Efficiency of Orange Yellow II Degradation by Synergistic Hydroxylamine with Fe2+ to Activate Peroxymonosulfate Oxidation: Machine Learning Prediction and Performance Optimization
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents and Instruments
2.2. Experimental Methods
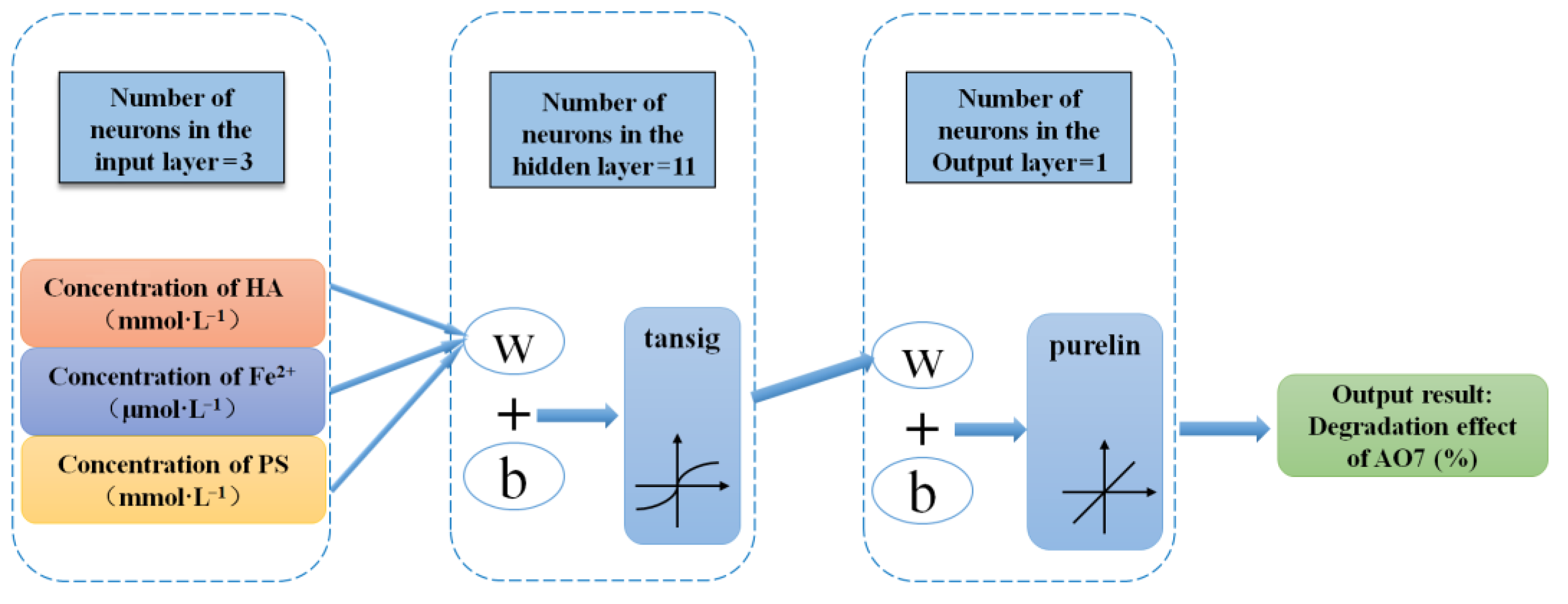
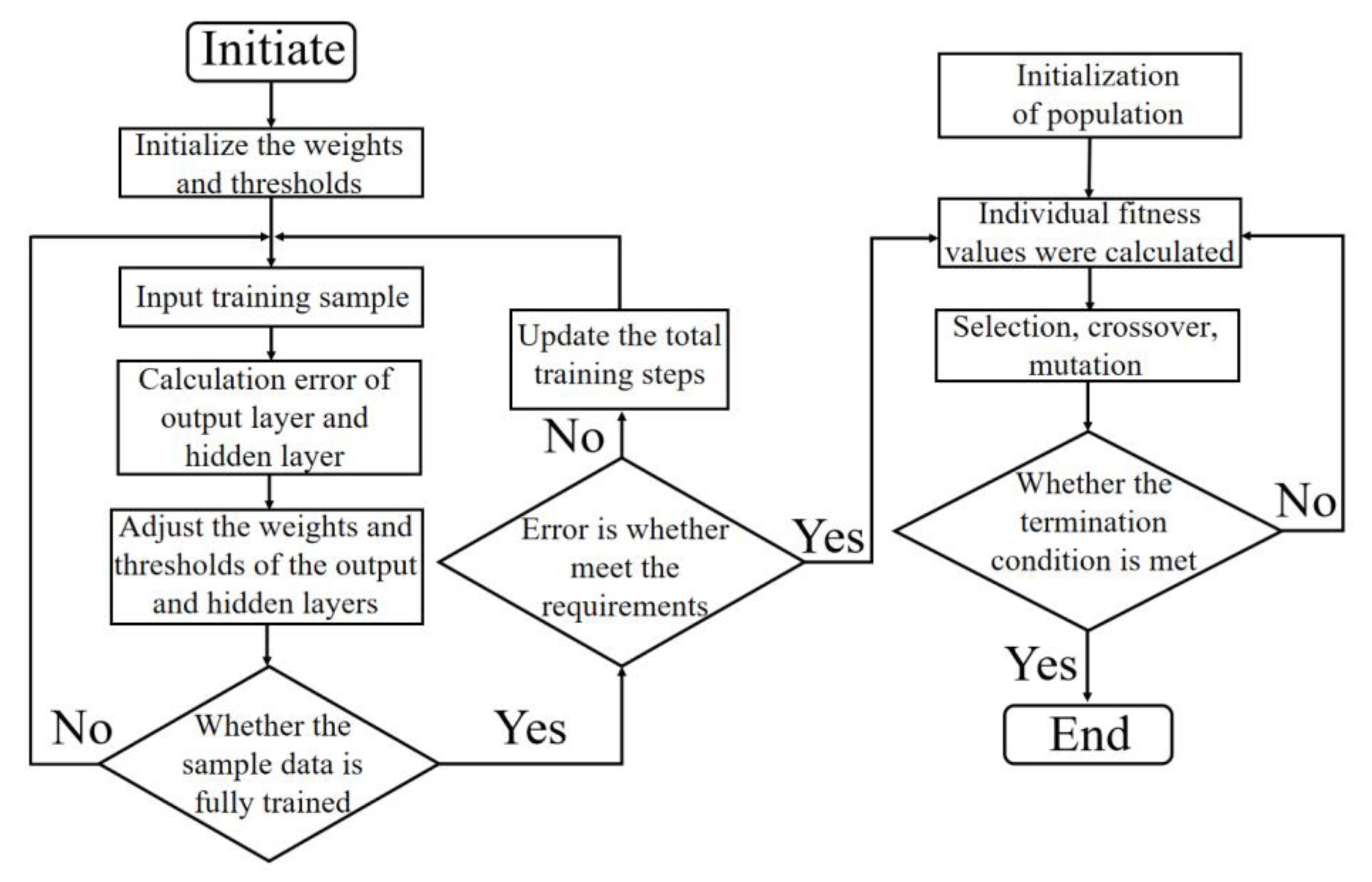
2.3. BP Neural Network Model
2.4. Garson and PaD2 Algorithms
3. Results and Discussion
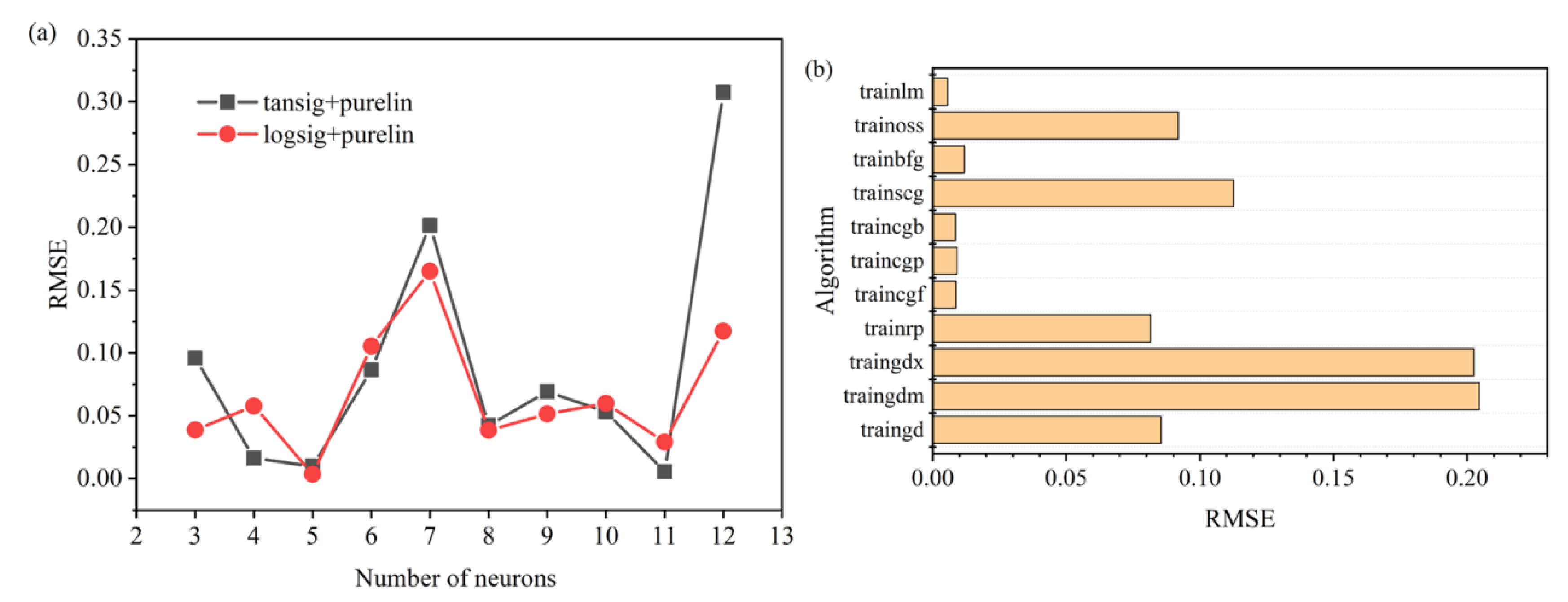
3.1. Determination of the BPNN Structure
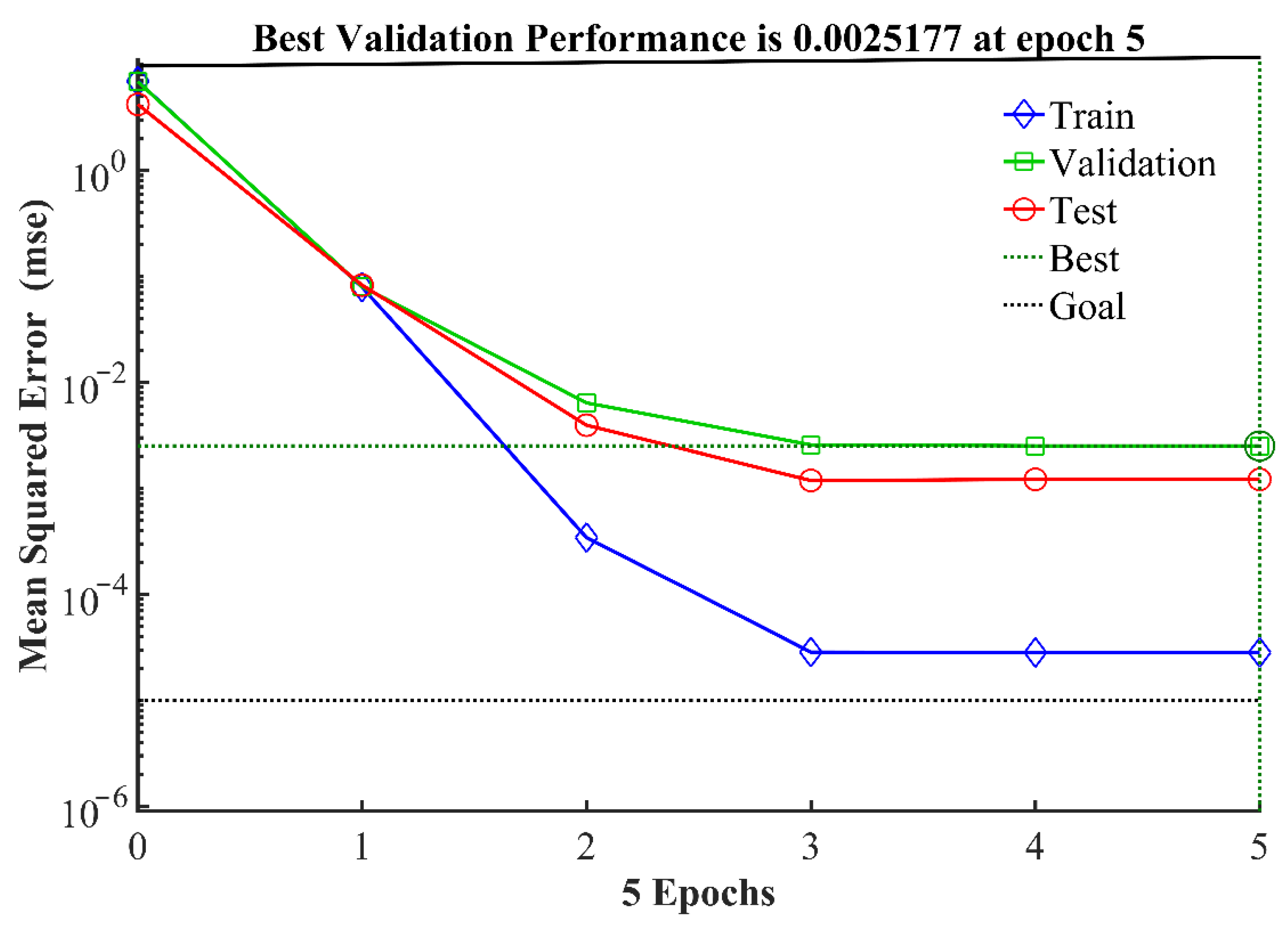
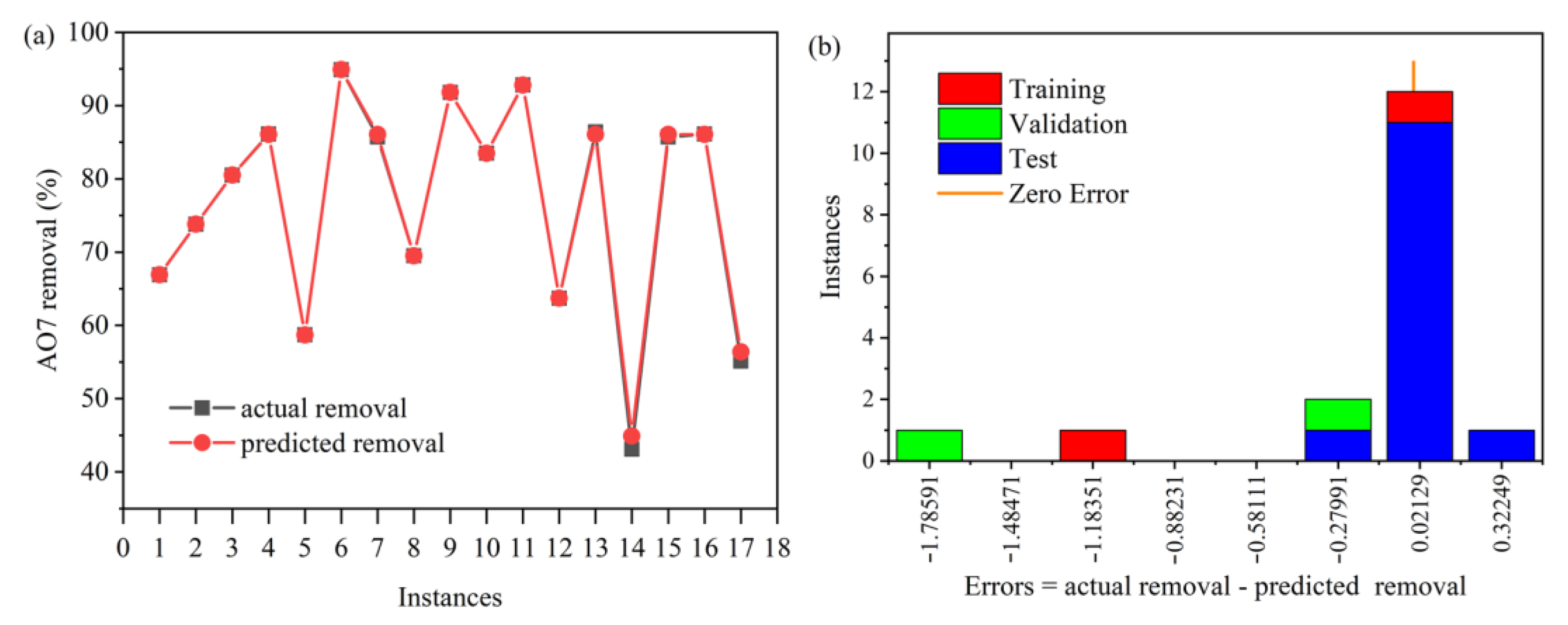
3.2. Performance Evaluation of the BPNN
3.3. Sensitivity Analysis of the BPNN
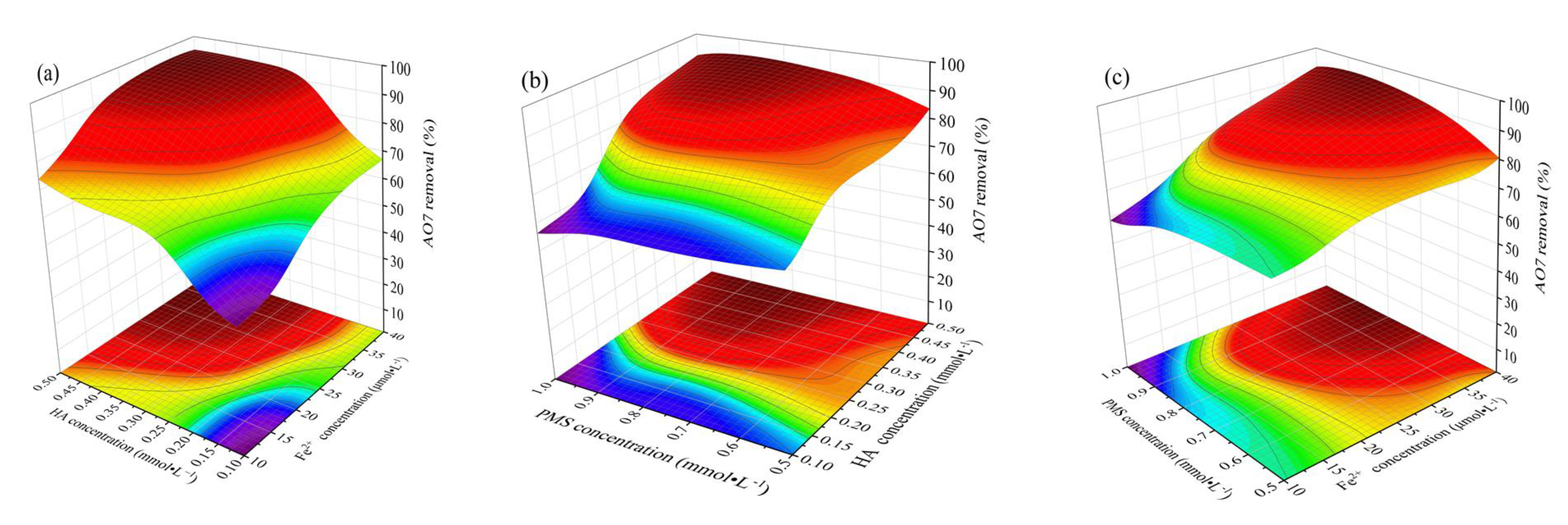
3.4. Influence of Concentrations of Fe2+, HA, and PMS on Degradation of AO7
3.5. Optimization of Process Parameters
4. Conclusions
- (1)
- The final BPNN topology was 3-11-1. The excitation functions used in the hidden and output layers were tansig and purelin, respectively, and the training function was trainlm. The R2 of the established BPNN model was 0.99852, and the data were distributed near the line y = x. The results show that the predicted value based on the BP neural network model was in good agreement with the measured value, and that there was a good fit of the model for the process of synergistic hydroxylamine with Fe2+ to activate PMS.
- (2)
- Using the Garson and PaD2 algorithms based on the neural network weights, the order of influence of factors and factor pairs on the degradation of AO7 was calculated as follows: concentration of HA > Fe2+ > PMS, and concentrations of Fe2+ and PMS > concentrations of HA and PMS > concentrations of Fe2+ and HA.
- (3)
- The optimization result obtained by the genetic algorithm was as follows: the concentration of Fe2+ was 35.33 μmol·L−1, HA was 0.46 mmol·L−1, and PMS was 0.93 mmol·L−1. According to the verification experiment, the degradation of AO7 was 95.7%, which was only 0.5% lower than the model’s predicted value, 96.2%.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Qu, S.; Wang, W.; Pan, X.; Li, C. Improving the Fenton catalytic performance of FeOCl using an electron mediator. J. Hazard. Mater. 2020, 384, 121494. [Google Scholar] [CrossRef] [PubMed]
- Qin, Q.D.; Qiao, N.; Liu, Y.H.; Wu, X. Spongelike porous CuO as an efficient peroxymonosulfate activator for degradation of Acid Orange 7. Appl. Surf. Sci. 2020, 521, 1464792020. [Google Scholar] [CrossRef]
- Al-Musawi, T.J.; Rajiv, P.; Mengelizadeh, N.; Mohammed, I.A.; Balarak, D. Development of sonophotocatalytic process for degradation of acid orange 7 dye by using titanium dioxide nanoparticles/graphene oxide nanocomposite as a catalyst. J. Environ. Manag. 2021, 292, 112777. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.H.; Li, S.; Ding, H.J.; Zhu, Y.H.; Wang, X.X.; Liu, H.F.; Zhang, Q.; Zhao, C. Electrochemical/Fe3+/peroxymonosulfate system for the degradation of Acid Orange 7 adsorbed on activated carbon fiber cathode. Chemosphere 2020, 241, 125125. [Google Scholar] [CrossRef]
- Zhu, K.M.; Wang, X.S.; Chen, D.; Ren, W.; Lin, H.; Zhang, H. Wood-based biochar as an excellent activator of peroxydisulfate for Acid Orange 7 decolorization. Chemosphere 2019, 231, 32–40. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.Z.; Yi, P.; Wang, X.; Gao, H.; Zhang, H. Degradation of Acid Orange 7 by an ultrasound/ZnO-GAC/persulfate process. Sep. Purif. Techno. 2018, 194, 181–187. [Google Scholar] [CrossRef]
- Zhu, S.R.; Gao, N.Y.; Lu, X.; Guo, Y.L. Degradation of bisphenol A in aqueous solution by thermally activated sulfate oxidation. Chin. Environ. Sci. 2017, 37, 188–194. [Google Scholar]
- Rahmani, A.; Salari, M.; Tari, K.; Shabanloo, A.; Shabanloo, N.; Bajalan, S. Enhanced degradation of furfural by heat-activated persulfate/nZVI-rGO oxidation system: Degradation pathway and improving the biodegradability of oil refinery wastewater. J. Environ. Chem. Eng. 2020, 8, 104468. [Google Scholar] [CrossRef]
- Li, Z.Y.; Wang, L.; Liu, Y.L.; He, P.N.; Ma, J. Overlooked enhancement of chloride ion on the transformation of reactive species in peroxymonosulfate/Fe(II)/NH2OH System. Water Res. 2021, 195, 116973. [Google Scholar] [CrossRef]
- Dhaka, S.; Kumar, R.; Khan, M.A.; Paeng, K.J.; Kurade, M.B.; Kim, S.J.; Jeon, B.H. Aqueous phase degradation of methyl paraben using UV-activated persulfate method. Chem. Eng. J. 2017, 321, 11–19. [Google Scholar] [CrossRef]
- Wang, H.B.; Wang, Q.; Liu, Y.Q.; Fu, Y.S.; Wu, P. Degradation of diclofenac by ferrous activated persulfate. Environ. Chem. 2020, 39, 869–875. [Google Scholar]
- Rahmani, A.; Shabanloo, A.; Shabanloo, N. A mini-review of recent progress in lead dioxide electrocatalyst for degradation of toxic organic pollutants. Mater. Today Chem. 2023, 27, 101311. [Google Scholar] [CrossRef]
- Rahmani, A.; Shabanloo, A.; Shabanloo, N.; Torkshavand, Z.; Dargahi, A.; Ansari, A. The integration of PbO2-based EAOPs with other advanced oxidation processes for improved treatment of water and wastewater. Curr. Opin. Electrochem. 2023, 37, 101204. [Google Scholar] [CrossRef]
- Ghanbari, F.; Moradi, M. Application of peroxymonosulfate and its activation methods for degradation of environmental organic pollutants: Review. Chem. Eng. J. 2016, 310, 307–315. [Google Scholar] [CrossRef]
- Wang, W.; Zhang, H.; Chen, Y.; Shi, H. Efficient Degradation of Tetracycline via Coupling of Photocatalysis and Photo-Fenton Processes over a 2D/2D α-Fe2O3/g-C3N4 S-Scheme Heterojunction Catalyst. Acta Phys.-Chim. Sin. 2022, 38, 2201008. [Google Scholar] [CrossRef]
- Su, C.; Li, R.; Li, C.; Wang, W. Piezo-promoted regeneration of Fe2+ boosts peroxydisulfate activation by Bi2Fe4O9 nanosheets. Appl. Catal. B-Environ. 2022, 310, 121330. [Google Scholar] [CrossRef]
- Li, C.W.; Chi, K.Y.; Yang, W.W.; Li, S.; Du, J. Remediation effect of alkali-activate persulfate in groundwater of a chlorinated hydrocarbon contaminated site. Chin. J. Environ. Eng. 2021, 15, 1916–1926. [Google Scholar]
- Li, X.; Feng, S.; Yang, J.; Xie, T.; Wang, J.; Chen, X.; Kong, D.; Chen, H. Tetracycline removal by a magnetic heterojunction Cu2O/CoFe2O4 activating peroxymonosulfate. Rare Metals 2023, 42, 862–874. [Google Scholar] [CrossRef]
- Xu, W.X.; Wu, J.F.; Xing, J.M.; Gong, Q.; Nie, H.; Wang, S.L. Degradation of acetaminophen by hydroxylamine enhanced Fe2+/persulfate system. Acta Sci. Circumstantiae 2019, 39, 3410–3417. [Google Scholar]
- Zhang, K.; Zhang, M.; Zhou, R.J.; Zhou, T. Hydroxylamine-enhanced Fe(II)-peroxymonosulfate activation for efficient degradation of organic pollutants: Optimization by response surface methodology. Water Sci. Technol. 2022, 86, 834–846. [Google Scholar] [CrossRef]
- Zhang, M.; Zhang, K.; Zhou, R.J.; Wang, J.P. Hydroxylamine enhanced activation of peroxymonosulfate by Fe(III)/Cu(II) bimetallic for high-efficiency degradation of AO7. Water Sci. Tech. 2022, 85, 2038–2050. [Google Scholar] [CrossRef]
- Luna, M.D.G.; Sablas, M.M.; Hung, C.M.; Chen, C.W.; Garcia-Segura, S.; Dong, C.D. Modeling and optimization of imidacloprid degradation by catalytic percarbonate oxidation using artificial neural network and Box-Behnken experimental design. Chemosphere 2020, 251, 126254. [Google Scholar] [CrossRef] [PubMed]
- Smaali, A.; Berkani, M.; Merouane, F.; Le, V.T.; Vasseghian, Y.; Rahim, N.; Kouachi, M. Photocatalytic-persulfate- oxidation for diclofenac removal from aqueous solutions: Modeling, optimization and biotoxicity test assessment. Chemosphere 2020, 266, 129158. [Google Scholar] [CrossRef] [PubMed]
- Soleymani, A.R.; Moradi, M. Performance and modeling of UV/Persulfate/Ce (IV) process as a dual oxidant photochemical treatment system: Kinetic study and operating cost estimation. Chem. Eng. J. 2018, 347, 243–251. [Google Scholar] [CrossRef]
- Almuntashiri, A.; Hosseinzadeh, A.; Volpin, F.; Ali, S.M.; Dorji, U.; Shon, H.; Phuntsho, S. Removal of pharmaceuticals from nitrified urine. Chemosphere 2021, 280, 130870. [Google Scholar] [CrossRef]
- Estahbanati, M.; Feilizadeh, M.; Iliuta, M.C. Photocatalytic valorization of glycerol to hydrogen: Optimization of operating parameters by artificial neural network. Appl. Catal. B-Environ. 2017, 209, 483–492. [Google Scholar] [CrossRef]
- Chen, J.Y.; Feng, J.W.; Lu, S.S.; Shen, Z.J.; Du, Y.L.; Peng, L.; Nian, P.; Yuan, S.J.; Zhang, A.Y. Non-thermal plasma and Fe2+ activated persulfate ignited degradation of aqueous crystal violet: Degradation mechanism and artificial neural network modeling. Sep. Purif. Technol. 2018, 191, 75–85. [Google Scholar] [CrossRef]
- Mohammadi, F.; Samaei, M.R.; Azhdarpoor, A.; Teiri, H.; Badeenezhad, A.; Rostami, S. Modelling and Optimizing Pyrene Removal from the Soil by Phytoremediation using Response Surface Methodology, Artificial Neural Networks, and Genetic Algorithm. Chemosphere 2019, 237, 124486. [Google Scholar] [CrossRef]
- Yu, M.Z.; Yuan, X.; Li, S.Y.; Hu, J.T.; Li, S.Y.; Zhao, Y.C. Laboratory and simulation study on the adsorption of caffeine onto river sediments and the influencing factors. Acta Sci. Circumstantiae 2018, 38, 560–569. [Google Scholar]
- Cai, Y.; Xing, Y.; Hu, D. On Sensitivity Aaalysis. J. Beijing Norm. Univ. (Nat. Sci.) 2008, 44, 9–16. [Google Scholar]
- Khan, S.U.; Khan, H.; Anwar, S.; Khan, S.; Zanoni, M.V.B.; Hussain, S. Computational and statistical modeling for parameters optimization of electrochemical decontamination of synozol red dye wastewater. Chemosphere 2020, 253, 126673. [Google Scholar] [CrossRef] [PubMed]
- Dong, H.Y.; Fan, S.; Jin, C.J.; Gao, M.C.; She, Z.L.; Zhao, Y.G. Degradation of microcystin-IR by electrochemically activated sulfate: Variable evaluation and model construction using artificial neural network. Environ. Chem. 2020, 39, 3390–3401. [Google Scholar]
- Liu, G.F.; Li, X.C.; Han, B.J.; Chen, L.W.; Zhu, L.N.; Campos, L.C. Efficient degradation of sulfamethoxazole by the Fe(II)/HSO5- process enhanced by hydroxylamine: Efficiency and mechanism. J. Hazard. Mater. 2017, 322, 461–468. [Google Scholar] [CrossRef] [PubMed]
- Qi, C.D.; Liu, X.T.; Ma, J.; Lin, C.Y.; Li, X.W.; Zhang, H.J. Activation of peroxymonosulfate by base: Implications for the degradation of organic pollutants. Chemosphere 2016, 151, 280–288. [Google Scholar] [CrossRef] [PubMed]
- Rastogi, A.; Al-Abed, S.R.; Dionysiou, D.D. Sulfate radical-based ferrous-peroxymonosulfate oxidative system for PCBs degradation in aqueous and sediment systems. Appl. Catal. B-Environ. 2009, 85, 171–179. [Google Scholar] [CrossRef]
- Ding, Y.M.; Zhang, W.L.; Yu, L.; Lu, K.H. The accuracy and efficiency of GA and PSO optimization schemes on estimating reaction kinetic parameters of biomass pyrolysis. Energy 2019, 176, 582–588. [Google Scholar] [CrossRef]
- Jin, J.L.; Wei, Y.M.; Ding, J. Projection pursuit model for comprehensive evaluation of water quality. Acta Sci. Circumstantiae 2001, 21, 431–434. [Google Scholar]

| Factors | Levels | ||
|---|---|---|---|
| −1 | 0 | +1 | |
| Concentration of Fe2+ (μmol·L−1) | 10 | 25 | 40 |
| Concentration of HA (mmol·L−1) | 0.1 | 0.3 | 0.5 |
| Concentration of PMS (mmol·L−1) | 0.5 | 0.75 | 1 |
| Runs | Fe2+ (μmol·L−1) | HA (mmol·L−1) | PMS (mmol·L−1) | RAO7 (%) | |
|---|---|---|---|---|---|
| Actual | Predicted | ||||
| 1 | 40 | 0.1 | 0.75 | 66.9 | 66.9 |
| 2 | 10 | 0.5 | 0.75 | 73.8 | 73.8 |
| 3 | 25 | 0.3 | 0.75 | 86.1 | 86.1 |
| 4 | 25 | 0.3 | 0.75 | 86.1 | 86.1 |
| 5 | 10 | 0.3 | 1 | 58.7 | 58.7 |
| 6 | 40 | 0.5 | 0.75 | 94.9 | 94.9 |
| 7 | 25 | 0.3 | 0.75 | 85.7 | 86.1 |
| 8 | 10 | 0.3 | 0.5 | 69.5 | 69.5 |
| 9 | 25 | 0.5 | 1 | 91.8 | 91.8 |
| 10 | 25 | 0.5 | 0.5 | 83.5 | 83.5 |
| 11 | 40 | 0.3 | 1 | 92.8 | 92.8 |
| 12 | 25 | 0.1 | 0.5 | 63.7 | 63.7 |
| 13 | 25 | 0.3 | 0.75 | 86.4 | 86.1 |
| 14 | 10 | 0.1 | 0.75 | 43.1 | 44.9 |
| 15 | 25 | 0.3 | 0.75 | 85.7 | 86.1 |
| 16 | 40 | 0.3 | 0.5 | 80.5 | 80.5 |
| 17 | 25 | 0.1 | 1 | 55.1 | 56.4 |
| Hidden Layer Neuron | Weight between Input and Hidden Layers | Threshold of Hidden Layer | Weight between Hidden and Output Layers | Threshold of Output Layer | ||
|---|---|---|---|---|---|---|
| Fe2+ | HA | PMS | ||||
| 1 | 2.8783 | −0.7327 | −1.1964 | −3.0276 | −0.1574 | −0.2901 |
| 2 | −2.1334 | −2.3543 | −0.2061 | 2.3871 | −0.0495 | |
| 3 | −1.8180 | 2.3485 | −0.3131 | 2.0163 | −0.1829 | |
| 4 | −0.1180 | 2.9386 | −0.7082 | 1.4494 | 0.5138 | |
| 5 | −2.9141 | −0.9853 | 0.4082 | 0.6374 | −0.0003 | |
| 6 | 1.6788 | 1.6654 | 2.0006 | 0.1730 | 0.2882 | |
| 7 | −1.1376 | −2.5627 | −1.2200 | −0.8487 | 0.0315 | |
| 8 | 3.0577 | 0.2525 | −0.3583 | 1.2881 | 0.2380 | |
| 9 | −0.7018 | 2.0271 | −2.2731 | −1.8713 | −0.0537 | |
| 10 | 1.0895 | 0.7200 | −2.6689 | 2.7863 | 0.3428 | |
| 11 | −2.4593 | −1.3092 | −1.6007 | −2.9865 | 0.0662 | |
| No. | Optimized Conditions | Predicted Degradation Rate | Actual Degradation Rate | Mean; Error |
|---|---|---|---|---|
| 1 | Fe2+: 35.33 μmol·L−1, HA: 0.46 mmol·L−1, PMS: 0.93 mmol·L−1 | 96.2% | 96.0% | 95.7%; −0.5% |
| 2 | 95.4% | |||
| 3 | 95.6% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, R.; Zhang, K.; Zhang, M. Efficiency of Orange Yellow II Degradation by Synergistic Hydroxylamine with Fe2+ to Activate Peroxymonosulfate Oxidation: Machine Learning Prediction and Performance Optimization. Water 2023, 15, 1931. https://doi.org/10.3390/w15101931
Zhou R, Zhang K, Zhang M. Efficiency of Orange Yellow II Degradation by Synergistic Hydroxylamine with Fe2+ to Activate Peroxymonosulfate Oxidation: Machine Learning Prediction and Performance Optimization. Water. 2023; 15(10):1931. https://doi.org/10.3390/w15101931
Chicago/Turabian StyleZhou, Runjuan, Kuo Zhang, and Ming Zhang. 2023. "Efficiency of Orange Yellow II Degradation by Synergistic Hydroxylamine with Fe2+ to Activate Peroxymonosulfate Oxidation: Machine Learning Prediction and Performance Optimization" Water 15, no. 10: 1931. https://doi.org/10.3390/w15101931





