1. Introduction
Extracellular polymeric substances (EPS) are commonly defined as a combination of high-molecular-weight secretions from microorganisms, products of cell lysis and hydrolysis of macromolecules, and adsorbed organic matter from activated sludge (AS). EPS is described as having significant influence on the physico-chemical properties of microbial aggregates, affecting their structure, surface charge, flocculation, settling properties, dewatering properties, and sorption ability. EPS can serve as a carbon or energy source during nutrient shortages and aid the formation of microbial aggregates by binding cells closely [
1].
There is agreement within the literature that more than half of organic matter contained in AS consists of EPS, but there is no consensus about what exactly constitutes EPS or how best to extract and characterize it. EPS has been grouped into loosely-bound EPS (LB-EPS), tightly-bound EPS (TB-EPS), and soluble EPS, also referred to as soluble microbial products (SMP). SMP can be separated by centrifugation alone, but the quantification of bound EPS requires physical or chemical extraction to affect the release of materials from the cell matrix prior to centrifugation [
2].
Biosorption is a biochemical process which occurs naturally when raw, untreated wastewater (WW) is mixed with AS. In this process, particulate and soluble organic matter contained in the WW absorbs onto the AS flocs [
3]. The high-rate biological contactor (HBC) is a new primary treatment process that removes particulate organic matter and a portion of the soluble organic matter by biosorption [
4]. This process operates with a short hydraulic retention time (HT) and low dissolved oxygen (DO) concentration to affect enhanced carbon diversion to anaerobic digestion for increased methane gas production, and reduced aeration for carbon oxidation in the secondary liquid-phase process. High biomethane potential (BMP) values that were found for the float in a pilot dissolved air flotation (DAF) unit separating raw municipal WW contacted with trickling filter/solids contact (TF/SC) waste activated sludge (WAS) as a biosorbent revealed that oxidation of the sorbed organic matter was at a minimum. However, the WW treatment operating parameters, such as DO, HT, and WAS total suspended solids concentrations, did not show a good correlation with the organic removal performance. In the prior study, EPS was not measured.
It is hypothesized that EPS contained in WAS has a large influence on the biosorption process because of the crucial role that it plays in the biosorption of heavy metals [
5], and because it has been shown that properties of the AS have a larger influence on biosorption than WW properties [
3]. Given the “sticky” nature of EPS, it is assumed here that a higher EPS concentration leads to more biosorption. The objectives of this study were to evaluate the relationship between EPS concentration and organic matter sorption and which component of the EPS has the dominant effect for a TF/SC WAS.
2. Materials and Methods
2.1. Procedure for Biosorption
WW and WAS were collected no more than 2 h prior to utilization in biosorption experiments and were stored at ambient temperature during transport. Samples were taken at a municipal wastewater treatment plant (WWTP) on the island of O‘ahu. The plant is a partial secondary treatment plant using a TF/SC process with a design annual average flow of 38 million gallons per day (MGD). Presently, the plant is treating 13 MGD to the secondary treatment level.
Bench-scale biosorption with DAF separation experiments were conducted in the same manner as previous studies where WAS and raw WW were mixed in an aerated contactor for 30 min while the DO was maintained at 1.0 mg L
−1. Following the contact period, the solids and liquids were separated via the DAF process [
6]. The bench-scale experiments are designed to mimic the HBC process. WW and WAS influent and DAF effluent were analyzed to determine the effects of biosorption. Total chemical oxygen demand (TCOD), soluble COD (sCOD, which is 1.5 μm filtered and thus includes colloids), and the truly soluble COD which is filtered and flocculated (ffCOD, which excludes colloids) were analyzed using Standard Method (SM) 5220D, and total suspended solids (TSS) were measured based on SM 2540D [
7]. Colloidal organic fraction (cCOD) and normalized biosorption were calculated according to previous studies [
6]. Statistical analyses, including
t-test, r
2, and ANOVA, were conducted using the Data Analysis ToolPak Add-in in Microsoft Excel.
2.2. Procedure for Extracellular Polymeric Substances Extraction and Characterization
The TF/SC WAS samples were analyzed for EPS fractionation so that biosorption could be related to the EPS fractions. The cation exchange resin (CER) method [
8] was used to extract EPS from the WAS and the float, and extractions were run for 0.75, 4, and 24 h. The CER method was selected due to its ubiquity in the literature and its relatively high yields compared to other extraction methods. The extracted EPS was characterized and measured using colorimetric methods in the literature [
9,
10] for concentrations of proteins [
11], carbohydrates [
12], humic acids [
8], DNA using a Qubit 4 Fluorometer, uronic acids [
13], and lipids [
14].
To produce EPS values that could be compared, measured weights of centrifuged WAS pellets from known volumes of WAS were added to the set volume of extraction solutions such that EPS could be expressed in mg EPS per l of WAS and as mg EPS per g WAS TSS (or VSS).
3. Results and Discussion
3.1. Waste Activated Sludge Total Suspended Solids Impact on Biosorption Performance
Eleven bench-scale biosorption experiments were conducted on different days using samples of raw wastewater and TF/SC WAS.
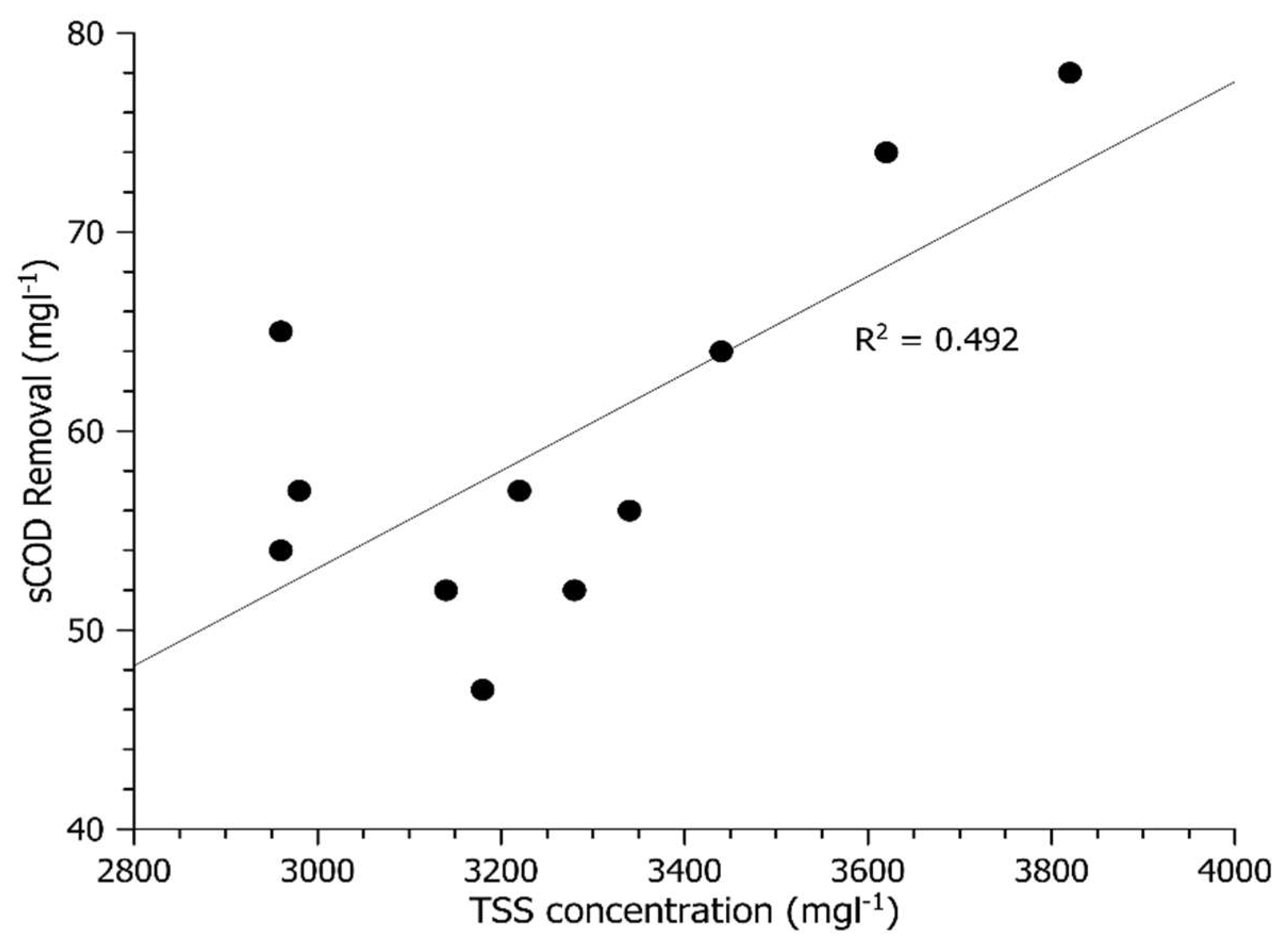
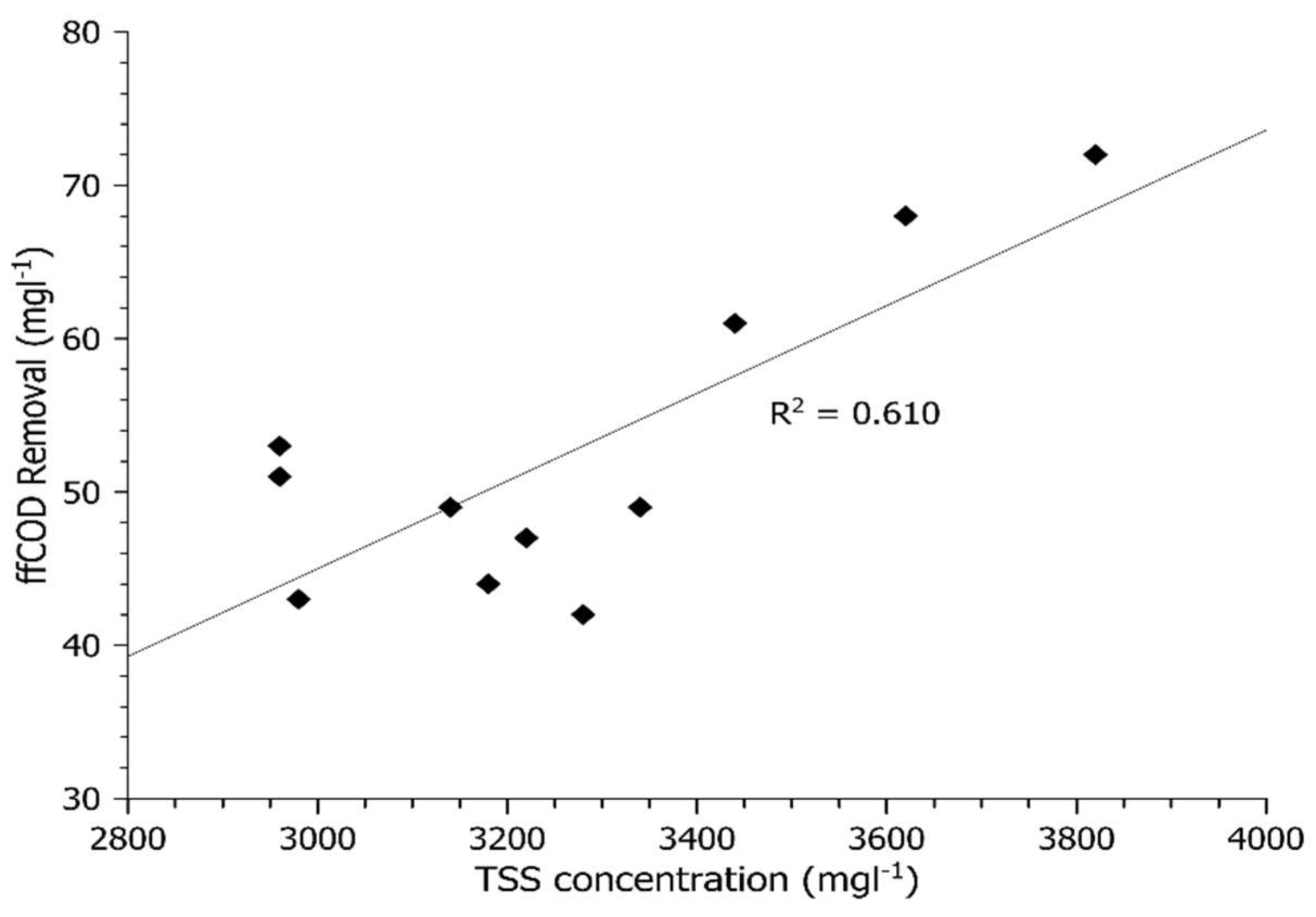
Figure 1 and
Figure 2 show sCOD and ffCOD biosorption in relation to the WAS TSS concentration, respectively. The data show increased sCOD biosorption with larger amounts of WAS TSS. These results were aligned with the pilot and bench-scale HBC experiments in the previous study [
4]. The r-squared value for ffCOD removal (r
2 = 0.61) in
Figure 2 is higher than that for sCOD (
Figure 1), indicating a better correlation with truly soluble COD. According to Modin et al. [
15], operating a reactor under a low DO concentration environment can promote storage by the microorganisms. Since the bench-scale tests were conducted with DO = 1 mg L
−1 and the ffCOD is the truly soluble fraction of organic matter, this suggests that the sCOD removal mechanism in the experiments is more soluble organic matter uptake/storage rather than sorption.
Figure 1 and
Figure 2 show that between about 45 and 75 mg L
−1 of COD can be sorbed in an HBC with a 30-min contact time.
3.2. Extracellular Polymeric Substances Extraction Times
In these experiments, the lipid concentrations in the extracted EPS were all less than 1 mg L
−1. Therefore, the lipid concentrations were assumed to be negligible, similar to other studies [
16]. The sum of the concentrations of the other measured fractions, namely proteins (P), carbohydrates (C), humic acids (H), DNA (D), and uronic acids (U), was therefore considered to represent the total EPS concentration (Equation (1)).
In accordance with Frolund et al. [
17], a longer extraction time can extract more EPS.
Table 1 shows the composition of the TF/SC WAS EPS for extraction times of 0.75, 4, and 24-h. The data represent the average +/− standard deviations for analyses of the 11 samples of WAS used in the experiments. Proteins, carbohydrates, and humic acids made up most of the total EPS, while DNA and uronic acids constitute only minor fractions. This is in agreement with other literature [
10]. The average total EPS concentrations were 90 mg g
−1 VSS, 158 mg g
−1 VSS, and 316 mg g
−1 VSS for the extraction times of 0.75 h, 4 h, and 24 h, respectively. The total EPS concentration for the 24 h extraction time was two times greater than for the extraction time of 4 h and almost four times greater than for the extraction time of 0.75 h. It is noted that most studies in the literature utilize a 0.75-h extraction time which clearly under-represents the total EPS. The data also revealed that as the extraction time increased, the fraction of proteins decreased and the fraction of humic acids increased, while carbohydrates, DNA, and uronic acids remained relatively constant. This could indicate that humic acids are extracted more slowly than other EPS components.
High levels of DNA in the EPS extract would indicate cellular lysis during the extraction process [
18]. Based on a review by Liu and Fang [
19], the DNA in the EPS for municipal WWTP ranges from 0 to 25 mg g
−1 of sludge VSS depending on the extraction method. The TF/SC WAS VSS used herein ranged from 2740 mg L
−1 to 3220 mg L
−1, with an average of 2940 mg L
−1. Since the DNA in all EPS extracted for this part of the study was between 2 and 10 mg g
−1 of the sludge VSS and the DNA ratios in the total EPS for all thee extracted times remained small (2%–3%), it is concluded that none to minimal cellular rapture occurred during the extractions. The 24-h extraction time was able to extract significantly more EPS than the shorter times, representing 31.6% of the mass of the VSS. Other additional extraction methods may be required to achieve the extraction of more EPS to obtain the estimated 50% of VSS represented by EPS.
3.3. Extracellular Polymeric Substances versus cCOD Removal
Table 2 presents the influent and effluent cCOD concentrations for the eleven biosorption experiments. The average influent TCOD and cCOD were 289 mg L
−1 and 35 mg L
−1, respectively. The data show an average of 25% sorption of cCOD. No correlation was observed between cCOD removals and the various EPS components or total EPS concentrations. This observation differs from the high-rate activated sludge (HAS) process, a process that is also promoted for carbon diversion. Jimenez et al. [
20] found that bioflocculation of cCOD and particulate COD (pCOD) increased with an increase in EPS content of the mixed liquor. In that study, total organic carbon (TOC) concentrations were assumed to represent the EPS.
3.4. Extracellular Polymeric Substances versus sCOD and ffCOD Removals
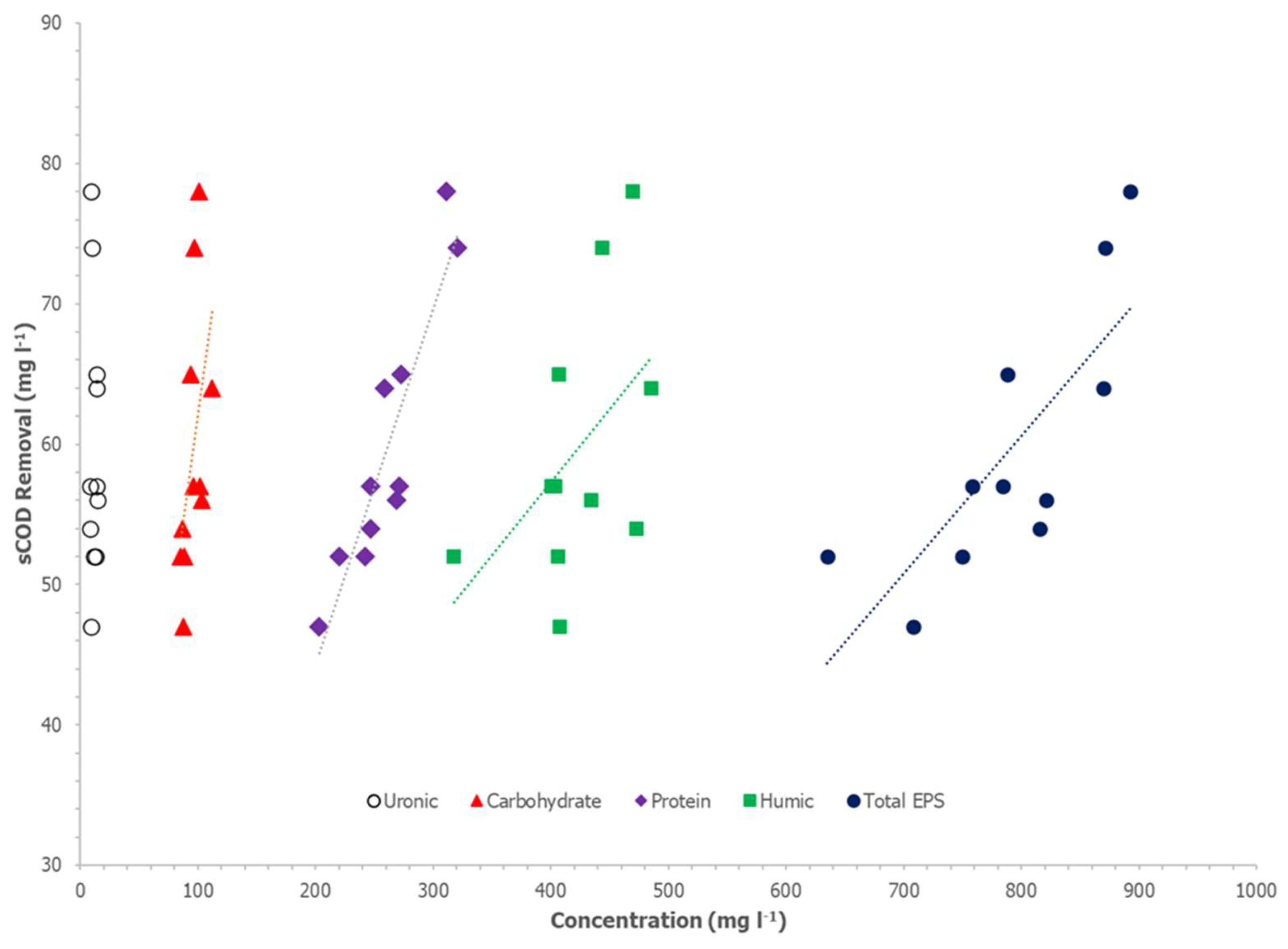
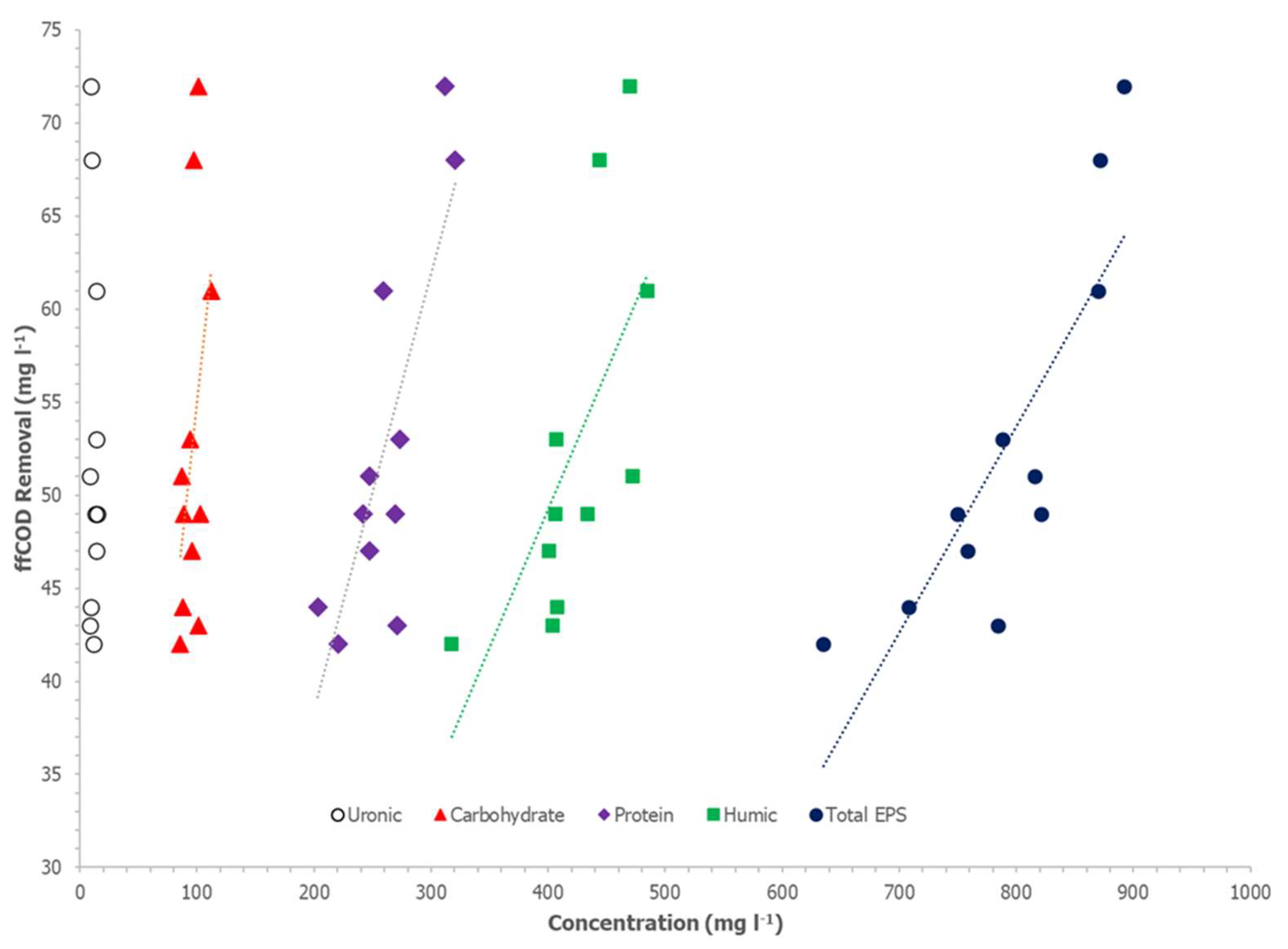
Table 3 and
Figure 3 and
Figure 4 show sCOD and ffCOD sorption in relation to various EPS components and total EPS concentrations. For sCOD, biosorption removal is significantly correlated to the total EPS and the proteins fraction of the EPS. The r-squared values for the total EPS and protein concentrations with respect to sCOD removals were 0.61 and 0.85, respectively, and the ANOVA significance F values for both variables were less than 0.01. Humic acids, carbohydrates, and uronic acids did not show a significant correlation with sCOD biosorption. Biosorption of ffCOD was similarly correlated with the total EPS and proteins concentrations in the WAS. The r-squared values for the total EPS and protein compositions were 0.66 and 0.70, respectively, and the ANOVA significance F values were less than 0.01. Similar to sCOD biosorption, humic acids, carbohydrates, and uronic acids did not show a significant correlation with ffCOD biosorption.
According to Parks et al. [
21], proteins in the EPS influence the surface and bulk properties of AS flocs. Thus, they influence the flocculation, dewaterability, and digestibility of the AS. The results herein suggest that the proteins in the EPS also influence the biosorption capability of the WAS. Based on the data here, measurement of just the amount of proteins in the extracted EPS of the WAS may provide an accurate estimation of sCOD and/or ffCOD biosorption.
3.5. Soluble Microbial Products and Extracellular Polymeric Substances in Influent and Effluent
The quantities of proteins, carbohydrates, humic acids, DNA, and uronic acids in WW and DAF underflow (effluent) were measured without any extraction to represent SMP. It is assumed that any EPS on the small quantity of suspended solids in WW and DAF effluent is negligible. It was desired to investigate the movement of EPS and SMP during the biosorption tests.
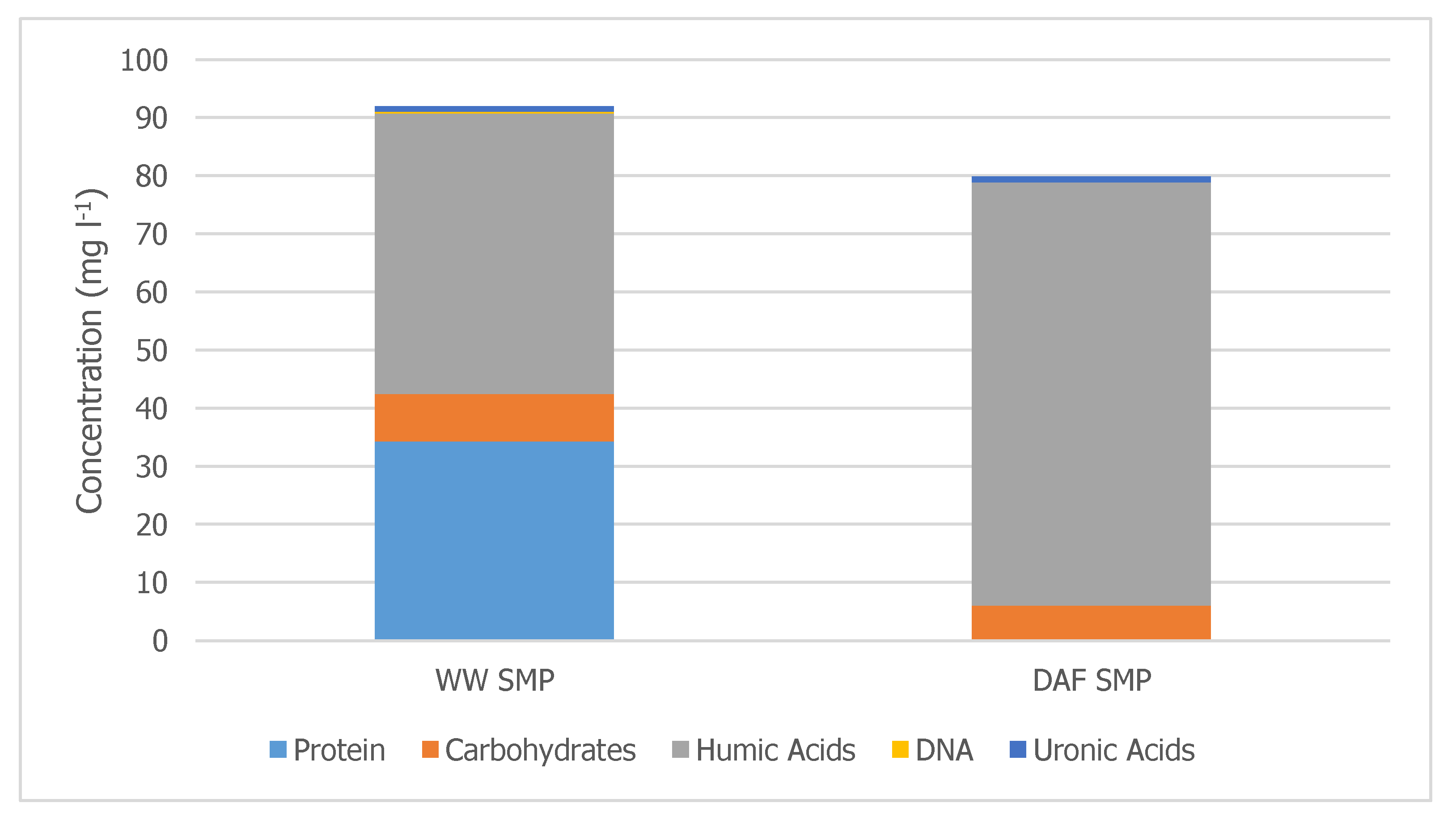
Figure 5 shows that 34 mg L
−1 of WW proteins and 2 mg L
−1 of WW carbohydrates were biosorbed. The data also show that the concentration of DAF underflow humic acids increased by 25 mg L
−1. Additional data are needed to confirm this observation and determine a mechanism.
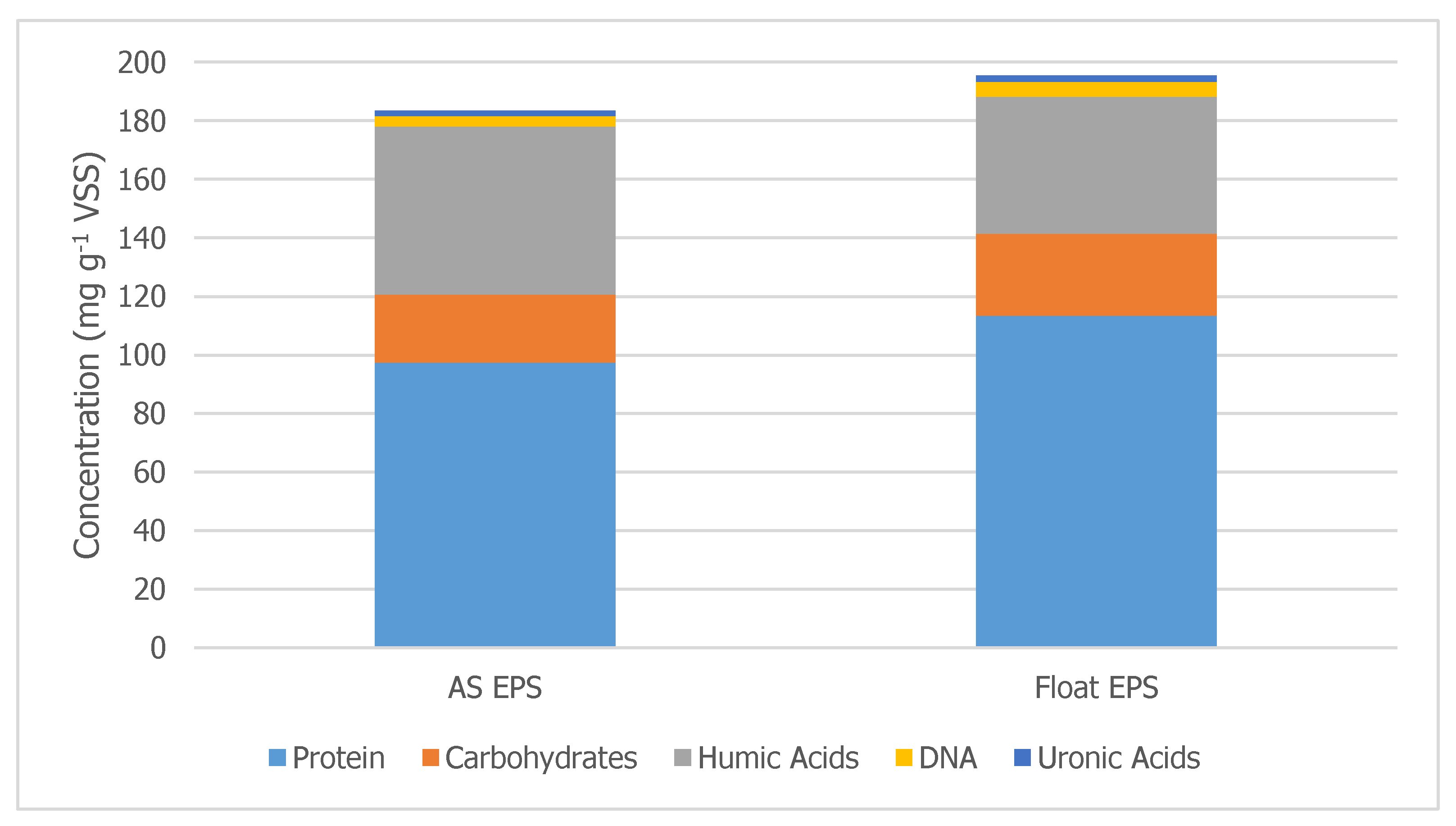
Figure 6 compares the EPS composition of TF/SC WAS and DAF float. A 0.75-h extraction time was utilized to extract EPS from the WAS and the DAF float. The float contained increased amounts of proteins and carbohydrates compared to the WAS, indicating sorption. Proteins increased by 16 mg g
−1 VSS. It is noted that the humic acid fraction of the Float EPS was smaller than that of the WAS EPS. The increase in the humic acids in the DAF underflow may be due to the desorption of humic acids from the WAS being measured as SMP, and/or some biological mechanism for humic acid production. The mechanism for this transformation requires further investigation.
4. Conclusions
The experiments herein have shown that extending CER extraction time increases the amount of EPS extracted. The 24-h extraction time extracted 3.5 times more EPS than the 0.75-h extraction time, which is the commonly used extraction time in the literature. This was true for all of the main EPS components, including carbohydrates, proteins, humic acids, and DNA, except uronic acids. Lipid concentrations were undetectable in the CER-extracted material. The experiments also indicate that humic acids are extracted more slowly than other EPS components. The amount of DNA in the total EPS for all thee extraction times remained low (2–3%) which indicated that minimal cellular rapture occurred during the extraction. The literature estimates that EPS may represent about 50% of the VSS in AS. With the CER extraction time of 45 min, the EPS extracted herein was only 9% of the VSS, and with a 24-h extraction time, the EPS extracted was almost 32%. Additional extraction time or different additional extraction methods may be necessary in order to approach the 50% of VSS estimation.
The assumption that larger WAS EPS concentrations lead to greater organic matter removal during biosorption has been confirmed. The results show that between 45 and 75 mg L−1 of COD is removed during a 30-min contact time. Experiments here have shown that the biosorption of sCOD and ffCOD are statistically correlated to, and could therefore be predicted by the total quantity of EPS and/or the protein fraction of EPS in the TF/SC WAS in contact with WW. The correlations comparing the total EPS and/or protein fraction of EPS in the TS/SC WAS with sCOD and ffCOD removals were better than using the WAS TSS concentrations. However, no correlation was found between EPS concentrations and cCOD removal.
During the biosorption process, the protein fraction of the SMP was absorbed or stored by the cells. Though the process, it was suspected that the humic acids were partly desorbed, which may be due to the mixing in the aeration stage. Further investigation is needed to determine why more humic acids were found in the DAF effluent.
Author Contributions
Conceptualization, T.P.W. and R.W.B.J.; methodology, T.U., J.S., B.H., T.P.W., and R.W.B.J.; validation, T.P.W. and R.W.B.J.; formal analysis, J.S., T.P.W., and R.W.B.J.; investigation, T.U., J.S., and B.H.; writing—original draft preparation, J.S. and T.P.W.; writing—review and editing, T.P.W. and R.W.B.J.; visualization, J.S., T.P.W., and R.W.B.J.; supervision, T.P.W. and R.W.B.J.; project administration, R.W.B.J.; funding acquisition, R.W.B.J. All authors have read and agreed to the published version of the manuscript.
Funding
This research received funding from R. M. Towill Corporation.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
References
- Sheng, G.P.; Yu, H.Q.; Li, X.Y. Extracellular polymeric substances (EPS) of microbial aggregates in biological wastewater treatment systems: A review. Biotechnol. Adv. 2010, 28, 882–894. [Google Scholar] [PubMed]
- Wingender, J.; Neu, T.R.; Flemming, H. Microbial Extracellular Polymeric Substances: Characterization, Structure and Function, 1st ed.; Springer: Berlin/Heidelberg, Germany, 1999; p. 58. [Google Scholar]
- Jorand, F.; Palmgren, R.; Block, J.C.; Nielsen, P.H.; Urbain, V.; Manem, J. Biosorption of wastewater organics by activated sludges. Récents Progrés En Génie Des Procédés 1995, 9, 61–67. [Google Scholar]
- Wong, T.P.; Babcock, R.W., Jr.; Hu, B.; Schneider, J.; Milan, S. High rate biological contactor system using waste activated sludge from trickling filter/solids contact process. Water Sci. Technol. 2020, 81, 2202–2210. [Google Scholar] [PubMed]
- Comte, S.; Guibaud, G.; Baudu, M. Biosorption properties of extracellular polymeric substances (EPS) resulting from activated sludge according to their type: Soluble or bound. Process Biochem. 2006, 41, 815–823. [Google Scholar]
- Schneider, J.; Babcock, R.W., Jr.; Wong, T.P.; Hu, B. Effects of Mixing Ratio, Contact Time and Dissolved Oxygen on Efficiency of Biosorption for Primary Carbon Diversion. Int. J. Water Wastewater Treat 2020, 6, 1–7. [Google Scholar]
- American Public Health Association/Amerian Water Works Association/Water Environment Federation. Standard Methods for the Examination of Water and Wastewater, 20th ed.; American Public Health Association/Amerian Water Works Association/Water Environment Federation: Washington, DC, USA, 1999. [Google Scholar]
- Frolund, B.; Griebe, T.; Nielsen, P.H. Enzymatic activity in the activated-sludge floc matrix. Appl. Microbiol. Biotechnol. 1995, 43, 755–761. [Google Scholar] [PubMed]
- Raunkjaer, K.; Hvitved-Jacobsen, T.; Nielsen, P.H. Measurement of Pools of Protein, Carbohydrate and Lipid in Domestic Wastewater. Water Res. 1994, 28, 251–262. [Google Scholar]
- Liu, H.; Fang, H.H.P. Extraction of extracellular polymeric substances (EPS) of sludges. J. Biotechnol. 2002, 95, 249–256. [Google Scholar] [PubMed]
- Waterborg, J.H.; Matthews, H.R. The Lowry Method for Protein Quantitation. In Methods in Molecular Biology, 1st ed.; Walker, J.M., Ed.; Humana Press: Clifton, NJ, USA, 1984; Volume 1, pp. 1–3. [Google Scholar]
- Gaudy, A.F., Jr. Colorimetric Determination of Protein and Carbohydrate. Ind. Water Wastes 1962, 7, 17–22. [Google Scholar]
- Kintner, P.K., III; Buren, J.P.V. Carbohydrate Interference and Its Correction in Pectin Analysis Using the m-Hydroxydiphenyl Method. J. Food Sci. 1982, 47, 756–759. [Google Scholar]
- Frings, C.S.; Frendley, T.W.; Dunn, R.T.; Queen, C.A. Improved Determination of Total Serum Lipids by the Sulfo-Phospho-Vanillin Reaction. Clin. Chem. 1972, 18, 673–674. [Google Scholar]
- Modin, O.; Persson, F.; Wilen, B.; Hermansson, M. Nonoxidative removal of organics in the activated sludge process. Crit. Rev. Environ. Sci. Technol. 2016, 46, 635–672. [Google Scholar] [PubMed] [Green Version]
- D’Abzac, P.; Bordas, F.; Hullebusch, E.V.; Lens, P.N.L.; Guibaud, G. Extraction of extracellular polymeric substances (EPS) from anaerobic granular sludges: Comparison of chemical and physical extraction protocols. Appl. Microbiol. Biotechnol. 2010, 85, 1589–1599. [Google Scholar] [PubMed]
- Frolund, B.; Palmgren, R.; Keiding, K.; Nielsen, P.H. Extraction of Extracellular Polymers from Activated Sludge using a Cation Exchange Resin. Water Res. 1996, 30, 1749–1758. [Google Scholar]
- Comte, S.; Guibaud, G.; Baudu, M. Relations between extraction protocols for activated sludge extracellular polymeric substances (EPS) and EPS complexation properties Part I. Comparison of the efficiency of eight EPS extraction methods. Enzym. Microb. Technol. 2006, 38, 237–245. [Google Scholar]
- Liu, Y.; Fang, H.H.P. Influences of extracellular polymeric substances (EPS) on flocculation, settling and dewatering of activated sludge. Crit. Rev. Environ. Sci. Technol. 2003, 33, 237–273. [Google Scholar]
- Jimenez, J.; Miller, M.; Bott, C.; Murthy, S.; Clippeleir, H.D.; Wett, B. High-rate activated sludge system for carbon management—Evaluation of crucial process mechanisms and design parameters. Water Res. 2015, 87, 476–482. [Google Scholar] [PubMed]
- Park, C.; Novak, J.T.; Helm, R.F.; Ahn, Y.; Esen, A. Evaluation of the extracellular proteins in full-scale activated sludges. Water Res. 2008, 42, 3879–3889. [Google Scholar] [PubMed]
| Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).