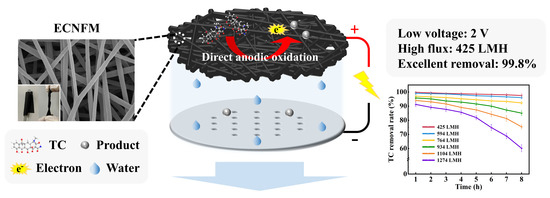
A High Flux Electrochemical Filtration System Based on Electrospun Carbon Nanofiber Membrane for Efficient Tetracycline Degradation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials and Reagents
2.2. Preparation of ECNFM
2.3. Characterization
2.4. Electrochemical Filtration Experiment
2.5. Analytical Methods
3. Results and Discussion
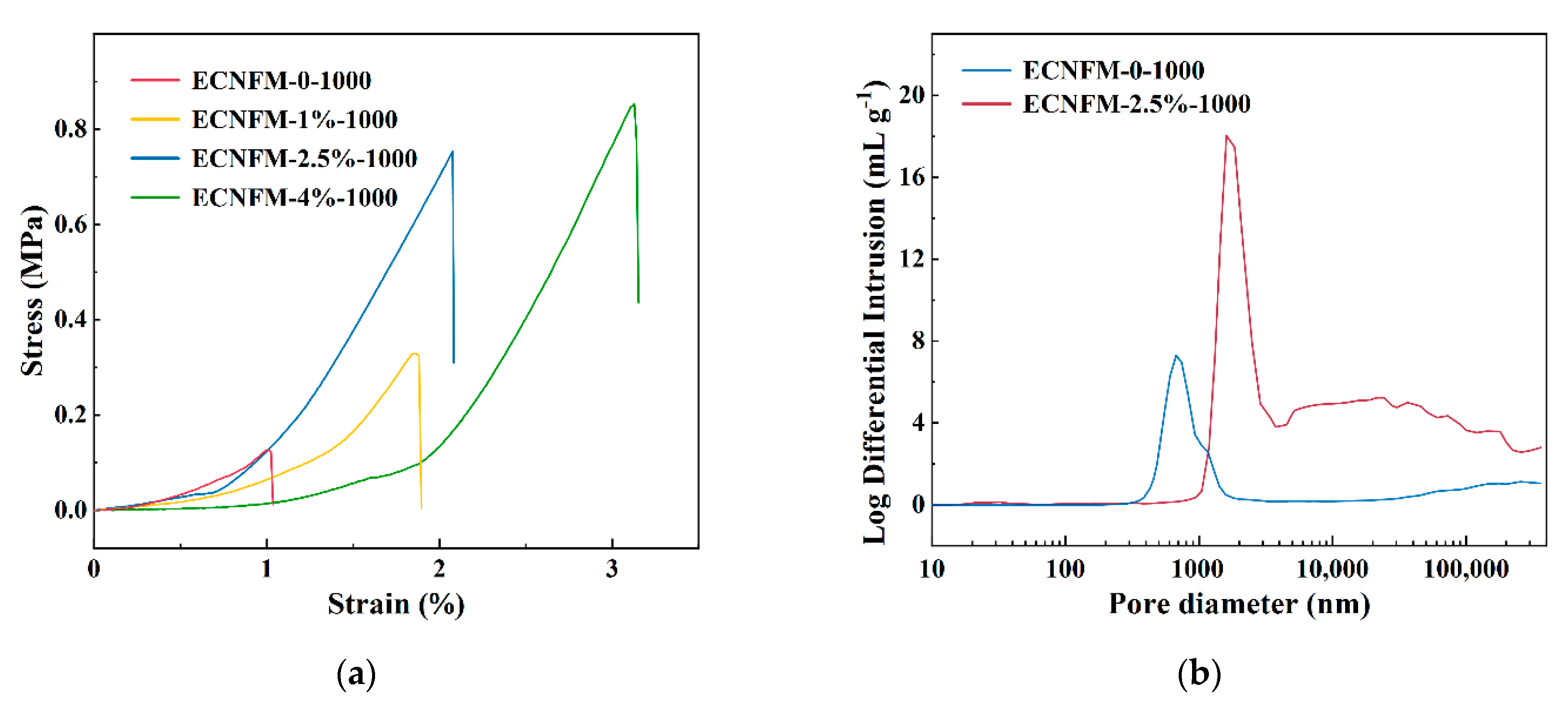
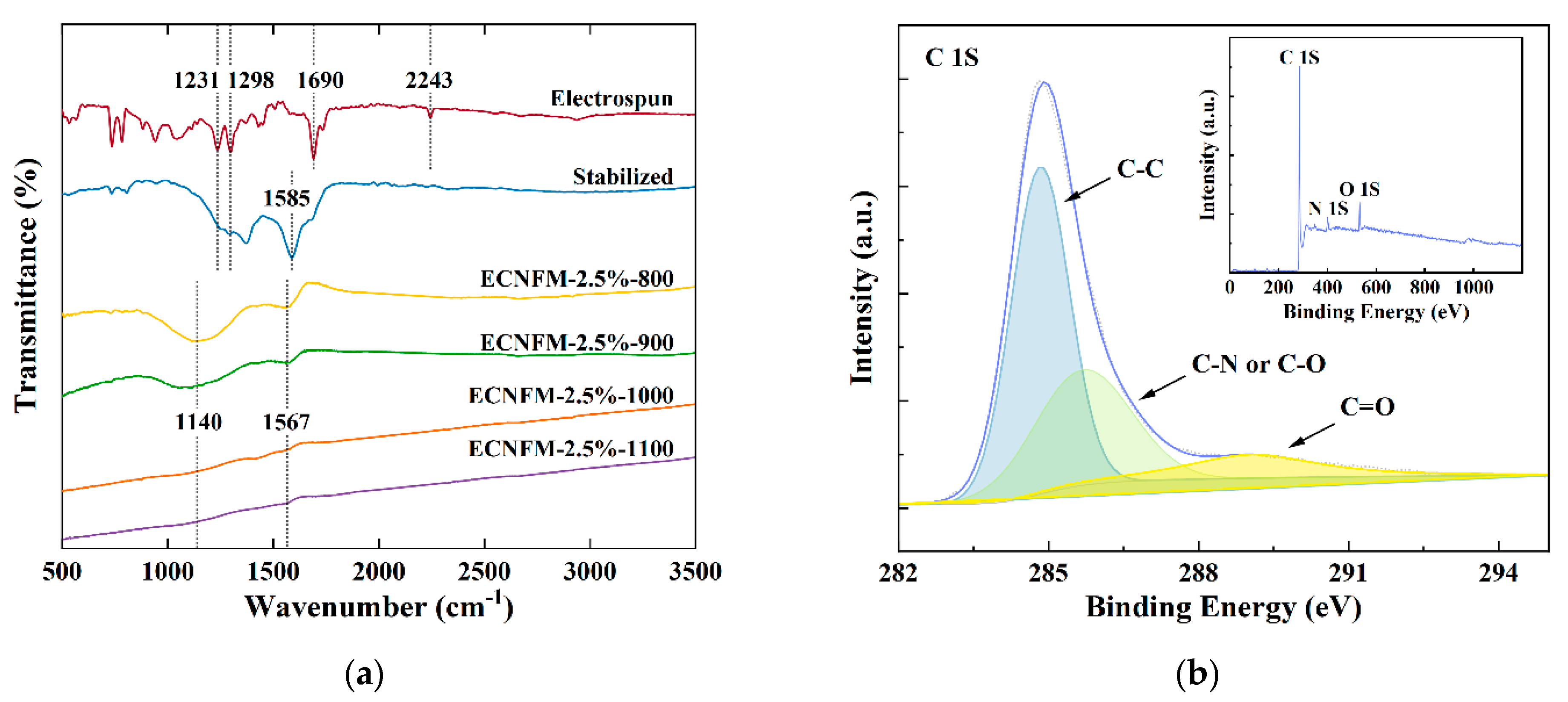
3.1. Structure, Morphology and Electrochemical Characteristics of ECNFM
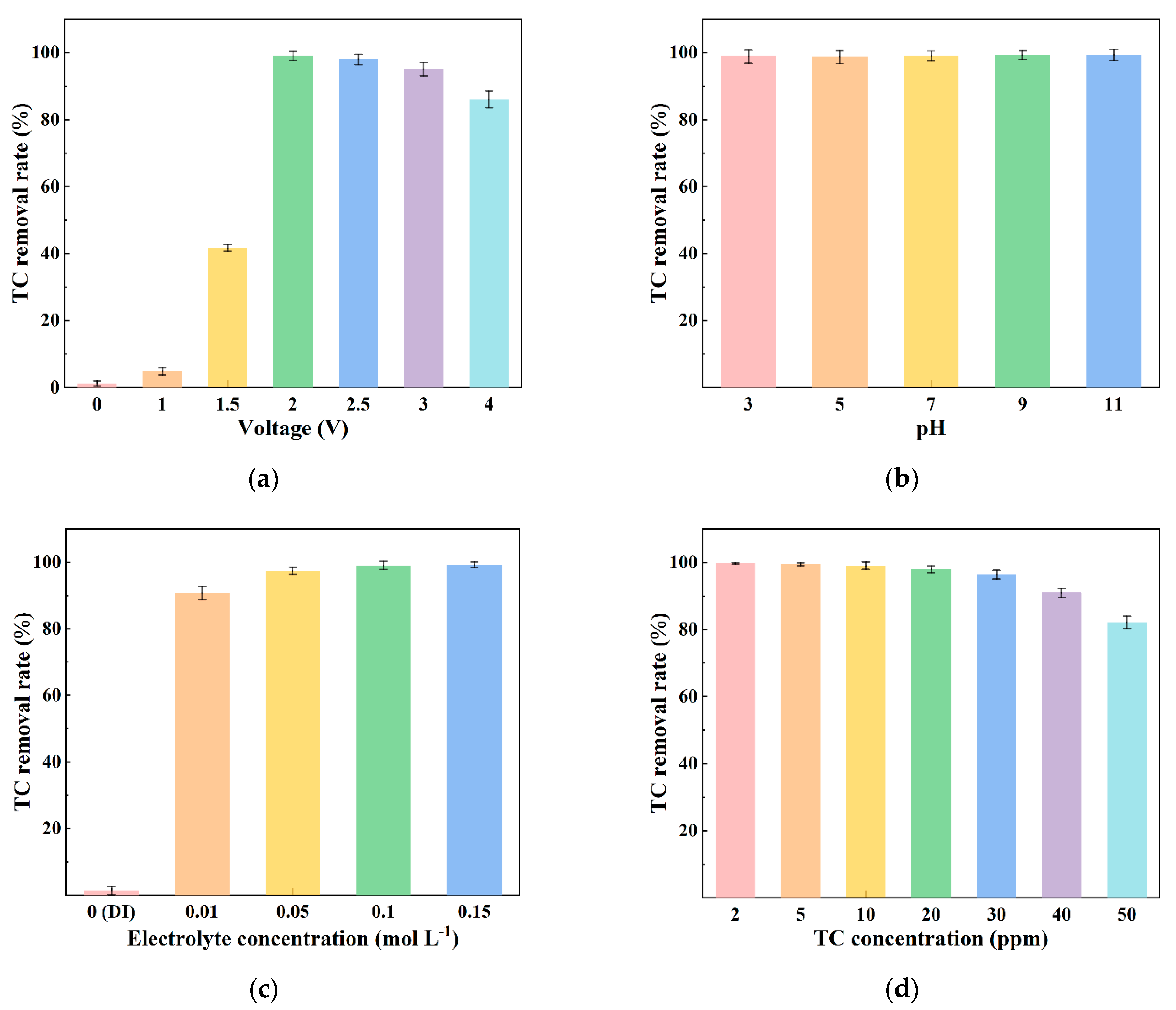
3.2. Effect of Operational Parameters on TC Removal during Electrochemical Filtration
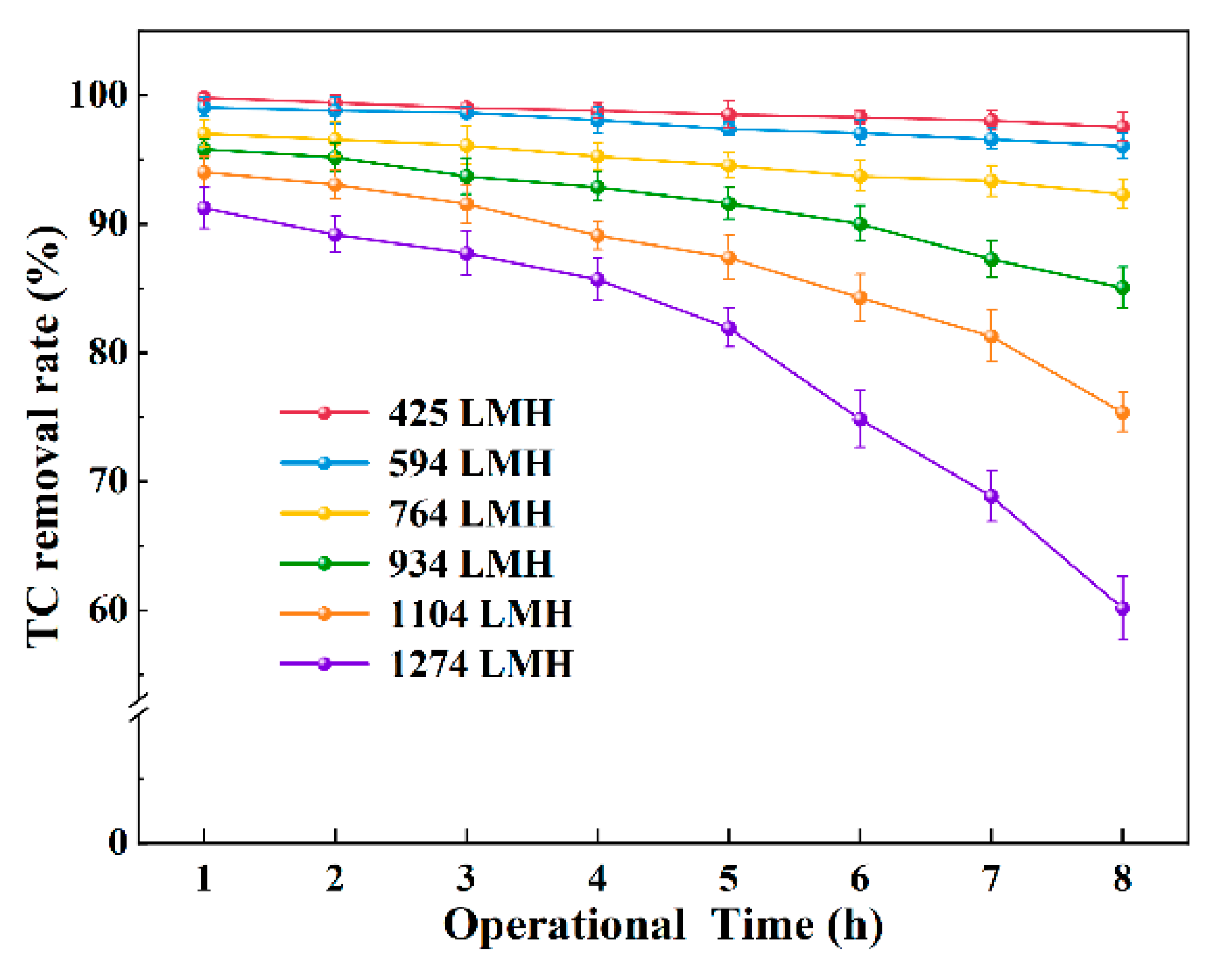
3.3. Long-Term Operation Performance for TC Removal
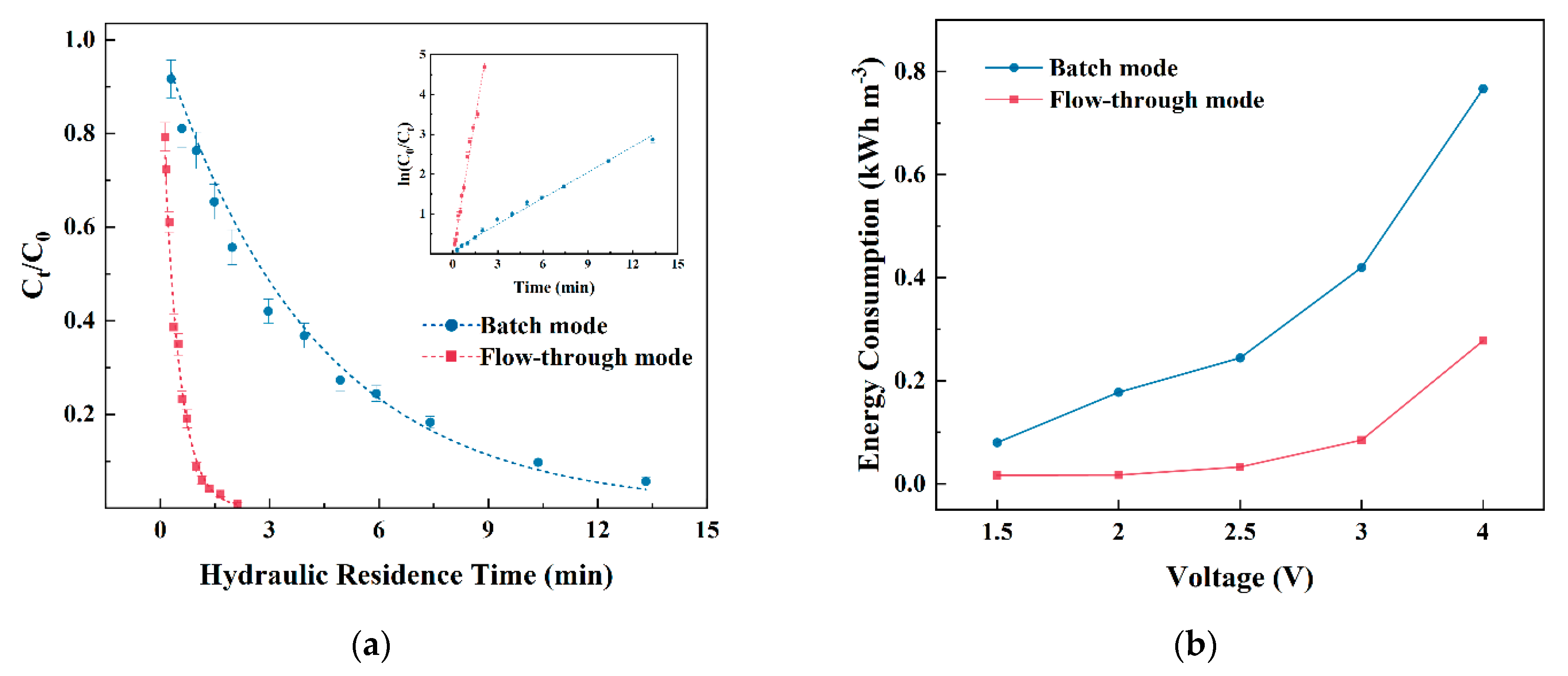
3.4. Comparison of Efficacy and Energy Consumption between Flow-Through and Batch Mode
3.5. The Removal Mechanism of TC
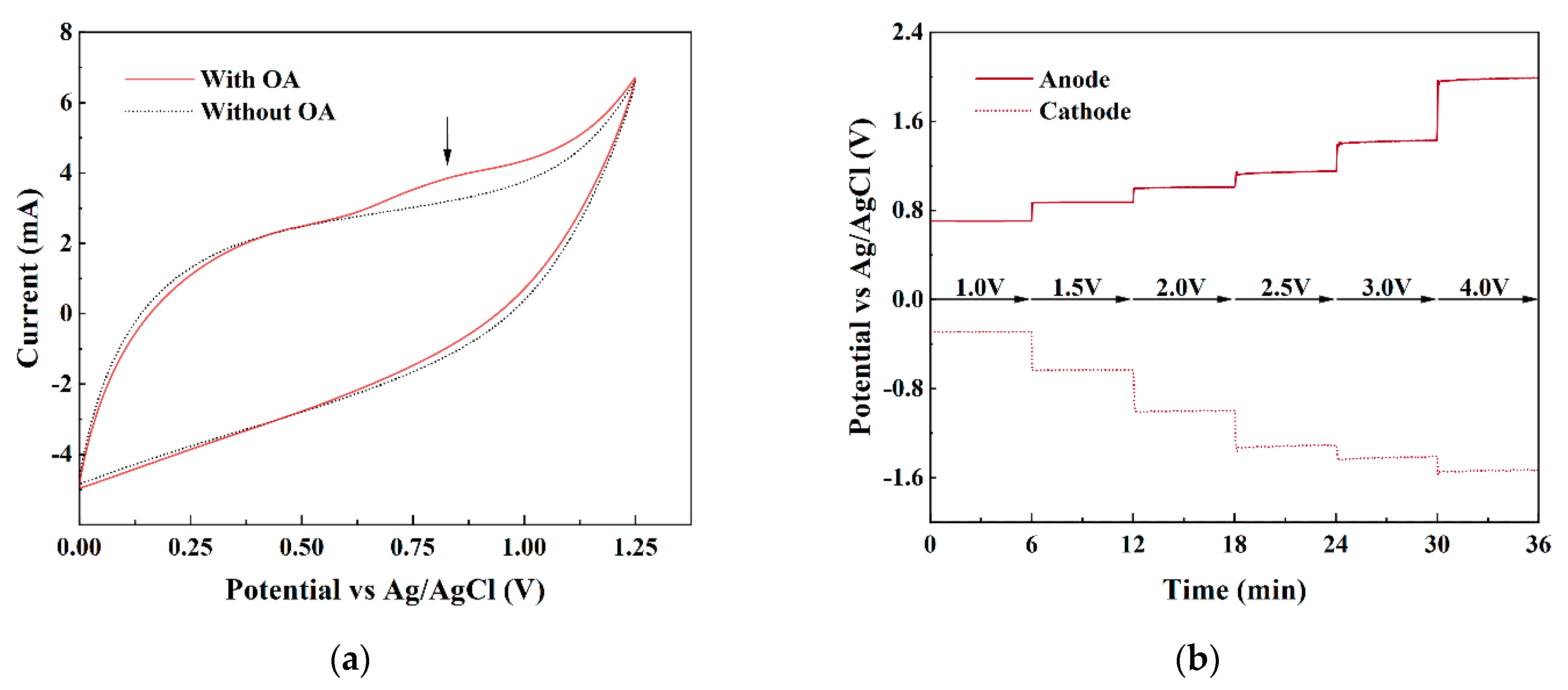
3.5.1. Electrochemical Oxidation Mechanism
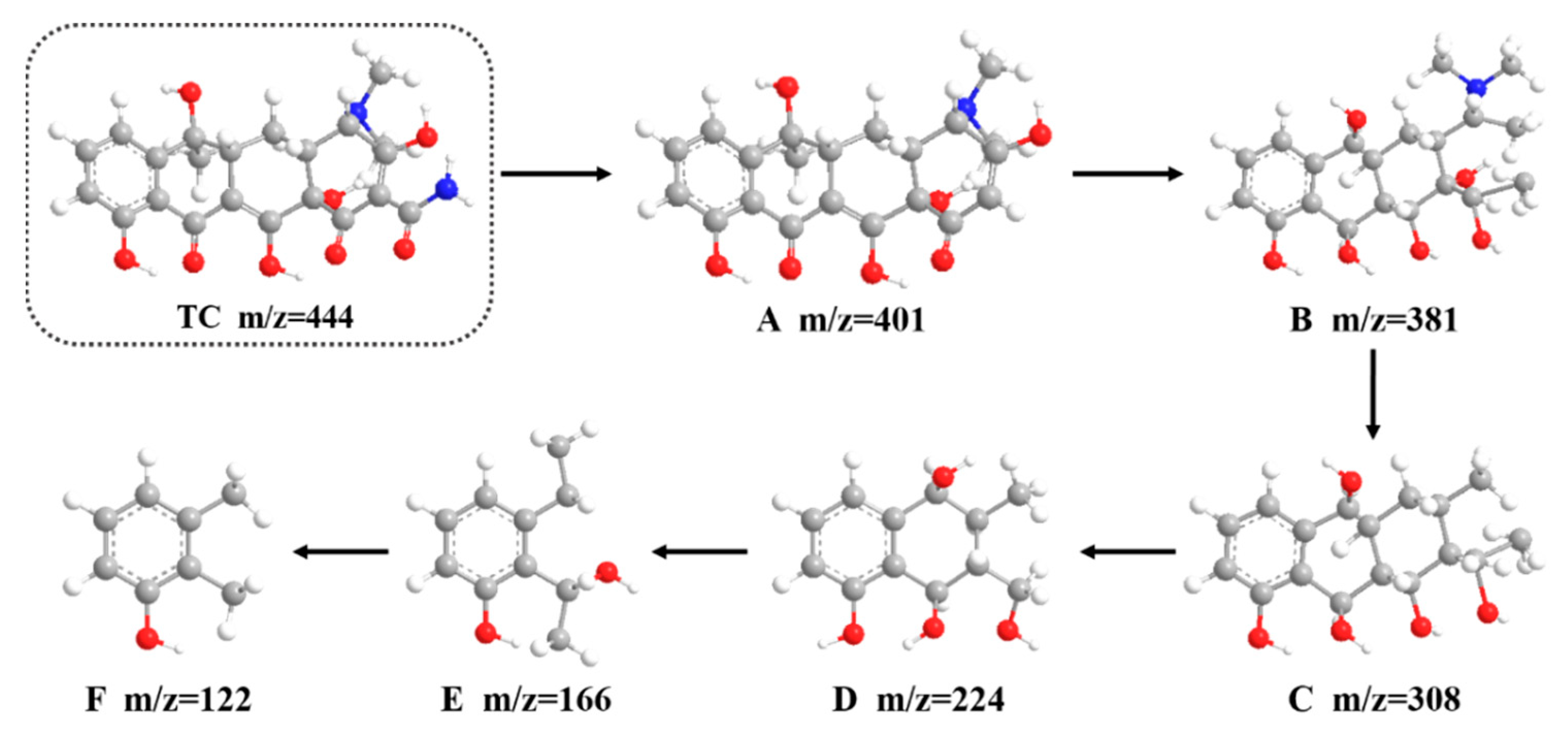
3.5.2. Proposed TC Degradation Pathway and Safety Evaluation
3.5.3. Removal Mechanism of TC by ECNFM-Based Electrochemical Filter
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kovaláková, P.; Cizmas, L.; McDonald, T.J.; Marsalek, B.; Feng, M.; Sharma, V.K. Occurrence and toxicity of antibiotics in the aquatic environment: A review. Chemosphere 2020, 251, 126351. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Zhuan, R. Degradation of antibiotics by advanced oxidation processes: An overview. Sci. Total Environ. 2020, 701, 135023. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Chen, Y.; Feng, M.; Chen, J.; Shen, W.; Zhang, S. Occurrence of antibiotics and antibiotic resistance genes and their correlations in river-type drinking water source, China. Environ. Sci. Pollut. Res. 2021, 28, 42339–42352. [Google Scholar] [CrossRef] [PubMed]
- He, P.; Wu, J.; Peng, J.; Wei, L.; Zhang, L.; Zhou, Q.; Wu, Z. Pharmaceuticals in drinking water sources and tap water in a city in the middle reaches of the Yangtze River: Occurrence, spatiotemporal distribution, and risk assessment. Environ. Sci. Pollut. Res. 2021, 29, 2365–2374. [Google Scholar] [CrossRef] [PubMed]
- Benotti, M.J.; Trenholm, R.A.; Vanderford, B.J.; Holady, J.C.; Stanford, B.D.; Snyder, S. Pharmaceuticals and endocrine disrupting compounds in US drinking water. Environ. Sci. Technol. 2009, 43, 597–603. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Padhye, L.; Yao, H.; Kung’U, F.T.; Huang, C.-H. Year-long evaluation on the occurrence and fate of pharmaceuticals, personal care products, and endocrine disrupting chemicals in an urban drinking water treatment plant. Water Res. 2014, 51, 266–276. [Google Scholar] [CrossRef]
- Reichert, G.; Hilgert, S.; Fuchs, S.; Azevedo, J.C.R. Emerging contaminants and antibiotic resistance in the different environmental matrices of Latin America. Environ. Pollut. 2019, 255, 113140. [Google Scholar] [CrossRef] [PubMed]
- Watkinson, A.; Murby, E.; Costanzo, S. Removal of antibiotics in conventional and advanced wastewater treatment: Implications for environmental discharge and wastewater recycling. Water Res. 2007, 41, 4164–4176. [Google Scholar] [CrossRef] [PubMed]
- Vieno, N.M.; Härkki, H.; Tuhkanen, T.; Kronberg, L. Occurrence of pharmaceuticals in river water and their elimination in a pilot-scale drinking water treatment plant. Environ. Sci. Technol. 2007, 41, 5077–5084. [Google Scholar] [CrossRef] [PubMed]
- Vieno, N.; Tuhkanen, T.; Kronberg, L. Removal of pharmaceuticals in drinking water treatment: Effect of chemical coagulation. Environ. Technol. 2006, 27, 183–192. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Sun, Q.; Zhang, C.; Li, H.; Song, W.; Zhang, N.; Jia, X. Removal of typical antibiotics in the advanced treatment process of productive drinking water. Desalin. Water Treat. 2016, 57, 11386–11391. [Google Scholar] [CrossRef]
- Xiang, Y.; Xu, Z.; Wei, Y.; Zhou, Y.; Yang, X.; Yang, Y.; Yang, J.; Zhang, J.; Luo, L.; Zhou, Z. Carbon-based materials as adsorbent for antibiotics removal: Mechanisms and influencing factors. J. Environ. Manag. 2019, 237, 128–138. [Google Scholar] [CrossRef] [PubMed]
- Xiao, C.; Li, X.; Li, Q.; Hu, Y.; Cheng, J.; Chen, Y. Ni-doped FeC2O4 for efficient photo-Fenton simultaneous degradation of organic pollutants and reduction of Cr(VI): Accelerated Fe(III)/Fe(II) cycle, enhanced stability and mechanism insight. J. Clean. Prod. 2022, 340, 130775. [Google Scholar] [CrossRef]
- Yang, G.; Zhang, D.; Wang, C.; Liu, H.; Qu, L.; Li, H. A novel nanocomposite membrane combining BN nanosheets and GO for effective removal of antibiotic in water. Nanomaterials 2019, 9, 386. [Google Scholar] [CrossRef] [Green Version]
- Huang, A.; Yan, M.; Lin, J.; Xu, L.; Gong, H.; Gong, H. A review of processes for removing antibiotics from breeding wastewater. Int. J. Environ. Res. Public Health 2021, 18, 4909. [Google Scholar] [CrossRef] [PubMed]
- Cristóvão, M.B.; Tela, S.; Silva, A.F.; Oliveira, M.; Bento-Silva, A.; Bronze, M.R.; Crespo, M.T.B.; Crespo, J.G.; Nunes, M.; Pereira, V.J. Occurrence of antibiotics, antibiotic resistance genes and viral genomes in wastewater effluents and their treatment by a pilot scale nanofiltration unit. Membranes 2021, 11, 9. [Google Scholar] [CrossRef] [PubMed]
- Alonso, J.J.S.; El Kori, N.; Melián-Martel, N.; Del Río-Gamero, B. Removal of ciprofloxacin from seawater by reverse osmosis. J. Environ. Manag. 2018, 217, 337–345. [Google Scholar] [CrossRef] [PubMed]
- Pan, Z.; Yu, F.; Li, L.; Song, C.; Yang, J.; Wang, C.; Pan, Y.; Wang, T. Electrochemical microfiltration treatment of bisphenol A wastewater using coal-based carbon membrane. Sep. Purif. Technol. 2019, 227, 115695. [Google Scholar] [CrossRef]
- Li, C.; Zhang, M.; Song, C.; Tao, P.; Sun, M.; Shao, M.; Wang, T. Enhanced treatment ability of membrane technology by integrating an electric field for dye wastewater treatment: A review. J. AOAC Int. 2018, 101, 1341–1352. [Google Scholar] [CrossRef]
- Li, Z.; Shen, C.; Liu, Y.; Ma, C.; Li, F.; Yang, B.; Huang, M.; Wang, Z.; Dong, L.; Wolfgang, S. Carbon nanotube filter functionalized with iron oxychloride for flow-through electro-Fenton. Appl. Catal. B Environ. 2020, 260, 118204. [Google Scholar] [CrossRef]
- Liu, Y.; Liu, H.; Zhou, Z.; Wang, T.; Ong, C.N.; Vecitis, C.D. Degradation of the common aqueous antibiotic tetracycline using a carbon nanotube electrochemical filter. Environ. Sci. Technol. 2015, 49, 7974–7980. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.; Wang, Z.; Ma, J.; Xu, S.; Wu, Z. Development of an electrochemical ceramic membrane filtration system for efficient contaminant removal from waters. Environ. Sci. Technol. 2018, 52, 4117–4126. [Google Scholar] [CrossRef]
- Misal, S.N.; Lin, M.-H.; Mehraeen, S.; Chaplin, B.P. Modeling electrochemical oxidation and reduction of sulfamethoxazole using electrocatalytic reactive electrochemical membranes. J. Hazard. Mater. 2020, 384, 121420. [Google Scholar] [CrossRef]
- Cunha, G.D.S.; de Souza-Chaves, B.M.; Bila, D.; Bassin, J.; Vecitis, C.D.; Dezotti, M. Insights into estrogenic activity removal using carbon nanotube electrochemical filter. Sci. Total Environ. 2019, 678, 448–456. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Zhang, X.; Liu, Z.; Deng, C.; Xu, H.; Wang, Y.; Zhu, M. Carbon nanotube-based electrocatalytic filtration membrane for continuous degradation of flow-through Bisphenol A. Sep. Purif. Technol. 2021, 265, 118503. [Google Scholar] [CrossRef]
- Trellu, C.; Coetsier, C.; Rouch, J.-C.; Esmilaire, R.; Rivallin, M.; Cretin, M.; Causserand, C. Mineralization of organic pollutants by anodic oxidation using reactive electrochemical membrane synthesized from carbothermal reduction of TiO2. Water Res. 2018, 131, 310–319. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, Y.; Mei, J.; Shen, C.; Huang, M.; Yang, M.; Wang, Z.; Sand, W.; Li, F. Rapid and selective electrochemical transformation of ammonia to N2 by substoichiometric TiO2-based electrochemical system. RSC Adv. 2020, 10, 1219–1225. [Google Scholar] [CrossRef] [Green Version]
- Li, D.; Tang, J.; Zhou, X.; Li, J.; Sun, X.; Shen, J.; Wang, L.; Han, W. Electrochemical degradation of pyridine by Ti/SnO2–Sb tubular porous electrode. Chemosphere 2016, 149, 49–56. [Google Scholar] [CrossRef]
- Yang, K.; Lin, H.; Liang, S.; Xie, R.; Lv, S.; Niu, J.; Chen, J.; Hu, Y. A reactive electrochemical filter system with an excellent penetration flux porous Ti/SnO2–Sb filter for efficient contaminant removal from water. RSC Adv. 2018, 8, 13933–13944. [Google Scholar] [CrossRef] [Green Version]
- Liu, Z.; Zhu, M.; Zhao, L.; Deng, C.; Ma, J.; Wang, Z.; Liu, H.; Wang, H. Aqueous tetracycline degradation by coal-based carbon electrocatalytic filtration membrane: Effect of nano antimony-doped tin dioxide coating. Chem. Eng. J. 2017, 314, 59–68. [Google Scholar] [CrossRef]
- Gao, G.; Zhang, Q.; Vecitis, C.D. CNT–PVDF composite flow-through electrode for single-pass sequential reduction–oxidation. J. Mater. Chem. A 2014, 2, 6185–6190. [Google Scholar] [CrossRef]
- Peng, L.; Liu, H.; Wang, W.-L.; Xu, Z.-B.; Ni, X.-Y.; Wu, Y.-H.; Wu, Q.-Y.; Hu, H.-Y. Degradation of methylisothiazolinone biocide using a carbon fiber felt-based flow-through electrode system (FES) via anodic oxidation. Chem. Eng. J. 2020, 384, 123239. [Google Scholar] [CrossRef]
- Yu, S.; Gao, Y.; Khan, R.; Liang, P.; Zhang, X.; Huang, X. Electrospun PAN-based graphene/SnO2 carbon nanofibers as anodic electrocatalysis microfiltration membrane for sulfamethoxazole degradation. J. Membr. Sci. 2020, 614, 118368. [Google Scholar] [CrossRef]
- Nataraj, S.; Yang, K.; Aminabhavi, T. Polyacrylonitrile-based nanofibers—A state-of-the-art review. Prog. Polym. Sci. 2012, 37, 487–513. [Google Scholar] [CrossRef]
- Li, X.; Shao, S.; Yang, Y.; Mei, Y.; Qing, W.; Guo, H.; Peng, L.E.; Wang, P.; Tang, C.Y. Engineering interface with a one-dimensional RuO2/TiO2 heteronanostructure in an electrocatalytic membrane electrode: Toward highly efficient micropollutant decomposition. ACS Appl. Mater. Interfaces 2020, 12, 21596–21604. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Cao, C.-Y.; Wei, F.-F.; Huang, P.-P.; Sun, Y.-B.; Jiang, L.; Song, W.-G. Flexible macroporous carbon nanofiber film with high oil adsorption capacity. J. Mater. Chem. A 2014, 2, 3557–3562. [Google Scholar] [CrossRef]
- Xie, L.; Shu, Y.; Hu, Y.; Cheng, J.; Chen, Y. SWNTs-PAN/TPU/PANI composite electrospun nanofiber membrane for point-of-use efficient electrochemical disinfection: New strategy of CNT disinfection. Chemosphere 2020, 251, 126286. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Zeng, L.; Yang, Z.; Gu, L.; Wang, J.; Liu, X.; Cheng, J.; Yu, Y. Free-standing and binder-free sodium-ion electrodes with ultralong cycle life and high rate performance based on porous carbon nanofibers. Nanoscale 2014, 6, 693–698. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Zhou, M.; Wang, J.; Ge, F.; Zhao, Y.; Komarneni, S.; Cai, Z. Flexible and internal series-connected supercapacitors with high working voltage using ultralight porous carbon nanofilms. J. Power Sources 2017, 342, 762–771. [Google Scholar] [CrossRef]
- Tang, Y.; Li, N.; Liu, A.; Ding, S.; Yi, C.; Liu, H. Effect of spinning conditions on the structure and performance of hydrophobic PVDF hollow fiber membranes for membrane distillation. Desalination 2012, 287, 326–339. [Google Scholar] [CrossRef]
- Trellu, C.; Chaplin, B.P.; Coetsier, C.; Esmilaire, R.; Cerneaux, S.; Causserand, C.; Cretin, M. Electro-oxidation of organic pollutants by reactive electrochemical membranes. Chemosphere 2018, 208, 159–175. [Google Scholar] [CrossRef] [Green Version]
- Liu, K.; Chen, L.; Chen, Y.; Wu, J.L.; Zhang, W.Y.; Chen, F.; Fu, Q. Preparation of polyester/reduced graphene oxide composites via in situ melt polycondensation and simultaneous thermo-reduction of graphene oxide. J. Mater. Chem. 2011, 21, 8612–8617. [Google Scholar] [CrossRef]
- Arbab, S.; Teimoury, A.; Mirbaha, H.; Adolphe, D.C.; Noroozi, B.; Nourpanah, P. Optimum stabilization processing parameters for polyacrylonitrile-based carbon nanofibers and their difference with carbon (micro) fibers. Polym. Degrad. Stab. 2017, 142, 198–208. [Google Scholar] [CrossRef]
- Zhang, L.; Aboagye, A.; Kelkar, A.D.; Lai, C.; Fong, H. A review: Carbon nanofibers from electrospun polyacrylonitrile and their applications. J. Mater. Sci. 2014, 49, 463–480. [Google Scholar] [CrossRef]
- Yu, M.; Xu, Y.; Wang, C.; Hu, X.; Zhu, B.; Qiao, K.; Yuan, H. Heredity and difference of multiple-scale microstructures in PAN-based carbon fibers and their precursor fibers. J. Appl. Polym. Sci. 2012, 125, 3159–3166. [Google Scholar] [CrossRef]
- Xu, W.; Xin, B.; Yang, X. Carbonization of electrospun polyacrylonitrile (PAN)/cellulose nanofibril (CNF) hybrid membranes and its mechanism. Cellulose 2020, 27, 3789–3804. [Google Scholar] [CrossRef]
- Li, X.; Wang, J.; Zhao, Y.; Ge, F.; Komarneni, S.; Cai, Z. Wearable solid-state supercapacitors operating at high working voltage with a flexible nanocomposite electrode. ACS Appl. Mater. Interfaces 2016, 8, 25905–25914. [Google Scholar] [CrossRef]
- Yang, T.; Du, M.; Zhu, H.; Zhang, M.; Zou, M. Immobilization of Pt nanoparticles in carbon nanofibers: Bifunctional catalyst for hydrogen evolution and electrochemical sensor. Electrochim. Acta 2015, 167, 48–54. [Google Scholar] [CrossRef]
- Li, X.; Xiao, C.; Ruan, X.; Hu, Y.; Zhang, C.; Cheng, J.; Chen, Y. Enrofloxacin degradation in a heterogeneous electro-Fenton system using a tri-metal-carbon nanofibers composite cathode. Chem. Eng. J. 2022, 427, 130927. [Google Scholar] [CrossRef]
- Xie, T.; Hu, H.; Chen, D.; Sun, P. Electrochemical degradation of tetracycline hydrochloride in aqueous medium by (B4C/C)-β-PbO2 electrode. Bull. Korean Chem. Soc. 2017, 38, 756–762. [Google Scholar] [CrossRef]
- Zhang, H.; Liu, F.; Wu, X.; Zhang, J.; Zhang, D. Degradation of tetracycline in aqueous medium by electrochemical method. Asia-Pac. J. Chem. Eng. 2009, 4, 568–573. [Google Scholar] [CrossRef]
- Schnoor, M.H.; Vecitis, C.D. Quantitative examination of aqueous ferrocyanide oxidation in a carbon nanotube electrochemical filter: Effects of flow rate, ionic strength, and cathode material. J. Phys. Chem. C 2013, 117, 2855–2867. [Google Scholar] [CrossRef]
- Yang, S.; Liu, Y.; Shen, C.; Li, F.; Yang, B.; Huang, M.; Yang, M.; Wang, Z.; Sand, W. Rapid decontamination of tetracycline hydrolysis product using electrochemical CNT filter: Mechanism, impacting factors and pathways. Chemosphere 2020, 244, 125525. [Google Scholar] [CrossRef] [PubMed]
- Panizza, M.; Cerisola, G. Direct and mediated anodic oxidation of organic pollutants. Chem. Rev. 2009, 109, 6541–6569. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Zhi, D.; Zhou, H.; He, X.; Zhang, D. Evaluating tetracycline degradation pathway and intermediate toxicity during the electrochemical oxidation over a Ti/Ti4O7 anode. Water Res. 2018, 137, 324–334. [Google Scholar] [CrossRef] [PubMed]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, X.; Li, X.; Hu, Y.; Cheng, J.; Chen, Y. A High Flux Electrochemical Filtration System Based on Electrospun Carbon Nanofiber Membrane for Efficient Tetracycline Degradation. Water 2022, 14, 910. https://doi.org/10.3390/w14060910
Yang X, Li X, Hu Y, Cheng J, Chen Y. A High Flux Electrochemical Filtration System Based on Electrospun Carbon Nanofiber Membrane for Efficient Tetracycline Degradation. Water. 2022; 14(6):910. https://doi.org/10.3390/w14060910
Chicago/Turabian StyleYang, Xue, Xian Li, Yongyou Hu, Jianhua Cheng, and Yuancai Chen. 2022. "A High Flux Electrochemical Filtration System Based on Electrospun Carbon Nanofiber Membrane for Efficient Tetracycline Degradation" Water 14, no. 6: 910. https://doi.org/10.3390/w14060910