Novel Magnetite Nanocomposites (Fe3O4/C) for Efficient Immobilization of Ciprofloxacin from Aqueous Solutions through Adsorption Pretreatment and Membrane Processes
Abstract
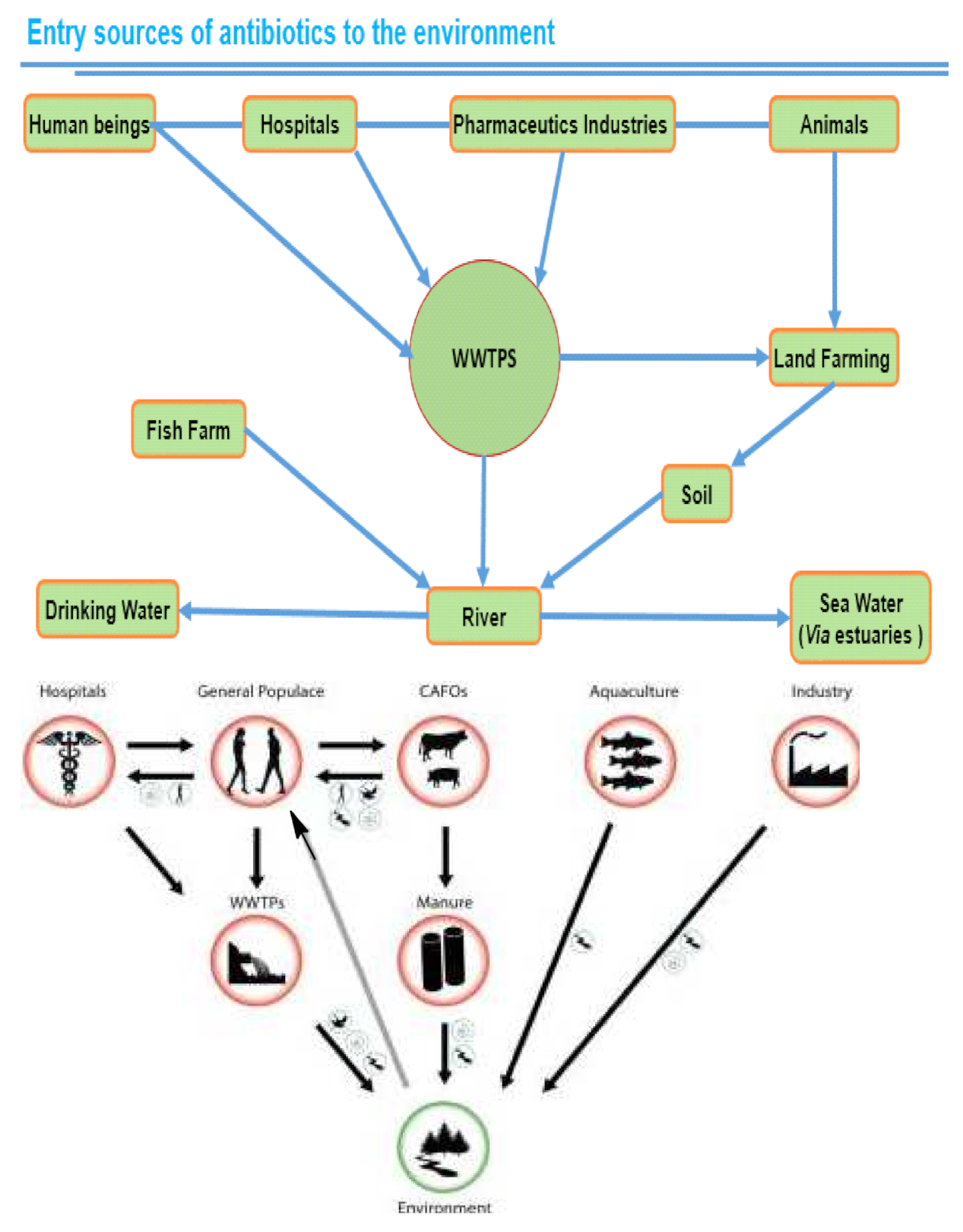
:1. Introduction
2. Materials and Methods
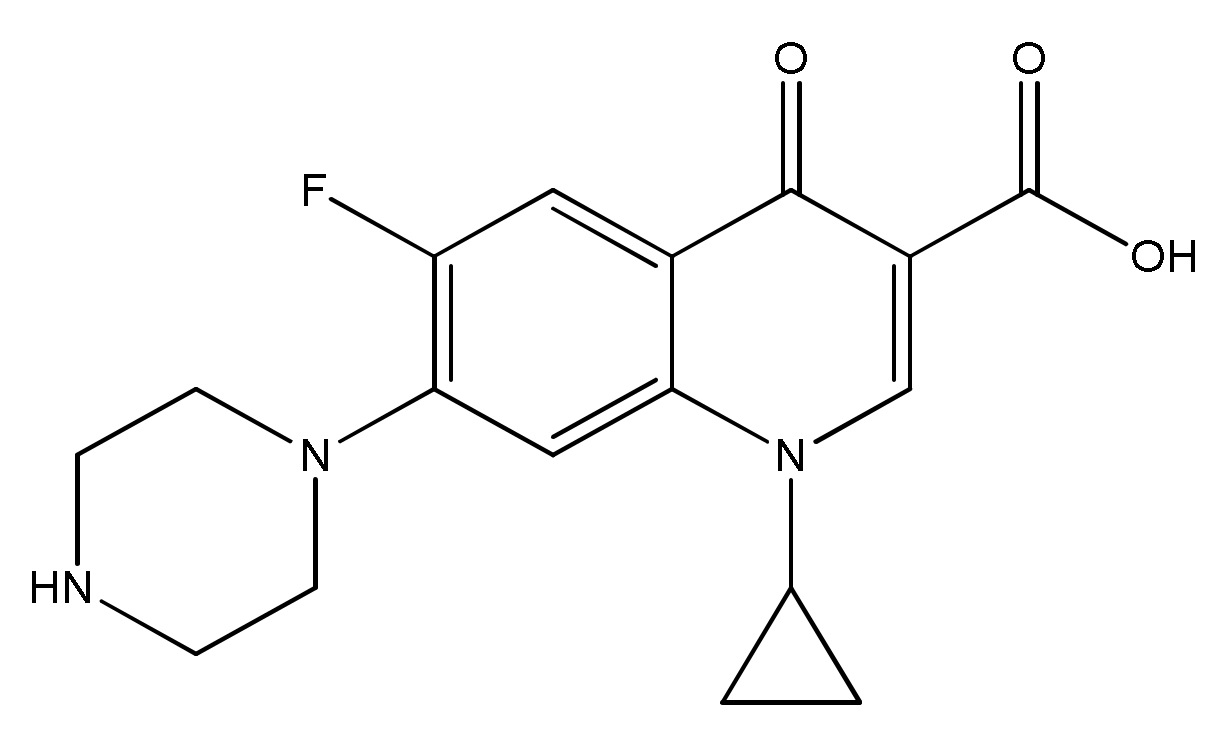
2.1. Reagents and Materials
2.2. Preparation of Magnetic Carbon Nanocomposites (Fe3O4/C)
2.3. Characterization of Fe3O4/C Composite
2.4. Batch Sorption Experiments
2.5. Equations and Model Used in the Study
2.6. Membranes’ Operation with and without Magnetic Composite
3. Results and Discussion
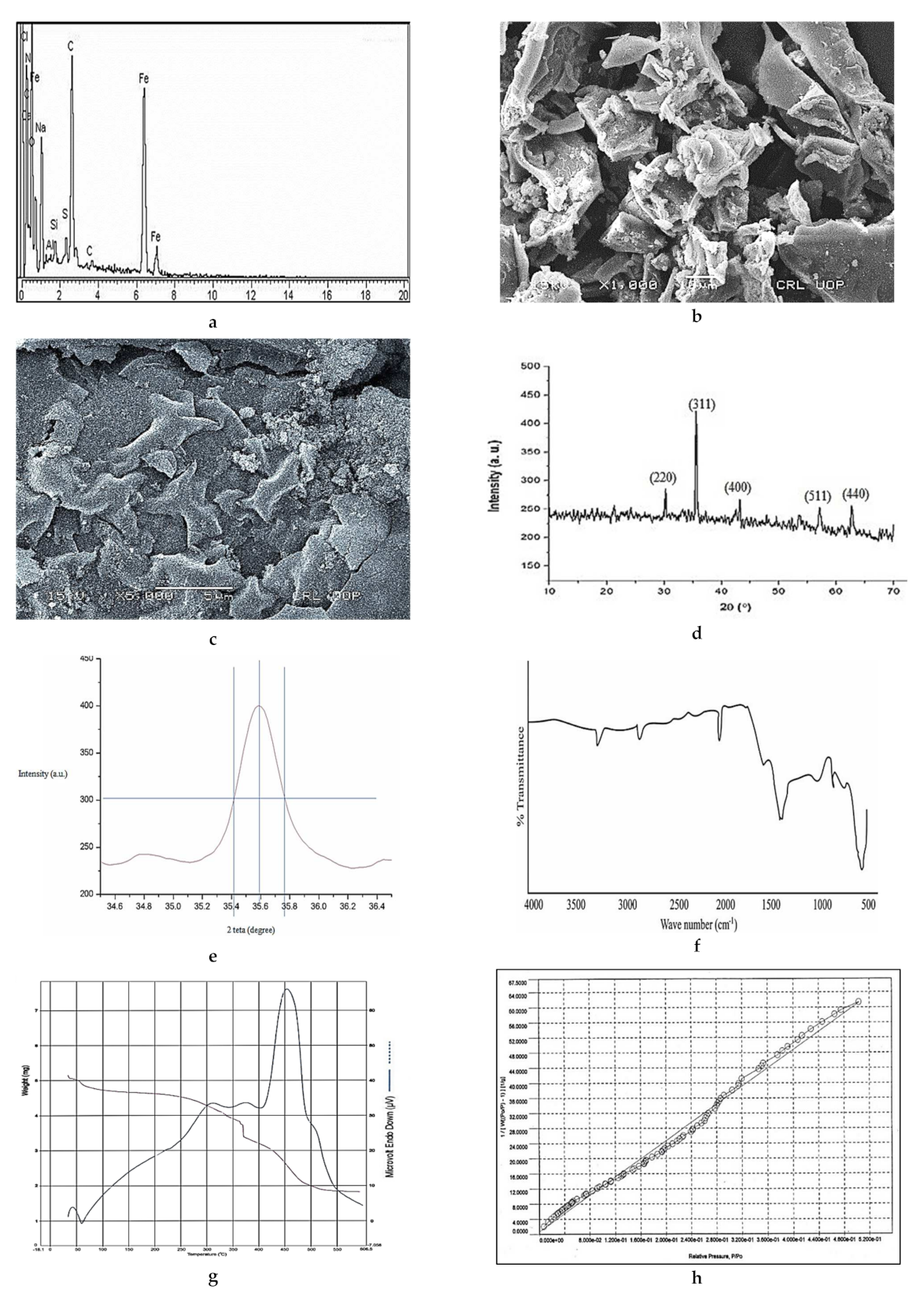
3.1. Characterization of the Fe3O4/C Adsorbent
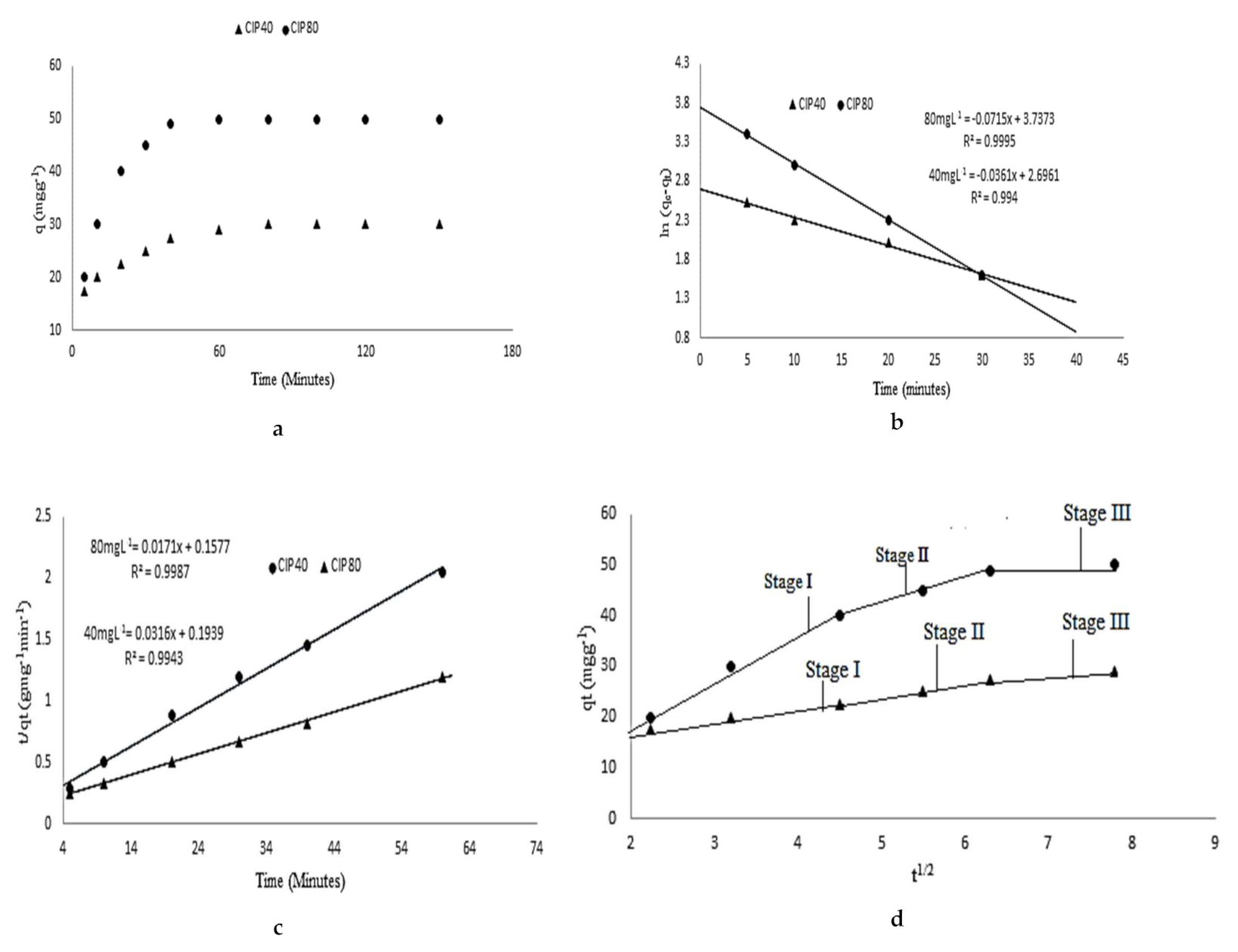
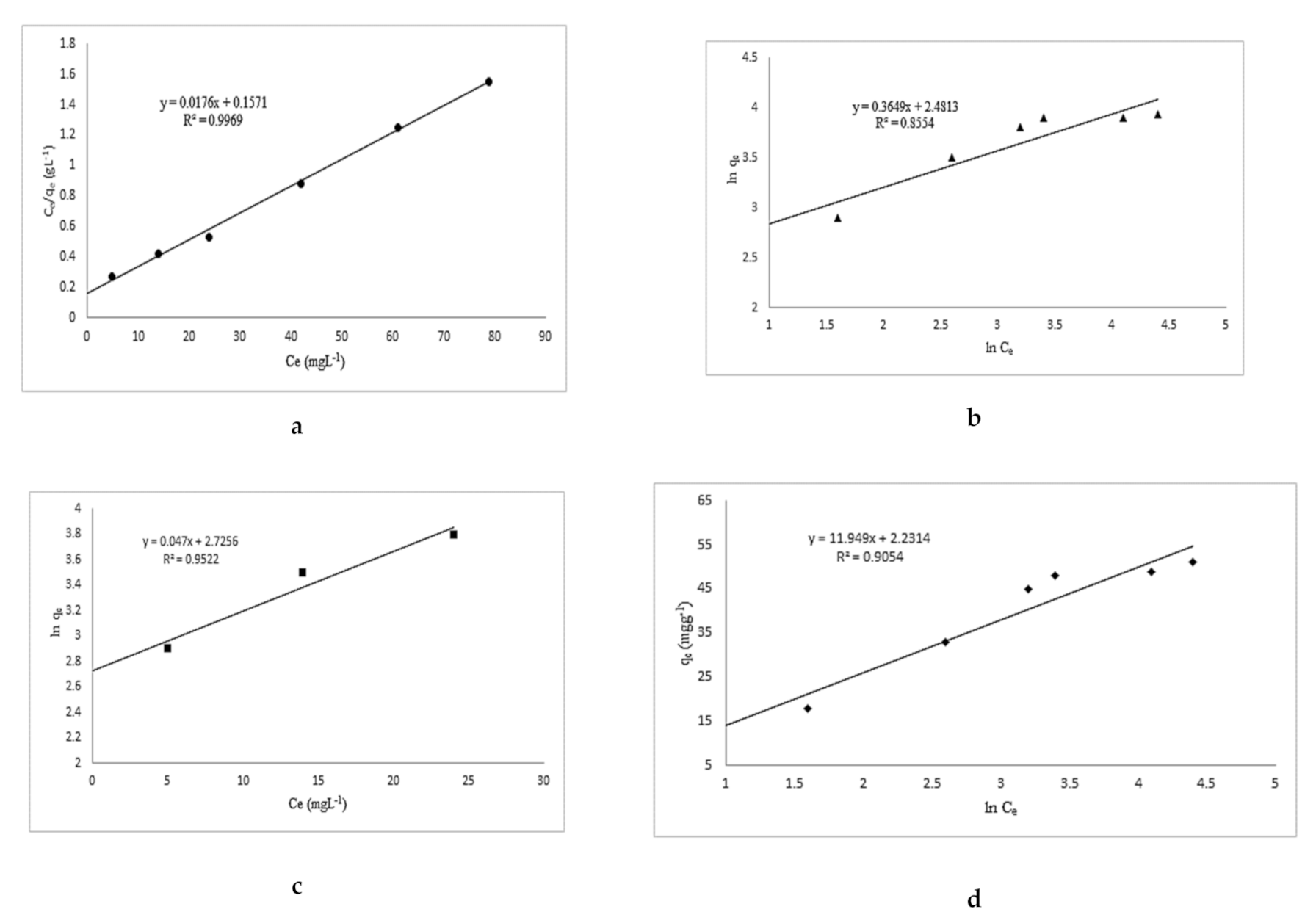
3.2. Isothermal and Kinetics Studies
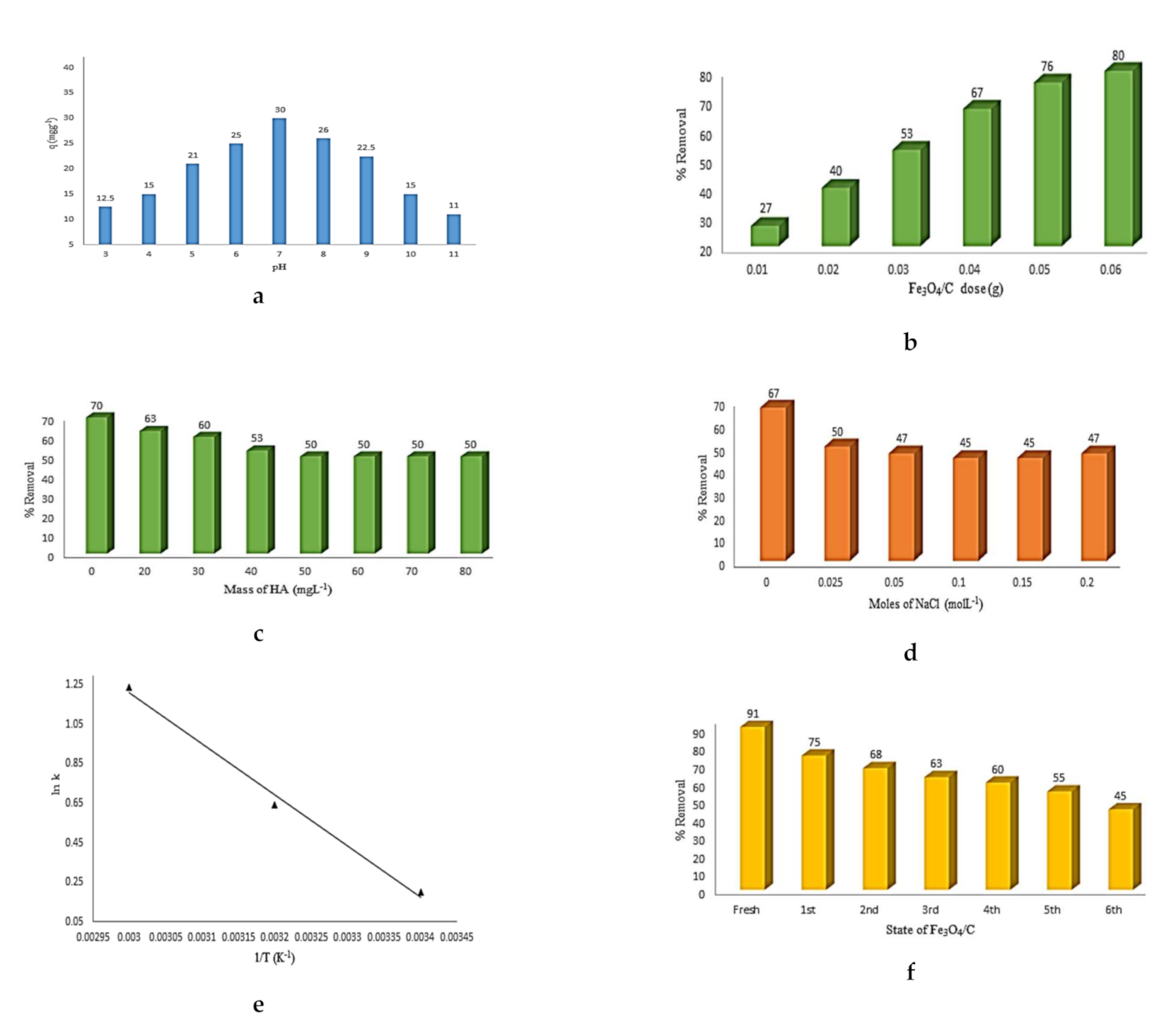
3.3. The Effect of pH and Adsorbent Dosage on the Adsorption of the CIPRO Molecules
3.4. Effects of Hums (Humic Acid) on CIPRO Adsorption
3.5. Effect of Ionic Strength (Sodium Chloride) on the CIPRO Adsorption
3.6. Thermodynamics Parameters
3.7. Reusability/Regeneration and Recycling of Fe3O4/C
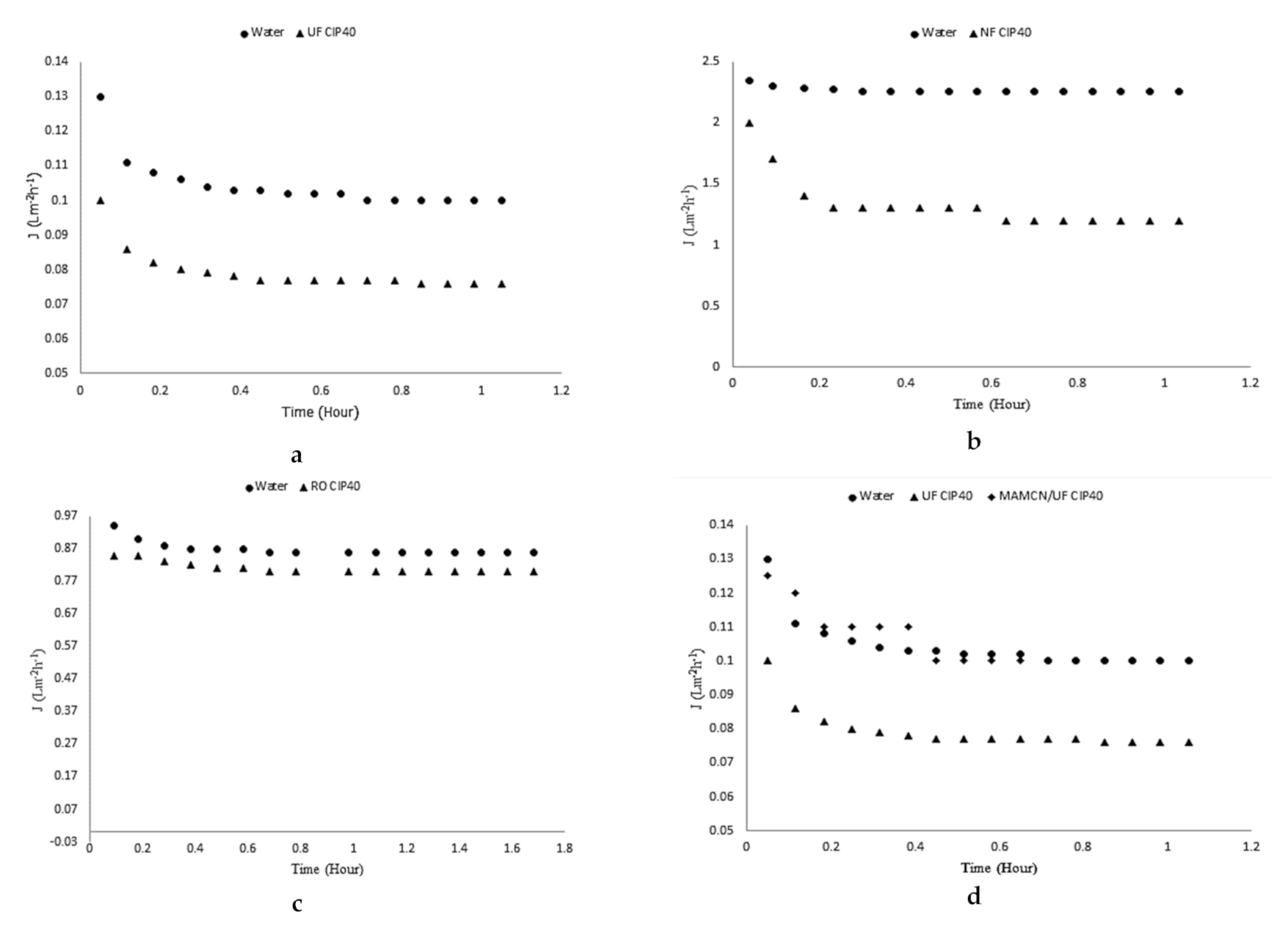
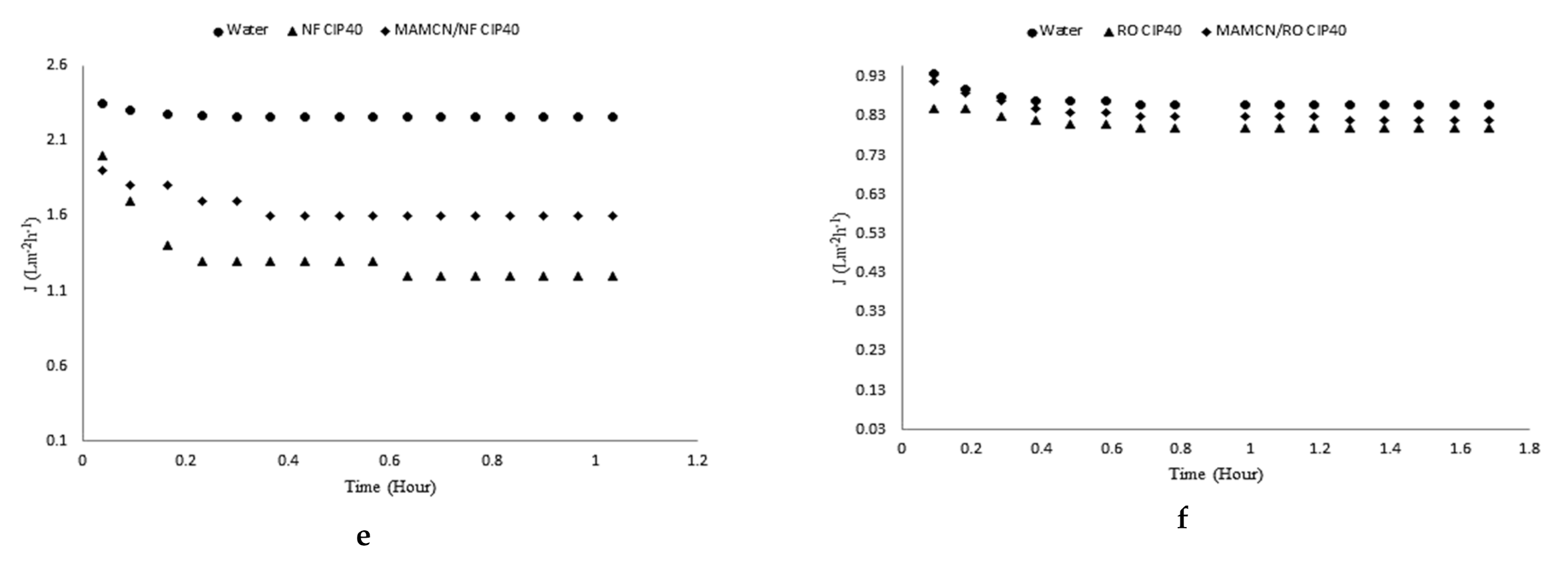
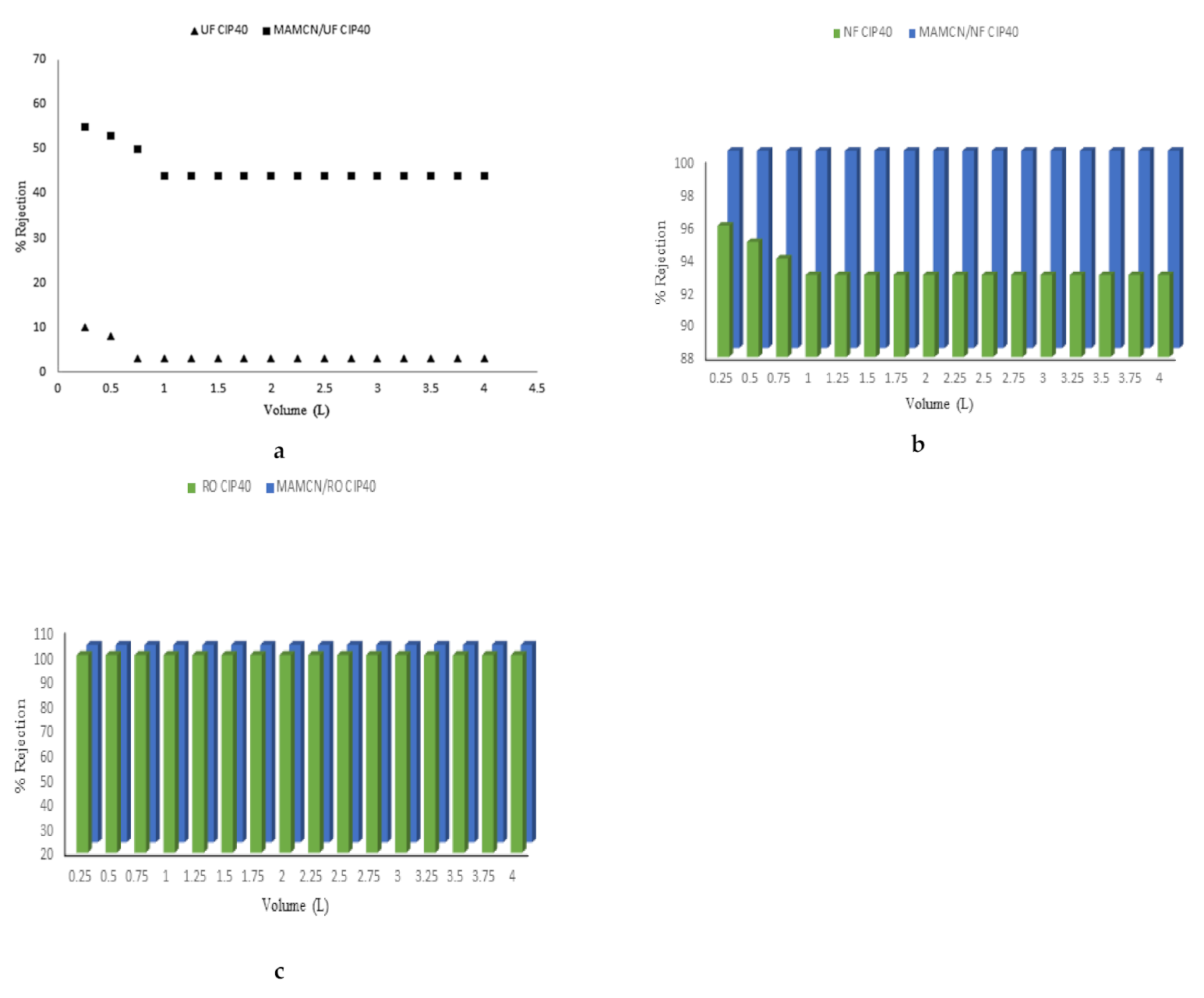
3.8. Membranes and Adsorption/Membrane Hybrid Processes
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sousa, A.R.; Ribeiro, M.O.; Barbosa, M.F.R.; Pereira, A.M.; Silva, A.M.T. A review on environmental monitoring of water organic pollutants identified by EU guidelines. J. Hazard. Mater. 2018, 344, 146–162. [Google Scholar] [CrossRef]
- Feng, X.; Wang, J.; Chen, R.; Qu, Y.; Sui, L.; Cizmas, Z.; Wang Sharma, V.K. Degradation of fluoroquinolone antibiotics by ferrate (VI): Effects of water constituents and oxidized products. Water Res. 2016, 103, 48–57. [Google Scholar] [CrossRef] [PubMed]
- Vithanage, M.; Rajapaksa, A.U.; Tang, X.; Thiele-Bruhn, S.; Kim, K.H.; Lee, S.E.; Ok, Y.S. Sorption and transport of sulfamethazine in agricultural soils amended with invasive-plant-derived biochar. J. Environ. Manag. 2014, 141, 95–103. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Lu, M.; Wan, J.; Sun, Y.; Lan, H.; Deng, X. Effects of pH, dissolved humic acid and Cu2+ on the adsorption of norfloxacin on montmorillonite-biochar composite derived from wheat straw. Biochem. Eng. J. 2018, 130, 104–112. [Google Scholar] [CrossRef]
- Li, H.; Zhang, D.; Han, X.; Xing, B. Adsorption of antibiotic ciprofloxacin on carbon nanotubes: pH dependence and thermodynamics. Chemosphere 2014, 95, 150–155. [Google Scholar] [CrossRef] [PubMed]
- Jiang, W.-T.; Chang, P.H.; Wang, Y.S.; Tsai, Y.; Jean, J.S.; Li, Z.; Krukowski, K. Removal of ciprofloxacin from water by birnessite. J. Hazard. Mater. 2013, 250, 362–369. [Google Scholar] [CrossRef] [PubMed]
- Yu, F.; Li, Y.; Han, S.; Ma, J. Adsorptive removal of antibiotics from aqueous solution using carbon materials. Chemosphere 2016, 153, 365–385. [Google Scholar] [CrossRef]
- Ullah, A.; Zahoor, M.; Alam, S. Removal of ciprofloxacin from water through magnetic nanocomposite/membrane hybrid processes. Desalin. Water Treat. 2019, 137, 260–272. [Google Scholar] [CrossRef]
- Di, J.; Xia, J.; Ji, M.; Yin, S.; Li, H.; Xu, H.; Zhang, Q.; Li, H. Controllable synthesis of Bi4O5Br2 ultrathin nano sheets for photocatalytic removal of ciprofloxacin and mechanism insight. J. Mater. Chem. A 2015, 3, 15108–15118. [Google Scholar] [CrossRef]
- Ahmadzadeh, S.; Asadipour, A.; Pournamdari, M.; Behnam, B.; Rahimi, H.R.; Dolatabadi, M. Removal of ciprofloxacin from hospital wastewater using electrocoagulation technique by aluminum electrode: Optimization and modelling through response surface methodology. Process Saf. Environ. 2017, 109, 538–547. [Google Scholar] [CrossRef]
- Alonso, J.J.S.; El Kori, N.; Melián-Martel, N.; Del Río-Gamero, B. Removal of ciprofloxacin from seawater by reverse osmosis. J. Environ. Manag. 2018, 217, 337–345. [Google Scholar] [CrossRef] [PubMed]
- Zahoor, M.; Ullah, A.; Alam, S. Removal of enrofloxacin from water through magnetic nanocomposites prepared from pineapple waste biomass. Surf. Eng. Appl. Electrochem. 2019, 55, 536–547. [Google Scholar] [CrossRef]
- Zhou, L.; Ji, L.; Ma, P.C.; Shao, Y.; Zhang, H.; Gao, W.; Li, Y. Development of carbon nanotubes/CoFe2O4 magnetic hybrid material for removal of tetrabromobisphenol A and Pb (II). J. Hazard. Mater. 2014, 265, 104–114. [Google Scholar] [CrossRef] [PubMed]
- Reddy, D.H.K.; Yun, Y.S. Spinel ferrite magnetic adsorbents: Alternative future materials for water purification. Coord. Chem. Rev. 2016, 315, 90–111. [Google Scholar] [CrossRef]
- Plakas, K.V.; Karabelas, A.J. Removal of pesticides from water by NF and RO membranes-A review. Desalin 2012, 287, 255–265. [Google Scholar] [CrossRef]
- Zahoor, M. Separation of surfactants from water by granular activated carbon/ultrafiltration hybrid process. Desalin. Water Treat. 2016, 57, 1988–1994. [Google Scholar] [CrossRef]
- Zahoor, M. Effect of granular activated carbon on percent retention of humic acid and permeate flux in GAC/UF membrane process. Desalin. Water Treat. 2016, 57, 23661–23665. [Google Scholar] [CrossRef]
- Zahoor, M.; Khan, F.A. Aflatoxin B1 detoxification by magnetic carbon nanostructures prepared from maize straw. Desalin. Water Treat. 2016, 57, 11893–11903. [Google Scholar] [CrossRef]
- Tran, N.B.T.; Duong, N.B.; Le, N.L. Synthesis and characterization of magnetic Fe3O4/Zeolite NaA nanocomposite for the adsorption removal of methylene blue potential in wastewater treatment. J. Chem. 2021, 2021, 6678588. [Google Scholar] [CrossRef]
- Ho, Y.S. Review of second-order models for adsorption systems. J. Hazard. Mater. 2006, 136, 681–689. [Google Scholar] [CrossRef] [Green Version]
- Guibal, E.; McCarrick, P.; Tobin, J.M. Comparison of the sorption of anionic dyes on activated carbon and chitosan derivatives from dilute solutions. Sep. Sci. Technol. 2003, 38, 3049–3073. [Google Scholar] [CrossRef]
- Liu, Z.; Zhang, F.S.; Sasai, R. Arsenate removal from water using Fe3O4-loaded activated carbon prepared from waste biomass. Chem. Eng. J. 2010, 160, 57–62. [Google Scholar] [CrossRef]
- Pang, Y.; Zeng, G.; Tang, L.; Zhang, Y.; Liu, Y.; Lei, X.; Li, Z.L.; Zhang, J.; Liu, Z.; Xiong, Y. Preparation and application of stability enhanced magnetic nanoparticles for rapid removal of Cr (VI). Chem. Eng. J. 2011, 175, 222–227. [Google Scholar] [CrossRef]
- Ali, N.; Hussain, A.; Ahmed, R.; Shamsuri, W.N.W.; Abdel-Salam, N.N.; Khanates, R. Fabrication and characterization of 150 nm tin antimony sulfide thin films, a promising window layer material for homojunction solar cells. Appl. Phys. A 2017, 123, 1–6. [Google Scholar] [CrossRef]
- Lalitha, S.; Sathyamoorthy, R.; Senthilarasu, S.; Subbarayan, A.; Natarajan, K. Characterization of Cd/Te thin film dependence of structural and optical properties on temperature and thickness. Sol. Energy Mater. Sol. Cells 2004, 82, 187–199. [Google Scholar] [CrossRef]
- Chander, S.; Dhaka, M.S. Influence of thickness on physical properties of vacuum evaporated polycrystalline Cd/Te thin films for solar cell applications. Physica E. Low-Dimen. Syst. Nanostructures 2016, 76, 52–59. [Google Scholar] [CrossRef]
- Dhanam, M.; Prabhu, R.R.; Manoj, P.K. Investigations on chemical bath deposited cadmium selenide thin films. Mater. Chem. Phys. 2008, 107, 289–296. [Google Scholar] [CrossRef]
- Zahoor, M.; Khan, F.A. Adsorption of aflatoxin B1 on magnetic carbon nanocomposites prepared from bagasse. Arab. J. Chem. 2018, 11, 729–738. [Google Scholar] [CrossRef] [Green Version]
- Mahdavi, M.; Ahmad, M.B.; Haron, M.J.; Namvar, F.; Nadi, B.; Rahman, M.Z.A.; Amin, J. Synthesis, surface modification and characterisation of biocompatible magnetic iron oxide nanoparticles for biomedical applications. Molecules 2013, 18, 7533–7548. [Google Scholar] [CrossRef] [Green Version]
- Oliveira, L.C.A.; Rios, R.V.R.A.; Fabris, J.D.; Garg, V.; Sapag, K.; Lago, R.M. Activated carbon/iron oxide magnetic composites for the adsorption of contaminants in water. Carbon 2002, 40, 2177–2183. [Google Scholar] [CrossRef]
- Anyika, C.; Asri, N.A.M.; Majid, Z.A.; Yahya, A.; Jaafar, J. Synthesis and characterization of magnetic activated carbon developed from palm kernel shells. Nanotechnol. Environ. Eng. 2017, 2, 16. [Google Scholar] [CrossRef]
- Chen, G.C.; Shan, X.Q.; Zhou, Y.Q.; Shen, X.; Huang, H.-L.; Khan, S.U. Adsorption kinetics, isotherms and thermodynamics of atrazine on surface oxidized multiwalled carbon nanotubes. J. Hazard. Mater. 2009, 169, 912–918. [Google Scholar] [CrossRef] [PubMed]
- Ibezim, E.C.; Ofoefule, S.I.; Ejeahalaka, C.N.; Orisakwe, O.E. In vitro adsorption of ciprofloxacin on activated charcoal and Talc. Am. J. Ther. 1999, 6, 199–201. [Google Scholar] [CrossRef] [PubMed]
- Jia, M.; Wang, F.; Bian, Y.; Jin, X.; Song, Y.; Kengara, F.O. Effects of pH and metal ions on oxytetracycline sorption to maize-straw-derived biochar. Bioresour. Technol. 2013, 136, 87–93. [Google Scholar] [CrossRef]
- Tanis, E.; Hanna, K.; Emmanuel, E. Experimental and modeling studies of sorption of tetracycline on to iron oxides-coated quartz. Colloids Surf. A: Physicochem. Eng. Asp. 2008, 327, 57–63. [Google Scholar] [CrossRef]
- Chen, D.; Zeng, Z.; Zheng, Y.; Zhang, F.; Wang, M. Removal of Methylene Blue and mechanism of magnetic γ Fe2O3/SiO2 nanocomposites from aqueous solution. Water Resour. Ind. 2016, 15, 1–13. [Google Scholar] [CrossRef] [Green Version]
- Mao, H.; Wang, S.; Lin, J.Y.; Wang, Z.; Ren, J. Modification of a magnetic carbon composite for ciprofloxacin adsorption. J. Environ. Sci. 2016, 49, 179–188. [Google Scholar] [CrossRef]
- Mezni, M.; Saied, T.; Horri, N.; Srasra, E. Removal of enrofloxacin from aqueous solutions using illite and synthetic zeolite X. Surf. Eng. App. Electrochem. 2017, 53, 89–97. [Google Scholar] [CrossRef]
- Wang, Y.X.; Ngo, H.H.; Guo, W.S. Preparation of a specific bamboo based activated carbon and its application for ciprofloxacin removal. Sci. Total Environ. 2015, 533, 32–39. [Google Scholar] [CrossRef]
- Wang, Y.H.; Gupta, K.; Li, J.R.; Yuan, B.; Yang, J.C.E.; Fu, M.L. Novel chalcogenide based magnetic adsorbent KMS-1/L-Cystein/Fe3O4 for the facile removal of ciprofloxacin from aqueous solution. Colloids Surf. A Physicochem. Eng. Asp. 2018, 538, 378–386. [Google Scholar] [CrossRef]

| UF Membrane | NF Membrane | RO Membrane | |||
|---|---|---|---|---|---|
| Parameters | Specification | Parameters | Specification | Parameters | Specification |
| Material | Polyether sulfone | Model | NF 270-2540 | Model | RO 270-2540 |
| Type | Capillary multi bores × 7 | Permeate Flow rate | 850 gallons/day (3.2 m3/day) | Membrane type | Thin film composite (Filmtech) |
| Diameter bores ID | 0.9 mm | Active surface area | 28 ft2 (3.2 m2) | Permeate Flow rate | 850 gallons/day (3.2 m3/day) |
| Diameter fiber OD | 4.2 mm | Applied pressure | 4.8 bar | Active surface area | 28 ft2 (3.2 m2) |
| Stabilized salt rejection | 10–20% | Stabilized salt rejection | >97% | Stabilized salt rejection | 100% |
| Surface area | 50 m2 | Surface area | 3.2 m2 | Surface area | 3.2 m2 |
| Maximum temperature | 40 °C | Maximum temperature | 40–180 °C | Maximum temperature | 40–180 °C |
| Maximum pressure | 109 psi | Maximum pressure | 100–1000 psi | Maximum pressure | 100–1000 psi |
| Membrane back wash pressure | 0.5–1 bar | Membrane back wash pressure | 50–800 psi | Membrane back wash pressure | 50–800 psi |
| Operator pH range | 3–10 | Operator pH range | 3–10 | Operator pH range | 3–10 |
| Back wash pH range | 1–13 | Back wash pH range | 1–12 | Back wash pH range | 1–12 |
| Disinfection chemicals | Hypo chloride and Hydrogen peroxide | Disinfection chemicals | Hydrogen peroxide and peracetic acid | Disinfection chemicals | Hydrogen peroxide and peracetic acid |
| MWCO | 100 KD | MWCO | 0.3–1 KD | MWCO | 0.1–1 KD |
| Pore size | 5–20 nm | Pore size | 1–5 nm | Pore size | 1–5 nm |
| No | Parameters | Value |
|---|---|---|
| 1 | Crystallite size (D) | 0.8132 |
| 2 | Strain (ε) | 3.65 × 10−3 |
| 3 | Full width at half maximum (FWHM) (β) | 0.010472 |
| 4 | Dislocation density (δ) | 3.46 × 1015 m−2 |
| 5 | Number of crystallite per unit area (N) | 3.5 × 1013 m−2 |
| Co. (mg L−1) | qe exp (mg g−1) | Pseudo 1st Order k1 R2 qe cal (mg g−1) | Pseudo 1st Order k2 R2 qe cal (mg g−1) | Intra Particle Diffusion k3 R2 C |
|---|---|---|---|---|
| 40 | 30 | 0.044 0.96 39.40 | 0.0410 0.993 30.10 | 2.3 0.91 3.10 |
| 80 | 50 | 0.051 0.96 93.00 | 0.0480 0.980 50.00 | 5.3 0.93 6.30 |
| Langmuir | Freundlich | Temkin | Jovanovich |
|---|---|---|---|
| qmax (mg g−1) kL (L mg−1) R2 | KF 1/n R2 | b β α R2 | KJ qmax (mg g−1) R2 |
| 56.82 0.112 0.997 | 12 0.37 0.86 | 207 11.95 1.2 0.91 | 0.047 15.33 0.95 |
| Temperature (K) | ΔH° (kJ mol−1) | ΔS° (J mol−1 K−1) | ΔG° (kJmol−1) |
|---|---|---|---|
| 298 | −21.6 | 75 | −2.40 |
| 313 | ….. | ….. | −2.60 |
| 333 | ….. | ….. | −2.73 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zahoor, M.; Ullah, A.; Alam, S.; Muhammad, M.; Hendroko Setyobudi, R.; Zekker, I.; Sohail, A. Novel Magnetite Nanocomposites (Fe3O4/C) for Efficient Immobilization of Ciprofloxacin from Aqueous Solutions through Adsorption Pretreatment and Membrane Processes. Water 2022, 14, 724. https://doi.org/10.3390/w14050724
Zahoor M, Ullah A, Alam S, Muhammad M, Hendroko Setyobudi R, Zekker I, Sohail A. Novel Magnetite Nanocomposites (Fe3O4/C) for Efficient Immobilization of Ciprofloxacin from Aqueous Solutions through Adsorption Pretreatment and Membrane Processes. Water. 2022; 14(5):724. https://doi.org/10.3390/w14050724
Chicago/Turabian StyleZahoor, Muhammad, Azmat Ullah, Sultan Alam, Mian Muhammad, Roy Hendroko Setyobudi, Ivar Zekker, and Amir Sohail. 2022. "Novel Magnetite Nanocomposites (Fe3O4/C) for Efficient Immobilization of Ciprofloxacin from Aqueous Solutions through Adsorption Pretreatment and Membrane Processes" Water 14, no. 5: 724. https://doi.org/10.3390/w14050724
APA StyleZahoor, M., Ullah, A., Alam, S., Muhammad, M., Hendroko Setyobudi, R., Zekker, I., & Sohail, A. (2022). Novel Magnetite Nanocomposites (Fe3O4/C) for Efficient Immobilization of Ciprofloxacin from Aqueous Solutions through Adsorption Pretreatment and Membrane Processes. Water, 14(5), 724. https://doi.org/10.3390/w14050724







