Determination of Groundwater Recharge Mechanism Based on Environmental Isotopes in Chahannur Basin
Abstract
:1. Introduction
2. Materials and Methods
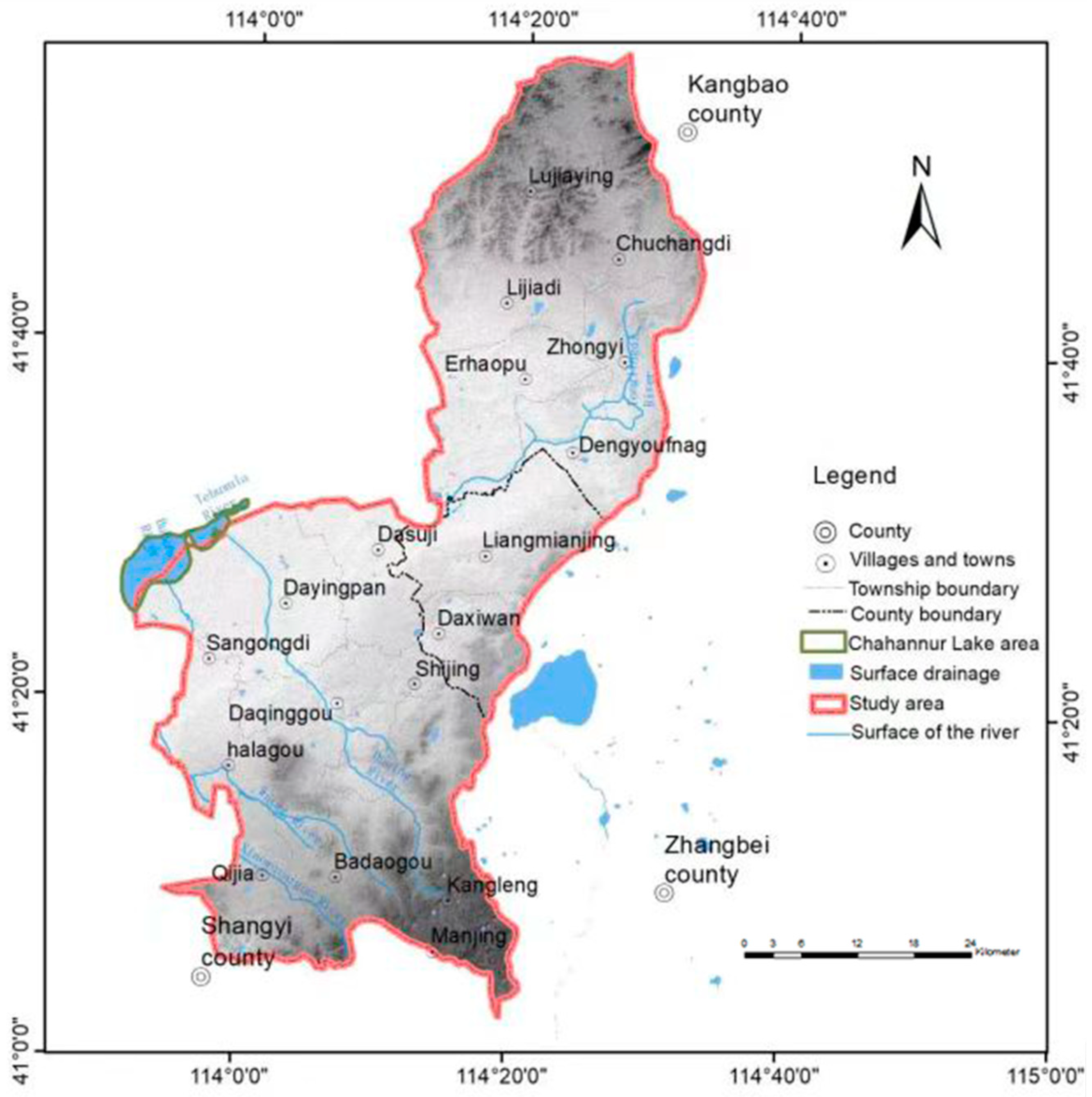
2.1. Study Area
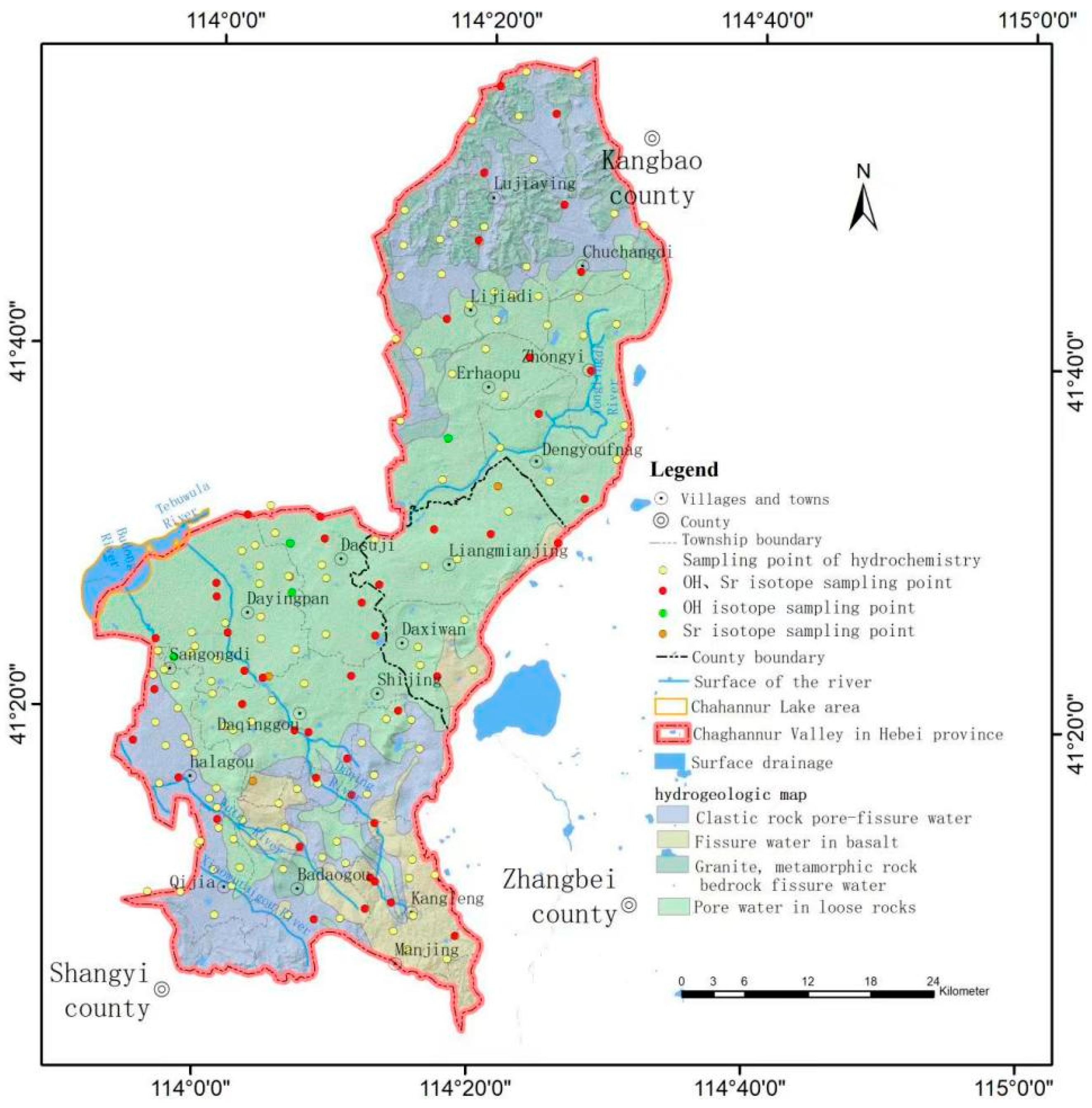
2.2. Data Collection
2.3. Groundwater Age Determination
2.3.1. 3H Method to Calculate Groundwater Age
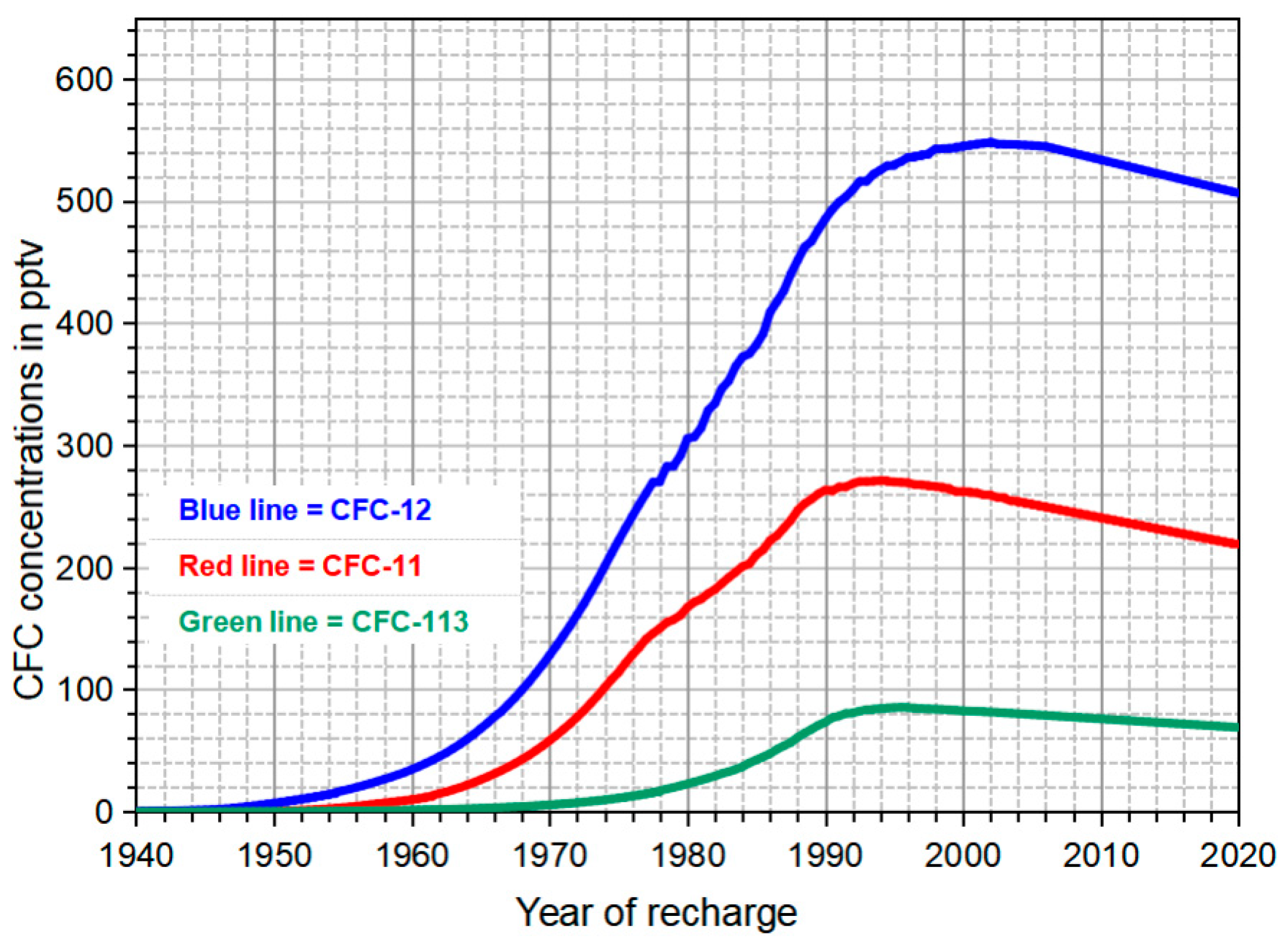
2.3.2. CFCs Dating Method
3. Results and Discussion
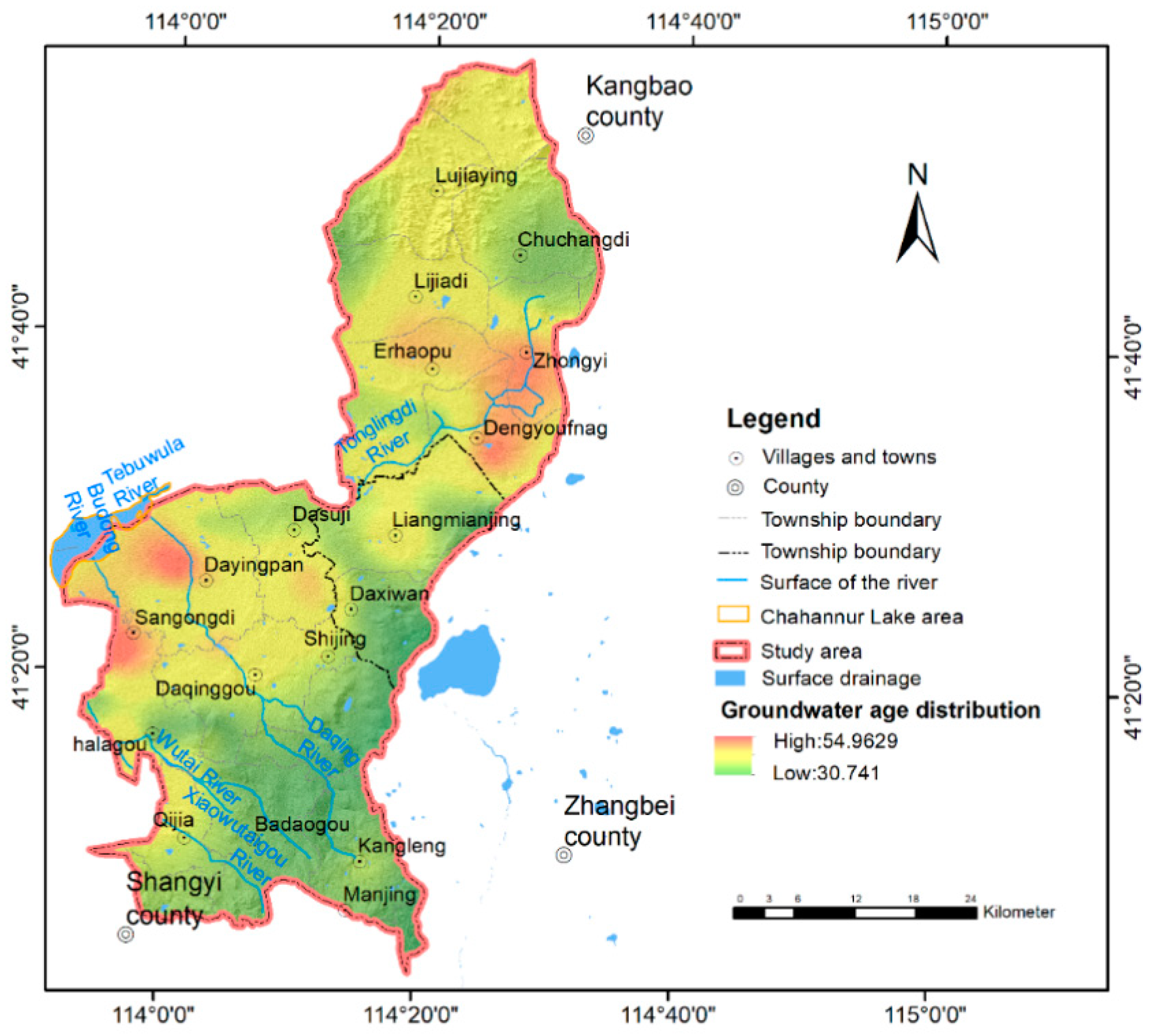
3.1. Age Characteristics of Groundwater in the Chahannur Basin
3.1.1. Groundwater Age Estimated by Tritium Isotope (3H)
3.1.2. Groundwater Ages Estimated by CFCs
3.2. Groundwater Recharge Flow Pattern in the Basin
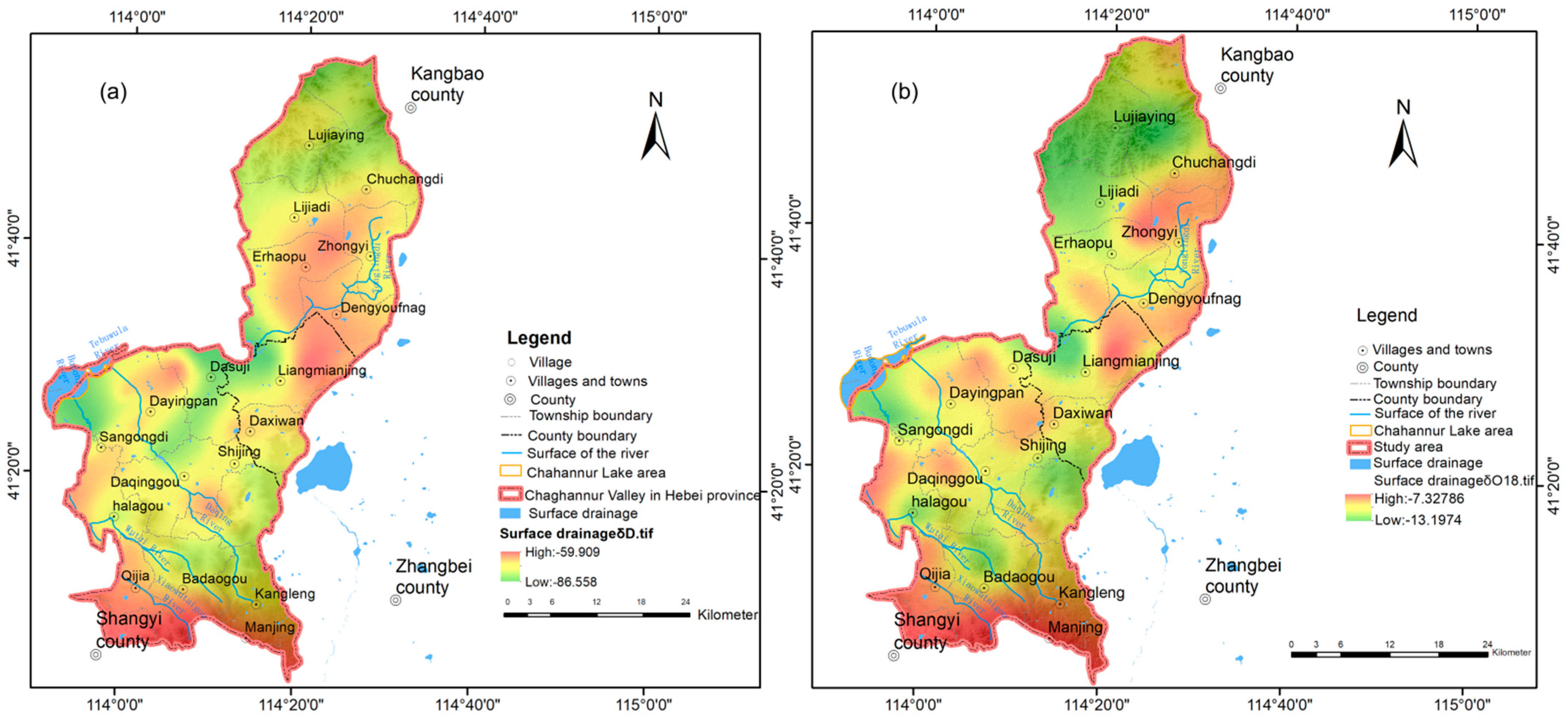
3.2.1. Characteristics of Water Cycle Indicated by Hydrogen and Oxygen Isotopes
Characteristics of Hydrogen and Oxygen Isotopes in Different Water Bodies
Recharge, Range, and Discharge Relationship of Groundwater Indicated by Hydrogen and Oxygen Isotopes
- Salt and Quagmire groundwater systems
- Chahannur groundwater system
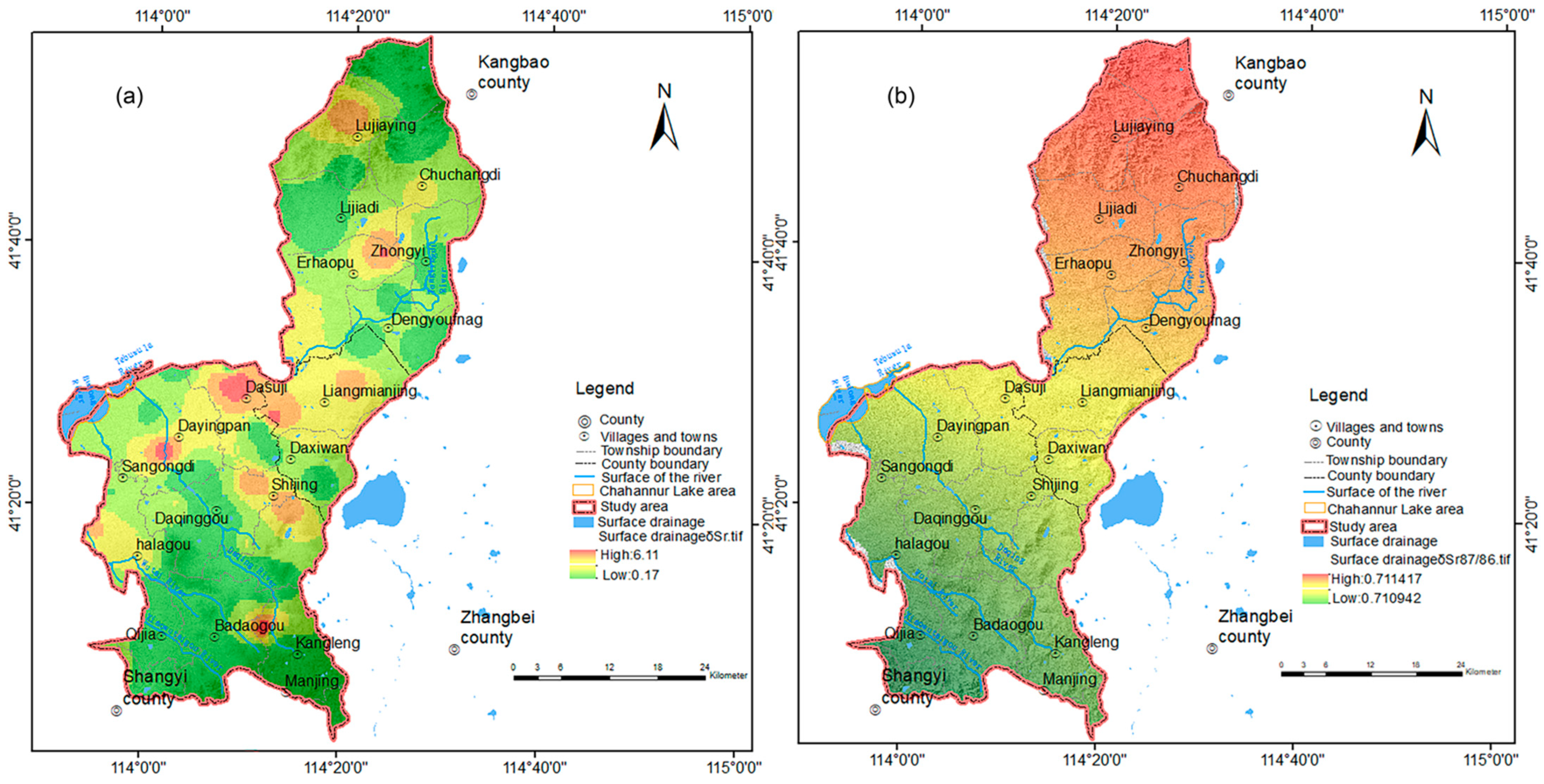
3.2.2. Characteristics of Water Cycle Indicated by Strontium Isotope
Spatial Distribution Characteristics of Strontium Isotopes
Characteristics of Groundwater Circulation as Indicated by Strontium Isotopes
4. Conclusions
- (1)
- Tritium isotope (3H) and Freon (CFCs) dating methods are suitable for the semi-quantitative dating of groundwater in the Chahannur Basin.
- (2)
- Hydrogen, oxygen, and strontium isotopes can be used to characterize the groundwater cycle in the Chahannur Basin.
- (3)
- The groundwater level around the Chahannur Lake area is low, and the groundwater in the Chahannur Basin flows from the southwest and northwest to the lake area.
- (4)
- The groundwater in the Chahannur Basin is mainly replenished by atmospheric precipitation, and the evaporation of groundwater and surface water is strong.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chen, M.; Li, Y.; Lei, X.; Li, N.; Gao, Q.; Wang, C. Quantitative assessment of spatio-temporal evolution characteristics and driving factors of groundwater level in Chahannur basin. Resour. Environ. Arid. Areas 2022, 36, 105–111. (In Chinese) [Google Scholar] [CrossRef]
- Prada, S.; Cruz, J.V.; Figueira, C. Using stable isotopes to characterize groundwater recharge sources in the volcanic island of Madeira, Portugal. J. Hydrol. 2016, 536, 409–425. [Google Scholar] [CrossRef]
- Wu, X.; Dong, W.; Lin, X. Evolution of wetland in Honghe ational Nature Reserve from the view of hydrogeology. Sci. Total Environ. 2017, 609, 1370–1380. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Li, P.; He, X. Interactions between surface water and groundwater in selected tributaries of the Wei River (China) revealed by hydrochemistry and stable isotopes. Hum. Ecol. Risk Assess. Int. J. 2021, 28, 1–21. [Google Scholar] [CrossRef]
- Matsumoto, T.; Chen, Z.; Wei, W.; Yang, G.-M.; Hu, S.-M.; Zhang, X. Application of combined 81Kr and 4He chronometers to the dating of old groundwater in a tectonically active region of the North China Plain. Earth Planet. Sci. Lett. 2018, 493, 208–217. [Google Scholar] [CrossRef]
- Anja, B.; Karsten, O.; Richard, G.; Janina, S.R.; Seifu, K.; Susanne, S. Groundwater origin and flow dynamics in active rift systems—A multi-isotope approach in the Main Ethiopian Rift. J. Hydrol. 2011, 402, 274–289. [Google Scholar]
- West, A.G.; February, E.C.; Bowen, G.J. Spatial analysis of hydrogen and oxygen stable isotopes (“isoscapes”) in ground water and tap water across South Africa. J. Geochem. Explor. 2014, 145, 213–222. [Google Scholar] [CrossRef] [Green Version]
- Vystavna, Y.; Diadin, D.; Huneau, F. Defining a stable water isotope framework for isotope hydrology application in a large trans-boundary watershed (Russian Federation/Ukraine). Isot. Environ. Health Stud. 2017, 54, 147–167. [Google Scholar] [CrossRef]
- Qiu, X.; Wang, Y.; Wang, Z.; Regenauer-Lieb, K.; Zhang, K.; Liu, J. Determining the origin, circulation path and residence time of geothermal groundwater using multiple isotopic techniques in the Heyuan Fault Zone of Southern China. J. Hydrol. 2018, 567, 339–350. [Google Scholar] [CrossRef]
- Taufiq, A.; Effendi, A.J.; Iskandar, I.; Hosono, T.; Hutasoit, L.M. Controlling factors and driving mechanisms of nitrate contamination in groundwater system of Bandung Basin, Indonesia, deduced by combined use of stable isotope ratios, CFC age dating, and socioeconomic parameters. Water Res. 2018, 148, 292–305. [Google Scholar] [CrossRef]
- Wu, X.; Chen, F.; Liu, X.; Wang, S.; Zhang, M.; Zhu, G.; Zhou, X.; Chen, J. The Significance of Hydrogen and Oxygen Stable Isotopes in the Water Vapor Source in Dingxi Area. Water 2021, 13, 2374. [Google Scholar] [CrossRef]
- Yuan, J.; Guo, Q.; Wang, Y. Geochemical behaviors of boron and its isotopes in aqueous environment of the Yangbajing and Yangyi geothermal fields, Tibet, China. J. Geochem. Explor. 2014, 140, 11–22. [Google Scholar] [CrossRef]
- Anovski, T.; Anovska-Jovcheva, E.; Lisichkov, K.; Dimitrovski, D.; Kuvendziev, S.; Leis, A.; Benischke, R.; Micevski, E.; Arsov, L. Application of environmental isotope techniques for determining the origin and mechanisms of recharge of the Rashche Spring. Environ. Earth Sci. 2019, 78, 656. [Google Scholar] [CrossRef]
- Wang, T.; Chen, J.; Zhang, C. Estimation of fossil groundwater mass fraction accounting for endogenic carbon input across California. J. Hydrol. 2021, 595, 126034. [Google Scholar] [CrossRef]
- Chandrajith, R.; Barth, J.A.; Subasinghe, N.; Merten, D.; Dissanayake, C. Geochemical and isotope characterization of geothermal spring waters in Sri Lanka: Evidence for steeper than expected geothermal gradients. J. Hydrol. 2013, 476, 360–369. [Google Scholar] [CrossRef]
- Massoudieh, A.; Ginn, T. The theoretical relation between unstable solutes and groundwater age. Water Resour. Res. 2011, 47, W10523. [Google Scholar] [CrossRef]
- Elliot, T.; Bonotto, D.M.; Andrews, J.N. Dissolved uranium, radium and radon evolution in the Continental Intercalaire aquifer, Algeria and Tunisia. J. Environ. Radioact. 2014, 137, 150–162. [Google Scholar] [CrossRef] [Green Version]
- Lee, K.-S.; Kim, J.-M.; Lee, D.-R.; Kim, Y.; Lee, D. Analysis of water movement through an unsaturated soil zone in Jeju Island, Korea using stable oxygen and hydrogen isotopes. J. Hydrol. 2007, 345, 199–211. [Google Scholar] [CrossRef]
- Woldemariyam, F.; Ayenew, T. Application of hydrochemical and isotopic techniques to understand groundwater recharge and flow systems in the Dawa River basin, southern Ethiopia. Environ. Earth Sci. 2016, 75, 1002. [Google Scholar] [CrossRef]
- Elmarami, H.; Meyer, H.; Massmann, G. Combined approach of isotope mass balance and hydrological water balance methods to constrain the sources of lake water as exemplified on the small dimictic lake Silbersee, northern Germany. Isot. Environ. Health Stud. 2017, 53, 184–197. [Google Scholar] [CrossRef]
- Zhen, Z.-L.; Li, C.-Y.; Zhang, S.; Li, W.-B.; Shi, X.-H.; Sun, B. Characteristics and indications of hydrogen and oxygen isotopes distribution in lake ice body. Water Sci. Technol. 2015, 71, 1065–1072. [Google Scholar] [CrossRef] [PubMed]
- Zhu, S.; Zhang, F.; Kung, H.-T.; Yushanjiang, A. Hydrogen and Oxygen Isotope Composition and Water Quality Evaluation for Different Water Bodies in the Ebinur Lake Watershed, Northwestern China. Water 2019, 11, 2067. [Google Scholar] [CrossRef] [Green Version]
- Kagabu, M.; Matsunaga, M.; Ide, K. Momoshima, N. Shimada, J.; Groundwater age determination using 85kr and multiple age tracers (sf6, cfcs, and 3H) to elucidate regional groundwater flow systems. J. Hydrol. Reg. Stud. 2017, 12, 165–180. [Google Scholar] [CrossRef]
- Bugai, D.A.; Fourre, E.; Jeancbaptiste, P.; Dapoigny, A.; Baumier, D.; Salle, K.L.G.L.; Lancelot, J.; Skalskyy, A.; Van Meir, N. Estimation of groundwater exchange at chernobyl site using the data of the isotope dating and hydrogeological modeling. Geol. J. 2010, 345, 119–124. [Google Scholar] [CrossRef]
- Okofo, L.; Adonadaga, M.; Martienssen, M. Groundwater age dating using multi-environmental tracers (SF6, CFC-11, CFC-12, δ18O, and δD) to investigate groundwater residence times and recharge processes in northeastern ghana. J. Hydrol. 2022, 610, 127821. [Google Scholar] [CrossRef]
- Kolbe, T.; MarcAis, J.; Thomas, Z.; Abbott, B.; Dreuzy, J.; Rousseau-Gueutin, P.; Aquilina, L.; Labasque, T.; Pinay, G. Coupling 3d groundwater modeling with cfc-based age dating to classify local groundwater circulation in an unconfined crystalline aquifer. J. Hydrol. 2016, 543, 31–46. [Google Scholar] [CrossRef] [Green Version]
- Bowen, G.J.; Kennedy, C.D.; Liu, Z.; Stalker, J. Water balance model for mean annual hydrogen and oxygen isotope distributions in surface waters of the contiguous United States. J. Geophys. Res. Earth Surf. 2011, 116, G04011. [Google Scholar] [CrossRef] [Green Version]
- Henderson, A.K.; Shuman, B.N. Hydrogen and oxygen isotopic compositions of lake water in the western United States. GSA Bull. 2009, 121, 1179–1189. [Google Scholar] [CrossRef]
- Janssen, S.E.; Johnson, M.W.; Blum, J.D.; Barkay, T.; Reinfelder, J.R. Separation of monomethylmercury from estuarine sediments for mercury isotope analysis. Chem. Geol. 2015, 411, 19–25. [Google Scholar] [CrossRef] [Green Version]
- He, X.; Li, P.; Ji, Y.; Wang, Y.; Su, Z.; Elumalai, V. Groundwater Arsenic and Fluoride and Associated Arsenicosis and Fluorosis in China: Occurrence, Distribution and Management. Expo. Health 2020, 12, 355–368. [Google Scholar] [CrossRef]
- Liu, J. Study on the variation trend of tritium concentration in Atmospheric precipitation over China in recent ten years. Investig. Sci. Technol. 2001, 4, 11–14. (In Chinese) [Google Scholar]
- Long, W.; Chen, H.; Duan, Q. Application of artificial neural network method in restoring tritium concentration in precipitation. Geol. Resour. 2008, 17, 208–212. (In Chinese) [Google Scholar]
- Wang, F. A regional model for the recovery of tritium concentration in precipitation in Jilin Province. Jilin Geol. 1998, 17, 75–81. (In Chinese) [Google Scholar]
- Vergnaud-Ayraud, V.; Aquilina, L.; Pauwels, H.; Labasque, T. Groundwater age determination by CFC analysis: A tool for groundwater sustainable management. Technol. Sci. Methodes 2008, 1, 37–44. [Google Scholar]
- Jing, L.; Fadong, L.; Song, S.; Zhang, Y.; Cai, W. Worldwide application and prospective of CFCs groundwater age-dating method. Ecol. Environ. Sci. 2012, 21, 1503–1508. [Google Scholar]
- Wang, P.; Chen, X.; Shen, L.; Wu, K.; Huang, M.; Xiao, Q. Geochemical features of the geothermal fluids from the Mapamyum non-volcanic geothermal system (Western Tibet, China). J. Volcanol. Geotherm. Res. 2016, 320, 29–39. [Google Scholar] [CrossRef]
- Kiro, Y.; Weinstein, Y.; Starinsky, A.; Yechieli, Y. Groundwater ages and reaction rates during seawater circulation in the Dead Sea aquifer. Geochim. Cosmochim. Acta 2013, 122, 17–35. [Google Scholar] [CrossRef]



| Tritium Content | Groundwater Age |
|---|---|
| <1TU | Old water (supplied before 1953) |
| 1~3 TU | Old water, new water (0~10 years supply), mixed |
| 3~10 TU | New water |
| 10~20 TU | Nuclear explosion 3H remained |
| >20 TU | Mainly supplied in the 1960s |
| Hole Number | Depth of the Water Table (m) | Depth of Monitoring Well (m) | Tritium Values | Age | Hole Number | Depth of the Water Table | Depth of Monitoring Well (m) | Tritium Values | Age | Hole Number | Depth of the Water Table | Depth of Monitoring Well (m) | Tritium Values | Age |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| KBA59 | 12.94 | 51.82 | 6.1 ± 0.7 | 23 | SYB20 | 3.72 | 51.51 | 7.2 ± 0.4 | 20 | SYC75 | 1.2 | 51.97 | 8.6 ± 0.6 | 17 |
| KBB41 | 27.77 | 51.69 | 1.3 ± 0.7 | 51 | SYB32 | 12.45 | 50.73 | 3.4 ± 0.6 | 33 | SYC76 | 230 | 50.58 | 13.9 ± 0.7 | 8 |
| KBB46 | 10.82 | 50.27 | <1.3 | >51 | SYB64 | 0.8 | 50.34 | 12.1 ± 0.5 | 11 | SYC78 | 330 | 50.47 | 13.9 ± 0.7 | 8 |
| KBB47 | 8.45 | 50.10 | 10.7 ± 0.9 | 13 | SYB66 | 2.13 | 51.90 | 11.3 ± 0.5 | 12 | SYC81 | 235 | 51.84 | 17.3 ± 0.9 | 4 |
| KBB49 | 11.47 | 50.32 | 3.6 ± 0.7 | 32 | SYB67 | 4.53 | 50.37 | 8.8 ± 0.7 | 16 | SYC82 | 13.4 | 50.58 | <1.3 | >51 |
| KBB51 | 10.5 | 51.93 | 7.2 ± 0.8 | 20 | SYBS08 | 34.82 | 51.75 | 5.9 ± 0.6 | 23 | SYCS36 | 25.9 | 51.18 | <1.3 | >51 |
| KBB53 | 10.62 | 51.30 | 4.6 ± 0.7 | 28 | SYBS19 | 39.05 | 50.85 | 4.1 ± 0.5 | 30 | Syx02 | 33.4 | 51.08 | 2.4 ± 0.5 | 40 |
| KBC40 | 6.31 | 50.23 | <1.3 | >51 | SYC07 | 15.72 | 50.29 | 7.7 ± 0.6 | 19 | Syx05 | 14.87 | 50.80 | 7.0 ± 0.6 | 20 |
| KBC48 | 27.7 | 51.87 | <1.3 | >51 | SYC20 | 12.01 | 51.19 | 2.6 ± 0.5 | 38 | Syx06 | 15.15 | 51.27 | <1.3 | >51 |
| KBC55 | 21.5 | 50.11 | 3.0 ± 0.9 | 36 | SYC26 | 9.6 | 52.00 | 4.9 ± 0.5 | 27 | Syx08 | 16.84 | 50.69 | <1.3 | >51 |
| R11 | 23.4 | 50.00 | <1.3 | >51 | SYC27 | 9.52 | 51.26 | 10.3 ± 0.6 | 13 | Syx09 | 7.6 | 51.92 | 3.9 ± 0.5 | 31 |
| SBY27 | 3.08 | 51.20 | <1.3 | >51 | SYC30 | 5.23 | 51.77 | 9.6 ± 0.6 | 15 | ZBA45 | 18.2 | 51.91 | 7.2 ± 0.6 | 20 |
| SYA23 | 1.1 | 51.62 | 9.8 ± 0.4 | 14 | SYC31 | 8.3 | 50.62 | 7.8 ± 0.6 | 18 | ZBA46 | 13.8 | 51.03 | 5.9 ± 0.5 | 23 |
| SYAS19 | 18.67 | 51.16 | 6.4 ± 0.4 | 22 | SYC58 | 30.65 | 51.18 | <1.3 | >51 | ZBA50- | 2.3 | 51.63 | 10.8 ± 0.6 | 13 |
| SYB03 | 38.8 | 50.41 | <1.3 | >51 | SYC59 | 26.5 | 51.20 | 4.1 ± 0.5 | 30 | ZBA51 | 3.3 | 50.79 | 11.9 ± 0.6 | 11 |
| SYB15 | 9.78 | 50.52 | 1.9 ± 0.3 | 44 | SYC60 | 14.75 | 51.08 | <1.3 | >51 | |||||
| SYB17 | 2.64 | 50.09 | <1.3 | >51 | SYC68 | 32.4 | 51.25 | 6.2 ± 0.6 | 23 |
| Point | CFC-11 | CFC-12 | CFC-113 | Age | Point | CFC-11 | CFC-12 | CFC-113 | Age |
|---|---|---|---|---|---|---|---|---|---|
| KBA37 | C | 34 | 35 | 35 | SYB20 | 33 | 38 | C | 36 |
| KBA41 | 52 | 49 | 43 | 48 | SYB50 | 49 | 54 | 38 | 47 |
| KBA43 | 36 | 38 | 37 | 37 | SYB55 | 47 | C | C | 47 |
| KBA44 | 36 | 43 | 33 | 37 | SYB65 | 43 | 45 | C | 44 |
| KBA59 | 36 | 37 | 34 | 36 | SYB66 | C | 37 | 36 | 37 |
| KBA63 | 45 | 45 | 42 | 44 | SYB67 | 41 | C | 40 | 41 |
| KBB33 | 51 | 47 | 41 | 46 | SYC04 | C | 35 | 34 | 35 |
| KBB35 | 43 | 38 | 38 | 40 | SYC07 | C | 38 | 36 | 37 |
| KBB36 | C | 50 | 34 | 42 | SYC08 | 35 | 36 | 34 | 35 |
| KBB45 | 49 | 45 | 40 | 45 | SYC27 | 49 | 46 | 40 | 45 |
| KBB46 | 55 | 55 | 47 | 52 | SYC29 | 47 | 45 | 37 | 43 |
| KBB47 | 35 | 37 | 38 | 37 | SYC30 | 46 | 45 | 38 | 43 |
| KBC40 | 49 | 49 | 38 | 45 | SYC35 | 48 | 46 | 38 | 44 |
| KBC45 | 64 | 49 | 41 | 51 | SYC61 | 45 | 53 | 38 | 45 |
| SYB60 | 34 | 36 | 33 | 34 | SYC63 | 49 | 47 | 40 | 45 |
| KBC52 | 51 | 41 | 39 | 44 | SYC65 | C | - | 33 | 33 |
| KBC54 | 36 | 43 | 35 | 38 | SYC67 | C | 32 | 34 | 33 |
| KBC55 | 47 | 42 | 39 | 43 | SYC70 | 36 | 29 | 34 | 33 |
| r8 | 32 | 30 | 35 | 32 | SYC75 | 37 | 30 | 33 | 33 |
| SBY27 | 55 | C | C | 55 | SYC84 | 48 | 44 | 38 | 43 |
| SYA18 | C | 37 | 35 | 36 | SYX09 | 37 | 40 | 35 | 37 |
| SYA29 | 55 | 61 | 49 | 55 | ZBA47 | 34 | 33 | 33 | 33 |
| SYA30 | 47 | C | 36 | 42 | ZBA50 | C | 37 | 36 | 37 |
| SYB15 | 30 | 37 | 33 | 33 | ZBA51 | 41 | 39 | 37 | 39 |
| SYB16 | 46 | 49 | 42 | 46 | ZBB52 | 43 | 36 | 38 | 39 |
| ZBC56 | 32 | 27 | 34 | 31 |
| Groundwater Type | Statistics | δ18O (‰) | δ2H (‰) |
|---|---|---|---|
| phreatic water | minimum value | −13.20 | −86.90 |
| maximum | −7.90 | −63.10 | |
| mean | −9.90 | −72.00 | |
| standard deviation | 1.30 | 6.40 |
| Type of Water Body | δD Value Variation Range | δ18O Value Variation Range |
|---|---|---|
| Spring water | −74.4~−71.3‰ | −11.4~−9.7‰ |
| River (reservoir) water | −50.8~−47.1‰ | −6.6~−4.5‰ |
| Loose rock pore water | −86.9~−63.1‰ | −13.2~−7.9‰ |
| Clastic rock fissure water | −81.9~−64.5‰ | −12.6~−8.2‰ |
| Basalt fissure hole water | −77.9~−67.7‰ | −11.7~−9.2‰ |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tian, X.; Gong, Z.; Fu, L.; You, D.; Li, F.; Wang, Y.; Chen, Z.; Zhou, Y. Determination of Groundwater Recharge Mechanism Based on Environmental Isotopes in Chahannur Basin. Water 2023, 15, 180. https://doi.org/10.3390/w15010180
Tian X, Gong Z, Fu L, You D, Li F, Wang Y, Chen Z, Zhou Y. Determination of Groundwater Recharge Mechanism Based on Environmental Isotopes in Chahannur Basin. Water. 2023; 15(1):180. https://doi.org/10.3390/w15010180
Chicago/Turabian StyleTian, Xizhao, Zhiqiang Gong, Lulu Fu, Di You, Fan Li, Yahui Wang, Zhi Chen, and Yahong Zhou. 2023. "Determination of Groundwater Recharge Mechanism Based on Environmental Isotopes in Chahannur Basin" Water 15, no. 1: 180. https://doi.org/10.3390/w15010180





