Degradation Efficiency Analysis of Sulfadiazine in Water by Ozone/Persulfate Advanced Oxidation Process
Abstract
:1. Introduction
2. Experimental Materials and Methods
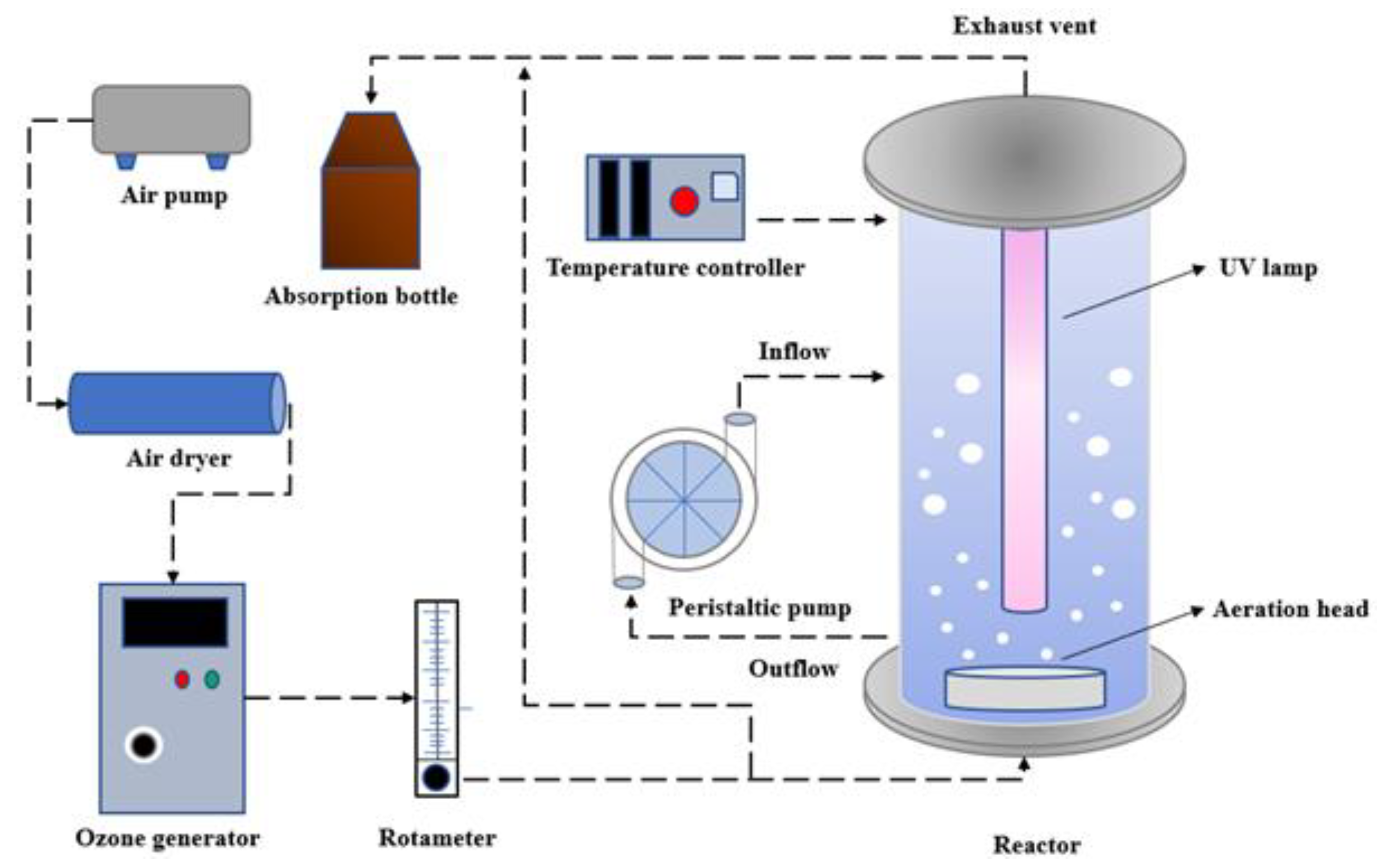
2.1. Experimental Device
2.2. Experimental Materials
2.3. Analytical Method
3. Results and Discussion
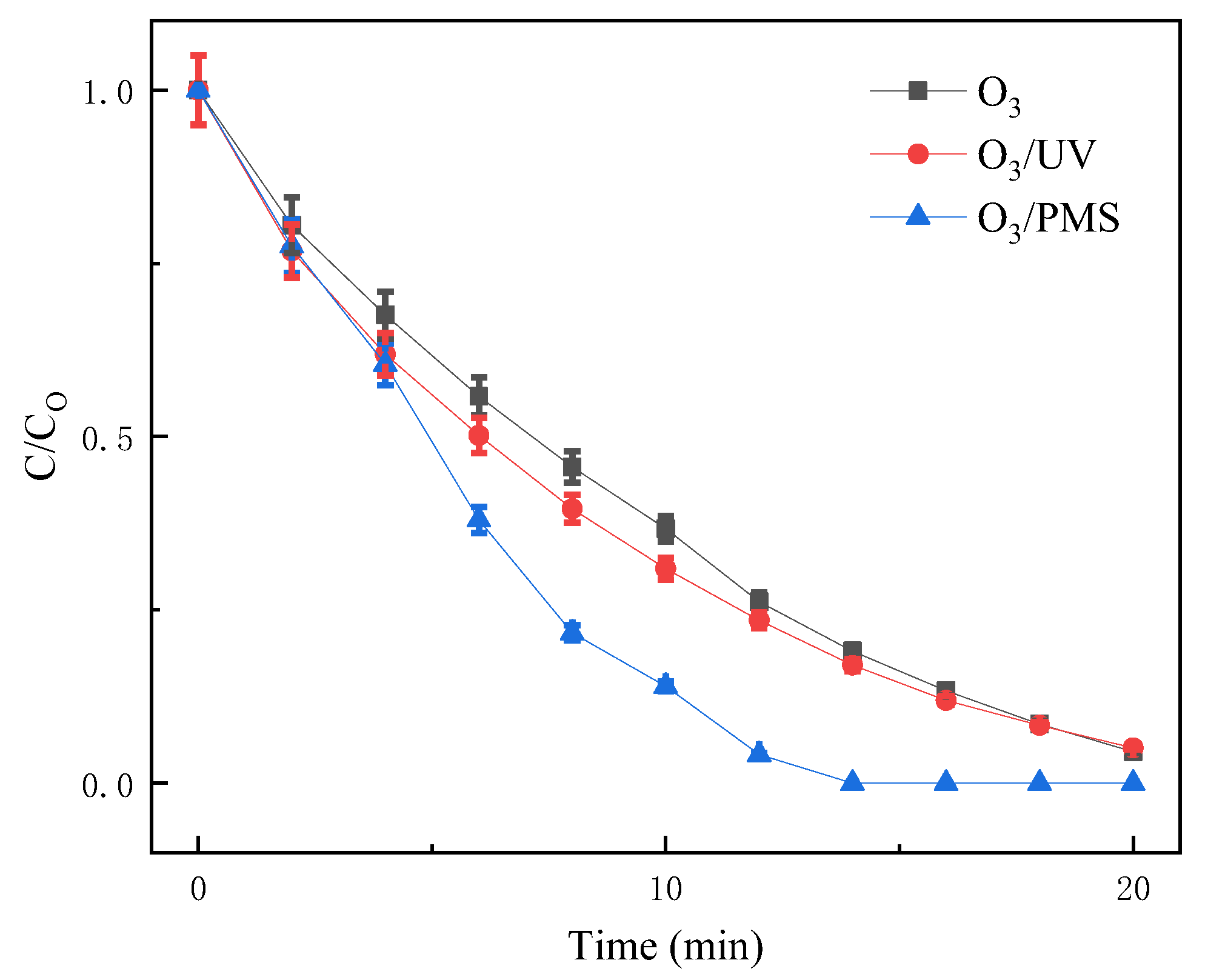
3.1. Comparison of SDZ Degradation Efficiency between O3/PMS and O3 and O3/UV
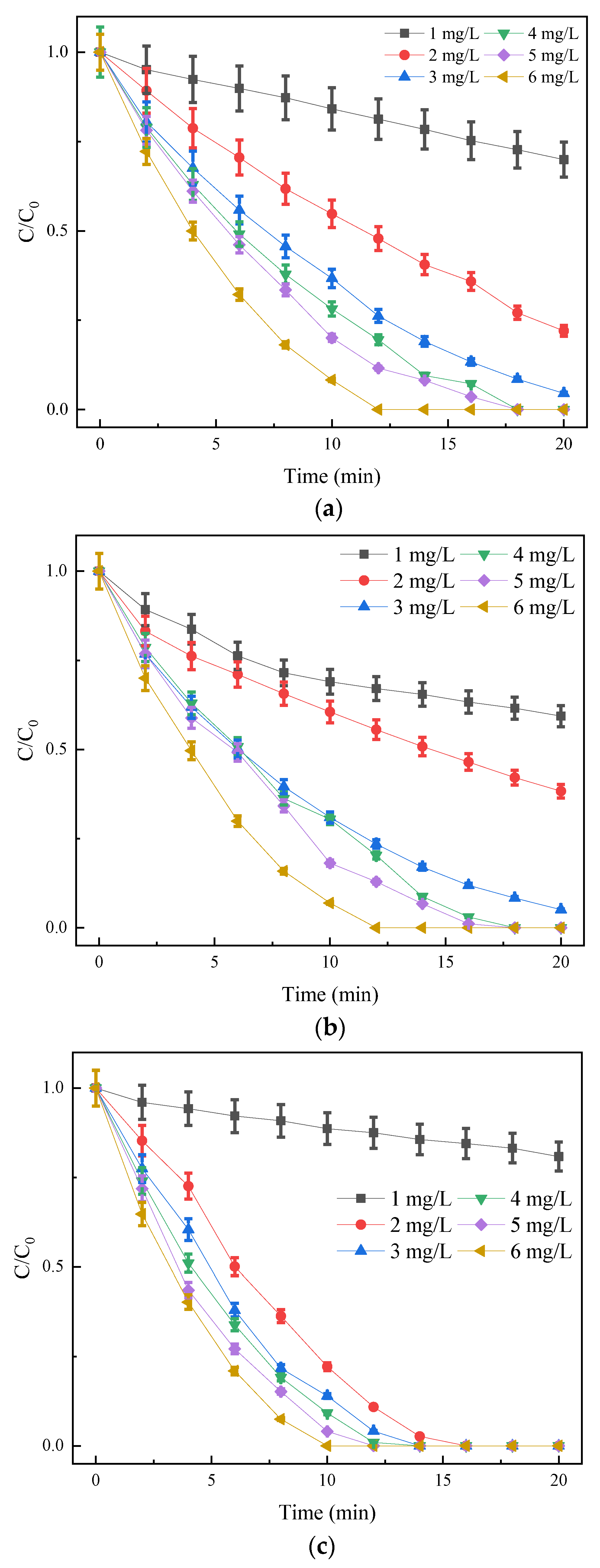
3.2. O3/PMS Single Factor Influence Experiment
3.2.1. Effect of Initial Mass Concentration of SDZ on Degradation Efficiency
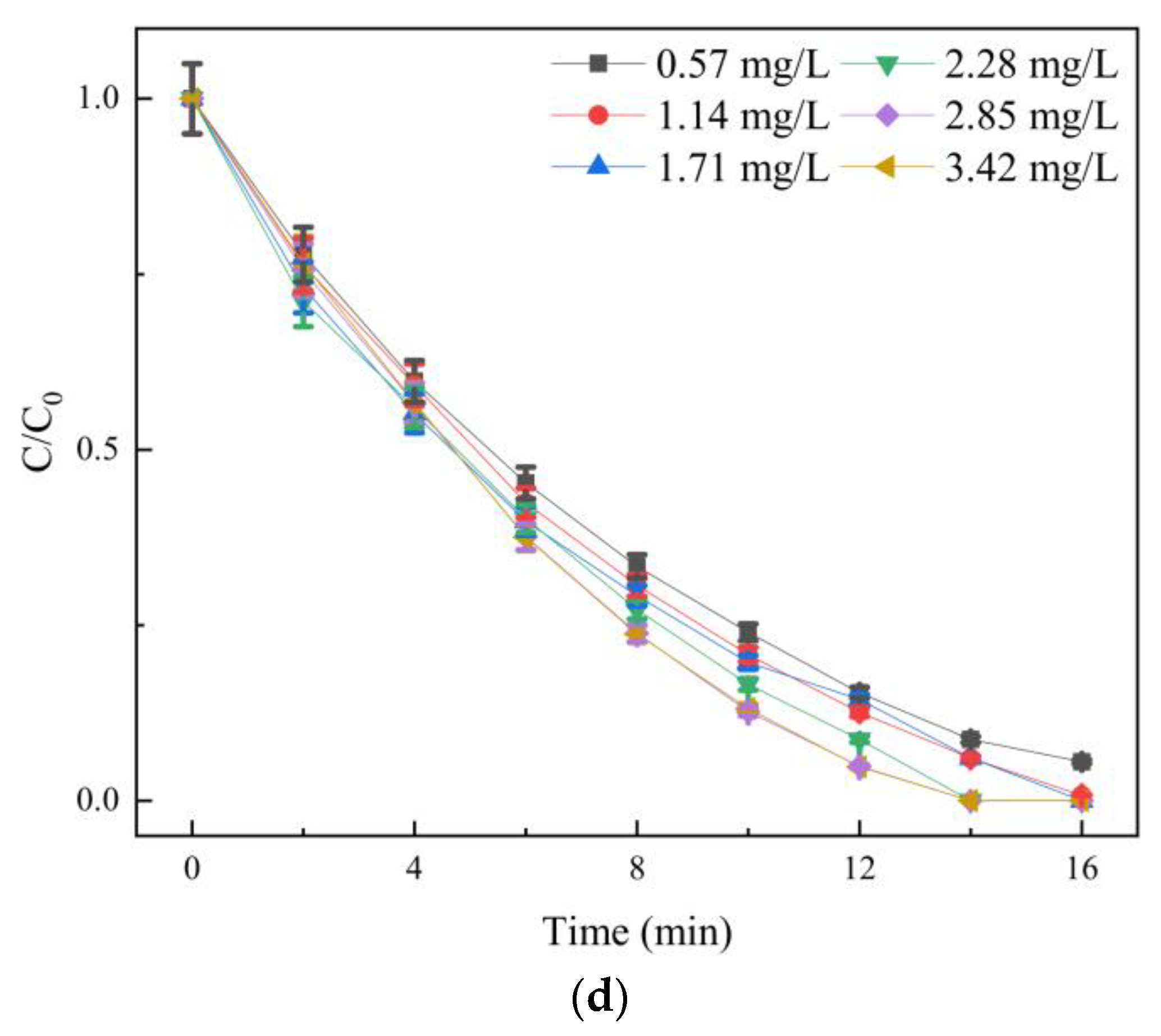
3.2.2. Effect of Oxidant Dosage on Degradation Efficiency
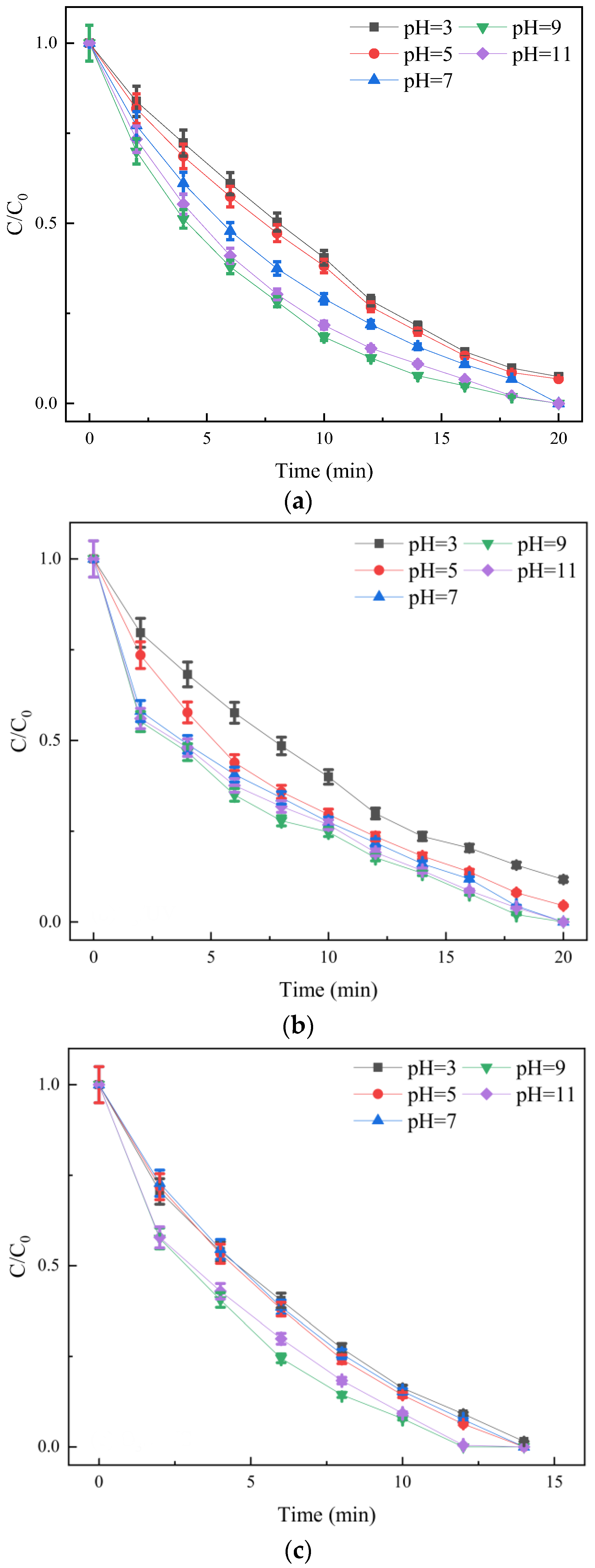
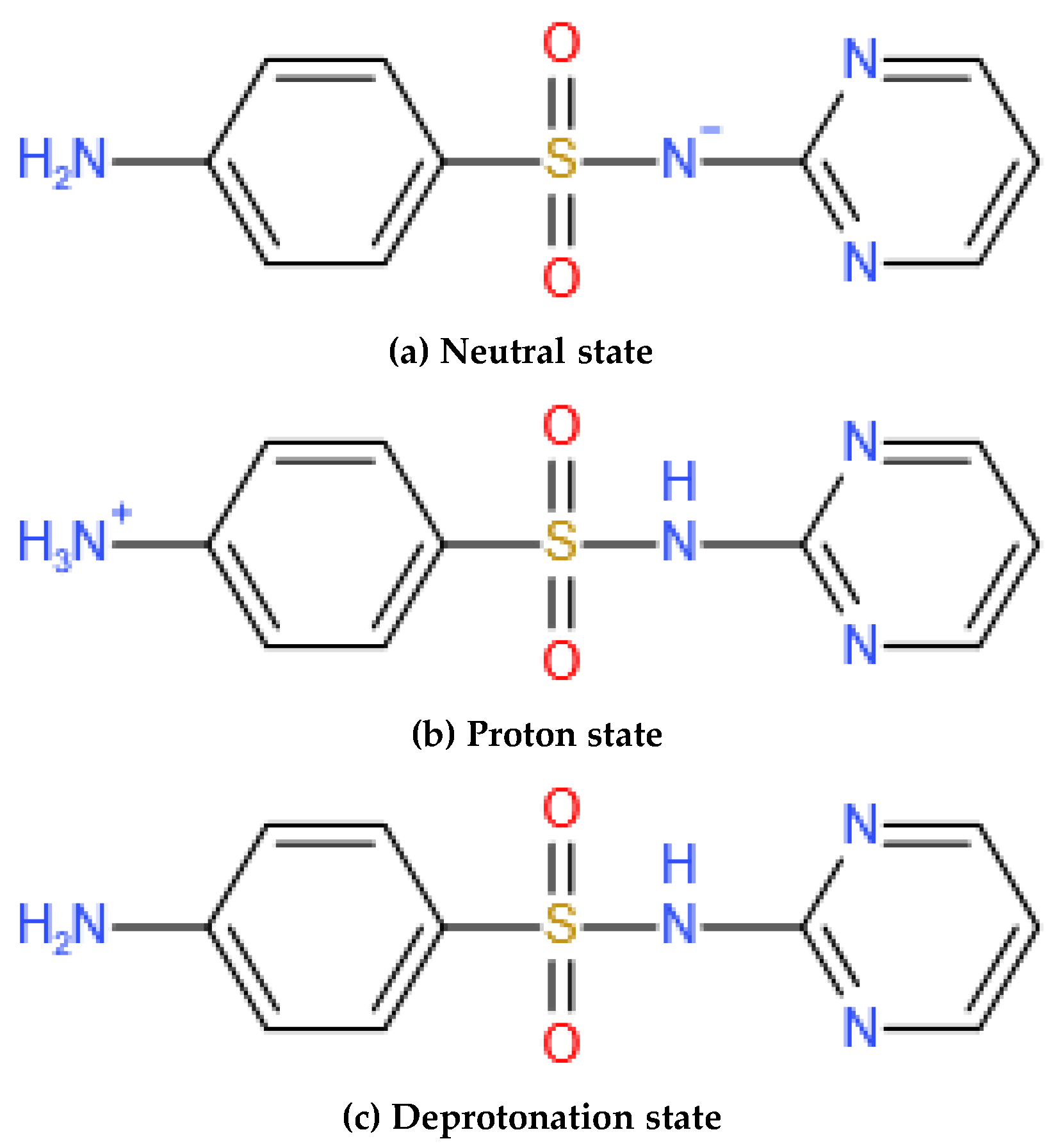
3.2.3. Effect of Initial pH on Degradation Efficiency
3.3. Mineralization Effect of O3, O3/UV, and O3/PMS on SDZ
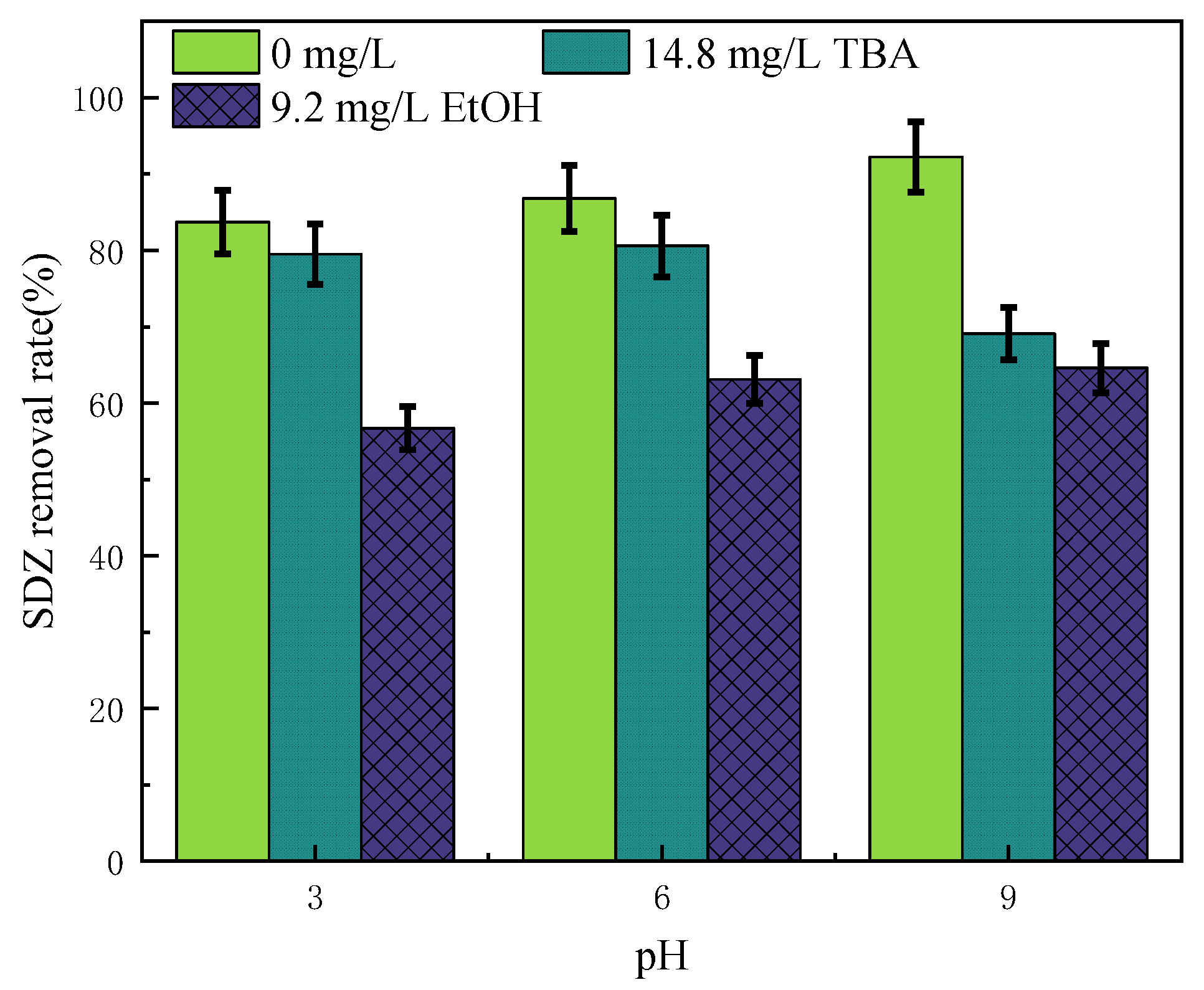
3.4. Radical Quenching Experiment of Sulfadiazine Degradation by Ozone/Persulfate
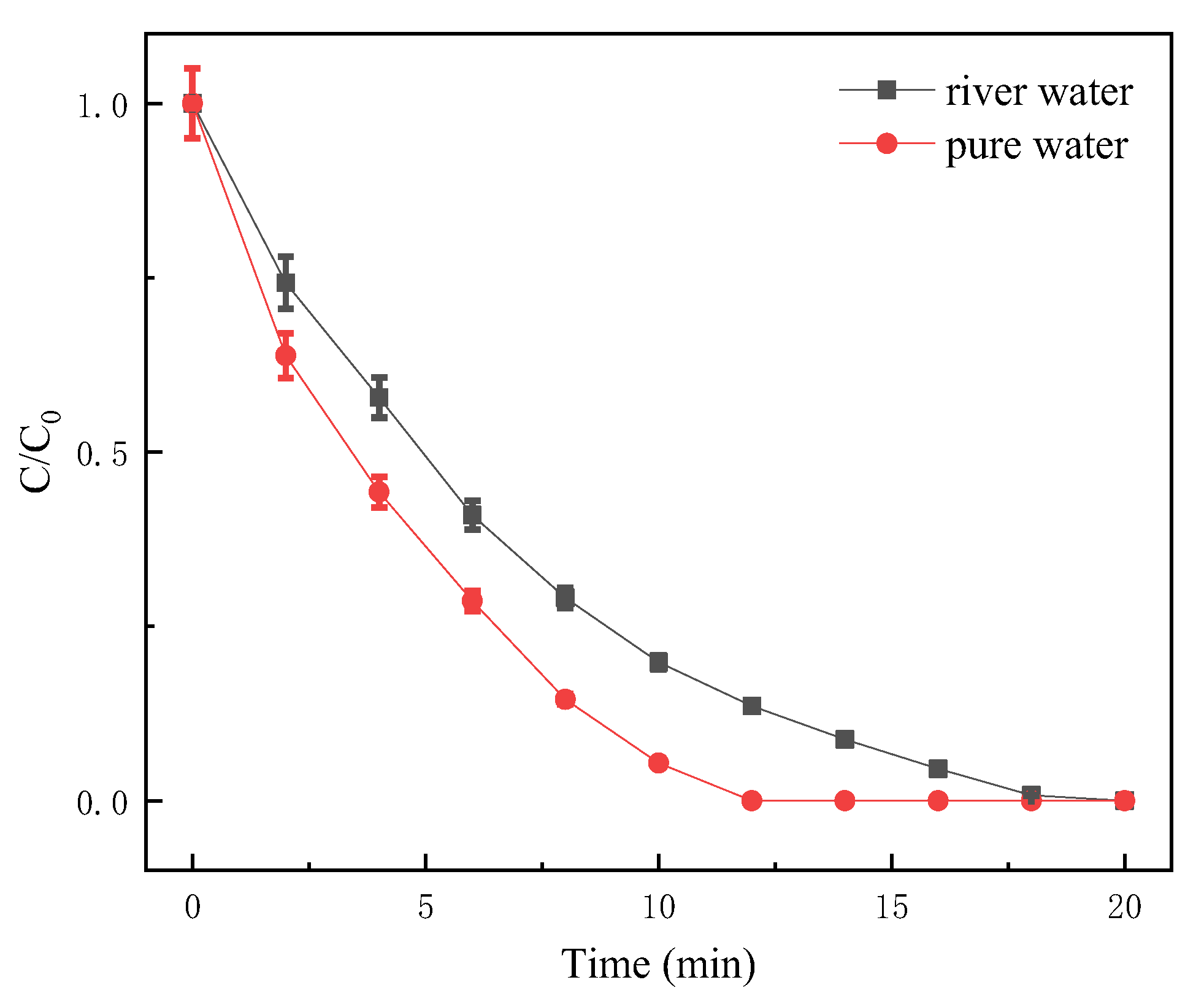
3.5. Degradation Test of SDZ in Natural Water
4. Conclusions
- (1)
- In this experiment, the O3/PMS process was used for the first time to degrade the typical antibiotic SDZ. The combination of O3 and PMS produced an apparent synergistic effect and improved the degradation efficiency of SDZ. Under the experimental conditions of initial SDZ concentration of 10 mg/L, ozone concentration of 3 mg/L, and pH of 6.8 ± 0.1, the time required for O3/PMS to degrade SDZ below the detection limit was 8 min less than that of O3 and O3/UV. Existing studies show that the treatment effect of a single UV factor was poor, and even the O3/UV combined process can only achieve limited improvement. The Kobs value increased from 0.1456 to 0.193 min−1, while the O3/PMS process Kobs was 0.2504 min−1.
- (2)
- The single factor experiment showed that the degradation rate of SDZ by O3/PMS decreased with the increase in the initial concentration of SDZ and increased with the increase in ozone concentration. There was an optimal concentration of PMS. The most suitable pH range was weak alkalinity, and the factors affecting the degradation of SDZ by the three processes agree with the pseudo-first-order reaction kinetic equation.
- (3)
- The strong oxidation ability of O3/PMS was attributed to the formation of ·OH and SO4−. When the reaction was carried out for 60 min, it had a nearly 54% mineralization effect on SDZ, slightly lower than 62% of O3/UV. When SDZ was degraded in natural water, O3/PMS showed efficient degradation of fluorescent organics, significantly better than other ozone-advanced oxidation processes.
- (4)
- Future research should focus on the insufficient mineralization ability of O3/PMS and apply it to the degradation of more sulfonamides to improve the stability and applicability of this technology. In addition, the influencing factors need to be modeled and calculated to provide data prediction and a theoretical analysis basis for the specific practical application of this method.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Qiao, M.; Chen, W.D.; Su, J.Q.; Zhang, B.; Zhang, C. Fate of tetracyclines in swine manure of three selected swine farms in China. J. Environ. Sci. 2012, 24, 1047–1052. [Google Scholar] [CrossRef]
- Chee-Sanford, J.C.; Mackie, R.I.; Koike, S.; Krapac, I.G.; Lin, Y.F.; Yannarell, A.C.; Maxwell, S.; Aminov, R.I. Fate and transport of antibiotic residues and antibiotic resistance genes following land application of manure waste. J. Environ. Qual. 2009, 38, 1086–1108. [Google Scholar] [CrossRef] [PubMed]
- Pan, X.; Qiang, Z.M.; Ben, W.W.; Chen, M.X. Residual veterinary antibiotics in swine manure from concentrated animal feeding operations in Shandong Province, China. Chemosphere 2011, 84, 695–700. [Google Scholar] [CrossRef] [PubMed]
- Hvistendahl, M. China takes aim ai rampant antibiotic resistance. Science 2012, 336, 795. [Google Scholar] [CrossRef]
- Meyer, E.; Gastmeier, P.; Deja, M.; Schwab, F. Antibiotic consumption and resistance: Data from Europe and Germany. Int. J. Med. Microbiol. 2013, 303, 388–395. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.G.; Johnson, T.A.; Su, J.Q.; Qiao, M.; Guo, G.X.; Stedtfeld, R.D.; Hashsham, S.A.; Tiedje, J.M. Diverse and abundant antibiotic resistance genes in China swine farms. Proc. Natl. Acad. Sci. USA 2013, 110, 3435–3440. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.Q.; Ying, G.G.; Pan, C.G.; Liu, Y.S.; Zhao, J.L. A comprehensive evaluation of antibiotics emission and fate in the river basins of China: Source analysis, multimedia modelling, and linkage to bacterial resistance. Environ. Sci. Technol. 2015, 49, 6772–6782. [Google Scholar] [CrossRef]
- Sassman, S.A.; Lee, L.S. Sorption of three tetracyclines by several soils: Assessing the role of pH and cation exchange. Environ. Sci. Technol. 2005, 39, 7452–7459. [Google Scholar] [CrossRef]
- Pruden, A.; Pei, R.; Storteboom, H.; Carlson, K.H. Antibiotic resistance genes as emerging contaminants: Studies in northern Colorado. Environ. Sci. Technol. 2006, 40, 7445–7450. [Google Scholar] [CrossRef]
- Gao, L.H.; Shi, Y.L.; Li, W.H.; Liu, J.M.; Cai, Y.Q. Occurrence, distribution and bioaccumulation of antibiotics in the Haihe River in China. J. Environ. Monit. 2012, 14, 1248. [Google Scholar] [CrossRef]
- Su, J.Q.; Huang, F.Y.; Zhu, Y.G. Antibiotic resistance genes in the environment. Biodivers. Sci. 2013, 21, 481–487. [Google Scholar]
- Jia, A.; Hu, J.Y.; Wu, X.Q.; Peng, H.; Wu, S.M.; Dong, Z.M. Occurrence and source apportionment of sulfonamides and their metabolites in Liaodong Bay and the adjacent Liao River basin, north China. Environ. Toxicol. Chem. 2011, 30, 1252–1260. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.Q.; Qian, M.R.; Lu, L.Z.; Tao, Z.R.; Li, G.Q.; Feng, S.L.; Chen, Z.M.; Li, R.; Fang, L.Z. Anitiobic residues in waste waters from main livestock farms in Zhejiang Province. Acta Agric. Zhejiangensis 2012, 24, 699–705. [Google Scholar]
- Chen, J.P.; Yang, Y.L.; Wu, Z.Q.; Peng, G.H. Pollution of antibiotics in livestock wastewater and the environmental water in Jiangxi Province. J. Anhui Agri Sci 2015, 43, 224–227. [Google Scholar]
- Peng, X.Z.; Wang, Z.D.; Kuang, W.X.; Tan, J.H.; Li, K. A preliminary study on the occurrence and behavior of sulfonamides, ofloxacin and chloramphenicol antimicrobials in wastewaters of two sewage treatment plants in Guangzhou, China. Sci. Total Environ. 2006, 371, 314–322. [Google Scholar] [CrossRef]
- Garcia-Galan, M.J.; Diaz-Cruz, M.S.; Barcelo, D. Determination of 19 sulfonamides in environmental water samples by automated on-line solid-phase extraction-liquid chromatography-tandem mass spectrometry (SPE-LC-MS/MS). Talanta 2010, 81, 355–366. [Google Scholar] [CrossRef]
- Wei, R.C.; Ge, F.; Huang, S.Y.; Chen, M.; Wang, R. Occurrence of veterinary antibiotics in animal wastewater and surface water around farms in Jiangsu Province, China. Chemosphere 2011, 82, 1408–1414. [Google Scholar] [CrossRef]
- Gao, L.H.; Shi, Y.L.; Li, W.H.; Niu, H.Y.; Liu, J.M.; Cai, Y.Q. Occurrence of antibiotics in eight sewage treatment plants in Beijing, China. Chemosphere 2012, 86, 665–671. [Google Scholar] [CrossRef]
- Lian, Z.R.; He, X.L.; Wang, J.T. Determination of Sulfadiazine in Jiaozhou Bay using molecularly imprinted solid-phase extraction followed by high-performance liquid chromatography with a diode-array detector. J. Chromatogr. B 2014, 957, 53–59. [Google Scholar] [CrossRef]
- Tlili, I.; Caria, G.; Ouddane, B.; Ghorbel-Abid, I.; Ternane, R.; Trabelsi-Ayadi, M.; Net, S. Simultaneous detection of antibiotics and other drug residues in the dissolved and particulate phases of water by an off-line SPE combined with on-line SPE-LC-MS/MS: Method development and application. Sci. Total Environ. 2016, 563–564, 424–433. [Google Scholar] [CrossRef]
- Chen, H.Y.; Jing, L.J.; Teng, Y.G.; Wang, J.S. Characterisation of antibiotic in a large-scale river system of China: Occurrence pattern, spatiotemporal distribution and environmental risks. Sci. Total Environ. 2018, 618, 409–418. [Google Scholar] [CrossRef] [PubMed]
- Afsa, S.; Hamden, K.; Martin, P.A.L.; Mansour, H.B. Occurrence of 40 pharmaceutically active compounds in hospital and urban wastewaters and their contribution to Mahdia coastal seawater contamination. Environ. Sci. Pollut. Res. 2020, 27, 1941–1955. [Google Scholar] [CrossRef]
- Ngigi, A.N.; Magu, M.M.; Muendo, B.M. Occurrence of antibiotics residues in hospital wastewater, wastewater treatment plant, and in surface water in Nairobi County, Kenya. Environ. Monit. Assess. 2020, 192, 18. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.; Pan, C.G.; Wang, Y.H.; Xiao, S.K.; Yu, K.F. Antibiotics in a subtropical food wed from the Beibu Gulf, South China: Occurrence, bioaccumulation and trophic transfer. Sci. Total Environ. 2021, 751, 141718. [Google Scholar] [CrossRef] [PubMed]
- Manna, M.; Sen, S. Advanced oxidation process: A sustainable technology for treating refractory organic compounds present in industrial wastewater. Environ. Sci. Pollut. Res. 2022. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Yang, Y.L.; Zheng, H.S.; Zheng, Y.J.; Jing, T.; Ma, J.; Nan, J.; Leong, Y.K.; Chang, J.S. Advanced oxidation process based on hydroxyl and sulfate radicals to degrade refractory organic pollutants in lanfill leachate. Chemosphere 2022, 297, 134214. [Google Scholar] [CrossRef] [PubMed]
- Hien, N.T.; Nguyen, L.H.; Van, H.T.; Nguyen, T.D.; Nguyen, T.H.V.; Chu, T.H.H.; Nguyen, T.V.; Trinh, V.T.; Vu, X.H.; Aziz, K.H.H. Heterogeneous catalyst ozonation of Direct Black 22 from aqueous solution in the presence of metal slags origi-nating from industrial solid wastes. Sep. Purif. Technol. 2020, 233, 115961. [Google Scholar] [CrossRef]
- Foroughi, M.; Khiadani, M.; Kakhki, S.; Kholghi, V.; Naderi, K.; Yektay, S. Effect of ozonation-based disinfection methods on the removal of antibiotic resistant bacteria and resistance genes (ARB/ARGs) in water and wastewater treatment: A systematic review. Sci. Total Environ. 2022, 811, 151404. [Google Scholar] [CrossRef]
- Issaka, E.; AMU-Darko, J.N.O.; Yakubu, S.; Fapohunda, F.O.; Ali, N. Bilal. M. Advanced catalytic ozonation for degradation of pharmaceutical pollutants-A review. Chemosphere 2022, 289, 133208. [Google Scholar] [CrossRef] [PubMed]
- Kanakaraju, D.; Glass, B.D.; Oelgemoller, M. Advanced oxidation process-mediated removal of pharmaceuticals from water: A review. J. Environ. Manag. 2018, 219, 189–207. [Google Scholar] [CrossRef] [PubMed]
- Pandis, P.K.; Kalogirou, C.; Kanellou, E.; Vaitsis, C.; Savvidou, M.G.; Sourkouni, G.; Zorpas, A.A.; Argirusis, C. Key Points of Advanced Oxidation Processes (AOPs) for Wastewater, Organic Pollutants and Pharmaceutical Waste Treatment: A Mini Review. ChemEngineering 2022, 6, 8. [Google Scholar] [CrossRef]
- Aziz, K.H.H. Application of different advanced oxidation processes for the removal of chloroacetic acids using a planar falling film reactor. Chemosphere 2019, 228, 377–383. [Google Scholar] [CrossRef]
- Asghar, A.; Lutze, H.V.; Tuerk, J.; Schmidt, T.C. Influence of water matrix on the degradation of organic micropollutants by ozone based processes: A review on oxidant scavenging mechanism. J. Hazard. Mater. 2022, 429, 128189. [Google Scholar] [CrossRef] [PubMed]
- Mora, A.S.; McBeath, S.T.; Cid, C.A.; Hoffmann, M.R.; Graham, N.J.D. Diamond electrode facilitated electrosynthesis of water and wastewater treatment oxidants. Curr. Opin. Electroche. 2022, 32, 100899. [Google Scholar] [CrossRef]
- Jaafarzadeh, N.; Ghanbari, F.; Ahmadi, M.; Omidinasab, M. Efficient integrated processes for pulp and paper wastewater treatment and phytotoxicity reduction: Permanganate, Electro-Fenton and Co3O4/UV/peroxymonosulfate. Chem. Eng. J. 2017, 308, 142–150. [Google Scholar] [CrossRef]
- Jaafarzadeh, N.; Ghanbari, F.; Omidinasab, M. Combined electrocoagulation and UV-based sulfate radical oxidation processes for treatment of pulp and paper wastewater. Process Saf. Environ. Prot. 2016, 102, 462–472. [Google Scholar] [CrossRef]
- Hu, P.D.; Long, M.C. Cobalt-catalyzed sulfate radical-based advanced oxidation: A review on heterogeneous catalysts and applications. Appl. Catal. B 2016, 181, 103–117. [Google Scholar] [CrossRef]
- Wen, G.; Wang, S.B.; Wang, T.; Feng, Y.B.; Chen, Z.H.; Lin, W.; Huang, T.L.; Ma, J. Inhibition of bromate formation in the O3/PMS process by adding low dosage of carbon materials: Efficiency and mechanism. Chem. Eng. J. 2020, 402, 126207. [Google Scholar] [CrossRef]
- Zhao, G.Y. Research on Emerging Contaminants Removal from Drinking Water by UV-Micro O3 Process; Southeast University: Nanjing, China, 2014. (In Chinese) [Google Scholar]
- Simonin, J.P. On the comparison of pseudo-second order rate laws in the modeling of adsorption kinetics. Chem. Eng. J. 2016, 300, 254–263. [Google Scholar] [CrossRef]
- Yang, Z.Z. Performance and Mechanism for Degradation of Sulfadiazine in Water by UV/O3 Process; Harbin Institute of Technology: Harbin, China, 2016. (In Chinese) [Google Scholar]
- Wang, Y.B.; Wang, S.P.; Wang, Z.; Zhou, R.F.; Chang, J. Degradation effect and kinetics of ibuprofen in water by UV/O3 advanced oxidation. Ind. Water Treat. 2020, 40, 40–43. [Google Scholar]
- Yuan, Z.; Sui, M.H.; Yuan, B.J.; Li, P.; Wang, J.Y.; Qin, J.; Xu, G.Y. Degradation of ibuprofen using Ozone combined with peroxymonosulfate. Environ. Sci. Water Res. Technol. 2017, 3, 960–969. [Google Scholar] [CrossRef]
- Maruthamuthu, P.; Neta, P. Radiolytic chain decomposition of peroxomonophosphoric and peroxomonosulfuric acids. J. Phys. Chem. B 1977, 81, 937–940. [Google Scholar] [CrossRef]
- Neta, P.; Huie, R.E.; Ross, A.B. Rate constants for reactions of inorganic radicals in aqueous solution. J. Phys. Chem. Ref. Data 1988, 17, 1027–1284. [Google Scholar] [CrossRef]
- Buxton, G.V.; Greenstock, C.L.; Helman, W.P.; Ross, A.B. Critical review of rate for reactions of hydrates electrons, hydrogen atoms and hydroxyl radicals (OH/·O−) in aqueous solution. J. Phys. Chem. Ref. Data 1988, 17, 513–886. [Google Scholar] [CrossRef]
- Klaning, U.K.; Sehested, K.; Appelman, E.H. Laser flash photolysis and pulse radiolysis of aqueous solutions of the fluoroxysulfate ion, SO4F−. Inorg. Chem. 1991, 30, 3582–3584. [Google Scholar] [CrossRef]
- Sehested, K.; Holcman, J.; Bjergbakke, E.; Hart, E.J. Formation of Ozone in the reaction of OH with O3− and the decay of the ozonide ion radical at pH 10–13. J. Phys. Chem. 1984, 88, 269–273. [Google Scholar] [CrossRef]
- Elliot, A.J.; Mccracken, D.R. Effect of temperature on O− reactions and equilibria: A pulse radiolysis study. Radiat. Phys. Chem. 1989, 33, 69–74. [Google Scholar] [CrossRef]
- Chen, T.Y.; Kao, C.M.; Hong, A.; Lin, C.E.; Liang, S.H. Application of Ozone on the decolorisation of reactive dyes-Orange-13 and Blue-19. Desalination 2009, 249, 1238–1242. [Google Scholar] [CrossRef]
- Chu, W.; Ma, C.W. Quantitative prediction of direct and indirect dye ozonation kinetics. Water Res. 2000, 34, 3153–3160. [Google Scholar] [CrossRef]
- Shu, H.Y.; Chang, M.C. Decolorization effects of six azo dyes by O3, UV/O3 and UV/H2O2 processes. Dyes. Pigm. 2005, 65, 25–31. [Google Scholar] [CrossRef]
- Rong, S.P.; Sun, Y.B.; Zhao, Z.H. Degradation of sulfadiazine antibiotics by water falling film dielectric barrier discharge. Chin. Chem. Lett. 2014, 25, 187–192. [Google Scholar] [CrossRef]
- Murphy, K.R.; Hambly, A.; Singh, S.; Henderson, R.K.; Baker, A.; Stuetz, R.; Khan, S.J. Organic matter fluorescence in municipal water recycling schemes: Toward a unified PARAFAC Model. Environ. Sci. Technol. 2011, 45, 2909–2916. [Google Scholar] [CrossRef] [PubMed]




| Water Quality Index | Value |
|---|---|
| COD (mg/L) | 38.81 |
| Total nitrogen (TN, mg/L) | 4.39 |
| NH4+-N (mg/L) | 1.73 |
| Total phosphorus (TP, mg/L) | 0.16 |
| Treatment Process | Kobs (min−1) | R2 |
|---|---|---|
| O3 | 0.1456 | 0.9599 |
| O3/UV | 0.193 | 0.7255 |
| O3/PMS | 0.2504 | 0.9326 |
| Treatment Process | Mass Concentration (mg/L) | Kobs (min−1) | R2 |
|---|---|---|---|
| O3 | 2 | 0.2814 | 0.9195 |
| 4 | 0.2228 | 0.8428 | |
| 6 | 0.2048 | 0.8731 | |
| 8 | 0.1261 | 0.9624 | |
| 10 | 0.1242 | 0.9556 | |
| O3/UV | 2 | 0.3872 | 0.9893 |
| 4 | 0.2303 | 0.8369 | |
| 6 | 0.1930 | 0.7255 | |
| 8 | 0.1625 | 0.9737 | |
| 10 | 0.1328 | 0.9203 | |
| O3/PMS | 2 | 0.5563 | 0.9935 |
| 4 | 0.3979 | 0.9793 | |
| 6 | 0.3551 | 0.8767 | |
| 8 | 0.3363 | 0.9802 | |
| 10 | 0.2497 | 0.9389 |
| Treatment Process | Mass Concentration (mg/L) | Kobs (min−1) | R2 |
|---|---|---|---|
| O3 | 1 | 0.0730 | 0.9821 |
| 2 | 0.0174 | 0.9975 | |
| 3 | 0.1456 | 0.9599 | |
| 4 | 0.1646 | 0.9666 | |
| 5 | 0.2019 | 0.9670 | |
| 6 | 0.2436 | 0.9716 | |
| O3/UV | 1 | 0.0241 | 0.9322 |
| 2 | 0.0447 | 0.9928 | |
| 3 | 0.1930 | 0.7255 | |
| 4 | 0.1946 | 0.8941 | |
| 5 | 0.2441 | 0.8802 | |
| 6 | 0.2613 | 0.9692 | |
| O3/PMS | 1 | 0.0098 | 0.9888 |
| 2 | 0.2359 | 0.8774 | |
| 3 | 0.2504 | 0.9326 | |
| 4 | 0.3025 | 0.9390 | |
| 5 | 0.3158 | 0.9649 | |
| 6 | 0.3408 | 0.8539 |
| Treatment Process | Mass Concentration (mg/L) | Kobs (min−1) | R2 |
|---|---|---|---|
| O3/PMS | 5 | 0.1790 | 0.9783 |
| 10 | 0.1851 | 0.9687 | |
| 15 | 0.1954 | 0.9743 | |
| 20 | 0.2570 | 0.8570 | |
| 25 | 0.2405 | 0.9502 | |
| 30 | 0.2408 | 0.9480 |
| Treatment Process | pH | Kobs (min−1) | R2 |
|---|---|---|---|
| O3 | 3 | 0.1320 | 0.9732 |
| 5 | 0.1366 | 0.9750 | |
| 7 | 0.1433 | 0.9873 | |
| 9 | 0.1903 | 0.9496 | |
| 11 | 0.2042 | 0.9719 | |
| O3/UV | 3 | 0.1051 | 0.9937 |
| 5 | 0.1401 | 0.9694 | |
| 7 | 0.1421 | 0.9305 | |
| 9 | 0.1531 | 0.9466 | |
| 11 | 0.1733 | 0.8975 | |
| O3/PMS | 3 | 0.2594 | 0.8776 |
| 5 | 0.2194 | 0.9654 | |
| 7 | 0.2074 | 0.9726 | |
| 9 | 0.3663 | 0.9947 | |
| 11 | 0.2486 | 0.7970 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lu, H.; Feng, W.; Li, Q. Degradation Efficiency Analysis of Sulfadiazine in Water by Ozone/Persulfate Advanced Oxidation Process. Water 2022, 14, 2476. https://doi.org/10.3390/w14162476
Lu H, Feng W, Li Q. Degradation Efficiency Analysis of Sulfadiazine in Water by Ozone/Persulfate Advanced Oxidation Process. Water. 2022; 14(16):2476. https://doi.org/10.3390/w14162476
Chicago/Turabian StyleLu, Hai, Weihao Feng, and Qingpo Li. 2022. "Degradation Efficiency Analysis of Sulfadiazine in Water by Ozone/Persulfate Advanced Oxidation Process" Water 14, no. 16: 2476. https://doi.org/10.3390/w14162476
APA StyleLu, H., Feng, W., & Li, Q. (2022). Degradation Efficiency Analysis of Sulfadiazine in Water by Ozone/Persulfate Advanced Oxidation Process. Water, 14(16), 2476. https://doi.org/10.3390/w14162476





