Uranium and Fluoride Removal from Aqueous Solution Using Biochar: A Critical Review for Understanding the Role of Feedstock Types, Mechanisms, and Modification Methods
Abstract
:1. Introduction
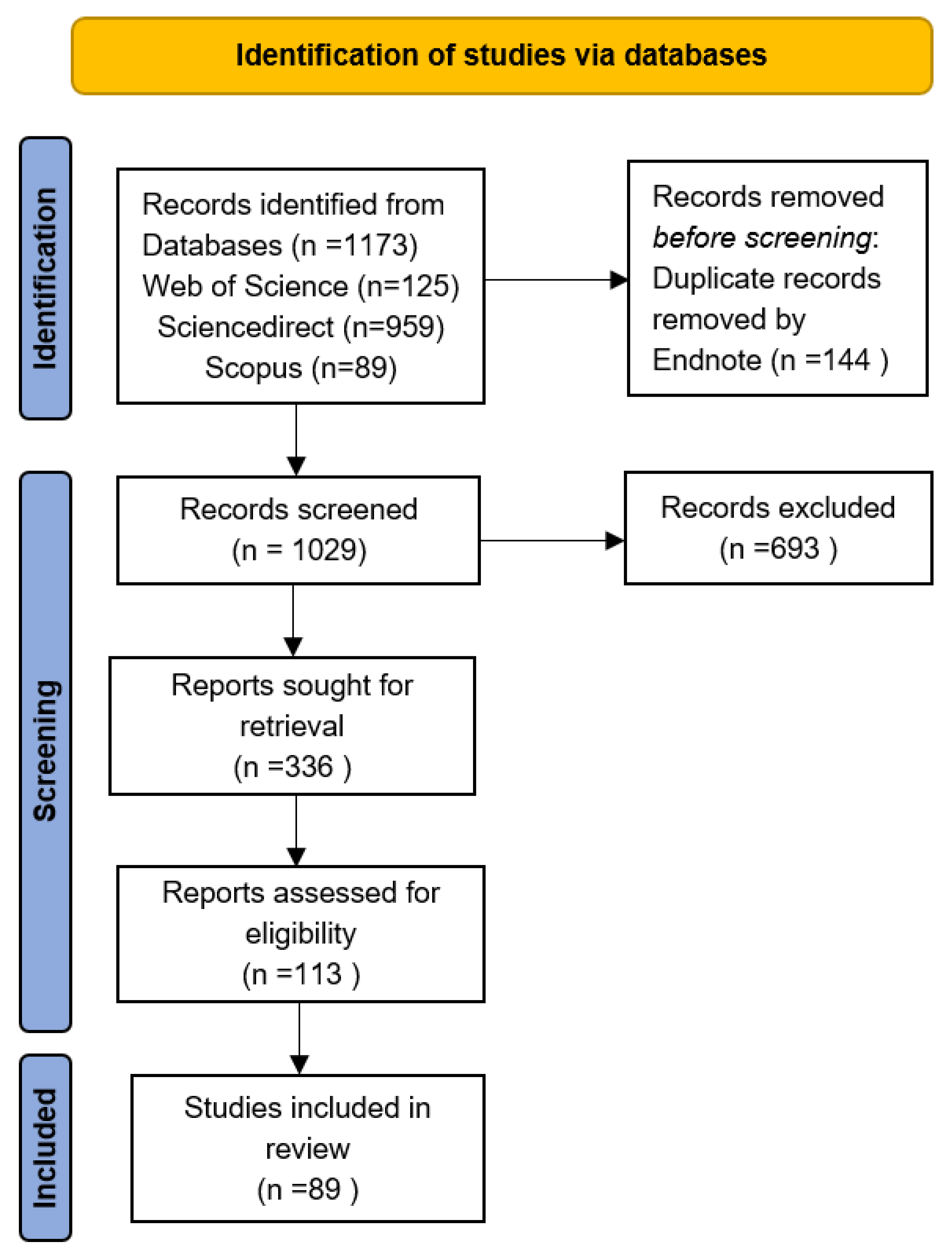
2. Methodology
3. Results and Discussion
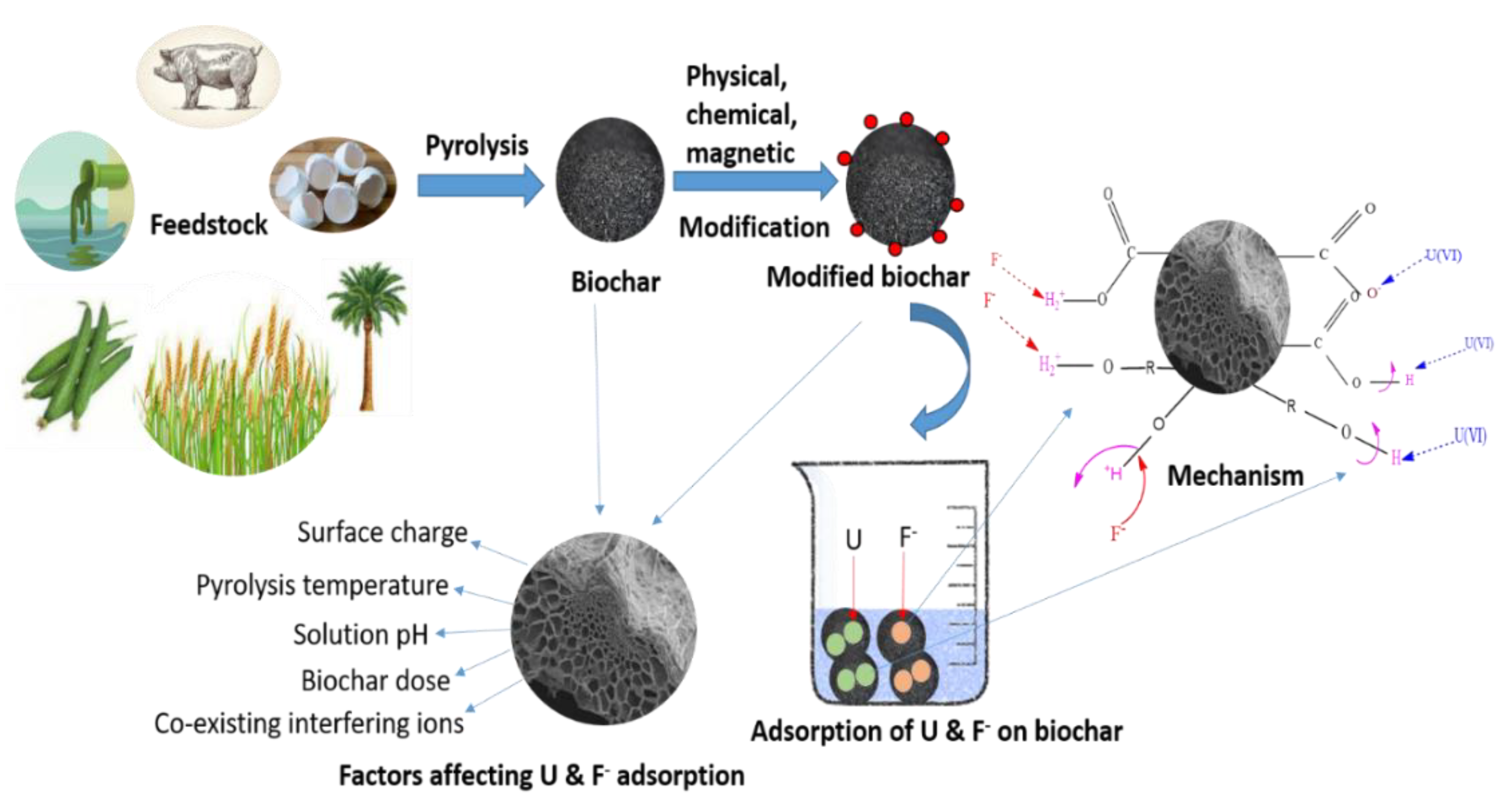
3.1. Role of Different Feedstock Types on Biochar Properties
3.2. Applications of Biochar in U and F− Remediation
3.3. Synthesis of Biochar
3.3.1. Pyrolysis
3.3.2. Gasification
3.3.3. Torrefaction
3.3.4. Hydrothermal Carbonization
3.4. Characteristics of Biochar
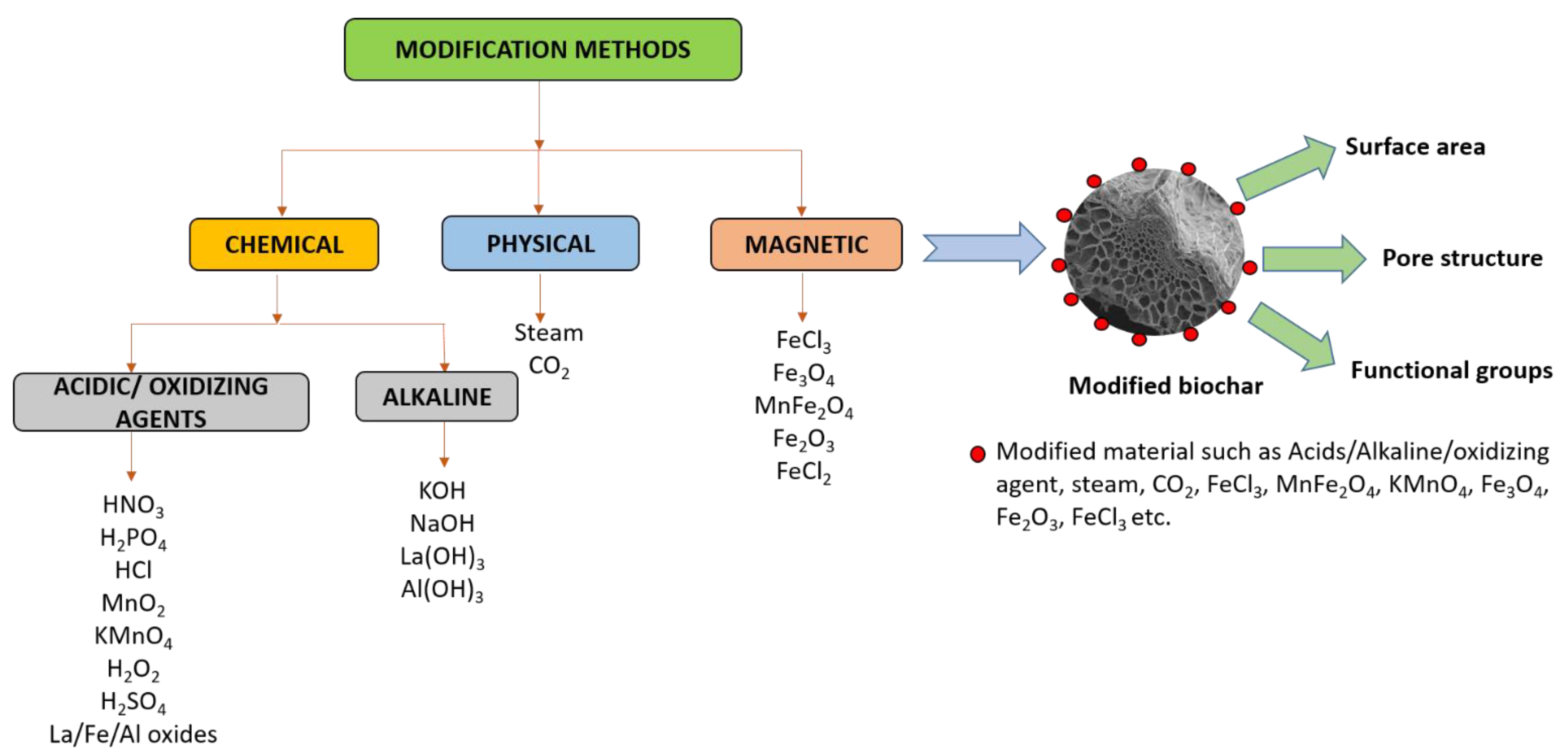
3.5. Modification Methods
| Feedstock | Modification | Target Contaminant | Surface Area (m2 g−1) | Pore Structure | Adsorption Capacity (mg g−1)/Removal Efficiency (%) | References | |
|---|---|---|---|---|---|---|---|
| Raw Biochar | Modified Biochar | ||||||
| Rice straw | Hydroxyapatite–biochar nanocomposite | U | 157.96 | Mesoporous | 110.56 | 428.25 | [131] |
| Rice straw | HNO3 oxidation | U | - | Mesoporous | 162.54 | 242.65 | [33] |
| Wheat straw | HNO3 oxidation | U | 290.1 | - | 8.7 | 355.6 | [82] |
| Rice husk | Magnetization by Siderite | U | 109.65 | Mesoporous | - | 52.63 | [125] |
| Rice husk | Silicon containing biochar-supported iron oxide nanoparticles | U | 62.88 | - | - | 138.88 | [81] |
| Rice husk | Magnetic modification using Fe2+/Fe3+ plus SO42− solution | U | 109 | - | 64 | 118 | [132] |
| Corn cob | Thermal air treatment at 300 °C | U | - | Mesoporous | 68.82 | 163.18 | [34] |
| Pine needles | Magnetization of oxidised biochar (HNO3 treated) by FeCl3 | U | - | - | - | 623.7 | [37] |
| Pine sawdust | MgO/biochar composite | U | 51.45 | Mesoporous | - | 514.72 | [133] |
| Macaúba palm | CO2 activation | U | 643.12 | Microporous | - | 488.7 | [36] |
| Palm tree fibres | HNO3 oxidation | U | - | - | - | 112 | [121] |
| Bamboo sawdust | Phytic acid | U | 1298 | - | 16.2 | 229.2 | [111] |
| Bamboo biomass | Phosphate impregnation biochar cross-linked Mg–Al layered double-hydroxide composite | U | 445.17 | Microporous | 15.869 | 274.15 | [134] |
| Cactus fibre | HNO3 oxidation | U | <5 | Microporous | - | 214 | [118] |
| Chinese banyan aerial root | KMnO4 modification | U | 284 | Mesoporous | 19.08 | 27.29 | [135] |
| Puncture vine | Magnetization by FeCl3 | U | - | Mesoporous | - | 17.24 | [39] |
| Hydrophyte biomass | Magnetization by FeCl2 | U | 92.43 | - | 52.36 | 54.35 | [136] |
| Hydrophyte | Phytic acid modification | U | 433 | - | - | 128.5 | [137] |
| Pig manure | KMnO4 | U | - | - | 369.9 | 979.3 | [107] |
| Pig manure | H2O2 | U | - | - | 369.9 | 661.7 | [107] |
| Pig manure | NaOH | U | 227.9 | Mesoporous | 369.9 | 952.5 | [138] |
| Pig manure | HCl | U | 36.3 | Mesoporous | 369.9 | 53.3 | [138] |
| Pig manure | NaOH | U | 345.7 | Microporous | 45.8 | 221.4 | [124] |
| Pig manure | H2O2 | U | 189 | Microporous | 45.8 | 145.1 | [124] |
| Horse manure | Bismuth impregnation | U | - | - | 186 | 516.5 | [108] |
| Horse manure | MgCl2 modification | U | - | - | - | 625.8 | [139] |
| Cow manure | HNO3 oxidation | U | 101.5 | - | 64 | 73.3 | [82] |
| Carp fish scales | KOH activation | U | 1074.73 | Microporous | 71.59 | 291.98 | [122] |
| Sewage sludge | Thermal air treatment | U | - | Mesoporous | 78.66 | 96.73 | [34] |
| Sewage sludge | Air roasting–oxidation | U | 623.09 | Mesoporous | 139.5 | 490.2 | [140] |
| Winery waste (grape peels) | Chemically modification by NaOH, Na2CO3 | U | - | - | - | 255 | [141] |
| Winery waste (grape peels) | Thermal modification at 650 °C and oxidized with HNO3 | U | 165 | - | - | 100 | [141] |
| Malt spent rootlets | HNO3 oxidation | U | 540 | Mesoporous | 547 | 500 | [120] |
| Coffee espresso residue | HNO3 oxidation | U | 700 | Mesoporous | 547 | 357 | [120] |
| Olive kernels | HNO3 oxidation | U | 510 | Mesoporous | 357 | 381 | [120] |
| Fungi | Sulfide nano zero valent iron | U | 102.7 | _ | - | 427.9 | [142] |
| Green algae | Mn impregnation | U | 63.7 | Mesoporous | - | 100.2 | [143] |
| Cyanobacteria | Magnetic modification using Fe3O4 | U | - | - | 58.05 | 52.06 | [144] |
| Sponge gourd | ZnO-modified biochar hydrogel | U | - | - | 239.21 | [83] | |
| Sponge gourd fibres | MnO2 oxidation | U | <5 | Microporous | 95 | 904 | [84] |
| Sponge gourd sponges | HNO3 oxidation | U | - | - | - | 92 | [119] |
| Sponge gourd sponge | Salophen modification | U | - | - | - | 833 | [145] |
| Sponge gourd residue | Functionalization by hummer method | U | - | - | - | 382 | [146] |
| Watermelon rind | Magnetization by co-precipitation | U | 86.35 | - | 135.86 | 323.56 | [85] |
| Watermelon seeds | MnFe2O4 modification | U | - | Mesoporous | 21.24 | 27.61 | [38] |
| Longan shell (fruit) | Nano zero valent iron | U | 1168.88 | Mesoporous | - | 331.13 | [77] |
| Orange peel | MnO2 modification | U | 273.25 | Mesoporous | 165.4 | 246.3 | [106] |
| Orange peel | Hydrogel | U | - | - | - | 263.2 | [147] |
| Tea waste | Iron manganese oxide | U | 12 | - | - | 510.8 | [148] |
| Rice straw | La/Fe/Al oxides impregnation | F− | 95.36 | Mesoporous | 10.85 | 111.11 | [89] |
| Wheat straw | Impregnation of aluminium and lanthanum hydroxide | F− | - | - | - | 51.28 | [90] |
| Rice husk | Chemical modification by iron | F− | 58.98 | Mesoporous | - | 4.45 | [149] |
| Rice husk | Nano-scale size reduction | F− | - | - | - | 17.3 | [24] |
| Rice husk | Magnetic biochar anchored with Al and Mg | F− | 114 | Mesoporous | - | 21.59 | [150] |
| Corn stover | Magnetization by Fe3+/Fe2+ solution | F− | 3.61 | Microporous | 6.42 | 4.11 | [91] |
| Chir pine | Calcium pretreated | F− | - | - | - | 16.72 | [151] |
| Mongolian scotch pine tree sawdust | Phosphoric acid-microwave method | F− | 339 | Microporous | - | 0.885 | [35] |
| Douglas fir (pine) | Magnetization by Fe2O3/Fe3O4 | F− | 494 | Microporous | - | 9.04 | [152] |
| Douglas fir (pine) | Iron-titanium biochar composite | F− | 576 | Microporous | - | 36 | [153] |
| Reed biomass | Ce-loaded biochar beads | F− | 236.84 | Mesoporous | - | 34.86 | [128] |
| Kashgar tamarisk | Lanthanum chloride | F− | 164.52 | Mesoporous | - | 164.23 | [40] |
| Tea oil plant (seed shells) | Impregnation of zirconium dioxide | F− | - | - | - | 11.04 | [93] |
| Sawdust | Chemical modification via cross-linking and protonation of the chitosan-sawdust biochar beads | F− | 57.97 | Microporous | - | 4.413 | [154] |
| Pongammia pinnata seed cake | Engineered biochar by HCl solution | F− | 10.1 | Microporous | - | 1.11 | [7] |
| Coconut | Steam activation | F− | 1054 | Mixture of micropores and mesopores | - | 82.45% | [92] |
| Pomelo peel | Impregnation of polypyrrole | F− | - | - | - | 18.52 | [95] |
| Peanut shell | MgO | F− | 182.3 | Mesoporous | - | 83.05 | [155] |
| Spent mushroom compost | Al(OH)3 coating | F− | 28.5 | - | - | 36.5 | [13] |
| Food waste | AlCl3 impregnation | F− | 20.95 | - | - | 123.4 | [156] |
| Tea waste | Chemical modification by H2SO4, NaNO3, KMnO4 | F− | 11.833 | Macroporous | - | 52.5 | [157] |
| Red algae seaweed | Spent biochar | F− | 319.47 | Microporous | - | 2.1 | [158] |
| Dairy manure | Calcium modification | F− | 2.6 | - | 0.11 | 0.42 | [109] |
| Tea waste | Magnetic modification | F− | 115.65 | Mesoporous | - | 18.78 | [159] |
| Peanut hull | Nil | F− | 98.2 | Mesoporous | 3.665 | - | [160] |
| Pinecone | AlCl3 | F− | - | - | - | 14.07 | [161] |
| Conocarpus erectus | Nil | F− | 9.88 | Microporous | 205.7 | - | [162] |
| Yak dung | FeCl2 | F− | - | - | - | 3.928 | [163] |
3.5.1. Physical Modification
3.5.2. Chemical Modification
3.5.3. Magnetic Modification
3.5.4. Thermal Air Treatment (TAT)
3.6. Raw vs. Modified Biochar
3.7. Factors Affecting the Adsorption of U and F− in Aqueous Solution
3.7.1. Influence of pH
| Feedstock | Solution pH | pHPZC | Target Pollutant | Speciation Adsorbed | Biochar Surface Charge | Adsorption Capacity (mg g−1) | References |
|---|---|---|---|---|---|---|---|
| Rice straw | 5.5 | 2.5 | U | UO22+, (UO2)3(OH)5+, (UO2)3(OH)42+, (UO2)2(OH)22+ | negative | 428.25 | [131] |
| Rice straw | 5.5 | 2.5 | U | negative | 242.65 | [33] | |
| Wheat straw | 6 | 3 | U | UO22+, (UO2)3(OH)5+, (UO2)2(OH)22+, UO2OH+, (UO2)4(OH)7+ | negative | 355.6 | [82] |
| Rice husk | 4 | 3.51 | U | UO22+, UO2OH+, (UO2)3(OH)5+ | negative | 52.63 | [125] |
| Rice husk | 5.5 | 4.17 | U | UO22+, (UO2)3(OH)5+, (UO2)2(OH)22+ | negative | 138.88 | [81] |
| Rice husk | 7 | 3.71 | U | UO2(OH)+, (UO2)2(OH)2, (UO2)3(OH)52+ | negative | 118 | [132] |
| Corn cob | 6 | _ | U | (UO2)3(OH)5+, (UO2)2(OH)22+, UO2OH+ | negative | 163.18 | [34] |
| Pine needles | 6 | 3.8 | U | _ | _ | 623.7 | [37] |
| Pine needles | 6 | _ | U | _ | _ | 62.7 | [172] |
| Pine sawdust | 4 | 2.98 | U | UO22+, (UO2)3(OH)5+, (UO2)2(OH)22+, (UO2)4(OH)7+ | negative | 514.72 | [133] |
| Macaúba palm | 3 | U | 488.7 | [36] | |||
| Palm tree fibres | 6 | _ | U | _ | _ | 112 | [121] |
| Bamboo sawdust | 4 | 2.73 | U | UO22+, (UO2)3(OH)5+, (UO2)2(OH)22+, UO2OH+ | negative | 229.2 | [111] |
| Bamboo biomass | 4 | 4.28 | U | UO22+, (UO2)3(OH)5+, (UO2)2(OH)22+, UO2OH+ | 274.15 | [134] | |
| Bamboo | 6 | _ | U | _ | _ | _ | [34] |
| Bamboo shoot shell | 4 | _ | U | UO22+, (UO2)2(OH)22+, UO2OH+, (UO2)4(OH)7+ | negative | 32.3 | [105] |
| Cactus fibre | 3 | _ | U | _ | _ | 214 | [118] |
| Camphor tree leaves | 6.5 | 5.76 | U | UO22+, (UO2)2(OH)22+, UO2OH+ | negative | 98.29 | [173] |
| Miswak branches | 4 | 2.79 | U | UO22+, (UO2)2(OH)22+, UO2OH+ | negative | 85.71 | [174] |
| Chinese banyan aerial root | 4 | _ | U | _ | _ | 27.29 | [135] |
| Eucalyptus wood | 5.5 | _ | U | (UO2)2OH3+, (UO2)3(OH)5+, (UO2)2(OH)22+, UO2OH+, (UO2)4(OH)7+ | negative | 27.2 | [175] |
| Puncture vine | 6 | 4 | U | UO22+, (UO2)3(OH)5+, (UO2)3(OH)42+, (UO2)2(OH)22+ | negative | 17.24 | [39] |
| Water hyacinth | 6 | _ | U | UO22+, (UO2)2(OH)22+, UO2OH+, (UO2)4(OH)7+ | negative | 138.57 | [176] |
| Hydrophyte biomass | 3 | 4.2 | U | UO22+ | 54.35 | [136] | |
| Hydrophyte | 4 | 2.46 | U | UO22+, (UO2)3(OH)42+, (UO2)2(OH)22+, UO2OH+ | negative | 128.5 | [137] |
| Switchgrass | 5.9 | _ | U | UO22+, (UO2)3(OH)5+, (UO2)2(OH)22+, UO2OH+ | negative | 4 | [177] |
| Pig manure | 4 | U | 979.3 | [107] | |||
| Pig manure | 4 | U | 661.7 | [107] | |||
| Pig manure | 4 | U | 952.5 | [138] | |||
| Pig manure | 4.5 | _ | U | UO22+,(UO2)2(OH)22+, UO2OH+ | 221.4 | [124] | |
| Horse manure | 4 | 9.05 | U | UO22+ | 516.5 | [108] | |
| Horse manure | 4 | U | UO22+ | negative | 625.8 | [139] | |
| Cow manure | 4.5 | 3 | U | UO22+, (UO2)3(OH)5+, (UO2)2(OH)22+, UO2OH+, (UO2)4(OH)7+ | negative | 73.3 | [82] |
| Carp fish scales | 5 | 2.87 | U | UO22+, (UO2)3(OH)5+, (UO2)2(OH)22+, UO2OH+ | negative | 291.98 | [122] |
| Sewage sludge | 6 | _ | U | (UO2)3(OH)5+, (UO2)2(OH)22+, UO2OH+ | negative | 96.73 | [34] |
| Sewage sludge | 6 | 3 | U | UO22+, (UO2)3(OH)5+, (UO2)2(OH)22+, UO2OH+ | negative | 490.2 | [140] |
| Winery waste (grape peels) | 4 | _ | U | UO22+ | negative | 255 | [141] |
| Winery waste (grape peels) | 4 | _ | U | UO22+ | negative | 100 | [141] |
| Malt spent rootlets (MSR) | 3 | _ | U | _ | _ | 547 | [120] |
| Coffee espresso residue | 3 | _ | U | _ | _ | 547 | [120] |
| Olive kernels | 3 | _ | U | _ | _ | 357 | [120] |
| Fungi | 5 | 6.41 | U | positive | 427.9 | [142] | |
| Green algae | 6 | 2.62 | U | (UO2)3(OH)5+, (UO2)2(OH)22+, (UO2)4(OH)7+ | negative | 100.2 | [143] |
| Cyanobacteria | 6 | 3.5 | U | (UO2)3(OH)5+, (UO2)2(OH)22+, UO2OH+ | negative | 58.05 | [144] |
| Sponge gourd | 5 | _ | U | (UO2)3(OH)5+, (UO2)2(OH)22+, UO2OH+ | negative | 239.21 | [83] |
| Sponge gourd fibres | 3 | U | 904 | [84] | |||
| Sponge gourd sponges | 3 | _ | U | _ | _ | 92 | [119] |
| Sponge gourd sponge | 5.5 | _ | U | _ | _ | 833 | [145] |
| Sponge gourd residue | 6 | 1.8 | U | UO22+, (UO2)3(OH)5+, (UO2)2(OH)22+, UO2OH+ | negative | 382 | [146] |
| Watermelon rind | 4 | 5.4 | U | _ | _ | 323.56 | [85] |
| Watermelon seeds | 4 | 2.5 | U | _ | _ | 27.61 | [38] |
| Longan shell (fruit) | 6 | 6.25 | U | 331.13 | [77] | ||
| Orange peel | 5.5 | 2.6 | U | UO22+, (UO2)3(OH)42+, (UO2)2(OH)22+, UO2OH+, (UO2)2OH3+, UO2(OH)2 | negative | 246.3 | [106] |
3.7.2. Effect of Biochar Dose on U and F− Adsorption
3.7.3. Influence of Initial Concentration
3.7.4. Influence of Co-Existing Ions
3.7.5. Influence of Pyrolysis Temperature on U and F− Adsorption
3.7.6. Influence of Different Feedstocks on U and F− Adsorption
3.8. Adsorption Mechanism of U and F−
4. Adsorption Isotherms, Kinetics, and Thermodynamics
4.1. Adsorption Isotherms
4.2. Adsorption Kinetics
4.3. Adsorption Thermodynamics
5. Regeneration of Biochar
6. Challenges and Limitations for Real U and F− Groundwater Treatment
7. Conclusions, Research Gaps, and Future Perspectives
- Most of the investigations have been carried out at lab scale through batch studies using synthetic water or simulated water, and limited studies have been performed using natural water. In order to scale up, column studies should be conducted for field applications, and future research should be focused on the treatment of natural water.
- Most studies have focused on the selective removal/extraction of either U or F− using biochar. However, actual waters contain multiple contaminants, so future research should focus on using biochar to remove multiple contaminants from water.
- There is a lack of studies for the treatment of drinking-water sources such as natural waters and groundwater using biochar. Most of the studies have focused on the remediation of wastewater.
- Most of the modifications are chemical modifications, and there is a need for environmentally friendly green methods/materials for modification.
- The effect of co-existing ions has not been studied in detail. There is no detailed study on the impact of multiple components in the solution that interferes with the U and F− adsorption.
- Most studies have considered U as a cation (UO22+), but in alkaline solutions, U exists as (UO2)2CO3 (OH)3− (anion). So, there is no study for treating anionic U using biochar from aqueous solution.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Salehi, M. Global Water Shortage and Potable Water Safety; Today’s Concern and Tomorrow’s Crisis. Environ. Int. 2022, 158, 106936. [Google Scholar] [CrossRef] [PubMed]
- Hering, J.G.; Maag, S.; Schnoor, J.L. A Call for Synthesis of Water Research to Achieve the Sustainable Development Goals by 2030. Environ. Sci. Technol. 2016, 50, 6122–6123. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aravinthasamy, P.; Karunanidhi, D.; Subramani, T.; Anand, B.; Roy, P.D.; Srinivasamoorthy, K. Fluoride Contamination in Groundwater of the Shanmuganadhi River Basin (South India) and Its Association with Other Chemical Constituents Using Geographical Information System and Multivariate Statistics. Chem. Erde 2020, 80, 125555. [Google Scholar] [CrossRef]
- Kaur, G.; Kumar, R.; Mittal, S.; Sahoo, P.K.; Vaid, U. Ground/Drinking Water Contaminants and Cancer Incidence: A Case Study of Rural Areas of South West Punjab, India. Hum. Ecol. Risk Assess. 2021, 27, 205–226. [Google Scholar] [CrossRef]
- Coyte, R.M.; Singh, A.; Furst, K.E.; Mitch, W.A.; Vengosh, A. Co-Occurrence of Geogenic and Anthropogenic Contaminants in Groundwater from Rajasthan, India. Sci. Total Environ. 2019, 688, 1216–1227. [Google Scholar] [CrossRef]
- Ma, M.; Wang, R.; Xu, L.; Xu, M.; Liu, S. Emerging Health Risks and Underlying Toxicological Mechanisms of Uranium Contamination: Lessons from the Past Two Decades. Environ. Int. 2020, 145, 106107. [Google Scholar] [CrossRef]
- De, D.; Santosha, S.; Aniya, V.; Sreeramoju, A.; Satyavathi, B. Assessing the Applicability of an Agro-Industrial Waste to Engineered Bio-Char as a Dynamic Adsorbent for Fluoride Sorption. J. Environ. Chem. Eng. 2018, 6, 2998–3009. [Google Scholar] [CrossRef]
- Balaram, V.; Rani, A.; Rathore, D.P.S. Uranium in Groundwater in Parts of India and World: A Comprehensive Review of Sources, Impact to the Environment and Human Health, Analytical Techniques, and Mitigation Technologies. Geosystems Geoenvironment 2022, 1, 100043. [Google Scholar] [CrossRef]
- Coyte, R.M.; Jain, R.C.; Srivastava, S.K.; Sharma, K.C.; Khalil, A.; Ma, L.; Vengosh, A. Large-Scale Uranium Contamination of Groundwater Resources in India. Environ. Sci. Technol. Lett. 2018, 5, 341–347. [Google Scholar] [CrossRef]
- Brugge, D.; Buchner, V. Health Effects of Uranium: New Research Findings. Rev. Environ. Health 2011, 26, 231–249. [Google Scholar] [CrossRef]
- Ali, S.; Thakur, S.K.; Sarkar, A.; Shekhar, S. Worldwide Contamination of Water by Fluoride. Environ. Chem. Lett. 2016, 14, 291–315. [Google Scholar] [CrossRef]
- Kumar, M.; Goswami, R.; Patel, A.K.; Srivastava, M.; Das, N. Scenario, Perspectives and Mechanism of Arsenic and Fluoride Co-Occurrence in the Groundwater: A Review. Chemosphere 2020, 249, 126126. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.J.; Peng, C.Y.; Fang, J.Y.; Dong, Y.Y.; Zhu, X.H.; Cai, H.M. Biosorption of Fluoride from Drinking Water Using Spent Mushroom Compost Biochar Coated with Aluminum Hydroxide. Desalin. Water Treat. 2016, 57, 12385–12395. [Google Scholar] [CrossRef]
- Shen, J.; Schäfer, A. Removal of Fluoride and Uranium by Nanofiltration and Reverse Osmosis: A Review. Chemosphere 2014, 117, 679–691. [Google Scholar] [CrossRef] [PubMed]
- Katsoyiannis, I.A.; Zouboulis, A.I. Removal of Uranium from Contaminated Drinking Water: A Mini Review of Available Treatment Methods. Desalin. Water Treat. 2013, 51, 2915–2925. [Google Scholar] [CrossRef]
- Bhatnagar, A.; Kumar, E.; Sillanpää, M. Fluoride Removal from Water by Adsorption—A Review. Chem. Eng. J. 2011, 171, 811–840. [Google Scholar] [CrossRef]
- Ghosh, D.; Sinha, M.K.; Purkait, M.K. A Comparative Analysis of Low-Cost Ceramic Membrane Preparation for Effective Fluoride Removal Using Hybrid Technique. Desalination 2013, 327, 2–13. [Google Scholar] [CrossRef]
- Ndiaye, P.I.; Moulin, P.; Dominguez, L.; Millet, J.C.; Charbit, F. Removal of Fluoride from Electronic Industrial Effluentby RO Membrane Separation. Desalination 2005, 173, 25–32. [Google Scholar] [CrossRef]
- Azbar, N.; Türkman, A. Defluoridation in Drinking Waters. Water Sci. Technol. 2000, 42, 403–407. [Google Scholar] [CrossRef]
- Chaturvedi, A.K.; Yadava, K.P.; Pathak, K.C.; Singh, V.N. Defluoridation of Water by Adsorption on Fly Ash. Water. Air. Soil Pollut. 1990, 49, 51–61. [Google Scholar] [CrossRef]
- Habuda-Stanić, M.; Ravančić, M.; Flanagan, A. A Review on Adsorption of Fluoride from Aqueous Solution. Materials 2014, 7, 6317–6366. [Google Scholar] [CrossRef] [PubMed]
- Samarghandi, M.R.; Khiadani, M.; Foroughi, M.; Zolghadr Nasab, H. Defluoridation of Water Using Activated Alumina in Presence of Natural Organic Matter via Response Surface Methodology. Environ. Sci. Pollut. Res. 2016, 23, 887–897. [Google Scholar] [CrossRef] [PubMed]
- Vithanage, M.; Bhattacharya, P. Fluoride in the Environment: Sources, Distribution and Defluoridation. Environ. Chem. Lett. 2015, 13, 131–147. [Google Scholar] [CrossRef]
- Goswami, R.; Kumar, M. Removal of Fluoride from Aqueous Solution Using Nanoscale Rice Husk Biochar. Groundw. Sustain. Dev. 2018, 7, 446–451. [Google Scholar] [CrossRef]
- Arfin, T.; Waghmare, S. Fluoride Removal from Water by Mixed Metal Oxide Adsorbent Materials: A State-of-the-Art Review. Int. J. Eng. Sci. Res. Technol. 2015, 4, 519–536. [Google Scholar]
- Velazquez-Jimenez, L.H.; Vences-Alvarez, E.; Flores-Arciniega, J.L.; Flores-Zuñiga, H.; Rangel-Mendez, J.R. Water Defluoridation with Special Emphasis on Adsorbents-Containing Metal Oxides and/or Hydroxides: A Review. Sep. Purif. Technol. 2015, 150, 292–307. [Google Scholar] [CrossRef]
- Shuibo, X.; Chun, Z.; Xinghuo, Z.; Jing, Y.; Xiaojian, Z.; Jingsong, W. Removal of Uranium (VI) from Aqueous Solution by Adsorption of Hematite. J. Environ. Radioact. 2009, 100, 162–166. [Google Scholar] [CrossRef]
- Camacho, L.M.; Deng, S.; Parra, R.R. Uranium Removal from Groundwater by Natural Clinoptilolite Zeolite: Effects of PH and Initial Feed Concentration. J. Hazard. Mater. 2010, 175, 393–398. [Google Scholar] [CrossRef]
- Alahabadi, A.; Singh, P.; Raizada, P.; Anastopoulos, I.; Sivamani, S.; Dotto, G.L.; Landarani, M.; Ivanets, A.; Kyzas, G.Z.; Hosseini-Bandegharaei, A. Activated Carbon from Wood Wastes for the Removal of Uranium and Thorium Ions through Modification with Mineral Acid. Colloids Surf. A Physicochem. Eng. Asp. 2020, 607, 125516. [Google Scholar] [CrossRef]
- (Ölmez) Aytaş, Ş.; Akyil, S.; Aslani, M.A.A.; Aytekin, U. Removal of Uranium from Aqueous Solutions by Diatomite (Kieselguhr). J. Radioanal. Nucl. Chem. 1999, 240, 973–976. [Google Scholar] [CrossRef]
- Inyang, M.I.; Gao, B.; Yao, Y.; Xue, Y.; Zimmerman, A.; Mosa, A.; Pullammanappallil, P.; Ok, Y.S.; Cao, X. A Review of Biochar as a Low-Cost Adsorbent for Aqueous Heavy Metal Removal. Crit. Rev. Environ. Sci. Technol. 2016, 46, 406–433. [Google Scholar] [CrossRef]
- Ahmad, M.; Rajapaksha, A.U.; Lim, J.E.; Zhang, M.; Bolan, N.; Mohan, D.; Vithanage, M.; Lee, S.S.; Ok, Y.S. Biochar as a Sorbent for Contaminant Management in Soil and Water: A Review. Chemosphere 2014, 99, 19–33. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, W.; Mehmood, S.; Qaswar, M.; Ali, S.; Khan, Z.H.; Ying, H.; Chen, D.Y.; Núñez-Delgado, A. Oxidized Biochar Obtained from Rice Straw as Adsorbent to Remove Uranium (VI) from Aqueous Solutions. J. Environ. Chem. Eng. 2021, 9, 105104. [Google Scholar] [CrossRef]
- Dai, L.; Li, L.; Zhu, W.; Ma, H.; Huang, H.; Lu, Q.; Yang, M.; Ran, Y. Post-Engineering of Biochar via Thermal Air Treatment for Highly Efficient Promotion of Uranium(VI) Adsorption. Bioresour. Technol. 2020, 298, 122576. [Google Scholar] [CrossRef] [PubMed]
- Guan, X.; Zhou, J.; Ma, N.; Chen, X.; Gao, J.; Zhang, R. Studies on Modified Conditions of Biochar and the Mechanism for Fluoride Removal. Desalin. Water Treat. 2015, 55, 440–447. [Google Scholar] [CrossRef]
- Guilhen, S.N.; Rovani, S.; de Araujo, L.G.; Tenório, J.A.S.S.; Mašek, O. Uranium Removal from Aqueous Solution Using Macauba Endocarp-Derived Biochar: Effect of Physical Activation. Environ. Pollut. 2021, 272, 116022. [Google Scholar] [CrossRef] [PubMed]
- Philippou, K.; Anastopoulos, I.; Dosche, C.; Pashalidis, I. Synthesis and Characterization of a Novel Fe3O4-Loaded Oxidized Biochar from Pine Needles and Its Application for Uranium Removal. Kinetic, Thermodynamic, and Mechanistic Analysis. J. Environ. Manage. 2019, 252, 109677. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, W.; Mehmood, S.; Núñez-Delgado, A.; Ali, S.; Qaswar, M.; Khan, Z.H.; Ying, H.; Chen, D.Y. Utilization of Citrullus Lanatus L. Seeds to Synthesize a Novel MnFe2O4-Biochar Adsorbent for the Removal of U(VI) from Wastewater: Insights and Comparison between Modified and Raw Biochar. Sci. Total Environ. 2021, 771, 144955. [Google Scholar] [CrossRef]
- Ahmed, W.; Mehmood, S.; Núñez-delgado, A.; Qaswar, M.; Ali, S.; Ying, H.; Liu, Z.; Mahmood, M.; Chen, D. Fabrication, Characterization and U (VI) Sorption Properties of a Novel Biochar Derived from Tribulus Terrestris via Two Different Approaches. Sci. Total Environ. 2021, 780, 146617. [Google Scholar] [CrossRef]
- Habibi, N.; Rouhi, P.; Ramavandi, B. Modification of Tamarix Hispida Biochar by Lanthanum Chloride for Enhanced Fluoride Adsorption from Synthetic and Real Wastewater. Environ. Prog. Sustain. Energy 2019, 38, S298–S305. [Google Scholar] [CrossRef]
- Liang, L.; Xi, F.; Tan, W.; Meng, X.; Hu, B.; Wang, X. Review of Organic and Inorganic Pollutants Removal by Biochar and Biochar-Based Composites. Biochar 2021, 3, 255–281. [Google Scholar] [CrossRef]
- Mohan, D.; Sarswat, A.; Ok, Y.S.; Pittman, C.U. Organic and Inorganic Contaminants Removal from Water with Biochar, a Renewable, Low Cost and Sustainable Adsorbent—A Critical Review. Bioresour. Technol. 2014, 160, 191–202. [Google Scholar] [CrossRef] [PubMed]
- Gwenzi, W.; Chaukura, N.; Noubactep, C.; Mukome, F.N.D. Biochar-Based Water Treatment Systems as a Potential Low-Cost and Sustainable Technology for Clean Water Provision. J. Environ. Manage. 2017, 197, 732–749. [Google Scholar] [CrossRef]
- Sizmur, T.; Fresno, T.; Akgül, G.; Frost, H.; Moreno-Jiménez, E. Biochar Modification to Enhance Sorption of Inorganics from Water. Bioresour. Technol. 2017, 246, 34–47. [Google Scholar] [CrossRef] [PubMed]
- Tan, X.; Liu, Y.; Zeng, G.; Wang, X.; Hu, X.; Gu, Y.; Yang, Z. Application of Biochar for the Removal of Pollutants from Aqueous Solutions. Chemosphere 2015, 125, 70–85. [Google Scholar] [CrossRef] [PubMed]
- Abbas, Z.; Ali, S.; Rizwan, M.; Zaheer, I.E.; Malik, A.; Riaz, M.A.; Shahid, M.R.; ur Rehman, M.Z.; Al-Wabel, M.I. A Critical Review of Mechanisms Involved in the Adsorption of Organic and Inorganic Contaminants through Biochar. Arab. J. Geosci. 2018, 11, 448. [Google Scholar] [CrossRef]
- Ambaye, T.G.; van Hullebusch, M.V.E.D.; Rtimi, A.A.S. Mechanisms and Adsorption Capacities of Biochar for the Removal of Organic and Inorganic Pollutants from Industrial Wastewater. Int. J. Environ. Sci. Technol. 2021, 18, 3273–3294. [Google Scholar] [CrossRef]
- Zeghioud, H.; Fryda, L.; Djelal, H.; Assadi, A.; Kane, A. A Comprehensive Review of Biochar in Removal of Organic Pollutants from Wastewater: Characterization, Toxicity, Activation/Functionalization and Influencing Treatment Factors. J. Water Process Eng. 2022, 47, 102801. [Google Scholar] [CrossRef]
- Zhang, M.; Song, G.; Gelardi, D.L.; Huang, L.; Khan, E.; Mašek, O.; Parikh, S.J.; Ok, Y.S. Evaluating Biochar and Its Modifications for the Removal of Ammonium, Nitrate, and Phosphate in Water. Water Res. 2020, 186, 116303. [Google Scholar] [CrossRef]
- Shakoor, M.B.; Ye, Z.L.; Chen, S. Engineered Biochars for Recovering Phosphate and Ammonium from Wastewater: A Review. Sci. Total Environ. 2021, 779, 146240. [Google Scholar] [CrossRef]
- Almanassra, I.W.; Mckay, G.; Kochkodan, V.; Ali Atieh, M.; Al-Ansari, T. A State of the Art Review on Phosphate Removal from Water by Biochars. Chem. Eng. J. 2021, 409, 128211. [Google Scholar] [CrossRef]
- Ambika, S.; Kumar, M.; Pisharody, L.; Malhotra, M.; Kumar, G.; Sreedharan, V.; Singh, L.; Nidheesh, P.V.; Bhatnagar, A. Modified Biochar as a Green Adsorbent for Removal of Hexavalent Chromium from Various Environmental Matrices: Mechanisms, Methods, and Prospects. Chem. Eng. J. 2022, 439, 135716. [Google Scholar] [CrossRef]
- Dai, Y.; Zhang, N.; Xing, C.; Cui, Q.; Sun, Q. The Adsorption, Regeneration and Engineering Applications of Biochar for Removal Organic Pollutants: A Review. Chemosphere 2019, 223, 12–27. [Google Scholar] [CrossRef] [PubMed]
- Lakshmi, D.; Akhil, D.; Kartik, A.; Gopinath, K.P.; Arun, J.; Bhatnagar, A.; Rinklebe, J.; Kim, W.; Muthusamy, G. Artificial Intelligence (AI) Applications in Adsorption of Heavy Metals Using Modified Biochar. Sci. Total Environ. 2021, 801, 149623. [Google Scholar] [CrossRef]
- Liu, T.; Lawluvy, Y.; Shi, Y.; Ighalo, J.O.; He, Y.; Zhang, Y.; Yap, P.S. Adsorption of Cadmium and Lead from Aqueous Solution Using Modified Biochar: A Review. J. Environ. Chem. Eng. 2022, 10, 106502. [Google Scholar] [CrossRef]
- Qiu, B.; Tao, X.; Wang, H.; Li, W.; Ding, X.; Chu, H. Biochar as a Low-Cost Adsorbent for Aqueous Heavy Metal Removal: A Review. J. Anal. Appl. Pyrolysis 2021, 155, 105081. [Google Scholar] [CrossRef]
- Sharma, P.K.; Kumar, R.; Singh, R.K.; Sharma, P.; Ghosh, A. Review on Arsenic Removal Using Biochar-Based Materials. Groundw. Sustain. Dev. 2022, 17, 100740. [Google Scholar] [CrossRef]
- Wang, L.; Wang, Y.; Ma, F.; Tankpa, V.; Bai, S.; Guo, X.; Wang, X. Mechanisms and Reutilization of Modified Biochar Used for Removal of Heavy Metals from Wastewater: A Review. Sci. Total Environ. 2019, 668, 1298–1309. [Google Scholar] [CrossRef]
- Zheng, C.; Yang, Z.; Si, M.; Zhu, F.; Yang, W.; Zhao, F.; Shi, Y. Application of Biochars in the Remediation of Chromium Contamination: Fabrication, Mechanisms, and Interfering Species. J. Hazard. Mater. 2021, 407, 124376. [Google Scholar] [CrossRef]
- Zoroufchi Benis, K.; Motalebi Damuchali, A.; Soltan, J.; McPhedran, K.N. Treatment of Aqueous Arsenic—A Review of Biochar Modification Methods. Sci. Total Environ. 2020, 739, 139750. [Google Scholar] [CrossRef]
- Godwin, P.M.; Pan, Y.; Xiao, H.; Afzal, M.T. Progress in Preparation and Application of Modified Biochar for Improving Heavy Metal Ion Removal From Wastewater. J. Bioresour. Bioprod. 2019, 4, 31–42. [Google Scholar] [CrossRef]
- Hussin, F.; Aroua, M.K.; Szlachta, M. Biochar Derived from Fruit By-Products Using Pyrolysis Process for the Elimination of Pb(II) Ion: An Updated Review. Chemosphere 2022, 287, 132250. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Dong, X.; da Silva, E.B.; de Oliveira, L.M.; Chen, Y.; Ma, L.Q. Mechanisms of Metal Sorption by Biochars: Biochar Characteristics and Modifications. Chemosphere 2017, 178, 466–478. [Google Scholar] [CrossRef] [PubMed]
- Berslin, D.; Reshmi, A.; Sivaprakash, B.; Rajamohan, N.; Kumar, P.S. Remediation of Emerging Metal Pollutants Using Environment Friendly Biochar- Review on Applications and Mechanism. Chemosphere 2022, 290, 133384. [Google Scholar] [CrossRef] [PubMed]
- Cheng, N.; Wang, B.; Wu, P.; Lee, X.; Xing, Y.; Chen, M.; Gao, B. Adsorption of Emerging Contaminants from Water and Wastewater by Modified Biochar: A Review. Environ. Pollut. 2021, 273, 116448. [Google Scholar] [CrossRef]
- Amusat, S.O.; Kebede, T.G.; Dube, S.; Nindi, M.M. Ball-Milling Synthesis of Biochar and Biochar–Based Nanocomposites and Prospects for Removal of Emerging Contaminants: A Review. J. Water Process Eng. 2021, 41, 101993. [Google Scholar] [CrossRef]
- Shahid, M.K.; Kashif, A.; Fuwad, A.; Choi, Y. Current Advances in Treatment Technologies for Removal of Emerging Contaminants from Water—A Critical Review. Coord. Chem. Rev. 2021, 442, 213993. [Google Scholar] [CrossRef]
- Biswal, B.K.; Vijayaraghavan, K.; Tsen-Tieng, D.L.; Balasubramanian, R. Biochar-Based Bioretention Systems for Removal of Chemical and Microbial Pollutants from Stormwater: A Critical Review. J. Hazard. Mater. 2022, 422, 126886. [Google Scholar] [CrossRef]
- Hassan, M.; Liu, Y.; Naidu, R.; Parikh, S.J.; Du, J.; Qi, F.; Willett, I.R. Influences of Feedstock Sources and Pyrolysis Temperature on the Properties of Biochar and Functionality as Adsorbents: A Meta-Analysis. Sci. Total Environ. 2020, 744, 140714. [Google Scholar] [CrossRef]
- Tomczyk, A.; Sokołowska, Z.; Boguta, P. Biochar Physicochemical Properties: Pyrolysis Temperature and Feedstock Kind Effects. Rev. Environ. Sci. Bio/Technol. 2020, 19, 191–215. [Google Scholar] [CrossRef] [Green Version]
- Yang, H.; Yan, R.; Chen, H.; Lee, D.H.; Zheng, C. Characteristics of Hemicellulose, Cellulose and Lignin Pyrolysis. Fuel 2007, 86, 1781–1788. [Google Scholar] [CrossRef]
- Yaashikaa, P.R.; Kumar, P.S.; Varjani, S.; Saravanan, A. A Critical Review on the Biochar Production Techniques, Characterization, Stability and Applications for Circular Bioeconomy. Biotechnol. Rep. 2020, 28, e00570. [Google Scholar] [CrossRef] [PubMed]
- Wijitkosum, S. Biochar Derived from Agricultural Wastes and Wood Residues for Sustainable Agricultural and Environmental Applications. Int. Soil Water Conserv. Res. 2022, 10, 335–341. [Google Scholar] [CrossRef]
- Ippolito, J.A.; Cui, L.; Kammann, C.; Wrage-Mönnig, N.; Estavillo, J.M.; Fuertes-Mendizabal, T.; Cayuela, M.L.; Sigua, G.; Novak, J.; Spokas, K.; et al. Feedstock Choice, Pyrolysis Temperature and Type Influence Biochar Characteristics: A Comprehensive Meta-Data Analysis Review. Biochar 2020, 2, 421–438. [Google Scholar] [CrossRef]
- Enaime, G.; Baçaoui, A.; Yaacoubi, A.; Lübken, M. Biochar for Wastewater Treatment—Conversion Technologies and Applications. Appl. Sci. 2020, 10, 3492. [Google Scholar] [CrossRef]
- Janu, R.; Mrlik, V.; Ribitsch, D.; Hofman, J.; Sedláček, P.; Bielská, L.; Soja, G. Biochar Surface Functional Groups as Affected by Biomass Feedstock, Biochar Composition and Pyrolysis Temperature. Carbon Resour. Convers. 2021, 4, 36–46. [Google Scholar] [CrossRef]
- Zhang, Q.; Wang, Y.; Wang, Z.; Zhang, Z.; Wang, X.; Yang, Z. Active Biochar Support Nano Zero-Valent Iron for Efficient Removal of U(VI) from Sewage Water. J. Alloy. Compd. 2021, 852, 156993. [Google Scholar] [CrossRef]
- Saleh, T.A.; Mustaqeem, M.; Khaled, M. Water Treatment Technologies in Removing Heavy Metal Ions from Wastewater: A Review. Environ. Nanotechnol. Monit. Manag. 2022, 17, 100617. [Google Scholar] [CrossRef]
- Dulama, M.; Iordache, M.; Deneanu, N. Treatment of Uranium Contaminated Wastewater a review. In Proceedings of NUCLEAR 2013 the 6th Annual International Conference on Sustainable Development through Nuclear Research and Education Part 3/3; 2013; pp. 80–87. Available online: http://inis.iaea.org/search/search.aspx?orig_q=RN:47056986 (accessed on 22 October 2022).
- Boraah, N.; Chakma, S.; Kaushal, P. Attributes of Wood Biochar as an Efficient Adsorbent for Remediating Heavy Metals and Emerging Contaminants from Water: A Critical Review and Bibliometric Analysis. J. Environ. Chem. Eng. 2022, 10, 107825. [Google Scholar] [CrossRef]
- Sen, K.; Mishra, D.; Debnath, P.; Mondal, A.; Mondal, N.K. Adsorption of Uranium (VI) from Groundwater by Silicon Containing Biochar Supported Iron Oxide Nanoparticle. Bioresour. Technol. Rep. 2021, 14, 100659. [Google Scholar] [CrossRef]
- Jin, J.; Li, S.; Peng, X.; Liu, W.; Zhang, C.; Yang, Y.; Han, L.; Du, Z.; Sun, K.; Wang, X. HNO3 Modified Biochars for Uranium ( VI ) Removal from Aqueous Solution. Bioresour. Technol. 2018, 256, 247–253. [Google Scholar] [CrossRef]
- Guo, Y.; Liu, X.; Xie, S.; Liu, H.; Wang, C.; Wang, L. 3D ZnO Modified Biochar-Based Hydrogels for Removing U(VI) in Aqueous Solution. Colloids Surf. A Physicochem. Eng. Asp. 2022, 642, 128606. [Google Scholar] [CrossRef]
- Ioannou, K.; Hadjiyiannis, P.; Liatsou, I.; Pashalidis, I. U(VI) Adsorption by Biochar Fiber–MnO2 Composites. J. Radioanal. Nucl. Chem. 2019, 320, 425–432. [Google Scholar] [CrossRef]
- Lingamdinne, L.P.; Choi, J.S.; Angaru, G.K.R.; Karri, R.R.; Yang, J.K.; Chang, Y.Y.; Koduru, J.R. Magnetic-Watermelon Rinds Biochar for Uranium-Contaminated Water Treatment Using an Electromagnetic Semi-Batch Column with Removal Mechanistic Investigations. Chemosphere 2022, 286, 131776. [Google Scholar] [CrossRef] [PubMed]
- Solanki, Y.S.; Agarwal, M.; Gupta, A.B.; Gupta, S.; Shukla, P. Fluoride Occurrences, Health Problems, Detection, and Remediation Methods for Drinking Water: A Comprehensive Review. Sci. Total Environ. 2022, 807, 150601. [Google Scholar] [CrossRef]
- Nizam, S.; Virk, H.S.; Sen, I.S. High Levels of Fluoride in Groundwater from Northern Parts of Indo-Gangetic Plains Reveals Detrimental Fluorosis Health Risks. Environ. Adv. 2022, 8, 100200. [Google Scholar] [CrossRef]
- Kashyap, S.J.; Sankannavar, R.; Madhu, G.M. Fluoride Sources, Toxicity and Fluorosis Management Techniques—A Brief Review. J. Hazard. Mater. Lett. 2021, 2, 100033. [Google Scholar] [CrossRef]
- Zhou, N.; Guo, X.; Ye, C.; Yan, L.; Gu, W.; Wu, X.; Zhou, Q.; Yang, Y.; Wang, X.; Cheng, Q. Enhanced Fluoride Removal from Drinking Water in Wide PH Range Using La/Fe/Al Oxides Loaded Rice Straw Biochar. Water Supply 2022, 22, 779–794. [Google Scholar] [CrossRef]
- Yan, L.; Gu, W.; Zhou, N.; Ye, C.; Yang, Y. Preparation and Characterization of Wheat Straw Biochar Loaded with Aluminium/Lanthanum Hydroxides: A Novel Adsorbent for Removing Fluoride from Drinking Water. Environ. Technol. 2021, 43, 2771–2784. [Google Scholar] [CrossRef]
- Mohan, D.; Kumar, S.; Srivastava, A. Fluoride Removal from Ground Water Using Magnetic and Nonmagnetic Corn Stover Biochars. Ecol. Eng. 2014, 73, 798–808. [Google Scholar] [CrossRef]
- Halder, G.; Khan, A.A.; Dhawane, S. Fluoride Sorption Onto a Steam-Activated Biochar Derived From Cocos Nucifera Shell. CLEAN–Soil Air Water 2016, 44, 124–133. [Google Scholar] [CrossRef]
- Mei, L.; Qiao, H.; Ke, F.; Peng, C.; Hou, R.; Wan, X.; Cai, H. One-Step Synthesis of Zirconium Dioxide-Biochar Derived from Camellia Oleifera Seed Shell with Enhanced Removal Capacity for Fluoride from Water. Appl. Surf. Sci. 2020, 509, 144685. [Google Scholar] [CrossRef]
- Sadhu, M.; Bhattacharya, P.; Vithanage, M.; Padmaja Sudhakar, P. Adsorptive Removal of Fluoride Using Biochar—A Potential Application in Drinking Water Treatment. Sep. Purif. Technol. 2022, 278, 119106. [Google Scholar] [CrossRef]
- Wang, J.; Chen, N.; Li, M.; Feng, C. Efficient Removal of Fluoride Using Polypyrrole-Modified Biochar Derived from Slow Pyrolysis of Pomelo Peel: Sorption Capacity and Mechanism. J. Polym. Environ. 2018, 26, 1559–1572. [Google Scholar] [CrossRef]
- Kumar, H.; Patel, M.; Mohan, D. Simplified Batch and Fixed-Bed Design System for E Ffi Cient and Sustainable Fluoride Removal from Water Using Slow Pyrolyzed Okra Stem and Black Gram Straw Biochars. ACS Omega 2019, 4, 19513–19525. [Google Scholar] [CrossRef]
- Sha, Q.; Xie, H.; Liu, W.; Yang, D.; He, Y.; Yang, C.; Wang, N. Removal of Fluoride Using Platanus Acerifoli Leaves Biochar—An Efficient and Low-Cost Application in Wastewater Treatment. Environ. Technol. 2021, 1–43. [Google Scholar] [CrossRef]
- Jha, S.; Nanda, S.; Acharya, B.; Dalai, A.K. A Review of Thermochemical Conversion of Waste Biomass to Biofuels. Energies 2022, 15, 6352. [Google Scholar] [CrossRef]
- Sun, Y.; Gao, B.; Yao, Y.; Fang, J.; Zhang, M.; Zhou, Y.; Chen, H.; Yang, L. Effects of Feedstock Type, Production Method, and Pyrolysis Temperature on Biochar and Hydrochar Properties. Chem. Eng. J. 2014, 240, 574–578. [Google Scholar] [CrossRef]
- Tripathi, M.; Sahu, J.N.; Ganesan, P. Effect of Process Parameters on Production of Biochar from Biomass Waste through Pyrolysis: A Review. Renew. Sustain. Energy Rev. 2016, 55, 467–481. [Google Scholar] [CrossRef]
- Asadi, H.; Ghorbani, M.; Rezaei-Rashti, M.; Abrishamkesh, S.; Amirahmadi, E.; Chengrong, C.; Gorji, M. Application of Rice Husk Biochar for Achieving Sustainable Agriculture and Environment. Rice Sci. 2021, 28, 325–343. [Google Scholar] [CrossRef]
- Windeatt, J.H.; Ross, A.B.; Williams, P.T.; Forster, P.M.; Nahil, M.A.; Singh, S. Characteristics of Biochars from Crop Residues: Potential for Carbon Sequestration and Soil Amendment. J. Environ. Manage. 2014, 146, 189–197. [Google Scholar] [CrossRef] [PubMed]
- Reguyal, F.; Sarmah, A.K.; Gao, W. Synthesis of Magnetic Biochar from Pine Sawdust via Oxidative Hydrolysis of FeCl2 for the Removal Sulfamethoxazole from Aqueous Solution. J. Hazard. Mater. 2017, 321, 868–878. [Google Scholar] [CrossRef] [PubMed]
- Umar, M.; Amru, A.R.; Amin, N.H.M.; Zaid, H.M.; Guan, B.H. Biomass Activated Carbon from Oil Palm Shell as Potential Material to Control Filtration Loss in Water-Based Drilling Fluid. Optim. Based Model Using Fuzzy Other Stat. Tech. Towar. Environ. Sustain. 2020, 55–65. [Google Scholar]
- Hu, H.; Zhang, X.; Wang, T.; Sun, L.; Wu, H.; Chen, X. Bamboo (Acidosasa longiligula) Shoot Shell Biochar: Its Potential Application to Isolation of Uranium(VI) from Aqueous Solution. J. Radioanal. Nucl. Chem. 2018, 316, 349–362. [Google Scholar] [CrossRef]
- Ying, D.; Hong, P.; Jiali, F.; Qinqin, T.; Yuhui, L.; Youqun, W.; Zhibin, Z.; Xiaohong, C.; Yunhai, L. Removal of Uranium Using MnO2/Orange Peel Biochar Composite Prepared by Activation and in-Situ Deposit in a Single Step. Biomass Bioenergy 2020, 142, 105772. [Google Scholar] [CrossRef]
- Liao, J.; Ding, L.; Zhang, Y.; Zhu, W. Efficient Removal of Uranium from Wastewater Using Pig Manure Biochar: Understanding Adsorption and Binding Mechanisms. J. Hazard. Mater. 2022, 423, 127190. [Google Scholar] [CrossRef]
- Liao, J.; He, X.; Zhang, Y.; Zhu, W.; Zhang, L.; He, Z. Bismuth Impregnated Biochar for Efficient Uranium Removal from Solution: Adsorption Behavior and Interfacial Mechanism. Sci. Total Environ. 2022, 819, 153145. [Google Scholar] [CrossRef]
- Wallace, A.R.; Su, C.; Choi, Y.-K.; Kan, E.; Sun, W. Removal of Fluoride from Water Using a Calcium-Modified Dairy Manure-Derived Biochar. J. Environ. Eng. 2020, 146, 1–10. [Google Scholar] [CrossRef]
- Xu, D.; Fan, X.; Chen, Q.; Qiao, S.; Zhang, J.; Yang, Y.; Wang, H.; Zhang, L.; Hou, J. Removal of Nitrogen and Phosphorus from Water by Sludge-Based Biochar Modified by Montmorillonite Coupled with Nano Zero-Valent Iron. Water Sci. Technol. 2022, 85, 2114–2128. [Google Scholar] [CrossRef]
- Hu, R.; Xiao, J.; Wang, T.; Chen, G.; Chen, L.; Tian, X. Engineering of Phosphate-Functionalized Biochars with Highly Developed Surface Area and Porosity for Efficient and Selective Extraction of Uranium. Chem. Eng. J. 2020, 379, 122388. [Google Scholar] [CrossRef]
- Alkurdi, S.S.A.; Al-juboori, R.A.; Bundschuh, J.; Bowtell, L. Effect of Pyrolysis Conditions on Bone Char Characterization and Its Ability for Arsenic and Fluoride Removal. Environ. Pollut. 2020, 262, 114221. [Google Scholar] [CrossRef] [PubMed]
- Thúy, N.; Chi, L.; Anto, S.; Shan, T.; Kumar, S.S.; Shanmugam, S.; Samuel, M.S.; Mathimani, T.; Brindhadevi, K.; Pugazhendhi, A. A Review on Biochar Production Techniques and Biochar Based Catalyst for Biofuel Production from Algae. Fuel 2020, 287, 119411. [Google Scholar] [CrossRef]
- Gabhane, J.W.; Bhange, V.P.; Patil, P.D.; Bankar, S.T.; Kumar, S. Recent Trends in Biochar Production Methods and Its Application as a Soil Health Conditioner: A Review. SN Appl. Sci. 2020, 2, 1307. [Google Scholar] [CrossRef]
- Wang, J.; Wang, S. Preparation, Modification and Environmental Application of Biochar: A Review. J. Clean. Prod. 2019, 227, 1002–1022. [Google Scholar] [CrossRef]
- Chen, D.; Zheng, Z.; Fu, K.; Zeng, Z.; Wang, J.; Lu, M. Torrefaction of Biomass Stalk and Its Effect on the Yield and Quality of Pyrolysis Products. Fuel 2015, 159, 27–32. [Google Scholar] [CrossRef]
- Heidenreich, S.; Müller, M.; Foscolo, P.U. Chapter 3—Biomass Pretreatment. In Advanced biomass gasification; Heidenreich, S., Müller, M., Foscolo, P.U.B.T.-A.B.G., Eds.; Academic Press: Cambridge, MA, USA, 2016; pp. 11–17. ISBN 978-0-12-804296-0. Available online: https://www.sciencedirect.com/science/article/pii/B9780128042960000038 (accessed on 22 October 2022).
- Hadjittofi, L.; Pashalidis, I. Uranium Sorption from Aqueous Solutions by Activated Biochar Fibres Investigated by FTIR Spectroscopy and Batch Experiments. J. Radioanal. Nucl. Chem. 2015, 304, 897–904. [Google Scholar] [CrossRef]
- Liatsou, I.; Michail, G.; Demetriou, M.; Pashalidis, I. Uranium Binding by Biochar Fibres Derived from Luffa Cylindrica after Controlled Surface Oxidation. J. Radioanal. Nucl. Chem. 2017, 311, 871–875. [Google Scholar] [CrossRef]
- Paschalidou, P.; Pashalidis, I.; Manariotis, I.D.; Karapanagioti, H.K. Hyper Sorption Capacity of Raw and Oxidized Biochars from Various Feedstocks for U(VI). J. Environ. Chem. Eng. 2020, 8, 103932. [Google Scholar] [CrossRef]
- Stasi, C.; Georgiou, E.; Ioannidis, I.; Pashalidis, I. Uranium Removal from Laboratory and Environmental Waters by Oxidised Biochar Prepared from Palm Tree Fibres. J. Radioanal. Nucl. Chem. 2022, 331, 375–381. [Google Scholar] [CrossRef]
- Xia, D.; Liu, Y.; Cheng, X.; Gu, P.; Chen, Q.; Zhang, Z. Applied Surface Science Temperature-Tuned Fish-Scale Biochar with Two-Dimensional Homogeneous Porous Structure: A Promising Uranium Extractant. Appl. Surf. Sci. 2022, 591, 153136. [Google Scholar] [CrossRef]
- Saikia, R.; Goswami, R.; Bordoloi, N.; Senapati, K.K.; Pant, K.K.; Kumar, M.; Kataki, R. Removal of Arsenic and Fluoride from Aqueous Solution by Biomass Based Activated Biochar: Optimization through Response Surface Methodology. J. Environ. Chem. Eng. 2017, 5, 5528–5539. [Google Scholar] [CrossRef]
- Han, L.; Zhang, E.; Yang, Y.; Sun, K.; Fang, L. Highly Efficient U(VI) Removal by Chemically Modified Hydrochar and Pyrochar Derived from Animal Manure. J. Clean. Prod. 2020, 264, 121542. [Google Scholar] [CrossRef]
- Li, M.; Liu, H.; Chen, T.; Dong, C.; Sun, Y. Synthesis of Magnetic Biochar Composites for Enhanced Uranium(VI) Adsorption. Sci. Total Environ. 2019, 651, 1020–1028. [Google Scholar] [CrossRef] [PubMed]
- Liao, J.; Xiong, T.; Ding, L.; Zhang, Y.; Zhu, W. Effective Separation of Uranium(VI) from Wastewater Using a Magnetic Carbon as a Recyclable Adsorbent. Sep. Purif. Technol. 2022, 282, 120140. [Google Scholar] [CrossRef]
- Ding, L.; Tan, W.; Xie, S.; Mumford, K.; Lv, J.; Wang, H.; Fang, Q.; Zhang, X.; Wu, X.; Li, M. Uranium Adsorption and Subsequent Re-Oxidation under Aerobic Conditions by Leifsonia sp.—Coated Biochar as Green Trapping Agent. Environ. Pollut. 2018, 242, 778–787. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Wang, L.; Wang, J. Cerium Alginate Cross-Linking with Biochar Beads for Fast Fluoride Removal over a Wide PH Range. Colloids Surf. A Physicochem. Eng. Asp. 2022, 636, 128161. [Google Scholar] [CrossRef]
- An, Q.; Li, X.Q.; Nan, H.Y.; Yu, Y.; Jiang, J.N. The Potential Adsorption Mechanism of the Biochars with Different Modification Processes to Cr(VI). Environ. Sci. Pollut. Res. 2018, 25, 31346–31357. [Google Scholar] [CrossRef]
- Chemerys, V.; Baltrėnaitė, E. Modified Biochar: A Review on Modifications of Biochar Towards Its Enhanced Adsorptive Properties. 2016. [Google Scholar]
- Ahmed, W.; Núñez-Delgado, A.; Mehmood, S.; Ali, S.; Qaswar, M.; Shakoor, A.; Chen, D.Y. Highly Efficient Uranium (VI) Capture from Aqueous Solution by Means of a Hydroxyapatite-Biochar Nanocomposite: Adsorption Behavior and Mechanism. Environ. Res. 2021, 201, 111518. [Google Scholar] [CrossRef]
- Wang, S.; Guo, W.; Gao, F.; Wang, Y.; Gao, Y. Lead and Uranium Sorptive Removal from Aqueous Solution Using Magnetic and Nonmagnetic Fast Pyrolysis Rice Husk Biochars. RSC Adv. 2018, 8, 13205–13217. [Google Scholar] [CrossRef] [Green Version]
- Chen, W.; Feng, J.; Liu, S.; Zhang, J.; Cai, Y.; Lv, Z.; Fang, M.; Tan, X. A Green and Economical MgO/Biochar Composite for the Removal of U(VI) from Aqueous Solutions. Chem. Eng. Res. Des. 2022, 180, 391–401. [Google Scholar] [CrossRef]
- Lyu, P.; Wang, G.; Wang, B.; Yin, Q.; Li, Y.; Deng, N. Adsorption and Interaction Mechanism of Uranium (VI) from Aqueous Solutions on Phosphate-Impregnation Biochar Cross-Linked MgAl Layered Double-Hydroxide Composite. Appl. Clay Sci. 2021, 209, 106146. [Google Scholar] [CrossRef]
- Li, N.; Yin, M.; Tsang, D.C.W.; Yang, S.; Liu, J.; Li, X.; Song, G.; Wang, J. Mechanisms of U(VI) Removal by Biochar Derived from Ficus Microcarpa Aerial Root: A Comparison between Raw and Modified Biochar. Sci. Total Environ. 2019, 697, 134115. [Google Scholar] [CrossRef]
- Hu, Q.; Zhu, Y.; Hu, B.; Lu, S.; Sheng, G. Mechanistic Insights into Sequestration of U ( VI ) toward Magnetic Biochar: Batch, XPS and EXAFS Techniques. J. Environ. Sci. 2018, 70, 217–225. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Xiao, J.; Hu, R.; Wang, T.; Shao, X.; Chen, G.; Chen, L.; Tian, X. Engineered Phosphorous-Functionalized Biochar with Enhanced Porosity Using Phytic Acid-Assisted Ball Milling for Efficient and Selective Uptake of Aquatic Uranium. J. Mol. Liq. 2020, 303, 112659. [Google Scholar] [CrossRef]
- Liao, J.; Chen, H.; Zhang, Y.; Zhu, W. Pyrolysis of Animal Manure under Nitrogen Atmosphere: An Environment Protection Way to Obtain Animal Manure Biochar for High-Efficient Adsorption of Uranium (VI). J. Anal. Appl. Pyrolysis 2022, 163, 105493. [Google Scholar] [CrossRef]
- Zheng, B.; Liao, J.; Ding, L.; Zhang, Y.; Zhu, W. High Efficiency Adsorption of Uranium in Solution with Magnesium Oxide Embedded Horse Manure-Derived Biochar. J. Environ. Chem. Eng. 2021, 9, 106897. [Google Scholar] [CrossRef]
- Sun, Y.; Zeng, B.; Dai, Y.; Liang, X.; Zhang, L.; Ahmad, R.; Su, X. Modification of Sludge-Based Biochar Using Air Roasting-Oxidation and Its Performance in Adsorption of Uranium(VI) from Aqueous Solutions. J. Colloid Interface Sci. 2022, 614, 547–555. [Google Scholar] [CrossRef]
- Noli, F.; Kapashi, E.; Pashalidis, I.; Margellou, A.; Karfaridis, D. The Effect of Chemical and Thermal Modifications on the Biosorption of Uranium in Aqueous Solutions Using Winery Wastes. J. Mol. Liq. 2022, 351, 118665. [Google Scholar] [CrossRef]
- Pang, H.; Diao, Z.; Wang, X.; Ma, Y.; Yu, S.; Zhu, H.; Chen, Z.; Hu, B.; Chen, J.; Wang, X. Adsorptive and Reductive Removal of U(VI) by Dictyophora Indusiate-Derived Biochar Supported Sulfide NZVI from Wastewater. Chem. Eng. J. 2019, 366, 368–377. [Google Scholar] [CrossRef]
- Wang, B.; Zheng, J.; Li, Y.; Zaidi, A.; Hu, Y.; Hu, B. Fabrication of δ-MnO2-Modified Algal Biochar for Efficient Removal of U(VI) from Aqueous Solutions. J. Environ. Chem. Eng. 2021, 9, 105625. [Google Scholar] [CrossRef]
- Wang, B.; Li, Y.; Zheng, J.; Hu, Y.; Wang, X.; Hu, B. Efficient Removal of U(VI) from Aqueous Solutions Using the Magnetic Biochar Derived from the Biomass of a Bloom-Forming Cyanobacterium (Microcystis aeruginosa). Chemosphere 2020, 254, 126898. [Google Scholar] [CrossRef] [PubMed]
- Liatsou, I.; Pashalidis, I.; Nicolaides, A. Triggering Selective Uranium Separation from Aqueous Solutions by Using Salophen-Modified Biochar Fibers. J. Radioanal. Nucl. Chem. 2018, 318, 2199–2203. [Google Scholar] [CrossRef]
- Ye, T.; Huang, B.; Wang, Y.; Zhou, L.; Liu, Z. Rapid Removal of Uranium(Ⅵ) Using Functionalized Luffa Rattan Biochar from Aqueous Solution. Colloids Surf. A Physicochem. Eng. Asp. 2020, 606, 125480. [Google Scholar] [CrossRef]
- Akl, Z.F.; Zaki, E.G.; ElSaeed, S.M. Green Hydrogel-Biochar Composite for Enhanced Adsorption of Uranium. ACS Omega 2021, 6, 34193–34205. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Ge, Y.; Wang, G.; Liu, Y.; Xu, X. Highly Efficient Removal of U(VI) in Aqueous Solutions by Tea Waste-Derived Biochar-Supported Iron-Manganese Oxide Composite. J. Radioanal. Nucl. Chem. 2021, 330, 871–882. [Google Scholar] [CrossRef]
- Yadav, K.; Jagadevan, S. Effect of Pyrolysis of Rice Husk–Derived Biochar on the Fuel Characteristics and Adsorption of Fluoride from Aqueous Solution. Bioenergy Res. 2021, 14, 964–977. [Google Scholar] [CrossRef]
- Shen, Z.; Jin, J.; Fu, J.; Yang, M.; Li, F. Anchoring Al- and/or Mg-Oxides to Magnetic Biochars for Co-Uptake of Arsenate and Fluoride from Water. J. Environ. Manage. 2021, 293, 112898. [Google Scholar] [CrossRef]
- Yadav, T.K.; Abhishek; Prasad, B.; Singh, D.; Prasad, K.S. Calcium Pretreated Pinus Roxburghii Wood Biochar for Adsorptive Removal of Fluoride from Aqueous Solution. Biointerface Res. Appl. Chem. 2022, 12, 4307–4316. [Google Scholar] [CrossRef]
- Bombuwala Dewage, N.; Liyanage, A.S.; Pittman, C.U.; Mohan, D.; Mlsna, T. Fast Nitrate and Fluoride Adsorption and Magnetic Separation from Water on A-Fe2O3 and Fe3O4 Dispersed on Douglas Fir Biochar. Bioresour. Technol. 2018, 263, 258–265. [Google Scholar] [CrossRef]
- Herath, A.; Navarathna, C.; Warren, S.; Perez, F.; Pittman, C.U.; Mlsna, T.E. Iron/Titanium Oxide-Biochar (Fe2TiO5/BC): A Versatile Adsorbent/Photocatalyst for Aqueous Cr(VI), Pb2+, F− and Methylene Blue. J. Colloid Interface Sci. 2022, 614, 603–616. [Google Scholar] [CrossRef]
- Rupasinghe, N.K.L.C.; Senanayake, S.M.A.E.; Nanayakkara, K.G.N. Development, Characterization and Mechanisms Study of Protonated Sawdust Biochar-Chitosan Composite Bead Biosorbent for Defluoridation of Contaminated Groundwater. Bioresour. Technol. Rep. 2022, 17, 100946. [Google Scholar] [CrossRef]
- Wan, S.; Lin, J.; Tao, W.; Yang, Y.; Li, Y.; He, F. Enhanced Fluoride Removal from Water by Nanoporous Biochar-Supported Magnesium Oxide. Ind. Eng. Chem. Res. 2019, 58, 9988–9996. [Google Scholar] [CrossRef]
- Meilani, V.; Lee, J.I.; Kang, J.K.; Lee, C.G.; Jeong, S.; Park, S.J. Application of Aluminum-Modified Food Waste Biochar as Adsorbent of Fluoride in Aqueous Solutions and Optimization of Production Using Response Surface Methodology; Elsevier Inc.: Amsterdam, The Netherlands, 2021; Volume 312, ISBN 0003212254. [Google Scholar] [CrossRef]
- Roy, S.; Sengupta, S.; Manna, S.; Das, P. Chemically Reduced Tea Waste Biochar and Its Application in Treatment of Fluoride Containing Wastewater: Batch and Optimization Using Response Surface Methodology. Process Saf. Environ. Prot. 2018, 116, 553–563. [Google Scholar] [CrossRef]
- Naga Babu, A.; Srinivasa Reddy, D.; Suresh Kumar, G.; Ravindhranath, K.; Krishna Mohan, G.V. Sequential Synergetic Sorption Analysis of Gracilaria Rhodophyta Biochar toward Aluminum and Fluoride: A Statistical Optimization Approach. Water Environ. Res. 2020, 92, 880–898. [Google Scholar] [CrossRef]
- Ashraf, I.; Li, R.; Chen, B.; Al-Ansari, N.; Rizwan Aslam, M.; Altaf, A.R.; Elbeltagi, A. Nanoarchitectonics and Kinetics Insights into Fluoride Removal from Drinking Water Using Magnetic Tea Biochar. Int. J. Environ. Res. Public Health 2022, 19, 13092. [Google Scholar] [CrossRef]
- Kumar, P.; Prajapati, A.K.; Dixit, S.; Yadav, V.L. Adsorption of Fluoride from Aqueous Solution Using Biochar Prepared from Waste Peanut Hull. Mater. Res. Express 2019, 6, 125553. [Google Scholar] [CrossRef]
- Khan, B.A.; Ahmad, M.; Iqbal, S.; Bolan, N.; Zubair, S.; Shafique, M.A.; Shah, A. Effectiveness of the Engineered Pinecone-Derived Biochar for the Removal of Fluoride from Water. Environ. Res. 2022, 212, 113540. [Google Scholar] [CrossRef]
- Papari, F.; Najafabadi, P.R.; Ramavandi, B. Fluoride Ion Removal from Aqueous Solution, Groundwater, and Seawater by Granular and Powdered Conocarpus Erectus Biochar. Desalin. Water Treat. 2017, 65, 375–386. [Google Scholar] [CrossRef]
- Chunhui, L.; Jin, T.; Puli, Z.; Bin, Z.; Duo, B.; Xuebin, L. Simultaneous Removal of Fluoride and Arsenic in Geothermal Water in Tibet Using Modified Yak Dung Biochar as an Adsorbent. R. Soc. Open Sci. 2018, 5, 181266. [Google Scholar] [CrossRef] [Green Version]
- Sajjadi, B.; Chen, W.Y.; Egiebor, N.O. A Comprehensive Review on Physical Activation of Biochar for Energy and Environmental Applications. Rev. Chem. Eng. 2019, 35, 735–776. [Google Scholar] [CrossRef]
- Zhao, F.; Shan, R.; Gu, J.; Zhang, Y.; Yuan, H.; Chen, Y. Magnetically Recyclable Loofah Biochar by KMnO4Modification for Adsorption of Cu(II) from Aqueous Solutions. ACS Omega 2022, 7, 8844–8853. [Google Scholar] [CrossRef]
- Zhai, W.; Sakthivel, T.; Chen, F.; Du, C.; Yu, H.; Dai, Z. Amorphous Materials for Elementary-Gas-Involved Electrocatalysis: An Overview. Nanoscale 2021, 13, 19783–19811. [Google Scholar] [CrossRef]
- Su, M.; Tsang, D.C.W.; Ren, X.; Shi, Q.; Tang, J.; Zhang, H.; Kong, L.; Hou, L.; Song, G.; Chen, D. Removal of U(VI) from Nuclear Mining Effluent by Porous Hydroxyapatite: Evaluation on Characteristics, Mechanisms and Performance. Environ. Pollut. 2019, 254, 112891. [Google Scholar] [CrossRef]
- Wang, X.; Guo, Z.; Hu, Z.; Zhang, J. Recent Advances in Biochar Application for Water and Wastewater Treatment: A Review. PeerJ 2020, 8, e9164. [Google Scholar] [CrossRef]
- Mühr-Ebert, E.L.; Wagner, F.; Walther, C. Speciation of Uranium: Compilation of a Thermodynamic Database and Its Experimental Evaluation Using Different Analytical Techniques. Appl. Geochem. 2019, 100, 213–222. [Google Scholar] [CrossRef]
- Kumar, A.; Tripathi, R.M.; Rout, S.; Mishra, M.K.; Ravi, P.M.; Ghosh, A.K. Characterization of Groundwater Composition in Punjab State with Special Emphasis on Uranium Content, Speciation and Mobility. Radiochim. Acta 2014, 102, 239–254. [Google Scholar] [CrossRef]
- Tan, Z.; Yuan, S.; Hong, M.; Zhang, L.; Huang, Q. Mechanism of Negative Surface Charge Formation on Biochar and Its Effect on the Fixation of Soil Cd. J. Hazard. Mater. 2020, 384, 121370. [Google Scholar] [CrossRef]
- Zhang, Z.B.; Cao, X.H.; Liang, P.; Liu, Y.H. Adsorption of Uranium from Aqueous Solution Using Biochar Produced by Hydrothermal Carbonization. J. Radioanal. Nucl. Chem. 2013, 295, 1201–1208. [Google Scholar] [CrossRef]
- Li, X.; Pan, H.; Yu, M.; Wakeel, M.; Luo, J.; Alharbi, N.S.; Liao, Q.; Liu, J. Macroscopic and Molecular Investigations of Immobilization Mechanism of Uranium on Biochar: EXAFS Spectroscopy and Static Batch. J. Mol. Liq. 2018, 269, 64–71. [Google Scholar] [CrossRef]
- Albayari, M.; Nazal, M.K.; Khalili, F.I.; Nordin, N.; Adnan, R. Biochar Derived from Salvadora Persica Branches Biomass as Low-Cost Adsorbent for Removal of Uranium(VI) and Thorium(IV) from Water. J. Radioanal. Nucl. Chem. 2021, 328, 669–678. [Google Scholar] [CrossRef]
- Mishra, V.; Sureshkumar, M.K.; Gupta, N.; Kaushik, C.P. Study on Sorption Characteristics of Uranium onto Biochar Derived from Eucalyptus Wood. Water. Air. Soil Pollut. 2017, 228, 309. [Google Scholar] [CrossRef]
- Xu, Z.; Xing, Y.; Ren, A.; Ma, D.; Li, Y.; Hu, S. Study on Adsorption Properties of Water Hyacinth-Derived Biochar for Uranium (VI). J. Radioanal. Nucl. Chem. 2020, 324, 1317–1327. [Google Scholar] [CrossRef]
- Kumar, S.; Loganathan, V.A.; Gupta, R.B.; Barnett, M.O. An Assessment of U(VI) Removal from Groundwater Using Biochar Produced from Hydrothermal Carbonization. J. Environ. Manage. 2011, 92, 2504–2512. [Google Scholar] [CrossRef]
- Yadav, K.; Jagadevan, S. Influence of Torrefaction and Pyrolysis on Engineered Biochar and Its Applicability in Defluoridation: Insight into Adsorption Mechanism, Batch Adsorber Design and Artificial Neural Network Modelling. J. Anal. Appl. Pyrolysis. 2021, 154, 105015. [Google Scholar] [CrossRef]
- Mohan, D.; Sharma, R.; Singh, V.K.; Steele, P.; Pittman, C.U. Fluoride Removal from Water Using Bio-Char, a Green Waste, Low-Cost Adsorbent: Equilibrium Uptake and Sorption Dynamics Modeling. Ind. Eng. Chem. Res. 2012, 51, 900–914. [Google Scholar] [CrossRef]
- Dong, Q.; Yang, D.; Luo, L.; He, Q.; Cai, F.; Cheng, S.; Chen, Y. Engineering Porous Biochar for Capacitive Fluorine Removal. Sep. Purif. Technol. 2021, 257, 117932. [Google Scholar] [CrossRef]
- Shahid, M.K.; Kim, J.Y.; Shin, G.; Choi, Y. Effect of Pyrolysis Conditions on Characteristics and Fluoride Adsorptive Performance of Bone Char Derived from Bone Residue. J. Water Process Eng. 2020, 37, 101499. [Google Scholar] [CrossRef]
- Mahmoud, M.E.; Khalifa, M.A.; El Wakeel, Y.M.; Header, M.S.; El-Sharkawy, R.M.; Kumar, S.; Abdel-Fattah, T.M. A Novel Nanocomposite of Liquidambar Styraciflua Fruit Biochar-Crosslinked-Nanosilica for Uranyl Removal from Water. Bioresour. Technol. 2019, 278, 124–129. [Google Scholar] [CrossRef]
- Ahmed, N.; Rahman, M.; Won, S.; Shim, S. Biochar Properties and Eco-Friendly Applications for Climate Change Mitigation, Waste Management, and Wastewater Treatment: A Review. Renew. Sustain. Energy Rev. 2017, 79, 255–273. [Google Scholar] [CrossRef]
- Kumar, R.; Sharma, P.; Yang, W.; Sillanpää, M.; Shang, J.; Bhattacharya, P.; Vithanage, M.; Maity, J.P. State-of-the-Art of Research Progress on Adsorptive Removal of Fluoride-Contaminated Water Using Biochar-Based Materials: Practical Feasibility through Reusability and Column Transport Studies. Environ. Res. 2022, 214, 114043. [Google Scholar] [CrossRef]
- Wang, J.; Chen, N.; Feng, C.; Li, M. Performance and Mechanism of Fluoride Adsorption from Groundwater by Lanthanum-Modified Pomelo Peel Biochar. Environ. Sci. Pollut. Res. 2018, 25, 15326–15335. [Google Scholar] [CrossRef]
- Zhou, J.; Liu, Y.; Han, Y.; Jing, F.; Chen, J. Bone-Derived Biochar and Magnetic Biochar for Effective Removal of Fluoride in Groundwater: Effects of Synthesis Method and Coexisting Chromium. Water Environ. Res. 2019, 91, 588–597. [Google Scholar] [CrossRef]
- Mandoreba, C.; Gwenzi, W.; Chaukura, N. Defluoridation of Drinking Water Using a Ceramic Filter Decorated with Iron Oxide-Biochar Composites. Int. J. Appl. Ceram. Technol. 2021, 18, 1321–1329. [Google Scholar] [CrossRef]


| Feedstock | Solution pH | pHPZC | Target Pollutant | Biochar Surface Charge | Adsorption Capacity (mg g−1) | References |
|---|---|---|---|---|---|---|
| Rice straw | 8 | 11 | F− | positive | 111.11 | [89] |
| Wheat straw | 7 | 4.8 | F− | negative | 51.28 | [90] |
| Rice husk | 4 | 6 | F− | positive | 4.45 | [178] |
| Rice husk | 7 | _ | F− | _ | 17.3 | [24] |
| Rice husk | 5.3 | F− | 21.59 | [150] | ||
| Rice husk | 6 | _ | F− | _ | 1.856 | [149] |
| Black gram straw | 2 | _ | F− | _ | 16 | [96] |
| Corn stover | 2 | 1.96 | F− | 4.11 | [91] | |
| Chir pine | 4.5 | _ | F− | _ | 16.72 | [151] |
| Mongolian scotch pine tree sawdust | 7 | _ | F− | _ | 0.885 | [35] |
| Pine bark | 2 | 9 | F− | positive | 9.77 | [179] |
| Pine wood | 2 | 9 | F− | positive | 7.66 | [179] |
| Douglas fir | 7 | 11 | F− | positive | 9.04 | [152] |
| Douglas fir | 6 | 6.4 | F− | positive | 36 | [153] |
| Reed biomass | 5.5 | 8.26 | F− | positive | 34.86 | [128] |
| Kashgar tamarisk | 6 | 6.6 | F− | positive | 164.23 | [40] |
| Tea oil plant (seed shells) | 6.8 | 4.45 | F− | negative | 11.04 | [93] |
| Sawdust | 7 | 2.2 | F− | negative | 4.413 | [154] |
| Pongammia pinnata seed cake | 7 | _ | F− | _ | 1.11 | [7] |
| Coconut | 6.5 | _ | F− | _ | _ | [92] |
| Cattail | _ | _ | F− | _ | 1.28 | [180] |
| Pomelo peel | 6.5 | 8.6 | F− | positive | 18.52 | [95] |
| Watermelon rind | 1 | 2.1 | F− | positive | 9.5 | [94] |
| Okra (lady finger) stem | 2 | _ | F− | _ | 20 | [96] |
| spent Mushroom compost | 10 | _ | F− | _ | 4.7 | [13] |
| Food waste | 7.1 | _ | F− | _ | 123.4 | [158] |
| Tea waste | 2 | _ | F− | _ | 52.5 | [109] |
| Red algae seaweed | 5 | 6.9 | F− | positive | 2.1 | [169] |
| Dairy manure | 5 | 8.8 | F− | positive | 0.42 | [109] |
| Sheep bone | _ | _ | F− | _ | 2.33 | [112] |
| Bone residues (chicken, cattle, and mixed bones) | _ | _ | F− | _ | 4.29 | [181] |
| Eggshell and platanus acerifoli leaves (5:1) | 5 | _ | F− | _ | 308 | [97] |
| Feedstock | Biochar Dose (g L−1) | Target Pollutant | Adsorption Capacity (mg g−1) | References |
|---|---|---|---|---|
| Rice straw | 5 mg/50 mL | U | 428.25 | [131] |
| Rice straw | 0.01g | U | 242.65 | [33] |
| Wheat straw | _ | U | 355.6 | [82] |
| Rice husk | 1 | U | 52.63 | [125] |
| Rice husk | 0.38 | U | 138.88 | [81] |
| Rice husk | 0.4 | U | 118 | [132] |
| Corn cob | 0.25 | U | 163.18 | [34] |
| Pine needles | 5 | U | 623.7 | [37] |
| Pine needles | 0.01 g/50 mL | U | 62.7 | [172] |
| Pine sawdust | 0.2 | U | 514.72 | [133] |
| Macaúba palm | 10 | U | 488.7 | [36] |
| Palm tree fibres | 0.1 g | U | 112 | [121] |
| Bamboo sawdust | 0.4 | U | 229.2 | [111] |
| Bamboo biomass | _ | U | 274.15 | [134] |
| Bamboo shoot shell | 2 | U | 32.3 | [105] |
| Cactus fibre | 0.01 | U | 214 | [118] |
| Camphor tree leaves | 0.25 | U | 98.29 | [173] |
| Miswak branches | 1 | U | 85.71 | [174] |
| Chinese banyan aerial root | 1 | U | 27.29 | [135] |
| Eucalyptus the Wood | 5 | U | 27.2 | [175] |
| Puncture vine | 0.5g | U | 17.24 | [39] |
| Water hyacinth | 0.2 | U | 138.57 | [176] |
| Hydrophyte biomass | 1 | U | 54.35 | [136] |
| Hydrophyte | 0.4 | U | 128.5 | [137] |
| Switchgrass | 0.1 | U | 4 | [177] |
| Pig manure | 0.3 | U | 979.3 | [107] |
| Pig manure | 0.3 | U | 661.7 | [107] |
| Pig manure | 0.1 | U | 952.5 | [138] |
| Pig manure | _ | U | 221.4 | [124] |
| Horse manure | 0.1 | U | 516.5 | [108] |
| Horse manure | 0.1 | U | 625.8 | [139] |
| Cow manure | _ | U | 73.3 | [82] |
| Carp fish scales | 0.1 | U | 291.98 | [122] |
| Sewage sludge | 0.25 | U | 96.73 | [34] |
| Sewage sludge | 0.13 | U | 490.2 | [140] |
| Winery waste (grape peels) | 1 | U | 255 | [141] |
| Winery waste (grape peels) | 1 | U | 100 | [141] |
| Malt spent rootlets | 0.01 | U | 547 | [120] |
| Coffee espresso residue | 0.01 | U | 547 | [120] |
| Olive kernels | 0.01 | U | 357 | [120] |
| Fungi | 0.05 | U | 427.9 | [142] |
| Green algae | 0.5 | U | 100.2 | [143] |
| Cyanobacteria | 0.5 | U | 58.05 | [144] |
| Sponge gourd | 5 mg/50 mL | U | 239.21 | [83] |
| Sponge gourd fibres | _ | U | 904 | [84] |
| Sponge gourd sponges | 0.01 g | U | 92 | [119] |
| Sponge gourd sponge | _ | U | 833 | [145] |
| Sponge gourd residue | 0.4 | U | 382 | [146] |
| Watermelon rind | 1 | U | 323.56 | [85] |
| Watermelon seeds | 1 | U | 27.61 | [38] |
| Longan shell (fruit) | 0.1 | U | 331.13 | [77] |
| Orange peel | 10 mg/50 mL | U | 246.3 | [106] |
| Rice straw | 1 | F− | 111.11 | [89] |
| Wheat straw | 1 | F− | 51.28 | [90] |
| Rice husk | 4 | F− | 4.45 | [178] |
| Rice husk | 1 | F− | 17.3 | [24] |
| Rice husk | 0.1 | F− | 21.59 | [150] |
| Rice husk | 10 | F− | 1.856 | [149] |
| Black gram straw | 2.5 | F− | 16 | [96] |
| Corn stover | 5 | F− | 4.11 | [91] |
| Chir pine | 2 | F− | 16.72 | [151] |
| Mongolian scotch pine tree sawdust | 3.6 g/100 mL | F− | 0.885 | [35] |
| Pine bark | 10 | F− | 9.77 | [179] |
| Pine wood | 10 | F− | 7.66 | [179] |
| Douglas fir (pine) | 0.05 g/25 mL | F− | 9.04 | [152] |
| Douglas fir (pine) | 25 mg | F− | 36 | [153] |
| Reed biomass | 1 | F− | 34.86 | [128] |
| Kashgar tamarisk | 5 | F− | 164.23 | [40] |
| Tea oil plant (seed shells) | 1.6 | F− | 11.04 | [93] |
| Sawdust | 5 | F− | 4.413 | [154] |
| Coconut | 7 | F− | _ | [92] |
| Cattail | _ | F− | 1.28 | [180] |
| Pongammia pinnata seed cake | 10 | F− | 1.11 | [7] |
| Pomelo peel | 2.5 | F− | 18.52 | [95] |
| Watermelon rind | 0.2 g | F− | 9.5 | [94] |
| Okra (lady finger) stem | 2.5 | F− | 20 | [96] |
| Spent mushroom compost | 2 | F− | 4.7 | [13] |
| Food waste | 0.1 g/30 mL | F− | 123.4 | [156] |
| Tea waste | 10 | F− | 52.5 | [157] |
| Red algae seaweed | 0.6 g/100 mL | F− | 2.1 | [158] |
| Dairy manure | 0.33 | F− | 0.51 | [109] |
| Sheep bone | 1 | F− | 2.33 | [112] |
| Bone residues (chicken, cattle, and mixed bones) | 1 | F− | 4.29 | [181] |
| Eggshell and platanus acerifoli leaves (5:1) | 1.6 | F− | 308 | [97] |
| Feedstock | Initial Conc. (mg L−1) | Target Pollutant | Adsorption Capacity (mg g−1) | References |
|---|---|---|---|---|
| Rice straw | 6 | F− | 111.11 | [89] |
| Wheat straw | 6 | F− | 51.28 | [90] |
| Rice husk | 5 | F− | 4.45 | [178] |
| Rice husk | 5 | F− | 17.3 | [24] |
| Rice husk | 2 | F− | 21.59 | [150] |
| Rice husk | 4 | F− | 1.856 | [149] |
| Black gram straw | 10 | F− | 16 | [96] |
| Corn stover | 100 | F− | 4.11 | [91] |
| Chir pine | 50 | F− | 16.72 | [151] |
| Mongolian scotch pine tree sawdust | 20 | F− | 0.885 | [35] |
| Pine bark | 100 | F− | 9.77 | [179] |
| Pine wood | 100 | F− | 7.66 | [179] |
| Douglas fir (pine) | 10 | F− | 9.04 | [152] |
| Douglas fir (pine) | 50 | F− | 36 | [153] |
| Reed biomass | 10 | F− | 34.86 | [128] |
| Kashgar tamarisk | 40 | F− | 164.23 | [40] |
| Tea oil plant (seed shells) | 70 | F− | 11.04 | [93] |
| Sawdust | 10 | F− | 4.413 | [154] |
| Coconut | 10 | F− | _ | [92] |
| Cattail | 20 | F− | 1.28 | [180] |
| Pongammia pinnata seed cake | 10 | F− | 1.11 | [7] |
| Pomelo peel | 10 | F− | 18.52 | [132] |
| Watermelon rind | 50 | F− | 9.5 | [94] |
| Okra (lady finger) stem | 10 | F− | 20 | [96] |
| Spent mushroom compost | 10 | F− | 4.7 | [13] |
| Food waste | 300 | F− | 123.4 | [156] |
| Tea waste | 50 | F− | 52.5 | [157] |
| Red algae seaweed | 15 | F− | 2.1 | [158] |
| Dairy manure | 5 | F− | 0.42 | [109] |
| Sheep bone | 10 | F− | 2.33 | [112] |
| Bone residues (chicken, cattle and mixed bones) | 10 | F− | 4.29 | [181] |
| Eggshell and platanus acerifoli leaves (5:1) | 500 | F− | 308 | [97] |
| Rice straw | 50 | U | 428.25 | [131] |
| Rice straw | 50 | U | 242.65 | [33] |
| Wheat straw | 10 | U | 355.6 | [82] |
| Rice husk | 10 | U | 52.63 | [125] |
| Rice husk | 3 | U | 138.88 | [81] |
| Rice husk | 80 | U | 118 | [132] |
| Corn cob | 25 | U | 163.18 | [34] |
| Pine needles | 11.9 | U | 623.7 | [37] |
| Pine needles | 50 | U | 62.7 | [172] |
| Pine sawdust | 10 | U | 514.72 | [133] |
| Macaúba palm | 5 | U | 488.7 | [36] |
| Palm tree fibres | 11.9 | U | 112 | [121] |
| Bamboo sawdust | 47.6 | U | 229.2 | [111] |
| Bamboo biomass | _ | U | 274.15 | [134] |
| Bamboo shoot shell | 50 | U | 32.3 | [105] |
| Cactus fibre | 119 | U | 214 | [118] |
| Camphor tree leaves | 50 | U | 98.29 | [173] |
| Miswak branches | 60 | U | 85.71 | [174] |
| Chinese banyan aerial root | 30 | U | 27.29 | [135] |
| Eucalyptus the Wood | 300 | U | 27.2 | [175] |
| Puncture vine | 50 | U | 17.24 | [39] |
| Water hyacinth | 30 | U | 138.57 | [176] |
| Hydrophyte biomass | _ | U | 54.35 | [136] |
| Hydrophyte | 47.6 | U | 128.5 | [137] |
| Switchgrass | 10 | U | 4 | [177] |
| Pig manure | 10 | U | 979.3 | [107] |
| Pig manure | 10 | U | 661.7 | [107] |
| Pig manure | 10 | U | 952.5 | [138] |
| Pig manure | 10 | U | 221.4 | [124] |
| Horse manure | 10 | U | 516.5 | [108] |
| Horse manure | 10 | U | 625.8 | [139] |
| Cow manure | 10 | U | 73.3 | [82] |
| Carp fish scales | 40 | U | 291.98 | [122] |
| Sewage sludge | 25 | U | 96.73 | [34] |
| Sewage sludge | 50 | U | 490.2 | [140] |
| Winery waste (grape peels) | 100 | U | 255 | [141] |
| Winery waste (grape peels) | 100 | U | 100 | [141] |
| Malt spent rootlets | _ | U | 547 | [120] |
| Coffee espresso residue | _ | U | 547 | [120] |
| Olive kernels | _ | U | 357 | [120] |
| Fungi | 10 | U | 427.9 | [142] |
| Green algae | 50 | U | 100.2 | [143] |
| Cyanobacteria | 50 | U | 58.05 | [144] |
| Sponge gourd | 5 | U | 239.21 | [83] |
| Sponge gourd fibres | U | 904 | [84] | |
| Sponge gourd sponges | 119 | U | 92 | [119] |
| Sponge gourd sponge | _ | U | 833 | [145] |
| Sponge gourd residue | 225 | U | 382 | [146] |
| Watermelon rind | 20 | U | 323.56 | [85] |
| Watermelon seeds | 30 | U | 27.61 | [38] |
| Longan shell (fruit) | 23.6 | U | 331.13 | [77] |
| Orange peel | 50 | U | 246.3 | [106] |
| Feedstock | Carbonization Temp. (°C) | Surface Area (m2 g−1) | Target Pollutant | Adsorption Capacity (mg g−1) | References |
|---|---|---|---|---|---|
| Rice straw | 500 | 157.96 | U | 428.25 | [131] |
| Rice straw | 500 | _ | U | 242.65 | [33] |
| Wheat straw | 450 | 290.1 | U | 355.6 | [82] |
| Rice husk | 500 | 109.65 | U | 52.63 | [125] |
| Rice husk | 300 | 62.88 | U | 138.88 | [81] |
| Rice husk | 500 | 109 | U | 118 | [132] |
| Corn cob | 800 | _ | U | 163.18 | [34] |
| Pine needles | 600 | _ | U | 623.7 | [37] |
| Pine needles | 180 | _ | U | 62.7 | [172] |
| Pine sawdust | 500 | 51.45 | U | 514.72 | [133] |
| Macaúba palm | 350 | 643.12 | U | 488.7 | [36] |
| palm tree fibres | 650 | _ | U | 112 | [121] |
| Bamboo sawdust | 450 | 1298 | U | 229.2 | [111] |
| Bamboo biomass | 700 | 445.17 | U | 274.15 | [134] |
| Bamboo shoot shell | 500 | 10.93 | U | 32.3 | [105] |
| Cactus fibre | 600 | <5 | U | 214 | [118] |
| Camphor tree leaves | 350 | 65.91 | U | 98.29 | [173] |
| Miswak branches | 400 | 9.05 | U | 85.71 | [174] |
| Chinese banyan aerial root | 600 | 284 | U | 27.29 | [135] |
| Eucalyptus wood | 400 | 20 | U | 27.2 | [175] |
| Puncture vine | 500 | _ | U | 17.24 | [39] |
| Water hyacinth | 400 | 50.545 | U | 138.57 | [176] |
| Hydrophyte biomass | 700 | 92.43 | U | 54.35 | [136] |
| Hydrophyte | 500 | 433 | U | 128.5 | [137] |
| Switchgrass | 300 | 2.9 | U | 4 | [177] |
| Pig manure | 500 | _ | U | 979.3 | [107] |
| Pig manure | 500 | _ | U | 661.7 | [107] |
| Pig manure | 500 | 227.9 | U | 952.5 | [138] |
| Pig manure | 250 | _ | U | 221.4 | [124] |
| Horse manure | 500 | _ | U | 516.5 | [108] |
| Horse manure | 500 | _ | U | 625.8 | [139] |
| Cow manure | 450 | 101.5 | U | 73.3 | [82] |
| Carp fish scales | 330 | 1074.73 | U | 291.98 | [122] |
| Sewage sludge | 500 | _ | U | 96.73 | [34] |
| Sewage sludge | 600 | 623.09 | U | 490.2 | [140] |
| Winery waste (grape peels) | 650 | _ | U | 255 | [141] |
| Winery waste (grape peels) | 650 | 165 | U | 100 | [141] |
| Malt spent rootlets (MSR) | 850 | 540 | U | 547 | [120] |
| Coffee espresso residue | 850 | 700 | U | 547 | [120] |
| Olive kernels | 850 | 510 | U | 357 | [120] |
| Fungi | 160 | 102.7 | U | 427.9 | [142] |
| Green algae | 180 | 63.7 | U | 100.2 | [143] |
| Cyanobacteria | 200 | _ | U | 58.05 | [144] |
| Sponge gourd | 400 | _ | U | 239.21 | [83] |
| Sponge gourd fibres | 650 | <5 | U | 904 | [84] |
| Sponge gourd sponges | 650 | _ | U | 92 | [119] |
| Sponge gourd sponge | 650 | _ | U | 833 | [145] |
| Sponge gourd residue | 200 | _ | U | 382 | [146] |
| Watermelon rind | 500 | 86.35 | U | 323.56 | [85] |
| Watermelon seeds | 350 | _ | U | 27.61 | [38] |
| Longan shell (fruit) | 800 | 1168.88 | U | 331.13 | [77] |
| Orange peel | 650 | 273.25 | U | 246.3 | [106] |
| rice straw | 500 | 95.36 | F− | 111.11 | [89] |
| Wheat straw | _ | _ | F− | 51.28 | [90] |
| Rice husk | 700 | 58.98 | F− | 4.45 | [178] |
| Rice husk | 600 | _ | F− | 17.3 | [24] |
| Rice husk | 600 | 114 | F− | 21.59 | [150] |
| Rice husk | 500 | 2.45 | F− | 1.856 | [149] |
| Black gram straw | 500 | 9.27 | F− | 16 | [96] |
| Corn stover | 500 | 3.61 | F− | 4.11 | [91] |
| Chir pine | 400 | _ | F− | 16.72 | [151] |
| Mongolian scotch pine tree sawdust | 550 | 339 | F− | 0.885 | [35] |
| Pine bark | 450 | 1.88 | F− | 9.77 | [179] |
| Pine wood | 450 | 2.73 | F− | 7.66 | [179] |
| Douglas fir (pine) | 1000 | 494 | F− | 9.04 | [152] |
| Douglas fir (pine) | 1000 | 576 | F− | 36 | [153] |
| Reed biomass | 600 | 236.84 | F− | 34.86 | [128] |
| Kashgar tamarisk | 350 | 164.52 | F− | 164.23 | [40] |
| Tea oil plant (seed shells) | 400 | _ | F− | 11.04 | [93] |
| Sawdust | 660 | 57.97 | F− | 4.413 | [154] |
| Pongammia pinnata seed cake | 550 | 10.1 | F− | 1.11 | [7] |
| Coconut | 700 | 1054 | F− | _ | [92] |
| Cattail | 800 | 733.62 | F− | 1.28 | [180] |
| Pomelo peel | 600 | _ | F− | 18.52 | [132] |
| Watermelon rind | 400 | 0.5365 | F− | 9.5 | [94] |
| Okra (lady finger) stem | 600 | 23.52 | F− | 20 | [96] |
| Spent mushroom compost | 500 | 28.5 | F− | 4.7 | [13] |
| Food waste | 600 | 20.95 | F− | 123.4 | [156] |
| Tea waste | 400 | 11.833 | F− | 52.5 | [157] |
| Red algae seaweed | 450 | 319.47 | F− | 2.1 | [158] |
| Dairy manure | 500 | 2.6 | F− | 0.42 | [109] |
| Sheep bone | 650 | 113.874 | F− | 2.33 | [112] |
| Bone residues (chicken, cattle, and mixed bones) | 350 | _ | F− | 4.29 | [181] |
| Bone residues (chicken, cattle, and mixed bones) | 700 | _ | F− | 2.91 | [181] |
| Eggshell and platanus acerifoli leaves (5:1) | 800 | 44.7 | F− | 308 | [97] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Thakur, A.; Kumar, R.; Sahoo, P.K. Uranium and Fluoride Removal from Aqueous Solution Using Biochar: A Critical Review for Understanding the Role of Feedstock Types, Mechanisms, and Modification Methods. Water 2022, 14, 4063. https://doi.org/10.3390/w14244063
Thakur A, Kumar R, Sahoo PK. Uranium and Fluoride Removal from Aqueous Solution Using Biochar: A Critical Review for Understanding the Role of Feedstock Types, Mechanisms, and Modification Methods. Water. 2022; 14(24):4063. https://doi.org/10.3390/w14244063
Chicago/Turabian StyleThakur, Anjali, Rakesh Kumar, and Prafulla Kumar Sahoo. 2022. "Uranium and Fluoride Removal from Aqueous Solution Using Biochar: A Critical Review for Understanding the Role of Feedstock Types, Mechanisms, and Modification Methods" Water 14, no. 24: 4063. https://doi.org/10.3390/w14244063







