δ34S, δ18O, and δ2H-δ18O as an Approach for Settling the Question of Groundwater Salinization in Neogene Basins: The North of Morocco in Focus
Abstract
:1. Introduction
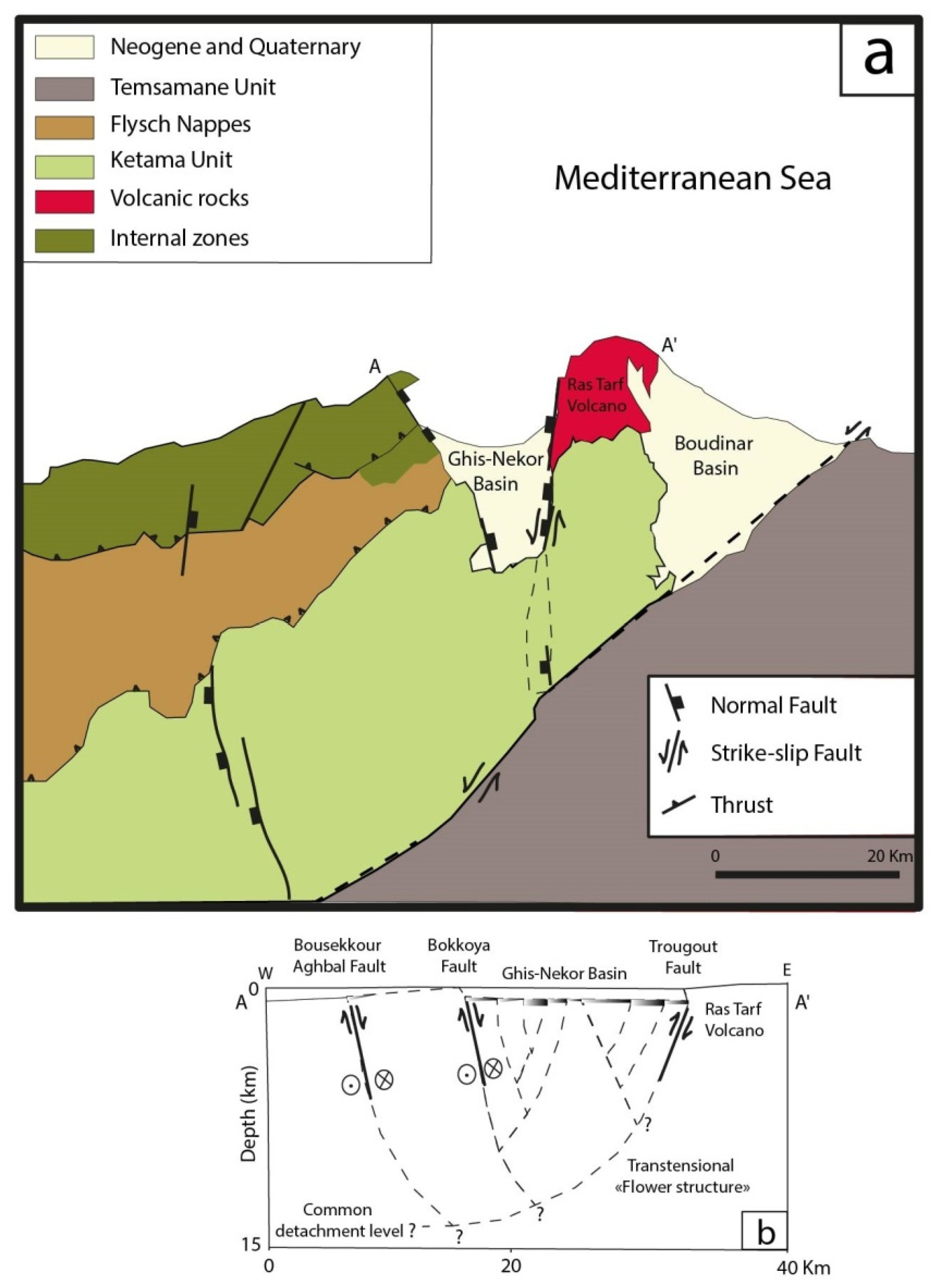
2. Regional Geology
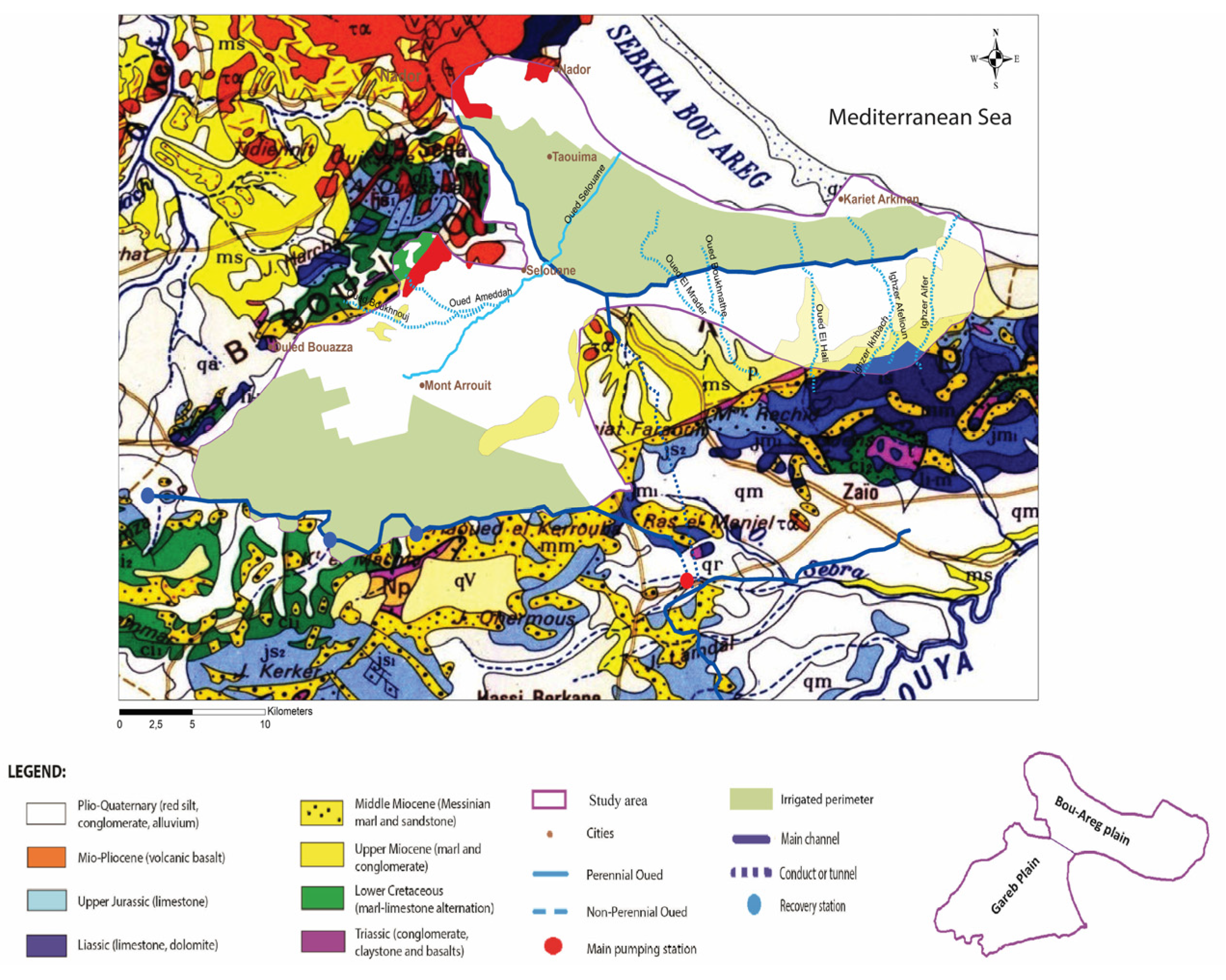
2.1. Local Geology and Hydrogeology
2.1.1. Ghis-Nekor Plain
2.1.2. BouAreg-Gareb Plain
2.1.3. Kert Plain
3. Materials and Methods
4. Results and Discussion
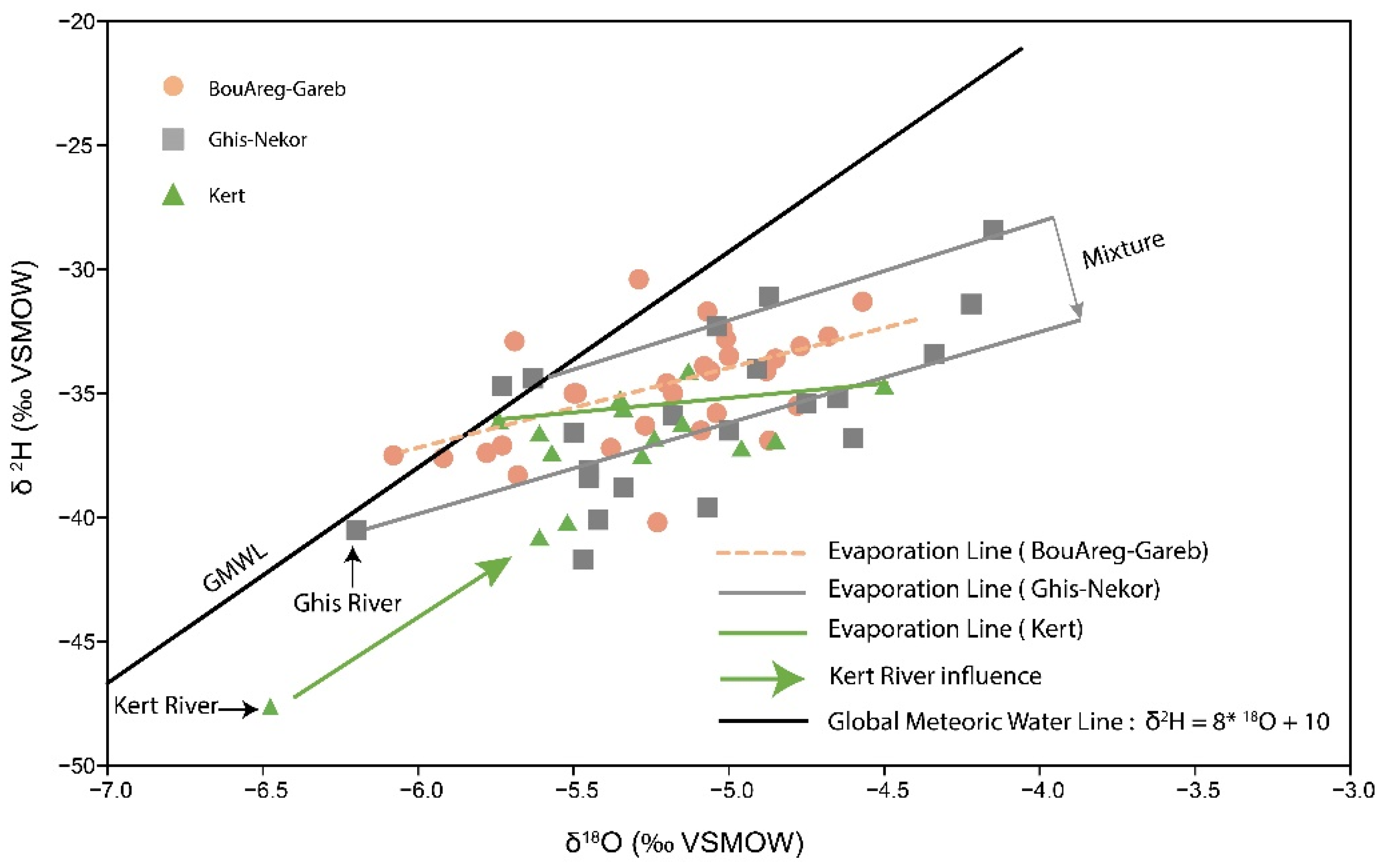
4.1. δ18O and δ2H
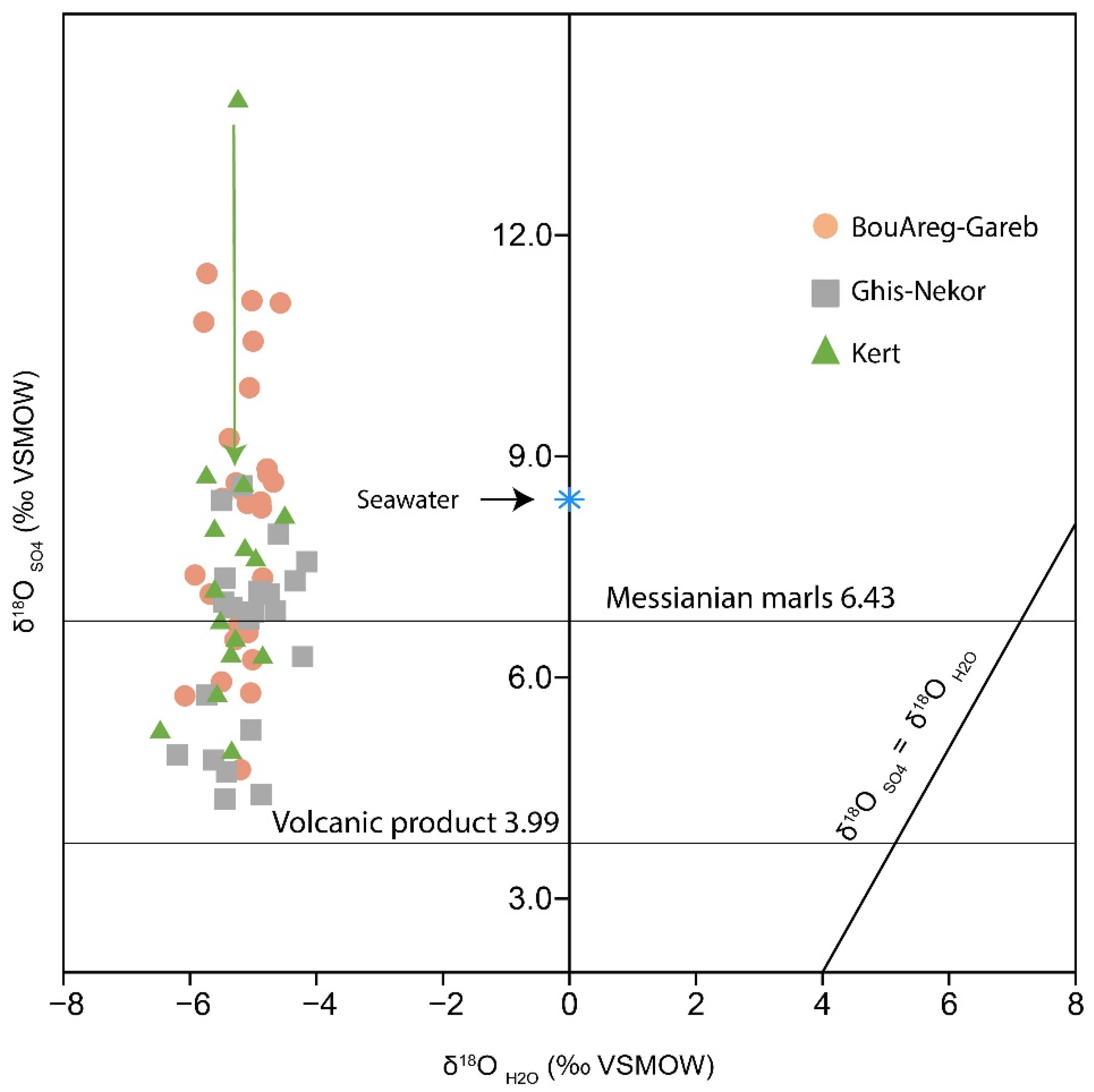
4.2. δ18OSO4 and δ18OH2O
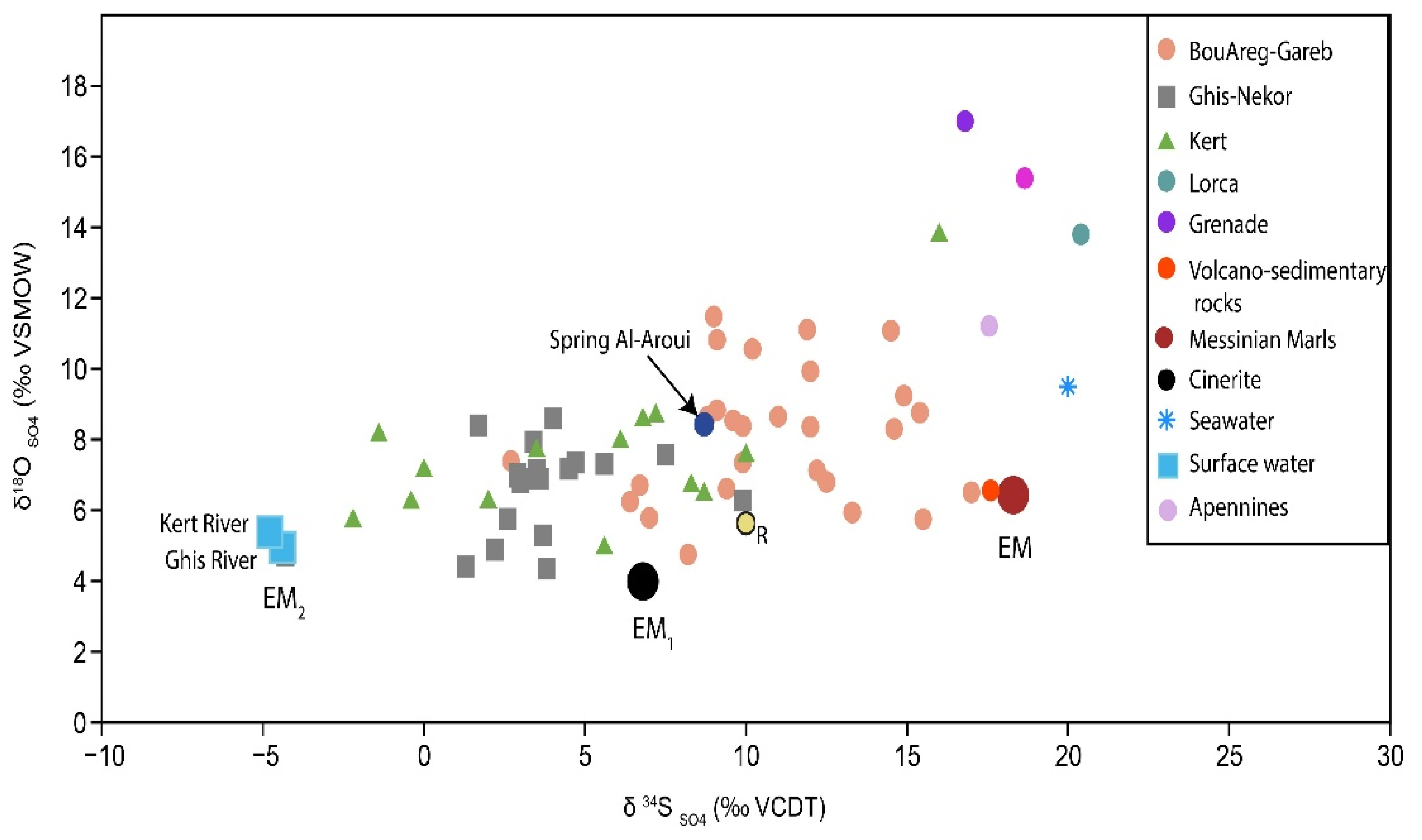
4.3. δ34SSO4 and δ18OSO4
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bouchaou, L.; Choukr-Allah, R.; Hirich, A.; Ennasr, M.S.; Malki, M.; Abahous, H.; Bouaakaz, B.; Nghira, A. Climate change and water valuation in Souss-Massa region: Management and adaptive measures. European Water 2017, 60, 203–209. [Google Scholar]
- Hssaisoune, M.; Bouchaou, L.; Sifeddine, A.; Bouimetarhan, I.; Chehbouni, A. Moroccan Groundwater Resources and Evolutionwith Global Climate Changes. Geosciences 2020, 10, 81. [Google Scholar] [CrossRef] [Green Version]
- Elgettafi, M.; Himi, M.; Elmandour, A.; Casas, A.; Boubker, E. Messinian salinity crisis impact on the groundwater quality in Kert aquifer NE Morocco: Hydrochemical and statistical approaches. Int. J. Water Res. Enviro. Eng. 2012, 11, 339–351. [Google Scholar]
- Hssaisoune, M.; Bouchaou, L.; Matsumoto, T.; Araguas, L.; Kraml, M.; Aggarwal, P. New evidences on groundwater dynamics from the Souss-Massa system (Morocco): Insights gained from dissolved noble gases. App. Geochem. 2019, 109, 104395. [Google Scholar] [CrossRef]
- Danni, S.O.; Bouchaou, L.; Elmouden, A.; Ait-Brahim, Y.; N’Da, B. Assessment of water quality and nitrate source in the Massa catchment (Morocco) using δ15N and δ18O tracers. Appl. Radiat. Isot. 2019, 154, 108859. [Google Scholar] [CrossRef]
- Re, V.; Sacchi, E.; Mas-Pla, J.; Menció, A.; El Amrani, N. Identifying the effects of human pressure on groundwater quality to support water management strategies in coastal regions: A multi-tracer and statistical approach (Bou-Areg region, Morocco). Sci. Total Environ. 2014, 500, 211–223. [Google Scholar] [CrossRef] [Green Version]
- Himi, M.; Tapias, J.; Benabdelouahab, S.; Salhi, A.; Rivero, L.; Elgettafi, M.; El Mandour, A.; Stitou, J.; Casas, A. Geophysical characterization of saltwater intrusion in a coastal aquifer: The case of Martil-Alila plain (North Morocco). J. Afr. Earth Sci. 2017, 126, 136–147. [Google Scholar] [CrossRef]
- Elgettafi, M.; Elmandour, A.; Himi, M.; Casas, A. The use of environmental markers to identify groundwater salinization sources in a Neogene basin, Kert aquifer case, NE Morocco. Int. J. Environ. Sci. Technol. 2013, 10, 719–728. [Google Scholar] [CrossRef] [Green Version]
- Karroum, M.; Elgettafi, M.; Elmandour, A.; Wilske, C.; Himi, M.; Casas, A. Geochemical processes controlling groundwater quality under semi-arid environment: A case study in central Morocco. Sci. Total Environ. 2017, 609, 1140–1151. [Google Scholar] [CrossRef]
- Chafouq, D.; El Mandour, A.; Elgettafi, M.; Himi, M.; Chouikri, I.; Casas, A. Hydrochemical and isotopic characterization of groundwater in the Ghis-Nekor plain (northern Morocco). J. Afr Earth Sci 2018, 139, 1–13. [Google Scholar] [CrossRef]
- Elmeknassi, M.; Bouchaou, L.; Elmandour, A.; Elgettafi, M.; Himi, M.; Casas, A. Multiple stable isotopes and geochemical approaches to elucidate groundwater salinity and contamination in the critical coastal zone: A case from the Bou-Areg and Gareb aquifers (North-Eastern Morocco). Environ. Pollut. 2022, 300, 118942. [Google Scholar] [CrossRef] [PubMed]
- Morel, J.L. Etats de contrainte et cinématique de la chaîne rifaine (Maroc) du Tortonienà l’actuel. Geodin. Acta 1989, 3, 283–294. [Google Scholar] [CrossRef]
- AitBrahim, L.; Chotin, P. Genèse et déformation des bassins néogènes du Rif central (Maroc) au cours du rapprochement Europe-Afrique. Geodin. Acta 1989, 3, 295–304. [Google Scholar] [CrossRef]
- Hsü, K.J.; Cita, M.B.; Ryan, W.B.F. The Origin of the Mediterranean Evaporites; Initial Reports of the Deep Sea Drilling Project; U.S. Govemment Printing Office: Washington, DC, USA, 1973; pp. 1203–1231. [Google Scholar]
- Montadert, L.; Sancho, J.; Fail, J.P.; Debyser, J.; Winnock, E. De l’âge tertiaire de la série salifère responsable des structures diapiriques en Méditerranée Occidentale (NordEst des Baléares). Comptes Rendus l’Académie Sci. 1970, 271, 812–815. [Google Scholar]
- Decima, A.; Wezel, F.C. Late Miocene Evaporites of the Central Sicilian Basin, Italy. Initial. Rep. Deep. Sea Drill. Proj. 1973, 13, 1234–1241. [Google Scholar]
- Clauzon, G.; Suc, J.P.; Gautier, F.; Berger, A.; Loutre, M.-F. Alternate interpretation of the Messinian Salinity Crisis: Controversy resolved? Geology 1996, 24, 363–366. [Google Scholar] [CrossRef]
- Krijgsman, W.; Langereis, C.G.; Zachariasse, W.J.; Boccaletti, M.; Moratti, G.; Gelati, R.; Iaccarino, S.; Papani, G.; Villa, G. Late Neogene evolution of the Taza-Guercif Basin (Rifian Corridor, Morocco) and implications for the Messinian salinity crisis. Mar. Geol. 1999, 153, 147–160. [Google Scholar] [CrossRef] [Green Version]
- Garcia-Castellanos, D.; Villaseñor, A. Messinian salinity crisis regulated by competing tectonics and erosion at the Gibraltar arc. Nature 2011, 480, 359–363. [Google Scholar] [CrossRef]
- Manzi, V.; Gennari, R.; Hilgen, F.; Krijgsman, W.; Lugli, S.; Roveri, M.; Sierro, F.J. Age refinement of the Messinian salinity crisis onset in the Mediterranean. Terra Nova 2013, 25, 315–322. [Google Scholar] [CrossRef]
- Pérez-Asensio, J.N.; Aguirre, J.; Jimenez-Moreno, G.; Schmiedl, G.; Civis, J. Glacioeustatic control on the origin and cessation of the Messinian salinity crisis. Glob. Planet. Chang 2013, 111, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Lourens, L.J.; Antonarakou, A.; Hilgen, F.J.; Van Hoof, A.A.M.; Vergnaud Grazzini, C.; Zachariasse, W.J. Evaluation of the Pliocene to early Pleistocene astronomical time scale. Paleoceanography 1996, 11, 391–413. [Google Scholar] [CrossRef]
- Baksi, A.K. A geomagnetic polarity timescale for the period 0-17Ma, based on 40Ar/39Ar plateau ages for selected field reversals. Geophys. Res. Lett. 1993, 20, 1607–1610. [Google Scholar] [CrossRef]
- Do Couto, D. Evolution Géodynamique de la Mer D’alboran par L’étude des Bassins Sédimentaires. Ph.D. Thesis, Université Pierre et Marie Curie, Paris, France, 2014. [Google Scholar]
- El Azzouzi, M.; Bernard-Griffiths, J.; Bellon, H.; Maury, R.C.; Pique, A.; Fourcade, S.; Cotten, J.; Hernandez, J. Evolution des sources du volcanisme marocain au cours du Neogene. CR Acad. Sci. Paris 1999, 329, 95–102. [Google Scholar] [CrossRef]
- D’Acremont, E.; Gutscher, M.A.; Rabaute, A.; Mercier de Lépinay, B.; Lafosse, M.; Poort, J.; Ammar, A.; Tahayt, A.; Le Roy, P.; Smit, J.; et al. High-resolution imagery of active faulting offshore Al Hoceima, Northern Morocco. Tectonophysics 2014, 632, 160–166. [Google Scholar] [CrossRef]
- El Alami, S.O.; Tadili, B.A.; Cherkaoui, T.E.; Medina, F.; Ramdani, M.; AitBrahim, L.; Harnaff, M. The Al Hoceima earthquake of May 26, 1994 and its aftershocks: A seismotectonic study. Ann. Geofis. 1998, 41, 519–537. [Google Scholar] [CrossRef]
- Medina, F.; El Alami, S.O. Focal mechanisms and state of stress in the Al Hoceima area (Central Rif, Morocco). Bull. l’Inst. Rabat Sect. Sci. Terre 2006, 28, 19–30. [Google Scholar]
- Poujol, A.; Ritz, J.F.; Tahayt, A.; Vernant, P.; Condomines, M.; Blard, P.H.; Billant, J.; Vacher, L.; Tibari, B.; Hni, L.; et al. Active tectonics of the northern Rif (Morocco) from geomorphic and geochronological data. J. Geodyn. 2014, 77, 70–88. [Google Scholar] [CrossRef]
- Elmandour, A.; El Yaouti, F.; Fakir, Y.; Zarhloule, Y.; Benavente, J. Evolution of groundwater salinity in the unconfined aquifer of BouAreg, Northeastern Mediterranian coast, Morocco. Environ. Geol. 2008, 54, 491–503. [Google Scholar] [CrossRef]
- El Yaouti, F.; Elmandour, A.; Khattach, D.; Benavente, J.; Kaufmann, O. Salinization processes in the unconfined aquifer of Bou-Areg (NE Morocco): A geostatistical, geochemical, and tomographic study. Appl. Geochem. 2009, 24, 16–31. [Google Scholar] [CrossRef]
- Frizon de Lamotte, D. La Structure du Rif Oriental (Maroc): Rôle de la Tectonique Longitudinale et Importance Des Fluides. Ph.D. Thesis, University of Piérre et Marie Curie, Paris, France, 1985. [Google Scholar]
- Elgettafi, M. Caractérisation des Processus de la Salinisation des Eaux Souterraines de la Plaine de Kert, Maroc, Nord Oriental: Approches Hydrogéologique, Géochimique et Géophysique. Ph.D. Thesis, Université de Marrakech, Marrakech, Morocco, 2011. [Google Scholar]
- Carlier, P. Carte Hydrogéologique au 1/50,000 de la Plaine du Moyen-Kerte, Province de Nador, Maroc Nord-Oriental: Notice Explicative; Éditions du Service Géologique du Maroc: Rabat, Morocco, 1973; Volume 250. [Google Scholar]
- Craig, H. Isotopic variation in meteoric waters. Science 1961, 133, 1702–1703. [Google Scholar] [CrossRef]
- Gamsjäger, H.; Murmann, R.K. Advances in Inorganic and Bioinorganic Mechanisms; Sykes, A.G., Ed.; Academic Press: London, UK, 1983; Volume 2, pp. 317–381. [Google Scholar]
- Boschetti, T.; Iacumin, P. Continuous-flow δ18O measurements: New approach to standardization, high-temperature thermodynamic and sulfate analysis. Rapid Commun. Mass Spectrom. 2005, 19, 3007–3014. [Google Scholar] [CrossRef] [PubMed]
- Van Stempvoort, D.R.; Krouse, H.R. Controls of δ18O in sulfate: Review of experimental data and application to specific environments. Environ. Geochem. Sulfide Oxid. 1994, 29, 446–480. [Google Scholar]
- Elkilany, A. Hydrologie et Cycles Biogéochimiques du Soufre Dans Deux Bassins Marginaux de Méditerranée Pendant la Crise de Salinité Messinienne. Ph.D. Thesis, Sorbonne University, Paris, France, 2018. [Google Scholar]
- Claypool, G.E.; Holser, W.T.; Kaplan, I.R.; Zak, I. The age curves of sulfur and oxygen isotope in marine sulfate and their mutual interpretations. Chem. Geol. 1980, 28, 199–260. [Google Scholar] [CrossRef]
- Urresti-Estala, B.; Vadillo-Pérez, I.; Jiménez-Gavilan, P.; Soler, A.; Sanchez-García, D.; Carrasco-Cantos, F. Application of stable isotopes (δ34S-SO4, δ18O-SO4, δ15N-NO3, δ18O-NO3) to determine natural background and contamination sources in the Guadalhorce River Basin (southern Spain). Sci. Total Environ. 2015, 506, 46–57. [Google Scholar] [CrossRef] [PubMed]
- Pierre, C. Teneurs en Isotopes Stables (18O, 2H, 13C, 34S) et Conditions de Genèse des Evaporites Marines: Application à Quelques Milieux Actuels et au Messinien de la Méditerranée. Ph.D. Thesis, University of Paris-Sud, Orsay, France, 1982; 226p. [Google Scholar]
- Boschetti, T.; Cortecchi, G.; Toscani, L.; Iacumin, P. Sulfur and oxygen isotope compositions of Upper Triassic sulfates from Northerm Apennines (Italy): Palaeogeographic and hidrogeochemical implications. Geol. Acta 2011, 9, 129–147. [Google Scholar]


| ID | EC | Na+ | Cl− | SO4 | NO3− | δ18OH2O | δ2H | δ18OSO4 | δ34S | Slope |
|---|---|---|---|---|---|---|---|---|---|---|
| µS/cm | mg/L | ‰ | ||||||||
| B1 | 2010 | 431 | 802 | 102 | 6 | −6.08 | −37.5 | 5.75 | 15.5 | 7.81 |
| B2 | 3840 | 851 | 1385 | 342 | 35 | −5.38 | −37.2 | 9.24 | 14.9 | 8.78 |
| B4 | 6210 | 1507 | 2490 | 324 | 14 | −5.29 | −30.4 | 6.51 | 17.0 | 7.63 |
| B5 | 6670 | 1219 | 2428 | 339 | 34 | −5.01 | −32.8 | 6.24 | 6.4 | 8.56 |
| B10 | 6880 | 1473 | 2344 | 472 | 34 | −5.68 | −38.3 | 7.13 | 12.2 | 8.51 |
| B15 | 6070 | 984 | 1448 | 594 | 25 | −4.87 | −36.9 | 8.30 | 14.6 | 9.62 |
| B17 | 6910 | 1783 | 2286 | 490 | 15 | −4.77 | −33.1 | 8.76 | 15.4 | 9.03 |
| B18 | 3430 | 523 | 748 | 602 | 30 | −5.09 | −36.5 | 8.36 | 12.0 | 9.13 |
| B21 | 5160 | 1156 | 1590 | 771 | 32 | −5.06 | −34.1 | 9.93 | 12.0 | 8.71 |
| B23 | 7840 | 346 | 398 | 538 | 28 | −4.78 | −35.5 | 8.83 | 9.1 | 9.52 |
| B26 | 8300 | 935 | 2171 | 780 | 18 | −5.23 | −40.2 | 6.71 | 6.7 | 9.60 |
| B27 | 6260 | 1337 | 1495 | 793 | 9 | −4.68 | −32.7 | 8.65 | 11.0 | 9.13 |
| B29 | 5330 | 932 | 795 | 681 | 41 | −5.27 | −36.3 | 8.64 | 8.8 | 8.78 |
| B31 | 22,600 | 2961 | 6759 | 810 | 5 | −4.57 | −31.3 | 11.08 | 14.5 | 9.03 |
| B32 | 17,000 | 2875 | 5112 | 339 | 8 | −5.50 | −35.0 | 5.94 | 13.3 | 8.18 |
| B33 | 4260 | 453 | 1068 | 579 | 62 | −5.07 | −31.7 | 6.80 | 12.5 | 8.22 |
| B34 | 4900 | 1081 | 994 | 882 | 45 | −5.00 | −33.5 | 10.56 | 10.2 | 8.70 |
| G1 | 6680 | 1783 | 2933 | 506 | 16 | −5.20 | −34.6 | 4.75 | 8.2 | 8.58 |
| G3 | 11,900 | 2034 | 3050 | 546 | 16 | −5.04 | −35.8 | 5.79 | 7.0 | 9.08 |
| G5 | 8040 | 1855 | 2627 | 866 | 27 | −4.85 | −33.6 | 7.35 | 9.9 | 8.98 |
| G6 | 4890 | 1507 | 1988 | 779 | 32 | −5.18 | −35.0 | 8.54 | 9.6 | 8.67 |
| G7 | 8055 | 1498 | 2370 | 879 | 39 | −4.88 | −34.1 | 8.38 | 9.9 | 9.03 |
| G8 | 5660 | 955 | 1590 | 572 | 37 | −5.73 | −37.1 | 11.48 | 9.0 | 8.22 |
| G9 | 5630 | 1070 | 1463 | 590 | 39 | −5.78 | −37.4 | 10.82 | 9.1 | 8.20 |
| G10 | 3970 | 598 | 1022 | 603 | 8 | −5.92 | −37.6 | 7.39 | 2.7 | 8.05 |
| G11 | 11,200 | 1679 | 3465 | 750 | 15 | −5.08 | −33.9 | 6.61 | 9.4 | 8.64 |
| G13 | 4810 | 449 | 909 | 562 | 29 | −5.02 | −32.4 | 11.11 | 11.9 | 8.45 |
| S | 19,700 | 3197 | 5853 | 398 | 8 | −5.49 | −35.0 | 8.43 | 8.7 | 8.20 |
| R | 1680 | 276 | 298 | 235 | 62 | −5.69 | −32.9 | 5.79 | 9.9 | 7.53 |
| GN3 | 2419 | 281 | 277 | 589 | 15 | −5.47 | −41.7 | 7.02 | 2.9 | 7.63 |
| GN8 | 2859 | 277 | 414 | 621 | 15 | −5.00 | −36.5 | 6.89 | 3.6 | 7.30 |
| GN9 | 4624 | 575 | 927 | 944 | 41 | −4.34 | −33.4 | 7.31 | 5.6 | 7.69 |
| GN20 | 4000 | 527 | 646 | 755 | 46 | −4.87 | −31.1 | 4.41 | 1.3 | 6.39 |
| GN23 | 5011 | 615 | 1063 | 407 | 123 | −4.15 | −28.4 | 7.57 | 7.5 | 6.84 |
| GN27 | 4062 | 527 | 775 | 485 | 485 | −5.04 | −32.3 | 5.29 | 3.7 | 6.41 |
| GN32 | 2561 | 204 | 333 | 561 | 9 | −5.42 | −40.1 | 4.72 | −4.3 | 7.40 |
| GN34 | 5958 | 759 | 1315 | 1031 | 10 | −5.50 | −36.6 | 8.40 | 1.7 | 6.65 |
| GN37 | 5117 | 620 | 839 | 1267 | 41 | −4.65 | −35.2 | 6.90 | 3.2 | 7.57 |
| GN39 | 2916 | 267 | 412 | 663 | 32 | −5.45 | −38.4 | 4.35 | 3.8 | 7.05 |
| GN40 | 10,575 | 1497 | 2976 | 990 | 112 | −4.22 | −31.4 | 6.28 | 9.9 | 7.45 |
| GN44 | 4008 | 472 | 548 | 1111 | 20 | −5.07 | −39.6 | 6.79 | 3.0 | 7.82 |
| GN45 | 2796 | 255 | 362 | 725 | 16 | −4.75 | −35.4 | 7.13 | 3.5 | 7.45 |
| GN46 | 3433 | 352 | 502 | 847 | 14 | −5.34 | −38.8 | 6.95 | 2.9 | 7.27 |
| GN48 | 3357 | 346 | 509 | 840 | 17 | −4.60 | −36.8 | 7.94 | 3.4 | 8.00 |
| GN49 | 4167 | 484 | 653 | 1101 | 11 | −5.18 | −35.9 | 8.60 | 4.0 | 6.94 |
| GN50 | 2723 | 300 | 394 | 500 | 21 | −5.73 | −34.7 | 5.76 | 2.6 | 6.05 |
| GN53 | 3400 | 376 | 558 | 522 | 30 | −5.63 | −34.4 | 4.88 | 2.2 | 6.11 |
| GN57 | 6760 | 1208 | 1101 | 1615 | 42 | −4.91 | −34.0 | 7.17 | 4.5 | 6.92 |
| GN59 | 2750 | 251 | 420 | 601 | 27 | −5.45 | −38.1 | 7.35 | 4.7 | 6.99 |
| GR | 2200 | 250 | 459 | 700 | 6 | −6.20 | −40.5 | 4.95 | −4.4 | 6.53 |
| K1 | 5670 | 822 | 1153 | 1397 | 66 | −5.61 | −36.6 | 7.21 | 0.0 | 6.52 |
| K2 | 5050 | 843 | 122 | 666 | 35 | −5.61 | −40.8 | 8.03 | 6.1 | 7.25 |
| K3 | 4680 | 792 | 974 | 639 | 30 | −5.74 | −36.1 | 8.76 | 7.2 | 6.28 |
| K4 | 6990 | 1189 | 1867 | 1168 | 36 | −4.96 | −37.2 | 7.63 | 10.0 | 7.50 |
| K5 | 7420 | 1658 | 1289 | 1462 | 0 | −5.24 | −36.8 | 13.86 | 16.0 | 7.00 |
| K6 | 1330 | 149 | 243 | 107 | 27 | −5.13 | −34.1 | 7.77 | 3.5 | 6.63 |
| K7 | 2830 | 581 | 458 | 248 | 12 | −5.34 | −35.6 | 5.02 | 5.6 | 6.65 |
| K8 | 6200 | 886 | 1517 | 1316 | 96 | −4.85 | −36.9 | 6.31 | −0.4 | 7.59 |
| K9 | 3690 | 664 | 739 | 428 | 17 | −5.15 | −36.2 | 8.64 | 6.8 | 7.02 |
| K10 | 5080 | 881 | 1064 | 1435 | 18 | −5.35 | −35.2 | 6.33 | 2.0 | 6.61 |
| K11 | 850 | 55 | 86 | 134 | 18 | −5.57 | −37.4 | 5.79 | −2.2 | 6.71 |
| K12 | 1020 | 172 | 64 | 55 | 24 | −5.52 | −40.2 | 6.78 | 8.3 | 7.57 |
| K13 | 4120 | 547 | 758 | 1063 | 94 | −4.50 | −34.7 | 8.21 | −1.4 | 7.69 |
| K14 | 4520 | 634 | 1121 | 512 | 65 | −5.28 | −37.5 | 6.54 | 8.7 | 7.08 |
| KR | 3828 | 3830 | 485 | 1476 | 21 | −6.47 | −47.6 | 5.30 | −4.6 | 7.35 |
| MM | 6.43 | 18.3 | ||||||||
| C | 3.99 | 6.8 | ||||||||
| VSD | 6.56 | 17.6 | ||||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Elgettafi, M.; Elmeknassi, M.; Elmandour, A.; Himi, M.; Lorenzo, J.M.; Casas, A. δ34S, δ18O, and δ2H-δ18O as an Approach for Settling the Question of Groundwater Salinization in Neogene Basins: The North of Morocco in Focus. Water 2022, 14, 3404. https://doi.org/10.3390/w14213404
Elgettafi M, Elmeknassi M, Elmandour A, Himi M, Lorenzo JM, Casas A. δ34S, δ18O, and δ2H-δ18O as an Approach for Settling the Question of Groundwater Salinization in Neogene Basins: The North of Morocco in Focus. Water. 2022; 14(21):3404. https://doi.org/10.3390/w14213404
Chicago/Turabian StyleElgettafi, Mohammed, Malak Elmeknassi, Abdenabi Elmandour, Mahjoub Himi, Juan M. Lorenzo, and Albert Casas. 2022. "δ34S, δ18O, and δ2H-δ18O as an Approach for Settling the Question of Groundwater Salinization in Neogene Basins: The North of Morocco in Focus" Water 14, no. 21: 3404. https://doi.org/10.3390/w14213404
APA StyleElgettafi, M., Elmeknassi, M., Elmandour, A., Himi, M., Lorenzo, J. M., & Casas, A. (2022). δ34S, δ18O, and δ2H-δ18O as an Approach for Settling the Question of Groundwater Salinization in Neogene Basins: The North of Morocco in Focus. Water, 14(21), 3404. https://doi.org/10.3390/w14213404








