Gloeotrichia cf. natans (Cyanobacteria) in the Continuous Permafrost Zone of Buotama River, Lena Pillars Nature Park, in Yakutia (Russia)
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Sampling
2.3. Algological Analysis
2.4. Water Chemistry Analysis
2.5. Detection of Cyanotoxins by Liquid Chromatography–Mass Spectrometry
2.6. PCR Analysis of Cyanotoxin Biosynthesis Genes
2.7. Molecular Approach to Species Identification
2.8. Molecular Analyses
3. Results
3.1. Water Chemistry
3.2. Field Algological Observations
3.3. Microscopic Survey
3.4. Phylogeny
3.5. Cyanotoxins Detection
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Codd, G.A.; Morrison, L.F.; Metcalf, J.S. Cyanobacterial toxins: Risk management for health protection. Toxicol. Appl. Pharmacol. 2005, 203, 264–272. [Google Scholar] [CrossRef] [PubMed]
- Paerl, H.W.; Huisman, J. Climate change: A catalyst for global expansion of harmful cyanobacterial blooms. Environ. Microbiol. 2009, 1, 27–37. [Google Scholar] [CrossRef] [PubMed]
- Neil, J.M.O.; Davis, T.W.; Burford, M.A.; Gobler, C.J. The rise of harmful cyanobacteria blooms: The potential roles of eutrophication and climate change. Harmful Algae 2012, 14, 313–334. [Google Scholar] [CrossRef]
- Gaget, V.; Hobson, P.; Keulen, A.; Newton, K.; Monis, P.; Humpage, A.; Weyrich, L.; Brookes, J. Toolbox for the sampling and monitoring of benthic cyanobacteria. Water Res. 2020, 169, 115222. [Google Scholar] [CrossRef]
- Wood, S.; Kelly, L.; Bouma-Gregson, K.; Humbert, J.; Laughinghouse, H.; Lazorchak, J.; McAllister, T.; McQueen, A.; Pokrzywinski, K.; Puddick, J.; et al. Toxic benthic freshwater cyanobacterial proliferations: Challenges and solutions for enhancing knowledge and improving monitoring and mitigation. Freshw. Biol. 2020, 65, 1824–1842. [Google Scholar] [CrossRef] [PubMed]
- Heino, J.; Virkkala, R.; Toivonen, H. Climate change and freshwater biodiversity: Detected patterns, future trends and adaptations in northern regions. Biol. Rev. 2009, 84, 39–54. [Google Scholar] [CrossRef]
- Winder, M.; Sommer, U. Phytoplankton response to a changing climate. Hydrobiologia 2012, 698, 5–16. [Google Scholar] [CrossRef]
- Korneva, L.G. Invasions of alien species of planktonic microalgae into the fresh waters of Holarctic (Review). Russ. J. Biol. Invasions 2014, 5, 65–81. [Google Scholar] [CrossRef]
- Lang-Yona, N.; Alster, A.; Cummings, D.; Freiman, Z.; Kaplan-Levy, R.; Lupu, A.; Malinsky-Rushansky, N.; Ninio, S.; Sukenik, A.; Viner-Mozzini, Y.; et al. Gloeotrichia pisum in Lake Kinneret: A successful epiphytic cyanobacterium. J. Phycol. 2023, 59, 97–110. [Google Scholar] [CrossRef]
- Komárek, J. Heterocytous Genera. Cyanoprokaryota; Springer Spektrum: Berlin, Germany, 2013; Volume 3, pp. 3–1130. [Google Scholar]
- Carey, C.C.; Haney, J.F.; Cottingham, K.L. First report of microcystin-LR in the cyanobacterium Gloeotrichia echinulate. Environ. Toxicol. 2007, 22, 337–339. [Google Scholar] [CrossRef]
- Carey, C.C.; Ewing, H.A.; Cottingham, K.L.; Weathers, K.C.; Thomas, R.Q.; Haney, J.F. Occurrence and toxicity of the cyanobacterium Gloeotrichia echinulata in low-nutrient lakes in the northeastern United States. Aquat. Ecol. 2012, 46, 395–409. [Google Scholar] [CrossRef]
- Stamann, S. “Toxin Production and Population Dynamics of Gloeotrichia echinulata with Considerations of Global Climate Change”. Master’s Thesis, Grand Valley State University, Allendale, Michigan, USA, 2015; p. 775. Available online: http://scholarworks.gvsu.edu/theses/775 (accessed on 25 May 2023).
- Cantoral Uriza, E.A.; Asencio, A.D.; Aboal, M. Are We Underestimating Benthic Cyanotoxins? Extensive Sampling Results from Spain. Toxins 2017, 9, 385. [Google Scholar] [CrossRef] [PubMed]
- Guiry, M.D.; Guiry, G.M. AlgaeBase; World-Wide Electronic Publication, National University of Ireland: Galway, Ireland. Available online: https://www.algaebase.org (accessed on 29 March 2023).
- Strunecky, O.; Ivanova, A. An updated classification of cyanobacterial orders and families based on phylogenomic and polyphasic analysis. J. Phycol. 2023, 59, 12–51. [Google Scholar] [CrossRef] [PubMed]
- Izyumenko, S.A. Climate of the Yakut ASSR (Atlas); Hydrometeorological Publishing House: Leningrad, Russia, 1968; 32p. (In Russian) [Google Scholar]
- State Water Register. Available online: http://textual.ru/gvr/index.php?card=243859 (accessed on 23 March 2023). (In Russian).
- Surface Water Resources of the USSR. Lensko-Indigirsky District; Gidrometeoizdat Publisher: Leningrad, Russia, 1972; 651p. (In Russian) [Google Scholar]
- Chistyakov, G.Y. Water Resources of the Rivers of Yakutia; Nauka: Moscow, Russia, 1964; 255p. (In Russian) [Google Scholar]
- Semenov, A.D. (Ed.) Guidance on the Chemical Analysis of Surface Waters of the Land; Gidrometeoizdat Publisher: Leningrad, Russia, 1977; 541p. (In Russian) [Google Scholar]
- Chernova, E.; Russkikh, I.; Voyakina, E.; Zhakovskaya, Z. Occurrence of microcystins and anatoxin-a in eutrophic lakes of Saint Petersburg, Northwestern Russia. Oceanol. Hydrobiol. Stud. 2016, 45, 466–484. [Google Scholar] [CrossRef]
- Jungblut, A.D.; Neilan, B.A. Molecular identification and evolution of the cyclic peptide hepatotoxins, microcystin and nodularin, synthetase genes in three orders of cyanobacteria. Arch. Microbiol. 2006, 185, 107–114. [Google Scholar] [CrossRef] [PubMed]
- Mihali, T.K.; Kellmann, R.; Muenchhoff, J.; Barrow, K.D.; Neilan, B.A. Characterization of the gene cluster responsible for cylindrospermopsin biosynthesis, Applied and Environmental. Microbiology 2008, 74, 716–722. [Google Scholar]
- Ballot, A.; Fastner, J.; Wiedner, C. Paralytic shellfish poisoning toxin-producing cyanobacterium Aphanizomenon gracile in Northeast Germany. Appl. Environ. Microbiol. 2010, 76, 1173–1180. [Google Scholar] [CrossRef]
- Rantala-Ylinen, A.; Känä, S.; Wang, H.; Rouhiainen, L.; Wahlsten, M.; Rizzi, E.; Berg, K.; Gugger, M.; Sivonen, K. Anatoxin–a synthetase gene cluster of the cyanobacterium Anabaena sp. strain 37 and molecular methods to detect potential producers. Appl. Environ. Microbiol. 2011, 77, 7271–7278. [Google Scholar] [CrossRef]
- Wilmotte, A.; Van Der Auwera, C.; De Wachter, R. Structure of the 16S ribosomal RNA of the thermophilic cyanobacteria Chlorogloeopsis HTF (‘Mastigocladus laminosus HTF’) strain PCC7518 and phylogenetic analysis. FEBS Lett. 1993, 317, 96–100. [Google Scholar] [CrossRef]
- Nübel, U.; Garcia-Pichel, F.; Muyzer, G. PCR primers to amplify 16S rRNA genes from cyanobacteria. Appl. Environ. Microbiol. 1997, 63, 3327–3332. [Google Scholar] [CrossRef]
- Wilmotte, A. Molecular evolution and taxonomy of the cyanobacteria. In The Molecular Biology of Cyanobacteria; Bryant, D.A., Ed.; Kluwer: Dordrecht, The Netherlands, 1994; pp. 1–25. [Google Scholar] [CrossRef]
- Hall, T.A. BioEdit: A User-Friendly Biological Sequence Alignment Editor and Analysis Program for Windows 95/98/NT. Nucleic Acids Symp. Ser. 1999, 41, 95–98. [Google Scholar]
- Benson, D.; Cavanaugh, M.; Clark, K.; Karsch-Mizrachi, I.; Lipman, D.; Ostell, J. Sayers EW. GenBank. Nucleic Acids Res. 2013, 41, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Lefler, F.; Berthold, D. Dail Laughinghouse H 4th. CyanoSeq: A database of cyanobacterial 16S rRNA sequences with curated taxonomy. J Phycol. 2023, 59, 470–480. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, L.-T.; Schmidt, H.A.; von Haeseler, A.; Minh, B.Q. IQ-TREE: A fast and effective stochastic algorithm for estimating maximum likelihood phylogenies. Mol. Biol. Evol. 2015, 32, 268–274. [Google Scholar] [CrossRef]
- Kalyaanamoorthy, S.; Minh, B.Q.; Wong, T.K.F.; von Haeseler, A.; Jermiin, L.S. ModelFinder: Fast model selection for accurate phylogenetic estimates. Nat. Methods 2017, 14, 587–589. [Google Scholar] [CrossRef]
- Hoang, D.T.; Chernomor, O.; von Haeseler, A.; Minh, B.Q.; Vinh, L.S. UFBoot2: Improving the ultrafast bootstrap approximation. Mol. Biol. Evol. 2018, 35, 518–522. [Google Scholar] [CrossRef]
- Tamura, K.; Stecher, G.; Kumar, S. MEGA11: Molecular Evolutionary Genetics Analysis version 11. Mol. Biol. Evol. 2021, 38, 3022–3027. [Google Scholar] [CrossRef]
- Yarza, P.; Yilmaz, P.; Pruesse, E.; Glöckner, L.W.; Schleifer, K.H.; Witman, W.B.; Euzéby, J.; Amann, R. Rosselló-Móra Uniting the classification of cultured and uncultured bacteria and archaea using 16S rRNA gene sequences. Nat. Rev. Microbiol. 2014, 12, 635–645. [Google Scholar] [CrossRef]
- Gabyshev, V.A.; Gabysheva, O.I. Phytoplankton of the Largest Rivers of Yakutia and Adjacent Territories of Eastern Siberia; Novosibirsk: SibAk, Russia, 2018; 414p. (In Russian) [Google Scholar]
- Getzen, M.V. Algae of Pechora River Basin; Nauka Publisher: Leningrad, Russia, 1973; 148p. (In Russian) [Google Scholar]
- Kiselev, G.A.; Balashova, N.B.; Ulanova, A.A. Ecological characteristics of algae from the lakes of Samoilovsky Island (Lena Delta Nature Reserve, Russia). In Proceedings of the IV International Conference «Advances in Modern Phycology», Kyiv, Ukraine, 23–25 May 2012. (In Russian). [Google Scholar]
- Melekhin, A.V.; Davydov, D.A.; Borovichev, E.A.; Shalygin, S.S.; Konstantinova, N.A. CRIS—Service for input, storage and analysis of the biodiversity data of the cryptogams. Folia Cryptogam. Est. 2019, 56, 99–108. [Google Scholar] [CrossRef]
- GBIF.org. GBIF Occurrence (7 April 2023). Available online: https://doi.org/10.15468/dl.85tp8k (accessed on 25 May 2023).
- Komarenko, L.Y.; Vasilyeva, I.I. Freshwater Diatoms and Blue-Green Algae of Water Bodies of Yakutia; Nauka: Moscow, Russia, 1975; 424p. (In Russian) [Google Scholar]
- Kopyrina, L.; Pshennikova, P.; Barinova, S. Diversity and ecological characteristic of algae and cyanobacteria of thermokarst lakes in Yakutia (northeastern Russia). Int. J. Oceanogr. Hydrobiol. 2020, 49, 99–122. [Google Scholar] [CrossRef]
- Zakharova, V.I.; Kuznetsova, L.V.; Ivanova, Y.I. Diversity of the flora of Yakutia; Publishing house of the Siberian Branch of the Russian Academy of Sciences: Novosibirsk, Russia, 2005; 328p. (In Russian) [Google Scholar]
- CYANOpro. Available online: http://kpabg.ru/cyanopro/?q=node/96073 (accessed on 23 March 2023).
- Elenkin, A.A. Kamchatka Expedition of Fyodor Pavlovich Ryabushinsky. Botanical Department. Issue. 2. Spore Plants of Kamchatka: 1) Algae, 2) Mushrooms; Printing House P.P. Ryabushinsky: Moscow, Russia, 1914; pp. 1–402. (In Russian) [Google Scholar]
- Medvedeva, L.A.; Nikulina, T.V. Catalogue of Freshwater Algae of the Southern Part of the Russian Far East; Vladivostok: Dalnauka, Russia, 2014; 271p. (In Russian) [Google Scholar]
- Gabyshev, V.A.; Tsarenko, P.M.; Ivanova, A.P. Diversity and features of the spatial structure of algal communities of water bodies and watercourses in the Lena River estuary. Inland Water Biol. 2019, 12, S1–S9. [Google Scholar] [CrossRef]
- Dodds, W.K. Trophic state, eutrophication and nutrient criteria in streams. Trends Ecol. Evol. 2007, 22, 669–676. [Google Scholar] [CrossRef] [PubMed]
- Vadeboncoeur, Y.; Moore, M.; Stewart, S.; Chandra, S.; Atkins, K.; Baron, J.; Bouma-Gregson, K.; Brothers, S.; Francoeur, S.; Genzoli, L.; et al. Blue Waters, Green Bottoms: Benthic Filamentous Algal Blooms Are an Emerging Threat to Clear Lakes Worldwide. Bioscience 2021, 71, 1011–1027. [Google Scholar] [CrossRef]
- DeNicola, D.M. Periphyton responses to temperature at different ecological levels. In Algal Ecology: Freshwater Benthic Ecosystem; Academic Press: San Diego, CA, USA, 1996; pp. 150–181. [Google Scholar]
- Vuglinsky, V.; Valatin, D. Changes in Ice Cover Duration and Maximum Ice Thickness for Rivers and Lakes in the Asian Part of Russia. Nat. Resour. 2018, 9, 73–87. [Google Scholar] [CrossRef]
- Gollerbakh, M.M.; Kosinskaya, Y.K.; Polyanskiy, V.I. Blue green algae. Identification guide to freshwater algae of the USSR; Nauka: Moscow, Russia, 1953; 650p. (In Russian) [Google Scholar]
- Starmach, K. Cyanophyta—Sinice. Glaucophyta—Glaukofity. Flora Słodkowodna Polski; PWN: Warszawa, Poland, 1966; Volume 2, 808p. (In Polish) [Google Scholar]
- Jüttner, F.; Watson, S. Biochemical and Ecological Control of Geosmin and 2-methylisoborneol in Source Waters. Appl. Environ. Microbiol. 2007, 73, 4395–4706. [Google Scholar] [CrossRef] [PubMed]
- Hitzfeld, B.C.; Lampert, C.S.; Spaeth, N.; Mountfort, D.; Kaspar, H.; Dietrich, D. Toxin production in cyanobacterial mats from ponds on the McMurdo ice shelf, Antarctica. Toxicon 2000, 38, 1731–1748. [Google Scholar] [CrossRef]
- Jungblut, A.D.; Hoeger, S.J.; Mountfort, D.; Hitzfeld, B.; Dietrich, D.; Neilan, B. Characterization of microcystin production in an Antarctic cyanobacterial mat community. Toxicon 2006, 47, 271–278. [Google Scholar] [CrossRef]
- Wood, S.; Mountfort, D.; Selwood, A.; Holland, P.; Puddick, J.; Cary, S. Widespread distribution and identification of eight novel microcystins in Antarctic cyanobacterial mats. Appl. Environ. Microbiol. 2008, 74, 7243–7251. [Google Scholar] [CrossRef]
- Kleinteich, J.; Wood, S.; Küpper, F.; Camacho, A.; Quesada, A.; Frickey, T.; Dietrich, D. Temperature-related changes in polar cyanobacterial mat diversity and toxin production. Nat. Clim. Chang. 2012, 2, 356–360. [Google Scholar] [CrossRef]
- Kleinteich, J.; Hildebrand, F.; Wood, S.; Cirés, S.; Agha, R.; Quesada, A.; Pearce, D.; Convey, P.; Küpper, F.; Dietrich, D. Diversity of toxin and non-toxin containing cyanobacterial mats of meltwater ponds on the Antarctic Peninsula: A pyrosequencing approach. Antarct. Sci. 2014, 26, 521–532. [Google Scholar] [CrossRef]
- Chrapusta, E.; Wegrzyn, M.; Zabaglo, K.; Kaminski, A.; Adamski, M.; Wietrzyk, P.; Bialczyk, J. Microcystins and anatoxin-a in Arctic biocrust cyanobacterial communities. Toxicon 2015, 101, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Gabyshev, V.A.; Sidelev, S.I.; Chernova, E.N.; Gabysheva, O.I.; Voronov, I.V.; Zhakovskaya, Z.A. Limnological characterization and first data on the occurrence of toxigenic cyanobacteria and cyanotoxins in the plankton of some lakes in the permafrost zone (Yakutia, Russia). Contemp. Probl. Ecol. 2023, 16, 89–102. [Google Scholar] [CrossRef]
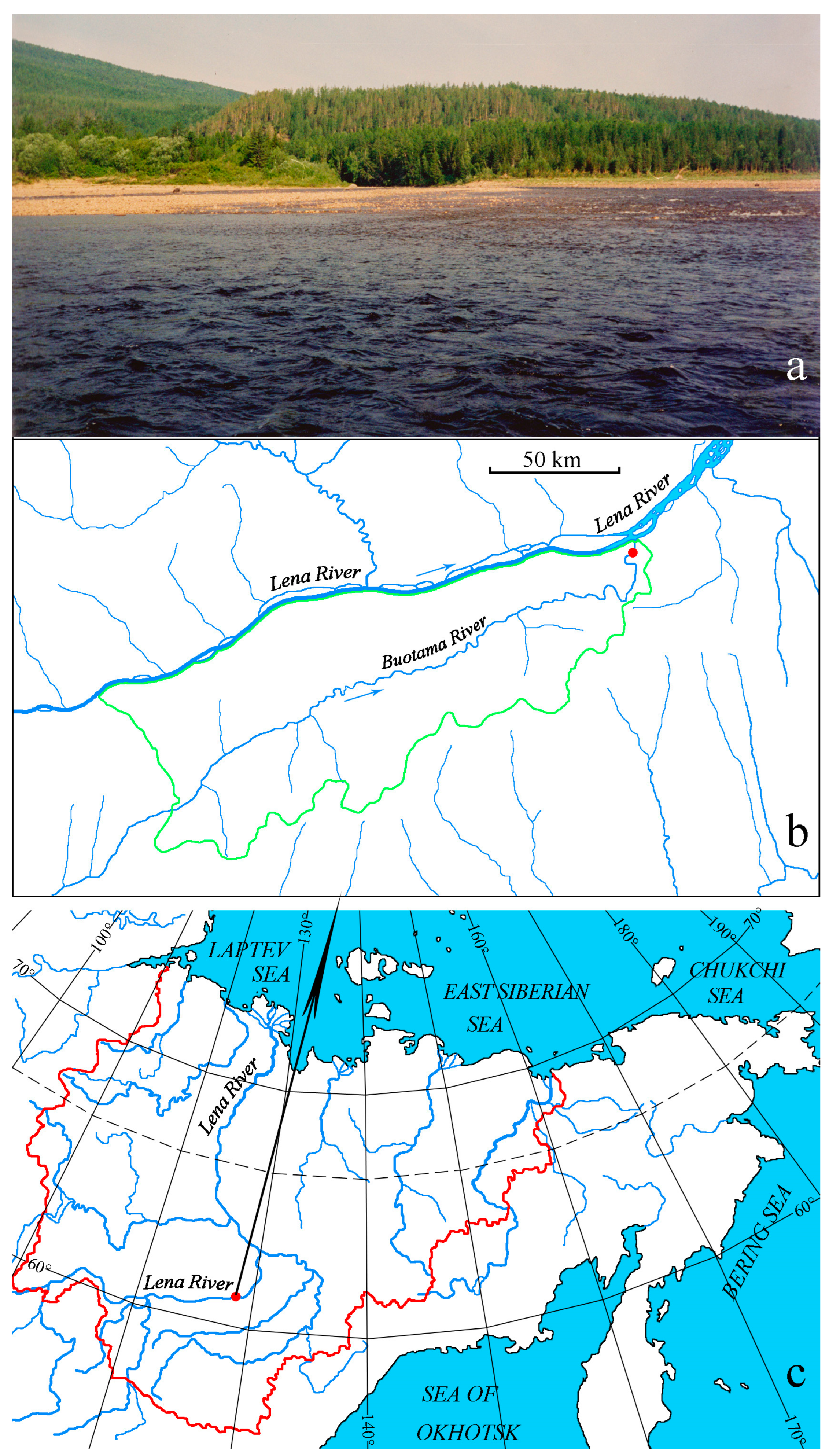

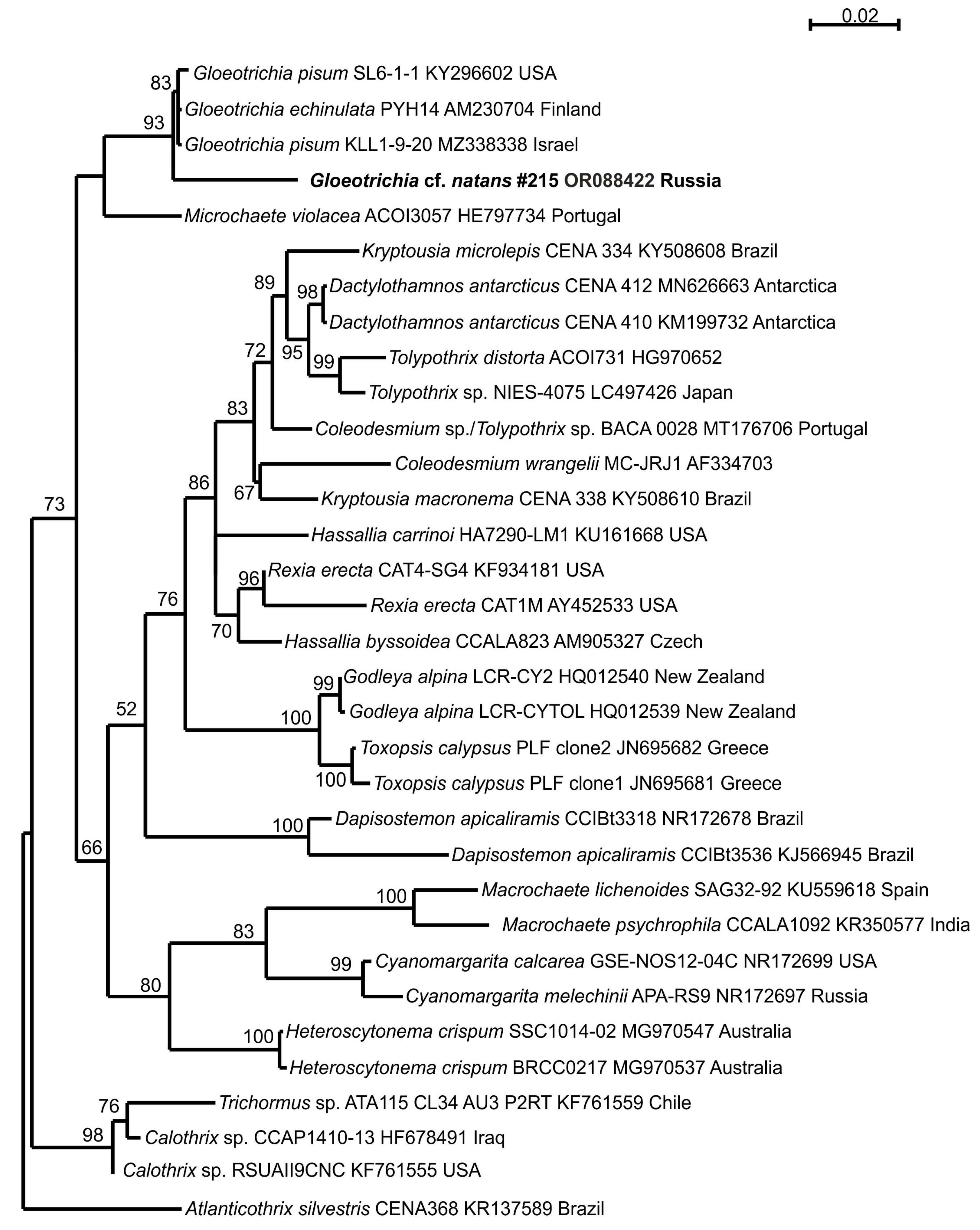
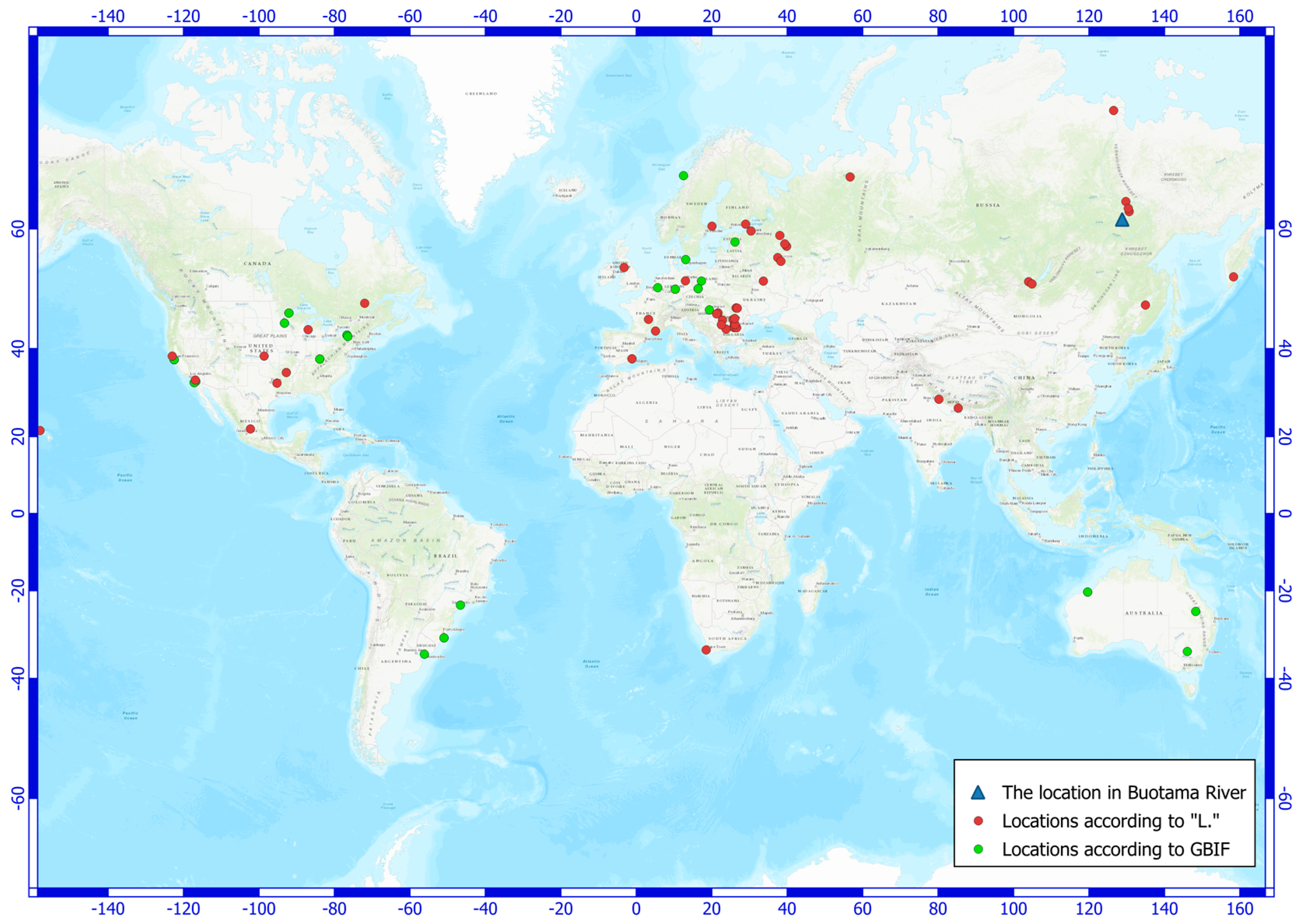
| Variable | Mean Value | Standard Deviation |
|---|---|---|
| pH | 8.47 | 0.0354 |
| TDS, mg L−1 | 185.76 | 1.8385 |
| Hardness, mmol. L−1 | 2.40 | 0.0141 |
| Ca, mg L−1 | 45.69 | 0.2687 |
| Mg, mg L−1 | 1.46 | 0.0566 |
| Na, mg L−1 | 1.19 | 0.0707 |
| K, mg L−1 | 0.58 | 0.0283 |
| HCO3, mg L−1 | 109.84 | 0.7071 |
| Cl, mg L−1 | 14.18 | 0.4243 |
| SO4, mg L−1 | 12.82 | 0.2828 |
| N-NH4, mg L-1 | 0.13 | 0.0071 |
| N-NO3, mg L−1 | 0.03 | 0.0071 |
| N-NO2, mg L−1 | 0.03 | 0.0 |
| Si-SiO2, mg L−1 | 2.04 | 0.2828 |
| P tot, mg L−1 | <0.04 | 0.0028 |
| PO4, mg L−1 | <0.02 | 0.0028 |
| Color, Pt/Co grad. | 44 | 4.2426 |
| COD, mg O L−1 | 20.20 | 2.1213 |
| Fe tot, mg L−1 | <0.05 | 0.0028 |
| Petrochemicals, mg L−1 | 0.005 | 0.0014 |
| Phenols, mg L−1 | 0.0005 | 0.0001 |
| Mn, µg L−1 | 33.00 | 5.6569 |
| Cu, µg L−1 | 6.00 | 0.7071 |
| Ni, µg L−1 | 4.40 | 0.7071 |
| Zn, µg L−1 | 13.70 | 1.1314 |
| Strain | OR088422 | MZ338338 | KY296602 |
|---|---|---|---|
| Gloeotrichia cf. natans 215 OR088422 Russia | |||
| G. pisum KLL1-9-20 MZ338338 Israel | 97.69% | ||
| G. pisum SL6-1-1 KY296602 USA | 97.54% | 99.71% | |
| G. echinulata PYH14 AM230704 Finland | 97.69% | 99.86% | 99.71% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gabyshev, V.; Davydov, D.; Vilnet, A.; Sidelev, S.; Chernova, E.; Barinova, S.; Gabysheva, O.; Zhakovskaya, Z. Gloeotrichia cf. natans (Cyanobacteria) in the Continuous Permafrost Zone of Buotama River, Lena Pillars Nature Park, in Yakutia (Russia). Water 2023, 15, 2370. https://doi.org/10.3390/w15132370
Gabyshev V, Davydov D, Vilnet A, Sidelev S, Chernova E, Barinova S, Gabysheva O, Zhakovskaya Z. Gloeotrichia cf. natans (Cyanobacteria) in the Continuous Permafrost Zone of Buotama River, Lena Pillars Nature Park, in Yakutia (Russia). Water. 2023; 15(13):2370. https://doi.org/10.3390/w15132370
Chicago/Turabian StyleGabyshev, Viktor, Denis Davydov, Anna Vilnet, Sergey Sidelev, Ekaterina Chernova, Sophia Barinova, Olga Gabysheva, and Zoya Zhakovskaya. 2023. "Gloeotrichia cf. natans (Cyanobacteria) in the Continuous Permafrost Zone of Buotama River, Lena Pillars Nature Park, in Yakutia (Russia)" Water 15, no. 13: 2370. https://doi.org/10.3390/w15132370
APA StyleGabyshev, V., Davydov, D., Vilnet, A., Sidelev, S., Chernova, E., Barinova, S., Gabysheva, O., & Zhakovskaya, Z. (2023). Gloeotrichia cf. natans (Cyanobacteria) in the Continuous Permafrost Zone of Buotama River, Lena Pillars Nature Park, in Yakutia (Russia). Water, 15(13), 2370. https://doi.org/10.3390/w15132370








