Preparation of Fe3O4/α-MnO2 Magnetic Nanocomposites for Degradation of 2,4-DCP through Persulfate Activation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents and Instruments
2.2. Experimental Methods
2.3. Analysis Methods
3. Results and Discussion
3.1. Characterization of the Fe3O4/α-MnO2 MNCs
3.2. Effectiveness of the Fe3O4/α-MnO2 MNCs in the Removal of 2,4-DCP by PS Activation
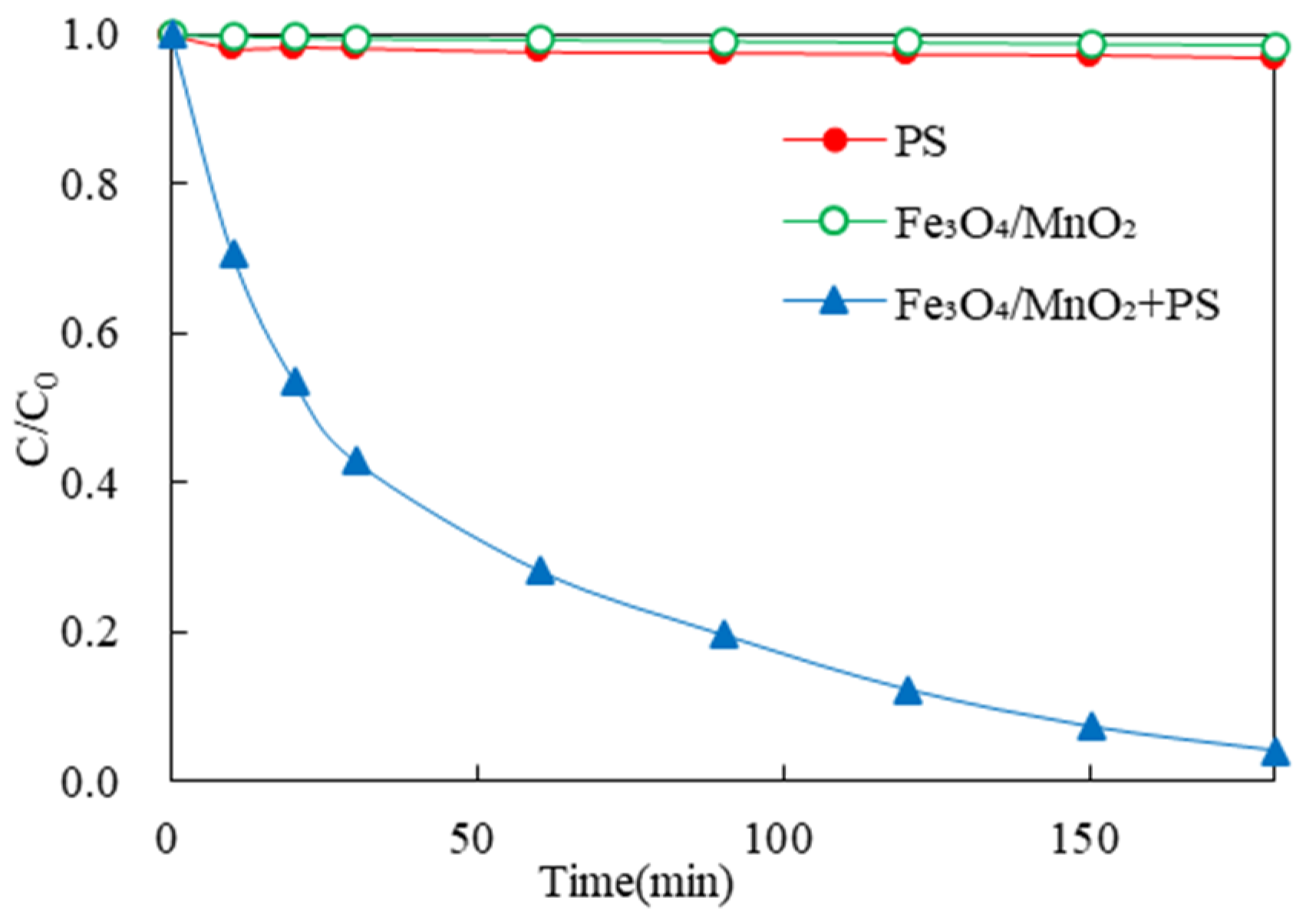
3.2.1. Comparison of 2,4-DCP Removal by Different Activated PS-Based Systems
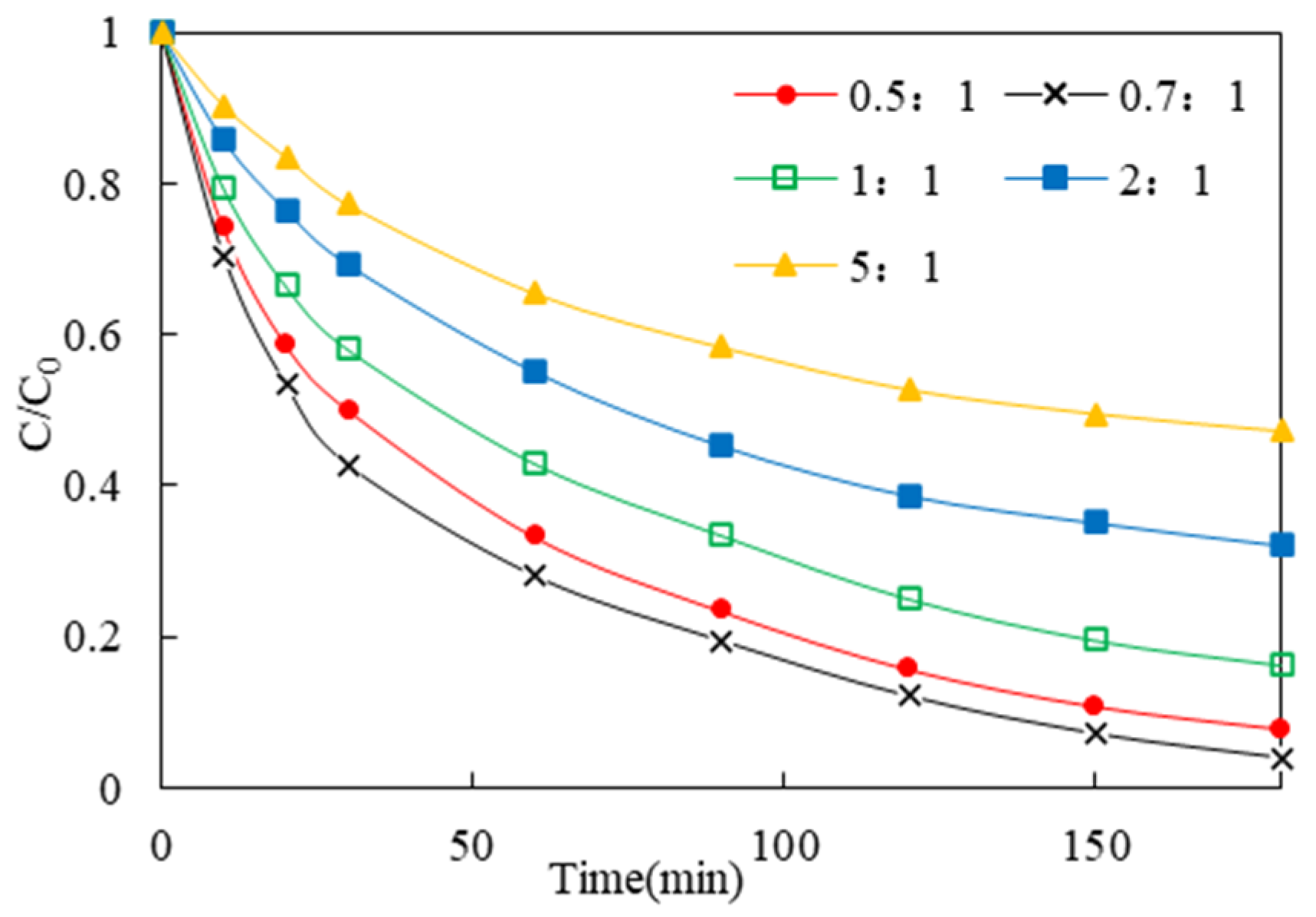
3.2.2. Optimization of the Loading Ratio of Fe3O4 on α-MnO2
3.3. Influencing Factors of 2,4-DCP Removal through the Activated PS by Fe3O4/α-MnO2 MNCs
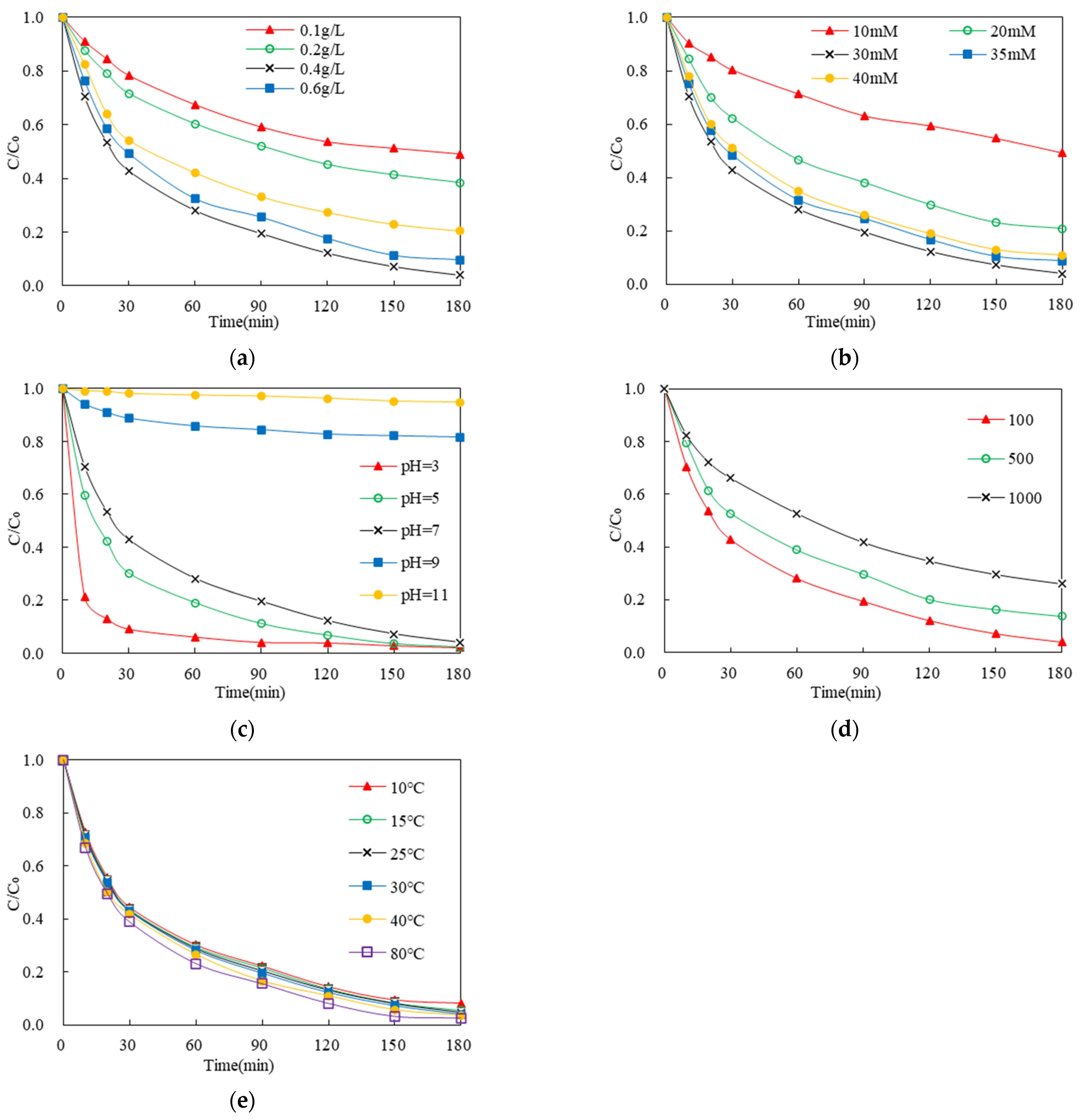
3.3.1. Effect of Activator Dosage
3.3.2. Effect of Oxidant Dosage
3.3.3. The Effect of the Initial pH of the System
3.3.4. Effect of Initial Pollutant Concentration in the System
3.3.5. Effect of Temperature
3.4. Reusability
3.5. Reaction Mechanism of 2,4-DCP Removal by the Fe3O4/α-MnO2-Activated PS System
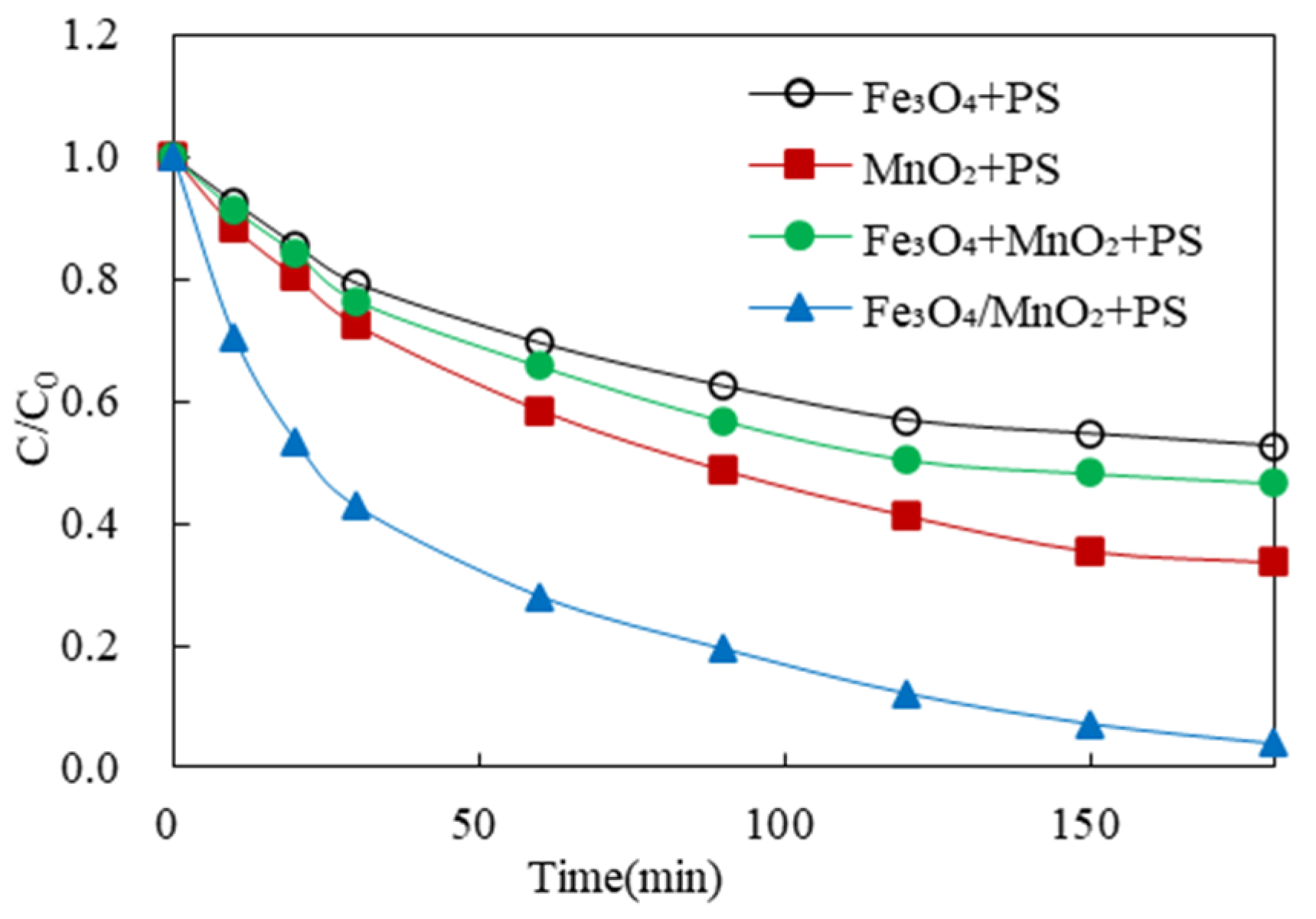
3.5.1. Comparison of 2,4-DCP Removal by Different Activated PS Systems
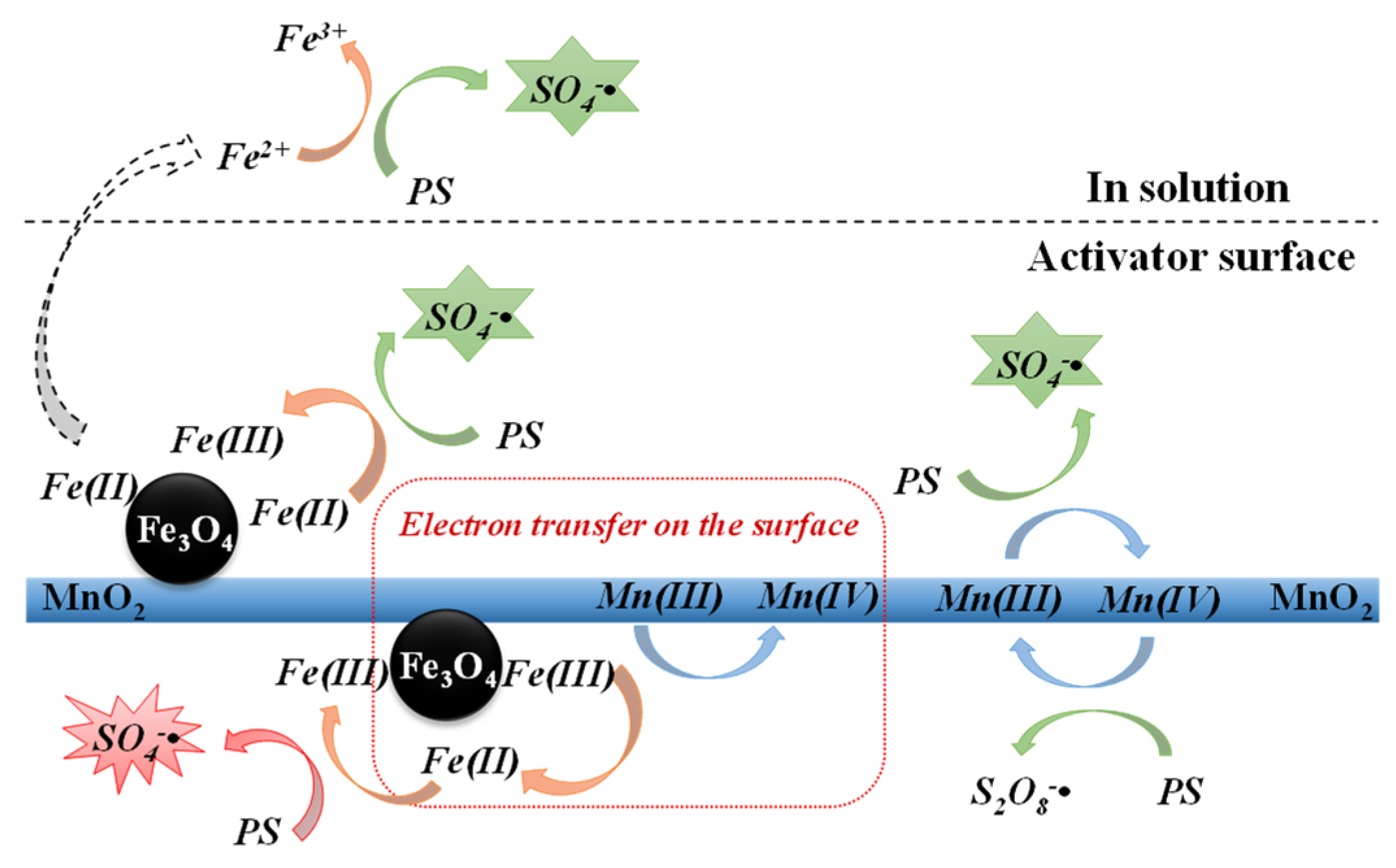
3.5.2. Speculation of the Reaction Mechanism for the Removal of 2,4-DCP by the Fe3O4/α-MnO2-Activated PS System
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ribeiro, A.R.; Nunes, O.C.; Pereira, M.F.R.; Silva, A.M.T. An overview on the advanced oxidation processes applied for the treatment of water pollutants defined in the recently launched Directive 2013/39/EU. Environ. Int. 2015, 75, 33–51. [Google Scholar] [CrossRef] [Green Version]
- Wang, J.L.; Xu, L.J. Advanced Oxidation Processes for Wastewater Treatment: Formation of Hydroxyl Radical and Application. Crit. Rev. Environ. Sci. Technol. 2012, 42, 251–325. [Google Scholar] [CrossRef]
- Asgari, E.; Esrafili, A.; Rostami, R.; Farzadkia, M. O3, O3/UV and O3/UV/ZnO for abatement of parabens in aqueous solutions: Effect of operational parameters and mineralization/biodegradability improvement. Process Saf. Environ. Prot. 2019, 125, 238–250. [Google Scholar] [CrossRef]
- Zhou, R.; Liu, S.; He, F.R.; Ren, H.J.; Han, Z.H. Alkylpolyglycoside modified MnFe2O4 with abundant oxygen vacancies boosting singlet oxygen dominated peroxymonosulfate activation for organic pollutants degradation. Chemosphere 2021, 285, 11. [Google Scholar] [CrossRef]
- Pera-Titus, M.; García-Molina, V.; Baños, M.A.; Giménez, J.; Esplugas, S. Degradation of chlorophenols by means of advanced oxidation processes: A general review. Appl. Catal. B 2004, 47, 219–256. [Google Scholar] [CrossRef]
- Jiang, G.; Zhu, B.; Sun, J.; Liu, F.; Wang, Y.; Zhao, C. Enhanced activity of ZnS (111) by N/Cu co-doping: Accelerated degradation of organic pollutants under visible light. J. Environ. Sci. 2023, 125, 244–257. [Google Scholar] [CrossRef]
- Oh, S.-Y.; Kim, H.-W.; Park, J.-M.; Park, H.-S.; Yoon, C. Oxidation of polyvinyl alcohol by persulfate activated with heat, Fe2+, and zero-valent iron. J. Hazard. Mater. 2009, 168, 346–351. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Bruton, T.A.; Doyle, F.M.; Sedlak, D.L. In Situ Chemical Oxidation of Contaminated Groundwater by Persulfate: Decomposition by Fe(III)- and Mn(IV)-Containing Oxides and Aquifer Materials. Environ. Sci. Technol. 2014, 48, 10330–10336. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oyekunle, D.T.; Cai, J.; Gendy, E.A.; Chen, Z. Impact of chloride ions on activated persulfates based advanced oxidation process (AOPs): A mini review. Chemosphere 2021, 280, 130949. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Yao, B.; Luo, Z.; Li, W.; Li, C.; Ye, Z.; Gong, X.; Yang, J.; Zhou, Y. Applications and influencing factors of the biochar-persulfate based advanced oxidation processes for the remediation of groundwater and soil contaminated with organic compounds. Sci. Total Environ. 2022, 836, 155421. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Xiao, S.; Zhong, H.; Yan, M.; Yang, X. Activation of persulfates by carbonaceous materials: A review. Chem. Eng. J. 2021, 418, 129297. [Google Scholar] [CrossRef]
- Oyekunle, D.T.; Gendy, E.A.; Ifthikar, J.; Chen, Z. Heterogeneous activation of persulfate by metal and non-metal catalyst for the degradation of sulfamethoxazole: A review. Chem. Eng. J. 2022, 437, 135277. [Google Scholar] [CrossRef]
- Saputra, E.; Muhammad, S.; Sun, H.; Ang, H.-M.; Tadé, M.O.; Wang, S. A comparative study of spinel structured Mn3O4, Co3O4 and Fe3O4 nanoparticles in catalytic oxidation of phenolic contaminants in aqueous solutions. J. Colloid Interface Sci. 2013, 407, 467–473. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jabbar, T.A.; Ammar, S.H. Core/shell phosphomolybdic acid-supported magnetic silica nanocomposite (Ni@SiO2-PMo): Synthesis, characterization and its application as a recyclable antibacterial agent. Colloid Interface Sci. Commun. 2019, 33, 100214. [Google Scholar] [CrossRef]
- Xu, L.; Wang, J. Fenton-like degradation of 2, 4-dichlorophenol using Fe3O4 magnetic nanoparticles. Appl. Catal. B 2012, 123, 117–126. [Google Scholar] [CrossRef]
- Ren, H.; Su, Y.; Han, X.; Zhou, R. Synthesis and characterization of saponin-modified Fe3O4 nanoparticles as heterogeneous Fenton-catalyst with enhanced degradation of p-nitrophenol. J. Chem. Technol. Biotechnol. 2016, 92, 1421–1427. [Google Scholar] [CrossRef]
- Zhou, R.; Lu, S.; Su, Y.; Li, T.; Ma, T.; Ren, H. Hierarchically fusiform CuO microstructures decorated with Fe3O4 nanoparticles as novel persulfate activators for 4-aminobenzenesulfonic acid degradation in aqueous solutions. J. Alloys Compd. 2020, 815, 152394. [Google Scholar] [CrossRef]
- Fang, G.-D.; Dionysiou, D.D.; Al-Abed, S.R.; Zhou, D.-M. Superoxide radical driving the activation of persulfate by magnetite nanoparticles: Implications for the degradation of PCBs. Appl. Catal. B 2013, 129, 325–332. [Google Scholar] [CrossRef]
- Li, R.; Jin, X.; Megharaj, M.; Naidu, R.; Chen, Z. Heterogeneous Fenton oxidation of 2,4-dichlorophenol using iron-based nanoparticles and persulfate system. Chem. Eng. J. 2015, 264, 587–594. [Google Scholar] [CrossRef]
- Jia, J.; Zhang, P.; Chen, L. The effect of morphology of α-MnO2 on catalytic decomposition of gaseous ozone. Catal. Sci. Technol. 2016, 6, 5841–5847. [Google Scholar] [CrossRef]
- Xu, L.; Xu, C.; Zhao, M.; Qiu, Y.; Sheng, G.D. Oxidative removal of aqueous steroid estrogens by manganese oxides. Water Res. 2008, 42, 5038–5044. [Google Scholar] [CrossRef]
- Saputra, E.; Muhammad, S.; Sun, H.; Ang, H.M.; Tade, M.; Wang, S. Different crystallographic one-dimensional MnO2 nanomaterials and their superior performance in catalytic phenol degradation. Environ. Sci. Technol. 2013, 47, 5882–5887. [Google Scholar] [CrossRef] [PubMed]
- Das, T.; Ganguly, S.; Remanan, S.; Ghosh, S.; Das, N. Mussel-inspired Ag/poly(norepinephrine)/MnO2 heterogeneous nanocatalyst for efficient reduction of 4-nitrophenol and 4-nitroaniline: An alternative approach. Res. Chem. Intermed. 2020, 46, 3629–3650. [Google Scholar] [CrossRef]
- Zhao, H.; Cui, H.-J.; Fu, M.-L. Synthesis of core–shell structured Fe3O4@ α-MnO2 microspheres for efficient catalytic degradation of ciprofloxacin. RSC Adv. 2014, 4, 39472–39475. [Google Scholar] [CrossRef]
- Wang, Y.; Ren, H.; Pan, H.; Liu, J.; Zhang, L. Enhanced tolerance and remediation to mixed contaminates of PCBs and 2, 4-DCP by transgenic alfalfa plants expressing the 2, 3-dihydroxybiphenyl-1, 2-dioxygenase. J. Hazard. Mater. 2015, 286, 269–275. [Google Scholar] [CrossRef] [PubMed]
- Saputra, E.; Muhammad, S.; Sun, H.; Patel, A.; Shukla, P.; Zhu, Z.; Wang, S. α-MnO2 activation of peroxymonosulfate for catalytic phenol degradation in aqueous solutions. Catal. Commun. 2012, 26, 144–148. [Google Scholar] [CrossRef]
- Jiang, Z.; Li, J.; Jiang, D.; Gao, Y.; Chen, Y.; Wang, W.; Cao, B.; Tao, Y.; Wang, L.; Zhang, Y. Removal of atrazine by biochar-supported zero-valent iron catalyzed persulfate oxidation: Reactivity, radical production and transformation pathway. Environ. Res. 2020, 184, 109260. [Google Scholar] [CrossRef]
- Wu, H.; Wang, J.; Liu, H.; Fan, X. Performance, reaction pathway and kinetics of the enhanced dechlorination degradation of 2,4-dichlorophenol by Fe/Ni nanoparticles supported on attapulgite disaggregated by a ball milling–freezing process. Materials 2022, 15, 3957. [Google Scholar] [CrossRef]
- Zhao, Y.; Zhao, Y.; Zhou, R.; Mao, Y.; Tang, W.; Ren, H. Insights into the degradation of 2,4-dichlorophenol in aqueous solution by alpha-MnO2 nanowire activated persulfate: Catalytic performance and kinetic modeling. RSC Adv. 2016, 6, 35441–35448. [Google Scholar] [CrossRef]
- Yan, J.; Lei, M.; Zhu, L.; Anjum, M.N.; Zou, J.; Tang, H. Degradation of sulfamonomethoxine with Fe3O4 magnetic nanoparticles as heterogeneous activator of persulfate. J. Hazard. Mater. 2011, 186, 1398–1404. [Google Scholar] [CrossRef]
- Xia, C.; Liu, Q.; Zhao, L.; Wang, L.; Tang, J. Enhanced degradation of petroleum hydrocarbons in soil by FeS@BC activated persulfate and its mechanism. Sep. Purif. Technol. 2022, 282, 120060. [Google Scholar] [CrossRef]
- Yuan, Y.; Tao, H.; Fan, J.; Ma, L. Degradation of p-chloroaniline by persulfate activated with ferrous sulfide ore particles. Chem. Eng. J. 2015, 268, 38–46. [Google Scholar] [CrossRef]
- Couttenye, R.; Huang, K.-C.; Hoag, G.; Suib, S. Evidence of Sulfate Free Radical (SO4-•) Formation under Heat-Assisted Persulfate Oxidation of MTBE. Bull. Educ. Res. Pract. 2003, 8, 345–350. [Google Scholar]
- Do, S.-H.; Kwon, Y.-J.; Kong, S.-H. Effect of metal oxides on the reactivity of persulfate/Fe(II) in the remediation of diesel-contaminated soil and sand. J. Hazard. Mater. 2010, 182, 933–936. [Google Scholar] [CrossRef]
- Hou, L.; Zhang, H.; Xue, X. Ultrasound enhanced heterogeneous activation of peroxydisulfate by magnetite catalyst for the degradation of tetracycline in water. Sep. Purif. Technol. 2012, 84, 147–152. [Google Scholar] [CrossRef]
- Xin Zhang, Y.; Long Guo, X.; Huang, M.; Dong Hao, X.; Yuan, Y.; Hua, C. Engineering birnessite-type MnO2 nanosheets on fiberglass for pH-dependent degradation of methylene blue. J. Phys. Chem. Solids 2015, 83, 40–46. [Google Scholar] [CrossRef]
- Liu, J.; Zhao, Z.; Shao, P.; Cui, F. Activation of peroxymonosulfate with magnetic Fe3O4-α-MnO2 core–shell nanocomposites for 4-chlorophenol degradation. Chem. Eng. J. 2015, 262, 854–861. [Google Scholar] [CrossRef]
- Prélot, B.; Poinsignon, C.; Thomas, F.; Schouller, E.; Villiéras, F. Structural–chemical disorder of manganese dioxides: 1. Influence on surface properties at the solid–electrolyte interface. J. Colloid Interface Sci. 2003, 257, 77–84. [Google Scholar] [CrossRef]
- Rahmani, A.; Salari, M.; Tari, K.; Shabanloo, A.; Shabanloo, N.; Bajalan, S. Enhanced degradation of furfural by heat-activated persulfate/nZVI-rGO oxidation system: Degradation pathway and improving the biodegradability of oil refinery wastewater. J. Environ. Chem. Eng. 2020, 8, 104468. [Google Scholar] [CrossRef]
- Liang, H.; Sun, H.; Patel, A.; Shukla, P.; Zhu, Z.H.; Wang, S. Excellent performance of mesoporous Co3O4/MnO2 nanoparticles in heterogeneous activation of peroxymonosulfate for phenol degradation in aqueous solutions. Appl. Catal. B 2012, 127, 330–335. [Google Scholar] [CrossRef]
- Gan, P.; Zhang, Z.; Hu, Y.; Li, Y.; Ye, J.; Tong, M.; Liang, J. Insight into the role of Fe in the synergetic effect of persulfate/sulfite and Fe2O3@g-C3N4 for carbamazepine degradation. Sci. Total Environ. 2022, 819, 152787. [Google Scholar] [CrossRef] [PubMed]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, Y.; Luo, F.; Zhou, R. Preparation of Fe3O4/α-MnO2 Magnetic Nanocomposites for Degradation of 2,4-DCP through Persulfate Activation. Water 2022, 14, 3312. https://doi.org/10.3390/w14203312
Zhao Y, Luo F, Zhou R. Preparation of Fe3O4/α-MnO2 Magnetic Nanocomposites for Degradation of 2,4-DCP through Persulfate Activation. Water. 2022; 14(20):3312. https://doi.org/10.3390/w14203312
Chicago/Turabian StyleZhao, Yan, Fei Luo, and Rui Zhou. 2022. "Preparation of Fe3O4/α-MnO2 Magnetic Nanocomposites for Degradation of 2,4-DCP through Persulfate Activation" Water 14, no. 20: 3312. https://doi.org/10.3390/w14203312
APA StyleZhao, Y., Luo, F., & Zhou, R. (2022). Preparation of Fe3O4/α-MnO2 Magnetic Nanocomposites for Degradation of 2,4-DCP through Persulfate Activation. Water, 14(20), 3312. https://doi.org/10.3390/w14203312






