Rice Straw Biochar and Magnetic Rice Straw Biochar for Safranin O Adsorption from Aqueous Solution
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Preparation of Biochar and Magnetic Biochar from Rice Straw
2.3. Characterization of Adsorbents
2.4. Batch Adsorption Experiments
3. Results
3.1. Characteristics of Adsorbent Materials
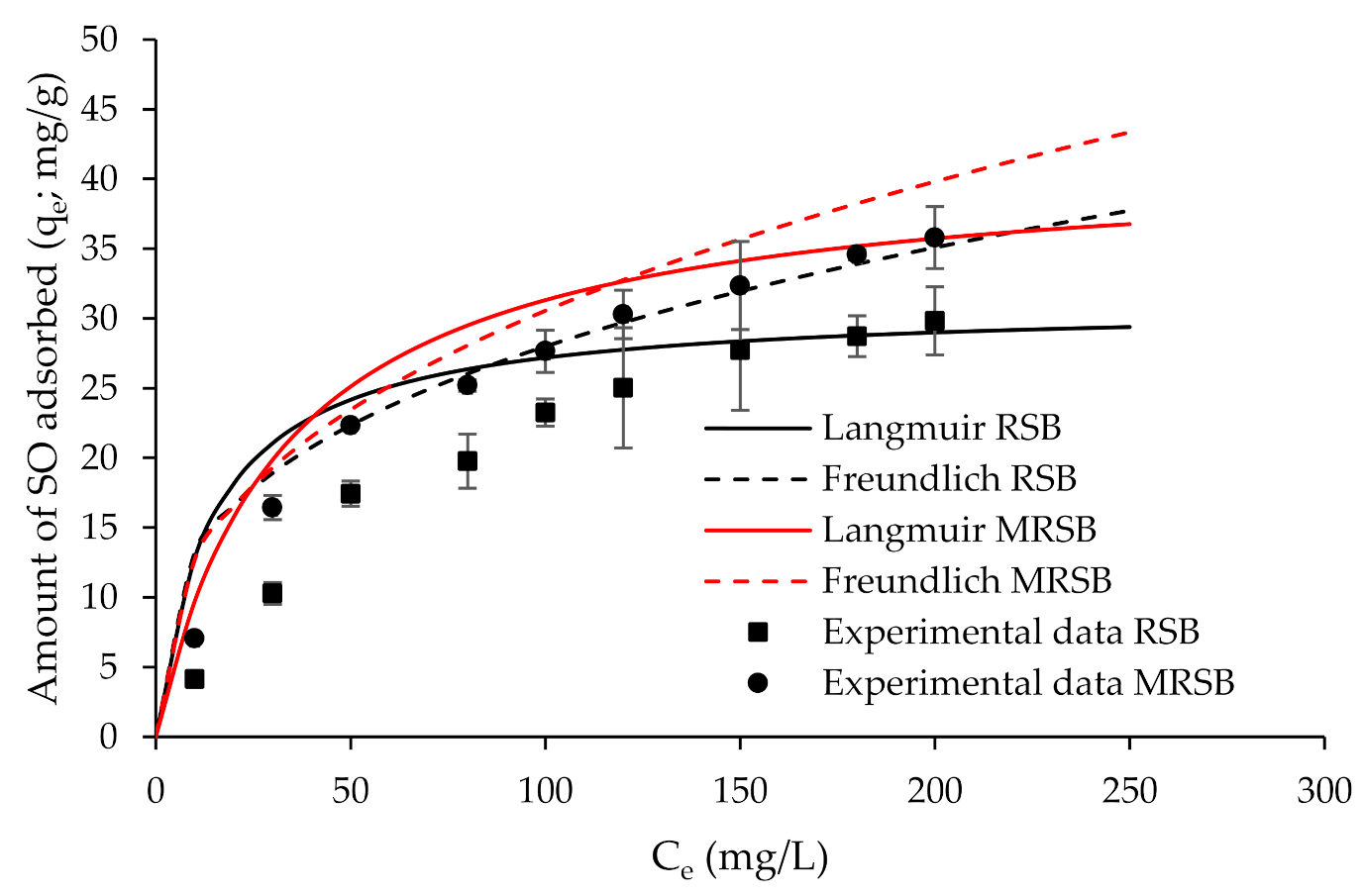
3.2. Adsorption Isotherm Analysis
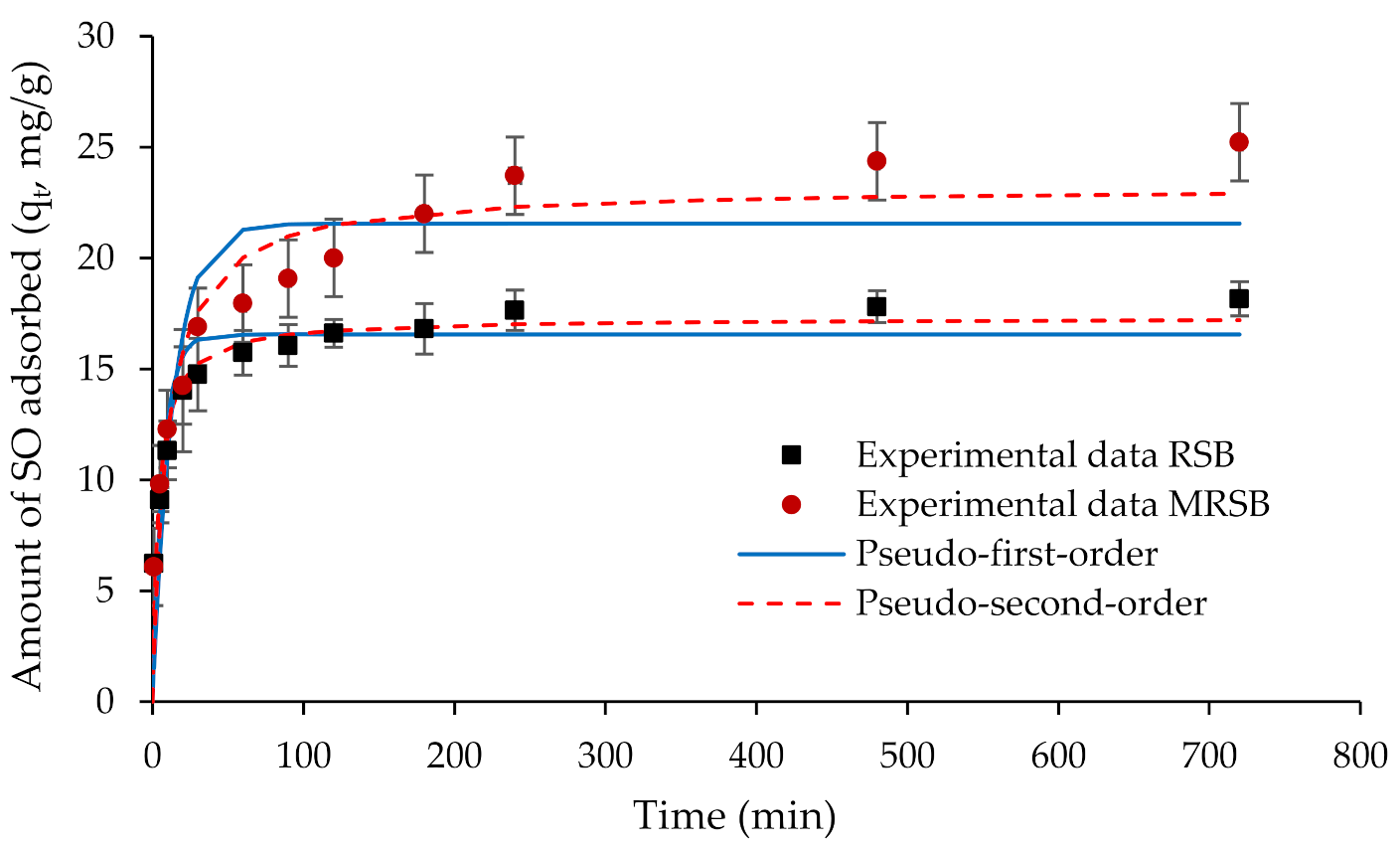
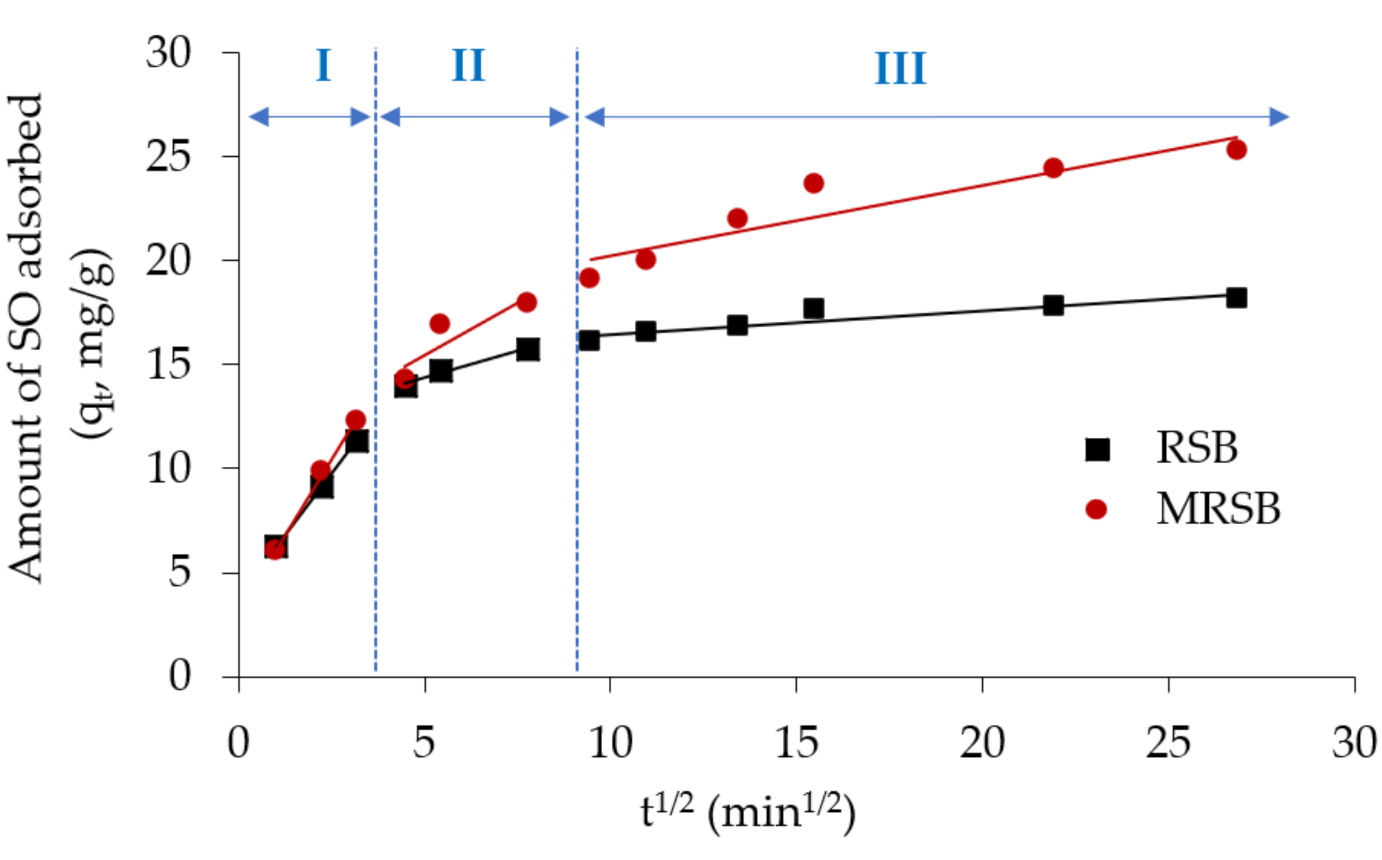
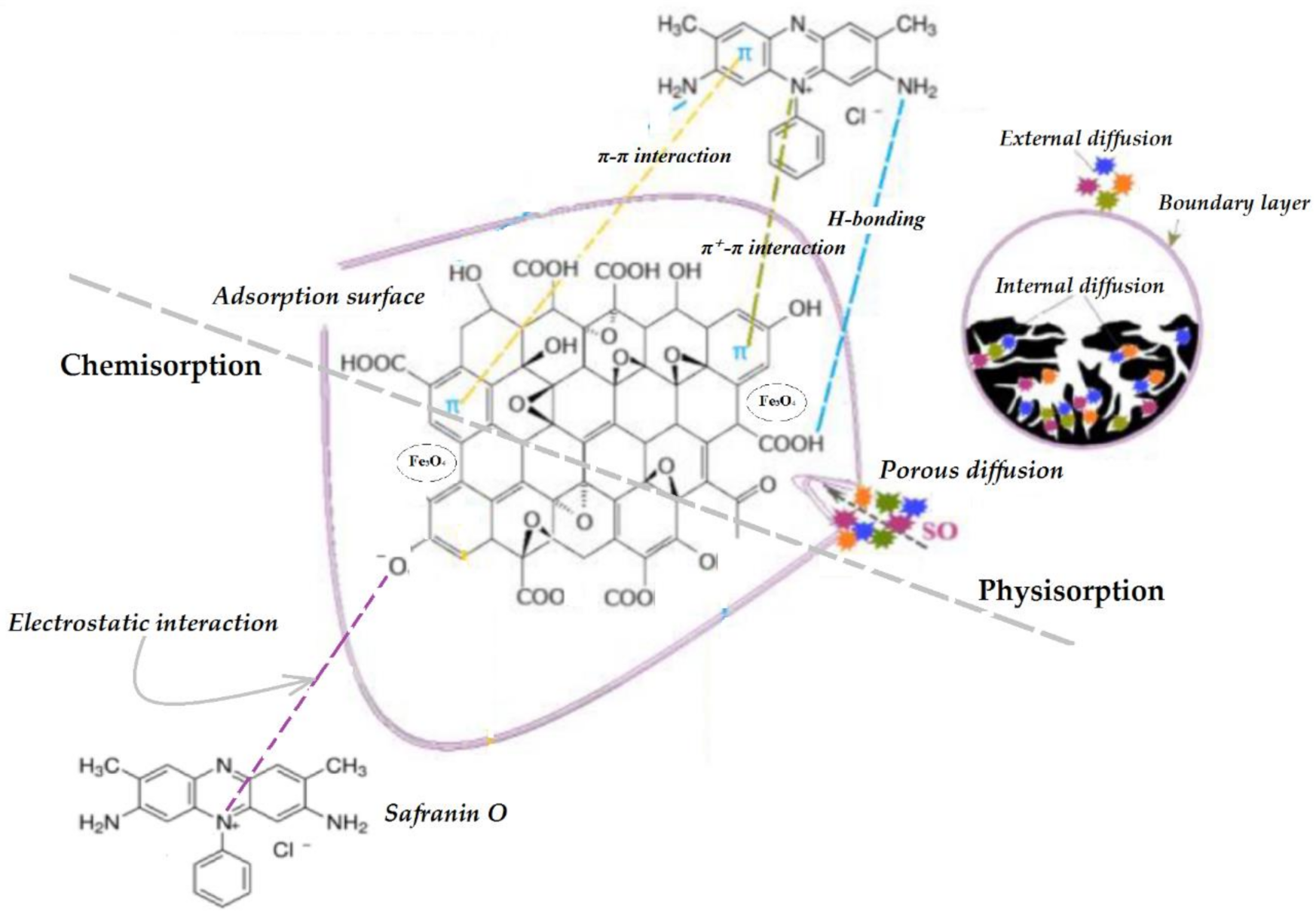
3.3. Adsorption Kinetic Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Malliga, P.; Bela, R.; Shanmugapriya, N. Conversion of textile effluent wastewater into fertilizer using marine cyanobacteria along with different agricultural waste. In Biovalorisation of Wastes to Renewable Chemicals and Biofuels; Elsevier: Amsterdam, The Netherlands, 2020; pp. 87–111. [Google Scholar]
- Verma, A.K.; Dash, R.R.; Bhunia, P. A review on chemical coagulation/flocculation technologies for removal of colour from textile wastewaters. J. Environ. Manag. 2012, 93, 154–168. [Google Scholar] [CrossRef] [PubMed]
- Ejder-Korucu, M.; Gürses, A.; Doǧar, Ç.; Sharma, S.K.; Açιkyιldιz, M. Removal of organic dyes from industrial effluents: An overview of physical and biotechnological applications. Green Chem. Dye. Remov. Wastewater Res. Trends Appl. 2015, 13, 1–34. [Google Scholar]
- Hagemann, N.; Spokas, K.; Schmidt, H.-P.; Kägi, R.; Böhler, M.A.; Bucheli, T.D. Activated Carbon, Biochar and Charcoal: Linkages and Synergies across Pyrogenic Carbon’s ABCs. Water 2018, 10, 182. [Google Scholar] [CrossRef] [Green Version]
- Hadi, P.; Sharma, S.K.; McKay, G. Removal of Dyes from Effluents Using Biowaste-Derived Adsorbents. Green Chem. Dye. Remov. Wastewater Res. Trends Appl. 2015, 6, 139–201. [Google Scholar]
- Phuong, D.; Loc, N.; Miyanishi, T. Efficiency of dye adsorption by biochars produced from residues of two rice varieties, Japanese Koshihikari and Vietnamese IR50404. Desalin. Water Treat. 2019, 165, 333–351. [Google Scholar] [CrossRef]
- Srivatsav, P.; Bhargav, B.S.; Shanmugasundaram, V.; Arun, J.; Gopinath, K.P.; Bhatnagar, A. Biochar as an eco-friendly and economical adsorbent for the removal of colorants (dyes) from aqueous environment: A review. Water 2020, 12, 3561. [Google Scholar] [CrossRef]
- Thines, K.; Abdullah, E.; Mubarak, N.; Ruthiraan, M. Synthesis of magnetic biochar from agricultural waste biomass to enhancing route for waste water and polymer application: A review. Renew. Sust. Energ. Rev. 2017, 67, 257–276. [Google Scholar] [CrossRef]
- Ruthiraan, M.; Mubarak, N.; Abdullah, E.; Khalid, M.; Nizamuddin, S.; Walvekar, R.; Karri, R.R. An overview of magnetic material: Preparation and adsorption removal of heavy metals from wastewater. Magn. Nanostruct. 2019, 15, 131–159. [Google Scholar]
- Shan, D.; Deng, S.; Zhao, T.; Wang, B.; Wang, Y.; Huang, J.; Yu, G.; Winglee, J.; Wiesner, M.R. Preparation of ultrafine magnetic biochar and activated carbon for pharmaceutical adsorption and subsequent degradation by ball milling. J. Hazar. Mater. 2016, 305, 156–163. [Google Scholar] [CrossRef] [Green Version]
- Zhang, J.; Liu, M.; Yang, T.; Yang, K.; Wang, H. A novel magnetic biochar from sewage sludge: Synthesis and its application for the removal of malachite green from wastewater. Water Sci. Tech. 2016, 74, 1971–1979. [Google Scholar] [CrossRef]
- Ren, Z.; Chen, F.; Wang, B.; Song, Z.; Zhou, Z.; Ren, D. Magnetic biochar from alkali-activated rice straw for removal of rhodamine B from aqueous solution. Environ. Eng. Res. 2020, 25, 536–544. [Google Scholar] [CrossRef]
- Yi, Y.; Tu, G.; Ying, G.; Fang, Z.; Tsang, E.P. Magnetic biochar derived from rice straw and stainless steel pickling waste liquor for highly efficient adsorption of crystal violet. Bioresour. Technol. 2021, 341, 125743. [Google Scholar] [CrossRef]
- Ma, H.; Li, J.-B.; Liu, W.-W.; Miao, M.; Cheng, B.-J.; Zhu, S.-W. Novel synthesis of a versatile magnetic adsorbent derived from corncob for dye removal. Bioresour. Technol. 2015, 190, 13–20. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.; Chen, Z.; Lv, S. A novel magnetic biochar efficiently sorbs organic pollutants and phosphate. Bioresour. Technol. 2011, 102, 716–723. [Google Scholar] [CrossRef]
- Sun, P.; Hui, C.; Khan, R.A.; Du, J.; Zhang, Q.; Zhao, Y.-H. Efficient removal of crystal violet using Fe 3 O 4-coated biochar: The role of the Fe 3 O 4 nanoparticles and modeling study their adsorption behavior. Sci. Rep. 2015, 5, 1–12. [Google Scholar]
- Yi, Y.; Huang, Z.; Lu, B.; Xian, J.; Tsang, E.P.; Cheng, W.; Fang, J.; Fang, Z. Magnetic biochar for environmental remediation: A review. Bioresour. Technol. 2020, 298, 122468. [Google Scholar] [CrossRef]
- Li, X.; Wang, C.; Zhang, J.; Liu, J.; Liu, B.; Chen, G. Preparation and application of magnetic biochar in water treatment: A critical review. Sci. Total Environ. 2020, 711, 134847. [Google Scholar] [CrossRef] [PubMed]
- Santhosh, C.; Daneshvar, E.; Tripathi, K.M.; Baltrėnas, P.; Kim, T.; Baltrėnaitė, E.; Bhatnagar, A. Synthesis and characterization of magnetic biochar adsorbents for the removal of Cr (VI) and Acid orange 7 dye from aqueous solution. Environ. Sci. Pollut. Res. 2020, 27, 32874–32887. [Google Scholar] [CrossRef]
- Silva, T.C.F.; VergÜtz, L.; Pacheco, A.A.; Melo, L.F.; Renato, N.S.; Melo, L.C. Characterization and application of magnetic biochar for the removal of phosphorus from water. Acad. Bras. Ciências 2020, 11, 92. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.H.; Pham, T.H.; Nguyen Thi, H.T.; Nguyen, T.N.; Nguyen, M.-V.; Tran Dinh, T.; Nguyen, M.P.; Do, T.Q.; Phuong, T.; Hoang, T.T. Synthesis of iron-modified biochar derived from rice straw and its application to arsenic removal. J. Chem. 2019, 2019, 5295610. [Google Scholar] [CrossRef]
- Zhang, X.; Lv, L.; Qin, Y.; Xu, M.; Jia, X.; Chen, Z. Removal of aqueous Cr (VI) by a magnetic biochar derived from Melia azedarach wood. Bioresour. Technol. 2018, 256, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Essandoh, M.; Wolgemuth, D.; Pittman, C.U.; Mohan, D.; Mlsna, T. Adsorption of metribuzin from aqueous solution using magnetic and nonmagnetic sustainable low-cost biochar adsorbents. Environ. Sci. Pollut. Res. 2017, 24, 4577–4590. [Google Scholar] [CrossRef]
- Wang, S.; Gao, B.; Zimmerman, A.R.; Li, Y.; Ma, L.; Harris, W.G.; Migliaccio, K.W. Removal of arsenic by magnetic biochar prepared from pinewood and natural hematite. Bioresour. Technol. 2015, 175, 391–395. [Google Scholar] [CrossRef] [PubMed]
- Suwunwong, T.; Patho, P.; Choto, P.; Phoungthong, K. Enhancement the rhodamine 6G adsorption property on Fe3O4-composited biochar derived from rice husk. Mate. Res. Express 2020, 7, 025511. [Google Scholar] [CrossRef]
- Agrafioti, E.; Kalderis, D.; Diamadopoulos, E. Ca and Fe modified biochars as adsorbents of arsenic and chromium in aqueous solutions. J. Environ. Manag. 2014, 146, 444–450. [Google Scholar] [CrossRef] [PubMed]
- Hubbe, M.A.; Azizian, S.; Douven, S. Implications of Apparent Pseudo-Second-Order Adsorption Kinetics onto Cellulosic Materials: A Review. BioResources 2019, 3, 14. [Google Scholar] [CrossRef]
- Pourhakkak, P.; Taghizadeh, A.; Taghizadeh, M.; Ghaedi, M.; Haghdoust, S. Fundamentals of adsorption technology. In Interface Science and Technology; Elsevier: Amsterdam, The Netherlands, 2021; Volume 33, pp. 1–70. [Google Scholar]
- Akbarnezhad, A.; Safa, F. Biochar-Based Magnetic Nanocomposite for Dye Removal from Aqueous Solutions: Response Surface Modeling and Kinetic Study. Russ. J. Appl. Chem. 2018, 91, 1856–1866. [Google Scholar] [CrossRef]
- Nizam, N.U.M.; Hanafiah, M.M.; Mahmoudi, E.; Halim, A.A.; Mohammad, A.W. The removal of anionic and cationic dyes from an aqueous solution using biomass-based activated carbon. Sci. Rep. 2021, 11, 1–17. [Google Scholar]
- Lashaki, M.J.; Atkinson, J.D.; Hashisho, Z.; Phillips, J.H.; Anderson, J.E.; Nichols, M. The role of beaded activated carbon’s surface oxygen groups on irreversible adsorption of organic vapors. J. Hazar. Mater. 2016, 317, 284–294. [Google Scholar] [CrossRef]
- Lu, X.; Shao, Y.; Gao, N.; Ding, L. Equilibrium, thermodynamic, and kinetic studies of the adsorption of 2, 4-dichlorophenoxyacetic acid from aqueous solution by MIEX resin. J. Chem. Eng. Data 2015, 60, 1259–1269. [Google Scholar] [CrossRef]
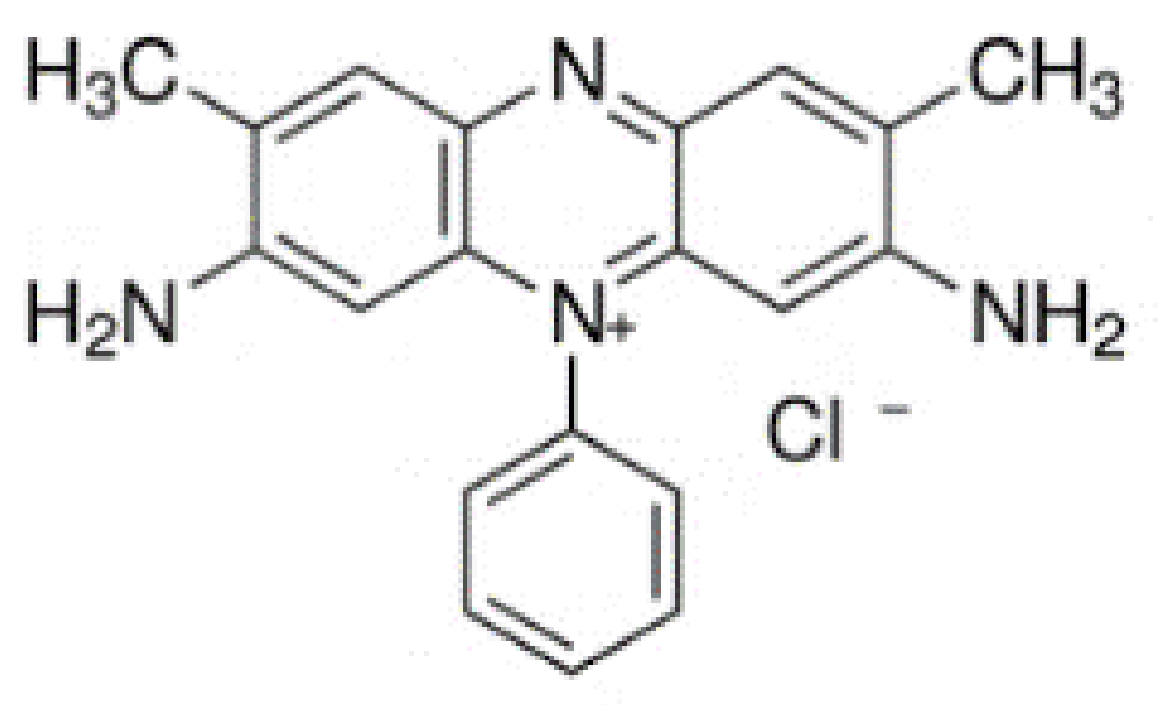
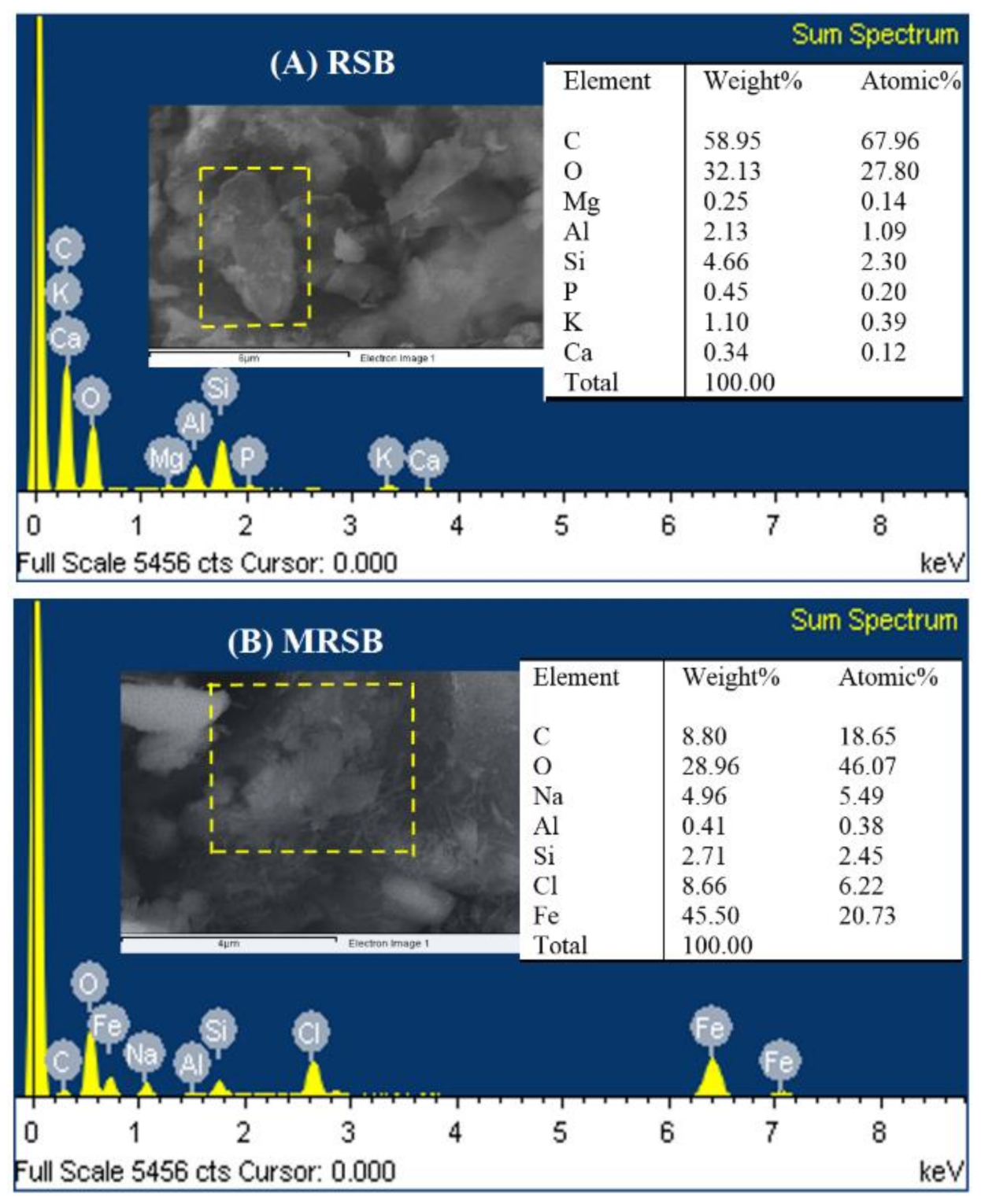
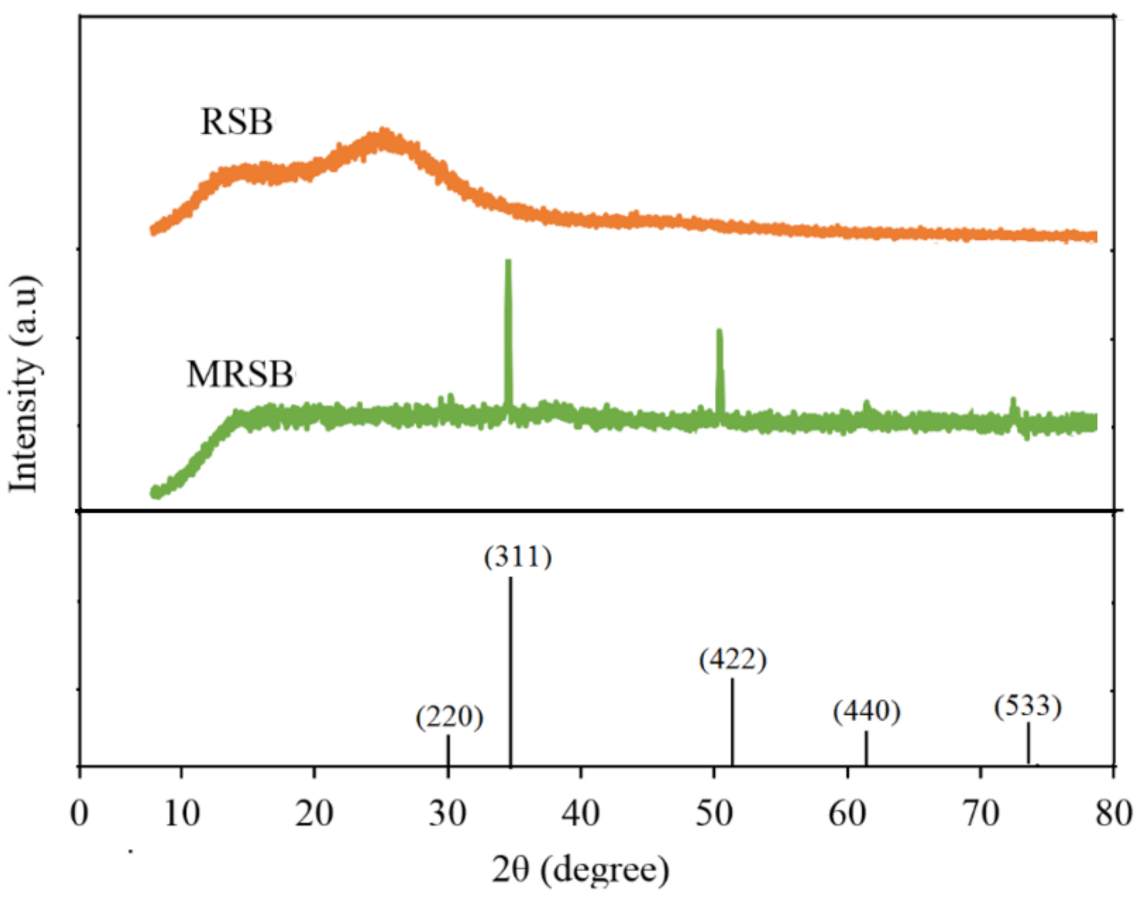
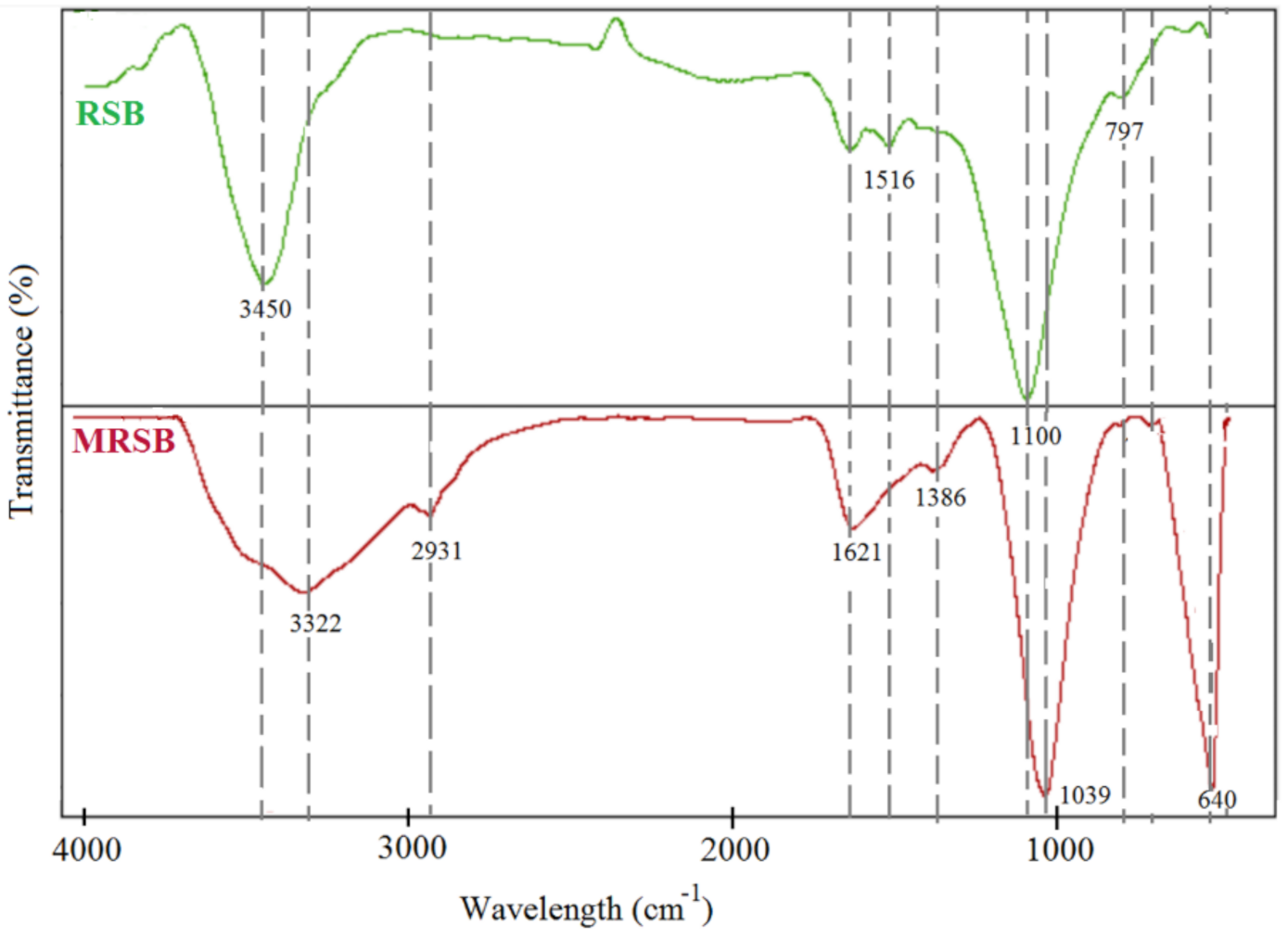

| RSB | MRSB | |
|---|---|---|
| Specific surface area (m2/g) | 269.93 | 337.77 |
| Average particle size (nm) | 2.26 | 1.74 |
| pHpzc | 6.90 | 7.69 |
| Adsorbent | Langmuir | Freundlich | ||||
|---|---|---|---|---|---|---|
| qm | kL | R2 | 1/n | kF | R2 | |
| mg/g | L/mg | (mg/kg)/(mg/L)n | ||||
| RSB | 31.06 | 0.07 | 0.956 | 3.07 | 6.24 | 0.970 |
| MRSB | 41.59 | 0.03 | 0.977 | 2.62 | 5.26 | 0.989 |
| Feedstock | Pyrolytic Temperature (°C) | Technique Used | Contaminants | Effect on Removal Capacity | Ref. |
|---|---|---|---|---|---|
| Corncob | 160 | Hydrothermal carbonization | Methylene blue | Near 1.6 times higher than pristine biochar | [14] |
| Corn stalks | 400 | Chemical co-precipitation | Crystal violet | Near 20 times higher than pristine biochar | [16] |
| Loblolly pine wood | 600 | Pyrolysis of hematite-treated biomass | As (V) | Near 1.6 times higher than pristine biochar | [24] |
| Rice husk | 400, 500 and 600 | Chemical co-precipitation | rhodamine 6G | Higher sorption capability than the pristine biochars | [25] |
| Orange peel | 250, 400 and 700 | Chemical co-precipitation | HOCs and phosphate | Higher sorption capability than the pristine biochars | [15] |
| Rice husk and the organic fraction of municipal solid wastes | 300 | The biomass was impregnated with calcium and iron agents before pyrolysis | As(V) and Cr(VI) | Much better As(V) removal capacity compared to the non-impregnated biochars | [26] |
| Rice straw | 500 | Chemical co-precipitation | Safranin O | Near 1.4 times higher than pristine biochar | This paper |
| Adsorbent | Pseudo-First-Order | Pseudo-Second-Order | ||||||
|---|---|---|---|---|---|---|---|---|
| qe,exp | qe,cal | k1 | R2 | qe,exp | qe,cal | k2 | R2 | |
| mg/g | mg/g | 1/min | mg/g | mg/g | g/mg·min | |||
| RSB | 18.16 | 16.55 | 0.14 | 0.81 | 18.16 | 17.29 | 0.01 | 0.92 |
| MRSB | 25.22 | 21.55 | 0.07 | 0.78 | 25.22 | 23.20 | 0.00 | 0.90 |
| Adsorbent | Intra-Particle Diffusion | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Stage I: t = 1–10 min | Stage II: t = 20–60 min | Stage III: t > 60 min | |||||||
| C1 | kid1 | R2 | C2 | kid2 | R2 | C3 | kid3 | R2 | |
| mg/g | mg/g·min1/2 | mg/g | mg/g·min1/2 | mg/g | mg/g·min1/2 | ||||
| RSB | 3.87 | 2.35 | 0.99 | 11.85 | 0.51 | 0.98 | 15.35 | 0.11 | 0.85 |
| MRSB | 3.24 | 2.88 | 0.99 | 10.37 | 1.02 | 0.80 | 16.89 | 0.34 | 0.84 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Phuong, D.T.M.; Loc, N.X. Rice Straw Biochar and Magnetic Rice Straw Biochar for Safranin O Adsorption from Aqueous Solution. Water 2022, 14, 186. https://doi.org/10.3390/w14020186
Phuong DTM, Loc NX. Rice Straw Biochar and Magnetic Rice Straw Biochar for Safranin O Adsorption from Aqueous Solution. Water. 2022; 14(2):186. https://doi.org/10.3390/w14020186
Chicago/Turabian StylePhuong, Do Thi My, and Nguyen Xuan Loc. 2022. "Rice Straw Biochar and Magnetic Rice Straw Biochar for Safranin O Adsorption from Aqueous Solution" Water 14, no. 2: 186. https://doi.org/10.3390/w14020186
APA StylePhuong, D. T. M., & Loc, N. X. (2022). Rice Straw Biochar and Magnetic Rice Straw Biochar for Safranin O Adsorption from Aqueous Solution. Water, 14(2), 186. https://doi.org/10.3390/w14020186






