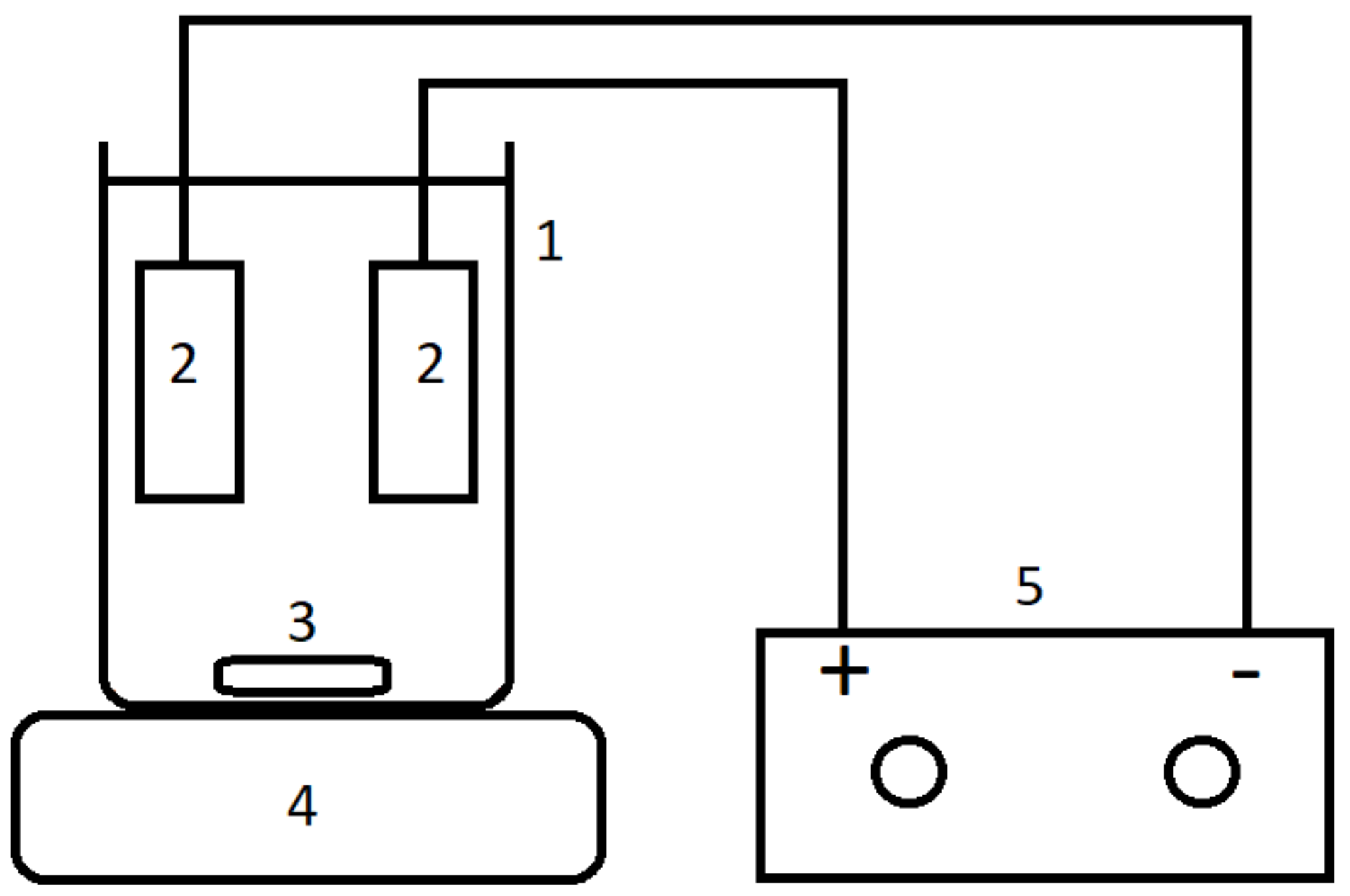
3.1. Precipitation Experiments
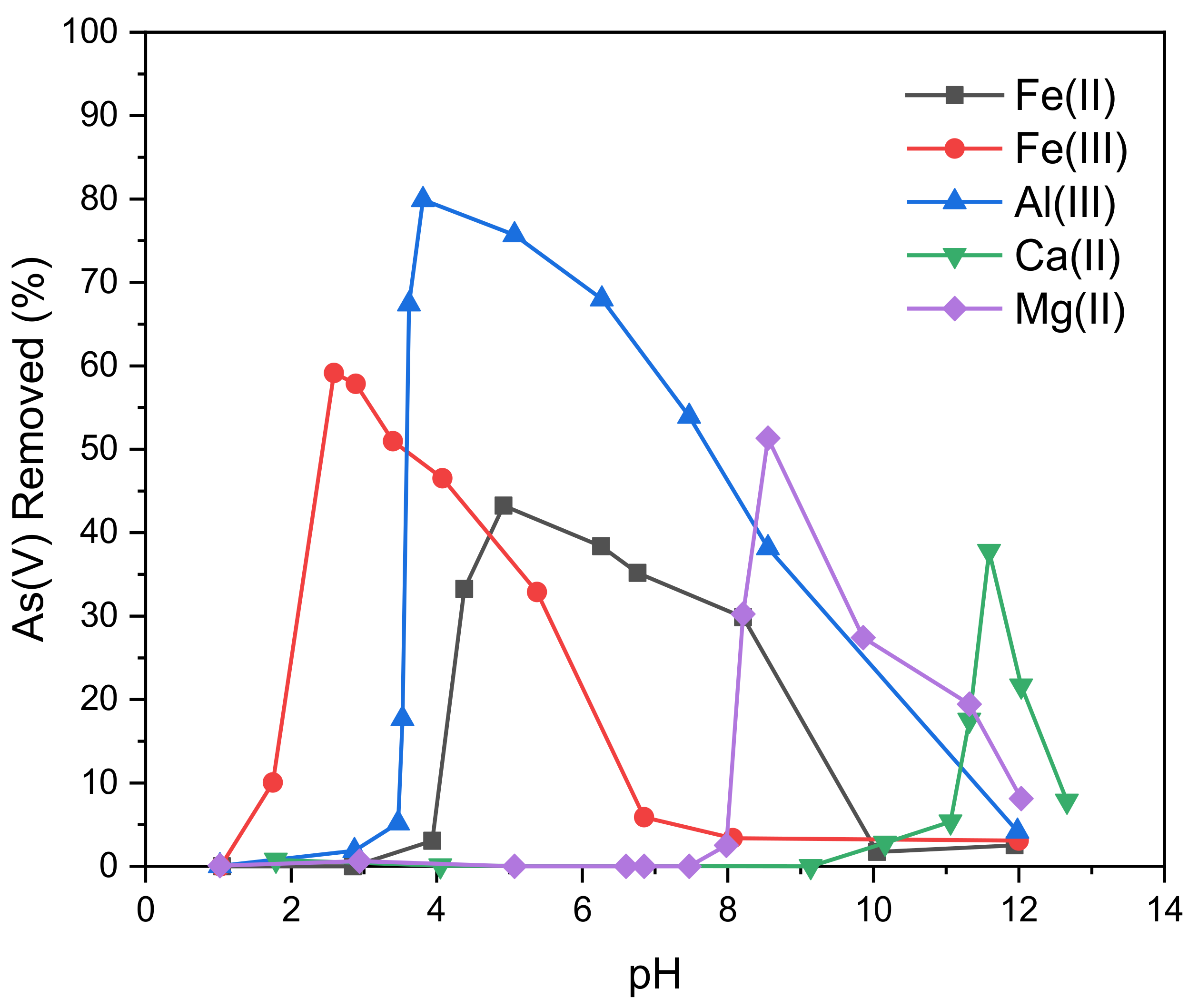
The influence of pH on the removal of As(V) through precipitation with various metal salts was tested, as revealed in
Figure 2. A downward shift in pH was observed after adding the coagulant for FeCl
3 and AlCl
3 and, to a lesser degree, FeCl
2. This effect, which was strongest for FeCl
3 and AlCl
3, is assumed to be due to the precipitation of metal hydroxide. Each of the metals added will form metal hydroxides which will bind OH
− from the solution, thus decreasing the pH. This is less noticeable for FeCl
2, CaCl
2 and MgCl
2 as they bind OH
− at a molar ratio of 2, in comparison to the ratio of 3 for FeCl
3 and AlCl
3.
Arsenic removal through coprecipitation with FeCl
2 took place between pH 4 and 10, but found to be highest between 4.4 and 8.2, where it reached a removal of ~30%. The highest removal of 43% took place at a pH of 4.9. The observed removal of As(V) through precipitation with FeCl
2 was on the lower end, with only CaCl
2 leading to a lower removal. The molar ratio of iron to arsenic was 0.67:1, which was lower than for other materials. Many papers find that optimum pH is significantly higher at a pH of 8.5 [
23]. Cui et al. [
24] found a slightly more similar optimum at a Fe:As molar ratio of 4:1, where complete precipitation took place between 6 and 7 when using NaOH for pH adjustment. The difference in optimum pH might be due to the use of ferrous sulphate, rather than ferrous chloride. Le Berre et al. [
25] showed that, for coprecipitation of As(V), the use of ferric nitrate and ferric sulfate influenced the optimum pH.
Precipitation of arsenic with FeCl
3 took place between pH 1.8 and 8.1 but was optimal at pH 2.6 with a removal of 59%, second only to precipitation with AlCl
3. Considering the molar ratio between Fe(III) and As(V) being 0.67:1, compared to 1.39:1 for Al(III):As(V), FeCl
3 seems to have the highest effectivity per mol. Nishimura et Tozawa [
26] confirmed that optimum precipitation takes place close to pH 3. Le Berre et al. [
25] found that precipitation with iron nitrate was optimal at pH 2 while iron sulfate was optimal at pH 3.
As(V) removal through coprecipitation with AlCl
3 took place from pH 1.8 to up to 8.7, the widest range found among metal chlorides. The removal of As(V) was also found to be the highest of all precipitates, with 80% of arsenic removed at a pH of 3.8. The removal remains high in the range of 3.6–7.5, at over 53%. It is noted that, at 50 mg/L Al(III), this was at a relatively high Al(III):As(V) ratio of 1.39:1. Violante et al. [
8] made precipitates of Al(III) and As(V) at a pH of 4, 7 and 10. In subsequent solubilization of the precipitate in HCl they found that the one generated at pH 4 released the most arsenic while the precipitate generated at pH 10 released the lowest amount. Arai et al. [
9] similarly found that, in the range of pH 4–9.5, adsorption of As(V) on Al
2O
3 was higher at lower pH, though the optimum was slightly under pH 5. Pantuzzo et al. [
10] found that, at pH over ~6.5 the fraction of aluminum and arsenic existing as AlAsO
4 decreased as it transformed into Al(OH)
3. This led to an increase in arsenic concentration, as adsorption onto Al(OH)
3 is less effective in removing As(V) than precipitation.
Removal through precipitation with CaCl
2 only took at a pH over 10.1, reaching over 37% at a pH of 11.6. This was the lowest removal found among metal chlorides used for precipitation, although the molar ratio was higher than for either iron chloride, at 0.93:1 of Ca(II):As(V). Zhang et al. [
12] experimented on arsenic removal from hydrometallurgical industry effluent using calcium, within a range of pH 3–10 the arsenic content in their precipitate went up with an increase in pH. Their tests also showed that, as the pH rises, gypsum transformed into calcium arsenate (Ca
3(AsO
4)
2). Bothe et Brown [
11] tested precipitation of various forms of calcium arsenates in the range of 7.32–12.72 and found that arsenic concentration in the solution went down with an increase in pH. At pH 11.2 only 3 mg/L arsenic with present and above 12 this was less than 0.5 mg/L. They found that this took place because the phases of calcium arsenate, with a rising pH, are Ca
4(OH)
2(AsO
4)
2·4H
2O, Ca
5(AsO
4)
3OH, Caa
3(AsO
4)
2·3
H
2O, Ca
3(AsO
4)
2·4
H
2O, Ca
5H
2(AsO
4)
4·9H
2O and CaHAsO
4·H
2O, with corresponding As:Ca molar ratios of 0.5, 0.6, 0.66, 0.66, 0.8 and 0.8 [
24]. This increase in molar ratio indicates that, at higher pH, more arsenic is bound per unit of calcium, which increases the removal.
Removal of arsenic through precipitation with MgCl
2 took place at a pH of over 8.0. This was highest at pH 8.6, reaching a removal of over 51%. At pH 12.0 this decreased to 8%. The removal of As(V) with MgCl
2 was third best, but molar ratio was the highest at 2.06:1 of Mg(II):As(V). Tresintsi et al. [
13] tested a two-step system to regenerate arsenic loaded FeOOH in a continuous flow system. In the range of pH 10–12 adsorption efficiency of As(V) on MgO was highest at 10 and decreased with increasing pH. Park et al. [
14] found that magnesium starts to precipitate into Mg(OH)
2 starting from a pH of ~6, and continues to being completely precipitated at pH 14. Arsenic was only removed through precipitation within the range of 6~11, however. This was optimal at pH 9~9.5, where arsenic existed in the form of Mg
3(AsO
4)
2. At higher pH that removal of As takes place through the mechanism of adsorption, rather than precipitation, which is less effective than through precipitation. The total removal of As(V), through combined precipitation and adsorption, seemed to take place at a pH of 10–11.
3.2. Electrocoagulation Experiments
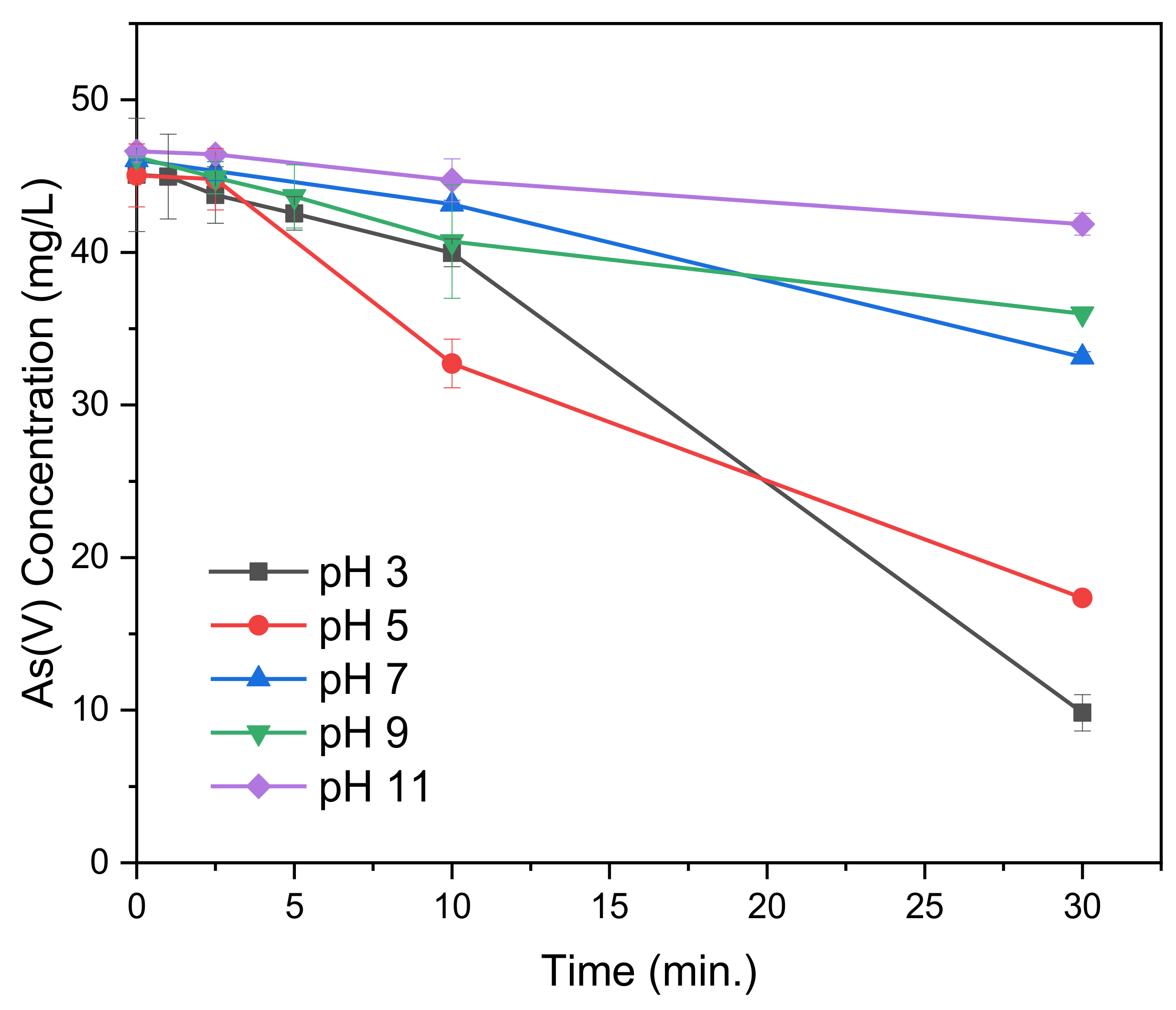
Removal of As(V) through electrocoagulation was tested at varying initial pH (pH
i) and a current of 0.03 A. The results, as listed in
Figure 3, reveal that the As(V) removal decreases with an increase in pH
i. At a pH
i of 3 only 9.8 mg/L As(V) remained, a removal of 78%. In comparison, pH
i of 5, 7, 9 and 11 led to a removal of 62%, 28%, 22% and 10%, respectively. After 10 min, however, it can be observed that the removal with pH
i 5 is higher than that with pH
i 3. The same can be observed for pH
i 7 and 9, where the initial removal at pH
i 9 is higher. This can be explained by the effect of pH during electrocoagulation. Can et al. [
27] found that a lower pH
i was better in the range of pH
i 4–8, but that a pH
i of 2 was the least effective. They found that optimum removal took place at a pH in the range of 8.0–8.5. In this range Fe(II) and Fe(III) exist either predominantly or as a significant fraction as Fe(OH)
2 and Fe(OH)
3 [
28] and As(V) as HAsO
42− [
29]. If the pH
i was set too low, the final pH never reached favorable conditions for adsorption. This explains why in this research the removal after 10 min is higher for higher pH
i, as this initial higher pH is more favorable for adsorption onto Fe(OH)
3. As the pH continues to rise, however, the experiments conducted at lower pH
i start to outperform those at higher pH
i as the former moves into or closer to optimum pH whereas the latter moves away from it.
Due to a current of 0.03 A not being able to achieve full removal of As(V), subsequent tests employ a doubled current of 0.06 A. To test the influence of metal chloride addition on electrocoagulation, either FeCl
2, FeCl
3, AlCl
3, CaCl
2 or MgCl
2 was added, while experiments only using electrocoagulation were performed as a control.
Figure 4 reveals that, as in
Figure 3, without metal addition a lower solution pH
i led to an increased As(V) removal. As mentioned, Can et al. [
27] found that optimum adsorption onto iron hydroxide took place at a pH range of 8.0–8.5.
Table 1 reveals the pH over time during the electrocoagulation process. The pH rises as electrocoagulation takes place, due to water splitting occurring at the cathode, which causes the OH
− concentration to increase. It is revealed that, after 30 min, the pH is closest to this range at a solution pH
i of 3. At solution pHi of 5 and 7 the final pH is 10.3 and 10.5 respectively, which is too high for efficient removal through both Fe(II) and Fe(III), as revealed in
Figure 2. The influence of each metal fraction (i.e., Fe(II), Fe(III), etc.) was tested at concentrations of 30 and 60 mg/L. In comparison, the iron generated during electrocoagulation can be calculated using Faraday’s Law as expressed in Equation (1):
here, w is weight of iron dissolved in grams, I is the current in amperes (0.06 A in this experiment), t the duration in seconds, M the molecular weight of iron (55.845 u), Z is the number of electrons involved in the oxidation/reduction (2 for Fe(II)) and F is Faraday’s constant of 96.485. Through this equation it is calculated that, at a 100% efficiency, 1.04 mg iron is dissolved every minute, with a total of 31.25 mg over 30 min. It is noted that this calculated concentration is only indicative, and the real concentration is affected by other factors. According to Huang et al. [
30] this current efficiency, that is reduced by a resistance layer forming at the electrodes, increases with NaCl concentration. They found that, at a concentration of 1000 mg/L, a current efficiency of 100% was reached. The current research employs a concentration of 750 mg/L, which is slightly lower, so it is suggested that the current efficiency is also high, though not necessarily 100%. Considering the reaction volume of 250 mL, this translates to a concentration of 4.17 mg/L each minute and 125.02 mg/L after 30 min or roughly 2 or 4 times the amount of metal added by metal chloride.
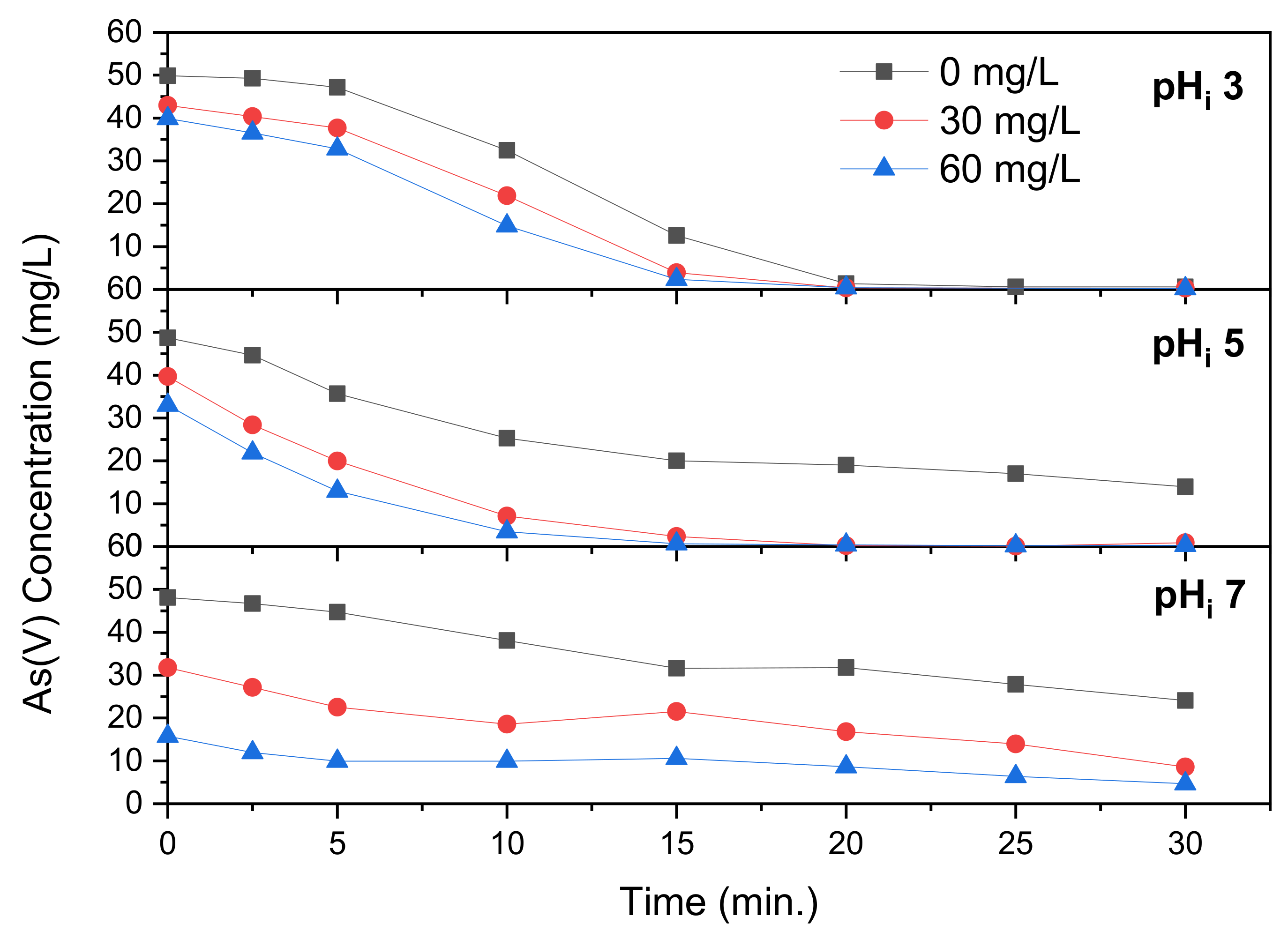
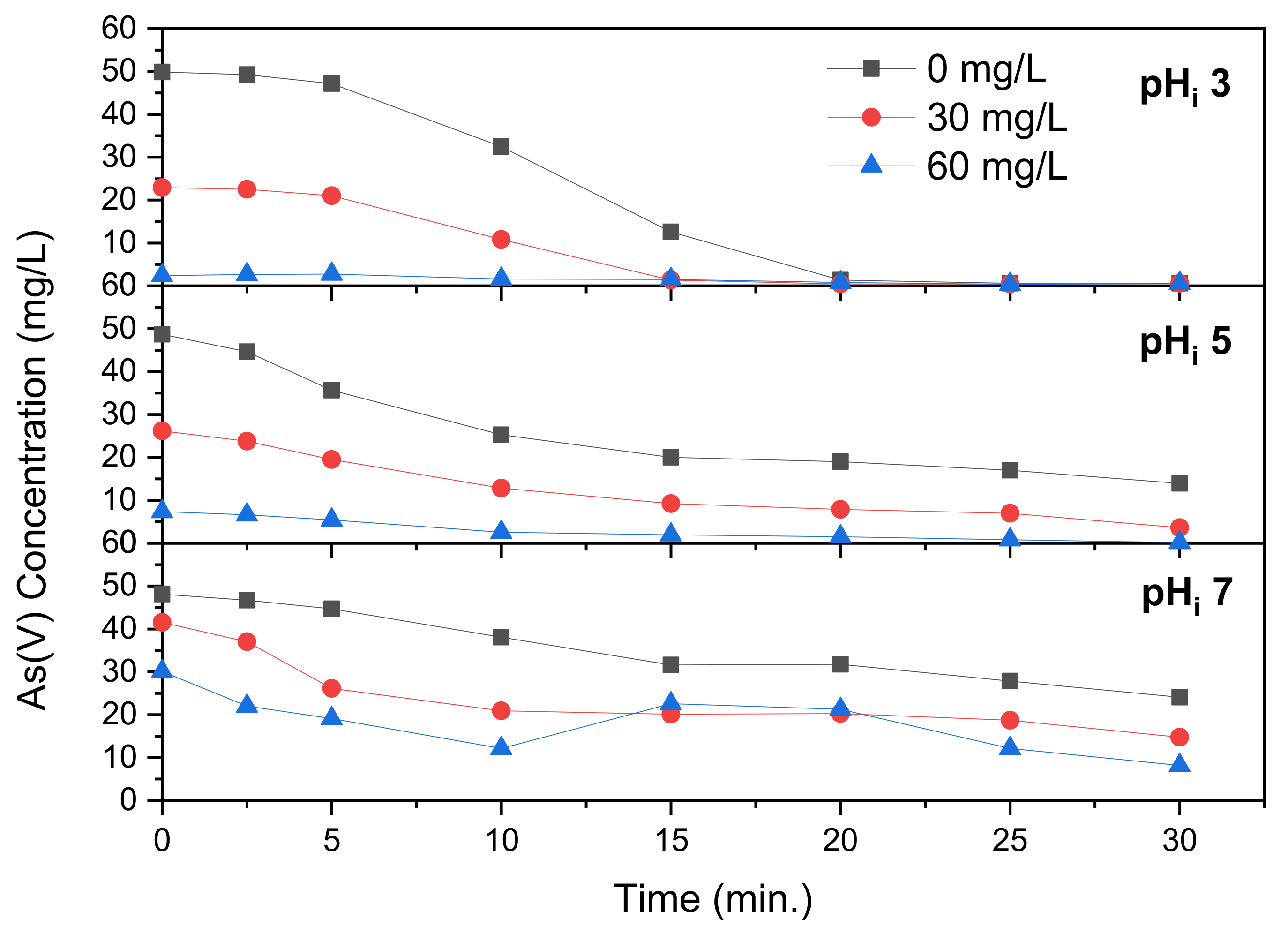
Electrocoagulation of As(V) was performed with addition of FeCl
2, as revealed in
Figure 4. Addition of FeCl
2 is demonstrated in
Figure 4 to increase As(V) removal. According to Ghernaout et al. [
28], Fe(II) is aqueous until a pH of ~5.5, after which it starts to precipitate into Fe(OH)
2, with the remaining soluble fraction transforming to Fe(OH)
+ and Fe(OH)
3− at higher pH.
Figure 4 shows the arsenic concentration over time at a starting pH of 3 at varying FeCl
2 concentration. A limited amount of precipitation, up to 20%, takes place prior to electrocoagulation, which was not witnessed at pH 3 in
Figure 2. It is demonstrated that FeCl
2 addition increases As(V) removal, with near complete removal after 15 min of electrocoagulation time. Complete removal, however, is reached after 20 min for all concentrations of FeCl
2.
Table 1 lists the pH over time where it is revealed that, without addition of FeCl
2, the pH increases from 3.0 to 7.2 over 30 min of electrocoagulation time. At concentrations of 30 and 60 mg/L FeCl
2, the pH ends up at 6.6 and 6.2, respectively. This indicates that FeCl
2 has a pH reducing effect during the electrocoagulation process, even after adjustment prior to the process. This takes place due to the formation of Fe(OH)
2, which binds hydroxide from the solution as it is formed, thus reducing the pH. A larger concentration of Fe(II) is able to bind more OH
− ions. Higher chlorine concentrations have been reported to affect electrocoagulation by increasing the release of aluminum because chlorine ions cause pitting erosion of the resistance layer on the anode [
30]. The effect of additional chlorine from added FeCl
2 must be low, however, due to the large initial NaCl concentration of 750 mg/L. It is demonstrated that, at pH
i 3, FeCl
2 addition does not greatly reduce the time needed for complete removal of As(V). This demonstrates that a pH
i of 3 is too low to benefit enough from FeCl
2 addition.
Figure 4 reveals that at pH
i 5 the influence of FeCl
2 is more pronounced. At this solution pH
i, complete removal is not reached after 30 min (reaching 14.0 mg/L) without FeCl
2 addition but is complete after 20 and 15 min with 30 and 60 mg/L addition, respectively. This demonstrates that addition of FeCl
2 benefits electrocoagulation more at the right solution pH. The final pH without addition of FeCl
2 is 10.3, with 30 and 60 mg/L Fe(II) this is significantly lower at 7.9 and 6.8. This difference is larger than that found at pH
i 3, as the higher pH is more favorable to precipitation of iron hydroxide. At pH
i 7, pictured in
Figure 4, complete As(V) removal does not take place within the runtime of 30 min, reaching 24.1, 8.6 and 4.7 mg/L at Fe(II) concentrations of 0, 30 and 60 mg/L. At these Fe(II) concentrations the final pH reached are 10.5, 10.5 and 10.1, respectively. The final pH without addition is only 0.2 higher than that at pH
i 5 which likely took place due to logarithmic nature of the pH scale, where the high concentration of OH
− leads to a smaller fraction being bound by Fe(II). This also explains the low difference between the final pH after 30 min at varying concentrations of FeCl
2. Majumder et Gupta [
21] tested the effect of iron concentration on the removal of arsenic by electrocoagulation, and showed its significance using a factorial design approach. However, the iron concentrations were not predetermined through chemical addition but calculated as theoretical release during electrocoagulation.
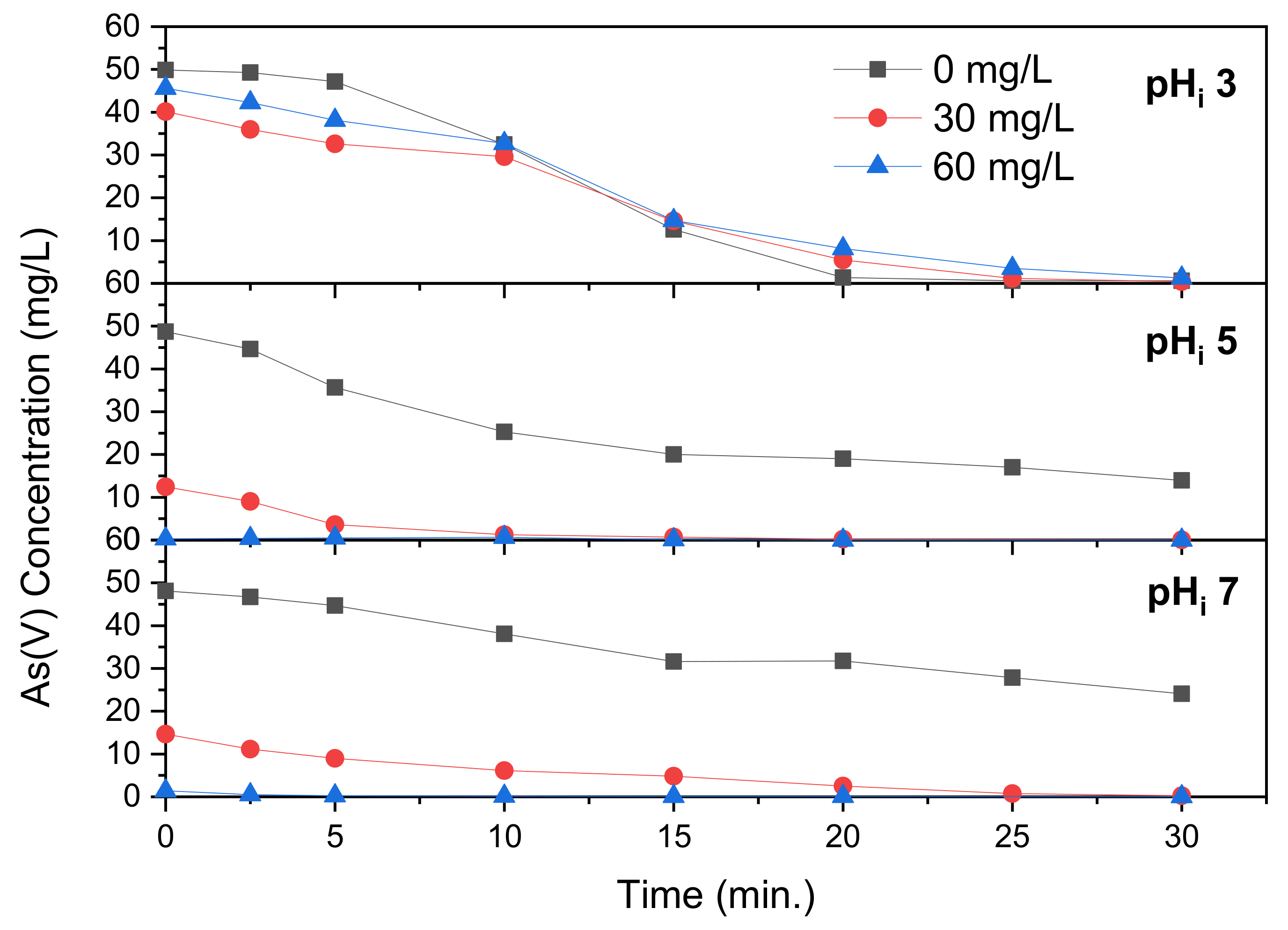
The removal of As(V) during electrocoagulation with addition of FeCl
3 is revealed in
Figure 5. At pH
i 3 the influence of FeCl
3 addition was significant, with precipitation removing the As(V) concentration to 23.0 and 2.4 mg/L at Fe(III) concentrations of 30 and 60 mg/L, respectively. This corresponds with the data discussed in
Section 3.1, where precipitation of As(V) with FeCl
3 was optimal at pH 2.6. Despite the initial precipitation taking place, removal to less than 1 mg/L As(V) takes 20 min when adding either concentration of FeCl
3, whereas this takes 25 min without addition. After 10 min the solution pH had already reached 5.2, at which the As(V) removal through FeCl
3 has already severely decreased, according to
Figure 2. This pH keeps rising, which explains why it takes so long for the As(V) concentration to decrease to less than 1 mg/L. The final pH after 30 min of electrocoagulation time was 7.0 and 6.7 at Fe(III) concentrations of 30 and 60 mg/L, compared to 7.2 without addition. This reveals that, after 30 min, the reduction in pH by addition of FeCl
3 is less pronounced than by addition of FeCl
2, because this pH is more favorable to precipitation of Fe(OH)
2. At pH
i 5, pictured in
Figure 5, the precipitation of As(V) prior to electrocoagulation is lower than at pH
i 3. Complete removal within 30 min only takes place with the addition of 60 mg/L Fe(III). The pH increases to 9.9 and 9.1 at 30 and 60 mg/L, compared to 10.3 mg/L without Fe(III) dosing. As with pH
i 3, this is well above optimum pH for coprecipitation found in
Figure 2. Electrocoagulation with addition of FeCl
3 at pH
i 7 is revealed in
Figure 5. As with FeCl
2 in
Figure 4, complete removal of As(V) is not achieved at this pH
i, with Fe(III) concentrations of 0, 30 and 60 mg/L leading to a final As(V) concentration of 24.1, 14.8 and 8.2 mg/L, respectively. With a dosing of 60 mg/L Fe(III), the As(V) concentration increased after 15 min of electrocoagulation and does not decrease until the same level until after 25 min. The pH, as seen in
Table 1, increases from 8.1 after 10 min to 8.4 after 20 min. This small increase in pH of only 0.3 indicates that this is when transformation of AsFeO
4 to Fe(OH)
3 took place, which binds OH
− ions from the solution and releases arsenic from the precipitate. As mentioned for Fe(II) addition, Majumder et Gupta showed a significantly improved arsenic removal when increasing the iron concentration but this iron resulted from electrocoagulation and was not added chemically, reducing its applicability in comparison to this study.
The removal of As(V) through electrocoagulation with addition of AlCl
3 was tested and revealed in
Figure 6. At pH
i 3 initially As(V) removal is better with addition of AlCl
3, due to precipitation. This precipitation is very low, which corresponds to the data in
Figure 2. After 15 min, however, the As(V) concentration as actually found to be lower without AlCl
3 addition. Whereas it takes 20 min remove As(V) to ~1 mg/L without addition of AlCl
3, this takes 25 and even 30 min at 30 and 60 mg/L, respectively. The pH after 20 min was 4.2 and 3.9 for 30 and 60 mg/L Al(III), respectively. Pantuzzo et al. [
10] showed that this pH is near optimal for precipitation of Al(III) and As(V) to AlAsO
4. In this research, electrocoagulation without addition of AlCl
3 led to a pH of 6.9, a pH that is more favorable to removal with both Fe(II) and Fe(III), according to
Figure 2. This suggests that, at a pH
i of 3, aluminum is less effective than the iron hydroxide generated by electrocoagulation. In addition, due to the binding of OH
− which reduces the pH, it even decreases the efficiency at which As(V) is removed.
Figure 6 reveals the As(V) removal at pH
i 5. Initially most As(V) is removed through precipitation, with only 12.4 and 0.3 mg/L left in the solution prior to electrocoagulation with the addition of 30 and 60 mg/L AlCl
3. It takes 10 min or electrocoagulation with 30 mg/L AlCl
3 to remove As(V) to ~1 mg/L, while this is amount of removal is never achieved at this pH
i without addition of Al(III). The final pH reached is 10.3, 9.4 and 8.0 for Al(III) concentrations of 0, 30 and 60 mg/L, which was not favorable to precipitation with AlCl
3. Pantuzzo et al. [
10] found that, at a pH over 6.5, the fraction of precipitated AlAsO
4 decreased as it transformed to Al(OH)
3, which removes As(V) through adsorption at a lower efficiency.
Figure 6 shows that the As(V) removal at pH
i 7 behaves nearly identical to that at pH
i 5. The main difference is that, at 30 mg/L Al(III), it takes 25 min for As(V) concentration to be less than 1 mg/L. The final solution pH after 30 min was 10.2 and 9.7 at Al(III) concentrations of 30 and 60 mg/L, respectively, significantly higher than at pH
i 5. This similarity reveals that, for both iron and aluminum, a variance in pH does not have a major effect on removal in this high range.
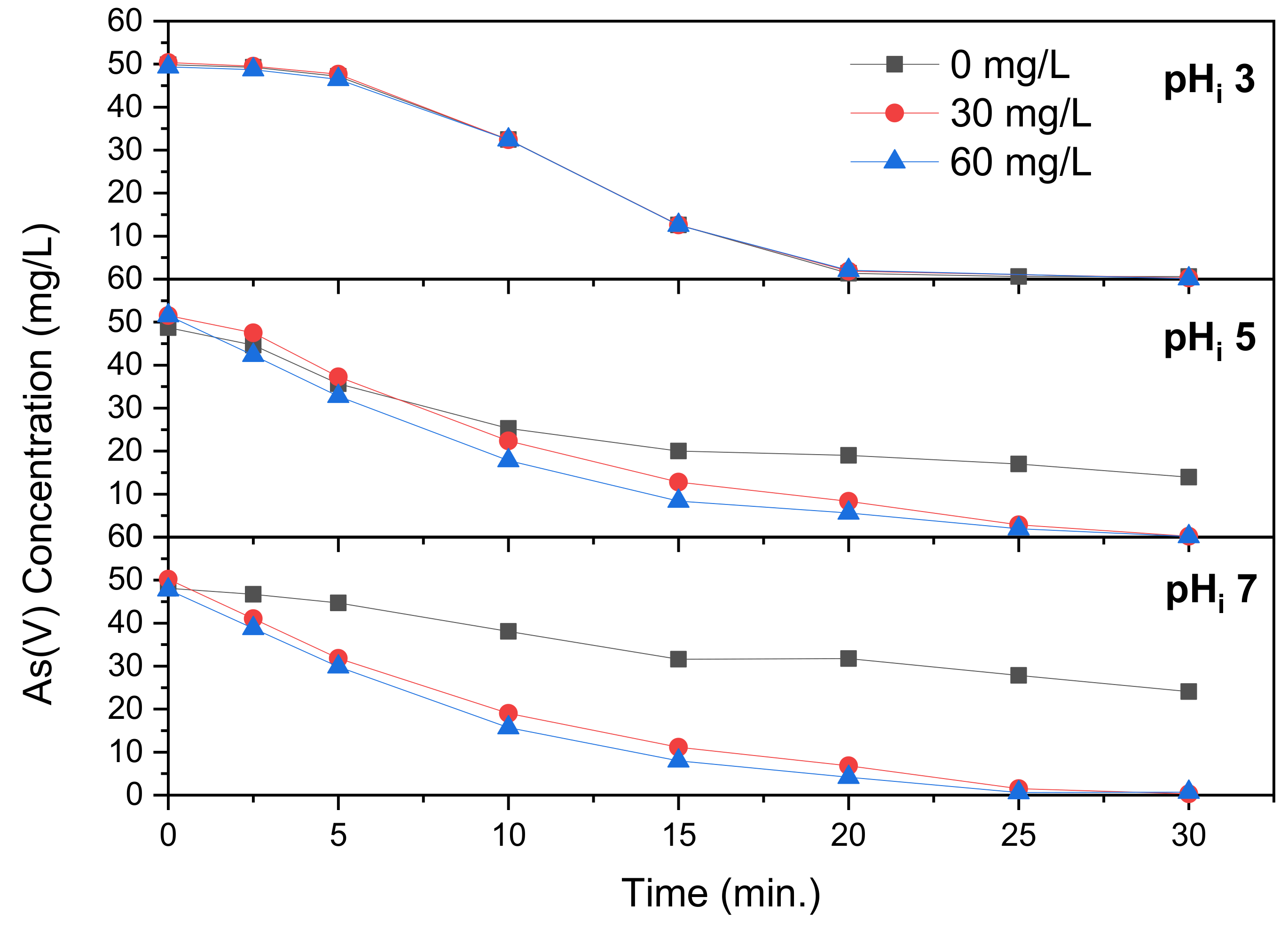
Figure 7 reveals the As(V) removal through electrocoagulation with addition of CaCl
2.
Figure 2 revealed that precipitation of As(V) with Ca(II) requires a pH, and according to literature, the molar ratio of arsenic to calcium increases from 0.5 to 0.8 as pH increases [
31]. At a solution pH
i of 3, revealed in
Figure 7, addition of CaCl
2 has no significant effect on the removal of As(V). The pH after 10 min is 5.9–6.0, seemingly unaffected by CaCl
2 dosage which corresponds to the data in
Figure 2. After 30 min the difference is, while slightly more visible, still relatively small in the range of 6.7–7.2. According to
Figure 2, coprecipitation of As(V) with Ca(II) did not take place at this pH.
Figure 7 reveals the removal of A(V) at solution pH
i 5. The addition of CaCl
2 ensures that complete removal is achieved, although this takes until the 30 min mark. The final pH is, similarly to pH
i 3, mostly unaffected by Ca(II) concentration, ending within a range of 9.9–10.3. According to the data in
Figure 2, only limited precipitation takes place. While the effect of Ca(II) concentration is larger in comparison to pH
i 3, the effect is still small.
Figure 7 reveals the As(V) removal through electrocoagulation with Ca(II) addition at pH
i 7. The As(V) removal with addition of CaCl
2 is nearly identical to that at solution pH
i 5. And similarly to pH
i 5, increasing the Ca(II) concentration from 30 to 60 mg/L has very limited benefit. The final pH was found to be in the range of 10.1–10.5, which is nearly identical to that at pH
i 5, which explains the similarity in As(V) removal. Hu et al. [
20] tested the removal of As(V) through electrocoagulation using aluminum electrodes under influence of varying calcium concentrations, added as CaCl
2. They found that the removal went up with the molar ratio of initial Ca(II) to As(V), but that higher ratios led to only marginal improvements. The effect of pH on this improvement was not discussed, however. Majumder et Gupta [
21] similarly found a significant increase in As removal when adding Ca(II) as CaCO
3.
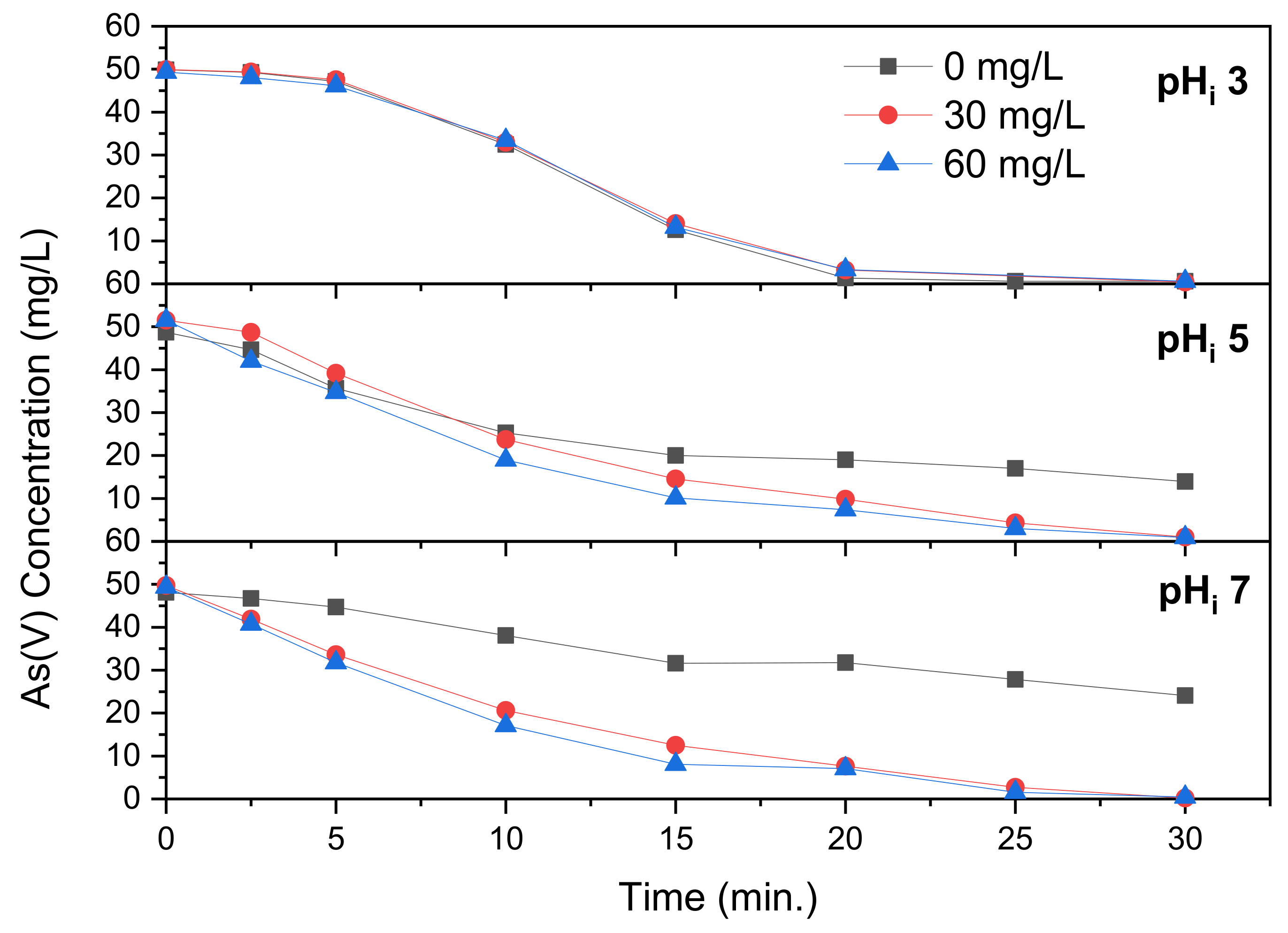
The effect of MgCl
2 addition on the removal of As(V) during electrocoagulation is displayed in
Figure 8. The As(V) concentration during electrocoagulation with addition of MgCl
2 addition is very similar to that with CaCl
2 addition. The pH is nearly identical as well, with a final pH between 6.7–6.8. This, as revealed by the data in
Figure 2, is too low for coprecipitation of arsenic and magnesium to occur.
Figure 8 reveals the As(V) concentration during electrocoagulation at a solution pH
i of 5. The As(V) concentration over time is again very similar that for electrocoagulation with CaCl
2. The final pH reached is 10.1 and 9.6 at a Mg(II) concentration of 30 and 60 mg/L, respectively. This is slightly lower in comparison to the control (at 10.3) and similar to Ca(II) (at 10.2 and 9.9). According to
Figure 2, removal is optimal somewhere between a pH of 8.2 and 9.9 but will take place to a lower degree up to a pH of at least 12.0. According to
Table 1, this optimum pH was reached between 10 and 20 min of electrocoagulation time. In
Figure 8, the As(V) removal at a Mg(II) concentration of 30 mg/L is initially the slowest until 5 min of electrocoagulation time. After 10 min, the As(V) concentration for 30 mg/L Mg(II) is lower in comparison to the control, and this difference proceeds to increase during the reaction. Park et al. [
14] showed that magnesium exists as Mg
2+ up to a pH of 5, after which it starts to form MgOH
+ between pH 5–12.5. Starting at pH 6, Mg
2+ starts to precipitate to Mg
3(AsO
4)
2. Starting at pH 9.5, the total fraction of magnesium precipitated increase, and this precipitate begins to transform into Mg(OH)
2. Above a pH of 11 the precipitate exists completely as Mg(OH)
2 but the amount of magnesium precipitated continues to increase with pH until a solution pH over 13, after which the magnesium is completely precipitated. The arsenic that is freed above pH 9.5, as Mg
3(AsO
4)
2 transforms into Mg(OH)
2, continues to be removed at a lower rate through adsorption onto Mg(OH)
2. The As(V) concentration at solution pH
i 7 is revealed in
Figure 8. As with CaCl
2 addition, the difference between As(V) concentration between pH
i 5 and 7 is very small. In addition, with pH
i 3 and 5, the As(V) concentration at pH
i 7 is very similar to that measured for electrocoagulation with CaCl
2. The final pH is 9.1 and 8.0 at a Mg(II) concentration of 30 and 60 mg/L, respectively. This is lower than the values found at pH
i 5 and meant that, after 20 min of electrocoagulation, the pH actually decreased. This is the same phenomenon that took place with addition of FeCl
2, FeCl
3 and AlCl
3 at lower pH, where formation of metal hydroxide binds OH
− ions from the solution. Mg(OH)
2 forms at a higher pH, which is why this phenomenon is most pronounced at pH
i 7. It is unclear, however, why this mechanism, that becomes more prominent at a higher pH, causes lower final pH than witnessed at pH
i 5. You and Han [
19] tested the effect of dissolved ions on the removal of As(III) through electrocoagulation. While Mg ion concentration up to 10 mg/L increased removal, this was not true at a concentration of 100 mg/L. They found that, at this concentration, while initial As(III) removal was higher in comparison to that at 0 and 1 mg/L Mg(II), after 10 min of reaction time the removal was worse than the control. This may be explained by the pH of the solution, as the researchers investigated the influence of initial pH only before addition of magnesium. As shown in the current research, addition of magnesium enhances the effect of initial pH, in particular at higher pH.
In this study, only mild steel electrodes were employed. A variation in electrode material is certain to affect the reaction. In particular, employing electrodes of varying materials could achieve similar affects to addition of metal chlorides. This has been tested for the combination of iron and zinc [
32] and aluminum and iron [
33]. This would be especially applicable in a more complex setup employing multiple anodes, where the metal ions are released into the solution, of varying metals. In addition, long-term electrode use should be tested as, in this study, electrodes were replaced between tests.
The voltage was measured over time during electrocoagulation, the average value of this is listed in
Table 1. When sorted by metal, the lowest average voltage was found for AlCl
3 (4.9 V), MgCl
2 (5.0 V), CaCl
2 (5.4 V), FeCl
3 (5.6 V), FeCl
2 (5.7 V) and finally without metal addition (5.8 V). Electrical conductivity is highest for aluminum, followed by calcium, magnesium and finally iron [
34]. In addition, 50 mg/L of each metal leads to a highest molarity for magnesium, followed by aluminum, calcium and iron. These combined effects explain the voltages found. The voltage required when using FeCl
3 is slightly lower than FeCl
2 as each metal ion supplies an extra electron. The effect of solution pH
i on conductivity was deemed insignificant, with the lowest voltage being in the order of pH
i 3 (5.2 V), 7 (5.4 V) and 5 (5.5 V). Voltage decreased with an increase in metal concentration at 5.8, 55 and 5.1 V at a concentration of 0, 30 and 60 mg/L, respectively. In previous studies a higher difference was found for the addition of NaCl at lower concentrations [
35] but, due to the background concentration of 750 mg/L NaCl, the influence of added solute is low.
At a neutral pH
i of 7, removal of As(V) was most improved by addition of AlCl
3, CaCl
2 and MgCl
2. Whereas industrial wastewater commonly contains significant concentrations of these metals, seawater exists as a cheap and reliable source for calcium and, particularly, magnesium. According to Turekian [
36], seawater contains a calcium concentration of 411 mg/L and a magnesium concentration of 1290 mg/L. For the minimum found concentration of 30 mg/L this means that seawater contains 13.7 times the required concentration of Ca and 43.0 times the required concentration of Mg. Another added benefit of seawater is the high concentration of Na at 10.8 g/L [
36], which is 36.6 times as high as the concentration used in all tests within this paper (750 mg/L NaCl = 295 mg/L Na). This indicates that even at low dosing of seawater (~2.7%) no further NaCl and MgCl
2 is required, which allows for effective As(V) removal at a pH
i of 7. Seawater is currently already employed for the precipitation of phosphates and ammonium from urine to struvite, which can be utilized as a fertilizer [
37,
38].