Pollutant Removal and Energy Recovery from Swine Wastewater Using Anaerobic Membrane Bioreactor: A Comparative Study with Up-Flow Anaerobic Sludge Blanket
Abstract
:1. Introduction
2. Materials and Methods
2.1. Swine Wastewater and Inoculum Sludge
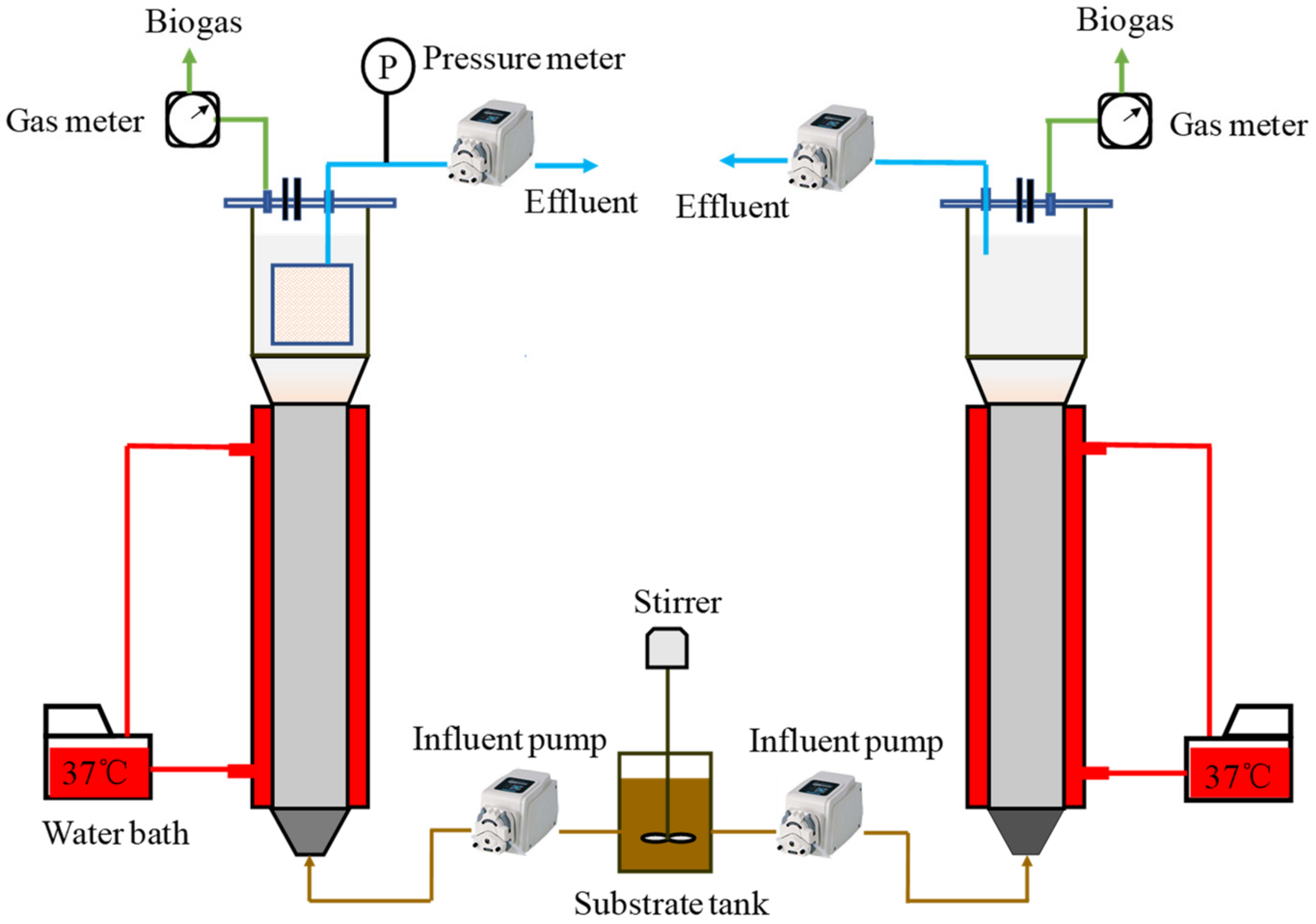
2.2. Reactors Design and Operation
2.3. Analytical Methods and Data Analysis
2.3.1. General Parameters
2.3.2. Specific Methanogenic Activity (SMA) Analysis
2.3.3. Microbial Community Analysis
2.3.4. Energy Balance Analysis
3. Results and Discussions
3.1. Pollutants Removal
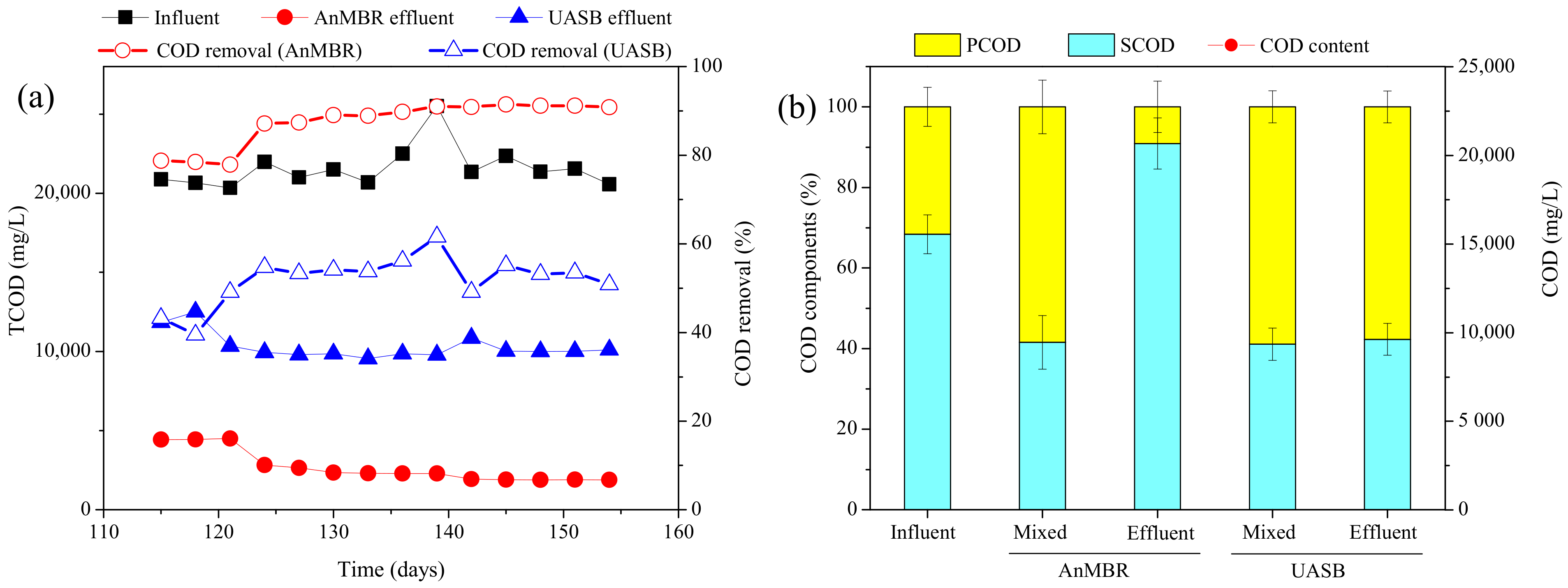
3.1.1. COD Removal
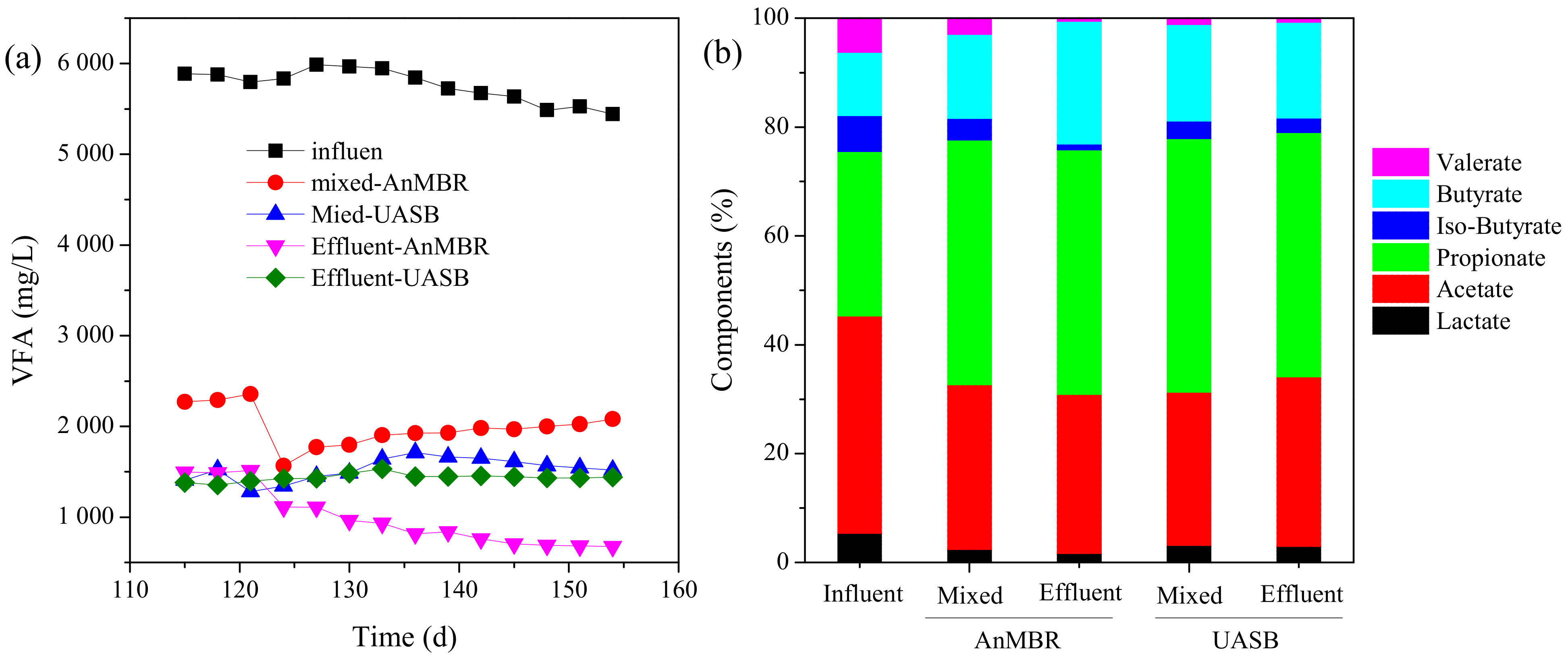
3.1.2. Volatile Fatty Acids
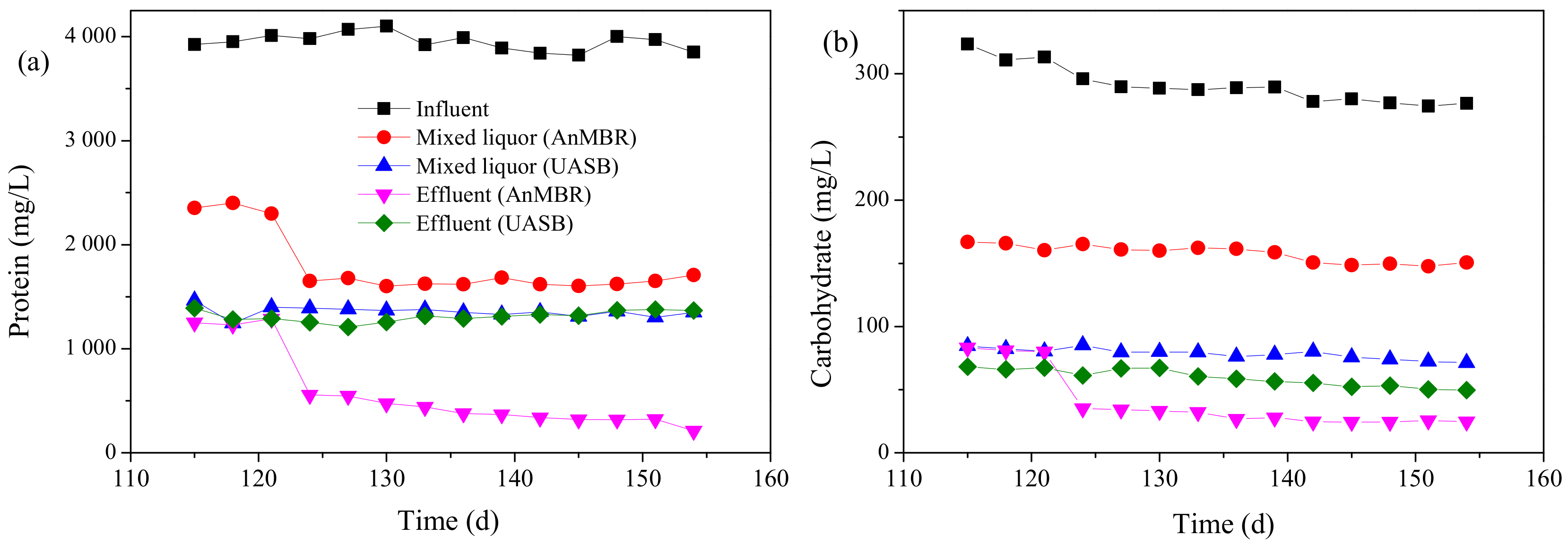
3.1.3. Proteins and Carbohydrates
3.2. Energy Recovery Properties
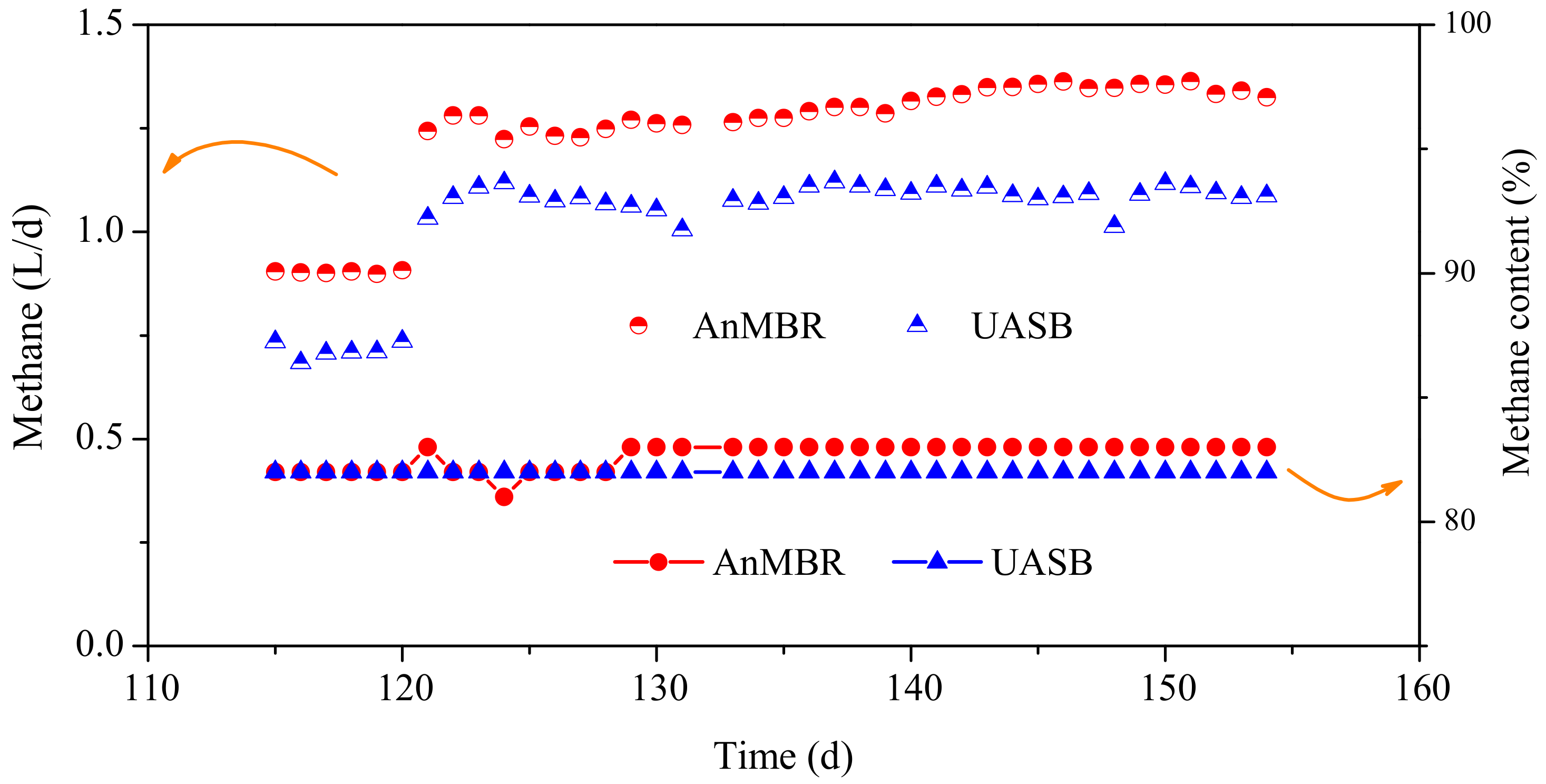
3.2.1. Methane Yield
3.2.2. Specific Methanogenic Activity
3.3. Microbial Community Analysis
3.3.1. Bacteria
3.3.2. Archaea
3.4. Energy Balance Analysis
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- National Bureau of Statistics of China. China Statistical Yearbook; National Bureau of Statistics of China, Ed.; China Statistics Press: Beijing, China, 2020. [Google Scholar]
- Cheng, D.; Ngo, H.H.; Guo, W.; Chang, S.W.; Nguyen, D.D.; Nguyen, Q.A.; Zhang, J.; Liang, S. Improving sulfonamide antibiotics removal from swine wastewater by supplying a new pomelo peel derived biochar in an anaerobic membrane bioreactor. Bioresour. Technol. 2021, 319, 124160. [Google Scholar] [CrossRef]
- Liu, Y.; Li, X.; Tan, Z.; Yang, C. Inhibition of tetracycline on anaerobic digestion of swine wastewater. Bioresour. Technol. 2021, 334, 125253. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Liu, Y.-S.; Zhao, J.-L.; Liu, W.-R.; Chen, J.; Zhang, Q.-Q.; He, L.-Y.; Ying, G.-G. Variations of antibiotic resistome in swine wastewater during full-scale anaerobic digestion treatment. Environ. Int. 2021, 155, 106694. [Google Scholar] [CrossRef]
- Zeng, Z.; Zheng, P.; Kang, D.; Li, Y.; Li, W.; Xu, D.; Chen, W.; Pan, C. The removal of copper and zinc from swine wastewater by anaerobic biological-chemical process: Performance and mechanism. J. Hazard. Mater. 2021, 401, 123767. [Google Scholar] [CrossRef] [PubMed]
- Domingues, E.; Fernandes, E.; Gomes, J.; Martins, R.C. Swine wastewater treatment by Fenton’s process and integrated methodologies involving coagulation and biofiltration. J. Clean. Prod. 2021, 293, 126105. [Google Scholar] [CrossRef]
- Guan, Q.; Zeng, G.; Gong, B.; Li, Y.; Ji, H.; Zhang, J.; Song, J.; Liu, C.; Wang, Z.; Deng, C. Phosphorus recovery and iron, copper precipitation from swine wastewater via struvite crystallization using various magnesium compounds. J. Clean. Prod. 2021, 328, 129588. [Google Scholar] [CrossRef]
- Dinnebier, H.C.F.; Matthiensen, A.; Michelon, W.; Tápparo, D.C.; Fonseca, T.G.; Favretto, R.; Steinmetz, R.L.R.; Treichel, H.; Antes, F.G.; Kunz, A. Phycoremediation and biomass production from high strong swine wastewater for biogas generation improvement: An integrated bioprocess. Bioresour. Technol. 2021, 332, 125111. [Google Scholar] [CrossRef]
- De Oliveira, M.G.; Mourão, J.M.M.; Silva, F.S.S.; dos Santos, A.B.; Pereira, E.L. Effect of microaerophilic treatment on swine wastewater (SWW) treatment: Engineering and microbiological aspects. J. Environ. Manag. 2021, 299, 113598. [Google Scholar] [CrossRef]
- Guimarães de Oliveira, M.; Marques Mourão, J.M.; Marques de Oliveira, A.K.; Bezerra dos Santos, A.; Lopes Pereira, E. Microaerophilic treatment enhanced organic matter removal and methane production rates during swine wastewater treatment: A long-term engineering evaluation. Renew. Energy 2021, 180, 691–699. [Google Scholar] [CrossRef]
- Jiang, M.; Westerholm, M.; Qiao, W.; Wandera, S.M.; Dong, R. High rate anaerobic digestion of swine wastewater in an anaerobic membrane bioreactor. Energy 2020, 193, 116783. [Google Scholar] [CrossRef]
- Cheng, H.-H.; Narindri, B.; Chu, H.; Whang, L.-M. Recent advancement on biological technologies and strategies for resource recovery from swine wastewater. Bioresour. Technol. 2020, 303, 122861. [Google Scholar] [CrossRef] [PubMed]
- Shahid, M.K.; Kashif, A.; Rout, P.R.; Aslam, M.; Fuwad, A.; Choi, Y.; Banu, J.R.; Park, J.H.; Kumar, G. A brief review of anaerobic membrane bioreactors emphasizing recent advancements, fouling issues and future perspectives. J. Environ. Manag. 2020, 270, 110909. [Google Scholar] [CrossRef]
- Lin, H.; Peng, W.; Zhang, M.; Chen, J.; Hong, H.; Zhang, Y. A review on anaerobic membrane bioreactors: Applications, membrane fouling and future perspectives. Desalination 2013, 314, 169–188. [Google Scholar] [CrossRef]
- Al-Asheh, S.; Bagheri, M.; Aidan, A. Membrane bioreactor for wastewater treatment: A review. Case Stud. Chem. Environ. Eng. 2021, 4, 100109. [Google Scholar] [CrossRef]
- Asante-Sackey, D.; Rathilal, S.; Tetteh, E.K.; Armah, E.K. Membrane Bioreactors for Produced Water Treatment: A Mini-Review. Membranes 2022, 12, 275. [Google Scholar] [CrossRef] [PubMed]
- Tomczak, W.; Gryta, M. Energy-Efficient AnMBRs Technology for Treatment of Wastewaters: A Review. Energies 2022, 15, 4981. [Google Scholar] [CrossRef]
- Jensen, P.; Yap, S.D.; Boyle-Gotla, A.; Janoschka, J.; Carney, C.; Pidou, M.; Batstone, D. Anaerobic membrane bioreactors enable high rate treatment of slaughterhouse wastewater. Biochem. Eng. J. 2015, 97, 132–141. [Google Scholar] [CrossRef] [Green Version]
- Pretel, R.; Robles, A.; Ruano, M.; Seco, A.; Ferrer, J. The operating cost of an anaerobic membrane bioreactor (AnMBR) treating sulphate-rich urban wastewater. Sep. Purif. Technol. 2014, 126, 30–38. [Google Scholar] [CrossRef]
- Lin, Y.-L. Membrane Fouling Control in Water Treatment. Membranes 2022, 12, 551. [Google Scholar] [CrossRef]
- Zhen, G.; Pan, Y.; Lu, X.; Li, Y.-Y.; Zhang, Z.; Niu, C.; Kumar, G.; Kobayashi, T.; Zhao, Y.; Xu, K. Anaerobic membrane bioreactor towards biowaste biorefinery and chemical energy harvest: Recent progress, membrane fouling and future perspectives. Renew. Sustain. Energy Rev. 2019, 115, 109392. [Google Scholar] [CrossRef]
- Berkessa, Y.W.; Yan, B.; Li, T.; Tan, M.; She, Z.; Jegatheesan, V.; Jiang, H.; Zhang, Y. Novel anaerobic membrane bioreactor (AnMBR) design for wastewater treatment at long HRT and high solid concentration. Bioresour. Technol. 2018, 250, 281–289. [Google Scholar] [CrossRef] [PubMed]
- Ni, J.; Ji, J.; Li, Y.-Y.; Kubota, K. Kubota, Microbial characteristics in anaerobic membrane bioreactor treating domestic sewage: Effects of HRT and process performance. J. Environ. Sci. 2022, 111, 392–399. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Lei, Z.; Dzakpasu, M.; Li, Q.; Li, Y.-Y.; Chen, R. Removal of trace organic contaminants in municipal wastewater by anaerobic membrane bioreactor: Efficiencies, fates and impact factors. J. Water Process Eng. 2021, 40, 101953. [Google Scholar] [CrossRef]
- Foglia, A.; Andreola, C.; Cipolletta, G.; Radini, S.; Akyol, C.; Eusebi, A.L.; Stanchev, P.; Katsou, E.; Fatone, F. Comparative life cycle environmental and economic assessment of anaerobic membrane bioreactor and disinfection for reclaimed water reuse in agricultural irrigation: A case study in Italy. J. Clean. Prod. 2021, 293, 126201. [Google Scholar] [CrossRef]
- Tomczak, W.; Gryta, M. Comparison of Polypropylene and Ceramic Microfiltration Membranes Applied for Separation of 1,3-PD Fermentation Broths and Saccharomyces cerevisiae Yeast Suspensions. Membranes 2021, 11, 44. [Google Scholar] [CrossRef]
- Jarvis, P.; Carra, I.; Jafari, M.; Judd, S. Ceramic vs polymeric membrane implementation for potable water treatment. Water Res. 2022, 215, 118269. [Google Scholar] [CrossRef]
- Yi, X.; Zhang, M.; Song, W.; Wang, X. Effect of Initial Water Flux on the Performance of Anaerobic Membrane Bioreactor: Constant Flux Mode versus Varying Flux Mode. Membranes 2021, 11, 203. [Google Scholar] [CrossRef]
- Annuar, A.M.; Nawi, N.I.M.; Bilad, M.R.; Jaafar, J.; Marbelia, L.; Nandianto, A.B.D. Improved bubbling for membrane fouling control in filtration of palm oil mill effluent anaerobic digester sludge. J. Water Process Eng. 2020, 36, 101350. [Google Scholar] [CrossRef]
- Tang, J.; Pu, Y.; Zeng, T.; Hu, Y.; Huang, J.; Pan, S.; Wang, X.C.; Li, Y.; Abomohra, A.E.-F. Enhanced methane production coupled with livestock wastewater treatment using anaerobic membrane bioreactor: Performance and membrane filtration properties. Bioresour. Technol. 2022, 345, 126470. [Google Scholar] [CrossRef]
- APHA. Standard Methods for the Examination of Water and Wastewater; American Public Health Association Inc.: Washington, DC, USA, 2005. [Google Scholar]
- Yang, Y.; Hu, Y.; Duan, A.; Wang, X.C.; Ngo, H.H.; Li, Y.-Y. Characterization of preconcentrated domestic wastewater toward efficient bioenergy recovery: Applying size fractionation, chemical composition and biomethane potential assay. Bioresour. Technol. 2021, 319, 124144. [Google Scholar] [CrossRef]
- Pu, Y.; Tang, J.; Wang, X.C.; Hu, Y.; Huang, J.; Zeng, Y.; Ngo, H.H.; Li, Y. Hydrogen production from acidogenic food waste fermentation using untreated inoculum: Effect of substrate concentrations. Int. J. Hydrog. Energy 2019, 44, 27272–27284. [Google Scholar] [CrossRef]
- Yang, Y.; Zang, Y.; Hu, Y.; Wang, X.C.; Ngo, H.H. Upflow anaerobic dynamic membrane bioreactor (AnDMBR) for wastewater treatment at room temperature and short HRTs: Process characteristics and practical applicability. Chem. Eng. J. 2020, 383, 123186. [Google Scholar] [CrossRef]
- Xiong, J.; Yu, S.; Hu, Y.; Yang, Y.; Wang, X. Applying a dynamic membrane filtration (DMF) process for domestic wastewater preconcentration: Organics recovery and bioenergy production potential analysis. Sci. Total Environ. 2019, 680, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Aslam, M.; McCarty, P.L.; Shin, C.; Bae, J.; Kim, J. Low energy single-staged anaerobic fluidized bed ceramic membrane bioreactor (AFCMBR) for wastewater treatment. Bioresour. Technol. 2017, 240, 33–41. [Google Scholar] [CrossRef] [PubMed]
- Aslam, A.; Khan, S.J.; Shahzad, H.M.A. Anaerobic membrane bioreactors (AnMBRs) for municipal wastewater treatment- potential benefits, constraints, and future perspectives: An updated review. Sci. Total Environ. 2022, 802, 149612. [Google Scholar] [CrossRef]
- Shao, S.; Feng, Y.; Yu, H.; Li, J.; Li, G.; Liang, H. Presence of an adsorbent cake layer improves the performance of gravity-driven membrane (GDM) filtration system. Water Res. 2017, 108, 240–249. [Google Scholar] [CrossRef]
- Ji, J.; Kakade, A.; Yu, Z.; Khan, A.; Liu, P.; Li, X. Anaerobic membrane bioreactors for treatment of emerging contaminants: A review. J. Environ. Manag. 2020, 270, 110913. [Google Scholar] [CrossRef]
- Xu, Y.; Wang, M.; Yu, Q.; Zhang, Y. Enhancing methanogenesis from anaerobic digestion of propionate with addition of Fe oxides supported on conductive carbon cloth. Bioresour. Technol. 2020, 302, 122796. [Google Scholar] [CrossRef]
- Li, Q.; Liu, Y.; Yang, X.; Zhang, J.; Lu, B.; Chen, R. Kinetic and thermodynamic effects of temperature on methanogenic degradation of acetate, propionate, butyrate and valerate. Chem. Eng. J. 2020, 396, 125366. [Google Scholar] [CrossRef]
- Balcıoğlu, G.; Yilmaz, G.; Gönder, Z.B. Evaluation of anaerobic membrane bioreactor (AnMBR) treating confectionery wastewater at long-term operation under different organic loading rates: Performance and membrane fouling. Chem. Eng. J. 2021, 404, 126261. [Google Scholar] [CrossRef]
- Ji, J.; Ni, J.; Ohtsu, A.; Isozumi, N.; Hu, Y.; Du, R.; Chen, Y.; Qin, Y.; Kubota, K.; Li, Y.-Y. Important effects of temperature on treating real municipal wastewater by a submerged anaerobic membrane bioreactor: Removal efficiency, biogas, and microbial community. Bioresour. Technol. 2021, 336, 125306. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Xie, L.; Luo, G.; Zhou, Q.; Angelidaki, I. Performance and microbial community analysis of the anaerobic reactor with coke oven gas biomethanation and in situ biogas upgrading. Bioresour. Technol. 2013, 146, 234–239. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shao, Z.; Guo, X.; Qu, Q.; Kang, K.; Su, Q.; Wang, C.; Qiu, L. Effects of chlorine disinfectants on the microbial community structure and the performance of anaerobic digestion of swine manure. Bioresource Technology 2021, 339, 125576. [Google Scholar] [CrossRef] [PubMed]
- Fu, J.; Yan, B.; Gui, S.; Fu, Y.; Xia, S. Anaerobic co-digestion of thermo-alkaline pretreated microalgae and sewage sludge: Methane potential and microbial community. J. Environ. Sci. 2023, 127, 133–142. [Google Scholar] [CrossRef]
- Pasalari, H.; Gholami, M.; Rezaee, A.; Esrafili, A.; Farzadkia, M. Perspectives on microbial community in anaerobic digestion with emphasis on environmental parameters: A systematic review. Chemosphere 2021, 270, 128618. [Google Scholar] [CrossRef]
- Ma, G.; Chen, Y.; Ndegwa, P. Association between methane yield and microbiota abundance in the anaerobic digestion process: A meta-regression. Renew. Sustain. Energy Rev. 2021, 135, 110212. [Google Scholar] [CrossRef]
- Zhang, L.; Loh, K.-C.; Zhang, J.; Mao, L.; Tong, Y.W.; Wang, C.-H.; Dai, Y. Three-stage anaerobic co-digestion of food waste and waste activated sludge: Identifying bacterial and methanogenic archaeal communities and their correlations with performance parameters. Bioresour. Technol. 2019, 285, 121333. [Google Scholar] [CrossRef]
- Niel, K.A.V. Sporosarcina. In Bergey’s Manual of Systematics of Archaea and Bacteria; Trujillo, M.E., Dedysh, S., DeVos, P., Hedlund, B., Kämpfer, P., Rainey, F.A., Whitman, W.B., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2015. [Google Scholar] [CrossRef]
- Zhang, L.; Tsui, T.-H.; Loh, K.-C.; Dai, Y.; Tong, Y.W. Effects of plastics on reactor performance and microbial communities during acidogenic fermentation of food waste for production of volatile fatty acids. Bioresour. Technol. 2021, 337, 125481. [Google Scholar] [CrossRef]
- Juni, E. Psychrobacter. In Bergey’s Manual of Systematics of Archaea and Bacteria; Trujillo, M.E., Dedysh, S., DeVos, P., Hedlund, B., Kämpfer, P., Rainey, F.A., Whitman, W.B., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2015. [Google Scholar] [CrossRef]
- Lin, R.; Cheng, J.; Ding, L.; Murphy, J.D. Improved efficiency of anaerobic digestion through direct interspecies electron transfer at mesophilic and thermophilic temperature ranges. Chem. Eng. J. 2018, 350, 681–691. [Google Scholar] [CrossRef]
- Pretel, R.; Durán, F.; Robles, A.; Ruano, M.V.; Ribes, J.; Serralta, J.; Ferrer, J. Designing an AnMBR-based WWTP for energy recovery from urban wastewater: The role of primary settling and anaerobic digestion. Sep. Purif. Technol. 2015, 156, 132–139. [Google Scholar] [CrossRef]



| Reactor | Accumulative Methane Volume (mL) | Retardation Time (h) | Maximal Methane Yiled (mL-CH4/h) | SMAvalue (mg-COD/g-VSS·h) |
|---|---|---|---|---|
| AnMBR | 17.2 ± 1.1 | 6.9 ± 0.2 | 1.02 ± 0.03 | 5.71 ± 0.8 |
| UASB | 15.8 ± 2.6 | 7.3 ± 0.1 | 0.83 ± 0.04 | 5.04 ± 0.6 |
| Type | Samples | Reads | OTUs | Shannon | Chao | Ace | Coverage |
|---|---|---|---|---|---|---|---|
| Bacteria | AnMBR | 56,542 | 711 | 3.61 | 804.59 | 813.76 | 0.998 |
| UASB | 52,226 | 612 | 2.78 | 715.21 | 726.47 | 0.998 | |
| Archaea | AnMBR | 65,678 | 53 | 0.58 | 54.67 | 57.14 | 0.999 |
| UASB | 73,942 | 50 | 0.36 | 52 | 53.31 | 0.999 |
| Parameters | AnMBR | UASB |
|---|---|---|
| Energy consumption | ||
| Influent flow rate (Q, 10−9 m3/s) | 3.01 | 3.01 |
| Water head (h, m) | 1.20 | 1.20 |
| Energy needed for influent (10−3 kWh/m3) | 5.04 | 5.04 |
| Effluent flow rate (Q, 10−9 m3/s) | 3.01 | 3.01 |
| Water head (h, m) | 2.00 | 0 |
| Energy needed for effluent (10−3 kWh/m3) | 8.40 | 0 |
| Energy for substrate mixing (kWh/m3) | 1.00 | 1.00 |
| Energy for water bath (kWh/m3) | 10.00 | 10.00 |
| Energy recovery | ||
| Methane production from wastewater (V/Q, m3/m3) | 4.70 | 3.99 |
| Energy from produced methane (kWh/m3) | 14.05 | 11.93 |
| Net energy recovery (kWh/m3) | 3.03 | 0.92 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pu, Y.; Tang, J.; Zeng, T.; Hu, Y.; Yang, J.; Wang, X.; Huang, J.; Abomohra, A. Pollutant Removal and Energy Recovery from Swine Wastewater Using Anaerobic Membrane Bioreactor: A Comparative Study with Up-Flow Anaerobic Sludge Blanket. Water 2022, 14, 2438. https://doi.org/10.3390/w14152438
Pu Y, Tang J, Zeng T, Hu Y, Yang J, Wang X, Huang J, Abomohra A. Pollutant Removal and Energy Recovery from Swine Wastewater Using Anaerobic Membrane Bioreactor: A Comparative Study with Up-Flow Anaerobic Sludge Blanket. Water. 2022; 14(15):2438. https://doi.org/10.3390/w14152438
Chicago/Turabian StylePu, Yunhui, Jialing Tang, Ting Zeng, Yisong Hu, Jixiang Yang, Xiaochang Wang, Jin Huang, and Abdelfatah Abomohra. 2022. "Pollutant Removal and Energy Recovery from Swine Wastewater Using Anaerobic Membrane Bioreactor: A Comparative Study with Up-Flow Anaerobic Sludge Blanket" Water 14, no. 15: 2438. https://doi.org/10.3390/w14152438









