Mechanism and Reactive Species in a Fountain-Strip DBD Plasma for Degrading Perfluorooctanoic Acid (PFOA)
Abstract
:1. Introduction
2. Experiments
2.1. Materials
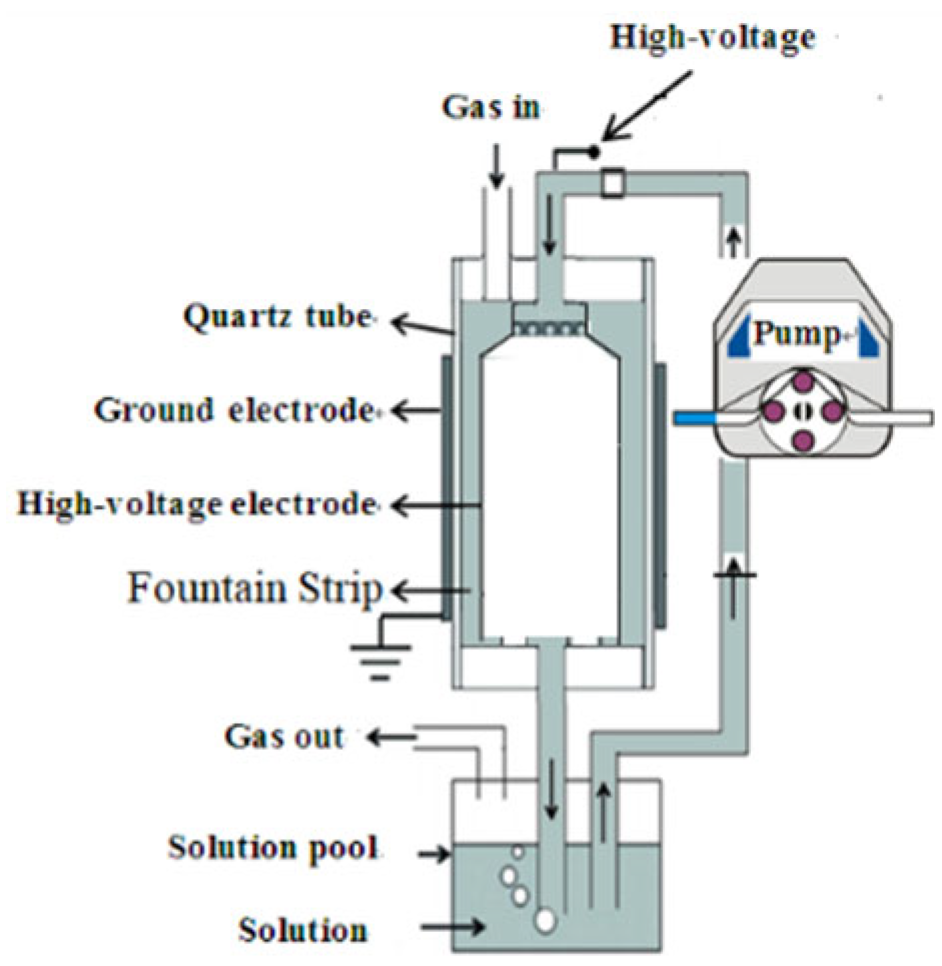
2.2. Experimental Setup
2.3. Analytical Methods
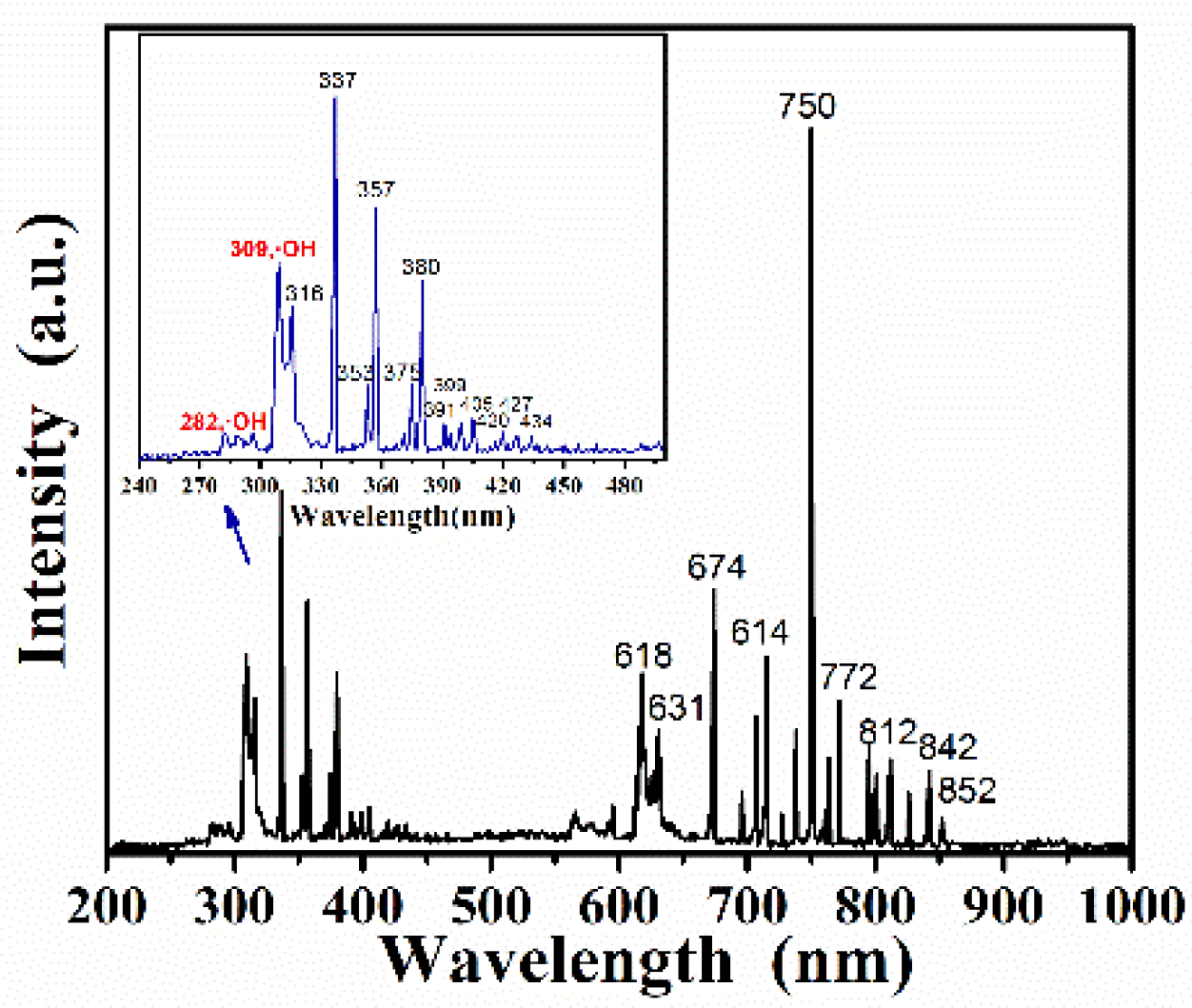
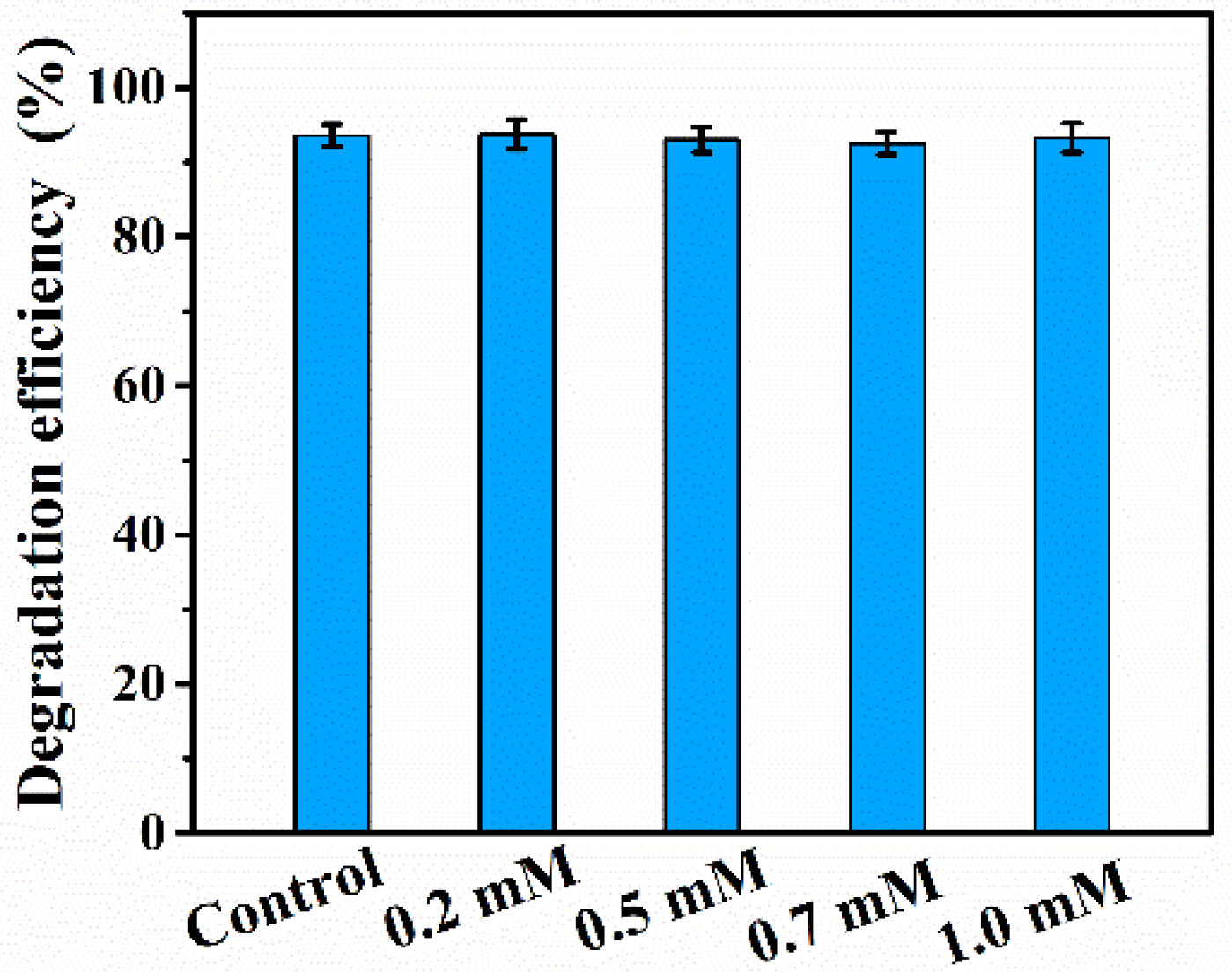
3. Results and Discussion
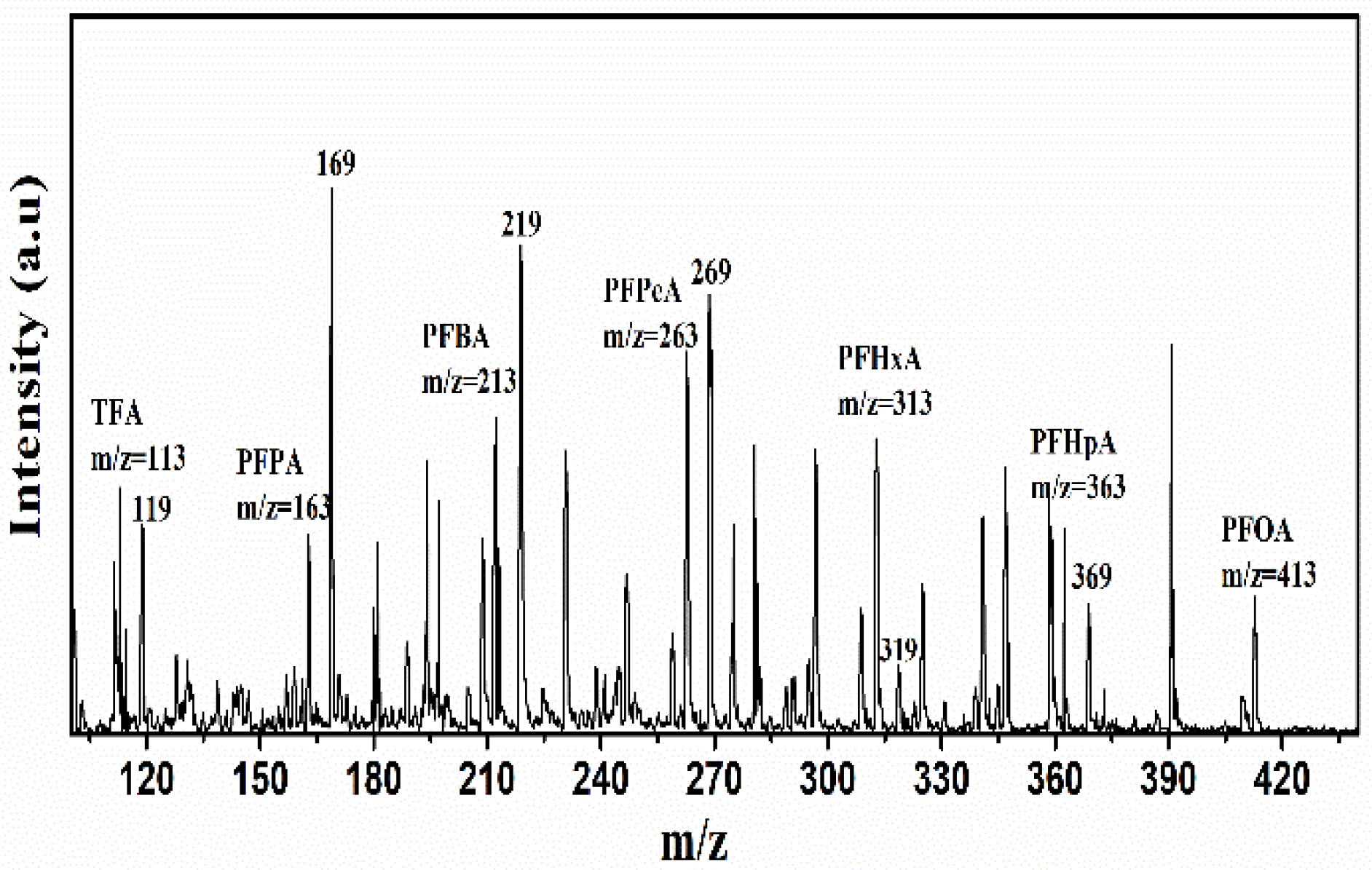
Decomposition Mechanism Analyzed from the Intermediates/Products
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Khan, M.J.; Jovicic, V.; Zbogar-Rasic, A.; Poser, A.; Freichels, K.; Delgado, A. Effectiveness of non-thermal plasma induced degradation of per- and polyfluoroalkyl substances from water. Water 2022, 14, 1408. [Google Scholar] [CrossRef]
- Wan, H.; Miills, R.; Qu, K.; Hower, J.C.; Mottaleb, M.A.; Bhattacharyya, D.; Xu, Z. Rapid removal of PFOA and PFOS via modified industrial solid waste: Mechanisms and influences of water matrices. Chem. Eng. J. 2022, 433, 133271. [Google Scholar] [CrossRef]
- Zhang, L.H.; Cheng, J.H.; You, X.; Hu, Y. Photochemical defluorination of aqueous perfluorooctanoic acid (PFOA) by Fe0/GAC micro-electrolysis and VUV-Fenton photolysis. Environ. Sci. Pollut. Res. 2016, 23, 13531–13542. [Google Scholar] [CrossRef]
- Maisonet, M.; Terrell, M.L.; McGeehin, M.A.; Christensen, K.Y.; Holmes, A.; Calafat, A.M.; Marcus, M. Maternal concentrations of polyfluoroalkyl compounds during pregnancy and fetal and postnatal growth in British girls. Environ. Health Perspect. 2012, 120, 1432–1437. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, J.Y.; Wang, X.; Pan, Z.Q.; Xukai Li Yu Ling Laisheng, L.i. Efficient degradation of perfluorooctanoic acid (PFOA) by photocatalytic ozonation. Chem. Eng. J. 2016, 296, 329–334. [Google Scholar] [CrossRef]
- Lee, Y.C.; Chen, M.J.; Huang, C.P.; Kuoc, J.; Lo, S.-L. Efficient sonochemical degradation of perfluorooctanoic acid using periodate. Ultrason. Sonochem 2016, 31, 499–505. [Google Scholar] [CrossRef]
- Yasuoka, K.; Sasaki, K.; Hayashi, R. An energy-efficient process for decomposing perfluorooctanoic and perfluorooctane sulfonic acids using dc plasmas generated within gas bubbles. Plasma Sources Sci. Technol. 2011, 20, 034009. [Google Scholar] [CrossRef]
- Mhadhbi, L.; Rial, D.; Pérez, S.; Beiras, R. Ecological risk assessment of perfluorooctanoic acid (PFOA) and perfluorooctanesulfonic acid (PFOS) in marine environment using Isochrysis galbana, Paracentrotus lividus, Siriella armata and Psetta maxima. J. Environ. Monit. 2012, 14, 1375–1382. [Google Scholar] [CrossRef] [Green Version]
- Qu, Y.; Zhang, C.; Li, F.; Bo, X.; Liu, G.; Zhou, Q. Equilibrium and kinetics study on the adsorption of perfluorooctanoic acid from aqueous solution onto powdered activated carbon. J. Hazard. Mater. 2009, 69, 146–152. [Google Scholar] [CrossRef]
- Lampert, D.J.; Frisch, M.A.; Speitel, G.E. Removal of Perfluorooctanoic Acid and Perfluorooctane Sulfonate from Wastewater by Ion Exchange. Pract. Period Hazard Toxic Radioact. Waste Manag. 2007, 11, 60–68. [Google Scholar] [CrossRef]
- Trojanowicz, M.; Bojanowska-Czajka, A.; Bartosiewicz, I.; Kulisa, K. Advanced oxidation/reduction processes treatment for aqueous perfluorooctanoate (PFOA) and perfluorooctanesulfonate (PFOS)–a review of recent advances. Chem. Eng. J. 2018, 336, 170–199. [Google Scholar] [CrossRef]
- Chen, J.; Zhang, P.Y.; Liu, J. Photodegradation of perfluorooctanoic acid by 185 nm vacuum ultraviolet light. J. Environ. Sci. 2007, 19, 387–390. [Google Scholar] [CrossRef]
- Moriwaki, H.; Takagi, Y.; Tanaka, M.; Tsuruho, K.; Okitsu, K.; Maeda, Y. Sonochemical decomposition of perfluorooctane sulfonate and perfluorooctanoic acid. Environ. Sci. Technol. 2005, 39, 3388–3392. [Google Scholar] [CrossRef]
- Lee, Y.C.; Lo, S.L.; Chiueh, P.T.; Liou, Y.H.; Chen, M.L. Microwave-hydrothermal decomposition of perfluorooctanoic acid in water by iron-activated persulfate oxidation. Water Res. 2010, 44, 886–892. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.; Niu, J.F.; Ding, S.Y.; Zhang, L.L. Electrochemical degradation of perfluorooctanoic acid (PFOA) by Ti/SnO2–Sb, Ti/SnO2–Sb/PbO2 and Ti/SnO2–Sb/MnO2 anodes. Water Res. 2012, 46, 2281–2289. [Google Scholar] [CrossRef]
- Trojanowicz, M.; Bartosiewicz, I.; Bojanowska-Czajka, A.; Kulisa, K.; Szreder, T.; Bobrowski, K.; Nichipor, H.; Garcia-Reyes, J.F.; Nałęcz-Jawecki, G.; Męczyńska-Wielgosz, S. Application of ionizing radiation in decomposition of perfluorooctanoate (PFOA) in waters. Chem. Eng. J. 2019, 357, 698–714. [Google Scholar] [CrossRef]
- Singh, R.K.; Fernando, S.; Baygi, S.F.; Multari, N.; Thagard, S.M.; Holsen, T.M. Breakdown products from perfluorinated alkyl substances (PFAS) degradation in a plasma-based water treatment process. Environ. Sci. Technol. 2019, 53, 2731–2738. [Google Scholar] [CrossRef]
- Hayashi, R.; Obo, H.; Takeuchi, N.; Yasuoka, K. Decomposition of perfluorinated compounds in water by DC plasma within oxygen bubbles. Electr. Eng. Jpn. 2015, 190, 9–16. [Google Scholar] [CrossRef]
- Stratton, G.R.; Dai, F.; Bellona, C.L.; Holsen, T.M.; Dickenson, E.R.; Thagard, M.S. Plasma-based water treatment: Efficient transformation of perfluoroalkyl substances in prepared solutions and contaminated groundwater. Environ. Sci. Technol. 2017, 51, 1643–1648. [Google Scholar] [CrossRef]
- Cheng, J.S.; Fan, Y.Y.; Pei, X.Y.; Tian, D.; Liu, Z.; Yang, L.; Feng, E.; Ji, H.-F.; Chen, Q. An Energy Efficient Process for Degrading Perfluorooctanoic Acid (PFOA) Using Strip Fountain Dielectric Barrier Discharge Plasma. Water 2022, 14, 2420. [Google Scholar] [CrossRef]
- Takeuchi, N.; Kitagawa, Y.; Kosugi, A.; Tachibana, K.; Obo, H.; Yasuoka, K. Plasma–liquid interfacial reaction in decomposition of perfluoro surfactants. J. Phys. D Appl. Phys. 2013, 47, 045203. [Google Scholar] [CrossRef]
- Panchangam, S.C.; Lin, A.-Y.C.; Shaik, K.L.; Lin, C.F. Decomposition of perfluorocarboxylic acids (PFCAs) by heterogeneous photocatalysis in acidic aqueous medium. Chemosphere 2009, 77, 242–248. [Google Scholar] [CrossRef] [PubMed]
- Horikoshi, S.; Sato, S.; Abe, M.; Serpone, N. A novel liquid plasma AOP device integrating microwaves and ultrasounds and its evaluation in defluorinating perfluorooctanoic acid in aqueous media. Ultrason. Sonochem. 2011, 18, 938–942. [Google Scholar] [CrossRef] [PubMed]
- Vanraes, P.; Willems, G.; Nikiforov, A.; Surmont, P.; Lynen, F.; Vandamme, J.; Van Durme, J.; Verheust, Y.P.; Van Hulle, S.W.; Dumoulin, A.; et al. Removal of atrazine in water by combination of activated carbon and dielectric barrier discharge. J. Hazard. Mater. 2015, 299, 647–655. [Google Scholar] [CrossRef] [PubMed]
- Lukes, P.; Dolezalova, E.; Sisrova, I.; Clupek, M. Aqueous-phase chemistry and bactericidal effects from an air discharge plasma in contact with water: Evidence for the formation of peroxynitrite through a pseudo-second-order post-discharge reaction of H2O2 and HNO2. Plasma Sources Sci. Technol. 2014, 23, 015019. [Google Scholar] [CrossRef]
- Nohara, K.; Toma, M.; Kutsuna, S.; Takeuchi, K.; Ibusuki, T. Cl atom-initiated oxidation of three homologous methyl perfluoroalkyl ethers. Environ. Sci. Technol. 2001, 35, 114–120. [Google Scholar] [CrossRef]
- Wallington, T.J.; Hurley, M.D.; Fracheboud, J.M.; Orlando, A.J.J.; Tyndall, G.S.; Sehested, J.; Møgelberg, T.E.; Nielsen, O.J. Role of excited CF3CFHO radicals in the atmospheric chemistry of HFC-134a. J. Phys. Chem. 1996, 100, 18116–18122. [Google Scholar] [CrossRef]
- Jorfi, S.; Kakavandi, B.; Motlagh, H.R.; Ahmadi, M.; Jaafarzadeh, N. A novel combination of oxidative degradation for benzotriazole removal using TiO2 loaded on FeIIFe2IIIO4@C as an efficient activator of peroxymonosulfate. Appl. Catal. B Environ. 2017, 219, 216–230. [Google Scholar] [CrossRef]
- Wang, Y.P.; Li, F.; Xue, T.S.; Liu, C.; Yuan, D.; Qi, F.; Xu, B. Heterogeneous activation of peroxymonosulfate by hierarchical CuBi2O4 to generate reactive oxygen species for refractory organic compounds degradation: Morphology and surface chemistry derived reaction and its mechanism. Environ. Sci. Pollut. Res. Int. 2018, 25, 4419–4434. [Google Scholar] [CrossRef]
- Liu, Z.C.; Liu, D.X.; Chen, C.; Li, D.; Yang, A.J.; Rong, M.Z.; Chen, H.L.; Kong, M.G. Physicochemical processes in the indirect interaction between surface air plasma and deionized water. J. Phys. D Appl. Phys. 2015, 48, 495201. [Google Scholar] [CrossRef]
- Peng, J.W.; Lee, S. Atmospheric Pressure Plasma Degradation of Azo Dyes in Water: pH and Structural Effects. Plasma Chem. Plasma Process. 2013, 33, 1063–1072. [Google Scholar] [CrossRef]
- Hong, Y.J.; Nam, C.J.; Song, K.B.; Cho, G.S.; Uhm, H.S.; Choi, D.I.; Choi, H. Measurement of hydroxyl radical density generated from the atmospheric pressure bioplasma jet. J. Instrum. 2012, 7, C03046. [Google Scholar] [CrossRef]
- Durantini, A.M.; Greene, L.E.; Lincoln, R.; Martinez, S.R.; Cosa, G. Reactive oxygen species mediated activation of a dormant singlet oxygen photosensitizer: From auto- catalytic singlet oxygen amplification to chemicontrolled photodynamic therapy. J. Am. Chem. Soc. 2016, 138, 1215–1225. [Google Scholar] [CrossRef]
- Mahyar, A.; Miessner, H.; Mueller, S.; Aziz, K.H.H.; Kalass, D.; Moeller, D.; Kretschmer, K.; Manuel, S.R.; Noack, J. Development and application of different non-thermal plasma reactors for the removal of perfluorosurfactants in water: A comparative study. Plasma Chem. Plasma Process. 2019, 39, 531–544. [Google Scholar] [CrossRef]
- Kiwi, J.; Lopez, A.; Nadtochenko, V. Mechanism and kinetics of the OH-radical intervention during Fenton oxidation in the presence of a significant amount of radical scavenger (Cl-). Environ. Sci. Technol. 2000, 34, 2162–2168. [Google Scholar] [CrossRef]
- Bruggeman, P.J.; Kushner, M.J.; Locke, B.R.; Gardeniers, J.G.E.; Graham, W.G.; Graves, D.B.; Hofman-Caris, R.C.H.M.; Maric, D.; Reid, J.P.; Ceriani, E.; et al. Plasma–liquid interactions: A review and roadmap. Plasma Sources Sci. Technol. 2016, 25, 053002. [Google Scholar] [CrossRef] [Green Version]
- Song, Z.; Tang, H.Q.; Wang, N.; Zhu, L.H. Reductive defluorination of perfluorooctanoic acid by hydrated electrons in a sulfite-mediated UV photochemical system. J. Hazard. Mater. 2013, 262, 332–338. [Google Scholar] [CrossRef]
- Li, X.; Ma, J.; Liu, G.; Fang, J.; Yue, S.; Guan, Y.; Chen, L.; Liu, X. Efficient reductive dechlorination of monochloroacetic acid by sulfite/UV process. Environ. Sci. Technol. 2012, 46, 7342–7349. [Google Scholar] [CrossRef]
- Buxton, G.V.; Greenstock, C.L.; Helman, W.P.; Ross, A.B. Critical review of rate constants for reactions of hydrated electrons, hydrogen atoms and hydroxyl radicals (OH/⋅O− in aqueous solution. J. Phys. Chem. Ref. Data 1988, 17, 513–886. [Google Scholar] [CrossRef] [Green Version]
- Rumbach, P.; Bartels, D.M.; Sankaran, R.M.; Go, D.B. The solvation of electrons by an atmospheric-pressure plasma. Nat. Commun. 2015, 6, 7248. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Craven, M.; Yu, X.; Ding, J.; Bryant, P.; Hualng, J.; Tu, X. Plasma-Enhanced Catalytic Synthesis of Ammonia over a Ni/Al2O3 Catalyst at Near-Room Temperature: Insights into the Importance of the Catalyst Surface on the Reaction Mechanism. ACS Catal. 2019, 9, 10780–10793. [Google Scholar] [CrossRef] [PubMed]

| C Number | Compound | Chemical Formula |
|---|---|---|
| 6 | Perfluoroheptanoic acid | C6F13COOH |
| 5 | Perfluorohexanoic acid, | C5F11COOH |
| 4 | Perfluoropentanoic acid | C4F9COOH |
| 3 | Perfluorobutanoic acid | C3F7COOH |
| 2 | Perfluoropropionic acid | C2F5COOH |
| 1 | Trifluoroacetic acid | CF3COOH |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cheng, J.; Fan, Y.; Pei, X.; Tian, D.; Liu, Z.; Wei, Z.Z.; Ji, H.-f.; Chen, Q. Mechanism and Reactive Species in a Fountain-Strip DBD Plasma for Degrading Perfluorooctanoic Acid (PFOA). Water 2022, 14, 3384. https://doi.org/10.3390/w14213384
Cheng J, Fan Y, Pei X, Tian D, Liu Z, Wei ZZ, Ji H-f, Chen Q. Mechanism and Reactive Species in a Fountain-Strip DBD Plasma for Degrading Perfluorooctanoic Acid (PFOA). Water. 2022; 14(21):3384. https://doi.org/10.3390/w14213384
Chicago/Turabian StyleCheng, Jiushan, Yangyang Fan, Xueyun Pei, Di Tian, Zhongwei Liu, Zachary Z. Wei, Hai-feng Ji, and Qiang Chen. 2022. "Mechanism and Reactive Species in a Fountain-Strip DBD Plasma for Degrading Perfluorooctanoic Acid (PFOA)" Water 14, no. 21: 3384. https://doi.org/10.3390/w14213384
APA StyleCheng, J., Fan, Y., Pei, X., Tian, D., Liu, Z., Wei, Z. Z., Ji, H.-f., & Chen, Q. (2022). Mechanism and Reactive Species in a Fountain-Strip DBD Plasma for Degrading Perfluorooctanoic Acid (PFOA). Water, 14(21), 3384. https://doi.org/10.3390/w14213384







