnesiophyceae). Hydrobiologia 1982, 96, 91–95. [Google Scholar] [CrossRef]
- Moestrup, Ø. Economic Aspects: ‘Blooms,’ Nuisance Species and Toxins. In The Haptophyte Algae; Green, J.C., Leadbeater, B.S.C., Eds.; Clarendon Press: Oxford, UK, 1994; pp. 265–285. [Google Scholar]
- Southard, G.M.; Fries, L.T.; Barkoh, A. Prymnesium parvum: The Texas Experience. J. Am. Water Resour. Assoc. 2010, 46, 14–23. [Google Scholar] [CrossRef]
- Free, G.; Van de Bund, W.; Gawlik, B.M.; Van Wijk, L.; Wood, M.; Guagnini, E.; Koutelos, K.; Annunziato, A.; Grizzetti, B.; Vigiak, O.; et al. An EU Analysis of the Ecological Disaster in the Oder River of 2022; Publications Office of the European Union: Luxembourg, 2023; ISBN 9789276993148. [Google Scholar]
- James, T.L.; De La Cruz, A. Prymnesium parvum Carter (Chrysophyceae) as a Suspect of Mass Mortalities of Fish and Shellfish Communities in Western Texas. Texas J. Sci. 1989, 41, 429–430. [Google Scholar]
- Rhodes, K.; Hubbs, C. Recovery of Pecos River Fishes from a Red Tide Fish Kill. Southwest. Nat. 1992, 37, 178–187. [Google Scholar] [CrossRef]
- Lutz-Carrillo, D.J.; Southard, G.M.; Fries, L.T. Global Genetic Relationships among Isolates of Golden Alga (Prymnesium parvum). J. Am. Water Resour. Assoc. 2010, 46, 24–32. [Google Scholar] [CrossRef]
- Triemer, R.E.; Zakryś, B. Photosynthetic Euglenoids. In Freshwater Algae of North America; Wehr, J.D., Sheath, R.G., Kociolek, J.P., Eds.; Academic Press: San Diego, California, USA, 2015; pp. 459–483. [Google Scholar]
- Zimba, P.V.; Rowan, M.; Triemer, R. Identification of Euglenoid Algae That Produce Ichthyotoxin(S). J. Fish Dis. 2004, 27, 115–117. [Google Scholar] [CrossRef]
- Zimba, P.V.; Huang, I.S.; Gutierrez, D.; Shin, W.; Bennett, M.S.; Triemer, R.E. Euglenophycin Is Produced in at Least Six Species of Euglenoid Algae and Six of Seven Strains of Euglena sanguinea. Harmful Algae 2017, 63, 79–84. [Google Scholar] [CrossRef]
- Zimba, P.V.; Moeller, P.D.; Beauchesne, K.; Lane, H.E.; Triemer, R.E. Identification of Euglenophycin—A Toxin Found in Certain Euglenoids. Toxicon 2010, 55, 100–104. [Google Scholar] [CrossRef]
- Janse van Vuuren, S.; Levanets, A. Mass Developments of Euglena sanguinea Ehrenberg in South Africa. Afr. J. Aquat. Sci. 2021, 46, 110–122. [Google Scholar] [CrossRef]
- Bevel, N. Some Notes on the Protozoa of Reelfoot Lake. J. Tennessee Acad. Sci. 1937, 13, 137–159. [Google Scholar]
- Johnson, L.P. Euglenae of Iowa. Trans. Am. Microsc. Soc. 1944, 2, 97–135. [Google Scholar] [CrossRef]
- Zakryś, B.; Walne, P.L. Floristic, Taxonomic and Phytogeographic Studies of Green Euglenophyta from the Southeastern United States, with Emphasis on New and Rare Species. Arch. Fur Hydrobiol. 1994, 72, 71–114. [Google Scholar]
- Gojdics, M. Some Observations of Euglena Sanguinea EHRBG. Trans. Am. Microsc. Soc. 1939, 58, 241–248. [Google Scholar] [CrossRef]
- Cirés, S.; Ballot, A. A Review of the Phylogeny, Ecology and Toxin Production of Bloom-Forming Aphanizomenon Spp. and Related Species within the Nostocales (Cyanobacteria). Harmful Algae 2016, 54, 21–43. [Google Scholar] [CrossRef]
- Li, X.; Dreher, T.W.; Li, R. An Overview of Diversity, Occurrence, Genetics and Toxin Production of Bloom-Forming Dolichospermum (Anabaena) Species. Harmful Algae 2016, 54, 54–68. [Google Scholar] [CrossRef]
- Harke, M.J.; Steffen, M.M.; Gobler, C.J.; Otten, T.G.; Wilhelm, S.W.; Wood, S.A.; Paerl, H.W. A Review of the Global Ecology, Genomics, and Biogeography of the Toxic Cyanobacterium, Microcystis spp. Harmful Algae 2016, 54, 4–20. [Google Scholar] [CrossRef]
- Wood, S.A.; Kelly, L.T.; Bouma-Gregson, K.; Humbert, J.F.; Laughinghouse, H.D.; Lazorchak, J.; McAllister, T.G.; McQueen, A.; Pokrzywinski, K.; Puddick, J.; et al. Toxic Benthic Freshwater Cyanobacterial Proliferations: Challenges and Solutions for Enhancing Knowledge and Improving Monitoring and Mitigation. Freshw. Biol. 2020, 65, 1824–1842. [Google Scholar] [CrossRef]
- Reinl, K.L.; Brookes, J.D.; Carey, C.C.; Harris, T.D.; Ibelings, B.W.; Morales-Williams, A.M.; De Senerpont Domis, L.N.; Atkins, K.S.; Isles, P.D.F.; Mesman, J.P.; et al. Cyanobacterial Blooms in Oligotrophic Lakes: Shifting the High-Nutrient Paradigm. Freshw. Biol. 2021, 66, 1846–1859. [Google Scholar] [CrossRef]
- Komárek, J.; Johansen, J.R. Coccoid Cyanobacteria. In Freshwater Algae of North America; Wehr, J.D., Sheath, R.G., Kociolek, J.P., Eds.; Academic Press: London, UK; Elsevier: Amsterdam, The Netherlands, 2015; pp. 75–133. [Google Scholar]
- Krztoń, W.; Kosiba, J.; Pociecha, A.; Wilk-Woźniak, E. The Effect of Cyanobacterial Blooms on Bio- and Functional Diversity of Zooplankton Communities. Biodivers. Conserv. 2019, 28, 1815–1835. [Google Scholar] [CrossRef]
- Falconer, I.R. Algal Toxins and Human Health. Qual. Treat. Drink. Water II 1998, 5, 53–82. [Google Scholar] [CrossRef]
- Meissner, K.; Dittmann, E.; Börner, T. Toxic and Non-Toxic Strains of the Cyanobacterium Microcystis aeruginosa Contain Sequences Homologous to Peptide Synthetase Genes. FEMS Microbiol. Lett. 1996, 135, 295–303. [Google Scholar] [CrossRef] [PubMed]
- Bertos-Fortis, M.; Farnelid, H.M.; Lindh, M.V.; Casini, M.; Andersson, A.; Pinhassi, J.; Legrand, C. Unscrambling Cyanobacteria Community Dynamics Related to Environmental Factors. Front. Microbiol. 2016, 7, 625. [Google Scholar] [CrossRef]
- Ribeiro, K.F.; Duarte, L.; Crossetti, L.O. Everything Is Not Everywhere: A Tale on the Biogeography of Cyanobacteria. Hydrobiologia 2018, 820, 23–48. [Google Scholar] [CrossRef]
- Kaplan-Levy, R.N.; Hadas, O.; Summers, M.L.; Rucker, J.; Sukenik, A. Akinetes: Dormant Cells of Cyanobacteria. In Dormancy and Resistance in Harsh Environments, Topics in Current Genetics; Lubenz, E., Ed.; Springer: Berlin/Heidelberg, Germany, 2010; Volume 21, ISBN 978-3-642-12421-1. [Google Scholar]
- Moustaka-Gouni, M.; Hiskia, A.; Genitsaris, S.; Katsiapi, M.; Manolidi, K.; Zervou, S.K.; Christophoridis, C.; Triantis, T.M.; Kaloudis, T.; Orfanidis, S. First Report of Aphanizomenon favaloroi Occurrence in Europe Associated with Saxitoxins and a Massive Fish Kill in Lake Vistonis, Greece. Mar. Freshw. Res. 2017, 68, 793–800. [Google Scholar] [CrossRef]
- Cirés, S.; Casero, M.C.; Quesada, A. Toxicity at the Edge of Life: A Review on Cyanobacterial Toxins from Extreme Environments. Mar. Drugs 2017, 15, 233. [Google Scholar] [CrossRef]
- Reynolds, C.S.; Oliver, R.L.; Walsby, A.E. Cyanobacterial Dominance: The Role of Buoyancy Regulation in Dynamic Lake Environments. N. Z. J. Mar. Freshw. Res. 1987, 21, 379–390. [Google Scholar] [CrossRef]
- Reynolds, C.S. What Factors Influence the Species Composition of Phytoplankton in Lakes of Different Trophic Status? Hydrobiologia 1998, 369–370, 11–26. [Google Scholar] [CrossRef]
- Graham, J.L.; Loftin, K.A.; Ziegler, A.C.; Meyer, M.T. Guidelines for Design and Sampling for Cyanobacterial Toxin and Taste-and-Odor Studies in Lakes and Reservoirs; U.S. Geological Survey Scientific Investigations Report 2008-5038; U.S. Geological Survey: Reston, VA, USA, 2008. [Google Scholar]
- Carmichael, W.W.; Boyer, G.L. Health Impacts from Cyanobacteria Harmful Algae Blooms: Implications for the North American Great Lakes. Harmful Algae 2016, 54, 194–212. [Google Scholar] [CrossRef]
- Kumar, K.; Mella-Herrera, R.A.; Golden, J.W. Cyanobacterial Heterocysts. Cold Spring Harb. Perspect. Biol. 2010, 2, a000315. [Google Scholar] [CrossRef]
- Meeks, J.C.; Elhai, J. Regulation of Cellular Differentiation in Filamentous Cyanobacteria in Free-Living and Plant-Associated Symbiotic Growth States. Microbiol. Mol. Biol. Rev. 2002, 66, 94–121. [Google Scholar] [CrossRef]
- Shih, P.M.; Wu, D.; Latifi, A.; Axen, S.D.; Fewer, D.P.; Talla, E.; Calteau, A.; Cai, F.; Tandeau de Marsac, N.; Rippka, R.; et al. Improving the Coverage of the Cyanobacterial Phylum Using Diversity-Driven Genome Sequencing. Proc. Natl. Acad. Sci. USA 2013, 110, 1053–1058. [Google Scholar] [CrossRef] [PubMed]
- Montgomery, B.L. Light-Dependent Governance of Cell Shape Dimensions in Cyanobacteria. Front. Microbiol. 2015, 6, 514. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.P.; Montgomery, B.L. Determining Cell Shape: Adaptive Regulation of Cyanobacterial Cellular Differentiation and Morphology. Trends Microbiol. 2011, 19, 278–285. [Google Scholar] [CrossRef] [PubMed]
- Mitsch, W.J.; Gosselink, J.G. Wetlands, 5th ed.; John Wiley & Sons: Hoboken, NJ, USA, 2015. [Google Scholar]
- Fofonoff, N.P.; Millard, R.C., Jr. Algorithms for Computation of Fundamental Properties of Seawater; UNESCO: Paris, France, 1983. [Google Scholar]
- Hill, K.D.; Dauphinee, T.M.; Woods, D.J. The Extension of the Practical Salinity Scale 1978 to Low Salinities. IEEE J. Ocean. Eng. 1986, 11, 109–112. [Google Scholar] [CrossRef]
- Carmichael, W.W.; Li, R. Cyanobacteria Toxins in the Salton Sea. Saline Syst. 2006, 2, 5. [Google Scholar] [CrossRef]
- Marcarelli, A.M.; Wurtsbaugh, W.A.; Griset, O. Salinity Controls Phytoplankton Response to Nutrient Enrichment in the Great Salt Lake, Utah, USA. Can. J. Fish. Aquat. Sci. 2006, 63, 2236–2248. [Google Scholar] [CrossRef]
- Galat, D.L.; Verdin, J.P.; Sims, L.L. Large-Scale Patterns of Nodularia spumigena Blooms in Pyramid Lake, Nevada, Determined from Landsat Imagery: 1972–1986. Hydrobiologia 1990, 197, 147–164. [Google Scholar] [CrossRef]
- Granéli, E.; Edvardsen, B.; Roelke, D.L.; Hagström, J.A. The Ecophysiology and Bloom Dynamics of Prymnesium spp. Harmful Algae 2012, 14, 260–270. [Google Scholar] [CrossRef]
- Binzer, S.B.; Svenssen, D.K.; Daugbjerg, N.; Alves-de-Souza, C.; Pinto, E.; Hansen, P.J.; Larsen, T.O.; Varga, E. A-, B- and C-Type Prymnesins Are Clade Specific Compounds and Chemotaxonomic Markers in Prymnesium parvum. Harmful Algae 2019, 81, 10–17. [Google Scholar] [CrossRef]
- Wisecaver, J.H.; Auber, R.P.; Pendleton, A.L.; Manning, S.R.; Moore, B.S.; Driscoll, W.W.; Wisecaver, J.H.; Auber, R.P.; Pendleton, A.L.; Watervoort, N.F.; et al. Article Extreme Genome Diversity and Cryptic Speciation in a Harmful Algal-Bloom-Forming Eukaryote. Curr. Biol. 2023, 33, 2246–2259.e8. [Google Scholar] [CrossRef]
- Edvardsen, B.; Paasche, E. Bloom Dynamics and Physiology of Prymnesium and Chrysochromulina. In Physiological Ecology of Harmful Algal Blooms; Anderson, D.M., Cembella, A.D., Hallegraeff, G.M., Eds.; Springer: Berlin, Germany, 1998; pp. 193–208. [Google Scholar]
- Medlin, L.K.; Kooistra, W.H.C.F.; Potter, D.; Saunders, G.W.; Andersen, R.A. Phylogenetic Relationships of the ‘Golden Algae’ (Haptophytes, Heterokont Chromophytes) and Their Plastids. In Origins of Algae and their Plastids. Plant Systematics and Evolution; Bhattacharya, D., Ed.; Springer: Vienna, Austria, 1997; pp. 187–219. [Google Scholar]
- Roelke, D.L.; Errera, R.M.; Riesling, R.; Brooks, B.W.; Grover, J.P.; Schwierzke, L.; Ureña-Boeck, F.; Baker, J.; Pinckney, J.L. Effects of Nutrient Enrichment on Prymnesium parvum Population Dynamics and Toxicity: Results from Field Experiments, Lake Possum Kingdom, USA. Aquat. Microb. Ecol. 2007, 46, 125–140. [Google Scholar] [CrossRef]
- VanLandeghem, M.M.; Denny, S.; Patiño, R. Predicting the Risk of Toxic Blooms of Golden Alga from Cell Abundance and Environmental Covariates. Limnol. Oceanogr. Methods 2015, 13, 568–586. [Google Scholar] [CrossRef]
- Driscoll, W.W.; Espinosa, N.J.; Eldakar, O.T.; Hackett, J.D. Allelopathy as an Emergent, Exploitable Public Good in the Bloom-Forming Microalga Prymnesium parvum. Evolution 2013, 67, 1582–1590. [Google Scholar] [CrossRef]
- Hambright, K.D.; Zamor, R.M.; Easton, J.D.; Glenn, K.L.; Remmel, E.J.; Easton, A.C. Temporal and Spatial Variability of an Invasive Toxigenic Protist in a North American Subtropical Reservoir. Harmful Algae 2010, 9, 568–577. [Google Scholar] [CrossRef]
- Clayton, J.B.; Patiño, R.; Rashel, R.H.; Tábora-Sarmiento, S. Water Quality Associations and Spatiotemporal Distribution of the Harmful Alga Prymnesium parvum in an Impounded Urban Stream System. J. Urban Ecol. 2021, 7, juab011. [Google Scholar] [CrossRef]
- Wagstaff, B.A.; Pratscher, J.; Rivera, P.P.L.; Hems, E.S.; Brooks, E.; Rejzek, M.; Todd, J.D.; Murrell, J.C.; Field, R.A. Assessing the Toxicity and Mitigating the Impact of Harmful Prymnesium Blooms in Eutrophic Waters of the Norfolk Broads. Environ. Sci. Technol. 2021, 55, 16538–16551. [Google Scholar] [CrossRef]
- Green, J.C.; Hibberd, D.J.; Pienaar, R.N. The Taxonomy of Prymnesium (Prymnesiophyceae) Including a Description of a New Cosmopolitan Species, P. Patellifera Sp. Nov., and Further Observations on P. Parvum N. Carter. Br. Phycol. J. 1982, 17, 363–382. [Google Scholar] [CrossRef]
- Li, W.K.W.; Andersen, R.A.; Gifford, D.J.; Incze, L.S.; Martin, J.L.; Pilskaln, C.H.; Rooney-Varga, J.N.; Sieracki, M.E.; Wilson, W.H.; Wolff, N.H. Planktonic Microbes in the Gulf of Maine Area. PLoS ONE 2011, 6, e20981. [Google Scholar] [CrossRef]
- Nelson, J.; Byrd, M. Occurrence of Prymnesium parvum (Golden Alga) in Texas Intertidal Waters; Texas Parks and Wildlife Department Management Data Series 264; 2011. Available online: https://texashistory.unt.edu/ark:/67531/metapth577898/m2/1/high_res_d/txca-0908.pdf (accessed on 1 March 2023).
- Roelke, D.L.; Schwierzke, L.; Brooks, B.W.; Grover, J.P.; Errera, R.M.; Valenti, T.W.; Pinckney, J.L. Factors Influencing Prymnesium parvum Population Dynamics during Bloom Initiation: Results from in-Lake Mesocosm Experiments. J. Am. Water Resour. Assoc. 2010, 46, 76–91. [Google Scholar] [CrossRef]
- Sager, R.; Barkoh, A.A.; Buzan, D.L.; Fries, L.T.; Guss, J.A.; Kurten, G.L.; Ralph, J.J.; Srnghurst, E.J.; Southard, G.M.; Swanson, E. Prymnesium parvum: A Potential Threat to U.S. Reservoirs. Am. Fish. Soc. Symp. 2008, 62, 261–273. [Google Scholar]
- Israël, N.M.D.; VanLandeghem, M.M.; Denny, S.; Ingle, J.; Patiño, R. Golden Alga Presence and Abundance Are Inversely Related to Salinity in a High-Salinity River Ecosystem, Pecos River, USA. Harmful Algae 2014, 39, 81–91. [Google Scholar] [CrossRef]
- Caron, D.A.; Lie, A.A.Y.; Buckowski, T.; Turneer, J.; Frabotta, K. The Effect of pH and Salinity on the Toxicity and Growth of the Golden Alga, Prymnesium parvum. Protist 2023, 174, 125927. [Google Scholar] [CrossRef]
- Hindák, F.; Wołowski, K.; Hindáková, A. Cysts and Their Formation in Some Neustonic Euglena Species. Ann. Limnol. 2000, 36, 83–93. [Google Scholar] [CrossRef]
- Graham, J.L.; Dubrovsky, N.M.; Eberts, S.M. Cyanobacterial Harmful Algal Blooms and U.S. Geological Survey Science Capabilities: U.S. Geological Survey Open-File Report 2016-1174; US Department of the Interior, US Geological Survey: Reston, VA, USA, 2016. [Google Scholar]
- Binoy, T.T.; Ponnachan, V.T.; Mavila, S.S. Outbreak of Harmful Algal Blooms (HABs) in the Sacred River of Achenkoil, Kerala, India during the Post Flood Season. Indian Hydrobiol. 2020, 19, 229–240. [Google Scholar]
- Rahman, M.M.; Jewel, M.A.S.; Khan, S.; Haque, M.M. Study of Euglenophytes Bloom and It’s Impact on Fish Growth in Bangladesh. Algae 2007, 22, 185–192. [Google Scholar] [CrossRef]
- Xavier, M.B.; Mainardes Pinto, C.S.R.; Takino, M. Euglena sanguinea Ehrenberg Bloom in a Fish-Breeding Tank (Pindamonhangaba, São Paulo, Brazil). Arch. Hydrobiol. Suppl. Algol. Stud. 1991, 26, 133–142. [Google Scholar]
- Zakryś, B.; Karnkowska-Ishikawa, A.; ŁUkomska-Kowalczyk, M.; Milanowski, R. A New Photosynthetic Euglenoid Isolated in Poland: Euglenaria clepsydroides Sp. Nov. (Euglenea). Eur. J. Phycol. 2013, 48, 260–267. [Google Scholar] [CrossRef]
- Paerl, H.W.; Fulton, R.S.; Moisander, P.H.; Dyble, J. Harmful Freshwater Algal Blooms, with an Emphasis on Cyanobacteria. Sci. World J. 2001, 1, 76–113. [Google Scholar] [CrossRef]
- Paerl, H.W.; Otten, T.G. Duelling “CyanoHABs”: Unravelling the Environmental Drivers Controlling Dominance and Succession among Diazotrophic and Non-N2-Fixing Harmful Cyanobacteria. Environ. Microbiol. 2016, 18, 316–324. [Google Scholar] [CrossRef]
- Graham, J.L.; Dubrovsky, N.M.; Foster, G.M.; King, L.R.; Loftin, K.A.; Rosen, B.H.; Stelzer, E.A. Cyanotoxin Occurrence in Large Rivers of the United States. INL Waters 2020, 10, 109–117. [Google Scholar] [CrossRef]
- Nietch, C.T.; Gains-germain, L.; Lazorchak, J.; Keely, S.P.; Youngstrom, G.; Urichich, E.M.; Astifan, B.; Dasilva, A.; Mayfield, H. Development of a Risk Characterization Tool for Harmful Cyanobacteria Blooms on the Ohio River. Water 2022, 14, 644. [Google Scholar] [CrossRef] [PubMed]
- U.S. Environmental Protection Agency. National Lakes Assessment 2017 Field Operations Manual EPA 841-B-16-002; U.S. Environmental Protection Agency: Washington, DC, USA, 2017. [Google Scholar]
- Bargu, S.; Justic, D.; White, J.R.; Lane, R.; Day, J.; Paerl, H.; Raynie, R. Mississippi River Diversions and Phytoplankton Dynamics in Deltaic Gulf of Mexico Estuaries: A Review. Estuar. Coast. Shelf Sci. 2019, 221, 39–52. [Google Scholar] [CrossRef]
- Lehman, P.W.; Boyer, G.; Hall, C.; Waller, S.; Gehrts, K. Distribution and Toxicity of a New Colonial Microcystis aeruginosa Bloom in the San Francisco Bay Estuary, California. Hydrobiologia 2005, 541, 87–99. [Google Scholar] [CrossRef]
- Metcalf, J.S.; Banack, S.A.; Powell, J.T.; Tymm, F.J.M.; Murch, S.J.; Brand, L.E.; Cox, P.A. Public Health Responses to Toxic Cyanobacterial Blooms: Perspectives from the 2016 Florida Event. Water Policy 2018, 20, 919–932. [Google Scholar] [CrossRef]
- Metcalf, J.S.; Banack, S.A.; Wessel, R.A.; Lester, M.; Pim, J.G.; Cassani, J.R.; Cox, P.A. Toxin Analysis of Freshwater Cyanobacterial and Marine Harmful Algal Blooms on the West Coast of Florida and Implications for Estuarine Environments. Neurotox. Res. 2021, 39, 27–35. [Google Scholar] [CrossRef]
- Sellner, K.G.; Lacouture, R.V.; Parrish, C.R. Effects of Increasing Salinity on a Cyanobacteria Bloom in the Potomac River Estuary. J. Plankton Res. 1988, 10, 49–61. [Google Scholar] [CrossRef]
- Ross, C.; Santiago-Vázquez, L.; Paul, V. Toxin Release in Response to Oxidative Stress and Programmed Cell Death in the Cyanobacterium Microcystis aeruginosa. Aquat. Toxicol. 2006, 78, 66–73. [Google Scholar] [CrossRef]
- Lehman, P.W.; Kurobe, T.; Teh, S.J. Impact of Extreme Wet and Dry Years on the Persistence of Microcystis Harmful Algal Blooms in San Francisco Estuary. Quat. Int. 2022, 621, 16–25. [Google Scholar] [CrossRef]
- Joshi, S.M.; Jackson, L.J. How Might Changing Climate Limit Cyanobacteria Growth in Shallow Prairie Lakes? An Empirical Space-for-Time Evaluation of the Potential Role of Increasing Sulfate. Adv. Environ. Eng. Res. 2022, 3. [Google Scholar] [CrossRef]
- Marino, R.; Howarth, R.W.; Shames, J.; Prepas, E. Molybdenum and Sulfate as Controls on the Abundance of Nitrogen-Fixing Cyanobacteria in Saline Lakes in Alberta. Limnol. Oceanogr. 1990, 35, 245–259. [Google Scholar] [CrossRef]
- Tanabe, Y.; Hodoki, Y.; Sano, T.; Tada, K.; Watanabe, M.M. Adaptation of the Freshwater Bloom-Forming Cyanobacterium Microcystis aeruginosa to Brackish Water Is Driven by Recent Horizontal Transfer of Sucrose Genes. Front. Microbiol. 2018, 9, 1150. [Google Scholar] [CrossRef] [PubMed]
- Albay, M.; Matthiensen, A.; Codd, G.A. Occurrence of Toxic Blue-Green Algae in the Kucukcekmece Lagoon (Istanbul, Turkey). Environ. Toxicol. 2005, 20, 277–284. [Google Scholar] [CrossRef]
- Bormans, M.; Savar, V.; Legrand, B.; Mineaud, E.; Robert, E.; Lance, E.; Amzil, Z. Cyanobacteria and Cyanotoxins in Estuarine Water and Sediment. Aquat. Ecol. 2020, 54, 625–640. [Google Scholar] [CrossRef]
- Trung, B.; Dao, T.S.; Faassen, E.; Lürling, M. Cyanobacterial Blooms and Microcystins in Southern Vietnam. Toxins 2018, 10, 471. [Google Scholar] [CrossRef] [PubMed]
- Olofsson, M.; Suikkanen, S.; Kobos, J.; Wasmund, N.; Karlson, B. Basin-Specific Changes in Filamentous Cyanobacteria Community Composition across Four Decades in the Baltic Sea. Harmful Algae 2020, 91, 101685. [Google Scholar] [CrossRef] [PubMed]
- Teikari, J.E.; Popin, R.V.; Hou, S.; Wahlsten, M.; Hess, W.R.; Sivonen, K. Insight into the Genome and Brackish Water Adaptation Strategies of Toxic and Bloom-Forming Baltic Sea Dolichospermum sp. UHCC 0315. Sci. Rep. 2019, 9, 4888. [Google Scholar] [CrossRef]
- Chen, H.; Lürling, M. Calcium Promotes Formation of Large Colonies of the Cyanobacterium Microcystis by Enhancing Cell-Adhesion. Harmful Algae 2020, 92, 101768. [Google Scholar] [CrossRef]
- Gu, P.; Li, Q.; Zhang, W.; Zheng, Z.; Luo, X. Effects of Different Metal Ions (Ca, Cu, Pb, Cd) on Formation of Cyanobacterial Blooms. Ecotoxicol. Environ. Saf. 2020, 189, 109976. [Google Scholar] [CrossRef]
- Shi, J.Q.; Wu, Z.X.; Song, L.R. Physiological and Molecular Responses to Calcium Supplementation in Microcystis aeruginosa (Cyanobacteria). N. Z. J. Mar. Freshw. Res. 2013, 47, 51–61. [Google Scholar] [CrossRef]
- Wei, K.; Jung, S.; Amano, Y.; Machida, M. Control of the Buoyancy of Microcystis aeruginosa via Colony Formation Induced by Regulating Extracellular Polysaccharides and Cationic Ions. SN Appl. Sci. 2019, 1, 1573. [Google Scholar] [CrossRef]
- Zhao, L.; Lu, L.; Li, M.; Xu, Z.; Zhu, W. Effects of Ca and Mg Levels on Colony Formation and EPS Content of Cultured M. aeruginosa. Procedia Environ. Sci. 2011, 10, 1452–1458. [Google Scholar] [CrossRef]
- Parker, D.L.; Kumar, H.D.; Rai, L.C.; Singh, J.B. Potassium Salts Inhibit Growth of the Cyanobacteria Microcystis Spp. in Pond Water and Defined Media: Implications for Control of Microcystin-Producing Aquatic Blooms. Appl. Environ. Microbiol. 1997, 63, 2324–2329. [Google Scholar] [CrossRef] [PubMed]
- Xiao, M.; Li, M.; Reynolds, C.S. Colony Formation in the Cyanobacterium Microcystis. Biol. Rev. 2018, 93, 1399–1420. [Google Scholar] [CrossRef] [PubMed]
- Howarth, R.W.; Marino, R. Nitrogen as the Limiting Nutrient for Eutrophication in Coastal Marine Ecosystems: Evolving Views over Three Decades. Limnol. Oceanogr. 2006, 51, 364–376. [Google Scholar] [CrossRef]
- Cole, J.J.; Lane, J.M.; Marino, R.; Howarth, R.W. Molybdenum Assimilation by Cyanobacteria and Phytoplankton in Freshwater and Salt Water. Limnol. Oceanogr. 1993, 38, 25–35. [Google Scholar] [CrossRef]
- Marino, R.; Howarth, R.W.; Chan, F.; Cole, J.J.; Likens, G.E. Sulfate Inhibition of Molybdenum-Dependent Nitrogen Fixation by Planktonic Cyanobacteria under Seawater Conditions: A Non-Reversible Effect. Hydrobiologia 2003, 500, 277–293. [Google Scholar] [CrossRef]
- Long, B.M. Evidence That Sulfur Metabolism Plays a Role in Microcystin Production by Microcystis aeruginosa. Harmful Algae 2010, 9, 74–81. [Google Scholar] [CrossRef]
- Chen, L.; Gin, K.Y.H.; He, Y. Effects of Sulfate on Microcystin Production, Photosynthesis, and Oxidative Stress in Microcystis aeruginosa. Environ. Sci. Pollut. Res. 2016, 23, 3586–3595. [Google Scholar] [CrossRef]
- VanLandeghem, M.M.; Farooqi, M.; Southard, G.M.; Patiño, R. Spatiotemporal Associations of Reservoir Nutrient Characteristics and the Invasive, Harmful Alga Prymnesium parvum in West Texas. J. Am. Water Resour. Assoc. 2015, 51, 487–501. [Google Scholar] [CrossRef]
- Hambright, K.D.; Beyer, J.E.; Easton, J.D.; Zamor, R.M.; Easton, A.C.; Hallidayschult, T.C. The Niche of an Invasive Marine Microbe in a Subtropical Freshwater Impoundment. ISME J. 2015, 9, 256–264. [Google Scholar] [CrossRef]
- Patiño, R.; Dawson, D.; VanLandeghem, M.M. Retrospective Analysis of Associations between Water Quality and Toxic Blooms of Golden Alga (Prymnesium parvum) in Texas Reservoirs: Implications for Understanding Dispersal Mechanisms and Impacts of Climate Change. Harmful Algae 2014, 33, 1–11. [Google Scholar] [CrossRef]
- Tábora-Sarmiento, S.; Patiño, R.; Portillo-Quintero, C.; Coldren, C. Air, Land, and Water Variables Associated with the First Appearance and Current Spatial Distribution of Toxic Prymnesium parvum Blooms in Reservoirs of the Southern Great Plains, USA. Sci. Total Environ. 2022, 836, 155567. [Google Scholar] [CrossRef] [PubMed]
- Hartman, K.J.; Wellman, D.I.; Kingsbury, J.W.; Cincotta, D.A.; Clayton, J.L.; Eliason, K.M.; Jernejcic, F.A.; Owens, N.V.; Smith, D.M. A Case Study of a Prymnesium parvum Harmful Algae Bloom in the Ohio River Drainage: Impact, Recovery and Potential for Future Invasions/Range Expansion. Water 2021, 13, 3233. [Google Scholar] [CrossRef]
- Johnsen, T.M.; Eikrem, W.; Olseng, C.D.; Tollefsen, K.E.; Bjerknes, V. Prymnesium parvum: The Norwegian Experience. J. Am. Water Resour. Assoc. 2010, 46, 6–13. [Google Scholar] [CrossRef]
- Guo, M.; Harrison, P.J.; Taylor, F.J.R. Fish Kills Related to Primnesium parvum N. Carter (Haptophyta) in the People’s Republic of China. J. Appl. Phycol. 1996, 8, 111–117. [Google Scholar] [CrossRef]
- Lemley, D.A.; Adams, J.B.; Rishworth, G.M.; Bouland, C. Phytoplankton Responses to Adaptive Management Interventions in Eutrophic Urban Estuaries. Sci. Total Environ. 2019, 693, 133601. [Google Scholar] [CrossRef]
- Rashel, R.H.; Patiño, R. Growth Response of the Ichthyotoxic Haptophyte, Prymnesium parvum Carter, to Changes in Sulfate and Fluoride Concentrations. PLoS ONE 2019, 14, e0223266. [Google Scholar] [CrossRef]
- VanLandeghem, M.M.; Farooqi, M.; Southard, G.M.; Patiño, R. Associations between Water Physicochemistry and Prymnesium parvum Presence, Abundance, and Toxicity in West Texas Reservoirs. J. Am. Water Resour. Assoc. 2015, 51, 471–486. [Google Scholar] [CrossRef]
- Ohio Environmental Protection Agency. Seeing Red: Emerging Harmful Algal Blooms; Spigot News; Ohio Environmental Protection Agency: Columbus, OH, USA, 2013; Volume 13. [Google Scholar]
- Paerl, H.W.; Otten, T.G.; Kudela, R. Mitigating the Expansion of Harmful Algal Blooms across the Freshwater-to-Marine Continuum. Environ. Sci. Technol. 2018, 52, 5519–5529. [Google Scholar] [CrossRef]
- Schindler, D.W.; Carpenter, S.R.; Chapra, S.C.; Hecky, R.E.; Orihel, D.M. Reducing Phosphorus to Curb Lake Eutrophication Is a Success. Environ. Sci. Technol. 2016, 50, 8923–8929. [Google Scholar] [CrossRef]
- Taranu, Z.E.; Gregory-Eaves, I.; Leavitt, P.R.; Bunting, L.; Buchaca, T.; Catalan, J.; Domaizon, I.; Guilizzoni, P.; Lami, A.; Mcgowan, S.; et al. Acceleration of Cyanobacterial Dominance in North Temperate-Subarctic Lakes during the Anthropocene. Ecol. Lett. 2015, 18, 375–384. [Google Scholar] [CrossRef] [PubMed]
- Heathcote, A.J.; Taranu, Z.E.; Tromas, N.; MacIntyre-Newell, M.; Leavitt, P.R.; Pick, F.R. Sedimentary DNA and Pigments Show Increasing Abundance and Toxicity of CyanoHABs during the Anthropocene. Freshw. Biol. 2023, 1–16. [Google Scholar] [CrossRef]
- Glibert, P.M. Harmful Algae at the Complex Nexus of Eutrophication and Climate Change. Harmful Algae 2020, 91, 101583. [Google Scholar] [CrossRef] [PubMed]
- Wurtsbaugh, W.A.; Paerl, H.W.; Dodds, W.K. Nutrients, Eutrophication and Harmful Algal Blooms along the Freshwater to Marine Continuum. Wiley Interdiscip. Rev. Water 2019, 6, e1373. [Google Scholar] [CrossRef]
- Doubek, J.P.; Carey, C.C.; Cardinale, B.J. Anthropogenic Land Use Is Associated with N-Fixing Cyanobacterial Dominance in Lakes across the Continental United States. Aquat. Sci. 2015, 77, 681–694. [Google Scholar] [CrossRef]
- Marion, J.W.; Zhang, F.; Cutting, D.; Lee, J. Associations between County-Level Land Cover Classes and Cyanobacteria Blooms in the United States. Ecol. Eng. 2017, 108, 556–563. [Google Scholar] [CrossRef]
- Beaver, J.R.; Manis, E.E.; Loftin, K.A.; Graham, J.L.; Pollard, A.I.; Mitchell, R.M. Land Use Patterns, Ecoregion, and Microcystin Relationships in U.S. Lakes and Reservoirs: A Preliminary Evaluation. Harmful Algae 2014, 36, 57–62. [Google Scholar] [CrossRef]
- Iiames, J.S.; Salls, W.B.; Mehaffey, M.H.; Nash, M.S.; Christensen, J.R.; Schaeffer, B.A. Modeling Anthropogenic and Environmental Influences on Freshwater Harmful Algal Bloom Development Detected by MERIS over the Central United States. Water Resour. Res. 2021, 57, e2020WR028946. [Google Scholar] [CrossRef]
- Rose, V.J.; Forney, W.M.; Norton, R.A.; Harrison, J.A. Catchment Characteristics, Water Quality, and Cyanobacterial Blooms in Washington and Oregon Lakes. Lake Reserv. Manag. 2019, 35, 51–63. [Google Scholar] [CrossRef]
- Ho, J.C.; Michalak, A.M.; Pahlevan, N. Widespread Global Increase in Intense Lake Phytoplankton Blooms since the 1980s. Nature 2019, 574, 667–670. [Google Scholar] [CrossRef]
- Brêda-Alves, F.; de Oliveira Fernandes, V.; Chia, M.A. Understanding the Environmental Roles of Herbicides on Cyanobacteria, Cyanotoxins, and CyanoHABs. Aquat. Ecol. 2021, 55, 347–361. [Google Scholar] [CrossRef]
- Harris, T.D.; Smith, V.H. Do Persistent Organic Pollutants Stimulate Cyanobacterial Blooms? INL Waters 2016, 6, 124–130. [Google Scholar] [CrossRef]
- Benbrook, C.M. Trends in Glyphosate Herbicide Use in the United States and Globally. Environ. Sci. Eur. 2016, 28, 3. [Google Scholar] [CrossRef] [PubMed]
- Hébert, M.P.; Fugère, V.; Gonzalez, A. The Overlooked Impact of Rising Glyphosate Use on Phosphorus Loading in Agricultural Watersheds. Front. Ecol. Environ. 2019, 17, 48–56. [Google Scholar] [CrossRef]
- Forlani, G.; Pavan, M.; Gramek, M.; Kafarski, P.; Lipok, J. Biochemical Bases for a Widespread Tolerance of Cyanobacteria to the Phosphonate Herbicide Glyphosate. Plant Cell Physiol. 2008, 49, 443–456. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.; Zhang, Z.; Chen, Y.; Zhang, Q.; Ke, M.; Lu, T.; Qian, H. The Mechanism of Different Cyanobacterial Responses to Glyphosate. J. Environ. Sci. 2023, 125, 258–265. [Google Scholar] [CrossRef]
- Xu, S.; Liu, Y.; Zhang, J.; Gao, B. Proteomic Mechanisms for the Combined Stimulatory Effects of Glyphosate and Antibiotic Contaminants on Microcystis aeruginosa. Chemosphere 2021, 267, 129244. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhou, H.; Li, Z.; Zhu, J.; Zhou, C.; Zhao, M. Effects of Glyphosate at Environmentally Relevant Concentrations on the Growth of and Microcystin Production by Microcystis aeruginosa. Environ. Monit. Assess. 2016, 188, 632. [Google Scholar] [CrossRef]
- Dabney, B.L.; Patiño, R. Low-Dose Stimulation of Growth of the Harmful Alga, Prymnesium parvum, by Glyphosate and Glyphosate-Based Herbicides. Harmful Algae 2018, 80, 130–139. [Google Scholar] [CrossRef]
- Sydney, E.B.; Schafranski, K.; Barretti, B.R.V.; Sydney, A.C.N.; Zimmerman, J.F.D.A.; Cerri, M.L.; Mottin Demiate, I. Biomolecules from Extremophile Microalgae: From Genetics to Bioprocessing of a New Candidate for Large-Scale Production. Process Biochem. 2019, 87, 37–44. [Google Scholar] [CrossRef]
- Qu, M.; Wang, L.; Xu, Q.; An, J.; Mei, Y.; Liu, G. Influence of Glyphosate and Its Metabolite Aminomethylphosphonic Acid on Aquatic Plants in Different Ecological Niches. Ecotoxicol. Environ. Saf. 2022, 246, 114155. [Google Scholar] [CrossRef] [PubMed]
- Battaglin, W.A.; Meyer, M.T.; Kuivila, K.M.; Dietze, J.E. Glyphosate and Its Degradation Product AMPA Occur Frequently and Widely in U.S. Soils, Surface Water, Groundwater, and Precipitation. J. Am. Water Resour. Assoc. 2014, 50, 275–290. [Google Scholar] [CrossRef]
- Coupe, R.H.; Kalkhoff, S.J.; Capel, P.D.; Gregoire, C. Fate and Transport of Glyphosate and Aminomethylphosphonic Acid in Surface Waters of Agricultural Basins. Pest Manag. Sci. 2011, 68, 16–30. [Google Scholar] [CrossRef] [PubMed]
- Mahler, B.J.; Van Metre, P.C.; Burley, T.E.; Loftin, K.A.; Meyer, M.T.; Nowell, L.H. Similarities and Differences in Occurrence and Temporal Fluctuations in Glyphosate and Atrazine in Small Midwestern Streams (USA) during the 2013 Growing Season. Sci. Total Environ. 2017, 579, 149–158. [Google Scholar] [CrossRef]
- Medalie, L.; Baker, N.T.; Shoda, M.E.; Stone, W.W.; Meyer, M.T.; Stets, E.G.; Wilson, M. Influence of Land Use and Region on Glyphosate and Aminomethylphosphonic Acid in Streams in the USA. Sci. Total Environ. 2020, 707, 136008. [Google Scholar] [CrossRef]
- Beecraft, L.; Rooney, R. Bioconcentration of Glyphosate in Wetland Biofilms. Sci. Total Environ. 2021, 756, 143993. [Google Scholar] [CrossRef]
- Vymazal, J.; Březinová, T. The Use of Constructed Wetlands for Removal of Pesticides from Agricultural Runoff and Drainage: A Review. Environ. Int. 2015, 75, 11–20. [Google Scholar] [CrossRef]
- Davidson, N.C. How Much Wetland Has the World Lost? Long-Term and Recent Trends in Global Wetland Area. Mar. Freshw. Res. 2014, 65, 934–941. [Google Scholar] [CrossRef]
- Bullerjahn, G.S.; McKay, R.M.; Davis, T.W.; Baker, D.B.; Boyer, G.L.; D’Anglada, L.V.; Doucette, G.J.; Ho, J.C.; Irwin, E.G.; Kling, C.L.; et al. Global Solutions to Regional Problems: Collecting Global Expertise to Address the Problem of Harmful Cyanobacterial Blooms. A Lake Erie Case Study. Harmful Algae 2016, 54, 223–238. [Google Scholar] [CrossRef]
- Mitsch, W.J. Solving Lake Erie’s Harmful Algal Blooms by Restoring the Great Black Swamp in Ohio. Ecol. Eng. 2017, 108, 406–413. [Google Scholar] [CrossRef]
- Mitsch, W.J. Restoring the Florida Everglades: Comments on the Current Reservoir Plan for Solving Harmful Algal Blooms and Restoring the Florida Everglades. Ecol. Eng. 2019, 3, 100009. [Google Scholar] [CrossRef]
- Tran, C.P.; Bode, R.W.; Smith, A.J.; Kleppel, G.S. Land-Use Proximity as a Basis for Assessing Stream Water Quality in New York State (USA). Ecol. Indic. 2010, 10, 727–733. [Google Scholar] [CrossRef]
- Sommer, U.; Adrian, R.; De Senerpont Domis, L.; Elser, J.J.; Gaedke, U.; Ibelings, B.; Jeppesen, E.; Lürling, M.; Molinero, J.C.; Mooij, W.M.; et al. Beyond the Plankton Ecology Group (PEG) Model: Mechanisms Driving Plankton Succession. Annu. Rev. Ecol. Evol. Syst. 2012, 43, 429–448. [Google Scholar] [CrossRef]
- Shan, K.; Song, L.; Chen, W.; Li, L.; Liu, L.; Wu, Y.; Jia, Y.; Zhou, Q.; Peng, L. Analysis of Environmental Drivers Influencing Interspecific Variations and Associations among Bloom-Forming Cyanobacteria in Large, Shallow Eutrophic Lakes. Harmful Algae 2019, 84, 84–94. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Li, L.; Zheng, L.; Dai, G.; Ma, H.; Shan, K.; Wu, H.; Zhou, Q.; Song, L. Patterns of Succession between Bloom-Forming Cyanobacteria Aphanizomenon flos-aquae and Microcystis and Related Environmental Factors in Large, Shallow Dianchi Lake, China. Hydrobiologia 2016, 765, 1–13. [Google Scholar] [CrossRef]
- Becker, V.; Caputo, L.; Ordóñez, J.; Marcé, R.; Armengol, J.; Crossetti, L.O.; Huszar, V.L.M. Driving Factors of the Phytoplankton Functional Groups in a Deep Mediterranean Reservoir. Water Res. 2010, 44, 3345–3354. [Google Scholar] [CrossRef]
- Christensen, V.G.; Maki, R.P.; Stelzer, E.A.; Norland, J.E.; Khan, E. Phytoplankton Community and Algal Toxicity at a Recurring Bloom in Sullivan Bay, Kabetogama Lake, Minnesota, USA. Sci. Rep. 2019, 9, 16129. [Google Scholar] [CrossRef]
- Huisman, J.; Codd, G.A.; Paerl, H.W.; Ibelings, B.W.; Verspagen, J.M.H.; Visser, P.M. Cyanobacterial Blooms. Nat. Rev. Microbiol. 2018, 16, 471–483. [Google Scholar] [CrossRef]
- Yamamoto, Y.; Nakahara, H. The Formation and Degradation of Cyanobacterium Aphanizomenon flos-aquae Blooms: The Importance of pH, Water Temperature, and Day Length. Limnology 2005, 6, 1–6. [Google Scholar] [CrossRef]
- Zepernick, B.N.; Gann, E.R.; Martin, R.M.; Pound, H.L.; Krausfeldt, L.E.; Chaffin, J.D.; Wilhelm, S.W. Elevated pH Conditions Associated with Microcystis Spp. Blooms Decrease Viability of the Cultured Diatom Fragilaria crotonensis and Natural Diatoms in Lake Erie. Front. Microbiol. 2021, 12, 598736. [Google Scholar] [CrossRef]
- Graham, J.L.; Jones, J.R.; Jones, S.B.; Clevenger, T.E. Spatial and Temporal Dynamics of Microcystin in a Missouri Reservoir. Lake Reserv. Manag. 2006, 22, 59–68. [Google Scholar] [CrossRef]
- Jones, J.R.; Argerich, A.; Obrecht, D.V.; Thorpe, A.P.; North, R.L. Missouri Lakes and Reservoirs Long-Term Limnological Dataset Ver 1. Environmental Data Initiative. Available online: https://portal.edirepository.org/nis/mapbrowse?packageid=edi.520.1 (accessed on 10 July 2023).
- Krztoń, W.; Kosiba, J. Variations in Zooplankton Functional Groups Density in Freshwater Ecosystems Exposed to Cyanobacterial Blooms. Sci. Total Environ. 2020, 730, 139044. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Duan, H.; Shi, X.; Yu, Y.; Kong, F. Contributions of Meteorology to the Phenology of Cyanobacterial Blooms: Implications for Future Climate Change. Water Res. 2012, 46, 442–452. [Google Scholar] [CrossRef]
- Reinl, K.L.; Harris, T.D.; North, R.L.; Almela, P.; Berger, S.A.; Bizic, M.; Burnet, S.H.; Grossart, H.P.; Ibelings, B.W.; Jakobsson, E.; et al. Blooms Also like It Cold. Limnol. Oceanogr. Lett. 2023, 8, 546–564. [Google Scholar] [CrossRef]
- Brutemark, A.; Granéli, E. Role of Mixotrophy and Light for Growth and Survival of the Toxic Haptophyte Prymnesium parvum. Harmful Algae 2011, 10, 388–394. [Google Scholar] [CrossRef]
- Baker, J.W.; Grover, J.P.; Brooks, B.W.; Ureña-Boeck, F.; Roelke, D.L.; Errera, R.; Kiesling, R.L. Growth and Toxicity of Prymnesium parvum (Haptophyta) as a Function of Salinity, Light, and Temperature. J. Phycol. 2007, 43, 219–227. [Google Scholar] [CrossRef]
- VanLandeghem, M.M.; Meyer, M.D.; Cox, S.B.; Sharma, B.; Patiño, R. Spatial and Temporal Patterns of Surface Water Quality and Ichthyotoxicity in Urban and Rural River Basins in Texas. Water Res. 2012, 46, 6638–6651. [Google Scholar] [CrossRef]
- Roelke, D.L.; Brooks, B.W.; Grover, J.P.; Gable, G.M.; Schwierzke-Wade, L.; Hewitt, N.C. Anticipated Human Population and Climate Change Effects on Algal Blooms of a Toxic Haptophyte in the South-Central USA. Can. J. Fish. Aquat. Sci. 2012, 69, 1389–1404. [Google Scholar] [CrossRef]
- VanLandeghem, M.M.; Farooqi, M.; Farquhar, B.; Patiño, R. Impacts of Golden Alga Prymnesium parvum on Fish Populations in Reservoirs of the Upper Colorado River and Brazos River Basins, Texas. Trans. Am. Fish. Soc. 2013, 142, 581–595. [Google Scholar] [CrossRef]
- Dawson, D.; VanLandeghem, M.M.; Asquith, W.H.; Patiño, R. Long-Term Trends in Reservoir Water Quality and Quantity in Two Major River Basins of the Southern Great Plains. Lake Reserv. Manag. 2015, 31, 254–279. [Google Scholar]
- Rashel, R.H.; Patiño, R. Influence of Genetic Background, Salinity, and Inoculum Size on Growth of the Ichthyotoxic Golden Alga (Prymnesium parvum). Harmful Algae 2017, 66, 97–104. [Google Scholar] [CrossRef]
- Rashel, R.H. Influence of Water Quality and Climate Variables on Growth of the Harmful Alga, Prymnesium parvum. Ph.D. Thesis, Texas Tech University, Lubbock, TX, USA, 2020. Available online: https://hdl.handle.net/2346/85858 (accessed on 1 March 2023).
- Danilov, R.A.; Ekelund, N.G.A. Effects of pH on the Growth Rate, Motility and Photosynthesis in Euglena gracilis. Folia Microbiol. 2001, 46, 549–554. [Google Scholar] [CrossRef]
- Zheng, X.; Wang, Y.; Yang, T.; He, Z.; Yan, Q. Size-Fractioned Aggregates within Phycosphere Define Functional Bacterial Communities Related to Microcystis aeruginosa and Euglena sanguinea Blooms. Aquat. Ecol. 2020, 54, 609–623. [Google Scholar] [CrossRef]
- Rahman, M.S.; Shahjahan, M.; Haque, M.M.; Khan, S. Control of Euglenophyte Bloom and Fish Production Enhancement Using Duckweed and Lime. Iran. J. Fish. Sci. 2012, 11, 602–617. [Google Scholar]
- Chernoff, N.; Hill, D.J.; Diggs, D.L.; Faison, B.D.; Francis, B.M.; Lang, J.R.; Larue, M.M.; Le, T.T.; Loftin, K.A.; Lugo, J.N.; et al. A Critical Review of the Postulated Role of the Non-Essential Amino Acid, β-N-Methylamino-L-Alanine, in Neurodegenerative Disease in Humans. J. Toxicol. Environ. Health Part B Crit. Rev. 2017, 20, 183–229. [Google Scholar] [CrossRef] [PubMed]
- Rutkowska, M.; Płotka-Wasylka, J.; Majchrzak, T.; Wojnowski, W.; Mazur-Marzec, H.; Namieśnik, J. Recent Trends in Determination of Neurotoxins in Aquatic Environmental Samples. TrAC Trends Anal. Chem. 2019, 112, 112–122. [Google Scholar] [CrossRef]
- Graham, J.L.; Loftin, K.A.; Meyer, M.T.; Ziegler, A.C. Cyanotoxin Mixtures and Taste-and-Odor Compounds in Cyanobacterial Blooms from the Midwestern United States. Environ. Sci. Technol. 2010, 44, 7361–7368. [Google Scholar] [CrossRef] [PubMed]
- Beaver, J.R.; Tausz, C.E.; Scotese, K.C.; Pollard, A.I.; Mitchell, R.M. Environmental Factors Influencing the Quantitative Distribution of Microcystin and Common Potentially Toxigenic Cyanobacteria in U.S. Lakes and Reservoirs. Harmful Algae 2018, 78, 118–128. [Google Scholar] [CrossRef]
- Kinnear, S. Cylindrospermopsin: A Decade of Progress on Bioaccumulation Research. Mar. Drugs 2010, 8, 542–564. [Google Scholar] [CrossRef]
- Miller, T.R.; Beversdorf, L.J.; Weirich, C.A.; Bartlett, S.L. Cyanobacterial Toxins of the Laurentian Great Lakes, Their Toxicological Effects, and Numerical Limits in Drinking Water. Mar. Drugs 2017, 15, 160. [Google Scholar] [CrossRef]
- Fiore, M.F.; de Lima, S.T.; Carmichael, W.W.; McKinnie, S.M.K.; Chekan, J.R.; Moore, B.S. Guanitoxin, Re-Naming a Cyanobacterial Organophosphate Toxin. Harmful Algae 2020, 92, 101737. [Google Scholar] [CrossRef]
- Christensen, V.G.; Khan, E. Freshwater Neurotoxins and Concerns for Human, Animal, and Ecosystem Health: A Review of Anatoxin-a and Saxitoxin. Sci. Total Environ. 2020, 736, 139515. [Google Scholar] [CrossRef] [PubMed]
- D’Anglada, L.V.; Hilborn, E.D.; Backer, L.C. Harmful Algal Blooms (HABs) and Public Health: Progress and Current Challenges. In Toxins; MDPI: Basel, Switzerland, 2016; ISBN 9783038421559. [Google Scholar]
- Kotak, B.G.; Zurawell, R.W. Cyanobacterial Toxins in Canadian Freshwaters: A Review. Lake Reserv. Manag. 2007, 23, 109–122. [Google Scholar] [CrossRef]
- Al-Sammak, M.A.; Hoagland, K.D.; Cassada, D.; Snow, D.D. Co-Occurrence of the Cyanotoxins BMAA, DABA and Anatoxin-a in Nebraska Reservoirs, Fish, and Aquatic Plants. Toxins 2014, 6, 488–508. [Google Scholar] [CrossRef]
- John, N.; Baker, L.; Ansell, B.R.E.; Newham, S.; Crosbie, N.D.; Jex, A.R. First Report of Anatoxin-a Producing Cyanobacteria in Australia Illustrates Need to Regularly up-Date Monitoring Strategies in a Shifting Global Distribution. Sci. Rep. 2019, 9, 10894. [Google Scholar] [CrossRef] [PubMed]
- McAllister, T.G.; Wood, S.A.; Hawes, I. The Rise of Toxic Benthic Phormidium Proliferations: A Review of Their Taxonomy, Distribution, Toxin Content and Factors Regulating Prevalence and Increased Severity. Harmful Algae 2016, 55, 282–294. [Google Scholar] [CrossRef]
- Gobler, C.J.; Doherty, O.M.; Hattenrath-Lehmann, T.K.; Griffith, A.W.; Kang, Y.; Litaker, R.W. Ocean Warming since 1982 Has Expanded the Niche of Toxic Algal Blooms in the North Atlantic and North Pacific Oceans. Proc. Natl. Acad. Sci. USA 2017, 114, 4975–4980. [Google Scholar] [CrossRef]
- Christensen, V.G.; Stelzer, E.A.; Eikenberry, B.C.; Olds, H.T.; LeDuc, J.F.; Maki, R.P.; Saley, A.M.; Norland, J.E.; Khan, E. Cyanotoxin Mixture Models: Relating Environmental Variables and Toxin Co-Occurrence to Human Exposure Risk. J. Hazard. Mater. 2021, 415, 125560. [Google Scholar] [CrossRef]
- Mitrovic, S.M.; Pflugmacher, S.; James, K.J.; Furey, A. Anatoxin-a Elicits an Increase in Peroxidase and Glutathione S-Transferase Activity in Aquatic Plants. Aquat. Toxicol. 2004, 68, 185–192. [Google Scholar] [CrossRef]
- Zhang, Y.; Vo Duy, S.; Munoz, G.; Sauvé, S. Phytotoxic Effects of Microcystins, Anatoxin-a and Cylindrospermopsin to Aquatic Plants: A Meta-Analysis. Sci. Total Environ. 2022, 810, 152104. [Google Scholar] [CrossRef]
- Pawlik-Skowrońska, B.; Toporowska, M.; Rechulicz, J. Simultaneous Accumulation of Anatoxin-a and Microcystins in Three Fish Species Indigenous to Lakes Affected by Cyanobacterial Blooms. Oceanol. Hydrobiol. Stud. 2012, 41, 53–65. [Google Scholar] [CrossRef]
- Scarlett, K.R.; Kim, S.; Lovin, L.M.; Chatterjee, S.; Scott, J.T.; Brooks, B.W. Global Scanning of Cylindrospermopsin: Critical Review and Analysis of Aquatic Occurrence, Bioaccumulation, Toxicity and Health Hazards. Sci. Total Environ. 2020, 738, 139807. [Google Scholar] [CrossRef] [PubMed]
- De La Cruz, A.A.; Hiskia, A.; Kaloudis, T.; Chernoff, N.; Hill, D.; Antoniou, M.G.; He, X.; Loftin, K.A.; O’Shea, K.; Zhao, C.; et al. A Review on Cylindrospermopsin: The Global Occurrence, Detection, Toxicity and Degradation of a Potent Cyanotoxin. Environ. Sci. Process. Impacts 2013, 15, 1979–2003. [Google Scholar] [CrossRef] [PubMed]
- Mazmouz, R.; Chapuis-Hugon, F.; Mann, S.; Pichon, V.; Méjean, A.; Ploux, O. Biosynthesis of Cylindrospermopsin and 7-Epicylindrospermopsin in Oscillatoria Sp. Strain PCC 6506: Identification of the Cyr Gene Cluster and Toxin Analysis. Appl. Environ. Microbiol. 2010, 76, 4943–4949. [Google Scholar] [CrossRef]
- Chiswell, R.K.; Shaw, G.R.; Eaglesham, G.; Smith, M.J.; Norris, R.L.; Seawright, A.A.; Moore, M.R. Stability of Cylindrospermopsin, the Toxin from the Cyanobacterium, Cylindrospermopsis raciborskii: Effect of pH, Temperature, and Sunlight on Decomposition. Environ. Toxicol. 1999, 14, 155–161. [Google Scholar] [CrossRef]
- Aguilera, A.; Haakonsson, S.; Martin, M.V.; Salerno, G.L.; Echenique, R.O. Bloom-Forming Cyanobacteria and Cyanotoxins in Argentina: A Growing Health and Environmental Concern. Limnologica 2018, 69, 103–114. [Google Scholar] [CrossRef]
- Seifert, M.; McGregor, G.; Eaglesham, G.; Wickramasinghe, W.; Shaw, G. First Evidence for the Production of Cylindrospermopsin and Deoxy-Cylindrospermopsin by the Freshwater Benthic Cyanobacterium, Lyngbya wollei (Farlow Ex Gomont) Speziale and Dyck. Harmful Algae 2007, 6, 73–80. [Google Scholar] [CrossRef]
- Antunes, J.T.; Leão, P.N.; Vasconcelos, V.M. Cylindrospermopsis raciborskii: Review of the Distribution, Phylogeography, and Ecophysiology of a Global Invasive Species. Front. Microbiol. 2015, 6, 473. [Google Scholar] [CrossRef]
- Sundaravadivelu, D.; Sanan, T.T.; Venkatapathy, R.; Mash, H.; Tettenhorst, D.; Danglada, L.; Frey, S.; Tatters, A.O.; Lazorchak, J. Determination of Cyanotoxins and Prymnesins in Water, Fish Tissue, and Other Matrices: A Review. Toxins 2022, 14, 213. [Google Scholar] [CrossRef]
- White, S.H.; Duivenvoorden, L.J.; Fabbro, L.D.; Eaglesham, G.K. Influence of Intracellular Toxin Concentrations on Cylindrospermopsin Bioaccumulation in a Freshwater Gastropod (Melanoides Tuberculata). Toxicon 2006, 47, 497–509. [Google Scholar] [CrossRef]
- Cabang, A.B.; De Mukhopadhyay, K.; Meyers, S.; Morris, J.; Zimba, P.V.; Wargovich, M.J. Therapeutic Effects of the Euglenoid Ichthyotoxin, Euglenophycin, in Colon Cancer. Oncotarget 2017, 8, 104347–104358. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez, D.B.; Rafalski, A.; Beauchesne, K.; Moeller, P.D.; Triemer, R.E.; Zimba, P.V. Quantitative Mass Spectrometric Analysis and Post-Extraction Stability Assessment of the Euglenoid Toxin Euglenophycin. Toxins 2013, 5, 1587–1596. [Google Scholar] [CrossRef] [PubMed]
- Kulczycka, A.; Łukomska-Kowalczyk, M.; Zakryś, B.; Milanowski, R. PCR Identification of Toxic Euglenid Species Euglena sanguinea. J. Appl. Phycol. 2018, 30, 1759–1763. [Google Scholar] [CrossRef]
- Díez-Quijada, L.; Prieto, A.I.; Guzmán-Guillén, R.; Jos, A.; Cameán, A.M. Occurrence and Toxicity of Microcystin Congeners Other than MC-LR and MC-RR: A Review. Food Chem. Toxicol. 2019, 125, 106–132. [Google Scholar] [CrossRef]
- Svirčev, Z.; Drobac, D.; Tokodi, N.; Mijović, B.; Codd, G.A.; Meriluoto, J. Toxicology of Microcystins with Reference to Cases of Human Intoxications and Epidemiological Investigations of Exposures to Cyanobacteria and Cyanotoxins. Arch. Toxicol. 2017, 91, 621–650. [Google Scholar] [CrossRef]
- Chernoff, N.; Hill, D.; Lang, J.; Schmid, J.; Le, T.; Farthing, A.; Huang, H. The Comparative Toxicity of 10 Microcystin Congeners Administered Orally to Mice: Clinical Effects and Organ Toxicity. Toxins 2020, 12, 403. [Google Scholar] [CrossRef] [PubMed]
- Sivonen, K.; Jones, G. Cyanobacterial Toxins. In Toxic Cyanobacteria in Water: A Guide to Their Public Health Consequences, Monitoring, and Management; Chorus, I., Bartram, J., Eds.; World Health Organization: London, UK, 1999; pp. 41–112. [Google Scholar]
- Nishiwaki-Matsushima, R.; Ohta, T.; Nishiwaki, S.; Suganuma, M.; Kohyama, K.; Ishikawa, T.; Carmichael, W.W.; Fujiki, H. Liver Tumor Promotion by the Cyanobacterial Cyclic Peptide Toxin Microcystin-LR. J. Cancer Res. Clin. Oncol. 1992, 118, 420–424. [Google Scholar] [CrossRef]
- Dyble, J.; Fahnenstiel, G.L.; Litaker, R.W.; Millie, D.F.; Tester, P.A. Microcystin Concentrations and Genetic Diversity of Microcystis in the Lower Great Lakes. Environ. Toxicol. 2008, 23, 507–516. [Google Scholar] [CrossRef]
- Hardy, F.J.; Johnson, A.; Hamel, K.; Preece, E. Cyanotoxin Bioaccumulation in Freshwater Fish, Washington State, USA. Environ. Monit. Assess. 2015, 187, 667. [Google Scholar] [CrossRef]
- Preece, E.P.; Hardy, F.J.; Moore, B.C.; Bryan, M. A Review of Microcystin Detections in Estuarine and Marine Waters: Environmental Implications and Human Health Risk. Harmful Algae 2017, 61, 31–45. [Google Scholar] [CrossRef]
- Woller-Skar, M.M.; Russell, A.L.; Gaskill, J.A.; Luttenton, M.R. Microcystin in Multiple Life Stages of Hexagenia limbata, with Implications for Toxin Transfer. J. Great Lakes Res. 2020, 46, 666–671. [Google Scholar] [CrossRef]
- Corbel, S.; Mougin, C.; Bouaïcha, N. Cyanobacterial Toxins: Modes of Actions, Fate in Aquatic and Soil Ecosystems, Phytotoxicity and Bioaccumulation in Agricultural Crops. Chemosphere 2014, 96, 1–15. [Google Scholar] [CrossRef]
- Manning, S.R.; La Claire, J.W. Prymnesins: Toxic Metabolites of the Golden Alga, Prymnesium parvum Carter (Haptophyta). Mar. Drugs 2010, 8, 678–704. [Google Scholar] [CrossRef] [PubMed]
- Lindholm, T.; Öhman, P.; Kurki-Helasmo, K.; Kincaid, B.; Meriluoto, J. Toxic Algae and Fish Mortality in a Brackish-Water Lake in Aland, SW Finland. Hydrobiologia 1999, 397, 109–120. [Google Scholar] [CrossRef]
- Rasmussen, S.A.; Meier, S.; Andersen, N.G.; Blossom, H.E.; Duus, J.Ø.; Nielsen, K.F.; Hansen, P.J.; Larsen, T.O. Chemodiversity of Ladder-Frame Prymnesin Polyethers in Prymnesium parvum. J. Nat. Prod. 2016, 79, 2250–2256. [Google Scholar] [CrossRef]
- Valenti, T.W.; James, S.V.; Lahousse, M.; Schug, K.A.; Roelke, D.L.; Grover, J.P.; Brooks, B.W. Influence of pH on Amine Toxicology and Implications for Harmful Algal Bloom Ecology. Toxicon 2010, 55, 1038–1043. [Google Scholar] [CrossRef]
- O’Neill, K.; Musgrave, I.F.; Humpage, A.R. Low Dose Extended Exposure to Saxitoxin and Its Potential Neurodevelopmental Effects: A Review. Environ. Toxicol. Pharmacol. 2016, 48, 7–16. [Google Scholar] [CrossRef] [PubMed]
- Ballot, A.; Bernard, C.; Fastner, J. Saxitoxin and Analogues. In Handbook of Cyanobacterial Monitoring and Cyanotoxin Analysis; Meriluoto, J., Spoof, L., Codd, G.A., Eds.; John Wiley and Sons, Ltd.: Chichester, UK, 2017; pp. 148–154. [Google Scholar]
- Wiese, M.; D’Agostino, P.M.; Mihali, T.K.; Moffitt, M.C.; Neilan, B.A. Neurotoxic Alkaloids: Saxitoxin and Its Analogs. Mar. Drugs 2010, 8, 2185–2211. [Google Scholar] [CrossRef]
- Onodera, H.; Satake, M.; Oshima, Y.; Yasumoto, T.; Carmichael, W.W. New Saxitoxin Analogues from the Freshwater Filamentous Cyanobacterium Lyngbya wollei. Nat. Toxins 1997, 5, 146–151. [Google Scholar] [CrossRef]
- Casali, S.P.; Dos Santos, A.C.A.; De Falco, P.B.; Do Carmo Calijuri, M. Influence of Environmental Variables on Saxitoxin Yields by Cylindrospermopsis raciborskii in a Mesotrophic Subtropical Reservoir. J. Water Health 2017, 15, 509–518. [Google Scholar] [CrossRef]
- Smith, Z.J.; Martin, R.M.; Wei, B.; Wilhelm, S.W.; Boyer, G.L. Spatial and Temporal Variation in Paralytic Shellfish Toxin Production by Benthic Microseira (Lyngbya) wollei in a Freshwater New York Lake. Toxins 2019, 11, 44. [Google Scholar] [CrossRef] [PubMed]
- Negri, A.P.; Jones, G.; Blackburn, S.I.; Oshima, Y.; Onodera, H. Effect of Culture and Bloom Development and of Sample Storage on Paralytic Shellfish Poisons in the Cyanobacterium Anabaena circinalis. J. Phycol. 1997, 33, 26–35. [Google Scholar] [CrossRef]
- Belykh, O.I.; Tikhonova, I.V.; Kuzmin, A.V.; Sorokovikova, E.G.; Fedorova, G.A.; Khanaev, I.V.; Sherbakova, T.A.; Timoshkin, O.A. First Detection of Benthic Cyanobacteria in Lake Baikal Producing Paralytic Shellfish Toxins. Toxicon 2016, 121, 36–40. [Google Scholar] [CrossRef]
- Carmichael, W.W.; Jones, C.L.A.; Mahmood, N.A.; Theiss, W.C. Algal Toxins and Water-Based Diseases. Crit. Rev. Environ. Control 1985, 15, 275–313. [Google Scholar] [CrossRef]
- Cusick, K.D.; Sayler, G.S. An Overview on the Marine Neurotoxin, Saxitoxin: Genetics, Molecular Targets, Methods of Detection and Ecological Functions. Mar. Drugs 2013, 11, 991–1018. [Google Scholar] [CrossRef]
- Negri, A.P.; Jones, G.J. Bioaccumulation of Paralytic Shellfish Poisoning (PSP) Toxins from the Cyanobacterium Anabaena circinalis by the Freshwater Mussel Alathyria condola. Toxicon 1995, 33, 667–678. [Google Scholar] [CrossRef]
- De Calado, S.L.M.; Santos, G.S.; Wojciechowski, J.; de Magalhães, V.F.; Silva de Assis, H.C. The Accumulation Dynamics, Elimination and Risk Assessment of Paralytic Shellfish Toxins in Fish from a Water Supply Reservoir. Sci. Total Environ. 2019, 651, 3222–3229. [Google Scholar] [CrossRef]
- Giovannardi, S.; Pollegioni, L.; Pomati, F.; Rossetti, C.; Sacchi, S.; Sessa, L.; Calamari, D. Toxic Cyanobacterial Blooms in Lake Varese (Italy): A Multidisciplinary Approach. Environ. Toxicol. 1999, 14, 127–134. [Google Scholar] [CrossRef]
- Lenz, K.A.; Miller, T.R.; Ma, H. Anabaenopeptins and Cyanopeptolins Induce Systemic Toxicity Effects in a Model Organism the Nematode Caenorhabditis elegans. Chemosphere 2019, 214, 60–69. [Google Scholar] [CrossRef]
- Cox, P.A.; Banack, S.A.; Murch, S.J.; Rasmussen, U.; Tien, G.; Bidigare, R.R.; Metcalf, J.S.; Morrison, L.F.; Codd, G.A.; Bergman, B. Diverse Taxa of Cyanobacteria Produce Beta-N-Methylamino-L-Alanine, a Neurotoxic Amino Acid. Proc. Natl. Acad. Sci. USA 2005, 102, 5074–5078. [Google Scholar] [CrossRef]
- Faassen, E.J.; Harkema, L.; Begeman, L.; Lurling, M. First Report of (Homo)Anatoxin-a and Dog Neurotoxicosis after Ingestion of Benthic Cyanobacteria in The Netherlands. Toxicon 2012, 60, 378–384. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.A. Analytical Techniques for the Detection of α-Amino-β-Methylaminopropionic Acid. Analyst 2012, 137, 1991–2005. [Google Scholar] [CrossRef]
- Monteiro, P.R.; Do Amaral, S.C.; Siqueira, A.S.; Xavier, L.P.; Santos, A.V. Anabaenopeptins: What We Know so Far. Toxins 2021, 13, 522. [Google Scholar] [CrossRef] [PubMed]
- Beversdorf, L.J.; Rude, K.; Weirich, C.A.; Bartlett, S.L.; Seaman, M.; Kozik, C.; Biese, P.; Gosz, T.; Suha, M.; Stempa, C.; et al. Analysis of Cyanobacterial Metabolites in Surface and Raw Drinking Waters Reveals More than Microcystin. Water Res. 2018, 140, 280–290. [Google Scholar] [CrossRef] [PubMed]
- Saker, M.L.; Fastner, J.; Dittmann, E.; Christiansen, G.; Vasconcelos, V.M. Variation between Strains of the Cyanobacterium Microcystis aeruginosa Isolated from a Portuguese River. J. Appl. Microbiol. 2005, 99, 749–757. [Google Scholar] [CrossRef]
- Cerasino, L.; Capelli, C.; Salmaso, N. A Comparative Study of the Metabolic Profiles of Common Nuisance Cyanobacteria in Southern Perialpine Lakes. Adv. Oceanogr. Limnol. 2017, 8, 22–32. [Google Scholar] [CrossRef]
- Mazur-Marzec, H.; Fidor, A.; Cegłowska, M.; Wieczerzak, E.; Kropidłowska, M.; Goua, M.; Macaskill, J.; Edwards, C. Cyanopeptolins with Trypsin and Chymotrypsin Inhibitory Activity from the Cyanobacterium Nostoc edaphicum CCNP1411. Mar. Drugs 2018, 16, 220. [Google Scholar] [CrossRef]
- Janssen, E.M.-L. Cyanobacterial Peptides beyond Microcystins—A Review on Co-Occurrence, Toxicity, and Challenges for Risk Assessment. Water Res. 2019, 151, 488–499. [Google Scholar] [CrossRef]
- Weirich, C.A.; Miller, T.R. Freshwater Harmful Algal Blooms: Toxins and Children’s Health. Curr. Probl. Pediatr. Adolesc. Health Care 2014, 44, 2–24. [Google Scholar] [CrossRef]
- Funari, E.; Testai, E. Human Health Risk Assessment Related to Cyanotoxins Exposure. Crit. Rev. Toxicol. 2008, 38, 97–125. [Google Scholar] [CrossRef]
- Faltermann, S.; Zucchi, S.; Kohler, E.; Blom, J.F.; Pernthaler, J.; Fent, K. Molecular Effects of the Cyanobacterial Toxin Cyanopeptolin (CP1020) Occurring in Algal Blooms: Global Transcriptome Analysis in Zebrafish Embryos. Aquat. Toxicol. 2014, 149, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Zhao, Y. Reduced Zebrafish Transcriptome Atlas toward Understanding Environmental Neurotoxicants. Environ. Sci. Technol. 2018, 52, 7120–7130. [Google Scholar] [CrossRef] [PubMed]
- Christensen, V.G.; Olds, H.T.; Norland, J.; Khan, E. Phytoplankton Community Interactions and Cyanotoxin Mixtures in Three Recurring Surface Blooms within One Lake. J. Hazard. Mater. 2022, 427, 128142. [Google Scholar] [CrossRef]
- Howard, M.D.A.; Kudela, R.M.; Hayashi, K.; Tatters, A.O.; Caron, D.A.; Theroux, S.; Oehrle, S.; Roethler, M.; Donovan, A.; Loftin, K.; et al. Multiple Co-Occurring and Persistently Detected Cyanotoxins and Associated Cyanobacteria in Adjacent California Lakes. Toxicon 2021, 192, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Smith, Z.J.; Conroe, D.E.; Schulz, K.L.; Boyer, G.L. Limnological Differences in a Two-Basin Lake Help to Explain the Occurrence of Anatoxin-a, Paralytic Shellfish Poisoning Toxins, and Microcystins. Toxins 2020, 12, 559. [Google Scholar] [CrossRef] [PubMed]
- Beversdorf, L.J.; Weirich, C.; Bartlett, S.; Miller, T. Variable Cyanobacterial Toxin and Metabolite Profiles across Six Eutrophic Lakes of Differing Physiochemical Characteristics. Toxins 2017, 9, 62. [Google Scholar] [CrossRef]
- Dietrich, D.R.; Fischer, A.; Michel, C.; Hoeger, S. Toxin Mixture in Cyanobacterial Blooms—A Critical Comparison of Reality with Current Procedures Employed in Human Health Risk Assessment. In Cyanobacterial Harmful Algal Blooms: State of the Science and Research Needs; Springer: New York, NY, USA, 2008; pp. 885–912. [Google Scholar]
- Burford, M.A.; Carey, C.C.; Hamilton, D.P.; Huisman, J.; Paerl, H.W.; Wood, S.A.; Wulff, A. Perspective: Advancing the Research Agenda for Improving Understanding of Cyanobacteria in a Future of Global Change. Harmful Algae 2020, 91, 101601. [Google Scholar] [CrossRef]
- Paerl, H.W. Impacts of Climate Change on Cyanobacteria in Aquatic Environments. In Climate Change and Microbial Ecology: Current Research and Future Trends; Marxsen, J., Ed.; Caister Academic Press: Wymondham, UK, 2016. [Google Scholar]
- Chapra, S.C.; Boehlert, B.; Fant, C.; Bierman, V.J.; Henderson, J.; Mills, D.; Mas, D.M.L.; Rennels, L.; Jantarasami, L.; Martinich, J.; et al. Climate Change Impacts on Harmful Algal Blooms in U. S. Freshwaters: A Screening-Level Assessment. Environ. Sci. Technol. 2017, 51, 8933–8943. [Google Scholar] [CrossRef]
- Brandenburg, K.M.; de Senerpont Domis, L.N.; Wohlrab, S.; Krock, B.; John, U.; van Scheppingen, Y.; van Donk, E.; Van de Waal, D.B. Combined Physical, Chemical and Biological Factors Shape Alexandrium ostenfeldii Blooms in the Netherlands. Harmful Algae 2017, 63, 146–153. [Google Scholar] [CrossRef]
- Kolman, M.A.; Torres, L.L.; Martin, M.L.; Salerno, G.L. Sucrose Synthase in Unicellular Cyanobacteria and Its Relationship with Salt and Hypoxic Stress. Planta 2012, 235, 955–964. [Google Scholar] [CrossRef]
- Sandrini, G.; Huisman, J.; Matthijs, H.C.P. Potassium Sensitivity Differs among Strains of the Harmful Cyanobacterium Microcystis and Correlates with the Presence of Salt Tolerance Genes. FEMS Microbiol. Lett. 2015, 362, fnv121. [Google Scholar] [CrossRef] [PubMed]
- Tanabe, Y.; Yamaguchi, H.; Sano, T.; Kawachi, M. A Novel Salt-Tolerant Genotype Illuminates the Sucrose Gene Evolution in Freshwater Bloom-Forming Cyanobacterium Microcystis aeruginosa. FEMS Microbiol. Lett. 2019, 366, fnz190. [Google Scholar] [CrossRef] [PubMed]
- Tonk, L.; Bosch, K.; Visser, P.M.; Huisman, J. Salt Tolerance of the Harmful Cyanobacterium Microcystis aeruginosa. Aquat. Microb. Ecol. 2007, 46, 117–123. [Google Scholar] [CrossRef]
- Des Aulnois, M.G.; Roux, P.; Caruana, A.; Réveillon, D.; Briand, E.; Hervé, F.; Savar, V.; Bormans, M.; Amzil, Z. Physiological and Metabolic Responses of Freshwater and Brackish-Water Strains of Microcystis aeruginosa Acclimated to a Salinity Gradient: Insight into Salt Tolerance. Appl. Environ. Microbiol. 2019, 85, e01614-19. [Google Scholar] [CrossRef]
- Rosen, B.H.; Loftin, K.A.; Graham, J.L.; Stahlhut, K.N.; Riley, J.M.; Johnson, B.D.; Senegal, S. Understanding the Effect of Salinity Tolerance on Cyanobacteria Associated with a Harmful Algal Bloom in Lake Okeechobee, Florida; U.S. Geological Survey Scientific Investigations Report 2018-5092; US Geological Survey: Reston, VA, USA, 2018. [Google Scholar]
- Tatters, A.O.; Smith, J.; Kudela, R.M.; Hayashi, K.; Da, M.; Donovan, A.R.; Loftin, K.A.; Caron, D.A. The Tide Turns: Episodic and Localized Cross-Contamination of a California Coastline with Cyanotoxins. Harmful Algae 2021, 103, 102003. [Google Scholar] [CrossRef]
- Melero-Jiménez, I.J.; Martín-Clemente, E.; García-Sánchez, M.J.; Bañares-España, E.; Flores-Moya, A. The Limit of Resistance to Salinity in the Freshwater Cyanobacterium Microcystis aeruginosa Is Modulated by the Rate of Salinity Increase. Ecol. Evol. 2020, 10, 5045–5055. [Google Scholar] [CrossRef]
- Lundgren, V.M.; Roelke, D.L.; Brooks, B.W.; Granéli, E.; Davis, S.L.; Baty, T.; Scott, W.C. Prymnesium parvum Invasion Success into Coastal Bays of the Gulf of Mexico: Galveston Bay Case Study. Harmful Algae 2015, 43, 31–45. [Google Scholar] [CrossRef]
- Richardson, E.T.; Patiño, R. Growth of the Harmful Alga, Prymnesium parvum (Prymnesiophyceae), after Gradual and Abrupt Increases in Salinity. J. Phycol. 2021, 57, 1335–1344. [Google Scholar] [CrossRef]




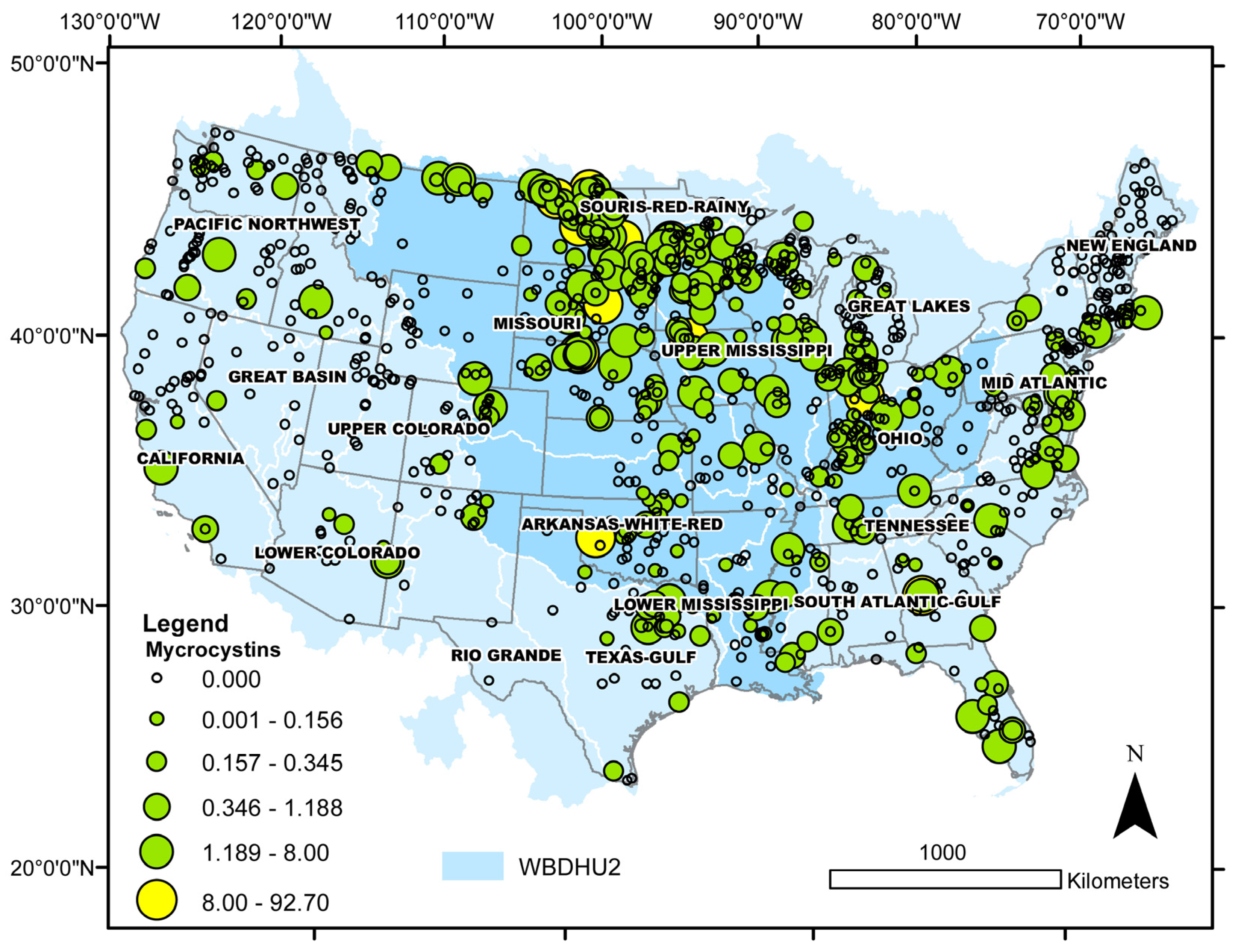
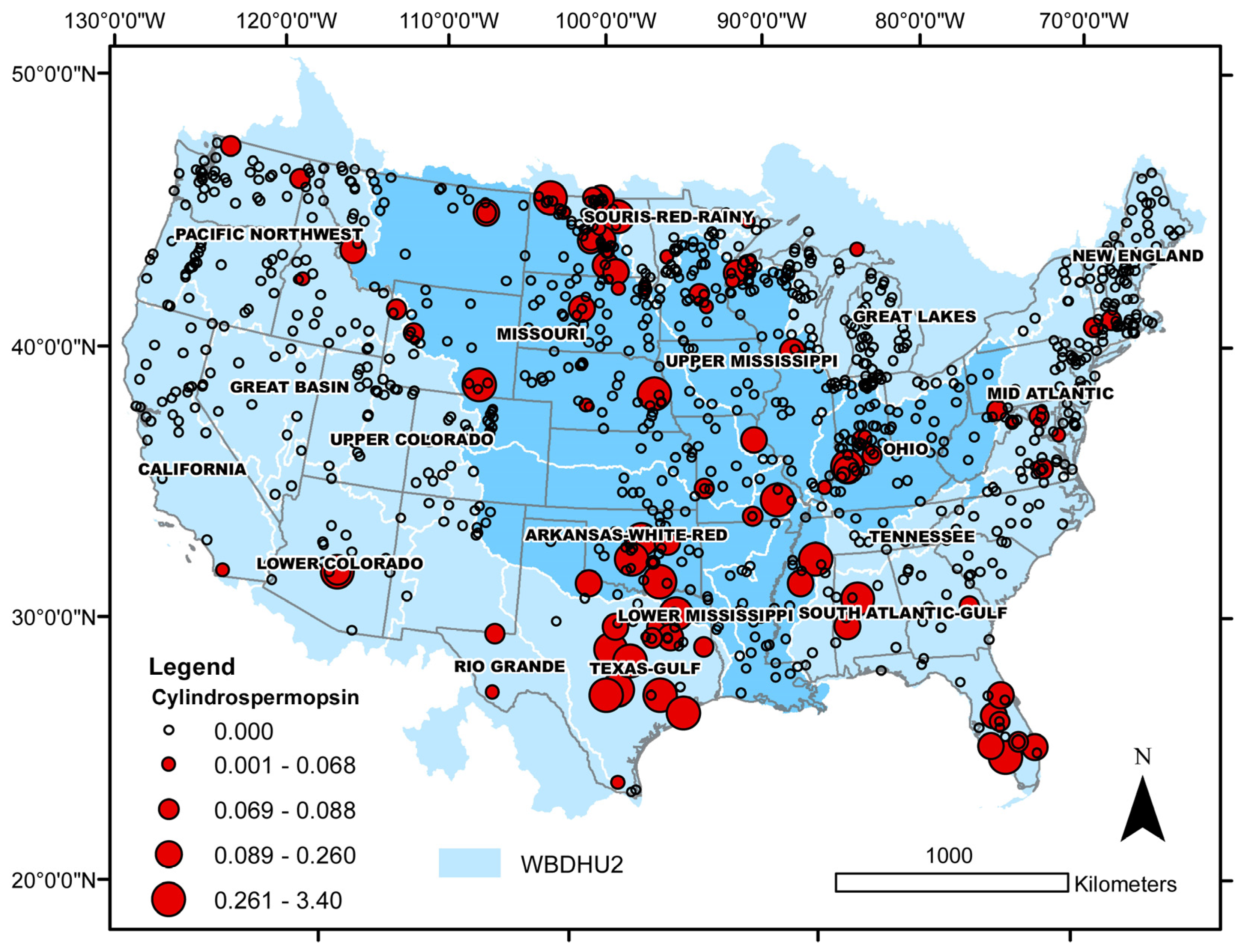
| Taxon | Number of Cases | Median Cell Density (min, max) × 104 | Median Salinity (min, max) | Median Sulfate (min, max) | Lake Salinity | Observed Distribution (%) | Binomial Test p-Value |
|---|---|---|---|---|---|---|---|
| Anabaena | 12 | 1.52 (1.09, 125) | 0.17 (0.02, 2.15) | 10.0 (0.25, 1483) | freshwater | 67 | 0.0433 |
| brackish | 33 | ||||||
| Aphanizomenon | 126 | 5.73 (1.00, 105) | 0.17 (0.00, 2.97) | 15.8 (0.02, 2343) | freshwater | 76 | <0.0001 |
| brackish | 24 | ||||||
| Dolichospermum | 81 | 2.73 (1.02, 39.2) | 0.16 (0.01, 2.25) | 12.4 (0.00, 1736) | freshwater | 81 | 0.0809 |
| brackish | 19 | ||||||
| Microcystis | 93 | 3.30 (1.03, 137) | 0.18 (0.01, 3.79) | 17.9 (0.00, 1525) | freshwater | 80 | 0.0146 |
| brackish | 20 | ||||||
| Planktothrix | 46 | 25.0 (1.07, 558) | 0.43 (0.02, 2.94) | 128 (0.27, 2888) | freshwater | 52 | <0.0001 |
| brackish | 48 | ||||||
| Euglena | 26 | 0.03 (0.006, 0.46) | 0.13 (0.01, 3.79) | 8.87 (0.22, 2888) | freshwater | 81 | 0.2218 |
| brackish | 19 |
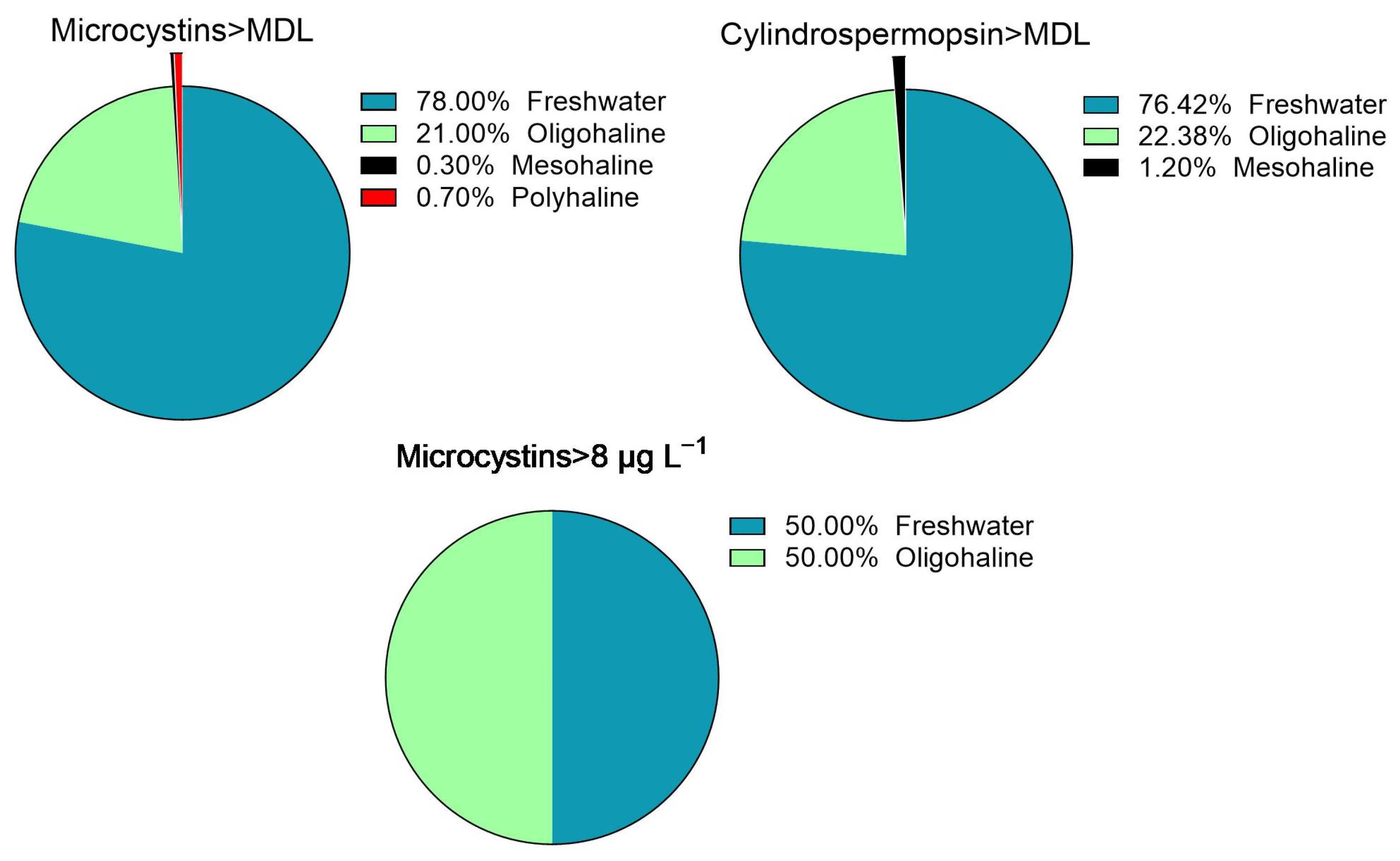
| Toxin | Number of Cases | Median Toxin Concentration (min, max) | Median Salinity (min, max) | Lake Salinity | Observed Distribution (%) | Binomial Test p-Value |
|---|---|---|---|---|---|---|
| Microcystins > MDL | 295 | 0.34 (0.10, 92.7) | 0.17 (0.00, 21.5) | freshwater | 78 | <0.0001 |
| brackish | 22 | |||||
| Cylindrospermopsin > MDL | 85 | 0.09 (0.05, 3.40) | 0.11 (0.00, 7.24) | freshwater | 76 | 0.002 |
| brackish | 24 | |||||
| Microcystins > 8 μg L−1 | 12 | 16.2 (8.02, 92.7) | 0.44 (0.01, 2.10) | freshwater | 50 | 0.0013 |
| brackish | 50 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Patiño, R.; Christensen, V.G.; Graham, J.L.; Rogosch, J.S.; Rosen, B.H. Toxic Algae in Inland Waters of the Conterminous United States—A Review and Synthesis. Water 2023, 15, 2808. https://doi.org/10.3390/w15152808
Patiño R, Christensen VG, Graham JL, Rogosch JS, Rosen BH. Toxic Algae in Inland Waters of the Conterminous United States—A Review and Synthesis. Water. 2023; 15(15):2808. https://doi.org/10.3390/w15152808
Chicago/Turabian StylePatiño, Reynaldo, Victoria G. Christensen, Jennifer L. Graham, Jane S. Rogosch, and Barry H. Rosen. 2023. "Toxic Algae in Inland Waters of the Conterminous United States—A Review and Synthesis" Water 15, no. 15: 2808. https://doi.org/10.3390/w15152808
APA StylePatiño, R., Christensen, V. G., Graham, J. L., Rogosch, J. S., & Rosen, B. H. (2023). Toxic Algae in Inland Waters of the Conterminous United States—A Review and Synthesis. Water, 15(15), 2808. https://doi.org/10.3390/w15152808









