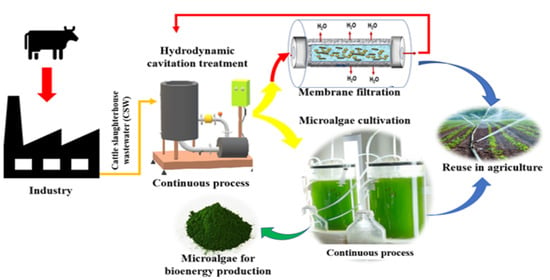
Continuous Cultivation of Microalgae in Cattle Slaughterhouse Wastewater Treated with Hydrodynamic Cavitation
Abstract
:1. Introduction
2. Materials and Method
2.1. Slaughterhouse Wastewater
2.2. Hydrodynamic Cavitation System
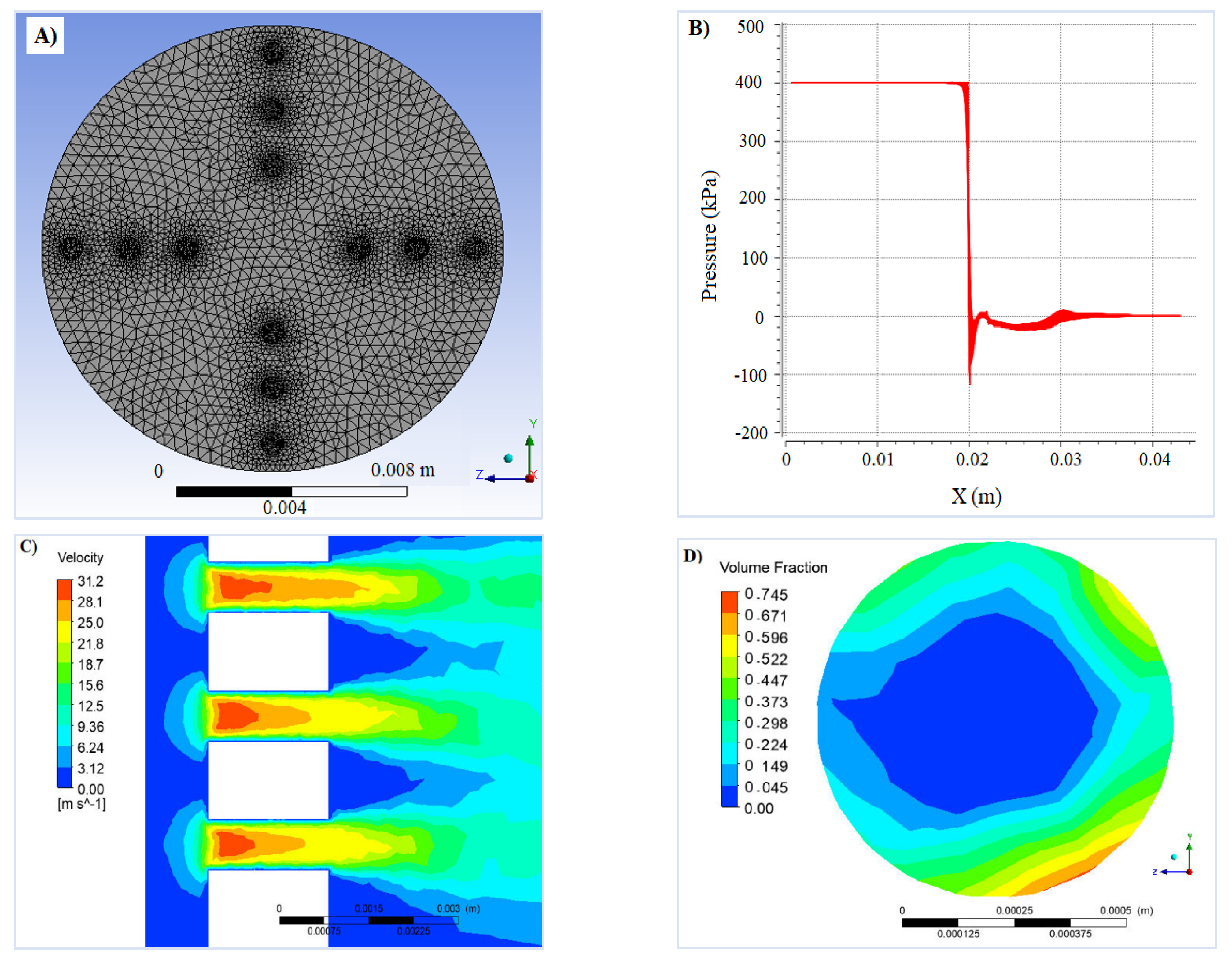
2.3. Computational Fluid Dynamics (CFD) Modeling
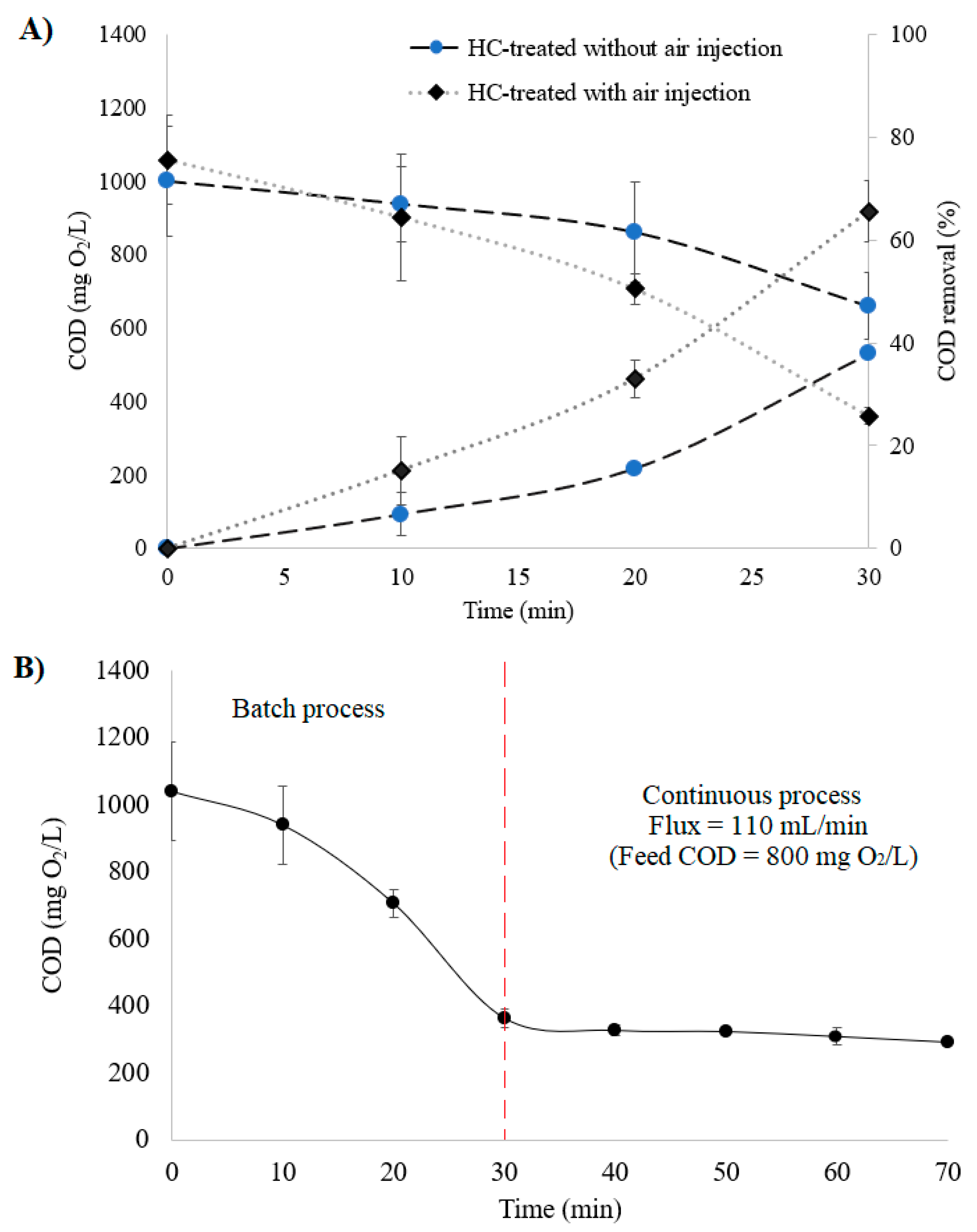
2.4. Hydrodynamic Cavitation Treatment of Cattle Slaughterhouse Wastewater
2.5. Membrane Filtration of HC Treated Water
2.6. Microalgae Cultivation in HC Treated Wastewater
3. Results and Discussion
3.1. Cattle Slaughterhouse Wastewater (CSW)
3.2. CFD Modelling and CSW Treatment Using HC
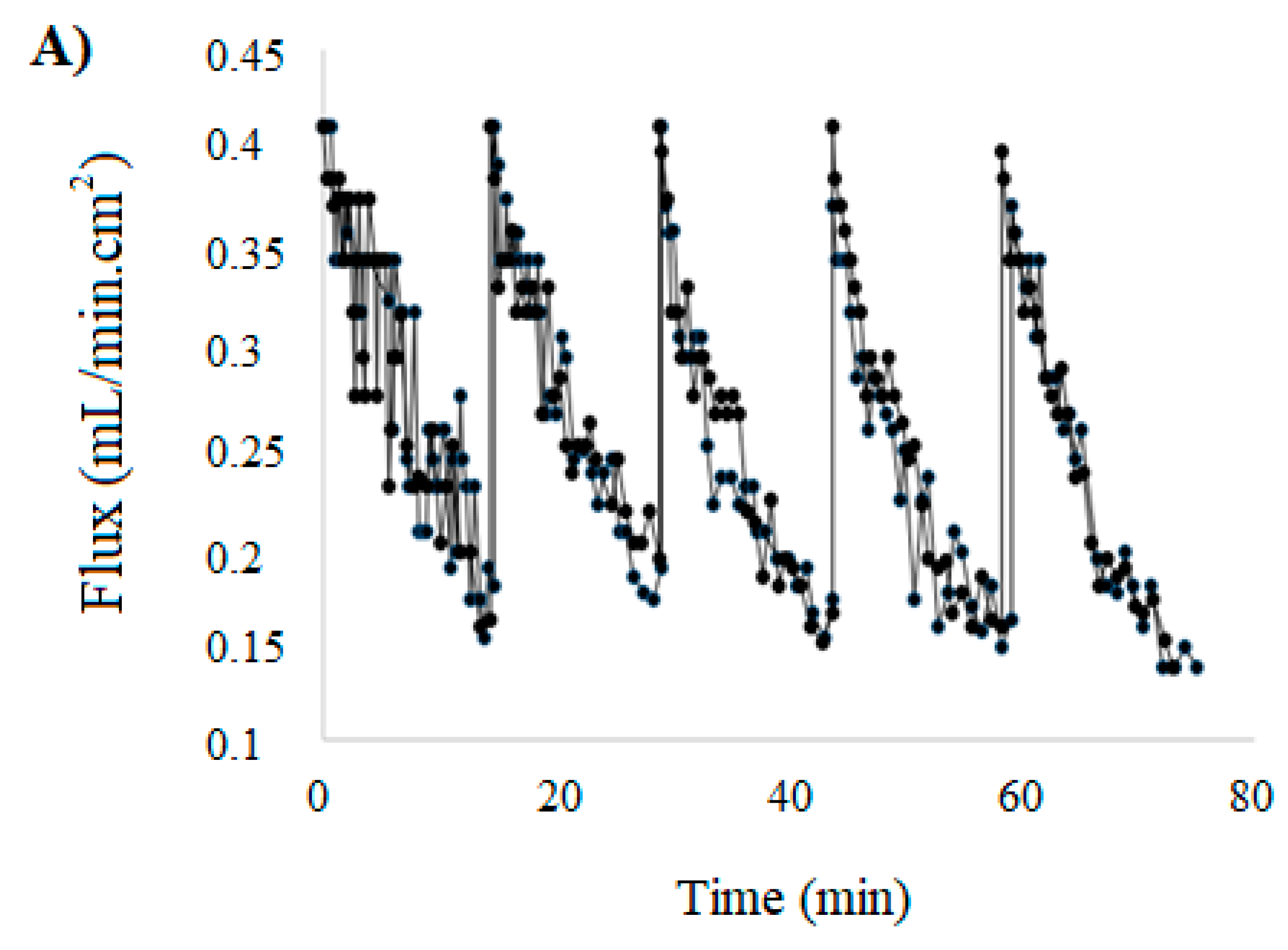
3.3. Membrane Filtration of HC-CSW
3.4. Microalgae Cultivation in HC-CSW
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- USDA Foreign Agricultural Service. Livestock and Poultry: World Markets and Trade. Available online: https://www.fas.usda.gov/data/livestock-and-poultry-world-markets-and-trade (accessed on 6 December 2021).
- Scopus. Analyze Search Results. Available online: https://www.scopus.com/term/analyzer.uri?sid=c99b618064d3edbb14619296c58c664b&origin=resultslist&src=s&s=TITLE-ABS-KEY%28hydrodynamic+cavitation%29&sort=plf-f&sdt=b&sot=b&sl=38&count=3624&analyzeResults=Analyze+results&txGid=01abb3754889fb68786b52042dac66 (accessed on 6 December 2021).
- Gogate, P.R.; Pandit, A.B. A Review and Assessment of Hydrodynamic Cavitation as a Technology for the Future. Ultrason. Sonochem. 2005, 12, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Badmus, K.O.; Irakoze, N.; Adeniyi, O.R.; Petrik, L. Synergistic Advance Fenton Oxidation and Hydrodynamic Cavitation Treatment of Persistent Organic Dyes in Textile Wastewater. J. Environ. Chem. Eng. 2020, 8, 103521. [Google Scholar] [CrossRef]
- Thanekar, P.; Panda, M.; Gogate, P.R. Degradation of Carbamazepine Using Hydrodynamic Cavitation Combined with Advanced Oxidation Processes. Ultrason. Sonochem. 2018, 40, 567–576. [Google Scholar] [CrossRef] [PubMed]
- Joshi, S.M.; Gogate, P.R. Intensification of Industrial Wastewater Treatment Using Hydrodynamic Cavitation Combined with Advanced Oxidation at Operating Capacity of 70 L. Ultrason. Sonochem. 2019, 52, 375–381. [Google Scholar] [CrossRef] [PubMed]
- Thanekar, P.; Gogate, P.R.; Znak, Z.; Sukhatskiy, Y.; Mnykh, R. Degradation of Benzene Present in Wastewater Using Hydrodynamic Cavitation in Combination with Air. Ultrason. Sonochem. 2021, 70, 105296. [Google Scholar] [CrossRef]
- Yi, C.; Lu, Q.; Wang, Y.; Wang, Y.; Yang, B. Degradation of Organic Wastewater by Hydrodynamic Cavitation Combined with Acoustic Cavitation. Ultrason. Sonochem. 2018, 43, 156–165. [Google Scholar] [CrossRef]
- Kovačič, A.; Škufca, D.; Zupanc, M.; Gostiša, J.; Bizjan, B.; Krištofelc, N.; Dolenc, M.S.; Heath, E. The Removal of Bisphenols and Other Contaminants of Emerging Concern by Hydrodynamic Cavitation: From Lab-Scale to Pilot-Scale. Sci. Total Environ. 2020, 743, 140724. [Google Scholar] [CrossRef]
- Wang, J.; Chen, H.; Yuan, R.; Wang, F.; Ma, F.; Zhou, B. Intensified Degradation of Textile Wastewater Using a Novel Treatment of Hydrodynamic Cavitation with the Combination of Ozone. J. Environ. Chem. Eng. 2020, 8, 103959. [Google Scholar] [CrossRef]
- Abramov, V.O.; Abramova, A.V.; Cravotto, G.; Nikonov, R.V.; Fedulov, I.S.; Ivanov, V.K. Flow-Mode Water Treatment under Simultaneous Hydrodynamic Cavitation and Plasma. Ultrason. Sonochem. 2021, 70, 105323. [Google Scholar] [CrossRef]
- Wang, B.; Su, H.; Zhang, B. Hydrodynamic Cavitation as a Promising Route for Wastewater Treatment—A Review. Chem. Eng. J. 2021, 412, 128685. [Google Scholar] [CrossRef]
- Terán, H.R.; Medeiros, D.R.; Sánchez, M.S.; Aline, P.C.; de Sousa Júnior, R.; da Silva, S.S.; Santos, J.C. Hydrodynamic Cavitation-Assisted Continuous Pre-Treatment of Sugarcane Bagasse for Ethanol Production: Effects of Geometric Parameters of the Cavitation Device. Ultrason. Sonochem. 2020, 63, 104931. [Google Scholar] [CrossRef] [PubMed]
- Zsirai, T.; Al-Jaml, A.K.; Qiblawey, H.; Al-Marri, M.; Ahmed, A.; Bach, S.; Watson, S.; Judd, S. Ceramic Membrane Filtration of Produced Water: Impact of Membrane Module. Sep. Purif. Technol. 2016, 165, 214–221. [Google Scholar] [CrossRef] [Green Version]
- Goswami, K.P.; Pugazhenthi, G. Treatment of Poultry Slaughterhouse Wastewater Using Tubular Microfiltration Membrane with Fly Ash as Key Precursor. J. Water Process Eng. 2020, 37, 101361. [Google Scholar] [CrossRef]
- Solmaz, A.; Işık, M. Optimization of Membrane Photobioreactor; the Effect of Hydraulic Retention Time on Biomass Production and Nutrient Removal by Mixed Microalgae Culture. Biomass Bioenergy 2020, 142, 105809. [Google Scholar] [CrossRef]
- Terán Hilares, R.; Garcia Bustos, K.A.; Sanchez Vera, F.P.; Colina Andrade, G.J.; Pacheco Tanaka, D.A. Acid Precipitation Followed by Microalgae (Chlorella vulgaris) Cultivation as a New Approach for Poultry Slaughterhouse Wastewater Treatment. Bioresour. Technol. 2021, 335, 125284. [Google Scholar] [CrossRef]
- Molinuevo-Salces, B.; Riaño, B.; Hernández, D.; Cruz García-González, M. Microalgae and Wastewater Treatment: Advantages and Disadvantages. In Microalgae Biotechnology for Development of Biofuel and Wastewater Treatment; Springer: Berlin/Heidelberg, Germany, 2019; pp. 505–533. [Google Scholar]
- Saravanan, A.; Kumar, P.S.; Varjani, S.; Jeevanantham, S.; Yaashikaa, P.R.; Thamarai, P.; Abirami, B.; George, C.S. A Review on Algal-Bacterial Symbiotic System for Effective Treatment of Wastewater. Chemosphere 2021, 271, 129540. [Google Scholar] [CrossRef]
- Salgueiro, J.L.; Pérez, L.; Maceiras, R.; Sánchez, Á.; Cancela, Á. Semicontinuous Culture of Chlorella Vulgaris Microalgae for Wastewater Treatment. Int. J. Environ. Res. 2018, 12, 765–772. [Google Scholar] [CrossRef]
- Gao, F.; Li, C.; Yang, Z.-H.; Zeng, G.-M.; Feng, L.-J.; Liu, J.; Liu, M.; Cai, H. Continuous Microalgae Cultivation in Aquaculture Wastewater by a Membrane Photobioreactor for Biomass Production and Nutrients Removal. Ecol. Eng. 2016, 92, 55–61. [Google Scholar] [CrossRef]
- De Mendonça, H.V.; Otenio, M.H.; Marchão, L.; Lomeu, A.; de Souza, D.S.; Reis, A. Biofuel Recovery from Microalgae Biomass Grown in Dairy Wastewater Treated with Activated Sludge: The next Step in Sustainable Production. Sci. Total Environ. 2022, 824, 153838. [Google Scholar] [CrossRef]
- American Public Health Association; WPCF, A.; Greenberg, W.E.F.I.A.E.; Clesceri, L.S.; Eaton, A. Standard Methods for the Examination of Water and Wastewater; American Public Health Association: Denver, CO, USA, 2017; Volume 2. [Google Scholar]
- Mali, C.R.; Patwardhan, A.W.; Pandey, G.K.; Banerjee, I.; Vinod, V. CFD Study on the Effect of Various Geometrical Parameters of Honeycomb Type Orifices on Pressure Drop and Cavitation Characteristics. Nucl. Eng. Des. 2020, 370, 110880. [Google Scholar] [CrossRef]
- Musa, M.A.; Idrus, S.; Che Man, H.; Nik Daud, N.N. Performance Comparison of Conventional and Modified Upflow Anaerobic Sludge Blanket (UASB) Reactors Treating High-Strength Cattle Slaughterhouse Wastewater. Water 2019, 11, 806. [Google Scholar] [CrossRef] [Green Version]
- Wu, P.F.; Mittal, G.S. Characterization of Provincially Inspected Slaughterhouse Wastewater in Ontario, Canada. Can. Biosyst. Eng. 2012, 54, 9–18. [Google Scholar] [CrossRef]
- Ziara, R.M.M.; Li, S.; Subbiah, J.; Dvorak, B.I. Characterization of Wastewater in Two US Cattle Slaughterhouses. Water Environ. Res. 2018, 90, 851–863. [Google Scholar] [CrossRef] [PubMed]
- Osman, H.; Sedrak, M.; Hosseini, S.H.; Elsayed, K. A Comprehensive Study of Hole-to-Hole Interaction for Multi-Hole Orifice (MHO) in Hydrodynamic Cavitation Process. Flow Meas. Instrum. 2022, 85, 102163. [Google Scholar] [CrossRef]
- Das, S.; Bhat, A.P.; Gogate, P.R. Degradation of Dyes Using Hydrodynamic Cavitation: Process Overview and Cost Estimation. J. Water Process Eng. 2021, 42, 102126. [Google Scholar] [CrossRef]
- Gore, M.M.; Saharan, V.K.; Pinjari, D.V.; Chavan, P.V.; Pandit, A.B. Degradation of Reactive Orange 4 Dye Using Hydrodynamic Cavitation Based Hybrid Techniques. Ultrason. Sonochem. 2014, 21, 1075–1082. [Google Scholar] [CrossRef]
- Bagal, M.V.; Gogate, P.R. Wastewater Treatment Using Hybrid Treatment Schemes Based on Cavitation and Fenton Chemistry: A Review. Ultrason. Sonochem. 2014, 21, 1–14. [Google Scholar] [CrossRef]
- Innocenzi, V.; Prisciandaro, M.; Centofanti, M.; Vegliò, F. Comparison of Performances of Hydrodynamic Cavitation in Combined Treatments Based on Hybrid Induced Advanced Fenton Process for Degradation of Azo-Dyes. J. Environ. Chem. Eng. 2019, 7, 103171. [Google Scholar] [CrossRef]
- Zampeta, C.; Bertaki, K.; Triantaphyllidou, I.-E.; Frontistis, Z.; Vayenas, D.V. Treatment of Real Industrial-Grade Dye Solutions and Printing Ink Wastewater Using a Novel Pilot-Scale Hydrodynamic Cavitation Reactor. J. Environ. Manag. 2021, 297, 113301. [Google Scholar] [CrossRef]
- Doltade, S.B.; Dastane, G.G.; Jadhav, N.L.; Pandit, A.B.; Pinjari, D.V.; Somkuwar, N.; Paswan, R. Hydrodynamic Cavitation as an Imperative Technology for the Treatment of Petroleum Refinery Effluent. J. Water Process Eng. 2019, 29, 100768. [Google Scholar] [CrossRef]
- Rajoriya, S.; Bargole, S.; Saharan, V.K. Degradation of Reactive Blue 13 Using Hydrodynamic Cavitation: Effect of Geometrical Parameters and Different Oxidizing Additives. Ultrason. Sonochem. 2017, 37, 192–202. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Xuan, X.; Ji, L.; Chen, S.; Liu, J.; Zhao, S.; Park, S.; Yoon, J.Y.; Om, A.S. A Novel Continuous Hydrodynamic Cavitation Technology for the Inactivation of Pathogens in Milk. Ultrason. Sonochem. 2021, 71, 105382. [Google Scholar] [CrossRef] [PubMed]
- Salierno, G.; Napoleone, S.; Maisterrena, M.A.; Cassanello, M.; Pellasio, M.; Doumic, L.; Ayude, M.A. Continuous Heterogeneous Fenton-Type Process for Dye Pollution Abatement Intensified by Hydrodynamic Cavitation. Ind. Eng. Chem. Res. 2021, 60, 16653–16664. [Google Scholar] [CrossRef]
- Kumar, R.V.; Goswami, L.; Pakshirajan, K.; Pugazhenthi, G. Dairy Wastewater Treatment Using a Novel Low Cost Tubular Ceramic Membrane and Membrane Fouling Mechanism Using Pore Blocking Models. J. Water Process Eng. 2016, 13, 168–175. [Google Scholar] [CrossRef]
- IFC. General Health and Safety Guidelines. Available online: https://www.ifc.org/wps/wcm/connect/topics_ext_content/ifc_external_corporate_site/sustainability-at-ifc/publications/publications_policy_ehs-general (accessed on 26 November 2021).
- FAO. Council Directive of 21 May 1991 Concerning Urban Waste Water Treatment (91/271/EEC). Available online: https://www.ecolex.org/details/legislation/council-directive-91271eec-concerning-urban-waste-water-treatment-lex-faoc013224/ (accessed on 10 February 2022).
- Gutiérrez-Mosquera, L.F.; Arias-Giraldo, S.; Zuluaga-Meza, A. Landfill Leachate Treatment Using Hydrodynamic Cavitation: Exploratory Evaluation. Heliyon 2022, 8, e09019. [Google Scholar] [CrossRef]
- Bhat, A.P.; Gogate, P.R. Cavitation-Based Pre-Treatment of Wastewater and Waste Sludge for Improvement in the Performance of Biological Processes: A Review. J. Environ. Chem. Eng. 2021, 9, 104743. [Google Scholar] [CrossRef]
- Vadiveloo, A.; Foster, L.; Kwambai, C.; Bahri, P.A.; Moheimani, N.R. Microalgae Cultivation for the Treatment of Anaerobically Digested Municipal Centrate (ADMC) and Anaerobically Digested Abattoir Effluent (ADAE). Sci. Total Environ. 2021, 775, 145853. [Google Scholar] [CrossRef]
- Spennati, E.; Mirizadeh, S.; Casazza, A.A.; Solisio, C.; Converti, A. Chlorella Vulgaris and Arthrospira Platensis Growth in a Continuous Membrane Photobioreactor Using Industrial Winery Wastewater. Algal Res. 2021, 60, 102519. [Google Scholar] [CrossRef]
- Bhatia, S.K.; Mehariya, S.; Bhatia, R.K.; Kumar, M.; Pugazhendhi, A.; Awasthi, M.K.; Atabani, A.E.; Kumar, G.; Kim, W.; Seo, S.-O.; et al. Wastewater Based Microalgal Biorefinery for Bioenergy Production: Progress and Challenges. Sci. Total Environ. 2021, 751, 141599. [Google Scholar] [CrossRef]



| Parameters | Unit | Cattle Slaughterhouse Wastewater (SCW) |
|---|---|---|
| True color | Pt-Co | 2175 ± 109 |
| Turbidity | NTU | 264 ± 5 |
| Total alkalinity | mg CaCO3/L | 1128 ± 62 |
| Conductivity | µS/cm | 3780 ± 255 |
| Ammoniacal nitrogen | mg NH3-N/L | 125 ± 9 |
| Total Kjeldahl nitrogen | mg/L | 296 ± 5 |
| Total phosphorous | mg P-PO4/L | 22 ± 0.4 |
| Biochemical oxygen demand (BOD) | mg/L | 1000 ± 48 |
| Chemical oxygen demand (COD) | mg O2/L | 3020 ± 20 |
| Oil and fat | mg/L | 24 ± 2 |
| Chloride | mg/L | 329 ± 2 |
| Nitrate | mg/L | 0.9 ± 0.1 |
| Parameters | Unit | Untreated Sample (Diluted) | After HC Treatment | After Membrane (600 nm) | Total Removal (%) *** | ||
|---|---|---|---|---|---|---|---|
| Value | Removal * (%) | Value | Removal ** (%) | ||||
| True color | UC | 544 ± 109 | 98.1 ± 19.6 | 82 | 48.0 ± 9.6 | 51 | 92 |
| Turbidity | NTU | 66.0 ± 5.3 | 14.8 ± 1.2 | 78 | 8.2 ± 0.7 | 45 | 88 |
| Total alkalinity | mg CaCO3/L | 282 ± 62 | 220 ± 49 | 22 | 211 ± 47 | 4 | 25 |
| Conductivity | µS/cm | 945 ± 255 | 883 ± 238 | 7 | 820 ± 221 | 7 | 13 |
| Ammoniacal nitrogen | mg NH3-N/L | 31.3 ± 8.5 | 40.0 ± 10.8 | - | 17.5 ± 4.7 | 56 | 44 |
| Total Kjeldahl nitrogen | mg/L | 74.0 ± 0.0 | 24.0 ± 0.0 | 68 | 15.0 ± 0.0 | 38 | 80 |
| Total phosphorous | mg P/L | 5.4 ± 0.4 | 3.6 ± 0.3 | 33 | 2.8 ± 0.2 | 22 | 48 |
| Biochemical oxygen demand (BOD) | mg/L | 250 ± 48 | 103 ± 20 | 59 | 82 ± 16 | 21 | 67 |
| Chemical oxygen demand (COD) | mg O2/L | 755 ± 20 | 229 ± 14 | 70 | 157 ± 13 | 31 | 79 |
| Oil and fat | mg/L | 5.9 ± 1.7 | 1.0 ± 0.3 | 83 | 1.0 ± 0.3 | 0 | 83 |
| Chloride | mg/L | 82.4 ± 2.1 | 85.9 ± 2.1 | ≈0 | 84.2 ± 2.1 | ≈0 | ≈0 |
| Nitrate | mg/L | 0.2 ± 0.1 | <0.1 | <0.1 | - | - | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Terán Hilares, R.; Sánchez Vera, F.P.; Colina Andrade, G.J.; Tejada Meza, K.; García, J.C.; Pacheco Tanaka, D.A. Continuous Cultivation of Microalgae in Cattle Slaughterhouse Wastewater Treated with Hydrodynamic Cavitation. Water 2022, 14, 1288. https://doi.org/10.3390/w14081288
Terán Hilares R, Sánchez Vera FP, Colina Andrade GJ, Tejada Meza K, García JC, Pacheco Tanaka DA. Continuous Cultivation of Microalgae in Cattle Slaughterhouse Wastewater Treated with Hydrodynamic Cavitation. Water. 2022; 14(8):1288. https://doi.org/10.3390/w14081288
Chicago/Turabian StyleTerán Hilares, Ruly, Fabio P. Sánchez Vera, Gilberto J. Colina Andrade, Kevin Tejada Meza, Jaime Cárdenas García, and David Alfredo Pacheco Tanaka. 2022. "Continuous Cultivation of Microalgae in Cattle Slaughterhouse Wastewater Treated with Hydrodynamic Cavitation" Water 14, no. 8: 1288. https://doi.org/10.3390/w14081288
APA StyleTerán Hilares, R., Sánchez Vera, F. P., Colina Andrade, G. J., Tejada Meza, K., García, J. C., & Pacheco Tanaka, D. A. (2022). Continuous Cultivation of Microalgae in Cattle Slaughterhouse Wastewater Treated with Hydrodynamic Cavitation. Water, 14(8), 1288. https://doi.org/10.3390/w14081288