Development of Lake from Acidification to Eutrophication in the Arctic Region under Reduced Acid Deposition and Climate Warming
Abstract
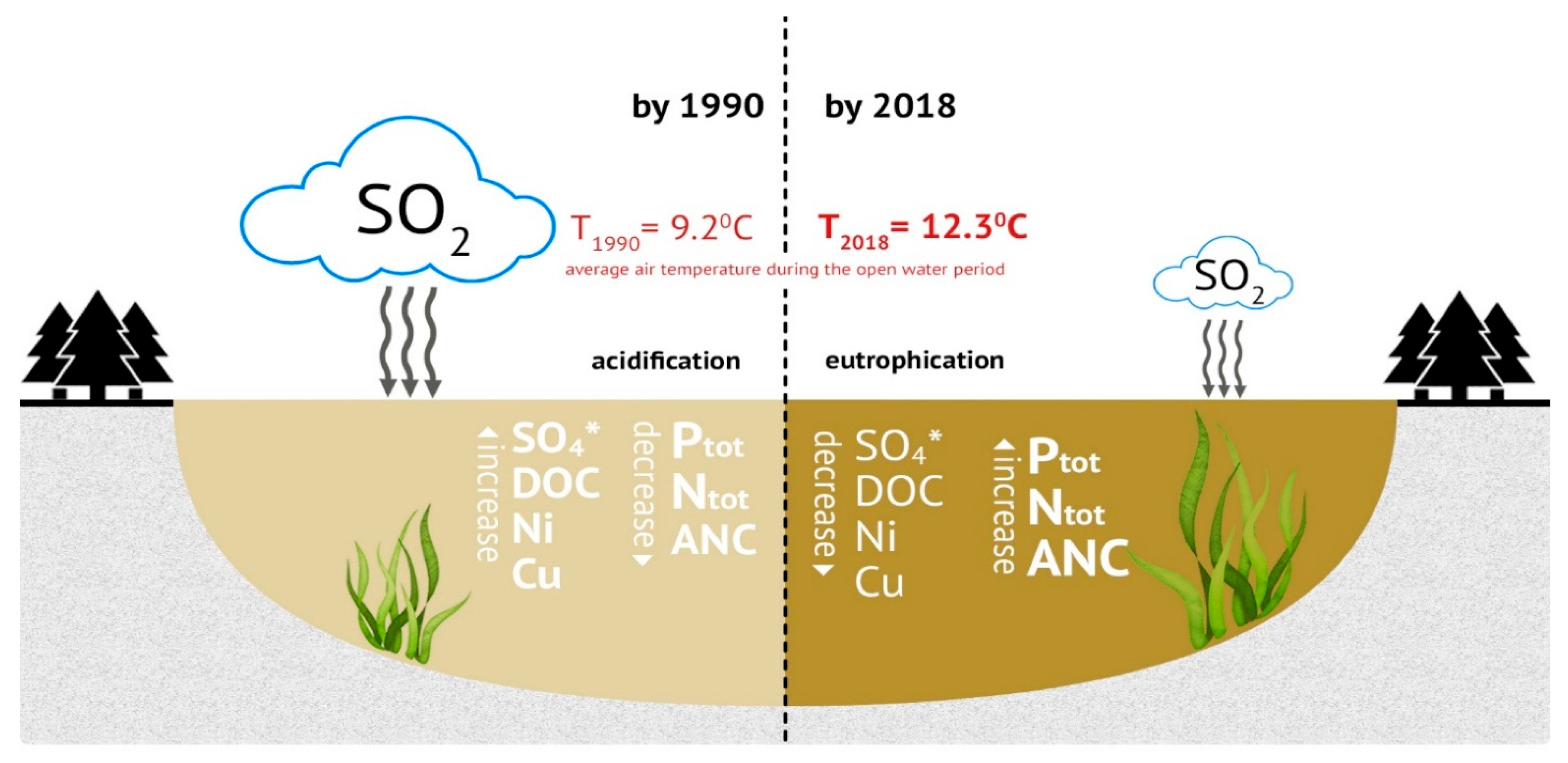
:1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Water Chemistry: Sampling and Analysis
2.3. Statistical Analysis
3. Results
3.1. Main Parameters of Water Chemistry
3.2. Regional Variations in Water Chemistry
3.3. Climatic Changes
4. Discussion
4.1. Response of Anthropogenic Sulfates, ANC, and Basic Cations
4.2. DOC and Nutrients
4.3. Effect of the Climate on the Evolution of the Lakes
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Brakke, D.F.; Landers, D.H. Chemical and Physical Characteristics of Lakes in the Northeastern United States. Environ. Sci. Technol. 1988, 222, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Evans, C.D.; Cullen, J.M.; Alewell, C.; Kopacek, J.; Marchetto, A.; Moldan, F.; Prechtel, A.; Rogora, M. Recovery from acidification in European surface waters. Hydrol. Earth Syst. Sci. 2001, 5, 283–298. [Google Scholar] [CrossRef]
- Galloway, J.N. Acid deposition: Perspectives in time and space. Water Air Soil Pollut. 1995, 85, 15–24. [Google Scholar] [CrossRef]
- Henriksen, A.; Kämäri, I.; Posh, M.; Wilander, A. Critical loads of acidity: Nordic surface waters. AMBIO 1992, 21, 356–363. [Google Scholar]
- Jeffries, D.S. Canadian Acid Rain Assessment, Aquatic Effects; Authority of the Minister of Environment: Burlington, ON, Canada, 1997; p. 270. [Google Scholar]
- Kuylenstierna, J.C.I.; Rodhe, M.; Cinderby, S.; Hicks, K. Acidification in developing countries: Ecosystem sensitivity and the critical load approach on a global scale. AMBIO 2001, 30, 20–28. [Google Scholar] [CrossRef] [PubMed]
- Reuss, J.O.; Cosby, B.J.; Wright, R.F. Chemical processes governing soil and water acidification. Nature 1987, 329, 27–32. [Google Scholar] [CrossRef]
- Stoddard, J.L.; Jeffries, D.S.; Lukewille, A.; Clair, T.A.; Dillon, P.J.; Driscoll, C.T.; Forsius, M. Regional trends in aquatic recovery from acidification in North America and Europe. Nature 1999, 401, 575–578. [Google Scholar] [CrossRef]
- Kvaeven, B.; Ulstein, M.J.; Skjelkvåle, B.L. ICP Waters—An international program for surface water monitoring. Water Air Soil Pollut. 2001, 130, 775–780. [Google Scholar] [CrossRef]
- Clair, T.A.; Dennis, I.F.; Vet, R. Water chemistry and dissolved organic carbon trends in lakes from Canada's Atlantic Provinces: No recovery from acidification measured after 25 years of lake monitoring. J. Fish. Aquat. Sci. 2011, 68, 663–674. [Google Scholar] [CrossRef]
- Monteith, D.T.; Stoddard, J.L.; Evans, C.D.; de Wit, H.A.; Forsius, M.; Høgåsen, T.; Wilander, A.; Skjelkvåle, B.L.; Jeffries, D.S.; Vuorenmaa, J.; et al. Dissolved organic carbon trends resulting from changes in atmospheric deposition chemistry. Nature 2007, 450, 537–539. [Google Scholar] [CrossRef] [PubMed]
- Skjelkvåle, B.L.; Stoddard, J.L.; Andersen, T. Trends in surface water acidification in Europe and North America (1989–1998). Water Air Soil Pollut. 2001, 130, 787–792. [Google Scholar] [CrossRef]
- Skjelkvåle, B.L.; Stoddard, J.L.; Jeffries, D.S.; Torseth, K.; Hogasen, T.J.; Bowmane, J.; Manniof, J.; Monteithg, D.T.; Moselloh, R.; Rogorah, M.; et al. Regional scale evidence for improvements in surface water chemistry 1990–2001. Environ. Pollut. 2005, 137, 165–176. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stoddard, J.L.; Van, Sickle, J.; Herlihy, A.T.; Brahney, J.; Paulsen, S.; Peck, D.V.; Mitchell, R.; Pollard, A.I. Continental-scale increase in lake and stream phosphorus: Are oligotrophic systems disappearing in the United States? Environ. Sci. Technol. 2016, 50, 3409–3415. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Strock, K.E.; Nelson, S.J.; Kahl, J.S.; Saros, J.E.; McDowell, W.H. Decadal trends reveal recent acceleration in the rate of recovery from acidification in the northeastern U.S. Environ. Sci. Technol. 2014, 48, 4681–4689. [Google Scholar] [CrossRef] [PubMed]
- Strock, K.E.; Theodore, N.; Gawley, W.G.; Ellsworth, A.C.; Saros, J.E. Increasing dissolved organic carbon concentrations in northern boreal lakes: Implications for lake water transparency and thermal structure. J. Geophys. Res. Biogeosci. 2017, 122, 1022–1035. [Google Scholar] [CrossRef]
- Driscoll, C.T.; Driscoll, K.M.; Roy, K.M.; Mitchell, M.J. Chemical response of lakes in the Adirondack Region of New York to declines in acidic deposition. Environ. Sci. Technol. 2003, 37, 2036–2042. [Google Scholar] [CrossRef] [Green Version]
- AR5 Synthesis Report: Climate Change. 2014. Available online: https://www.ipcc.ch/report/ar5/syr/ (accessed on 3 October 2022).
- Clark, J.M.; Bottrell, S.H.; Evans, C.D.; Monteith, D.T.; Bartlett, R.; Rose, R.; Newton, R.J.; Chapman, P.J. The importance of the relationship between scale and process in understanding long-term DOC dynamics. Sci. Total Environ. 2010, 408, 2768. [Google Scholar] [CrossRef]
- Gavin, A.L.; Nelson, S.J.; Klemmer, A.J.; Fernandez, I.J.; Strock, K.E.; McDowell, W.H. Acidification and climate linkages to increased dissolved organic carbon in high elevation lakes. Water Resour. Res. 2018, 54, 5187–5877. [Google Scholar]
- Watmough, S.A.; Eimers, C.; Baker, S. Impediments to recovery from acid deposition. Atmos. Environ. 2016, 146, 15–27. [Google Scholar]
- Houle, D.; Couture, S.; Gagnon, C. Relative role of decreasing precipitation sulfate and climate on recent lake recovery. Global Biogeochem. Cycles 2010, 24, 4029. [Google Scholar] [CrossRef]
- Corman, J.R.; Bertolet, B.L.; Casson, N.J.; Sebestyen, S.D.; Kolka, R.K.; Stanley, E.H. Nitrogen and phosphorus loads to temperate seepage lakes associated with allochthonous dissolved organic carbon loads. Geophys. Res. Let. 2018, 45, 5481–5490. [Google Scholar] [CrossRef]
- Komov, V.T.; Lazareva, V.I.; Stepanova, I.K. Anthropogenic pollution of small lakes of the Northern European Part of Russia. Biol. Vnutr. Vod. 1997, 3, 5–17. [Google Scholar]
- Moiseenko, T.I. Acidification and Critical Loads in Surface Waters: Kola, Northern Russia. AMBIO 1994, 23, 418–424. [Google Scholar]
- Moiseenko, T.I.; Dinu, M.I.; Gashkina, N.A.; Jones, V.; Khoroshavin, V.Y.; Kremleva, T.A. Present status of water chemistry and acidification under nonpoint sources of pollution across European Russia and West Siberia. Environ. Res. Lett. 2018, 13, 105007. [Google Scholar] [CrossRef] [Green Version]
- Moiseenko, T.I.; Gashkina, N.A.; Dinu, M.I.; Kremleva, T.A.; Khoroshavin, V.Y. Water Chemistry of Arctic Lakes under Airborne Contamination of Watersheds. Water 2020, 12, 1659. [Google Scholar] [CrossRef]
- Moiseenko, T.I.; Dinu, M.I.; Bazova, M.M.; de Wit, H.A. Long-Term Changes in the Water Chemistry of Arctic Lakes as a Response to Reduction of Air Pollution: Case Study in the Kola, Russia. Water Air Soil Pollut. 2015, 226, 98. [Google Scholar] [CrossRef]
- Intergovernmental Panel for Global Climate Change (IPCC). The Physical Science Basis. Climate Change. 2013. Available online: http://www.ipcc.ch/report/ar5/wg1 (accessed on 30 September 2022).
- Noori, R.; Woolway, R.I.; Saari, M.; Pulkkanen, M.; Klove, B. Six decades of thermal change in a pristine lake situated north of the Arctic Circle. Water Resour. Res. 2022, 58, 2021WR031543. [Google Scholar] [CrossRef]
- Noori, R.; Bateni, S.M.; Saari, M.; Almazroui, M. Torabi Haghighi, A. Strong warming rates in the surface and bottom layers of a boreal lake: Results from approximately six decades of measurements (1964–2020). Earth Space Sci. 2022, 9, e2021EA001973. [Google Scholar] [CrossRef]
- Durov, A.G. Atlas of the Murmansk Region; The Head Office of Geodesy and Cartography: Moscow, Russian, 1971; p. 33. (In Russian) [Google Scholar]
- Pogilenko, V.I.; Gavrilenko, B.V.; Girov, D.V.; Gabin, S.V. The Geology of Ore Districts of the Murmansk Region Apatity Kola Scientific Center; Academy of Sciences: Russian, Moscow, 2002; p. 359. [Google Scholar]
- Reports on the State and Environmental Protection of the Murmansk Region in 1990–2018. 2019. Available online: https://gov-murman.ru/region/environmentstate/ (accessed on 3 October 2022).
- State Reports 2019. On State and Protection of the Environment of the Russian Federation (2003–2011). Available online: https://www.mnr.gov.ru/docs/gosudarstvennye_doklady (accessed on 30 September 2022).
- Henriksen, A.; Skjelvåle, B.L.; Mannio, J.; Wilander, A.; Harriman, R.; Curtis, C.; Jensen, J.P.; Fjeld, E.; Moiseenko, T. Northern European Lake Survey, Finland, Norway, Sweden, Denmark, Russian Kola, Russian Karelia, Scotland and Wales. AMBIO 1998, 27, 80–91. [Google Scholar]
- ICP-waters. Waters Programme Manual. Report105/2010, International Cooperative Programme on Assessment and Monitoring Effects of Air Pollution on Rivers and Lakes. 2010. Available online: https://niva.brage.unit.no/niva-xmlui/handle/11250/215220?locale-ttribute=en (accessed on 30 September 2022).
- Eaton, A.; Arnold, E.; Archie, A.E.; Rice, E.W.; Clesceri, L.S. Standard Methods for the Examination of Water and Wastewater, 17th ed.; American Public Health Association (APHA): Washington, DC, USA, 1992. [Google Scholar]
- Intercomparison 2034: pH, Cond, HCO3 NO3+NO2, C1, SO4, Ca, Mg, Na, K, Fe, Mn, Cd, Pb, Cu, Ni, and Zn. In International Cooperative Programme on Assessment and Monitoring of Acidification of Rivers and Lakes; Reports of Norwegian Institute of Water Research: Oslo, Norway, 2020.
- Weather Archive: Murmansk Region. Knowledge Management Portal “Pogoda i Klimat”. 2019. Available online: http://www.pogodaiklimat.ru/archive.php?id=ru®ion=51 (accessed on 30 September 2022).
- Driscoll, C.T.; Driscoll, K.M.; Fakhraei, H.; Civerolo, K. Long-term temporal trends and spatial patterns in the acid-base chemistry of lakes in the Adirondack region of New York in response to decreases in acidic deposition. Atmos. Environ. 2016, 146, 5–14. [Google Scholar] [CrossRef] [Green Version]
- Kline, K.M.; Eshleman, K.N.; Garlitz, J.E.; U'Ren, S.H. Long-term response of surface water acid neutralizing capacity in a central Appalachian (USA) river basin to declining acid deposition. Atmos. Environ. 2016, 146, 195–205. [Google Scholar] [CrossRef] [Green Version]
- Galloway, J.N.; Norton, S.A.; Church, M.R. Freshwater acidification from atmospheric deposition of sulfuric acid: A conceptual model. Environ. Sci. Technol. 1983, 17, 541A–545A. [Google Scholar] [CrossRef] [PubMed]
- Rogora, M.; Colombo, L.; Marchetto, A.; Mosello, R.; Steingruber, S. Temporal and spatial patterns in the chemistry of wet deposition in Southern Alps. Atmosph. Environ. 2016, 146, 44–54. [Google Scholar] [CrossRef] [Green Version]
- San Clements, M.D.; Fernandez, I.J.; Lee, R.H.; Roberti, J.A.; Adams, M.B.; Rue, G.A.; McKnight, D.M. Long-Term Experimental Acidification Drives Watershed Scale Shift in Dissolved Organic Matter Composition and Flux. Environ Sci. Technol. 2018, 52, 2649–2657. [Google Scholar] [CrossRef] [PubMed]
- De Wit, H.A.; Stoddard, J.L.; Monteith, D.T.; Sample, J.E.; Austnes, K.; Couture, S.; Fölster, J.; Higgins, S.N.; Houle, D.; Hruška, J.; et al. Cleaner air reveals growing influence of climate on dissolved organic carbon trends in northern headwaters. Environ. Res. Lett. 2021, 16, 104009. [Google Scholar] [CrossRef]
- Evans, C.D.; Monteith, D.T.; Reynolds, B.; Clark, J.M. Buffering of recovery from acidification by organic acids. Sci. Total Environ. 2008, 404, 316–325. [Google Scholar] [CrossRef]
- Fakhraei, H.; Driscoll, C.T. Proton and Aluminum Binding Properties of Organic Acids in Surface Waters of the Northeastern U.S. Environ. Sci. Technol. 2015, 49, 2939–2947. [Google Scholar] [CrossRef]
- de Wit, H.A.; Valinia, S.; Weyhenmeyer, G.A.; Futter, M.N.; Kortelainen, P.; Austnes, K.; Hessen, D.O.; Räike, A.; Laudon, H.; Vuorenmaa, J. Current browning of surface waters will be further promoted by wetter climate. Environ. Sci. Technol. Lett. 2016, 3, 430–435. [Google Scholar] [CrossRef] [Green Version]
- Meingast, K.M.; Kane, E.; Coble, A.A.; Marcarelli, A.M.; Toczydlowski, D. Climate, snowmelt dynamics and atmospheric deposition interact to control dissolved organic carbon export from a northern forest stream over 26 years. Environ. Res. Lett. 2020, 15, 104034. [Google Scholar] [CrossRef]
- Garmo, O.G.; Skjelkvåle, B.L.; de Wit, H.A.; Colombo, L.; Curtis, C.; Folster, J.; Hoffmann, A. Trends in surface water chemistry in acidified areas in Europe and North America from 1990 to 2008. Water Air Soil Pollut. 2014, 225, 1880. [Google Scholar] [CrossRef] [Green Version]
- Campbell, J.L.; Rustad, L.E.; Boyer, E.W.; Christopher, S.F.; Driscoll, C.T.; Fernandez, I.J.; Groffman, P.M.; Houle, D.; Kiekbusch, J.; Magill, A.H.; et al. Consequences of climate change for biogeochemical cycling in forests of northeastern North America. Can. J. For. Res. 2009, 39, 264–284. [Google Scholar] [CrossRef]
- Melillo, J.M.; Richmond, T.C.; Yohe, G.W. Climate Change Impacts in the United States: The Third National Climate Assessment; U.S. Global Change Research Program; U.S. Government Printing Office: Washington, DC, USA, 2014; p. 841. [Google Scholar]
- Grouza, G.V.; Rankova, E.Y. The Observed and Expected Climate Change of RUSSIA: Air Temperature; Academy of Sciences, Obninsk Institute of Global Climate and Ecology: Russian, Moscow, 2012; p. 194. [Google Scholar]
- Sommaruga-Wӧgrath, S.; Koinig, K.A.; Schmidt, R.; Sommaruga, R.; Tessadri, R.; Psenner, R. Temperature effects on the acidity of remote alpine lakes. Nature 1997, 387, 64–67. [Google Scholar] [CrossRef]
- Moiseenko, T.; Sharov, A. Large Russian lakes Ladoga, Onega, and Imandra under strong pollution and in the period of revitalization: A review. Geosciences 2019, 9, 492. [Google Scholar] [CrossRef]
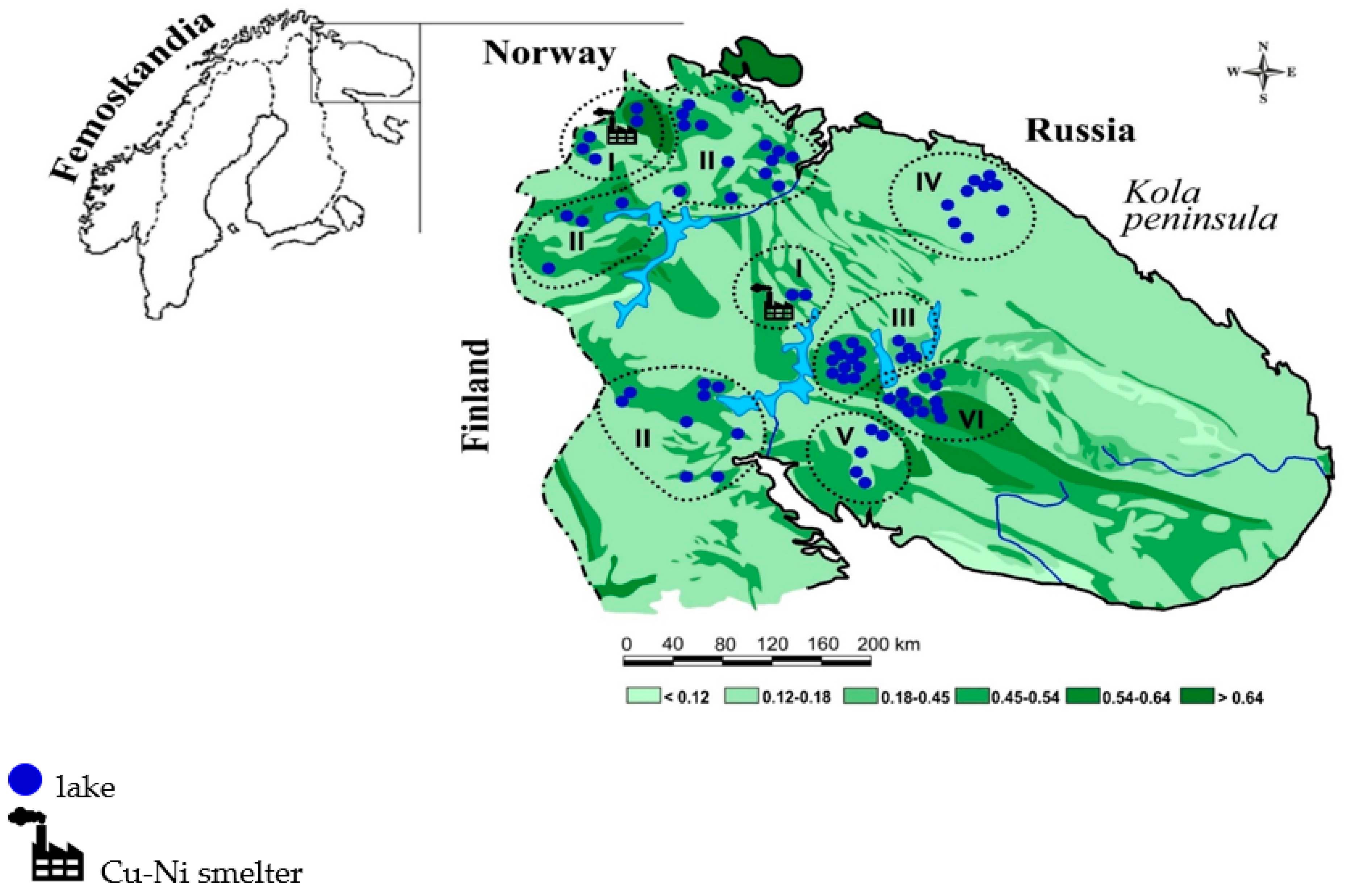
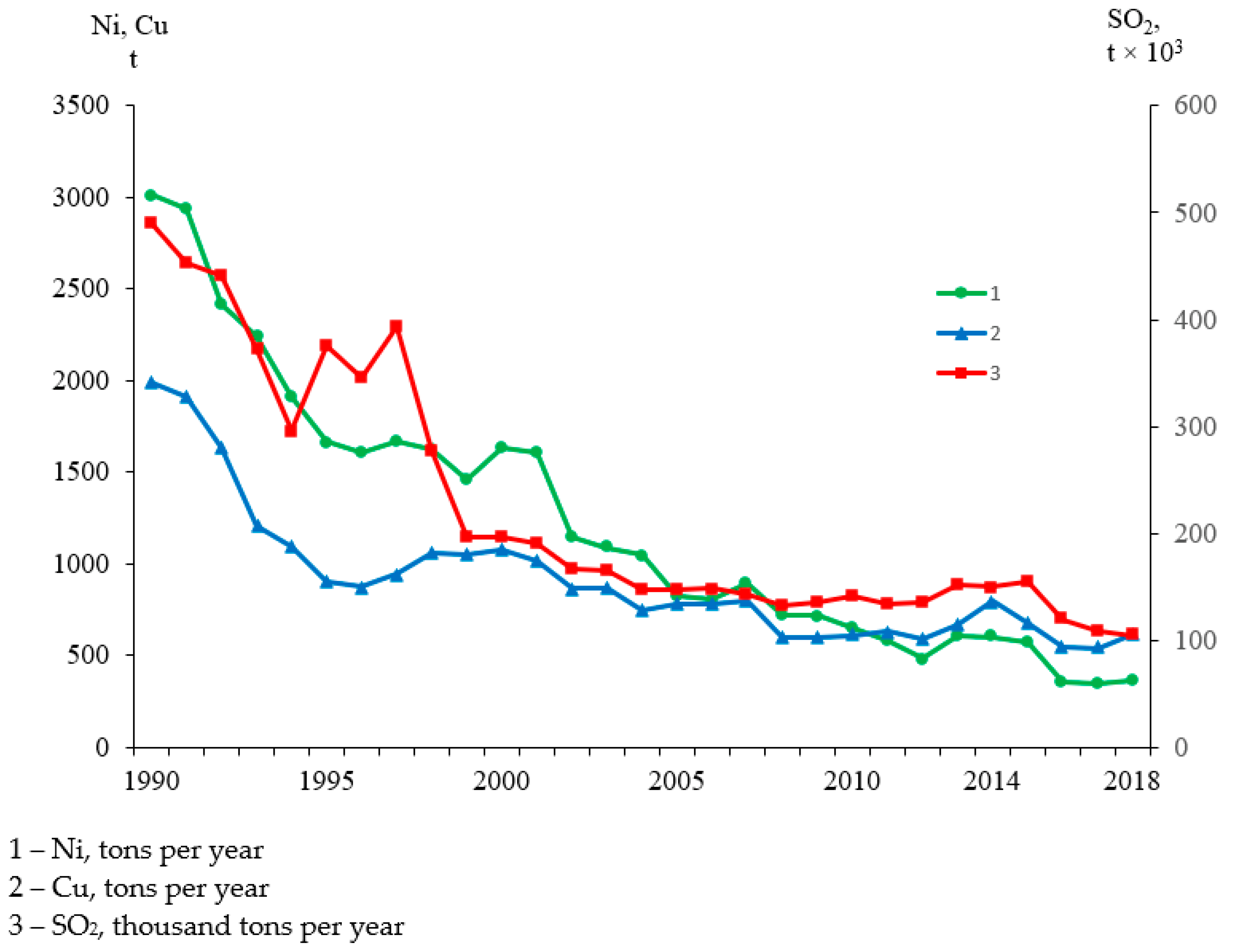

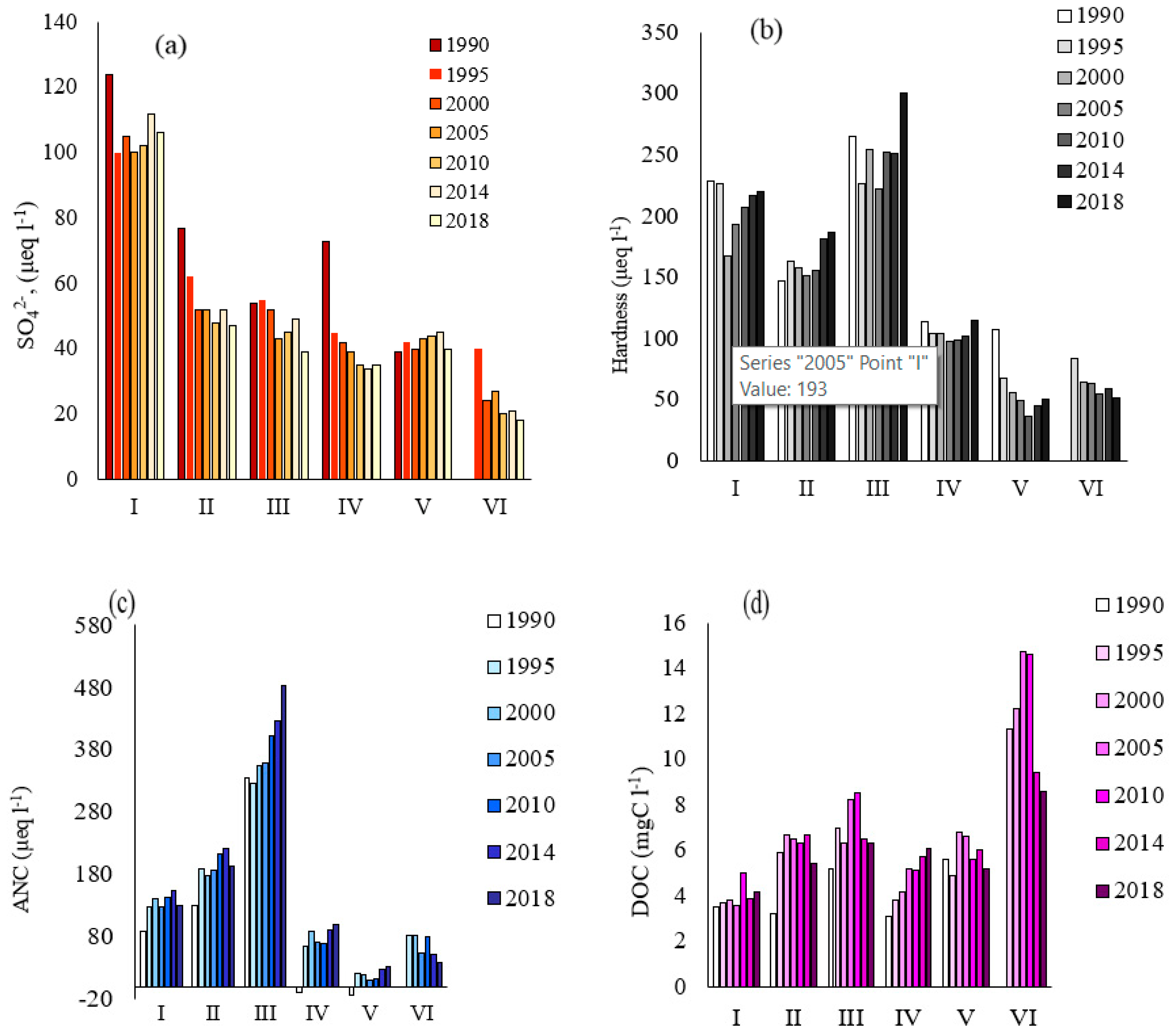

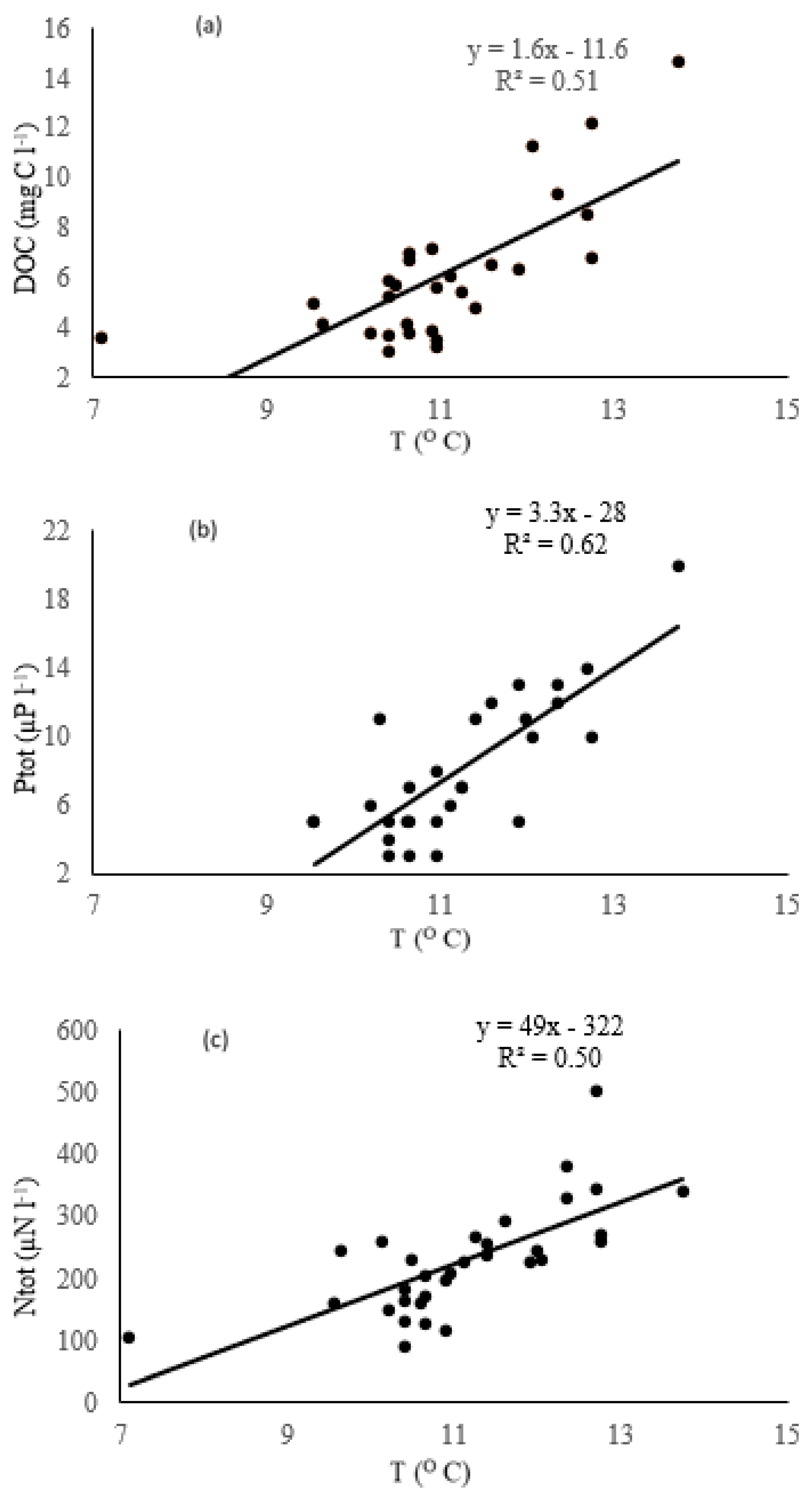
| Subregion | Major Rock Type (%) | Forest (%) | Peat (%) | Distance |
|---|---|---|---|---|
| from the Smelters, km | ||||
| I | basaltic (70), gneiss (10), | 12 | 13 | <40 km |
| amphibolites (10), slantsy (10) | 2–58 | 5–17 | ||
| II | basaltic and gabbros (90), | 63 | 15 | 40–120 km |
| gneiss (10) | 38–79 | 6–30 | ||
| III | nepheline syenite (80), | 57 | 18 | 40–80 km |
| foyaites and urtite (20) | 21–88 | 3–42 | ||
| IV | granite (100) | 8 | 7 | >150 km |
| 1–17 | 2–17 | |||
| V | quartz sands (100) | 54 | 22 | >200 km |
| 47–75 | 4–29 | |||
| VI | gneiss (20), diorites (20), | 74 | 23 | >200 km |
| migmatites (20), tonalities (20), | 56–81 | 16–46 | ||
| pyroxenes (20) |
| Parameters | 1990 | 1995 | 2000 | 2005 | 2010 | 2014 | 2018 |
|---|---|---|---|---|---|---|---|
| pH | 6.79 | 6.7 | 6.85 | 6.41 | 6.58 | 6.55 | 6.6 |
| 4.32–7.18 | 4.64–7.59 | 4.85–7.45 | 4.64–7.21 | 4.99–7.33 | 4.49–7.34 | 4.55–7.23 | |
| ANC | 89 | 128 | 140 | 128 | 143 | 170 | 168 |
| μeq l−1 | [−23]–410 | [−10]–648 | [−10]–682 | [−11]–843 | [−4]–789 | 16–856 | 11–724 |
| BC | 305 | 319 | 309 | 280 | 293 | 290 | 299 |
| μeq l−1 | 137–527 | 57–717 | 54–629 | 53–646 | 51–695 | 47–634 | 54–602 |
| Alk | 125 | 131 | 137 | 111 | 96 | 135 | 151 |
| μeq l−1 | 0–400 | 0–639 | 0–694 | 0–839 | 0–793 | 0–866 | 0–683 |
| SO4* | 121 | 65 | 49 | 46 | 45 | 44 | 41 |
| μeq l−1 | 48–158 | 22–239 | 13–209 | 15–199 | 0–192 | 0–198 | 11–246 |
| Cl | 46 | 38 | 37 | 36 | 40 | 37 | 42 |
| μeq l−1 | 8–290 | 14–307 | 7–287 | 11–270 | 12–265 | 7–281 | 11–274 |
| Color | 49 | 35 | 27 | 28 | 19 | 24 | 20 |
| 0 Pt-Co | 0–88 | 0–156 | 0–143 | 3–123 | 3–165 | 2–128 | 3–136 |
| DOC | 4.9 | 5.6 | 5.9 | 6.9 | 7.4 | 6.4 | 5.2 |
| mgC L−1 | 1.2–8.9 | 2.6–15.1 | 1.6–14.5 | 2.4–18.7 | 2.2–22.5 | 2.2–20.6 | 2.6–19.3 |
| Ntot | 184 | 202 | 170 | 223 | 214 | 220 | 304 |
| μgN l−1 | 55–376 | 33–216 | 57–484 | 87–429 | 70–428 | 94–639 | 108–836 |
| NO3− | 1 | 1 | 1 | 2 | 2 | 1 | 1 |
| μgN l−1 | 0–3 | 0–23 | 0–17 | 1–17 | 0–30 | 0–31 | 0–28 |
| Ptot | 2 | 4 | 8 | 9 | 5 | 10 | 8 |
| μgP l−1 | 0–12 | 1–39 | 1–54 | 2–258 | 2–230 | 5–76 | 4–93 |
| PO43− | 4 | 1 | 1 | 1 | 2 | 4 | 2 |
| μgP l−1 | 2–10 | 0–29 | 0–34 | 0–98 | 1–36 | 0–41 | 0–38 |
| Al | 43 | 33 | 39 | 47 | 54 | 20 | 39 |
| μg l−1 | 0–149 | 3–330 | 4–186 | 10–448 | 9–416 | 8–170 | 5–352 |
| Fe | 55 | 41 | 45 | 70 | 77 | 88 | 60 |
| μg l−1 | 0–99 | 4–370 | 3–340 | 5–509 | 11–580 | 4–480 | 10–510 |
| Cu | 2.2 | 0.7 | 0.7 | 0.8 | 1.1 | 0.9 | 0.8 |
| μg l−1 | 0.5–9 | 0.1–14 | 0.2–12 | 0.2–8.9 | 0.3–12 | 0.1–14.2 | 0.3–12.5 |
| Ni | 3.9 | 0.6 | 0.8 | 0.9 | 0.9 | 0.7 | 0.8 |
| μg l−1 | 0.7–65 | 0.1–65 | 0.2–60 | 0.2–54 | 0.2–74 | 0.1–79 | 0.2–65 |
| SO4* Trend | Hardness (Ca2+ + Mg2+) Trend | ANC Trend | DOC Trend | Ntot Trend | Ptot Trend | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Slope | p | Slope | p | Slope | p | Slope | p | Slope | p | Slope | p | |
| μeq L−1 | μeq L−1 | μeq L−1 | mgC L−1 | μgN L−1 | μgP L−1 | |||||||
| I-around the smelters Kola, n = 7 | ||||||||||||
| 1990–2018 | −0.62 ◊ | 0.53 | −0.49 ◊ | 0.62 | +0.93 ◊ | 0.35 | +0.11 ◊ | 0.91 | +1.93 * | <0.05 | +1.21 ◊ | 0.23 |
| sustainable (II + III), n = 42 | ||||||||||||
| 1990–2018 | −4.03 *** | <0.001 | +3.14 ** | <0.01 | +6.63 *** | <0.001 | +3.07 ** | <0.01 | +5.62 *** | <0.001 | +2.16 * | <0.05 |
| acid-sensitive (IV + V), n = 14 | ||||||||||||
| 1990–2018 | −2.61 ** | <0.01 | −2.25 * | <0.05 | +2.25 * | <0.05 | +2.32 * | <0.05 | +2.43 * | <0.01 | +0.31 * | 0.75 |
| VI-forest and wetland landscapes, n = 12 | ||||||||||||
| 1990–2018 | −0.06 ◊ | 0.95 | −0.53 ◊ | 0.59 | −0.69 ◊ | 0.49 | −0.83 ◊ | 0.41 | +2.16 * | <0.05 | +2.65 ** | <0.01 |
| all region, n = 75 | ||||||||||||
| 1990–2018 | −5.85 *** | <0.001 | +1.99 * | <0.05 | +7.15 *** | <0.001 | +2.29 * | <0.05 | +2.42 * | <0.01 | +2.74 * | 0.03 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moiseenko, T.I.; Bazova, M.M.; Gashkina, N.A. Development of Lake from Acidification to Eutrophication in the Arctic Region under Reduced Acid Deposition and Climate Warming. Water 2022, 14, 3467. https://doi.org/10.3390/w14213467
Moiseenko TI, Bazova MM, Gashkina NA. Development of Lake from Acidification to Eutrophication in the Arctic Region under Reduced Acid Deposition and Climate Warming. Water. 2022; 14(21):3467. https://doi.org/10.3390/w14213467
Chicago/Turabian StyleMoiseenko, Tatyana I., Mariya M. Bazova, and Natalia A. Gashkina. 2022. "Development of Lake from Acidification to Eutrophication in the Arctic Region under Reduced Acid Deposition and Climate Warming" Water 14, no. 21: 3467. https://doi.org/10.3390/w14213467
APA StyleMoiseenko, T. I., Bazova, M. M., & Gashkina, N. A. (2022). Development of Lake from Acidification to Eutrophication in the Arctic Region under Reduced Acid Deposition and Climate Warming. Water, 14(21), 3467. https://doi.org/10.3390/w14213467








