Assessment of Reed Grasses (Phragmites australis) Performance in PFAS Removal from Water: A Phytoremediation Pilot Plant Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Area
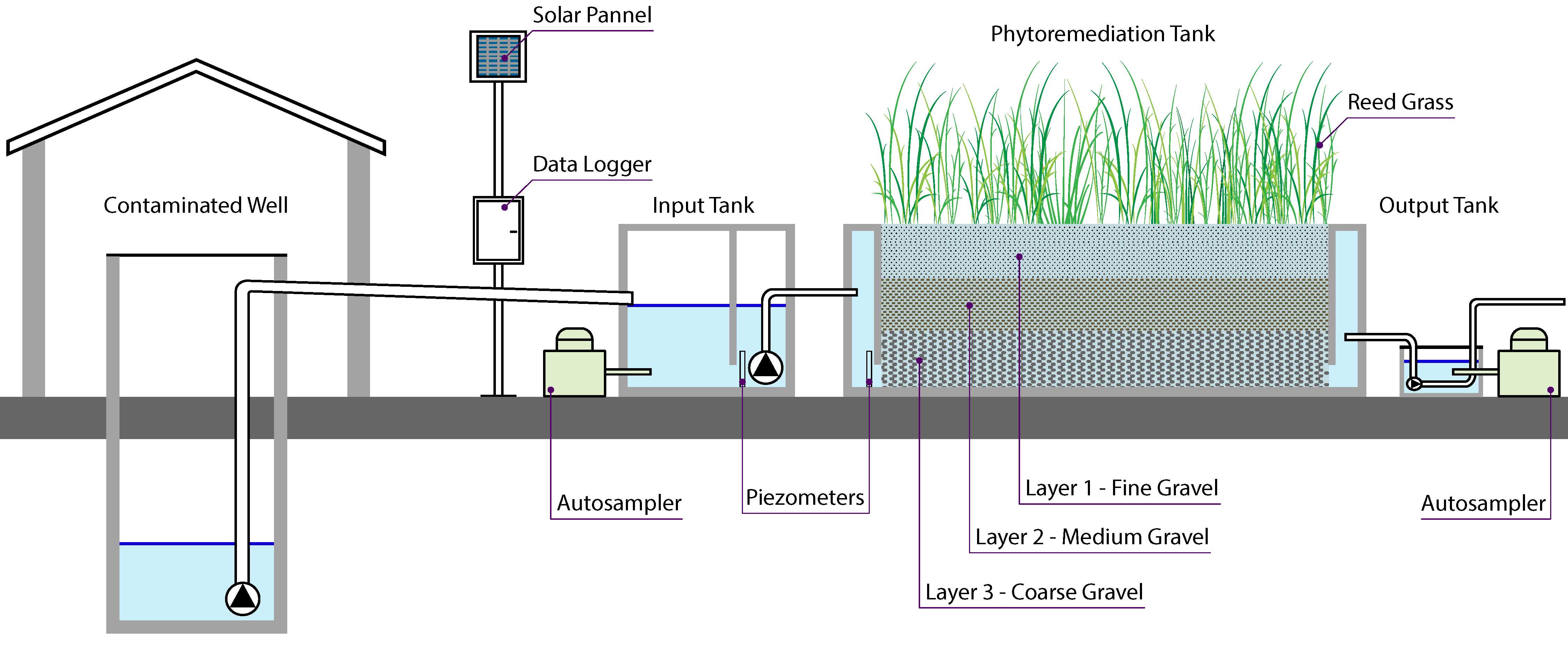
2.2. The Phytoremediation Pilot Plant
2.3. Experimental Activities
2.4. Chemical Analysis and Standards
2.5. Sample Extraction
2.6. PFAA Determination
3. Results and Discussion
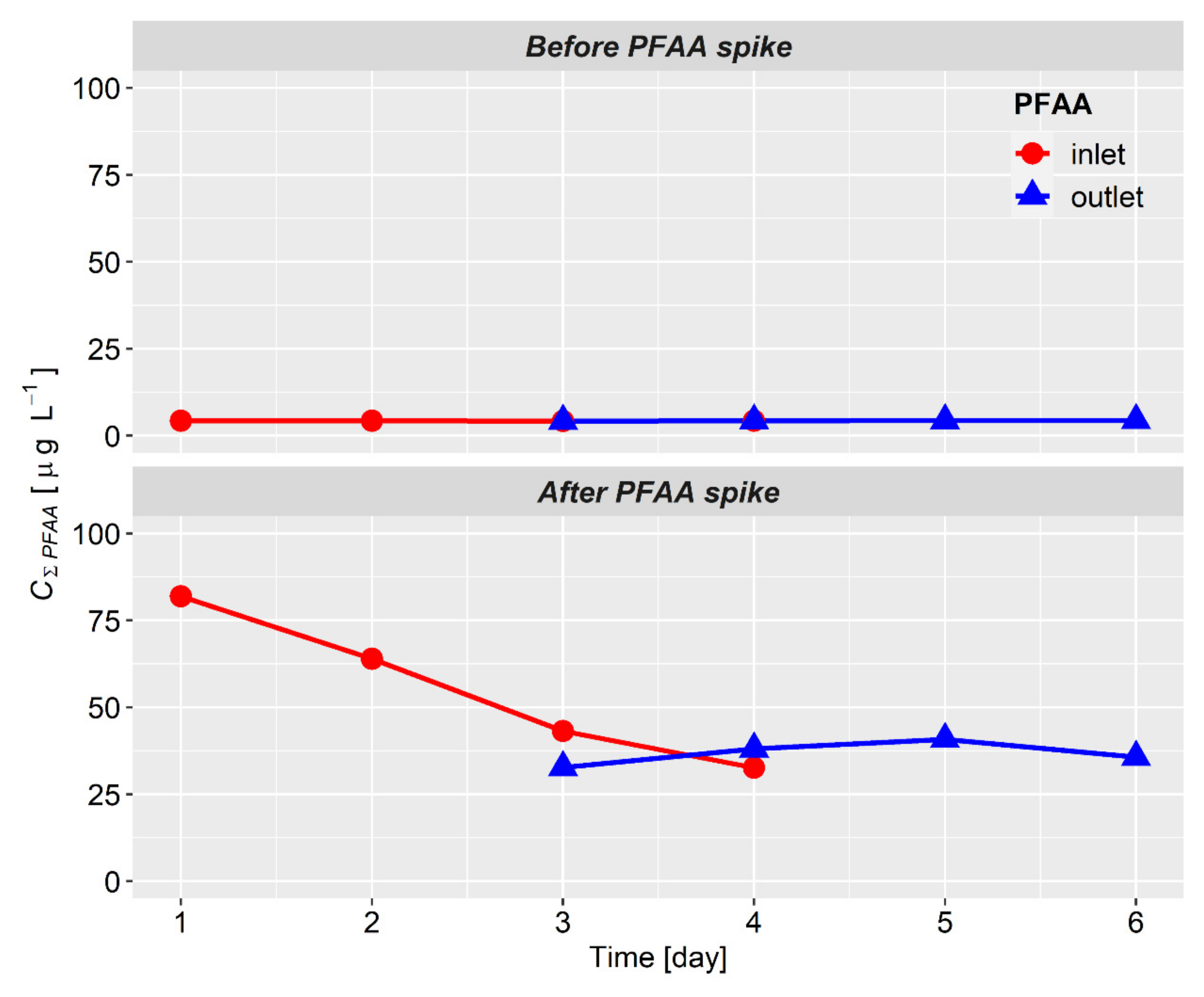
3.1. PFAAs in Water
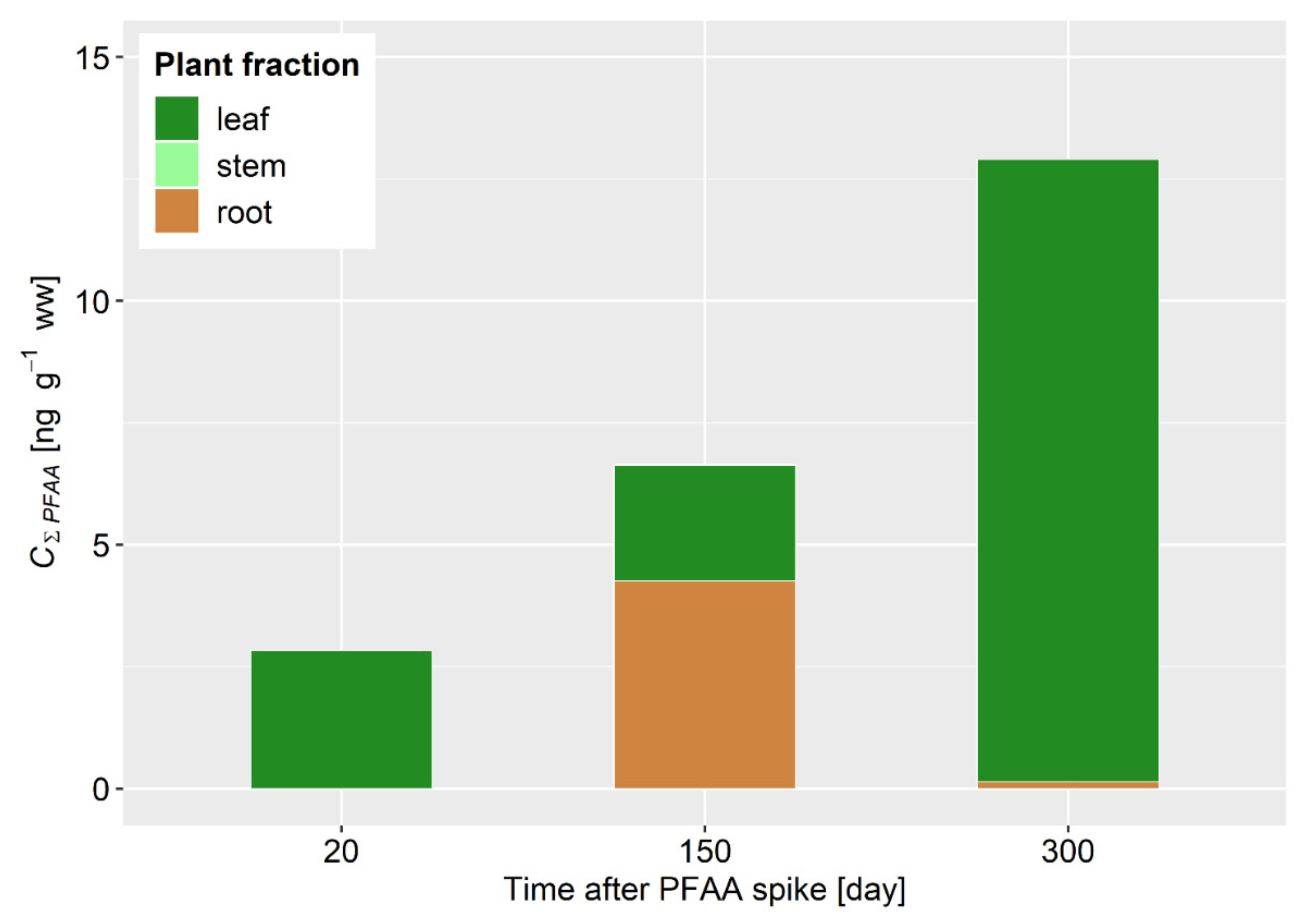
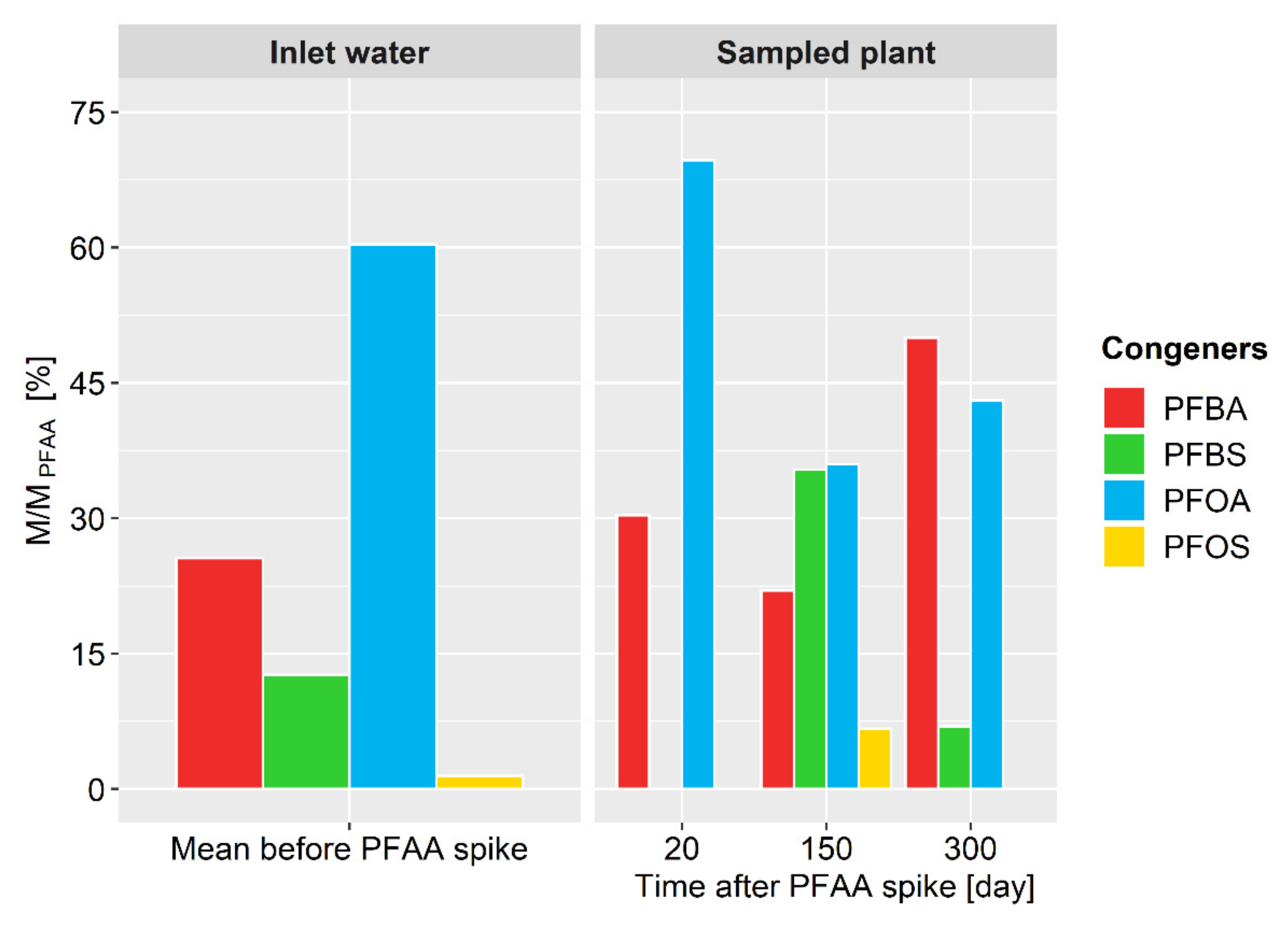
3.2. PFAAs in Reed Grass
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wang, Z.; Buser, A.M.; Cousins, I.T.; Demattio, S.; Drost, W.; Johansson, O.; Ohno, K.; Patlewicz, G.; Richard, A.M.; Walker, G.W.; et al. A New OECD Definition for Per- and Polyfluoroalkyl Substances. Environ. Sci. Technol. 2021, 1–4. [Google Scholar] [CrossRef]
- Krafft, M.P.; Riess, J.G. Per- and polyfluorinated substances (PFASs): Environmental challenges. Curr. Opin. Colloid Interface Sci. 2015, 20, 192–212. [Google Scholar] [CrossRef]
- Liu, J.; Mejia Avendaño, S. Microbial degradation of polyfluoroalkyl chemicals in the environment: A review. Environ. Int. 2013, 61, 98–114. [Google Scholar] [CrossRef] [PubMed]
- Xiao, F. Emerging poly- and perfluoroalkyl substances in the aquatic environment: A review of current literature. Water Res. 2017, 124, 482–495. [Google Scholar] [CrossRef]
- Bonato, M.; Corrà, F.; Bellio, M.; Guidolin, L.; Tallandini, L.; Irato, P.; Santovito, G. Pfas environmental pollution and antioxidant responses: An overview of the impact on human field. Int. J. Environ. Res. Public Health 2020, 17, 8020. [Google Scholar] [CrossRef]
- Ahrens, L.; Bundschuh, M. Fate and effects of poly- and perfluoroalkyl substances in the aquatic environment: A review. Environ. Toxicol. Chem. 2014, 33, 1921–1929. [Google Scholar] [CrossRef]
- Lau, C.; Anitole, K.; Hodes, C.; Lai, D.; Pfahles-Hutchens, A.; Seed, J. Perfluoroalkyl acids: A review of monitoring and toxicological findings. Toxicol. Sci. 2007, 99, 366–394. [Google Scholar] [CrossRef] [Green Version]
- Buck, R.C.; Franklin, J.; Berger, U.; Conder, J.M.; Cousins, I.T.; De Voogt, P.; Jensen, A.A.; Kannan, K.; Mabury, S.A.; van Leeuwen, S.P.J. Perfluoroalkyl and polyfluoroalkyl substances in the environment: Terminology, classification, and origins. Integr. Environ. Assess. Manag. 2011, 7, 513–541. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, C.; Kappleman, W.; DiGuiseppi, W. Ecological Considerations of Per- and Polyfluoroalkyl Substances (PFAS). Curr. Pollut. Reports 2017, 3, 289–301. [Google Scholar] [CrossRef]
- Fang, X.; Wang, Q.; Zhao, Z.; Tang, J.; Tian, C.; Yao, Y.; Yu, J.; Sun, H. Distribution and dry deposition of alternative and legacy perfluoroalkyl and polyfluoroalkyl substances in the air above the Bohai and Yellow Seas, China. Atmos. Environ. 2018, 192, 128–135. [Google Scholar] [CrossRef]
- Valsecchi, S.; Rusconi, M.; Mazzoni, M.; Viviano, G.; Pagnotta, R.; Zaghi, C.; Serrini, G.; Polesello, S. Occurrence and sources of perfluoroalkyl acids in Italian river basins. Chemosphere 2015, 129, 126–134. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, M.B.; Johir, M.A.H.; McLaughlan, R.; Nguyen, L.N.; Xu, B.; Nghiem, L.D. Per- and polyfluoroalkyl substances in soil and sediments: Occurrence, fate, remediation and future outlook. Sci. Total Environ. 2020, 748, 141251. [Google Scholar] [CrossRef] [PubMed]
- Valsecchi, S.; Babut, M.; Mazzoni, M.; Pascariello, S.; Ferrario, C.; De Felice, B.; Bettinetti, R.; Veyrand, B.; Marchand, P.; Polesello, S. Per- and Polyfluoroalkyl Substances (PFAS) in Fish from European Lakes: Current Contamination Status, Sources, and Perspectives for Monitoring. Environ. Toxicol. Chem. 2021, 40, 658–676. [Google Scholar] [CrossRef] [PubMed]
- Giesy, J.P.; Kannan, K. Global distribution of perfluorooctane sulfonate in wildlife. Environ. Sci. Technol. 2001, 35, 1339–1342. [Google Scholar] [CrossRef]
- Houde, M.; Martin, J.W.; Letcher, R.J.; Solomon, K.R.; Muir, D.C.G. Biological monitoring of polyfluoroalkyl substances: A review. Environ. Sci. Technol. 2006, 40, 3463–3473. [Google Scholar] [CrossRef]
- Ghisi, R.; Vamerali, T.; Manzetti, S. Accumulation of perfluorinated alkyl substances (PFAS) in agricultural plants: A review. Environ. Res. 2019, 169, 326–341. [Google Scholar] [CrossRef]
- Ferrario, C.; Valsecchi, S.; Lava, R.; Bonato, M.; Polesello, S. Determination of perfluoroalkyl acids in different tissues of graminaceous plants. Anal. Methods 2021, 13, 1643–1650. [Google Scholar] [CrossRef]
- Da Silva, B.F.; Ahmadireskety, A.; Aristizabal-Henao, J.J.; Bowden, J.A. A rapid and simple method to quantify per- and polyfluoroalkyl substances (PFAS) in plasma and serum using 96-well plates. MethodsX 2020, 7, 101111. [Google Scholar] [CrossRef]
- Ankley, G.T.; Cureton, P.; Hoke, R.A.; Houde, M.; Kumar, A.; Kurias, J.; Lanno, R.; McCarthy, C.; Newsted, J.; Salice, C.J.; et al. Assessing the Ecological Risks of Per- and Polyfluoroalkyl Substances: Current State-of-the Science and a Proposed Path Forward. Environ. Toxicol. Chem. 2021, 40, 564–605. [Google Scholar] [CrossRef]
- Gredelj, A.; Nicoletto, C.; Valsecchi, S.; Ferrario, C.; Polesello, S.; Lava, R.; Zanon, F.; Barausse, A.; Palmeri, L.; Guidolin, L.; et al. Uptake and translocation of perfluoroalkyl acids (PFAA) in red chicory (Cichorium intybus L.) under various treatments with pre-contaminated soil and irrigation water. Sci. Total Environ. 2020, 708, 134766. [Google Scholar] [CrossRef]
- Gredelj, A.; Nicoletto, C.; Polesello, S.; Ferrario, C.; Valsecchi, S.; Lava, R.; Barausse, A.; Zanon, F.; Palmeri, L.; Guidolin, L.; et al. Uptake and translocation of perfluoroalkyl acids (PFAAs) in hydroponically grown red chicory (Cichorium intybus L.): Growth and developmental toxicity, comparison with growth in soil and bioavailability implications. Sci. Total Environ. 2020, 720, 137333. [Google Scholar] [CrossRef]
- Brendel, S.; Fetter, É.; Staude, C.; Vierke, L.; Biegel-Engler, A. Short-chain perfluoroalkyl acids: Environmental concerns and a regulatory strategy under REACH. Environ. Sci. Eur. 2018, 30. [Google Scholar] [CrossRef]
- Ritscher, A.; Wang, Z.; Scheringer, M.; Boucher, J.M.; Ahrens, L.; Berger, U.; Bintein, S.; Bopp, S.K.; Borg, D.; Buser, A.M.; et al. Zürich Statement on Future Actions on Per- and Polyfluoroalkyl Substances. Environ. Health Perspect. 2018, 126, 084502. [Google Scholar] [CrossRef]
- Wang, Z.; Dewitt, J.C.; Higgins, C.P.; Cousins, I.T. A Never-Ending Story of Per- and Polyfluoroalkyl Substances (PFASs)? Environ. Sci. Technol. 2017, 51, 2508–2518. [Google Scholar] [CrossRef]
- Schrenk, D.; Bignami, M.; Bodin, L.; Chipman, J.K.; del Mazo, J.; Grasl-Kraupp, B.; Hogstrand, C.; Hoogenboom, L.; Leblanc, J.C.; Nebbia, C.S.; et al. Risk to human health related to the presence of perfluoroalkyl substances in food. EFSA J. 2020, 18. [Google Scholar] [CrossRef]
- Ahmed, M.B.; Alam, M.M.; Zhou, J.L.; Xu, B.; Johir, M.A.H.; Karmakar, A.K.; Rahman, M.S.; Hossen, J.; Hasan, A.T.M.K.; Moni, M.A. Advanced treatment technologies efficacies and mechanism of per- and poly-fluoroalkyl substances removal from water. Process Saf. Environ. Prot. 2020, 136, 1–14. [Google Scholar] [CrossRef]
- Cao, M.H.; Wang, B.B.; Yu, H.S.; Wang, L.L.; Yuan, S.H.; Chen, J. Photochemical decomposition of perfluorooctanoic acid in aqueous periodate with VUV and UV light irradiation. J. Hazard. Mater. 2010, 179, 1143–1146. [Google Scholar] [CrossRef]
- Hori, H.; Yamamoto, A.; Hayakawa, E.; Taniyasu, S.; Yamashita, N.; Kutsuna, S.; Kiatagawa, H.; Arakawa, R. Efficient decomposition of environmentally persistent perfluorocarboxylic acids by use of persulfate as a photochemical oxidant. Environ. Sci. Technol. 2005, 39, 2383–2388. [Google Scholar] [CrossRef]
- Hori, H.; Nagaoka, Y.; Yamamoto, A.; Sano, T.; Yamashita, N.; Taniyasu, S.; Kutsuna, S.; Osaka, I.; Arakawa, R. Efficient decomposition of environmentally persistent perfluorooctanesulfonate and related fluorochemicals using zerovalent iron in subcritical water. Environ. Sci. Technol. 2006, 40, 1049–1054. [Google Scholar] [CrossRef]
- Hori, H.; Yamamoto, A.; Koike, K.; Kutsuna, S.; Osaka, I.; Arakawa, R. Photochemical decomposition of environmentally persistent short-chain perfluorocarboxylic acids in water mediated by iron(II)/(III) redox reactions. Chemosphere 2007, 68, 572–578. [Google Scholar] [CrossRef]
- Hori, H.; Nagaoka, Y.; Murayama, M.; Kutsuna, S. Efficient decomposition of perfluorocarboxylic acids and alternative fluorochemical surfactants in hot water. Environ. Sci. Technol. 2008, 42, 7438–7443. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.C.; Lo, S.L.; Chiueh, P.T.; Liou, Y.H.; Chen, M.L. Microwave-hydrothermal decomposition of perfluorooctanoic acid in water by iron-activated persulfate oxidation. Water Res. 2010, 44, 886–892. [Google Scholar] [CrossRef] [PubMed]
- Kucharzyk, K.H.; Darlington, R.; Benotti, M.; Deeb, R.; Hawley, E. Novel treatment technologies for PFAS compounds: A critical review. J. Environ. Manag. 2017, 204, 757–764. [Google Scholar] [CrossRef] [PubMed]
- Sorlini, S.; Collivignarelli, M.C.; Miino, M.C. Technologies for the control of emerging contaminants in drinking water treatment plants. Environ. Eng. Manag. J. 2019, 18, 2203–2216. [Google Scholar]
- Lu, D.; Sha, S.; Luo, J.; Huang, Z.; Zhang Jackie, X. Treatment train approaches for the remediation of per- and polyfluoroalkyl substances (PFAS): A critical review. J. Hazard. Mater. 2020, 386, 121963. [Google Scholar] [CrossRef] [PubMed]
- Deng, S.; Yu, Q.; Huang, J.; Yu, G. Removal of perfluorooctane sulfonate from wastewater by anion exchange resins: Effects of resin properties and solution chemistry. Water Res. 2010, 44, 5188–5195. [Google Scholar] [CrossRef]
- Dixit, F.; Dutta, R.; Barbeau, B.; Berube, P.; Mohseni, M. PFAS removal by ion exchange resins: A review. Chemosphere 2021, 272, 129777. [Google Scholar] [CrossRef]
- Stoiber, T.; Evans, S.; Temkin, A.M.; Andrews, D.Q.; Naidenko, O.V. PFAS in drinking water: An emergent water quality threat. Water Solut. 2020, 1, e49. [Google Scholar]
- Du, Z.; Deng, S.; Bei, Y.; Huang, Q.; Wang, B.; Huang, J.; Yu, G. Adsorption behavior and mechanism of perfluorinated compounds on various adsorbents-A review. J. Hazard. Mater. 2014, 274, 443–454. [Google Scholar] [CrossRef]
- Kennedy, A.M.; Reinert, A.M.; Knappe, D.R.U.; Ferrer, I.; Summers, R.S. Full- and pilot-scale GAC adsorption of organic micropollutants. Water Res. 2015, 68, 238–248. [Google Scholar] [CrossRef]
- McCleaf, P.; Englund, S.; Östlund, A.; Lindegren, K.; Wiberg, K.; Ahrens, L. Removal efficiency of multiple poly- and perfluoroalkyl substances (PFASs) in drinking water using granular activated carbon (GAC) and anion exchange (AE) column tests. Water Res. 2017, 120, 77–87. [Google Scholar] [CrossRef]
- Sonmez Baghirzade, B.; Zhang, Y.; Reuther, J.F.; Saleh, N.B.; Venkatesan, A.K.; Apul, O.G. Thermal Regeneration of Spent Granular Activated Carbon Presents an Opportunity to Break the Forever PFAS Cycle. Environ. Sci. Technol. 2021, 55, 5608–5619. [Google Scholar] [CrossRef]
- Radjenovic, J.; Duinslaeger, N.; Avval, S.S.; Chaplin, B.P. Facing the Challenge of Poly- And Perfluoroalkyl Substances in Water: Is Electrochemical Oxidation the Answer? Environ. Sci. Technol. 2020, 54, 14815–14829. [Google Scholar] [CrossRef]
- Nzeribe, B.N.; Crimi, M.; Mededovic Thagard, S.; Holsen, T.M. Physico-Chemical Processes for the Treatment of Per- And Polyfluoroalkyl Substances (PFAS): A review. Crit. Rev. Environ. Sci. Technol. 2019, 49, 866–915. [Google Scholar] [CrossRef]
- Sahu, S.P.; Qanbarzadeh, M.; Ateia, M.; Torkzadeh, H.; Maroli, A.S.; Cates, E.L. Rapid Degradation and Mineralization of Perfluorooctanoic Acid by a New Petitjeanite Bi3O(OH)(PO4)2 Microparticle Ultraviolet Photocatalyst. Environ. Sci. Technol. Lett. 2018, 5, 533–538. [Google Scholar] [CrossRef]
- Yamada, T.; Taylor, P.H.; Buck, R.C.; Kaiser, M.A.; Giraud, R.J. Thermal degradation of fluorotelomer treated articles and related materials. Chemosphere 2005, 61, 974–984. [Google Scholar] [CrossRef]
- Stoiber, T.; Evans, S.; Naidenko, O.V. Disposal of products and materials containing per- and polyfluoroalkyl substances (PFAS): A cyclical problem. Chemosphere 2020, 260, 127659. [Google Scholar] [CrossRef]
- Krusic, P.J.; Roe, D.C. Gas-phase NMR technique for studying the thermolysis of materials: Thermal decomposition of ammonium perfluorooctanoate. Anal. Chem. 2004, 76, 3800–3803. [Google Scholar] [CrossRef]
- McKay, G. Dioxin characterisation, formation and minimisation during municipal solid waste (MSW) incineration: Review. Chem. Eng. J. 2002, 86, 343–368. [Google Scholar] [CrossRef]
- Tuppurainen, K.; Halonen, I.; Ruokojärvi, P.; Tarhanen, J.; Ruuskanen, J. Formation of PCDDs and PCDFs in municipal waste incineration and its inhibition mechanisms: A review. Chemosphere 1998, 36, 1493–1511. [Google Scholar] [CrossRef]
- Boano, F.; Caruso, A.; Costamagna, E.; Ridolfi, L.; Fiore, S.; Demichelis, F.; Galvão, A.; Pisoeiro, J.; Rizzo, A.; Masi, F. A review of nature-based solutions for greywater treatment: Applications, hydraulic design, and environmental benefits. Sci. Total Environ. 2020, 711, 134731. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, A.; Tondera, K.; Pálfy, T.G.; Dittmer, U.; Meyer, D.; Schreiber, C.; Zacharias, N.; Ruppelt, J.P.; Esser, D.; Molle, P.; et al. Constructed wetlands for combined sewer overflow treatment: A state-of-the-art review. Sci. Total Environ. 2020, 727, 138618. [Google Scholar] [CrossRef] [PubMed]
- Lavrnić, S.; Alagna, V.; Iovino, M.; Anconelli, S.; Solimando, D.; Toscano, A. Hydrological and hydraulic behaviour of a surface flow constructed wetland treating agricultural drainage water in northern Italy. Sci. Total Environ. 2020, 702. [Google Scholar] [CrossRef] [PubMed]
- Kadlec, R.H.; Wallace, S. Treatment Wetlands, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2009; ISBN 9781566705264. [Google Scholar]
- Colares, G.S.; Dell’Osbel, N.; Wiesel, P.G.; Oliveira, G.A.; Lemos, P.H.Z.; da Silva, F.P.; Lutterbeck, C.A.; Kist, L.T.; Machado, Ê.L. Floating treatment wetlands: A review and bibliometric analysis. Sci. Total Environ. 2020, 714, 136776. [Google Scholar] [CrossRef] [PubMed]
- Arslan, M.; El-Din, M.G. Removal of per- and poly-fluoroalkyl substances (PFASs) by wetlands: Prospects on plants, microbes and the interplay. Sci. Total Environ. 2021, 800, 149570. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Bai, Y.; Qu, J. The Phragmites root-inhabiting microbiome: A critical review on its composition and environmental application. Engineering, 2021; in press. [Google Scholar] [CrossRef]
- Chen, Y.C.; Lo, S.L.; Lee, Y.C. Distribution and fate of perfl uorinated compounds (PFCs) in a pilot constructed wetland. Desalin. Water Treat. 2012, 37, 178–184. [Google Scholar] [CrossRef]
- Rezania, S.; Park, J.; Rupani, P.F.; Darajeh, N.; Xu, X.; Shahrokhishahraki, R. Phytoremediation potential and control of Phragmites australis as a green phytomass: An overview. Environ. Sci. Pollut. Res. 2019, 26, 7428–7441. [Google Scholar] [CrossRef]
- Huang, X.; Wang, L.; Zhu, S.; Ho, S.H.; Wu, J.; Kalita, P.K.; Ma, F. Unraveling the effects of arbuscular mycorrhizal fungus on uptake, translocation, and distribution of cadmium in Phragmites australis (Cav.) Trin. ex Steud. Ecotoxicol. Environ. Saf. 2018, 149, 43–50. [Google Scholar] [CrossRef]
- Carvalho, P.N.; Basto, M.C.P.; Almeida, C.M.R. Potential of Phragmites australis for the removal of veterinary pharmaceuticals from aquatic media. Bioresour. Technol. 2012, 116, 497–501. [Google Scholar] [CrossRef]
- Almeida, C.M.R.; Santos, F.; Ferreira, A.C.F.; Gomes, C.R.; Basto, M.C.P.; Mucha, A.P. Constructed wetlands for the removal of metals from livestock wastewater—Can the presence of veterinary antibiotics affect removals? Ecotoxicol. Environ. Saf. 2017, 137, 143–148. [Google Scholar] [CrossRef]
- Liu, X.; Huang, S.; Tang, T.; Liu, X.; Scholz, M. Growth characteristics and nutrient removal capability of plants in subsurface vertical flow constructed wetlands. Ecol. Eng. 2012, 44, 189–198. [Google Scholar] [CrossRef]
- Marsik, P.; Podlipna, R.; Vanek, T. Study of praziquantel phytoremediation and transformation and its removal in constructed wetland. J. Hazard. Mater. 2017, 323, 394–399. [Google Scholar] [CrossRef]
- Topal, M. Uptake of tetracycline and degradation products by Phragmites australis grown in stream carrying secondary effluent. Ecol. Eng. 2015, 79, 80–85. [Google Scholar] [CrossRef]
- Wang, A.; Chi, J. Phthalic acid esters in the rhizosphere sediments of emergent plants from two shallow lakes. J. Soils Sediments 2012, 12, 1189–1196. [Google Scholar] [CrossRef]
- Schaller, J.; Brackhage, C.; Paasch, S.; Brunner, E.; Bäucker, E.; Dudel, E.G. Silica uptake from nanoparticles and silica condensation state in different tissues of Phragmites australis. Sci. Total Environ. 2013, 442, 6–9. [Google Scholar] [CrossRef]
- Kankiliç, G.B.; Metin, A.Ü.; Tüzün, I. Phragmites australis: An alternative biosorbent for basic dye removal. Ecol. Eng. 2016, 86, 85–94. [Google Scholar] [CrossRef]
- Haddaji, D.; Bousselmi, L.; Saadani, O.; Nouairi, I.; Ghrabi-Gammar, Z. Enzymatic degradation of azo dyes using three macrophyte species: Arundo donax, Typha angustifolia and Phragmites australis. Desalin. Water Treat. 2015, 53, 1129–1138. [Google Scholar]
- Lv, T.; Zhang, Y.; Zhang, L.; Carvalho, P.N.; Arias, C.A.; Brix, H. Removal of the pesticides imazalil and tebuconazole in saturated constructed wetland mesocosms. Water Res. 2016, 91, 126–136. [Google Scholar] [CrossRef]
- Hechmi, N.; Aissa, N.B.; Abdenaceur, H.; Jedidi, N. Evaluating the phytoremediation potential of Phragmites australis grown in pentachlorophenol and cadmium co-contaminated soils. Environ. Sci. Pollut. Res. 2014, 21, 1304–1313. [Google Scholar] [CrossRef]
- Mudumbi, J.B.N.; Ntwampe, S.K.O.; Muganza, M.; Okonkwo, J.O. Susceptibility of Riparian Wetland Plants to Perfluorooctanoic Acid (PFOA) Accumulation. Int. J. Phytoremediat. 2014, 16, 926–936. [Google Scholar] [CrossRef]
- Wang, P.; Zhang, M.; Lu, Y.; Meng, J.; Li, Q.; Lu, X. Removal of perfluoalkyl acids (PFAAs) through fluorochemical industrial and domestic wastewater treatment plants and bioaccumulation in aquatic plants in river and artificial wetland. Environ. Int. 2019, 129, 76–85. [Google Scholar] [CrossRef]
- Li, Y.; He, L.; Lv, L.; Xue, J.; Wu, L.; Zhang, Z.; Yang, L. Review on plant uptake of PFOS and PFOA for environmental cleanup: Potential and implications. Environ. Sci. Pollut. Res. 2021, 28, 30459–30470. [Google Scholar] [CrossRef] [PubMed]
- Castiglioni, S.; Valsecchi, S.; Polesello, S.; Rusconi, M.; Melis, M.; Palmiotto, M.; Manenti, A.; Davoli, E.; Zuccato, E. Sources and fate of perfluorinated compounds in the aqueous environment and in drinking water of a highly urbanized and industrialized area in Italy. J. Hazard. Mater. 2015, 282, 51–60. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Keeping Our Water Clean: The Case of Water Contamination in Veneto Region, Italy; WHO: Geneva, Switzerland, 2017.
- Pitter, G.; Da Re, F.; Canova, C.; Barbieri, G.; Jeddi, M.Z.; Daprà, F.; Manea, F.; Zolin, R.; Bettega, A.M.; Stopazzolo, G.; et al. Serum levels of perfluoroalkyl substances (PFAS) in adolescents and young adults exposed to contaminated drinking water in the Veneto region, Italy: A cross-sectional study based on a health surveillance program. Environ. Health Perspect. 2020, 128, 027007. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Milke, J.; Gałczyńska, M.; Wróbel, J. The importance of biological and ecological properties of Phragmites australis (Cav.) Trin. Ex steud., in phytoremendiation of aquatic ecosystems-The review. Water 2020, 12, 1770. [Google Scholar] [CrossRef]
- Allen, R.G.; Pereira, L.S.; Raes, D.; Smith, M. Crop Evapotraspiration Guidelines for Computing Crop Water Requirements; FAO: Rome, Italy, 1998; ISBN 9251042195. [Google Scholar]
- Hargreaves, G.H.; Samani, Z.A. Reference crop evapotranspiration from temperature. Appl. Eng. Agric. 1985, 1, 96–99. [Google Scholar] [CrossRef]
- Paredes, P.; Pereira, L.S.; Almorox, J.; Darouich, H. Reference grass evapotranspiration with reduced data sets: Parameterization of the FAO Penman-Monteith temperature approach and the Hargeaves-Samani equation using local climatic variables. Agric. Water Manag. 2020, 240, 106210. [Google Scholar] [CrossRef]
- Talebmorad, H.; Ahmadnejad, A.; Eslamian, S.; Ostad-Ali-Askari, K.; Singh, V.P. Evaluation of uncertainty in evapotranspiration values by FAO56-Penman-Monteith and Hargreaves-Samani methods. Int. J. Hydrol. Sci. Technol. 2020, 10, 135–147. [Google Scholar] [CrossRef]
- Bettio, D. Analysis and Modelling of the Functioning of a Phytoremediation Pilot Plant for the Extraction of PFAS COMPOUNDS from Water. Master’s Thesis, University of Padova, Padova, Italy, 2018. [Google Scholar]
- ASTM-D7979; ASTM Standard Test Method for Determination of Perfluorinated Compounds in Water, Sludge, Influent, Effluent and Wastewater by Liquid Chromatography Tandem Mass Spectrometry (LC/MS/MS); ASTM International: West Conshohocken, PA, USA, 2016.
- Metcalf, L.; Eddy, H.P. Wastewater Engineering: Treatment, Disposal, and Reuse, 4th ed.; McGraw-Hill: New York, NY, USA, 2002. [Google Scholar]
- Scher, D.P.; Kelly, J.E.; Huset, C.A.; Barry, K.M.; Hoffbeck, R.W.; Yingling, V.L.; Messing, R.B. Occurrence of perfluoroalkyl substances (PFAS) in garden produce at homes with a history of PFAS-contaminated drinking water. Chemosphere 2018, 196, 548–555. [Google Scholar] [CrossRef]
- Huang, J.; Reneau, R.B., Jr.; Hagedorn, C. Nitrogen removal in constructed wetlands employed to treat domestic wastewater. Water Res. 2000, 34, 2582–2588. [Google Scholar] [CrossRef]
- Knight, R.L.; Payne, V.W.E.; Borer, R.E.; Clarke, R.A.; Pries, J.H. Constructed wetlands for livestock wastewater management. Ecol. Eng. 2000, 15, 41–55. [Google Scholar] [CrossRef]
- Kumari, M.; Tripathi, B.D. Efficiency of Phragmites australis and Typha latifolia for heavy metal removal from wastewater. Ecotoxicol. Environ. Saf. 2015, 112, 80–86. [Google Scholar] [CrossRef]
- Akratos, C.S.; Tsihrintzis, V.A. Effect of temperature, HRT, vegetation and porous media on removal efficiency of pilot-scale horizontal subsurface flow constructed wetlands. Ecol. Eng. 2007, 29, 173–191. [Google Scholar] [CrossRef]
- Rodríguez, M.; Brisson, J. Pollutant removal efficiency of native versus exotic common reed (Phragmites australis) in North American treatment wetlands. Ecol. Eng. 2015, 74, 364–370. [Google Scholar] [CrossRef]
- Lee, B.H.; Scholz, M. What is the role of Phragmites australis in experimental constructed wetland filters treating urban runoff? Ecol. Eng. 2007, 29, 87–95. [Google Scholar] [CrossRef]
- Zhao, H.; Guan, Y.; Qu, B. PFCA uptake and translocation in dominant wheat species (Triticum aestivum L.). Int. J. Phytoremediat. 2018, 20, 68–74. [Google Scholar] [CrossRef]
- Gobelius, L.; Lewis, J.; Ahrens, L. Plant Uptake of Per- and Polyfluoroalkyl Substances at a Contaminated Fire Training Facility to Evaluate the Phytoremediation Potential of Various Plant Species. Environ. Sci. Technol. 2017, 51, 12602–12610. [Google Scholar] [CrossRef]
- Mulkeen, C.J.; Williams, C.D.; Gormally, M.J.; Healy, M.G. Seasonal patterns of metals and nutrients in Phragmites australis (Cav.) Trin. ex Steudel in a constructed wetland in the west of Ireland. Ecol. Eng. 2017, 107, 192–197. [Google Scholar] [CrossRef]
- Saeed, T.; Sun, G. A review on nitrogen and organics removal mechanisms in subsurface flow constructed wetlands: Dependency on environmental parameters, operating conditions and supporting media. J. Environ. Manag. 2012, 112, 429–448. [Google Scholar] [CrossRef]


| 0.275 | 0.175 | 5.1 |
| Day | Date | Tank | Concentration (μg L−1) | ||||
|---|---|---|---|---|---|---|---|
| PFBA | PFBS | PFOA | PFOS | ΣPFAA | |||
| Before PFAA spike | |||||||
| 1 | 24 September 2018 | Input | 1.00 | 0.52 | 2.70 | 0.08 | 4.30 |
| 2 | 25 September 2018 | Input | 1.01 | 0.50 | 2.71 | 0.07 | 4.30 |
| 3 | 26 September 2018 | Input | 1.02 | 0.54 | 2.61 | 0.07 | 4.23 |
| 4 | 27 September 2018 | Input | 0.97 | 0.52 | 2.71 | 0.07 | 4.26 |
| 3 | 26 September 2018 | Output | 1.02 | 0.53 | 2.6 | 0.07 | 4.22 |
| 4 | 27 September 2018 | Output | 1.00 | 0.53 | 2.7 | 0.07 | 4.31 |
| 5 | 28 September 2018 | Output | 0.99 | 0.54 | 2.8 | 0.06 | 4.38 |
| 6 | 29 September 2018 | Output | 1.00 | 0.57 | 2.8 | 0.07 | 4.42 |
| After PFAA spike | |||||||
| 1 | 1 October 2018 | Input | 34.32 | 15.59 | 23.62 | 8.43 | 81.95 |
| 2 | 2 October 2018 | Input | 24.92 | 13.26 | 20.26 | 5.53 | 63.96 |
| 3 | 3 October 2018 | Input | 16.47 | 9.29 | 14.06 | 3.33 | 43.14 |
| 4 | 4 October 2018 | Input | 11.82 | 6.93 | 11.64 | 2.22 | 32.61 |
| 3 | 3 October 2018 | Output | 12.12 | 7.84 | 11.44 | 1.34 | 32.74 |
| 4 | 4 October 2018 | Output | 14.52 | 8.31 | 13.27 | 1.99 | 38.08 |
| 5 | 5 October 2018 | Output | 15.62 | 8.59 | 14.47 | 2.16 | 40.84 |
| 6 | 6 October 2018 | Output | 13.09 | 8.25 | 12.54 | 1.80 | 35.69 |
| Date | Height | Weight | Root | Stem | Leaf | |||
|---|---|---|---|---|---|---|---|---|
| (cm) | (g ww) | Moisture (%) | Fraction of Plant (%) | Moisture (%) | Fraction of Plant (%) | Moisture (%) | Fraction of Plant (%) | |
| 12 September 2018 | 128.0 | 63.7 | - | - | - | - | - | - |
| 24 October 2018 | 140.4 | 138.3 | 33.0 | 44.7 | 7.6 | 34.8 | 3.7 | 20.5 |
| 7 March 2019 | 163.7 | 166.1 | 37.7 | 31.5 | 22.3 | 38.6 | 14.5 | 28.9 |
| 14 August 2019 | 187.0 | 194.0 | 86.4 | 18.3 | 28.3 | 44.4 | 15.7 | 37.3 |
| Abbreviation | Chemical (Common) Name | No. of Perfluorinated Carbons (CnF2n) | Functional Group | Molar Mass (g mol−1) | CAS Number |
|---|---|---|---|---|---|
| Short-chain PFAA | |||||
| PFBA | Perfluorobutanoic acid | 3 | –COOH | 214 | 375-22-4 |
| PFBS | Perfluorobutanesulfonic acid | 4 | – | 300 | 375-73-5 |
| Long-chain PFAA | |||||
| PFOA | Perfluorooctanoic acid | 7 | –COOH | 414 | 335-67-1 |
| PFOS | Perfluorooctanesulfonic acid | 8 | – | 500 | 1763-23-1 |
| PFBA | PFBS | PFOA | PFOS | ΣPFAA | |
|---|---|---|---|---|---|
| Before PFAA spike | 36.7 | 32.8 | 34.5 | 50.4 | 35.1 |
| After PFAA spike | 71.0 | 64.9 | 61.0 | 83.7 | 68.3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ferrario, C.; Peruzzi, C.; Cislaghi, A.; Polesello, S.; Valsecchi, S.; Lava, R.; Zanon, F.; Santovito, G.; Barausse, A.; Bonato, M. Assessment of Reed Grasses (Phragmites australis) Performance in PFAS Removal from Water: A Phytoremediation Pilot Plant Study. Water 2022, 14, 946. https://doi.org/10.3390/w14060946
Ferrario C, Peruzzi C, Cislaghi A, Polesello S, Valsecchi S, Lava R, Zanon F, Santovito G, Barausse A, Bonato M. Assessment of Reed Grasses (Phragmites australis) Performance in PFAS Removal from Water: A Phytoremediation Pilot Plant Study. Water. 2022; 14(6):946. https://doi.org/10.3390/w14060946
Chicago/Turabian StyleFerrario, Claudia, Cosimo Peruzzi, Alessio Cislaghi, Stefano Polesello, Sara Valsecchi, Roberto Lava, Francesca Zanon, Gianfranco Santovito, Alberto Barausse, and Marco Bonato. 2022. "Assessment of Reed Grasses (Phragmites australis) Performance in PFAS Removal from Water: A Phytoremediation Pilot Plant Study" Water 14, no. 6: 946. https://doi.org/10.3390/w14060946
APA StyleFerrario, C., Peruzzi, C., Cislaghi, A., Polesello, S., Valsecchi, S., Lava, R., Zanon, F., Santovito, G., Barausse, A., & Bonato, M. (2022). Assessment of Reed Grasses (Phragmites australis) Performance in PFAS Removal from Water: A Phytoremediation Pilot Plant Study. Water, 14(6), 946. https://doi.org/10.3390/w14060946










