BioH2 Production Using Microalgae: Highlights on Recent Advancements from a Bibliometric Analysis
Abstract
:1. Introduction
- What trends can be detected when analyzing literature investigating bioH2 production from microalgae?
- Who are the major contributors to this research area?
- What are the recent advancements and research gaps?
2. Background
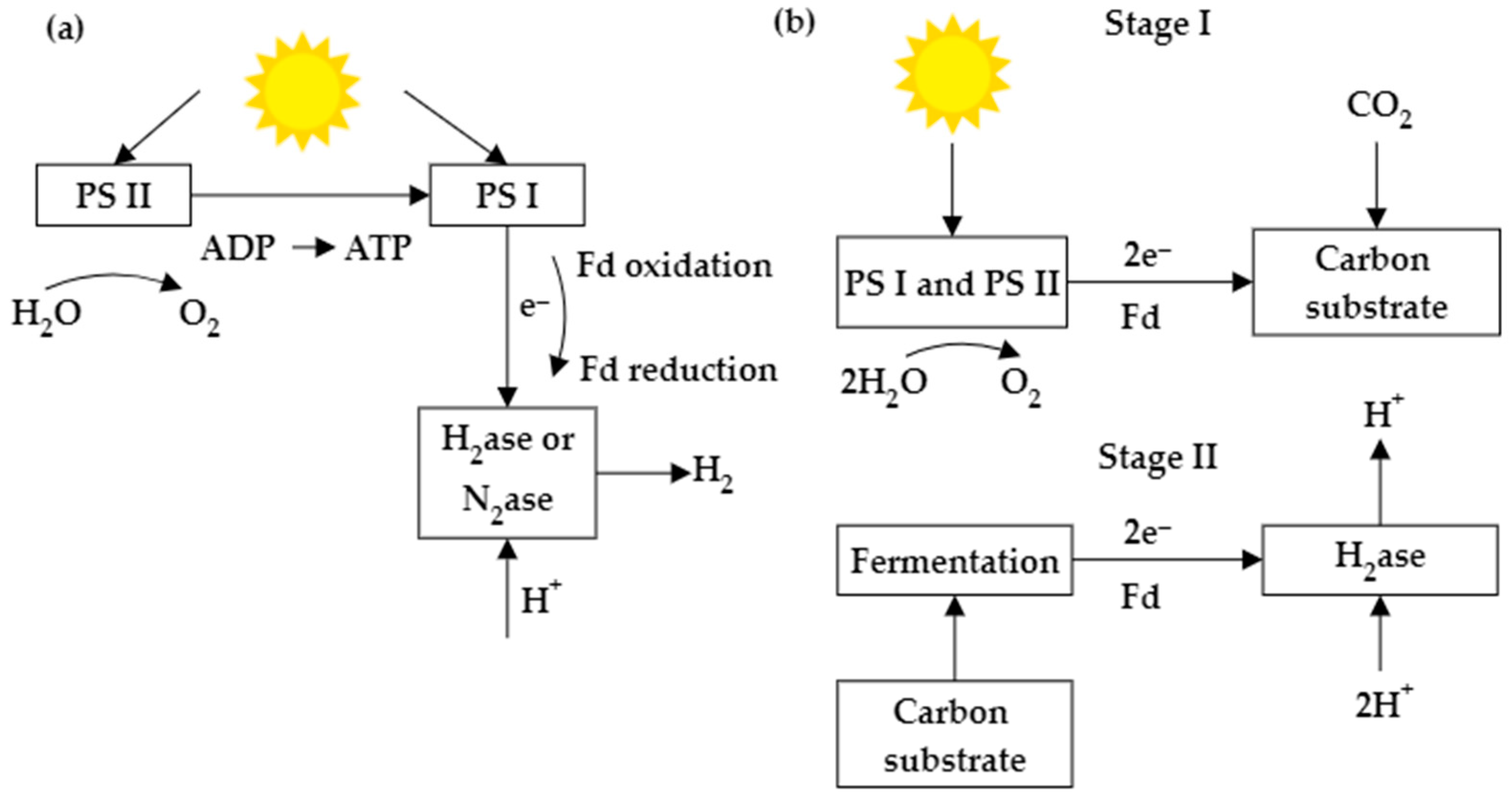
2.1. The Techniques to Produce BioH2 Using Microalgae
2.2. Factors Affecting BioH2 Production Using Microalgae
2.3. Strategies to Improve the BioH2 Production from Microalgae
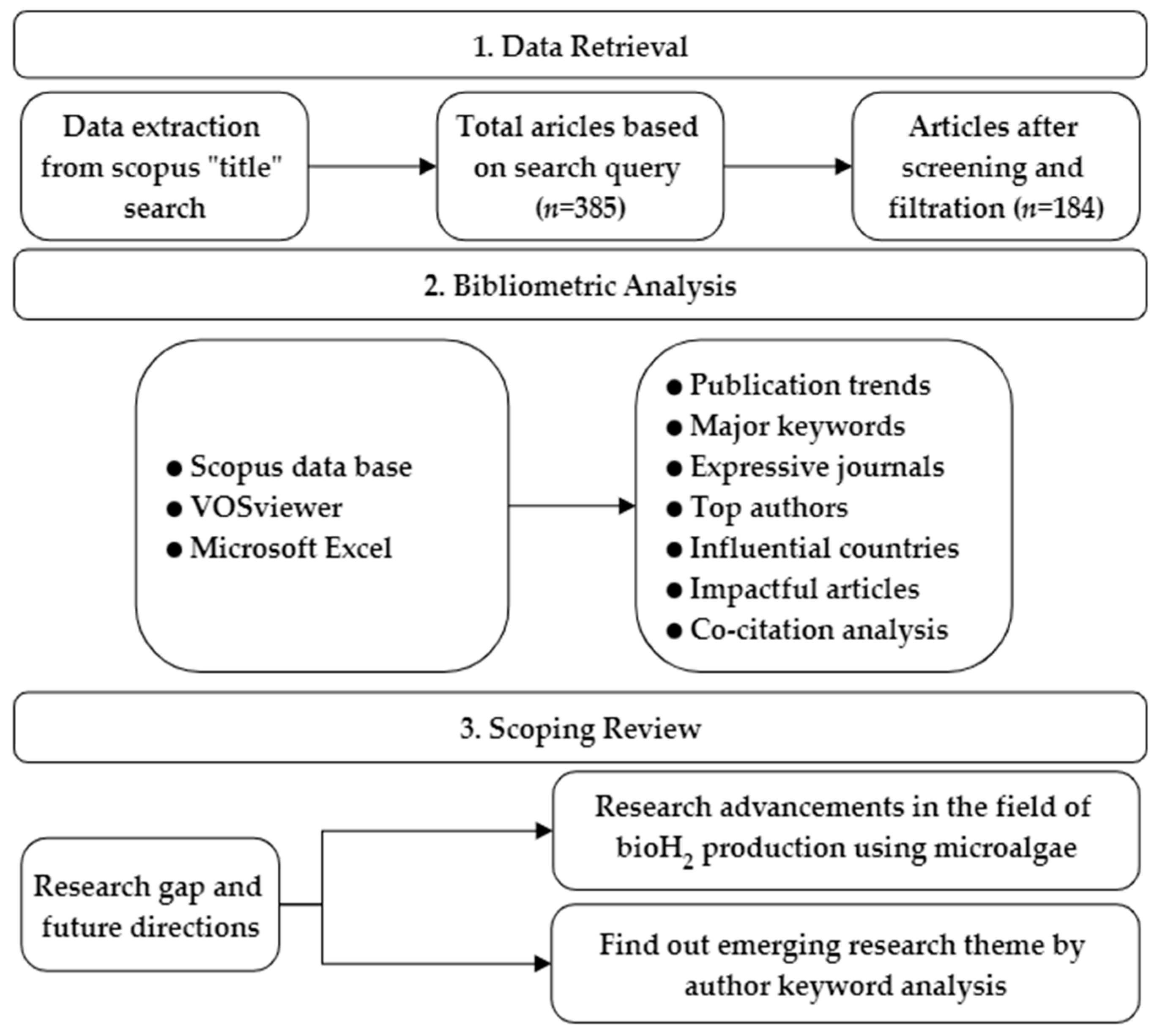
3. Methodology
3.1. Data Collection
3.2. Software Selection
3.3. Data Analysis
4. Results
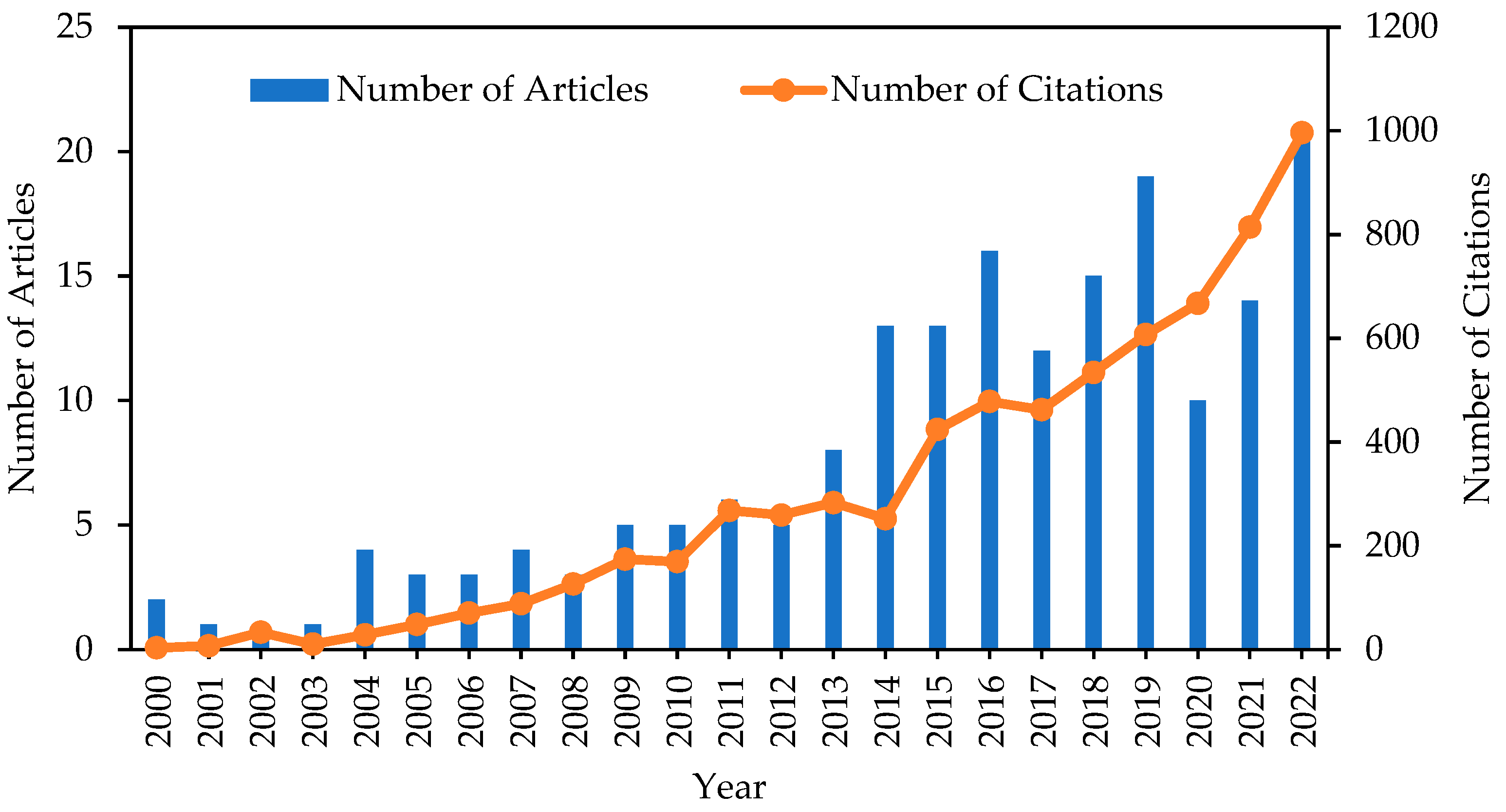
4.1. Publication and Citations Trends
4.2. Keyword Analysis
4.2.1. Most Used Keywords
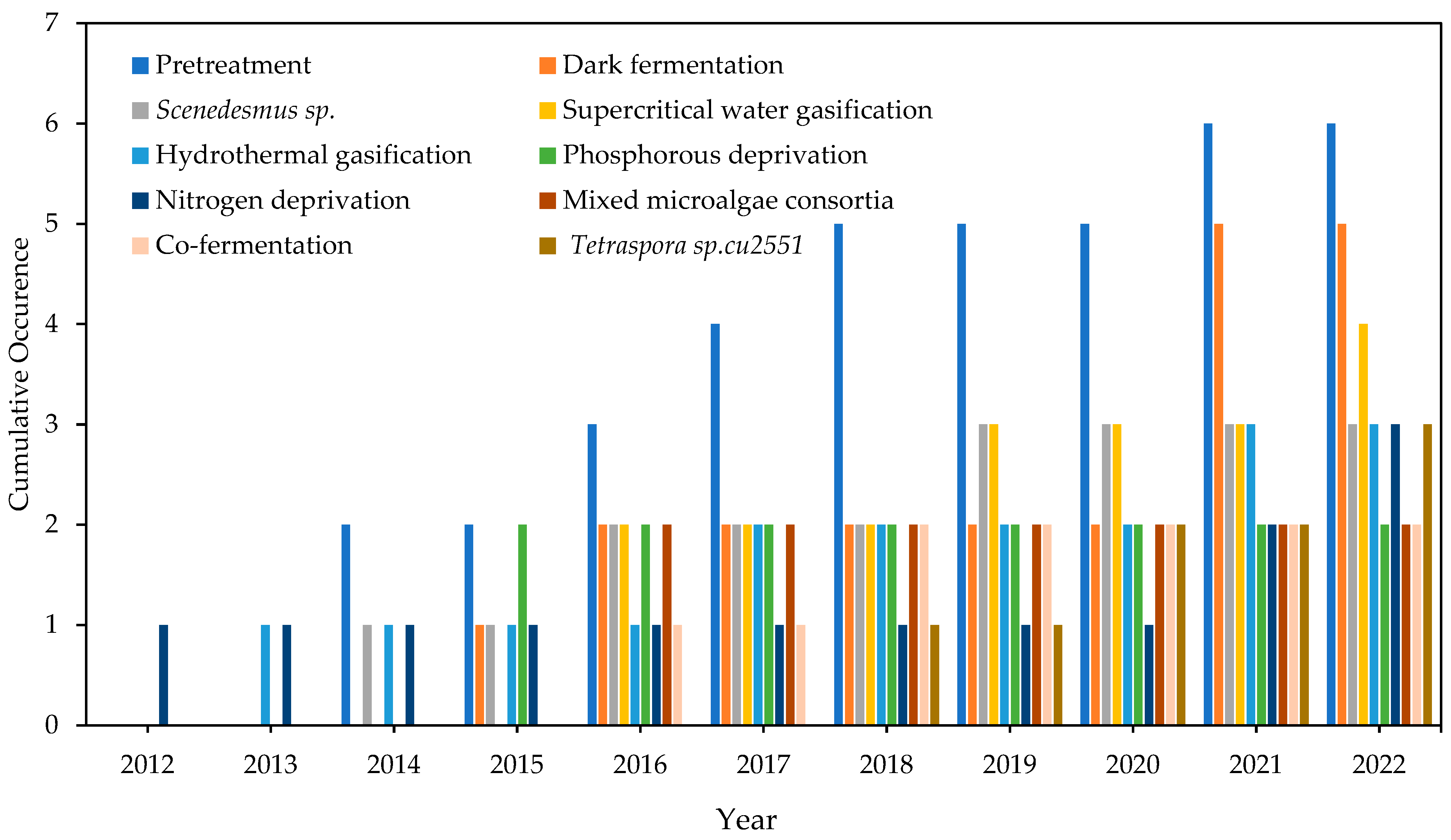
4.2.2. Emerging Keywords
4.3. Major Contributors to the Field
4.4. Nature of Collaboration
4.5. Co-Citation Analysis
5. Discussion of Recent Developments
- Designing customized photobioreactors for investigating direct biophotolysis.
- Developing techniques to prevent the inhibition of biophotolysis by limiting high light intensity and O2 formation.
- Assessing the possibility of limiting the amount of accumulated biomass during the growth phase and improving light transformation efficiency in indirect biophotolysis.
- Investigating alternative methods to improve substrate transformation efficiency and H2–CO2 mixture separation and to control O2 accumulation during dark fermentation.
- Improving H2 conversion efficiency, light transformation efficiency, and control inhomogeneity in the light distribution in photo fermentation.
- Exploring different strains of microalgae for higher bioH2 yield.
6. Conclusions and Future Directions
- More investigations on identifying genetic strategies to reduce the O2 sensitivity of the H2ase enzyme.
- Investigations to find different co-culture methods, such as algae-bacteria consortium for a continuous bioH2 yield.
- Emphasis on reducing the operation cost and realistic pilot studies for scaling up the process.
- Investigations to find effective pretreatment combinations.
- Studying the potential of nanoparticles to enhance bioH2 yield.
- Focusing on Life Cycle Assessments to scale up microalgal bioH2 production.
- Exploring the possibilities to incorporate genetic engineering.
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ampese, L.C.; Sganzerla, W.G.; Di Domenico Ziero, H.; Mudhoo, A.; Martins, G.; Forster-Carneiro, T. Research Progress, Trends, and Updates on Anaerobic Digestion Technology: A Bibliometric Analysis. J. Clean. Prod. 2022, 331, 130004. [Google Scholar] [CrossRef]
- Xia, A.; Jacob, A.; Tabassum, M.R.; Herrmann, C.; Murphy, J.D. Production of Hydrogen, Ethanol and Volatile Fatty Acids through Co-Fermentation of Macro- and Micro-Algae. Bioresour. Technol. 2016, 205, 118–125. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Xu, J.; Xue, H.; Li, C.; Liu, H.; Gu, S.; Miao, H.; Ruan, W. Improving Hydrogen Recovery from Anaerobic Co-Digestion of Algae and Food Waste by High-Pressure Homogenisation Pre-Treatment. Environ. Chem. Lett. 2021, 19, 3497–3504. [Google Scholar] [CrossRef]
- Srirangan, K.; Pyne, M.E.; Perry Chou, C. Biochemical and Genetic Engineering Strategies to Enhance Hydrogen Production in Photosynthetic Algae and Cyanobacteria. Bioresour. Technol. 2011, 102, 8589–8604. [Google Scholar] [CrossRef] [PubMed]
- Vanyan, L.; Cenian, A.; Trchounian, K. Biogas and Biohydrogen Production Using Spent Coffee Grounds and Alcohol Production Waste. Energies 2022, 15, 5935. [Google Scholar] [CrossRef]
- Jensen, L.S.; Kaul, C.; Juncker, N.B.; Thomsen, M.H.; Chaturvedi, T. Biohydrogen Production in Microbial Electrolysis Cells Utilizing Organic Residue Feedstock: A Review. Energies 2022, 15, 8396. [Google Scholar] [CrossRef]
- Tamburic, B.; Zemichael, F.W.; Maitland, G.C.; Hellgardt, K. Parameters Affecting the Growth and Hydrogen Production of the Green Alga Chlamydomonas reinhardtii. Int. J. Hydrog. Energy 2011, 36, 7872–7876. [Google Scholar] [CrossRef]
- Gholkar, P.; Shastri, Y.; Tanksale, A. Renewable Hydrogen and Methane Production from Microalgae: A Techno-Economic and Life Cycle Assessment Study. J. Clean. Prod. 2021, 279, 123726. [Google Scholar] [CrossRef]
- Aziz, M. Integrated Hydrogen Production and Power Generation from Microalgae. Int. J. Hydrog. Energy 2016, 41, 104–112. [Google Scholar] [CrossRef]
- Shobana, S.; Saratale, G.D.; Pugazhendhi, A.; Arvindnarayan, S.; Periyasamy, S.; Kumar, G.; Kim, S.-H. Fermentative Hydrogen Production from Mixed and Pure Microalgae Biomass: Key Challenges and Possible Opportunities. Int. J. Hydrog. Energy 2017, 42, 26440–26453. [Google Scholar] [CrossRef]
- Raheem, A.; Ji, G.; Memon, A.; Sivasangar, S.; Wang, W.; Zhao, M.; Taufiq-Yap, Y.H. Catalytic Gasification of Algal Biomass for Hydrogen-Rich Gas Production: Parametric Optimization via Central Composite Design. Energy Convers. Manag. 2018, 158, 235–245. [Google Scholar] [CrossRef]
- Maswanna, T.; Lindblad, P.; Maneeruttanarungroj, C. Improved Biohydrogen Production by Immobilized Cells of the Green Alga Tetraspora sp., CU2551 Incubated under Aerobic Conditions. J. Appl. Phycol. 2020, 32, 2937–2945. [Google Scholar] [CrossRef]
- Maswanna, T.; Phunpruch, S.; Lindblad, P.; Maneeruttanarungroj, C. Enhanced Hydrogen Production by Optimization of Immobilized Cells of the Green Alga Tetraspora sp., CU2551 Grown under Anaerobic Condition. Biomass Bioenergy 2018, 111, 88–95. [Google Scholar] [CrossRef]
- Vargas, S.R.; Santos, P.V.D.; Giraldi, L.A.; Zaiat, M.; Calijuri, M.d.C. Anaerobic Phototrophic Processes of Hydrogen Production by Different Strains of Microalgae Chlamydomonas sp. FEMS Microbiol. Lett. 2018, 365, fny073. [Google Scholar] [CrossRef] [Green Version]
- Ahmed, S.F.; Mofijur, M.; Nahrin, M.; Chowdhury, S.N.; Nuzhat, S.; Alherek, M.; Rafa, N.; Ong, H.C.; Nghiem, L.D.; Mahlia, T.M.I. Biohydrogen Production from Wastewater-Based Microalgae: Progresses and Challenges. Int. J. Hydrog. Energy 2021, 47, 37321–37342. [Google Scholar] [CrossRef]
- Rahmani, A.; Zerrouki, D.; Djafer, L.; Ayral, A. Hydrogen Recovery from the Photovoltaic Electroflocculation-Flotation Process for Harvesting Chlorella pyrenoidosa Microalgae. Int. J. Hydrog. Energy 2017, 42, 19591–19596. [Google Scholar] [CrossRef]
- Alalayah, W.M.; Al-Zahrani, A.; Edris, G.; Demirbas, A. Kinetics of Biological Hydrogen Production from Green Microalgae Chlorella vulgaris using Glucose as Initial Substrate. Energy Sources Part A Recovery Util. Environ. Eff. 2017, 39, 1210–1215. [Google Scholar] [CrossRef]
- Yagi, T.; Yamashita, K.; Okada, N.; Isono, T.; Momose, D.; Mineki, S.; Tokunaga, E. Hydrogen Photoproduction in Green Algae Chlamydomonas reinhardtii Sustainable over 2 Weeks with the Original Cell Culture without Supply of Fresh Cells nor Exchange of the Whole Culture Medium. J. Plant Res. 2016, 129, 771–779. [Google Scholar] [CrossRef]
- Das, A.A.K.; Esfahani, M.M.N.; Velev, O.D.; Pamme, N.; Paunov, V.N. Artificial Leaf Device for Hydrogen Generation from Immobilised C. reinhardtii Microalgae. J. Mater. Chem. A 2015, 3, 20698–20707. [Google Scholar] [CrossRef] [Green Version]
- Oncel, S.S.; Kose, A.; Faraloni, C.; Imamoglu, E.; Elibol, M.; Torzillo, G.; Vardar Sukan, F. Biohydrogen Production from Model Microalgae Chlamydomonas reinhardtii: A Simulation of Environmental Conditions for Outdoor Experiments. Int. J. Hydrog. Energy 2015, 40, 7502–7510. [Google Scholar] [CrossRef]
- Chen, Y. Global Potential of Algae-Based Photobiological Hydrogen Production. Energy Environ. Sci. 2022, 15, 2843–2857. [Google Scholar] [CrossRef]
- Faraji, M.; Saidi, M. Hydrogen-Rich Syngas Production via Integrated Configuration of Pyrolysis and Air Gasification Processes of Various Algal Biomass: Process Simulation and Evaluation Using Aspen Plus Software. Int. J. Hydrog. Energy 2021, 46, 18844–18856. [Google Scholar] [CrossRef]
- Adnan, M.A.; Xiong, Q.; Muraza, O.; Hossain, M.M. Gasification of Wet Microalgae to Produce H2-Rich Syngas and Electricity: A Thermodynamic Study Considering Exergy Analysis. Renew. Energy 2020, 147, 2195–2205. [Google Scholar] [CrossRef]
- Li, S.; Li, F.; Zhu, X.; Liao, Q.; Chang, J.-S.; Ho, S.-H. Biohydrogen Production from Microalgae for Environmental Sustainability. Chemosphere 2022, 291, 132717. [Google Scholar] [CrossRef]
- Musa Ardo, F.; Wei Lim, J.; Ramli, A.; Kee Lam, M.; Kiatkittipong, W.; Alaaeldin Abdelfattah, E.; Kashif Shahid, M.; Usman, A.; Wongsakulphasatch, S.; Tasnim Sahrin, N. A Review in Redressing Challenges to Produce Sustainable Hydrogen from Microalgae for Aviation Industry. Fuel 2022, 330, 125646. [Google Scholar] [CrossRef]
- Sridhar, A.; Ponnuchamy, M.; Senthil Kumar, P.; Kapoor, A.; Xiao, L. Progress in the Production of Hydrogen Energy from Food Waste: A Bibliometric Analysis. Int. J. Hydrog. Energy 2022, 47, 26326–26354. [Google Scholar] [CrossRef]
- Sillero, L.; Sganzerla, W.G.; Forster-Carneiro, T.; Solera, R.; Perez, M. A Bibliometric Analysis of the Hydrogen Production from Dark Fermentation. Int. J. Hydrog. Energy 2022, 47, 27397–27420. [Google Scholar] [CrossRef]
- Maneeruttanarungroj, C.; Lindblad, P.; Incharoensakdi, A. A Newly Isolated Green Alga, Tetraspora sp., CU2551, from Thailand with Efficient Hydrogen Production. Int. J. Hydrog. Energy 2010, 35, 13193–13199. [Google Scholar] [CrossRef]
- Mona, S.; Kumar, S.S.; Kumar, V.; Parveen, K.; Saini, N.; Deepak, B.; Pugazhendhi, A. Green Technology for Sustainable Biohydrogen Production (Waste to Energy): A Review. Sci. Total Environ. 2020, 728, 138481. [Google Scholar] [CrossRef]
- Chen, J.; Li, J.; Li, Q.; Wang, S.; Wang, L.; Liu, H.; Fan, C. Engineering a Chemoenzymatic Cascade for Sustainable Photobiological Hydrogen Production with Green Algae. Energy Environ. Sci. 2020, 13, 2064–2068. [Google Scholar] [CrossRef]
- Dalena, F.; Senatore, A.; Tursi, A.; Basile, A. Bioenergy Production from Second- and Third-Generation Feedstocks. In Bioenergy Systems for the Future; Elsevier: Amsterdam, The Netherlands, 2017; pp. 559–599. ISBN 978-0-08-101031-0. [Google Scholar]
- Goswami, R.K.; Mehariya, S.; Obulisamy, P.K.; Verma, P. Advanced Microalgae-Based Renewable Biohydrogen Production Systems: A Review. Bioresour. Technol. 2021, 320, 124301. [Google Scholar] [CrossRef] [PubMed]
- Javed, M.A.; Zafar, A.M.; Aly Hassan, A.; Zaidi, A.A.; Farooq, M.; El Badawy, A.; Lundquist, T.; Mohamed, M.M.A.; Al-Zuhair, S. The Role of Oxygen Regulation and Algal Growth Parameters in Hydrogen Production via Biophotolysis. J. Environ. Chem. Eng. 2022, 10, 107003. [Google Scholar] [CrossRef]
- Show, K.-Y.; Yan, Y.; Zong, C.; Guo, N.; Chang, J.-S.; Lee, D.-J. State of the Art and Challenges of Biohydrogen from Microalgae. Bioresour. Technol. 2019, 289, 121747. [Google Scholar] [CrossRef] [PubMed]
- Jiao, J.-L.; Wang, F.; Duan, P.-G.; Xu, Y.-P.; Yan, W.-H. Catalytic Hydrothermal Gasification of Microalgae for Producing Hydrogen and Methane-Rich Gas. Energy Sources Part A Recovery Util. Environ. Eff. 2017, 39, 851–860. [Google Scholar] [CrossRef]
- Kumar, M.; Oyedun, A.O.; Kumar, A. A Parametric Study through the Modelling of Hydrothermal Gasification for Hydrogen Production from Algal Biomass. Can. J. Chem. Eng. 2021, 99, S39–S54. [Google Scholar] [CrossRef]
- Demirbas, A. Hydrogen from Mosses and Algae via Pyrolysis and Steam Gasification. Energy Sources Part A Recovery Util. Environ. Eff. 2009, 32, 172–179. [Google Scholar] [CrossRef]
- Chader, S.; Mahmah, B.; Chetehouna, K.; Amrouche, F.; Abdeladim, K. Biohydrogen Production Using Green Microalgae as an Approach to Operate a Small Proton Exchange Membrane Fuel Cell. Int. J. Hydrog. Energy 2011, 36, 4089–4093. [Google Scholar] [CrossRef] [Green Version]
- Chatzitakis, A.; Nikolakaki, E.; Sotiropoulos, S.; Poulios, I. Hydrogen Production Using an Algae Photoelectrochemical Cell. Appl. Catal. B Environ. 2013, 142–143, 161–168. [Google Scholar] [CrossRef]
- Saifuddin, N.; Ong, M.Y.; Priatharsini, P. Optimization of Photosynthetic Hydrogen Gas Production by Green Alga in Sulfur Deprived Condition. Indian J. Sci. Technol. 2016, 9, 93390. [Google Scholar] [CrossRef]
- Mujalin Pholchan, K.K. Effect of Light Intensities and Atmospheric Gas Conditions on Biohydrogen Production of Microalgae Isolated from Fisheries Wastewater. Environ. Nat. Resour. J. 2017, 15, 21–29. [Google Scholar] [CrossRef]
- Raman, R.; Nair, V.K.; Prakash, V.; Patwardhan, A.; Nedungadi, P. Green-Hydrogen Research: What Have We Achieved, and Where Are We Going? Bibliometrics Analysis. Energy Rep. 2022, 8, 9242–9260. [Google Scholar] [CrossRef]
- Aria, M.; Cuccurullo, C. Bibliometrix: An R-Tool for Comprehensive Science Mapping Analysis. J. Informetr. 2017, 11, 959–975. [Google Scholar] [CrossRef]
- Nazarpour, M.; Taghizadeh-Alisaraei, A.; Asghari, A.; Abbaszadeh-Mayvan, A.; Tatari, A. Optimization of Biohydrogen Production from Microalgae by Response Surface Methodology (RSM). Energy 2022, 253, 124059. [Google Scholar] [CrossRef]
- Gomes, J.G.; Mitoura, J.; Guirardello, R. Thermodynamic Analysis for Hydrogen Production from the Reaction of Subcritical and Supercritical Gasification of the C. vulgaris Microalgae. Energy 2022, 260, 125030. [Google Scholar] [CrossRef]
- Cheng, J.; Yue, L.; Ding, L.; Li, Y.-Y.; Ye, Q.; Zhou, J.; Cen, K.; Lin, R. Improving Fermentative Hydrogen and Methane Production from an Algal Bloom through Hydrothermal/Steam Acid Pretreatment. Int. J. Hydrog. Energy 2019, 44, 5812–5820. [Google Scholar] [CrossRef]
- Wang, Q.; Gong, Y.; Liu, S.; Wang, D.; Liu, R.; Zhou, X.; Nghiem, L.D.; Zhao, Y. Free Ammonia Pretreatment To Improve Bio-Hydrogen Production from Anaerobic Dark Fermentation of Microalgae. ACS Sustain. Chem. Eng. 2019, 7, 1642–1647. [Google Scholar] [CrossRef]
- Kumar, G.; Sivagurunathan, P.; Thi, N.B.D.; Zhen, G.; Kobayashi, T.; Kim, S.-H.; Xu, K. Evaluation of Different Pretreatments on Organic Matter Solubilization and Hydrogen Fermentation of Mixed Microalgae Consortia. Int. J. Hydrog. Energy 2016, 41, 21628–21640. [Google Scholar] [CrossRef]
- Skjånes, K.; Knutsen, G.; Källqvist, T.; Lindblad, P. H2 Production from Marine and Freshwater Species of Green Algae during Sulfur Deprivation and Considerations for Bioreactor Design. Int. J. Hydrog. Energy 2008, 33, 511–521. [Google Scholar] [CrossRef]
- Onwudili, J.A.; Lea-Langton, A.R.; Ross, A.B.; Williams, P.T. Catalytic Hydrothermal Gasification of Algae for Hydrogen Production: Composition of Reaction Products and Potential for Nutrient Recycling. Bioresour. Technol. 2013, 127, 72–80. [Google Scholar] [CrossRef]
- Melis, A.; Zhang, L.; Forestier, M.; Ghirardi, M.L.; Seibert, M. Sustained Photobiological Hydrogen Gas Production upon Reversible Inactivation of Oxygen Evolution in the Green Alga Chlamydomonas reinhardtii. Plant Physiol. 2000, 122, 127–136. [Google Scholar] [CrossRef]
- Zhang, L.; Happe, T.; Melis, A. Biochemical and Morphological Characterization of Sulfur-Deprived and H2-Producing Chlamydomonas reinhardtii (Green Alga). Planta 2002, 214, 552–561. [Google Scholar] [CrossRef] [PubMed]
- Sirawattanamongkol, T.; Maswanna, T.; Maneeruttanarungroj, C. A Newly Isolated Green Alga Chlorella sp. KLSc59: Potential for Biohydrogen Production. J. Appl. Phycol. 2020, 32, 2927–2936. [Google Scholar] [CrossRef]
- Unpaprom, Y. A Newly Isolated Green Alga, Scenedesmus acuminatus, from Thailand with Efficient Hydrogen Production. Chiang Mai J. Sci. 2017, 44, 1270–1278. [Google Scholar]
- Winkler, M.; Kuhlgert, S.; Hippler, M.; Happe, T. Characterization of the Key Step for Light-Driven Hydrogen Evolution in Green Algae. J. Biol. Chem. 2009, 284, 36620–36627. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kruse, O.; Rupprecht, J.; Bader, K.-P.; Thomas-Hall, S.; Schenk, P.M.; Finazzi, G.; Hankamer, B. Improved Photobiological H2 Production in Engineered Green Algal Cells. J. Biol. Chem. 2005, 280, 34170–34177. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Antal, T.K.; Krendeleva, T.E.; Laurinavichene, T.V.; Makarova, V.V.; Ghirardi, M.L.; Rubin, A.B.; Tsygankov, A.A.; Seibert, M. The Dependence of Algal H2 Production on Photosystem II and O2 Consumption Activities in Sulfur-Deprived Chlamydomonas einhardtii Cells. Biochim. Et Biophys. Acta (BBA) Bioenerg. 2003, 1607, 153–160. [Google Scholar] [CrossRef] [Green Version]
- Guan, Y.; Deng, M.; Yu, X.; Zhang, W. Two-Stage Photo-Biological Production of Hydrogen by Marine Green Alga Platymonas subcordiformis. Biochem. Eng. J. 2004, 19, 69–73. [Google Scholar] [CrossRef]
- Lakatos, G.; Deák, Z.; Vass, I.; Rétfalvi, T.; Rozgonyi, S.; Rákhely, G.; Ördög, V.; Kondorosi, É.; Maróti, G. Bacterial Symbionts Enhance Photo-Fermentative Hydrogen Evolution of Chlamydomonas Algae. Green Chem. 2014, 16, 4716–4727. [Google Scholar] [CrossRef]
- Duman, G.; Uddin, M.A.; Yanik, J. Hydrogen Production from Algal Biomass via Steam Gasification. Bioresour. Technol. 2014, 166, 24–30. [Google Scholar] [CrossRef] [Green Version]
- Hwang, J.-H.; Kim, H.-C.; Choi, J.-A.; Abou-Shanab, R.A.I.; Dempsey, B.A.; Regan, J.M.; Kim, J.R.; Song, H.; Nam, I.-H.; Kim, S.-N.; et al. Photoautotrophic Hydrogen Production by Eukaryotic Microalgae under Aerobic Conditions. Nat. Commun. 2014, 5, 3234. [Google Scholar] [CrossRef] [Green Version]
- Nguyen, A.V.; Thomas-Hall, S.R.; Malnoë, A.; Timmins, M.; Mussgnug, J.H.; Rupprecht, J.; Kruse, O.; Hankamer, B.; Schenk, P.M. Transcriptome for Photobiological Hydrogen Production Induced by Sulfur Deprivation in the Green Alga Chlamydomonas reinhardtii. Eukaryot. Cell 2008, 7, 1965–1979. [Google Scholar] [CrossRef] [PubMed]
- United Nations Environment Programme. Emissions Gap Report 2022: The Closing Window—Climate Crisis Calls for Rapid Transformation of Societies. Nairobi. 2022. Available online: https://www.unep.org/emissions-Gap-Report-2022 (accessed on 1 November 2022).
- Leung, X.Y.; Sun, J.; Bai, B. Bibliometrics of Social Media Research: A Co-Citation and Co-Word Analysis. Int. J. Hosp. Manag. 2017, 66, 35–45. [Google Scholar] [CrossRef]
- Melis, A. Photosynthetic H2 Metabolism in Chlamydomonas reinhardtii (Unicellular Green Algae). Planta 2007, 226, 1075–1086. [Google Scholar] [CrossRef] [PubMed]
- Kosourov, S.; Tsygankov, A.; Seibert, M.; Ghirardi, M.L. Sustained Hydrogen Photoproduction by Chlamydomonas reinhardtii: Effects of Culture Parameters. Biotechnol. Bioeng. 2002, 78, 731–740. [Google Scholar] [CrossRef]
- Hallenbeck, P. Biological Hydrogen Production; Fundamentals and Limiting Processes. Int. J. Hydrog. Energy 2002, 27, 1185–1193. [Google Scholar] [CrossRef]
- Gaffron, H.; Rubin, J. Fermentative and Photochemical Production of Hydrogen in Algae. J. Gen. Physiol. 1942, 26, 219–240. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, N.; Srivastava, M.; Singh, R.; Syed, A.; Bahadur Pal, D.; Elgorban, A.M.; Kushwaha, D.; Mishra, P.K.; Gupta, V.K. Co-Fermentation of Residual Algal Biomass and Glucose under the Influence of Fe3O4 Nanoparticles to Enhance Biohydrogen Production under Dark Mode. Bioresour. Technol. 2021, 342, 126034. [Google Scholar] [CrossRef]
- Sun, C.; Xia, A.; Liao, Q.; Fu, Q.; Huang, Y.; Zhu, X.; Wei, P.; Lin, R.; Murphy, J.D. Improving Production of Volatile Fatty Acids and Hydrogen from Microalgae and Rice Residue: Effects of Physicochemical Characteristics and Mix Ratios. Appl. Energy 2018, 230, 1082–1092. [Google Scholar] [CrossRef]
- Xu, J.; Upcraft, T.; Tang, Q.; Guo, M.; Huang, Z.; Zhao, M.; Ruan, W. Hydrogen Generation Performance from Taihu Algae and Food Waste by Anaerobic Codigestion. Energy Fuels 2019, 33, 1279–1289. [Google Scholar] [CrossRef]
- Hwang, J.-H.; Lee, M.; Kang, E.H.; Lee, W.H. Renewable Algal Photo H2 Production without S Control Using Acetate Enriched Fermenter Effluents. Int. J. Hydrog. Energy 2021, 46, 1740–1751. [Google Scholar] [CrossRef]
- Kim, D.-H.; Yoon, J.-J.; Kim, S.-H.; Park, J.-H. Effect of Conductive Material for Overcoming Inhibitory Conditions Derived from Red Algae-Based Substrate on Biohydrogen Production. Fuel 2021, 285, 119059. [Google Scholar] [CrossRef]
- Zhao, M.; Liu, Z.; Xu, J.; Liu, H.; Dai, X.; Gu, S.; Ruan, W. Dosing Effect of Nano Zero Valent Iron (nZVI) on the Dark Hydrogen Fermentation Performance via Lake Algae and Food Waste Co-Digestion. Energy Rep. 2020, 6, 3192–3199. [Google Scholar] [CrossRef]
- Chang, K.-L.; Lin, Y.-C.; Shangdiar, S.; Chen, S.-C.; Hsiao, Y.-H. Hydrogen Production from Dry Spirulina Algae with Downstream Feeding in Microwave Plasma Reactor Assisted under Atmospheric Pressure. J. Energy Inst. 2020, 93, 1597–1601. [Google Scholar] [CrossRef]
- Nurdiawati, A.; Zaini, I.N.; Irhamna, A.R.; Sasongko, D.; Aziz, M. Novel Configuration of Supercritical Water Gasification and Chemical Looping for Highly-Efficient Hydrogen Production from Microalgae. Renew. Sustain. Energy Rev. 2019, 112, 369–381. [Google Scholar] [CrossRef]
- Nurdiawati, A.; Zaini, I.N.; Aziz, M. Efficient Hydrogen Production from Algae and Its Conversion to Methylcyclohexane. Chem. Eng. Trans. 2018, 70, 1507–1512. [Google Scholar] [CrossRef]
- Xie, L.-F.; Duan, P.-G.; Jiao, J.-L.; Xu, Y.-P. Hydrothermal Gasification of Microalgae over Nickel Catalysts for Production of Hydrogen-Rich Fuel Gas: Effect of Zeolite Supports. Int. J. Hydrog. Energy 2019, 44, 5114–5124. [Google Scholar] [CrossRef]
- Raheem, A.; Cui, X.; Mangi, F.H.; Memon, A.A.; Ji, G.; Cheng, B.; Dong, W.; Zhao, M. Hydrogen-Rich Energy Recovery from Microalgae (Lipid-Extracted) via Steam Catalytic Gasification. Algal Res. 2020, 52, 102102. [Google Scholar] [CrossRef]
- Phunpruch, S. Biomass and Biohydrogen Production by Unicellular Green Alga Chlorella vulgaris var. vulgaris TISTR 8261 Using Frozen Food Industrial Wastewater. Asia-Pac. J. Sci. Technol. 2022, 27, 9-Jan. [Google Scholar] [CrossRef]
- Maliutina, K.; Tahmasebi, A.; Yu, J. Pressurized Entrained-Flow Pyrolysis of Microalgae: Enhanced Production of Hydrogen and Nitrogen-Containing Compounds. Bioresour. Technol. 2018, 256, 160–169. [Google Scholar] [CrossRef]
- Rosha, P.; Kumar, S.; Vikram, S.; Ibrahim, H.; Al-Muhtaseb, A.H. H2-Enriched Gaseous Fuel Production via Co-Gasification of an Algae-Plastic Waste Mixture Using Aspen PLUS. Int. J. Hydrog. Energy 2022, 47, 26294–26302. [Google Scholar] [CrossRef]
- Ban, S.; Lin, W.; Luo, J. Ca2+ Enhances Algal Photolysis Hydrogen Production by Improving the Direct and Indirect Pathways. Int. J. Hydrog. Energy 2019, 44, 1466–1473. [Google Scholar] [CrossRef]
- Chen, J.; Li, Y.; Li, M.; Shi, J.; Wang, L.; Luo, S.; Liu, H. Chemical Flocculation-Based Green Algae Materials for Photobiological Hydrogen Production. ACS Appl. Bio Mater. 2022, 5, 897–903. [Google Scholar] [CrossRef] [PubMed]
- Sharma, P.; Sivaramakrishnaiah, M.; Deepanraj, B.; Saravanan, R.; Reddy, M.V. A Novel Optimization Approach for Biohydrogen Production using Algal Biomass. Int. J. Hydrog. Energy 2022. [Google Scholar] [CrossRef]
- Sittijunda, S.; Sitthikitpanya, N.; Plangklang, P.; Reungsang, A. Two-Stage Anaerobic Codigestion of Crude Glycerol and Micro-Algal Biomass for Biohydrogen and Methane Production by Anaerobic Sludge Consortium. Fermentation 2021, 7, 175. [Google Scholar] [CrossRef]
- Pewnual, T.; Jampapetch, N.; Saladtook, S.; Raksajit, W.; Klinsalee, R.; Maneeruttanarungroj, C. Response of Green Alga Tetraspora sp., CU2551 under Potassium Deprivation: A New Promising Strategy for Hydrogen Production. J. Appl. Phycol. 2022, 34, 811–819. [Google Scholar] [CrossRef]
- Manoyan, J.; Samovich, T.; Kozel, N.; Demidchik, V.; Gabrielyan, L. Growth Characteristics, Biohydrogen Production and Photochemical Activity of Photosystems in Green Microalgae Parachlorella kessleri Exposed to Nitrogen Deprivation. Int. J. Hydrog. Energy 2022, 47, 16815–16823. [Google Scholar] [CrossRef]
- Bechara, R.; Azizi, F.; Boyadjian, C. Process Simulation and Optimization for Enhanced Biophotolytic Hydrogen Production from Green Algae Using the Sulfur Deprivation Method. Int. J. Hydrog. Energy 2021, 46, 14096–14108. [Google Scholar] [CrossRef]
- Laokua, N.; Rittiyan, N.; Kornrawudaphikasama, Y.; Klinsalee, R.; Tonawut, Y.; Preechaphonkul, N.; Raksajit, W.; Khetkorn, W.; Dejtisakdi, W.; Maneeruttanarungroj, C. Optimal Conditions for Maximized H2 Yield from a New Green Algal Strain Chlorella sp., KLSc61. J. Appl. Phycol. 2022, 34, 1909–1919. [Google Scholar] [CrossRef]
- Manoyan, J.; Gabrielyan, L.; Kozel, N.; Trchounian, A. Regulation of Biohydrogen Production by Protonophores in Novel Green Microalgae Parachlorella Kessleri. J. Photochem. Photobiol. B Biol. 2019, 199, 111597. [Google Scholar] [CrossRef]
- Paramesh, K.; Lakshmana Reddy, N.; Shankar, M.V.; Chandrasekhar, T. Enhancement of Biological Hydrogen Production Using Green Alga Chlorococcum minutum. Int. J. Hydrog. Energy 2018, 43, 3957–3966. [Google Scholar] [CrossRef]
- Kanygin, A.; Smith, A.; Nagy, V.; Tóth, S.Z.; Redding, K.E. Interplay between Hydrogen Production and Photosynthesis in a Green Alga Expressing an Active Photosystem I-Hydrogenase Chimera. Int. J. Hydrog. Energy 2022, 47, 21969–21983. [Google Scholar] [CrossRef]
- Rai, A.; Khan, M.J.; Ahirwar, A.; Deka, R.; Singh, N.; Schoefs, B.; Marchand, J.; Varjani, S.; Vinayak, V. Hydrogen Economy and Storage by Nanoporous Microalgae Diatom: Special Emphasis on Designing Photobioreactors. Int. J. Hydrog. Energy 2022, 47, 42099–42121. [Google Scholar] [CrossRef]
- Cheng, J.; Yue, L.; Hua, J.; Dong, H.; Li, Y.-Y.; Zhou, J.; Lin, R. Hydrothermal Heating with Sulphuric Acid Contributes to Improved Fermentative Hydrogen and Methane Co-Generation from Dianchi Lake Algal Bloom. Energy Convers. Manag. 2019, 192, 282–291. [Google Scholar] [CrossRef]
- Homburg, S.V.; Kruse, O.; Patel, A.V. Viability, Growth, and Hydrogen Production of Green Microalgae in Novel Silica Hydrogels. Chem. Ing. Tech. 2018, 90, 1162. [Google Scholar] [CrossRef] [Green Version]
- Ban, S.; Lin, W.; Wu, F.; Luo, J. Algal-Bacterial Cooperation Improves Algal Photolysis-Mediated Hydrogen Production. Bioresour. Technol. 2018, 251, 350–357. [Google Scholar] [CrossRef] [PubMed]
- Hupp, B.; Pap, B.; Farkas, A.; Maróti, G. Development of a Microalgae-Based Continuous Starch-to-Hydrogen Conversion Approach. Fermentation 2022, 8, 294. [Google Scholar] [CrossRef]
- Shetty, P.; Boboescu, I.Z.; Pap, B.; Wirth, R.; Kovács, K.L.; Bíró, T.; Futó, Z.; White, R.A.; Maróti, G. Exploitation of Algal-Bacterial Consortia in Combined Biohydrogen Generation and Wastewater Treatment. Front. Energy Res. 2019, 7, 52. [Google Scholar] [CrossRef]
- Jokel, M.; Nagy, V.; Tóth, S.Z.; Kosourov, S.; Allahverdiyeva, Y. Elimination of the Flavodiiron Electron Sink Facilitates Long-Term H2 Photoproduction in Green Algae. Biotechnol. Biofuels 2019, 12, 280. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Zhuang, X.; Chen, M.; Zeng, Z.; Cai, X.; Li, H.; Hu, Z. An Endogenous MicroRNA (MiRNA1166.1) Can Regulate Photobio-H2 Production in Eukaryotic Green Alga Chlamydomonas reinhardtii. Biotechnol. Biofuels 2018, 11, 126. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Jiang, X.; Hu, C.; Sun, T.; Zeng, Z.; Cai, X.; Li, H.; Hu, Z. Optogenetic Regulation of Artificial MicroRNA Improves H2 Production in Green Alga Chlamydomonas reinhardtii. Biotechnol. Biofuels 2017, 10, 257. [Google Scholar] [CrossRef] [Green Version]
- Elman, T.; Hoai Ho, T.T.; Milrad, Y.; Hippler, M.; Yacoby, I. Enhanced Chloroplast-Mitochondria Crosstalk Promotes Ambient Algal-H2 Production. Cell Rep. Phys. Sci. 2022, 3, 100828. [Google Scholar] [CrossRef]
- Liu, J.-Z.; Ge, Y.-M.; Sun, J.-Y.; Chen, P.; Addy, M.; Huo, S.-H.; Li, K.; Cheng, P.-F.; Ruan, R. Exogenic Glucose as an Electron Donor for Algal Hydrogenases to Promote Hydrogen Photoproduction by Chlorella pyrenoidosa. Bioresour. Technol. 2019, 289, 121762. [Google Scholar] [CrossRef] [PubMed]
- Phunpruch, S. Effect of Cell Density and Nutrient Deprivation on Hydrogen Production by Unicellular Green Alga Scenedesmus sp., KMITL-OVG1. Asia-Pac. J. Sci. Technol. 2019, 24, 1–9. [Google Scholar] [CrossRef]
- Sengmee, D.; Cheirsilp, B.; Suksaroge, T.T.; Prasertsan, P. Biophotolysis-Based Hydrogen and Lipid Production by Oleaginous Microalgae Using Crude Glycerol as Exogenous Carbon Source. Int. J. Hydrog. Energy 2017, 42, 1970–1976. [Google Scholar] [CrossRef]
- Gholkar, P.; Shastri, Y.; Tanksale, A. Catalytic Reactive Flash Volatilisation of Microalgae to Produce Hydrogen or Methane-Rich Syngas. Appl. Catal. B Environ. 2019, 251, 326–334. [Google Scholar] [CrossRef]
- Duangjan, K.; Nakkhunthod, W.; Pekkoh, J.; Pumas, C. Comparison of Hydrogen Production in Microalgae under Autotrophic and Mixotrophic Media. Bot. Lith. 2017, 23, 169–177. [Google Scholar] [CrossRef] [Green Version]
- Narravula Raga, S.; Duddela, V.; Pallaval Veera, B.; Poda, S.; Thummala, C. Effects of Various Factors on Biomass, Bioethanol, and Biohydrogen Production in Green Alga Chlamydomonas reinhardtii. J. App. Biol. Biotech. 2021, 9, 152–156. [Google Scholar] [CrossRef]

| Rank | Keywords | Occurrences | Total Link Strength |
|---|---|---|---|
| 1 | Microalgae | 34 | 22 |
| 2 | Green algae | 24 | 20 |
| 3 | Chlamydomonas reinhardtii | 18 | 15 |
| 4 | Hydrogenase | 12 | 19 |
| 5 | Algae | 11 | 10 |
| 6 | Methane | 10 | 8 |
| 7 | Photosynthesis | 10 | 18 |
| 8 | Renewable energy | 7 | 7 |
| 9 | Sulfur deprivation | 7 | 11 |
| 10 | Algal biomass | 6 | 2 |
| 11 | Biomass | 6 | 6 |
| 12 | Chlamydomonas | 6 | 8 |
| 13 | Photobioreactor | 6 | 6 |
| 14 | Pretreatment | 6 | 6 |
| 15 | Biofuels | 5 | 5 |
| 16 | Dark fermentation | 5 | 4 |
| 17 | Green alga | 5 | 8 |
| 18 | Fermentation | 4 | 6 |
| 19 | Gasification | 4 | 4 |
| 20 | Optimization | 4 | 3 |
| 21 | Platymonas subcordiformis | 4 | 5 |
| 22 | Pyrolysis | 4 | 2 |
| 23 | Supercritical water gasification | 4 | 3 |
| Ranked by TP * | Ranked by TC * | Ranked by CPP * | ||||||
|---|---|---|---|---|---|---|---|---|
| Rank | Journal Name | TP | Rank | Journal Name | TC | Rank | Journal Name | CPP |
| 1 | Int. J. Hydrog. Energy | 47 | 1 | Int. J. Hydrog. Energy | 1194 | 1 | Plant Physiology | 873 |
| 2 | Bioresource Technology | 17 | 2 | Bioresource Technology | 1016 | 2 | Planta | 341 |
| 3 | Biotechnology for Biofuels | 7 | 3 | Plant Physiology | 873 | 3 | J. Biological Chemistry | 195 |
| 4 | Energy | 5 | 4 | J. Biological Chemistry | 391 | 4 | BBA—Bioenergetics | 165 |
| 5 | Fuel | 4 | 5 | Planta | 341 | 5 | Eukaryotic cell | 111 |
| 6 | J. Applied Phycology | 4 | 6 | Biotechnology for Biofuels | 247 | 6 | Dalton Transactions | 97 |
| 7 | Algal Research | 3 | 7 | Biochemical Eng. J | 175 | 7 | J. Bioscience and Bio. Eng | 94 |
| 8 | Asia-Pacific J. Sci. Technol. | 3 | 8 | BBA—Bioenergetics | 165 | 8 | Biochemical Eng. J | 87 |
| 9 | Biotechnology Letters | 3 | 9 | Fuel | 125 | 9 | Nature Communications | 80 |
| 10 | Biotechnology Progress | 3 | 10 | PLOS One | 124 | 10 | Water Research | 78 |
| Ranked by TP | Ranked by TC | Ranked by CPP | ||||||
|---|---|---|---|---|---|---|---|---|
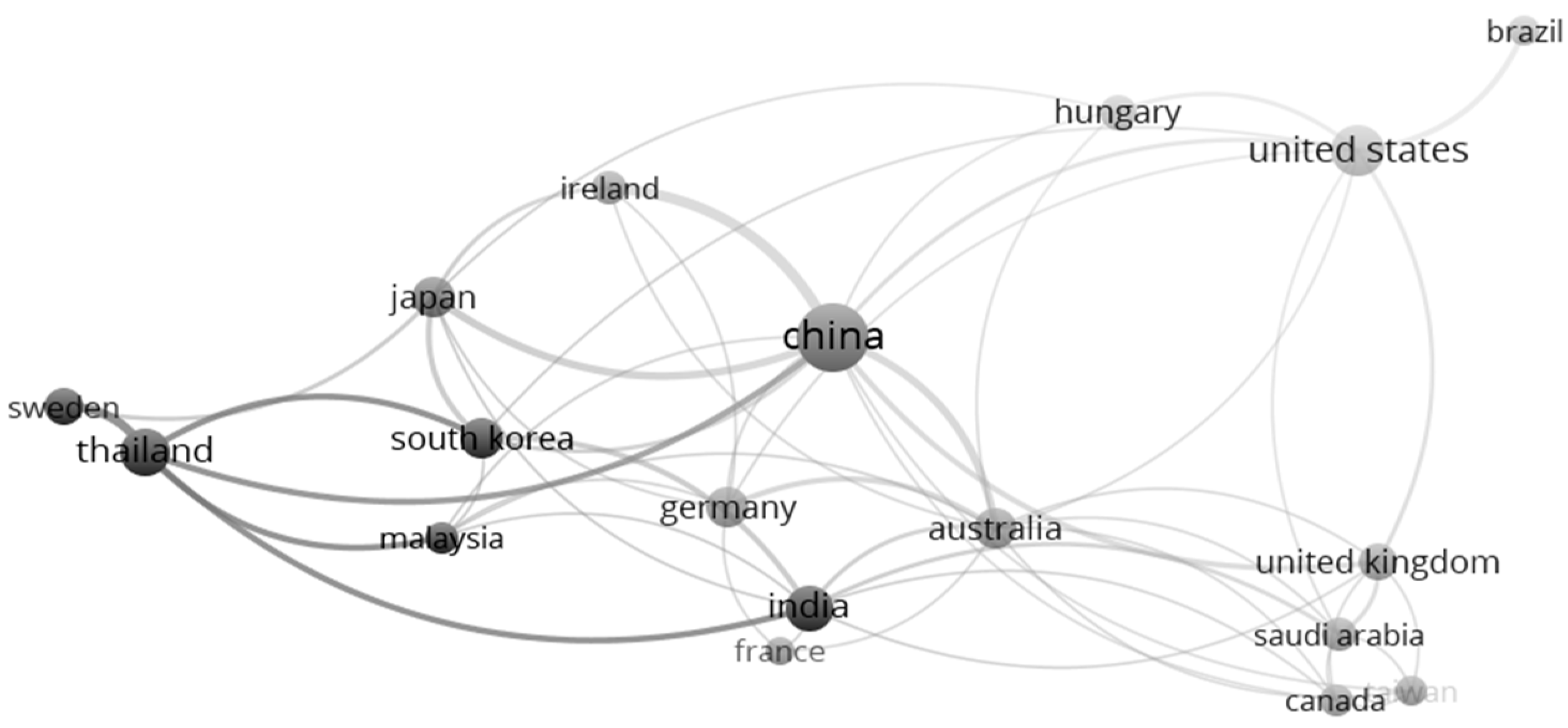
| Rank | Country | TP | Rank | Country | TC | Rank | Country | CPP |
| 1 | China | 51 | 1 | USA | 1944 | 1 | Germany | 96 |
| 2 | USA | 26 | 2 | China | 1636 | 2 | USA | 75 |
| 3 | Thailand | 17 | 3 | Germany | 1255 | 3 | France | 71 |
| 4 | India | 16 | 4 | Australia | 897 | 4 | Australia | 64 |
| 5 | Australia | 14 | 5 | Japan | 581 | 5 | Ireland | 50 |
| 6 | Germany | 13 | 6 | United Kingdom | 421 | 6 | Japan | 45 |
| 7 | Japan | 13 | 7 | Ireland | 397 | 7 | United Kingdom | 38 |
| 8 | United Kingdom | 11 | 8 | France | 355 | 8 | South Korea | 33 |
| 9 | South Korea | 10 | 9 | India | 335 | 9 | China | 32 |
| 10 | Hungary | 9 | 10 | South Korea | 326 | 10 | Canada | 31 |
| Ranked by Normalized Citation Score (NCS) | ||||
|---|---|---|---|---|
| Rank | Author | Affiliation | Country | NCS |
| 1 | Jerry D Murphy | University College Cork | Ireland | 10 |
| 2 | Ao Xia | Chongqing University | China | 9 |
| 3 | Maria Lucia Ghirardi | National Renewable Energy Lab. | USA | 9 |
| 4 | Jie Cheng | Zhejiang University | China | 8 |
| 5 | Lin Zhengyan | Zhejiang University | China | 8 |
| 6 | Mei Zhao | Jiangnan University | China | 7 |
| 7 | Michael Seibert | National Renewable Energy Lab. | USA | 6 |
| 8 | Ben Hankamer | The University of Queensland | Australia | 6 |
| 9 | Gopalakrishnan Kumar | University of Stavanger | Norway | 6 |
| 10 | Thomas Happe | Ruhr-Universitat Bochum | Germany | 6 |
| Ranked by Global Citations | Ranked by Local Citations | ||||
|---|---|---|---|---|---|
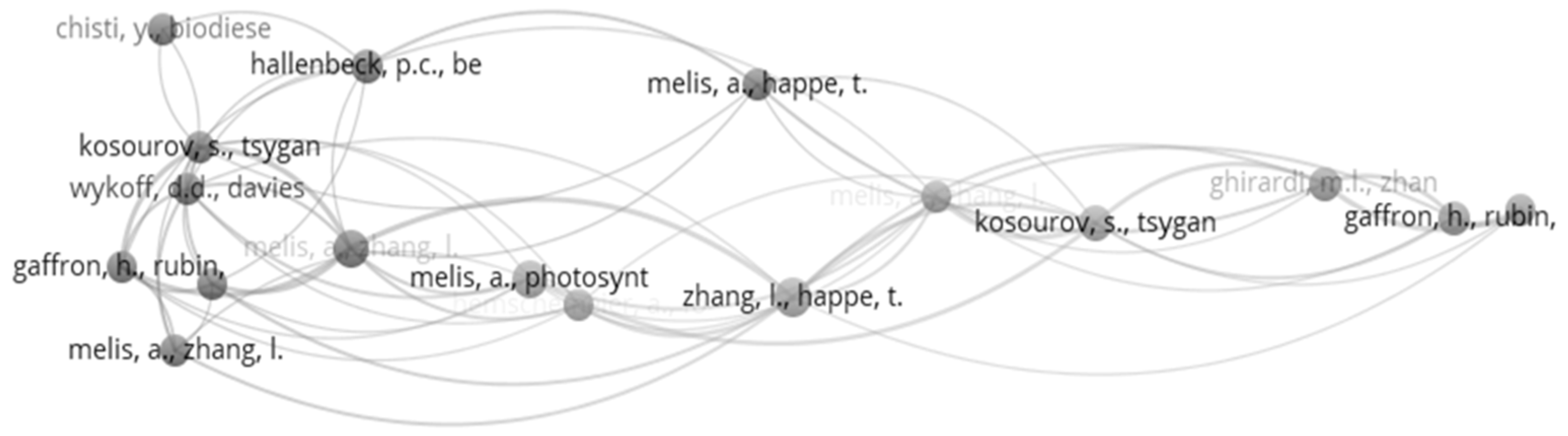
| Rank | Article | Global Citations | Rank | Article | Local Citations |
| 1 | Melis et al. (2000) [51] | 873 | 1 | Melis et al. (2000) [51] | 60 |
| 2 | Zhang et al. (2002) [52] | 341 | 2 | Zhang et al. (2002) [52] | 20 |
| 3 | Kruse et al. (2005) [56] | 286 | 3 | Kruse et al. (2005) [56] | 12 |
| 4 | Antal et al. (2003) [57] | 165 | 4 | Guan et al. (2004) [58] | 11 |
| 5 | Onwudili et al. (2013) [50] | 147 | 5 | Maneeruttanarungroj et al. (2010) [28] | 9 |
| 6 | Guan et al. (2004) [58] | 142 | 6 | Lakatos et al. (2014) [59] | 7 |
| 7 | Xia et al. (2016) [2] | 134 | 7 | Skjanes et al. (2008) [49] | 7 |
| 8 | Duman et al. (2014) [60] | 124 | 8 | Hwang et al. (2014) [61] | 7 |
| 9 | Nguyen et al. (2008) [62] | 111 | 9 | Antal et al. (2003) [57] | 7 |
| 10 | Srirangan et al. (2011) [4] | 110 | 10 | Onwudili et al. (2013) [50] | 6 |
| Research Theme | Recent Advancement |
|---|---|
| Techniques to produce bioH2 using microalgae | Co-fermentation of cyanobacteria and glucose with Fe3O4 nanoparticles [69], co-fermentation of pretreated rice residue and microalgae [70], anaerobic co-digestion of Taihu Algae and food waste [71], bioH2 production without sulfur using fermenter effluents enriched in acetate [72]. Effect of magnetite (Fe3O4) supplement on bioH2 production of Clostridium Butyricum DSM 10702 by anaerobic fermentation [73]. Dosing of nano zero-valent iron (nZVI) in the dark co-fermentation system [74]. Pyrolysis of dry spirulina algae in an atmospheric pressure microwave plasma reactor [75].Integrated supercritical water gasification (SCWG), syngas chemical looping (SCL), and H2ation system [76,77]. Catalytic SCWG with Nickel (Ni)/zeolite catalysts [78], catalytic and non-catalytic gasification with a hybrid functional mixture of waste eggshell-derived Calcium oxide (CaO), Ni catalyst, and Yttrium oxide Y2O3 [79], Catalytic hydrothermal gasification using noble metals Platinum (Pt) and Ruthenium (Ru) [35]. BioH2 production by Chlorella vulgaris var. vulgaris TISTR 8261 using frozen food industrial wastewater [80]. Pressurized entrained flow pyrolysis of Chlorella vulgaris microalgae [81].Co-gasification of algae-plastic waste for bioH2 production by Aspen Plus simulation model [82]. Application of Calcium ions (Ca2+) to enhance algal photolysis bioH2 production [83]. Chemical flocculation of the green algae Chlorella pyrenoidosa to form aggregates for 11 days of continuous photobiological hydrogen production [84]. |
| Optimization of factors affecting bioH2 production from microalgae | Response surface methodology to optimize sulfur concentration, run time, and algal biomass concentration [44,85], response surface methodology with central composite design for optimization of factors such as crude glycerol, microalgal biomass, and inoculum of bioH2 production from co-digestion of crude glycerol and microalgae by anaerobic sludge [86]. Potassium deprivation conditions to enhance bioH2 production [87]. Nitrogen-deprived conditions gave 4–5-fold more bioH2 [88]. Optimization of conditions for supercritical gasification using GAMS software [45]. Optimization of essential process parameters such as reaction time, sulfur concentrations, and the medium pH for bio photolytic cyclic process [89]. Optimization of temperature, catalyst loading reaction time for catalytic gasification by central composite design [11], integrated pyrolysis and air gasification processes of algal waste, Chlorella vulgaris, Rhizoclonium sp., and Spirogyra by simulation model developed using Aspen Plus software to optimize the gasifier temperature, gasifier pressure, and air flowrate [22]. |
| Alternative and potential microalgal species | Newly isolated green algae Chlorella sp. KLSc59: [53], Chlorella sp. KLSc61 [90]. Another newly isolated green alga, Scenedesmus acuminatus from Thailand [54], Parachlorella kessleri isolated from Armenia [91], and Chlorococcum minutum [92]. Chlamydomonas reinhardtii having PSI-H2ase chimera polypeptide for enhanced H2 production [93]. H2 generation from the diatoms by culturing it in various solar panel photobioreactors [94]. |
| Strategies to improve the bioH2 production from microalgae | Anaerobic co-digestion of food waste and high-pressure homogenization pretreated Taihu lake wet algae [3], hydrothermal sulfuric acid pretreatment to improve fermentative bioH2 of Dianchi Lake algal bloom [46,95]. An innovative free ammonia pretreatment technology [47]. Immobilized cells of Tetraspora sp. CU2551 in alginate matrix under aerobic sulfur deprivation and under anaerobic sulfur deprivation to maximize bioH2 production [12,13]. The entrapment of microalgae in silica gels [96]. Microalgae-bacteria consortiums such as Chlamydomonas sp. and Pseudomonas sp. strain D [97], Chlamydomonas and Chlorella genera with a starch-degrading bacterium from the Bacillus genus [98], Chlorella vulgaris MACC360, and Archaea [99]. Removal of flavodiiron proteins in Chlamydomonas reinhardtii showed increased bioH2 [100], regulation of photo bioH2 production in Chlamydomonas reinhardtii by an endogenous microRNA [101], Optogenetic regulation of artificial microRNA in Chlamydomonas reinhardtii [102] and Chlamydomonas defective in thylakoid proton gradient overcame the limitation due to O2 and carbon fixations [103]. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Arimbrathodi, S.P.; Javed, M.A.; Hamouda, M.A.; Aly Hassan, A.; Ahmed, M.E. BioH2 Production Using Microalgae: Highlights on Recent Advancements from a Bibliometric Analysis. Water 2023, 15, 185. https://doi.org/10.3390/w15010185
Arimbrathodi SP, Javed MA, Hamouda MA, Aly Hassan A, Ahmed ME. BioH2 Production Using Microalgae: Highlights on Recent Advancements from a Bibliometric Analysis. Water. 2023; 15(1):185. https://doi.org/10.3390/w15010185
Chicago/Turabian StyleArimbrathodi, Shirin P., Muhammad Asad Javed, Mohamed A. Hamouda, Ashraf Aly Hassan, and Mahmoud E. Ahmed. 2023. "BioH2 Production Using Microalgae: Highlights on Recent Advancements from a Bibliometric Analysis" Water 15, no. 1: 185. https://doi.org/10.3390/w15010185
APA StyleArimbrathodi, S. P., Javed, M. A., Hamouda, M. A., Aly Hassan, A., & Ahmed, M. E. (2023). BioH2 Production Using Microalgae: Highlights on Recent Advancements from a Bibliometric Analysis. Water, 15(1), 185. https://doi.org/10.3390/w15010185










