Manganese Pollution in Mining-Influenced Rivers and Lakes: Current State and Forecast under Climate Change in the Russian Arctic
Abstract
1. Introduction
2. Materials and Methods
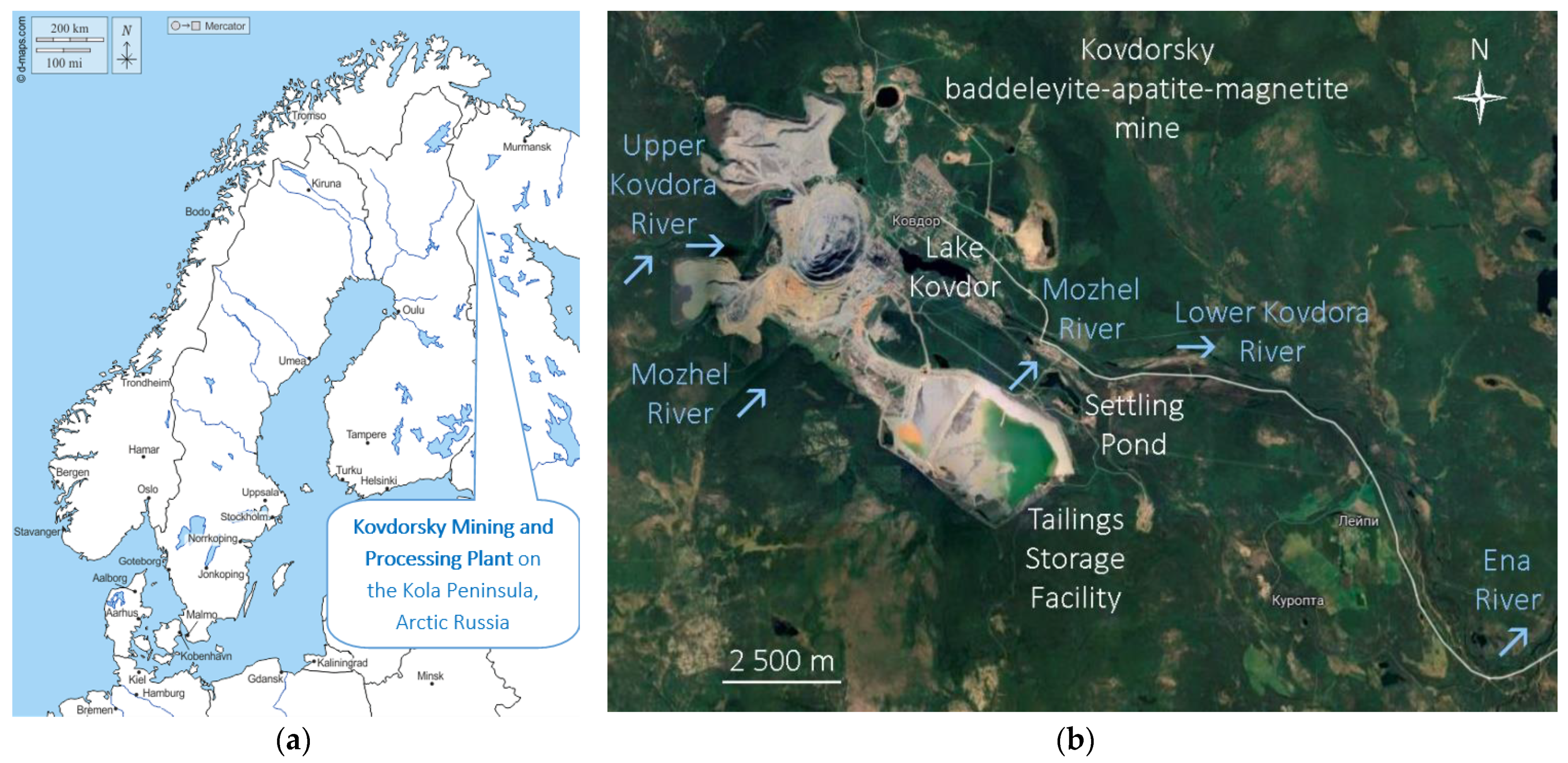
2.1. Study Area
2.2. Sampling and Analysis of the Background and Polluted Water
- Discharge No. 1 annually releases 3.5 million m3 of the quarry waters from the western water-reducing wells into the Upper Kovdora River;
- Discharge No. 2 annually releases 4.0 million m3 of the quarry waters from the northern water-reducing wells into the drainage channel of the Upper Kovdora River;
- Discharge No. 3 annually releases 25.1 million m3 of the quarry waters, treated industrial water from the power plant and storm sewer of the industrial site, from the pit water sump into Lake Kovdor.
2.3. Sampling and Analysis of Bottom Sediments
3. Results and Discussion
3.1. Pollution of Aquatic Ecosystems
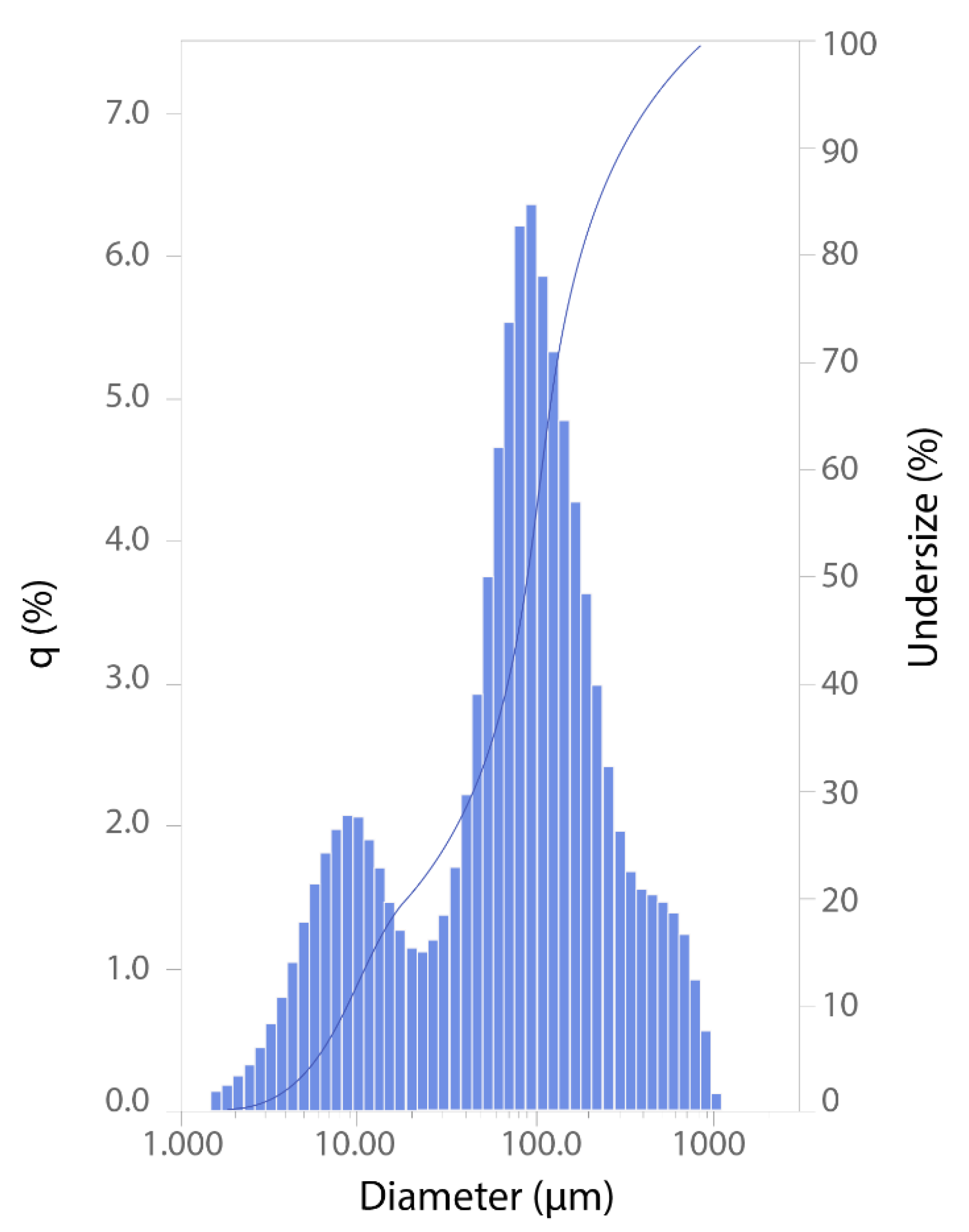
3.2. Pollution of Bottom Sediments
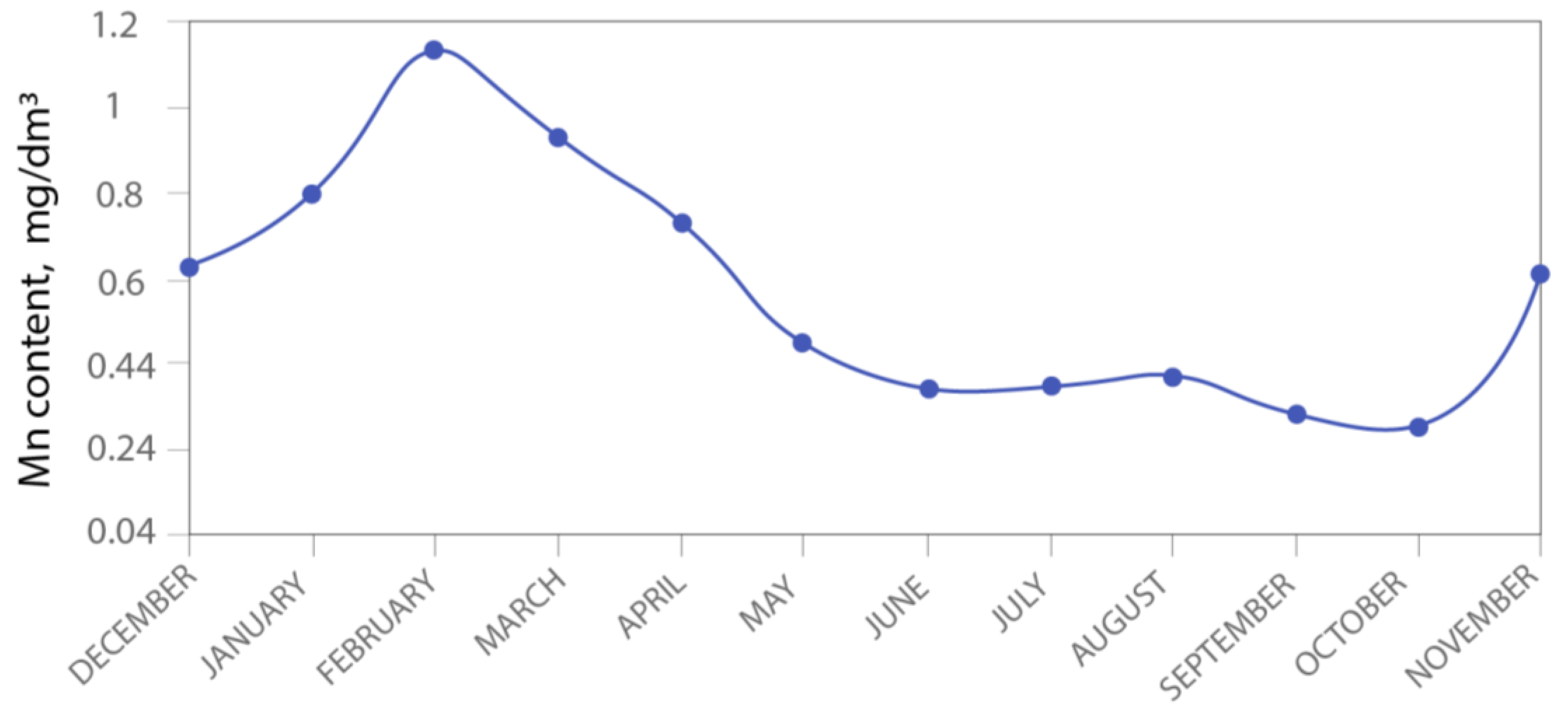
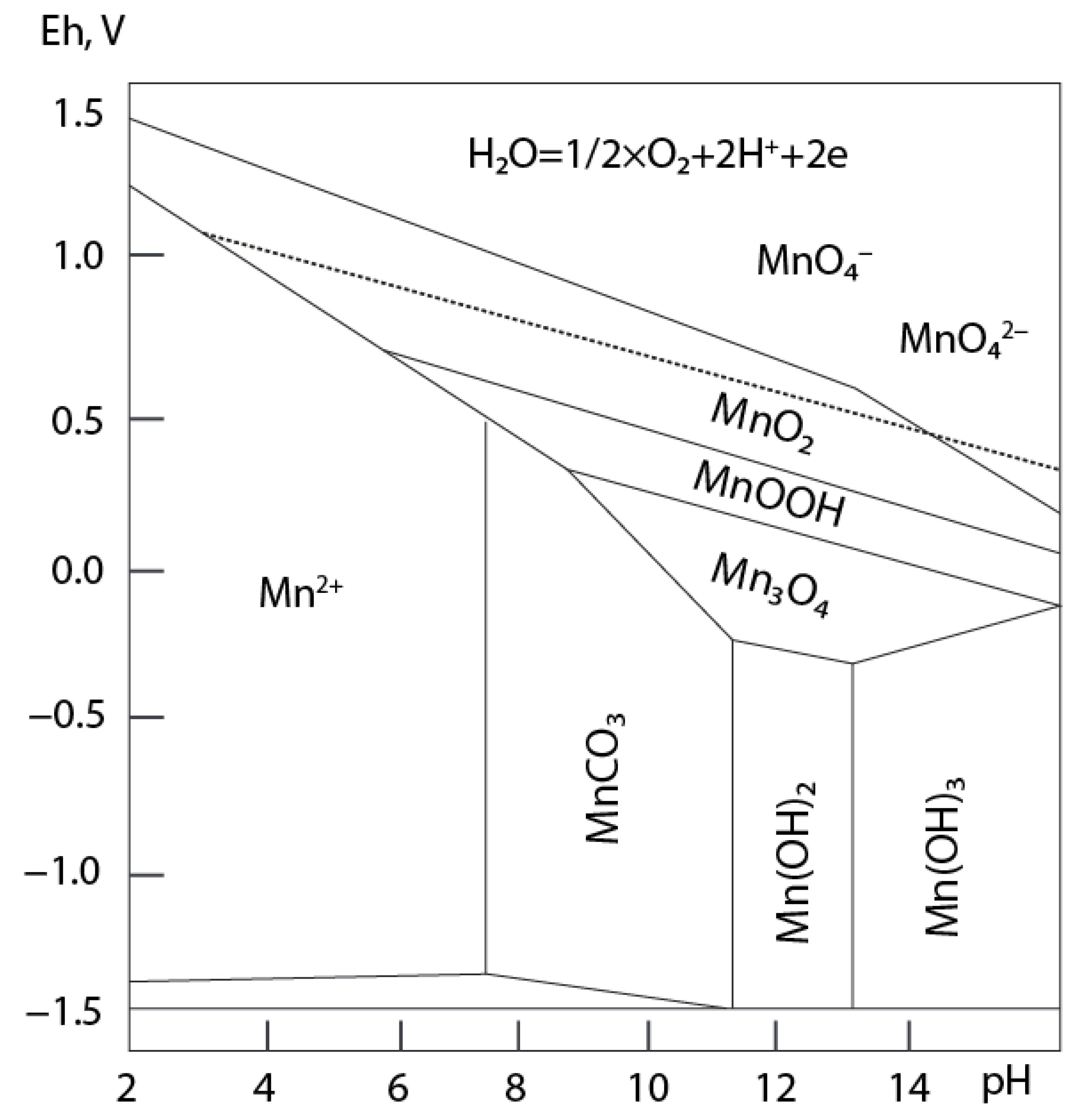
3.3. Migration and Accumulation of Mn in Mining-Influenced Aquatic Ecosystems
3.4. Potential Hazard and Fate of Mn in the Aquatic Ecosystems under Climate Change
- Pollutant accumulation in water bodies increasingly reaches toxic levels, with negative impacts on aquatic ecosystems;
- Changes in the trophic status of aquatic ecosystems and an increase in the rate of eutrophication, especially in case of relatively warm wastewater discharge;
- Changes in the direction and speed of plant succession;
- The reduction of the ecosystem stability and increasing risks of irreversible degradation.
- Formation of insoluble compounds from the wastewater of the mining enterprise as a result of interaction with mineral salts;
- Formation of insoluble complexes and sorption of Mn on organic components of suspended solids and bottom sediments. Taking into account the increase in water flow from the catchment area, the mass of organic matter in water bodies will increase significantly;
- Formation of soluble organic and inorganic complex Mn compounds with the subsequent pollutant migration.
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hülsmann, S.; Sušnik, J.; Rinke, K.; Langan, S.; van Wijk, D.; Janssen, A.B.; Mooij, W.M. Integrated modelling and management of water resources: The ecosystem perspective on the nexus approach. Curr. Opin. Environ. Sustain. 2019, 40, 14–20. [Google Scholar] [CrossRef]
- Alexakis, D.E. Linking DPSIR Model and Water Quality Indices to Achieve Sustainable Development Goals in Groundwater Resources. Hydrology 2021, 8, 90. [Google Scholar] [CrossRef]
- Culp, J.M.; Brua, R.B.; Luiker, E.; Glozier, N.E. Ecological causal assessment of benthic condition in the oil sands region, Athabasca River, Canada. Sci. Total Environ. 2020, 749, 141393. [Google Scholar] [CrossRef]
- Golovina, E.I. Strategic issues of groundwater extraction management in Russia. J. Ecol. Eng. 2017, 18, 13–21. [Google Scholar] [CrossRef][Green Version]
- Kuznetsov, V.S.; Petrov, D.S. Assessing the environmental condition of minor rivers in urban areas. J. Ecol. Eng. 2017, 18, 110–114. [Google Scholar] [CrossRef]
- Lebedeva, Y.; Kotiukov, P.; Lange, I. Study of the geo-ecological state of groundwater of metropolitan areas under the conditions of intensive contamination thereof. J. Ecol. Eng. 2020, 21, 157–165. [Google Scholar] [CrossRef]
- Mello, K.D.; Taniwaki, R.H.; Paula, F.R.D.; Rodrigues, C.B.; Hughes, R.M. Multiscale land use impacts on water quality: Assessment, planning, and future perspectives in Brazil. J. Environ. Manag. 2020, 270, 110879. [Google Scholar] [CrossRef] [PubMed]
- Michalik, B.; Wysocka, M.; Bonczyk, M.; Samolej, K.; Chmielewska, I. Long term behaviour of radium rich deposits in a lake ecosystem. J. Environ. Radioact. 2020, 222, 106349. [Google Scholar] [CrossRef]
- Bauer, A.E.; Hewitt, L.M.; Parrott, J.L.; Dixon, D.G.; Frank, R.A. The toxicity of organic fractions from aged oil sands process-affected water to aquatic species. Sci. Total Environ. 2019, 669, 702–710. [Google Scholar] [CrossRef] [PubMed]
- De Biase, M.; Chidichimo, F.; Maiolo, M.; Micallef, A. The Impact of Predicted Climate Change on Groundwater Resources in a Mediterranean Archipelago: A Modelling Study of the Maltese Islands. Water 2021, 13, 3046. [Google Scholar] [CrossRef]
- Monaci, F.; Ancora, S.; Bianchi, N.; Paoli, L.; Loppi, S. Combined use of native and transplanted moss for post-mining characterization of metal(loid) river contamination. Sci. Total Environ. 2021, 750, 141669. [Google Scholar] [CrossRef] [PubMed]
- Newton, A.; Icely, J.; Cristina, S.; de Lima, R.; Kuenzer, C. Anthropogenic, Direct Pressures on Coastal Wetlands. Front. Ecol. Evol. 2020, 8, 144. [Google Scholar] [CrossRef]
- Sánchez-Castro, I.; Martínez-Rodríguez, P.; Jroundi, F.; Descostes, M.; Merroun, M.L. High-efficient microbial immobilization of solved U(VI) by the Stenotrophomonas strain Br8. Water Res. 2020, 183, 116110. [Google Scholar] [CrossRef] [PubMed]
- Bazhin, V.Y.; Beloglazov, I.I.; Feshchenko, R.Y. Deep conversion and metal content of Russian coals. Eurasian Min. 2016, 2, 28–32. [Google Scholar] [CrossRef]
- Corella, J.P.; Sierra, M.J.; Garralón, A.; Saiz-Lopez, A.; Valero-Garcés, B.L. Recent and historical pollution legacy in high altitude Lake Marboré (Central Pyrenees): A record of mining and smelting since pre-Roman times in the Iberian Peninsula. Sci. Total Environ. 2021, 751, 141557. [Google Scholar] [CrossRef] [PubMed]
- Egbueri, J.C.; Enyigwe, M.T. Pollution and Ecological Risk Assessment of Potentially Toxic Elements in Natural Waters from the Ameka Metallogenic District in Southeastern Nigeria. Anal. Lett. 2020, 53, 2812–2839. [Google Scholar] [CrossRef]
- Golovanova, O.A.; Maslovskaya, E.A. Dynamics of heavy metal ion contamination of surface waters of rivers in the Siberian region. Bull. Omsk Univ. 2016, 3, 64–73. [Google Scholar]
- Khormali, A.; Sharifov, A.R.; Torba, D.I. Increasing efficiency of calcium sulfate scale prevention using a new mixture of phosphonate scale inhibitors during waterflooding. J. Pet. Sci. Eng. 2018, 164, 245–258. [Google Scholar] [CrossRef]
- Kuznetsov, V.S.; Suprun, I.K. The assessment of alumina production waste impact on natural water. J. Ecol. Eng. 2018, 19, 154–158. [Google Scholar] [CrossRef]
- Marinelli, E.; Radini, S.; Akyol, Ç.; Sgroi, M.; Eusebi, A.L.; Bischetti, G.B.; Mancini, A.; Fatone, F. Water-Energy-Food-Climate Nexus in an Integrated Peri-Urban Wastewater Treatment and Reuse System: From Theory to Practice. Sustainability 2021, 13, 10952. [Google Scholar] [CrossRef]
- Nevskaya, M.; Seleznev, S.; Masloboev, V.; Klyuchnikova, E.; Makarov, D. Involving small and medium-sized mining industry businesses in mining waste processing in the Russian Federation. Miner. Econ. 2021, 34, 81–86. [Google Scholar] [CrossRef]
- Safiullin, R.N.; Afanasyev, A.S.; Reznichenko, V.V. The concept of development of monitoring systems and management of intelligent technical complexes. J. Min. Inst. 2019, 237, 322–330. [Google Scholar] [CrossRef]
- Santana, C.S.; Montalván Olivares, D.M.; Silva, V.H.C.; Velasco, F.G.; de Jesus, R.M. Assessment of water resources pollution associated with mining activity in a semi-arid region. J. Environ. Manag. 2020, 273, 111148. [Google Scholar] [CrossRef] [PubMed]
- World Water Quality Assessment: First Global Display of a Water Quality Baseline; World Water Quality Alliance: Nairobi, Kenya, 2021.
- Nedosekin, A.O.; Rejshahrit, E.I.; Kozlovskiy, A.N. Strategic approach to assessing economic sustainability objects of mineral resources sector of Russia. J. Min. Inst. 2019, 237, 354–360. [Google Scholar] [CrossRef]
- Rikhvanov, L.P.; Soktoev, B.R.; Baranovskaya, N.V.; Ageeva, E.V.; Belyanovskaya, A.I.; Deriglazova, M.A.; Yusupov, D.V.; Epova, E.S.; Solodukhina, M.A.; Zamana, L.V.; et al. Comprehensive geochemical research of the environmental components in endemic areas of Transbaikalia. Bull. Tomsk Polytech. Univ. Geo Assets Eng. 2021, 332, 7–25. [Google Scholar] [CrossRef]
- Sizyakova, E.V.; Ivanov, P.V. Multifunctional coagulants based on hidrocarboaluminates. J. Ecol. Eng. 2017, 18, 16–20. [Google Scholar] [CrossRef][Green Version]
- Strizhenok, A.V.; Korelskiy, D.S.; Kuznetsov, V.S. The wastewater disposal system modernization during processing of amber deposit as a way to reduce the anthropogenic load on the Baltic Sea ecosystem. J. Ecol. Eng. 2019, 20, 30–35. [Google Scholar] [CrossRef]
- Yungmeister, D.A.; Sudarikov, S.M.; Kireev, K.A. Feasibility of type of deep-water technologies for the extraction of marine ferro-manganese nodules. J. Min. Inst. 2019, 235, 88–95. [Google Scholar] [CrossRef]
- Novikov, A.I.; Shirokaya, A.A.; Slukovskaya, M.V. Elemental Concentrations of Major and Trace Elements in the Spring Waters of the Arctic Region of Russia. Minerals 2022, 12, 8. [Google Scholar] [CrossRef]
- Kashulin, N.A.; Dauvalter, V.A.; Denisov, D.B.; Valkova, S.A.; Vandysh, O.I.; Terentyev, P.M.; Kashulin, A.N. Some aspects of the current state of freshwater resources of the Murmansk region. Bull. Mosc. State Tech. Univ. 2013, 16, 98–107. [Google Scholar]
- Ministry of Natural Recourses and Ecology of Russian Federation. State Report. In On the State and Protection of the Environment of the Russian Federation in 2018; Ministry of Natural Recourses and Ecology of Russian Federation: Moscow, Russia, 2019. [Google Scholar]
- Zhukovskiy, Y.; Tsvetkov, P.; Buldysko, A.; Malkova, Y.; Stoianova, A.; Koshenkova, A. Scenario Modeling of Sustainable Development of Energy Supply in the Arctic. Resources 2021, 10, 124. [Google Scholar] [CrossRef]
- Danilov, A.S.; Smirnov, Y.D.; Pashkevich, M.A. Use of biological adhesive for effective dust suppression in mining operations. J. Ecol. Eng. 2015, 16, 9–14. [Google Scholar] [CrossRef][Green Version]
- Varotsos, C.A.; Krapivin, V.F. Pollution of Arctic waters has reached a critical point: An innovative approach to this problem. Water Air Soil Pollut. 2018, 229, 343. [Google Scholar] [CrossRef]
- Moiseenko, T.I.; Dauvalter, V.A.; Lukin, A.A.; Kudryavtseva, L.P.; Ilyashchuk, B.P.; Ilyashchuk, L.I.; Sandimirov, S.S.; Kagan, L.Y.; Vandysh, O.M.; Sharova, Y.N.; et al. Anthropogenic Modifications of the Ecosystem of the Lake Imandra; Nauka: Moscow, Russia, 2002; 403p. [Google Scholar]
- Kashulin, N.A.; Dauvalter, V.A.; Kashulina, T.G.; Sandimirov, S.S.; Ratkin, N.E.; Kudryavtseva, L.P.; Koroleva, I.M.; Vandysh, O.I.; Mokrotovarova, O.I. Anthropogenic Changes in the Running Water Ecosystems of the Murmansk Oblast. Part I: Kovdorsky District; Kola Science Center RAS: Apatity, Russia, 2005; 234p. [Google Scholar]
- Mazukhina, S.I.; Sandimirov, S.S.; Koroleva, I.M. Assessment of the impact of technogenic effluents on fresh water. Ecol. Chem. 2003, 12, 97–104. [Google Scholar]
- Ivanyuk, G.Y.; Kalashnikov, A.O.; Pakhomovsky, Y.A.; Mikhailova, J.A.; Yakovenchuk, V.N.; Konopleva, N.G.; Goryainov, P.M. Economic minerals of the Kovdor baddeleyite-apatite-magnetite deposit, Russia: Mineralogy, spatial distribution and ore processing optimization. Ore Geol. Rev. 2016, 77, 279–311. [Google Scholar] [CrossRef]
- Moiseenko, T.I.; Rodyushkin, I.V.; Dauvalter, V.A.; Kudryavtseva, L.P. Formation of Water Quality and Bottom Sediments under Anthropogenic Loads on Reservoirs of the Arctic Basin (on the Example of the North Kola); Kola Science Center RAS: Apatity, Russia, 1996; 263p. [Google Scholar]
- Li, N.; Zhou, Y.; Liu, J.; Chen, Z.; Chen, D. Persistent thallium contamination in river sediments, source apportionment and environmental implications. Ecotoxicol. Environ. Saf. 2020, 202, 110874. [Google Scholar] [CrossRef] [PubMed]
- Meshref, M.N.A.; Ibrahim, M.D.; Huang, R.; Mahaffey, A.; Gamal El-Din, M. Fourier transform infrared spectroscopy as a surrogate tool for the quantification of naphthenic acids in oil sands process water and groundwater. Sci. Total Environ. 2020, 734, 139191. [Google Scholar] [CrossRef] [PubMed]
- Unnikrishnan, B.; Lien, C.-W.; Chu, H.-W.; Huang, C.-C. A review on metal nanozyme-based sensing of heavy metal ions: Challenges and future perspectives. J. Hazard. Mater. 2021, 401, 123397. [Google Scholar] [CrossRef] [PubMed]
- Kashulin, N.A.; Sandimirov, S.S.; Dauvalter, V.A.; Kudryavtseva, L.P.; Terentyev, P.M.; Denisov, D.B.; Vandysh, O.I.; Valkova, S.A. Annotated Ecological Catalog of Lakes of the Murmansk Oblast: Eastern Part (Barents Sea Basin); Kola Science Center RAS: Apatity, Russia, 2010. [Google Scholar]
- Moiseenko, T.I. Acidification of surface waters of the Kola North: Critical loads and their excess. Water Resour. 1996, 23, 200–211. [Google Scholar]
- Petrova, T.A.; Pashkevich, M.A. The state of water bodies in the zone of influence of mining and processing enterprises on the example of JSC Kovdorsky GOK. Alm. Mod. Sci. Educ. 2011, 4, 107–109. [Google Scholar]
- ISO 5667-3:2018; Water Quality—Sampling—Part 3: Preservation and Handling of Water Samples. ISO: Geneva, Switzerland, 2018.
- M-02-2406-13; Quantitative Chemical Analysis Technique for Shimadzu Spectrophotometers. Determination of Elements in Drinking, miner Al, Natural, and Waste Water and Atmospheric Precipitation by Atomic Absorption Method. Mendeleev Institute for Metrology: Saint Petersburg, Russia, 2013.
- Dauvalter, V.A.; Kashulin, N.A. Chemical composition of sediments of the Pasvik River system under conditions of global and local pollution. Her. Kola Sci. Cent. RAS 2014, 2, 106–120. [Google Scholar]
- Dauvalter, V.A.; Kashulin, N.A.; Sandimirov, S.S. Tendencies in changes of the chemical composition of sediments in freshwater Subarctic and Arctic reservoirs under the influence of natural and anthropogenic factors. Trans. Kola Sci. Cent. 2012, 2, 54–87. [Google Scholar]
- Förstner, U.; Salamons, W. Trace Metal Analyses on Polluted Sediments; Delft, The Netherlands, 1980; pp. 1–13. Available online: https://edepot.wur.nl/214350 (accessed on 8 February 2022).
- Håkanson, L. Sediment sampling in different aquatic environments: Statistical aspects. Water Res. 1984, 20, 41–46. [Google Scholar] [CrossRef]
- M-MVI-80-2008; Method for Performing Measurements of the Mass Fraction of Elements in Samples of Soils and Bottom Sediments by Methods of Atomic Emission and Atomic Absorption Spectrometry. St. Petersburg Press: St. Petersburg, Russia, 2008.
- Water Quality Standards for Water Bodies of Fishery Significance, including Standards for Maximum Permissible Concentrations of Harmful Substances; Decree No. 552, as amended on 10 March 2020; Ministry of Agriculture of the Russian Federation: Moscow, Russia, 2020.
- Maximum Permissible Concentrations and Approximately Permissible Concentrations of Chemical Substances in Soil; Sanitary rules and norms, 1.2.3685-21; Decree No. 2, 28 February 2021; Chief State Sanitary Doctor of the Russian Federation: Moscow, Russia, 2021.
- Alekseenko, V.; Alekseenko, A. The abundances of chemical elements in urban soils. J. Geochem. Explor. 2014, 147, 245–249. [Google Scholar] [CrossRef]
- MacDonald, D.D.; Ingersoll, C.G.; Berger, T.A. Development and Evaluation of Consensus-Based Sediment Quality Guidelines for Freshwater Ecosystems. Arch. Environ. Contam. Toxicol. 2000, 39, 20–31. [Google Scholar] [CrossRef] [PubMed]
- Koptsik, G.N.; Koptsik, S.V.; Smirnova, I.E.; Sinichkina, M.A. Effect of Soil Degradation and Remediation in Technogenic Barrens on the Uptake of Nutrients and Heavy Metals by Plants in the Kola Subarctic. Eurasian Soil Sci. 2021, 54, 1252–1264. [Google Scholar] [CrossRef]
- Paatero, J.; Dauvalter, V.; Derome, J.; Lehto, J.; Pasanen, J.; Vesala, T.; Miettinen, J.; Makkonen, U.; Kyrö, E.-M.; Jernström, J.; et al. Effects of Kola Air Pollution on the Environment in the Western Part of the Kola Peninsula and Finnish Lapland–Final Report; Finnish Meteorological Institute: Helsinki, Finland, 2008; Available online: https:/helda.helsinki.fi/bitstream/handle/10138/1202/2008nro6.pdf (accessed on 10 February 2022).
- Slukovskaya, M.V.; Vasenev, V.I.; Ivashchenko, K.V.; Morev, D.V.; Drogobuzhskaya, S.V.; Ivanova, L.A.; Kremenetskaya, I.P. Technosols on mining wastes in the Subarctic: Efficiency of remediation under Cu-Ni atmospheric pollution. Int. Soil Water Conserv. Res. 2019, 7, 297–307. [Google Scholar] [CrossRef]
- Barkan, V.S.; Lyanguzova, I.V. Changes in the Degree of Contamination of Organic Horizons of Al–Fe-Humus Podzols upon a Decrease in Aerotechnogenic Loads, the Kola Peninsula. Eurasian Soil Sci. 2018, 51, 327–335. [Google Scholar] [CrossRef]
- Moiseenko, T.I.; Dinu, M.I.; Bazova, M.M.; de Wit, H.A. Long-Term Changes in the Water Chemistry of Arctic Lakes as a Response to Reduction of Air Pollution: Case Study in the Kola, Russia. Water Air Soil Pollut. 2015, 226, 98. [Google Scholar] [CrossRef]
- Khalifa, A.A.; Bazhin, V.Y.; Ustinova, Y.V.; Shalabi, M.E.H. Research on the reduction of iron oxide from red mud pellets using coke. Obogashchenie Rud 2021, 4, 46–51. [Google Scholar] [CrossRef]
- Bamforth, S.M.; Manning, D.A.; Singleton, I.; Younger, P.L.; Johnson, K.L. Manganese removal from mine waters—Investigating the occurrence and importance of manganese carbonates. Appl. Geochem. 2006, 21, 1274–1287. [Google Scholar] [CrossRef]
- Schaller, T.; Wehrli, B. Geochemical-focusing of manganese in lake sediments—An indicator of deep-water oxygen conditions. Aquat. Geochem. 1996, 2, 359–378. [Google Scholar] [CrossRef]
- Shackleton, R.T. Loss of land and livelihoods from mining operations: A case in the Limpopo Province, South Africa. Land Use Policy 2020, 99, 104825. [Google Scholar] [CrossRef]
- Spellman, C.D.; Tasker, T.L.; Strosnider, W.H.J.; Goodwill, J.E. Abatement of circumneutral mine drainage by Co-treatment with secondary municipal wastewater. J. Environ. Manag. 2020, 271, 110982. [Google Scholar] [CrossRef] [PubMed]
- Zdravković, A.; Cvetković, V.; Šarić, K.; Rosić, A.; Erić, S. Waste rocks and medieval slag as sources of environmental pollution in the area of the Pb-Zn Mine Rudnik (Serbia). J. Geochem. Explor. 2020, 218, 106629. [Google Scholar] [CrossRef]
- Ingri, J.; Pekka, L.; Dauvalter, V.; Rodushkin, I.; Peinerud, E. Manganese redox cycling in Lake Imandra: Impact on nitrogen and the trace metal sediment record. Biogeosciences Discuss. 2011, 8, 273–321. [Google Scholar] [CrossRef]
- Kim, Y. Geochemical Behavior of Potentially Toxic Elements in Riverbank-Deposited Weathered Tailings and Their Environmental Effects: Weathering of Pyrite and Manganese Pyroxene. Minerals 2020, 10, 413. [Google Scholar] [CrossRef]
- El-Jaoual, T.; Cox, D.A. Manganese toxicity in plants. J. Plant Nutr. 1998, 21, 353–386. [Google Scholar] [CrossRef]
- Crossgrove, J.; Zheng, W. Manganese toxicity upon overexposure. NMR Biomed. Int. J. Devoted Dev. Appl. Magn. Reson. Vivo 2004, 17, 544–553. [Google Scholar] [CrossRef] [PubMed]
- Frisbie, S.H.; Mitchell, E.J.; Dustin, H.; Maynard, D.M.; Sarkar, B. World Health Organization discontinues its drinking-water guideline for manganese. Environ. Health Perspect. 2012, 120, 775–778. [Google Scholar] [CrossRef]
- Goss, M.J.; Carvalho, M.J.G.P.R.; Cosimini, V.; Fearnhead, M.L. An approach to the identification of potentially toxic concentrations of manganese in soils. Soil Use Manag. 1992, 8, 40–43. [Google Scholar] [CrossRef]
- Luo, X.; Ren, B.; Hursthouse, A.S.; Thacker, J.R.M.; Wang, Z. Soil from an Abandoned Manganese Mining Area (Hunan, China): Significance of Health Risk from Potentially Toxic Element Pollution and Its Spatial Context. Int. J. Environ. Res. Public Health 2020, 17, 6554. [Google Scholar] [CrossRef]
- Semenkov, I.N.; Koroleva, T.V. Guideline Values for the Content of Chemical Elements in Soils of Urban Functional Zones: A Review. Eurasian Soil Sci. 2022, 55, 81–89. [Google Scholar] [CrossRef]
- Ljung, K.; Vahter, M. Time to re-evaluate the guideline value for manganese in drinking water? Environ. Health Perspect. 2007, 115, 1533–1538. [Google Scholar] [CrossRef]
- World Health Organization. Manganese in Drinking Water: Background Document for Development of WHO Guidelines for Drinking-Water Quality; World Health Organization: Geneva, Switzerland, 2021. [Google Scholar]
- Alekseenko, V.A.; Shvydkaya, N.V.; Puzanov, A.V.; Nastavkin, A.V. Landscape monitoring studies of the North Caucasian geochemical province. J. Min. Inst. 2020, 243, 371–378. [Google Scholar] [CrossRef]
- Batbayar, G.; Pfeiffer, M.; von Tümpling, W.; Kappas, M.; Karthe, D. Chemical water quality gradients of the sub catchments of the Mongolian Selenga River basin. Env. Monit Assess 2017, 189, 420. [Google Scholar] [CrossRef]
- Luo, X.; Ren, B.; Hursthouse, A.S.; Jiang, F.; Deng, R. Potentially toxic elements (PTEs) in crops, soil, and water near Xiangtan manganese mine, China: Potential risk to health in the foodchain. Env. Geochem Health 2020, 42, 1965–1976. [Google Scholar] [CrossRef] [PubMed]
- Lucchini, R.G.; Aschner, M.; Kim, Y. Manganese. In Handbook on the Toxicology of Metals, 5th ed.; Nordberg, G., Costa, M., Eds.; Academic Press: Cambridge, MA, USA, 2022; pp. 501–538. [Google Scholar] [CrossRef]
- Brown, M.T.; Foos, B. Assessing children’s exposures and risks to drinking water contaminants: A manganese case study. Hum. Ecol. Risk Assess. 2009, 15, 923–947. [Google Scholar] [CrossRef]
- O’Neal, S.L.; Zheng, W. Manganese toxicity upon overexposure: A decade in review. Curr. Environ. Health Rep. 2015, 2, 315–328. [Google Scholar] [CrossRef]
- Nakhshina, E.P. Trace Elements in the Reservoirs of the Dnieper; Naukova Dumka: Kiev, Ukraine, 1983; 160p. [Google Scholar]
- Takeno, N. Atlas of Eh-pH Diagrams; Geological Survey of Japan Open File Report; Academia: San Francisco, CA, USA, 2005; 102p. [Google Scholar]
- Yudovich, Y.E.; Ketris, M.P. Geochemistry of Manganese; Geoprint: Syktyvkar, Russia, 2014; 538p. [Google Scholar]
- Listova, L.P. Physico-Chemical Analysis of Formation of the Oxide and Carbonate Ores of Manganese; Academy of Sciences of the Soviet Union: Moscow, Russia, 1961; 119p. [Google Scholar]
- Gilkes, R.J.; McKenzie, R.M. Geochemistry and mineralogy of manganese in soils. In Manganese in Soils and Plants; Graham, R.D., Hannam, R.J., Uren, N.C., Eds.; Springer: Dordrecht, The Netherlands, 1988; pp. 23–35. [Google Scholar]
- Grigorieva, T.N.; Dubinina, G.A.; Moroz, T.N.; Palchik, N.A. Mineral composition of Mn oxides synthesized with the participation of microorganisms. Pac. Geol. 1993, 17, 59–64. [Google Scholar]
- Pyrolusite: Mineral Information, Data and Localities. Available online: https://www.mindat.org/min-3318.html (accessed on 25 January 2022).
- Herndon, E.M.; Havig, J.R.; Singer, D.M.; McCormick, M.L.; Kump, L.R. Manganese and iron geochemistry in sediments underlying the redox-stratified Fayetteville Green Lake. Geochim. Cosmochim. Acta 2018, 231, 50–63. [Google Scholar] [CrossRef]
- Nakhshina, E.P. Manganese in freshwaters. Hydrobiol. J. 1975, 11, 98–115. [Google Scholar]
- Tityunova, F.I. Hydrogeochemistry of Technogenesis; Nauka: Moscow, Russia, 1987; 335p. [Google Scholar]
- Manganite: Mineral Information, Data and Localities. Available online: https://www.mindat.org/min-2519.html (accessed on 25 January 2022).
- Birnessite: Mineral Information, Data and Localities. Available online: https://www.mindat.org/min-680.html (accessed on 25 January 2022).
- Vernadite: Mineral Information, Data and Localities. Available online: https://www.mindat.org/min-4171.html (accessed on 25 January 2022).
- Makarov, D.; Svetlov, A.; Goryachev, A.; Masloboev, V.; Minenko, V.; Samusev, A.; Krasavtseva, E. Mine Waters of the Mining Enterprises of the Murmansk Region: Main Pollutants, Perspective Treatment Technologies. In Mine Water–Technological and Ecological Challenges; Khayrulina, E., Wolkersdorfer, C., Polyakova, S., Bogush, A., Eds.; Perm State University: Perm, Russia, 2019; pp. 206–211. [Google Scholar]
- Moiseenko, T.I.; Morgunov, B.A.; Gashkina, N.A.; Megorskiy, V.V.; Pesiakova, A.A. Ecosystem and human health assessment in relation to aquatic environment pollution by heavy metals: Case study of the Murmansk region, northwest of the Kola Peninsula, Russia. Environ. Res. Lett. 2018, 13, 065005. [Google Scholar] [CrossRef]
- Moiseenko, T.I.; Dinu, M.I.; Gashkina, N.A.; Kremleva, T.A.; Khoroshavin, V.Y. Geochemical features of elements distributions in the lake waters of the Arctic region. Geochem. Intern. 2020, 58, 613–623. [Google Scholar] [CrossRef]
- Jarsjö, J.; Andersson-Skold, Y.; Froberg, M.; Pietron, J.; Borgstrom, R.; Lov, A.; Kleja, D.B. Projecting impacts of climate change on metal mobilization at contaminated sites: Controls by the groundwater level. Sci. Total Environ. 2020, 712, 135560. [Google Scholar] [CrossRef]
- North: Further Warming or New Ice Age? Available online: https://ria.ru/20140926/1025725993.html (accessed on 25 January 2022).
- Avellan, C.T.; Ardakanian, R.; Gremillion, P. The Role of Constructed Wetlands for Biomass Production within the Water-Soil-Waste Nexus. Water Sci. Technol. 2017, 75, 2237–2245. [Google Scholar] [CrossRef] [PubMed]
- Vasilenko, A.N.; Magritskiy, D.V.; Frolova, N.L. Patterns of changes in average annual water temperature of the rivers of the arctic zone of Russia associated with climate change. Water Sect. Russ. 2020, 2, 8–22. [Google Scholar] [CrossRef]
- Jackson, F.L.; Fryer, R.J.; Hannah, D.M.; Millar, C.P.; Malcolm, I.A. A spatio-temporal statistical model of maximum daily river temperatures to inform the management of Scotland’s Atlantic salmon rivers under climate change. Sci. Total Environ. 2018, 612, 1543–1558. [Google Scholar] [CrossRef] [PubMed]
- Filatov, N.N.; Nazarova, L.E.; Georgiev, A.P.; Semenov, A.V.; Antsiferova, A.R.; Ozhigina, V.V.; Bogdan, M.I. Changes and climate variability in the European North of Russia and their impact on water bodies. Arct. Ecol. Econ. 2012, 2, 80–93. [Google Scholar]
- Avellán, T.; Roidt, M.; Emmer, A.; Von Koerber, J.; Schneider, P.; Raber, W. Making the Water–Soil–Waste Nexus Work: Framing the Boundaries of Resource Flows. Sustainability 2017, 9, 1881. [Google Scholar] [CrossRef]
- Benavides, L.; Avellán, T.; Caucci, S.; Hahn, A.; Kirschke, S.; Müller, A. Assessing Sustainability of Wastewater Management Systems in a Multi-Scalar, Transdisciplinary Manner in Latin America. Water 2019, 11, 249. [Google Scholar] [CrossRef]
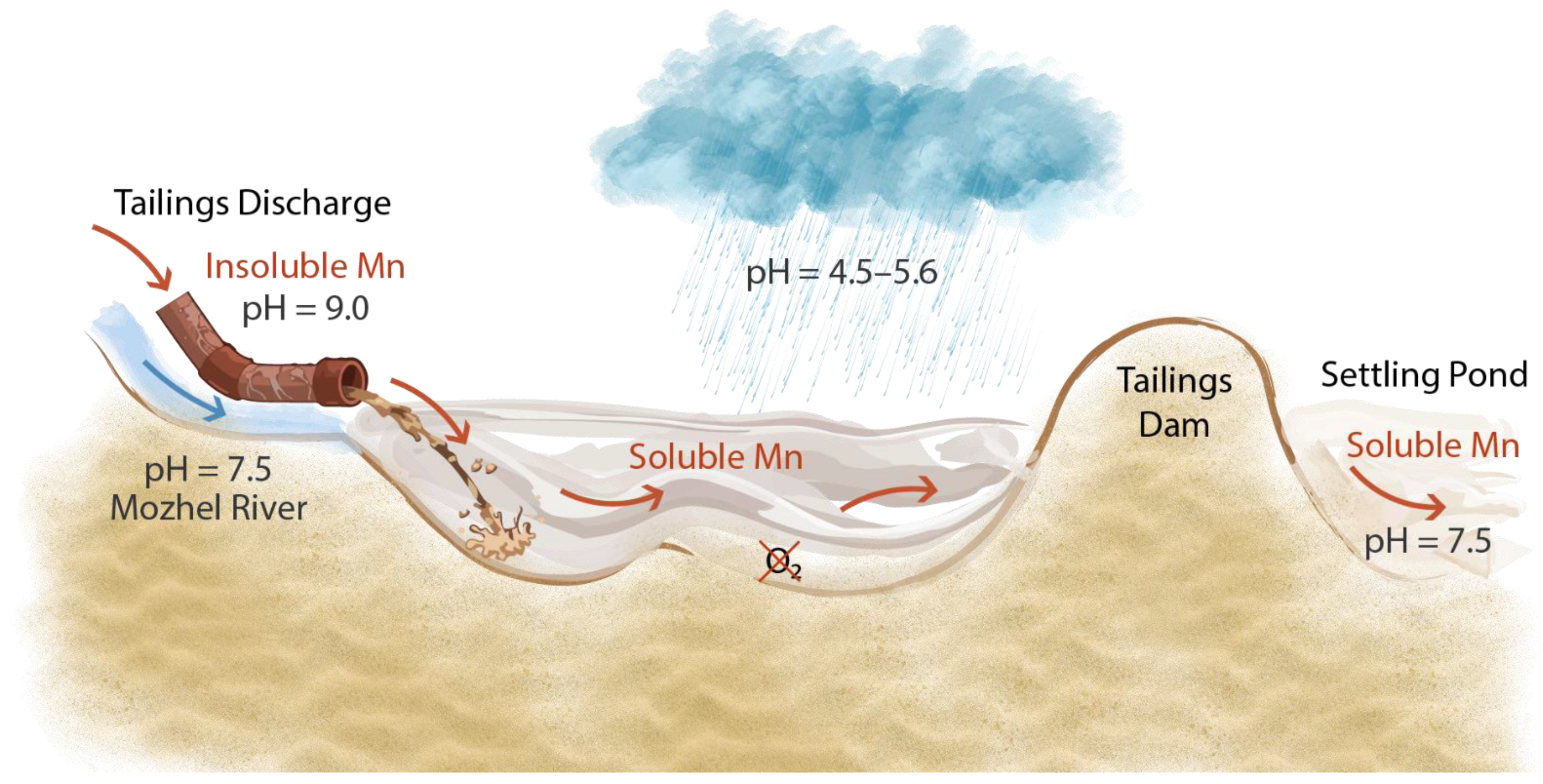
| Parameters | Upper Kovdora | Lower Kovdora | Mozhel |
|---|---|---|---|
| Length, km | 13.0 | 21.0 | 11.7 |
| Catchment area, km2 | 84.7 | 110.0 | 47.2 |
| Depth, m | 0.5–1.0 | 0.5–1.0 | 0.2–1.0 |
| Width, m | 5–25 | 20–50 | 2–8 |
| Average stream velocity, m/s | 0.50 | 0.05 | 0.91 |
| Average multi-year flow, m3/s | 0.49 | 0.90 | 0.18 |
| Element | Fe | Ca | Mg | Mn | Cu | Zn | Sr | Ni | Co | Cd | Cr |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Limit of detection in dissolved phase | mg/dm3 | µg/dm3 | |||||||||
| 0.01 | 2.0 | 0.1 | 0.005 | 0.0005 | 0.005 | 0.5 | 0.002 | 0.0025 | 0.0005 | 0.005 | |
| The relative total measurement error in dissolved phase at the confidence level of p = 0.95 (%) | 11 | 13 | 10 | 15 | 16 | 10 | 15 | 19 | 33 | 17 | 26 |
| Element | Fe | Ca | Mg | Mn | Cu | Zn | Sr | Ni | Co | Cd | Cr |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Measurement Units | mg/dm3 | µg/dm3 | |||||||||
| Russian surface water standard [54] | |||||||||||
| 0.05 | 180.0 | 40.0 | 0.01 for Mn2+ | 0.001 | 0.01 | 0.4 | 10.0 | 10.0 | 5.0 | 20.0 for Cr6+, 70.0 for Cr3+ | |
| River water | |||||||||||
| Upper Kovdora River (UKR), background | 0.06 | 10.0 | 4.3 | 0.007 | 0.002 | 0.003 | 0.18 | 0.1 | 0.2 | 0.05 | 0.6 |
| UKR, 100 m downstream from the confluence with the Bystry Current | 0.04 | 16.0 | 5.5 | 0.010 | 0.002 | 0.001 | 0.40 | 0.6 | 0.8 | 0.22 | 0.8 |
| UKR, 500 m downstream from the Discharge No. 1 | 0.05 | 30.1 | 12.8 | 0.008 | 0.002 | 0.003 | 0.16 | 0.2 | 0.2 | 0.07 | 0.8 |
| UKR, 200 m downstream from the Discharge No. 2 | 0.06 | 29.0 | 12.2 | 0.010 | 0.002 | 0.002 | 0.70 | 0.5 | 0.5 | 0.15 | 0.6 |
| UKR, 500 m downstream from the Discharge No. 3 | 0.09 | 33.1 | 16.4 | 0.015 | 0.002 | 0.002 | 1.03 | 0.5 | 1.0 | 0.5 | 0.2 |
| Mozhel River (MR), background | 0.01 | 15.0 | 6.1 | 0.008 | 0.002 | 0.002 | 0.26 | 0.1 | 0.2 | 0.05 | 0.5 |
| MR, 250 m downstream from the Discharge No. 6 | 0.09 | 64.2 | 46.0 | 0.910 | 0.002 | 0.002 | 2.57 | 1.0 | 1.2 | 0.56 | 0.2 |
| Lower Kovdora River (LKR), 200 m upstream from the mouth of MR | 0.06 | 36.1 | 16.4 | 0.009 | 0.002 | 0.009 | 1.14 | 0.1 | 0.9 | 0.32 | 0.2 |
| LKR, 500 m downstream from the mouth of MR | 0.09 | 29.1 | 7.9 | 0.080 | 0.002 | 0.005 | 0.80 | 0.1 | 0.2 | 0.11 | 0.2 |
| Ena River (ER), upstream from LKR | 0.08 | 6.4 | 2.1 | 0.002 | 0.001 | 0.001 | 0.04 | 0.1 | 0.2 | 0.05 | 0.5 |
| ER, downstream from LKR | 0.09 | 6.9 | 2.4 | 0.021 | 0.002 | 0.001 | 0.07 | 0.1 | 0.5 | 0.05 | 0.4 |
| Wastewater | |||||||||||
| Discharge No. 1 | 0.03 | 165 | 69 | 0.005 | 0.001 | - | 0.18 | 0.1 | 0.2 | 0.05 | 0.8 |
| Discharge No. 2 | 0.02 | 27 | 10 | 0.004 | 0.001 | - | 1.00 | 0.5 | 0.5 | 0.21 | 0.7 |
| Discharge No. 3 | 0.04 | 40 | 32 | 0.017 | 0.002 | - | 2.35 | 0.3 | 0.7 | 0.68 | 0.2 |
| Discharge No. 6 | 0.09 | 64 | 49 | 0.930 | 0.002 | 0.010 | 2.62 | 0.4 | 1.0 | 0.98 | 0.2 |
| Discharge of drainage wastewater into the first settling pond | 0.06 | 57 | 24 | 0.070 | 0.002 | - | 1.65 | 0.9 | 1.0 | 0.58 | 0.2 |
| Discharge of filtration wastewater into the second settling pond | 0.12 | 69 | 52 | 1.390 | 0.002 | 0.019 | 2.98 | 0.6 | 1.8 | 1.00 | 0.2 |
| Discharge of mine wastewater into the first settling pond | 0.06 | 60 | 48 | 0.030 | 0.002 | - | 2.70 | 1.6 | 2.8 | 1.10 | 0.2 |
| Analyzed Samples | Recycled Water of the TSF | Wastewater after Filtering through the Tailings Dam | Settling Pond | Discharge from the Settling Pond | Mouth of the Mozhel River, Wastewater-Receiver |
|---|---|---|---|---|---|
| Mn | |||||
| Liquid (obtained after filtration) | 0.009 | 1.380 | 1.100 | 0.930 | 0.072 |
| Suspended fraction (filtered residue on 0.45 µm pore size membrane) | 0.013 | 0.073 | 0.017 | 0.018 | 0.017 |
| Sr | |||||
| Liquid (obtained after filtration) | 0.820 | 2.940 | 2.640 | 2.610 | 2.510 |
| Suspended fraction (filtered residue on 0.45 µm pore size membrane) | 0.020 | 0.027 | 0.006 | 0.008 | 0.002 |
| Sampling Sites | Total Content (T) or Content of Mobile Fractions (M) | % | mg/kg | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Mn | Sr | Cd | Co | Cr | Cu | Ni | Zn | ||
| Settling pond upstream from the separation dam | T | 0.29 | 0.17 | 3.0 | 40 | 23 | 200 | 30 | 90 |
| M | 0.16 | 0.07 | 0.4 | 2 | 1 | 18 | 4 | 10 | |
| Settling pond downstream from the separation dam | T | 0.17 | 0.10 | 4.0 | 23 | 60 | 80 | 28 | 40 |
| M | 0.12 | 0.04 | 0.3 | 2 | 1 | 5 | 2 | 3 | |
| Lower Kovdora River upstream from the confluence with the Mozhel River | T | 0.07 | 0.04 | 4.0 | 29 | 75 | 210 | 130 | 160 |
| M | 0.03 | 0.01 | 0.2 | 3 | 2 | 20 | 4 | 40 | |
| Mozhel River downstream from the wastewater discharge | T | 4.40 | 0.15 | 7.0 | 40 | 80 | 80 | 50 | 80 |
| M | 3.00 | 0.05 | 0.8 | 7 | 5 | 5 | 9 | 19 | |
| Mozhel River 250 m downstream from the wastewater discharge | T | 3.80 | 0.11 | 5.0 | 25 | 50 | 50 | 40 | 50 |
| M | 2.50 | 0.04 | 0.7 | 4 | 3 | 3 | 7 | 11 | |
| Mozhel River upstream from the confluence with the Lower Kovdora River | T | 4.40 | 0.14 | 9.0 | 45 | 130 | 100 | 70 | 90 |
| M | 0.30 | 0.08 | 0.6 | <0.5 | 2 | 5 | 4 | 10 | |
| Lower Kovdora River 100 m downstream from the confluence with the Mozhel River | T | 0.35 | 0.07 | 8.0 | 51 | 230 | 230 | 210 | 180 |
| M | 0.25 | 0.02 | 0.5 | 5 | 6 | 6 | 5 | 17 | |
| Lower Kovdora River 250 m downstream from the confluence with the Mozhel River | T | 0.25 | 0.03 | 4.0 | 28 | 90 | 90 | 100 | 90 |
| M | 0.18 | 0.01 | 0.4 | 4 | 5 | 5 | 4 | 16 | |
| Lower Kovdora River 1000 m downstream from the confluence with the Mozhel River | T | 0.12 | 0.03 | 6.0 | 21 | 70 | 70 | 70 | 80 |
| M | 0.10 | 0.01 | 0.3 | 3 | 4 | 4 | 3 | 14 | |
| Russian Maximum Permissible Concentration in soils [55] | T (except for Co and Cr) | 1.50 | – | 1.0 | 5 (M) | 6 (Cr3+, M) | 66 | 40 | 110 |
| Mean concentration in urban soils worldwide [56] | T | 0.73 | 0.05 | 0.9 | 14 | 80 | 39 | 33 | 158 |
| Threshold Effect Concentration [57] | T | – | – | 0.99 | – | 43.4 | 31.6 | 22.7 | 12 |
| Strata, cm | LOI | Cu | Ni | Zn | Co | Cd | Pb | Mn | Fe | K | Na | Sr | Ca | Mg | Cr | Al |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 0–1 | 11.13 | 149 | 173 | 213 | 92 | 2.59 | 61.7 | 933 | 43,156 | 6882 | 1780 | 1401 | 26,542 | 49,127 | 100 | 29,542 |
| 1–2 | 8.67 | 184 | 165 | 210 | 86 | 1.84 | 21.9 | 331 | 39,968 | 6080 | 1352 | 1533 | 30,649 | 57,359 | 108 | 31,804 |
| 2–3 | 9.50 | 178 | 156 | 190 | 67 | 2.18 | 18.0 | 273 | 45,028 | 4438 | 1091 | 1103 | 19,451 | 46,689 | 112 | 30,081 |
| 3–4 | 9.14 | 175 | 156 | 181 | 48 | 2.01 | 14.1 | 282 | 48,476 | 3724 | 1009 | 935 | 13,876 | 44,068 | 117 | 28,763 |
| 4–5 | 8.04 | 128 | 158 | 135 | 45 | 1.11 | 9.4 | 276 | 42,447 | 3220 | 900 | 866 | 12,343 | 40,852 | 103 | 25,494 |
| 5–6 | 14.98 | 68 | 79 | 99 | 22 | 0.87 | 15.1 | 181 | 24,212 | 2121 | 588 | 546 | 7556 | 19,685 | 86 | 17,025 |
| 6–7 | 19.04 | 45 | 52 | 74 | 17 | 0.75 | 10.5 | 120 | 16,376 | 1693 | 504 | 486 | 5888 | 12,666 | 82 | 13,457 |
| 7–8 | 20.34 | 38 | 48 | 64 | 13 | 0.74 | 8.4 | 95 | 14,504 | 1441 | 452 | 455 | 5493 | 10,969 | 81 | 12,273 |
| 8–9 | 20.36 | 34 | 38 | 64 | 11 | 0.50 | 7.7 | 84 | 11,600 | 1234 | 424 | 416 | 5143 | 16,448 | 89 | 10,978 |
| 9–10 | 20.76 | 27 | 34 | 55 | 11 | 0.50 | 7.1 | 83 | 10,340 | 1228 | 437 | 405 | 5117 | 14,878 | 83 | 10,923 |
| 10–11 | 21.14 | 21 | 31 | 56 | 10 | 0.42 | 5.0 | 58 | 8143 | 1115 | 388 | 292 | 5237 | 7109 | 81 | 10,967 |
| 11–12 | 21.76 | 16 | 21 | 58 | 6 | 0.40 | 4.4 | 46 | 6912 | 922 | 338 | 248 | 4763 | 4256 | 73 | 8706 |
| Strata, cm | LOI | Cu | Ni | Zn | Co | Cd | Pb | Mn | Fe | K | Na | Sr | Ca | Mg | Cr | Al |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 0–1 | 8.75 | 252 | 73 | 146 | 91 | 2.75 | 18.4 | 3652 | 16,428 | 6600 | 3037 | 1394 | 119,221 | 106,233 | 28 | 15,486 |
| 1–2 | 3.20 | 242 | 70 | 120 | 91 | 3.40 | 24.3 | 2389 | 7144 | 7157 | 2877 | 1674 | 159,029 | 119,450 | 24 | 14,863 |
| 2–3 | 2.92 | 237 | 65 | 99 | 82 | 3.89 | 19.5 | 1465 | 6430 | 7645 | 3000 | 1526 | 178,874 | 122,052 | 18 | 13,729 |
| 3–4 | 1.33 | 259 | 66 | 103 | 80 | 3.46 | 24.8 | 1360 | 6722 | 8481 | 3870 | 1662 | 122,113 | 134,641 | 16 | 15,150 |
| 4–5 | 1.30 | 265 | 66 | 100 | 83 | 3.46 | 19.8 | 1296 | 6724 | 9056 | 4487 | 1485 | 112,286 | 128,931 | 16 | 15,953 |
| 5–6 | 1.20 | 241 | 58 | 97 | 71 | 3.47 | 14.9 | 1232 | 7665 | 8241 | 4242 | 1709 | 64,135 | 130,745 | 16 | 15,569 |
| 6–7 | 1.25 | 253 | 61 | 103 | 71 | 3.96 | 19.9 | 1231 | 7475 | 8731 | 4240 | 1686 | 163,456 | 111,812 | 13 | 15,162 |
| 7–8 | 0.95 | 332 | 64 | 94 | 74 | 3.48 | 19.9 | 1235 | 6561 | 8262 | 3532 | 1646 | 174,129 | 112,120 | 10 | 14,409 |
| 8–9 | 1.25 | 261 | 63 | 96 | 71 | 3.47 | 19.9 | 1167 | 8795 | 7990 | 3697 | 1663 | 174,705 | 113,031 | 13 | 13,965 |
| Complex Iron Ores | Low-Iron Ores | ||||||
|---|---|---|---|---|---|---|---|
| Averaged Complex Iron Ores | Magnetite | Apatite- Forsterite- Magnetite | Forsterite- Magnetite | Apatite- Carbonate- Magnetite | Carbonate- Forsterite- Magnetite | Apatite- Forsterite | Apatite- Carbonatite |
| 0.37 | 0.45 | 0.32 | 0.47 | 0.35 | 0.4 | 0.2 | 0.12 |
| Horizon | LH-1 | LH-2 | LH-3 | LH-4 | LH-5 |
|---|---|---|---|---|---|
| Concentration | 0.11 | 0.11 | 0.12 | 0.13 | 0.15 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Matveeva, V.A.; Alekseenko, A.V.; Karthe, D.; Puzanov, A.V. Manganese Pollution in Mining-Influenced Rivers and Lakes: Current State and Forecast under Climate Change in the Russian Arctic. Water 2022, 14, 1091. https://doi.org/10.3390/w14071091
Matveeva VA, Alekseenko AV, Karthe D, Puzanov AV. Manganese Pollution in Mining-Influenced Rivers and Lakes: Current State and Forecast under Climate Change in the Russian Arctic. Water. 2022; 14(7):1091. https://doi.org/10.3390/w14071091
Chicago/Turabian StyleMatveeva, Vera A., Alexey V. Alekseenko, Daniel Karthe, and Alexander V. Puzanov. 2022. "Manganese Pollution in Mining-Influenced Rivers and Lakes: Current State and Forecast under Climate Change in the Russian Arctic" Water 14, no. 7: 1091. https://doi.org/10.3390/w14071091
APA StyleMatveeva, V. A., Alekseenko, A. V., Karthe, D., & Puzanov, A. V. (2022). Manganese Pollution in Mining-Influenced Rivers and Lakes: Current State and Forecast under Climate Change in the Russian Arctic. Water, 14(7), 1091. https://doi.org/10.3390/w14071091







