Synergistically Improved Catalytic Ozonation Process Using Iron-Loaded Activated Carbons for the Removal of Arsenic in Drinking Water
Abstract
:1. Introduction
2. Experimentation
2.1. Materials and Reagents
2.2. Sampling Study area and Analysis
2.3. Catalyst Preparation
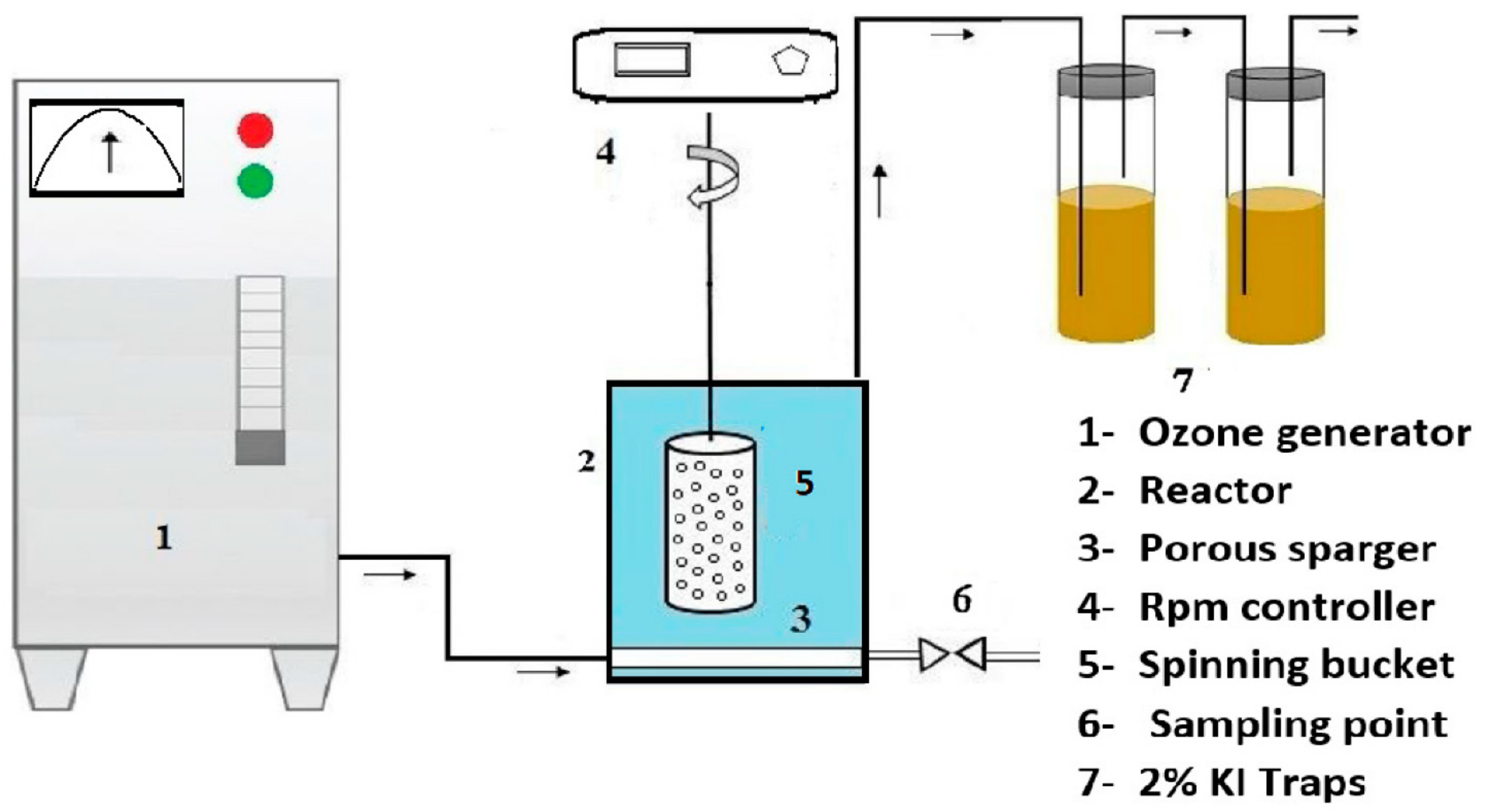
2.4. Ozonation and Catalytic Ozonation
2.5. Determination of Ozone Dose
3. Results and Discussion
3.1. Characterizations
3.2. Treatment of Arsenic
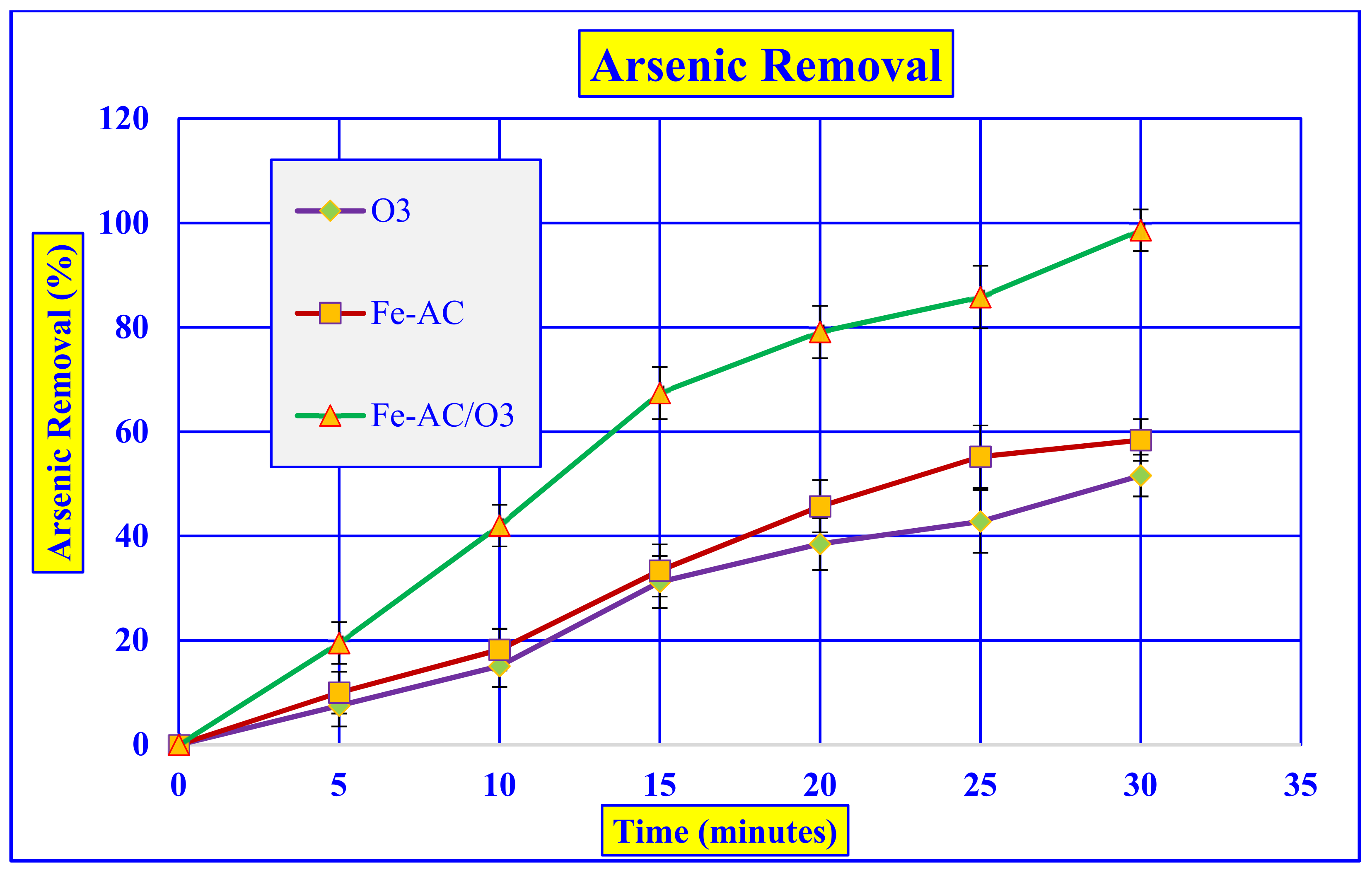
3.2.1. Comparison of O3, Adsorption, and Catalytic Ozonation Processes
3.2.2. Fe-AC Dose Effect
3.2.3. Ozone Dose Effect
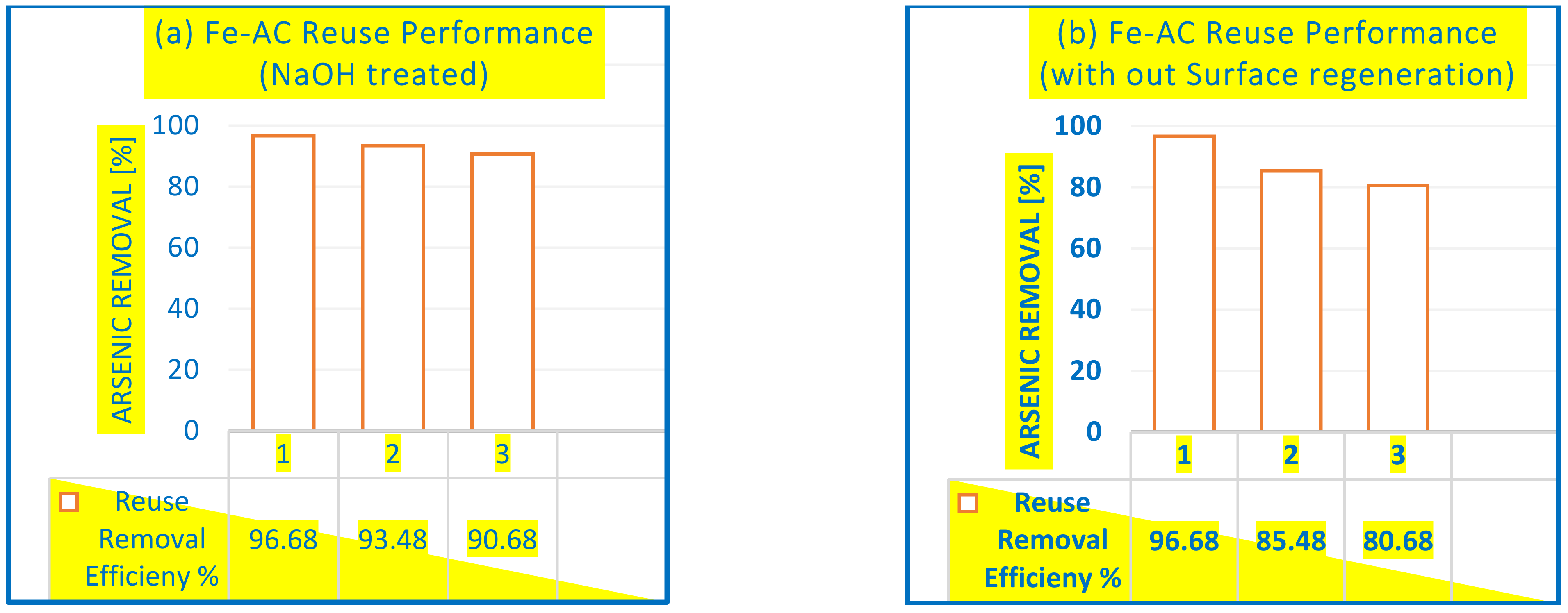
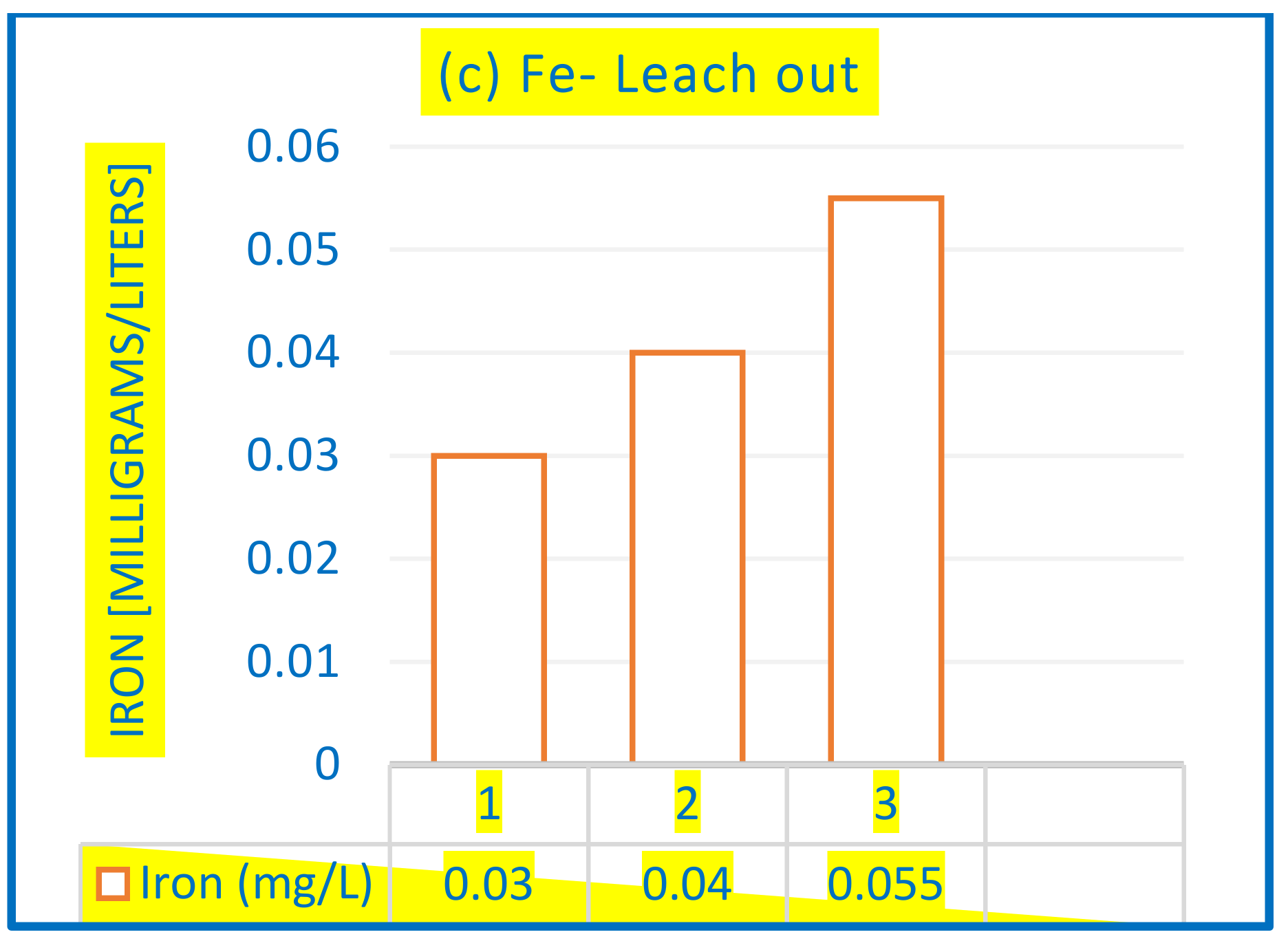
3.2.4. Removal of As in Fe-AC/O3 from Drinking Water
3.3. Arsenic Removal Mechanism on Fe-AC/O3 Process
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Javaid, R.; Qazi, U.Y. Catalytic oxidation process for the degradation of synthetic dyes: An overview. Int. J. Environ. Res. Public Health 2019, 16, 2066. [Google Scholar] [CrossRef] [Green Version]
- Masood, Z.; Ikhlaq, A.; Akram, A.; Qazi, U.Y.; Rizvi, O.S.; Javaid, R.; Alazmi, A.; Madkour, M.; Qi, F. Application of Nanocatalysts in Advanced Oxidation Processes for Wastewater Purification: Challenges and Future Prospects. Catalysts 2022, 12, 741. [Google Scholar] [CrossRef]
- Ikhlaq, A.; Javed, F.; Akram, A.; Qazi, U.Y.; Masood, Z.; Ahmed, T.; Arshad, Z.; Khalid, S.; Qi, F. Treatment of leachate through constructed wetlands using Typha angustifolia in combination with catalytic ozonation on Fe-zeolite A. Int. J. Phytoremediation 2021, 23, 809–817. [Google Scholar] [CrossRef]
- Azizullah, A.; Khattak, M.N.K.; Richter, P.; Hader, D.-P. Water pollution in Pakistan and its impact on public healthea review. Environ. Int. 2011, 37, 491–497. [Google Scholar] [CrossRef]
- Javaid, R.; Qazi, U.Y.; Kawasaki, S. Highly efficient decomposition of Remazol Brilliant Blue R using tubular reactor coated with thin layer of PdO. J. Environ. Manag. 2016, 180, 551–556. [Google Scholar] [CrossRef]
- Madkour, M.; Abdelmonem, Y.; Qazi, U.Y.; Javaid, R.; Vadivel, S. Efficient Cr(vi) photoreduction under natural solar irradiation using a novel step-scheme ZnS/SnIn4S8 nanoheterostructured photocatalysts. RSC Adv. 2021, 11, 29433–29440. [Google Scholar] [CrossRef]
- Tabasum, A.; Alghuthaymi, M.; Qazi, U.Y.; Shahid, I.; Abbas, Q.; Javaid, R.; Nadeem, N.; Zahid, M. UV-Accelerated Photocatalytic Degradation of Pesticide over Magnetite and Cobalt Ferrite Decorated Graphene Oxide Composite. Plants 2020, 10, 6. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Hu, C.; Nie, Y.; Qu, J. Catalytic ozonation of selected pharmaceuticals over mesoporous alumina-supported manganese oxide. Environ. Sci. Technol. 2009, 43, 2525–2529. [Google Scholar] [CrossRef] [PubMed]
- Javaid, R.; Qazi, U.Y.; Ikhlaq, A.; Zahid, M.; Alazmi, A. Subcritical and supercritical water oxidation for dye decomposition. J. Environ. Manag. 2021, 290, 112605. [Google Scholar] [CrossRef] [PubMed]
- Javaid, R.; Kawasaki, S.; Ookawara, R.; Sato, K.; Nishioka, M.; Suzuki, A.; Suzuki, T.M. Continuous dehydrogenation of aqueous formic acid under sub-critical conditions by use of hollow tubular reactor coated with thin palladium oxide layer. J. Chem. Eng. Jpn. 2013, 46, 751–758. [Google Scholar] [CrossRef]
- Javaid, R.; Kawanami, H.; Chatterjee, M.; Ishizaka, T.; Suzuki, A.; Suzuki, T.M. Fabrication of microtubular reactors coated with thin catalytic layer (M = Pd, Pd-Cu, Pt, Rh, Au). Catalysis Commun. 2010, 11, 1160–1164. [Google Scholar] [CrossRef]
- Rahman, M.U.; Qazi, U.Y.; Hussain, T.; Nadeem, N.; Zahid, M.; Bhatti, H.N.; Shahid, I. Solar driven photocatalytic degradation potential of novel graphitic carbon nitride based nano zero-valent iron doped bismuth ferrite ternary composite. Opt. Mater. 2021, 120, 111408. [Google Scholar] [CrossRef]
- Baig, J.A.; Kazi, T.G.; Arain, M.B.; Afridi, H.I.; Kandhro, G.A.; Sarfraz, R.A.; Jamal, M.K.; Shah, A.Q. Evaluation of arsenic and other physico-chemical parameters of surface and ground water of Jamshoro, Pakistan. J. Hazard. Mater. 2009, 166, 662–669. [Google Scholar] [CrossRef] [PubMed]
- Nickson, R.; McArthur, J.; Shrestha, B.; Kyaw-Myint, T.; Lowry, D. Arsenic and other drinking water quality issues, Muzaffargarh District, Pakistan. Appl. Geochem. 2005, 20, 55–68. [Google Scholar] [CrossRef]
- Baig, J.A.; Kazi, T.G.; Mustafa, M.A.; Solangi, I.B.; Mughal, M.J.; Afridi, H.I. Arsenic exposure in children through drinking water in different districts of Sindh, Pakistan. Biol. Trace Elem. Res. 2016, 173, 35–46. [Google Scholar] [CrossRef] [PubMed]
- Sultana, J.; Farooqi, A.; Ali, U. Assessment. Arsenic concentration variability, health risk assessment, and source identification using multivariate analysis in selected villages of public water system, Lahore, Pakistan. Environ. Monit. Assess. 2014, 186, 1241–1251. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Wei, D.; Wei, G.; Du, Y. Transformation of cefazolin during chlorination process: Products, mechanism and genotoxicity assessment. J. Hazard Mater. 2013, 262, 48–54. [Google Scholar] [CrossRef]
- Smedley, P.L.; Kinniburgh, D.G. A review of the source, behavior, and distribution of arsenic in natural water. Appl. Geochem. 2002, 17, 517–568. [Google Scholar]
- Wang, J.; Chen, H. Catalytic ozonation for water and wastewater treatment: Recent advances and perspective. Sci. Total Environ. 2020, 704, 135249. [Google Scholar] [CrossRef] [PubMed]
- Qazi, U.Y.; Iftikhar, R.; Ikhlaq, A.; Riaz, I.; Jaleel, R.; Nusrat, R.; Javaid, R. Application of Fe-RGO for the removal of dyes by catalytic ozonation process. Environ. Sci. Pollut. Res. 2022, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Charoenngam, N.; Shirvani, A.; Kalajian, T.A.; Song, A.; Holick, M.F. The effect of various doses of oral vitamin D3 supplementation on gut microbiota in healthy adults: A randomized, double-blinded, dose-response study. Anticancer Res. 2020, 40, 551–556. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Kumar, S. Arsenic exposure with reference to neurological impairment: An overview. Rev. Environ. Health 2019, 34, 403–414. [Google Scholar] [CrossRef]
- Addo Ntim, S.; Mitra, S. Removal of trace arsenic to meet drinking water standards using iron oxide coated multiwall carbon nanotubes. J. Chem. Eng. Data 2011, 56, 2077–2083. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.-M.; Zou, S.-W.; Nanayakkara, K.N.; Matsuura, T.; Chen, J.P. Adsorptive removal of arsenic from aqueous solution by a PVDF/zirconia blend flat sheet membrane. J. Membr. Sci. 2011, 374, 1–11. [Google Scholar] [CrossRef]
- Kobya, M.; Gebologlu, U.; Ulu, F.; Oncel, S.; Demirbas, E. Removal of arsenic from drinking water by the electrocoagulation using Fe and Al electrodes. Electrochim. Acta 2011, 56, 5060–5070. [Google Scholar] [CrossRef]
- Moreira, V.R.; Lebron, Y.A.R.; de Souza Santos, L.V.; Amaral, M.C.S. Dead-end ultrafiltration as a cost-effective strategy for improving arsenic removal from high turbidity waters in conventional drinking water facilities. Chem. Eng. J. 2021, 417, 128132. [Google Scholar] [CrossRef]
- Ikhlaq, A.; Fatima, R.; Qazi, U.Y.; Javaid, R.; Akram, A.; Ibn Shamsah, S.; Qi, F. Combined iron-loaded zeolites and ozone-based process for the purification of drinking water in a novel hybrid reactor: Removal of faecal coliforms and arsenic. Catalysts 2021, 11, 373. [Google Scholar] [CrossRef]
- El Araby, R.; Hawash, S.; El Diwani, G. Treatment of iron and manganese in simulated groundwater via ozone technology. Desalination 2009, 249, 1345–1349. [Google Scholar] [CrossRef]
- Boddu, V.M.; Abburi, K.; Talbott, J.L.; Smith, E.D.; Haasch, R. Removal of arsenic (III) and arsenic (V) from aqueous medium using chitosan-coated biosorbent. Water Res. 2008, 42, 633–642. [Google Scholar] [CrossRef]
- Xu, Y.-h.; Nakajima, T.; Ohki, A. Adsorption and removal of arsenic (V) from drinking water by aluminum-loaded Shirasu-zeolite. J. Hazard. Mater. 2002, 92, 275–287. [Google Scholar] [CrossRef]
- Gupta, S.K.; Chen, K.Y. Arsenic removal by adsorption. J. Water Pollut. Control. Fed. 1978, 50, 493–506. [Google Scholar]
- Vaclavikova, M.; Gallios, G.P.; Hredzak, S.; Jakabsky, S. Removal of arsenic from water streams: An overview of available techniques. Clean Technol. Environ. Policy 2008, 10, 89–95. [Google Scholar] [CrossRef]
- Nguyen, V.; Vigneswaran, S.; Ngo, H.; Shon, H.; Kandasamy, J. Arsenic removal by a membrane hybrid filtration system. Desalination 2009, 236, 363–369. [Google Scholar] [CrossRef] [Green Version]
- Brandhuber, P.; Amy, G. Alternative methods for membrane filtration of arsenic from drinking water. Desalination 1998, 117, 1–10. [Google Scholar] [CrossRef]
- Fogarassy, E.; Galambos, I.; Bekassy-Molnar, E.; Vatai, G. Treatment of high arsenic content wastewater by membrane filtration. Desalination 2009, 240, 270–273. [Google Scholar] [CrossRef]
- Ali, I.; Khan, T.A.; Asim, M. Removal of arsenic from water by electrocoagulation and electrodialysis techniques. Sep. Purif. Rev. 2011, 40, 25–42. [Google Scholar] [CrossRef]
- Nidheesh, P.; Singh, T.A. Arsenic removal by electrocoagulation process: Recent trends and removal mechanism. Chemosphere 2017, 181, 418–432. [Google Scholar] [CrossRef] [PubMed]
- Demirbas, A. Agricultural based activated carbons for the removal of dyes from aqueous solutions: A review. J. Hazard. Mater. 2009, 167, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Hug, S.J.; Canonica, L.; Wegelin, M.; Gechter, D.; Von Gunten, U. Solar oxidation and removal of arsenic at circumneutral pH in iron containing waters. Environ. Sci. Technol. 2001, 35, 2114–2121. [Google Scholar] [CrossRef]
- Bissen, M.; Frimmel, F.H. Arsenic—A review. Part II: Oxidation of arsenic and its removal in water treatment. Acta Hydrochim. Et Hydrobiol. 2003, 31, 97–107. [Google Scholar] [CrossRef]
- Fatima, N.; Qazi, U.Y.; Mansha, A.; Bhatti, I.A.; Javaid, R.; Abbas, Q.; Nadeem, N.; Rehan, Z.A.; Noreen, S.; Zahid, M. Recent developments for antimicrobial applications of graphene-based polymeric composites: A review. J. Ind. Eng. Chem. 2021, 100, 40–58. [Google Scholar] [CrossRef]
- Javaid, R.; Tanaka, D.A.P.; Kawanami, H.; Suzuki, T.M. Silica capillary with thin metal (Pd and Pt) inner wall: Application to continuous decomposition of hydrogen peroxide. Chem. Lett. 2009, 38, 146–147. [Google Scholar] [CrossRef]
- Nawrocki, J.; Kasprzyk-Hordern, B. The efficiency and mechanisms of catalytic ozonation. Appl. Catal. B Environ. 2010, 99, 27–42. [Google Scholar] [CrossRef]
- Ikhlaq, A.; Javaid, R.; Akram, A.; Qazi, U.Y.; Erfan, J.; Madkour, M.; Abdelbagi, M.E.M.; Ibn Shamsah, S.M.; Qi, F. Application of Attapulgite Clay-Based Fe-Zeolite 5A in UV-Assisted Catalytic Ozonation for the Removal of Ciprofloxacin. J. Chem. 2022, 2022, 2846453. [Google Scholar] [CrossRef]
- Irfan, M.; Zahid, M.; Tahir, N.; Yaseen, M.; Qazi, U.; Javaid, R.; Shahid, I. Enhanced photo-Fenton degradation of Rhodamine B using iodine-doped iron tungstate nanocomposite under sunlight. Int. J. Environ. Sci. Technol. 2022, 1–16. [Google Scholar] [CrossRef]
- Javaid, R.; Kawasaki, S.-i.; Suzuki, A.; Suzuki, T.M. Simple and rapid hydrogenation of p-nitrophenol with aqueous formic acid in catalytic flow reactors. Beilstein J. Org. Chem. 2013, 9, 1156–1163. [Google Scholar] [CrossRef] [Green Version]
- Preočanin, T.; Kallay, N. Point of zero charge and surface charge density of TiO2 in aqueous electrolyte solution as obtained by potentiometric mass titration. Croat. Chem. Acta 2006, 79, 95–106. [Google Scholar]
- Eaton, A.D.; Clesceri, L.S.; Rice, E.W.; Greenberg, A.E.; Franson, M.A.H. Standard Methods for the Examination of Water and Wastewater, 21st ed.; American Public Health Association: Washington, DC, USA; American Water Works Association: Denver, CO, USA; Water Environment Federation: Alexandria, VA, USA, 2005. [Google Scholar]
- Mahmood, K.; Rana, A.D.; Tariq, S.; Kanwal, S.; Ali, R.; Haidar, A. Groundwater levels susceptibility to degradation in Lahore metropolitan. Depression 2013, 25, 123–126. [Google Scholar]
- Mahmood, S.; Irshad, A.; Nasir, J.M.; Sharif, F.; Farooqi, S.H. Spatiotemporal analysis of dengue outbreaks in Samanabad town, Lahore metropolitan area, using geospatial techniques. Environ. Monit. Assess. 2019, 191, 55. [Google Scholar] [CrossRef]
- Su, Y.-F.; Cheng, Y.-L.; Shih, Y.-H. Removal of trichloroethylene by zerovalent iron/activated carbon derived from agricultural wastes. J. Environ. Manag. 2013, 129, 361–366. [Google Scholar] [CrossRef] [PubMed]
- Kazmi, M.; Hayder, S.; Rashid, H.; Rustam, M.; Sulheri, A.W.; Saeed, A. Evaluation of Drinking water quality parameters in the areas of East-Lahore Pakistan: A case study. J. Fac. Eng. Technol. 2015, 21, 41–53. [Google Scholar]
- Ikhlaq, A.; Brown, D.R.; Kasprzyk-Hordern, B. Mechanisms of catalytic ozonation on alumina and zeolites in water: Formation of hydroxyl radicals. Appl. Catal. B Environ. 2012, 123, 94–106. [Google Scholar] [CrossRef]
- Xu, Y.; Wang, Q.; Yoza, B.A.; Li, Q.X.; Kou, Y.; Tang, Y.; Ye, H.; Li, Y.; Chen, C. Catalytic ozonation of recalcitrant organic chemicals in water using vanadium oxides loaded ZSM-5 zeolites. Front. Chem. 2019, 7, 384. [Google Scholar] [CrossRef] [PubMed]
- Ikhlaqa, A.; Aslama, T.; Zafara, A.M.; Javedb, F.; Munirc, H.M.S. Combined ozonation and adsorption system for the removal of heavy metals from municipal wastewater: Effect of COD removal. Desalination Water Treat. 2019, 159, 304–309. [Google Scholar] [CrossRef] [Green Version]
- Payne, K.B.; Abdel-Fattah, T.M. Adsorption of arsenate and arsenite by iron-treated activated carbon and zeolites: Effects of pH, temperature, and ionic strength. J. Environ. Sci. Health 2005, 40, 723–749. [Google Scholar] [CrossRef] [PubMed]
- Hou, Y.; Li, X.; Zhao, Q.; Chen, G.; Raston, C.L. Role of hydroxyl radicals and mechanism of Escherichia coli inactivation on Ag/AgBr/TiO2 nanotube array electrode under visible light irradiation. Environ. Sci. Technol. 2012, 46, 4042–4050. [Google Scholar] [CrossRef]
- Moldovan, M.; Gomez, M.M.; Palacios, M.A.; Cámara, C. Arsenic speciation in water and human urine by HPLC–ICP-MS and HPLC–MO–HG-AAS. Microchem. J. 1998, 59, 89–99. [Google Scholar] [CrossRef]
- Einaga, H.; Futamura, S. Catalytic oxidation of benzene with ozone over Mn ion-exchanged zeolites. Catal. Commun. 2007, 8, 557–560. [Google Scholar] [CrossRef]
- Li, Z.; Jean, J.-S.; Jiang, W.-T.; Chang, P.-H.; Chen, C.-J.; Liao, L. Removal of arsenic from water using Fe-exchanged natural zeolite. J. Hazard. Mater. 2011, 187, 318–323. [Google Scholar] [CrossRef]
- Kim, M.-J.; Nriagu, J. Oxidation of arsenite in groundwater using ozone and oxygen. Sci. Total Environ. 2000, 247, 71–79. [Google Scholar] [CrossRef]
- Zhu, H.; Jia, Y.; Wu, X.; Wang, H. Removal of arsenic from water by supported nano zero-valent iron on activated carbon. J. Hazard. Mater. 2009, 172, 1591–1596. [Google Scholar] [CrossRef]
- Nishimura, T.; Umetsu, Y. Oxidative precipitation of arsenic(III) with manganese(II) and iron(II) in dilute acidic solution by ozone. Hydrometallurgy 2001, 62, 83–92. [Google Scholar] [CrossRef]
- Du, J.; Zhang, B.; Li, J.; Lai, B. Decontamination of heavy metal complexes by advanced oxidation processes: A review. Chin. Chem. Lett. 2020, 31, 2575–2582. [Google Scholar] [CrossRef]
- Xiong, W.; Chen, N.; Feng, C.; Liu, Y.; Ma, N.; Deng, J.; Xing, L.; Gao, Y. Ozonation catalyzed by iron-and/or manganese-supported granular activated carbons for the treatment of phenol. Environ. Sci. Pollut. Res. 2019, 26, 21022–21033. [Google Scholar] [CrossRef]
- Javaid, R.; Qazi, U.Y.; Kawasaki, S. Efficient and continuous decomposition of hydrogen peroxide using silica capillary coated with thin palladium or platinum layer. Bull. Chem. Soc. Jpn. 2015, 88, 976–980. [Google Scholar] [CrossRef] [Green Version]
- Qazi, U.Y.; Javaid, R.; Tahir, N.; Jamil, A.; Afzal, A. Design of advanced self-supported electrode by surface modificationof copper foam with transition metals for efficient hydrogen evolution reaction. Int. J. Hydrog. Energy 2020, 45, 33396–33406. [Google Scholar] [CrossRef]
- Arshad, Z.; Shakir, S.; Khoja, A.H.; Javed, A.H.; Anwar, M.; Rehman, A.; Javaid, R.; Qazi, U.Y.; Farrukh, S. Performance analysis of calcium-dopped titania (TiO2) as an effective electron transport layer (ETL) for pervoskite solar cells. Energies 2022, 15, 1408. [Google Scholar] [CrossRef]
- Qazi, U.Y.; Shervani, Z.; Javaid, R.; Kajimoto, S.; Furukawa, H. Formation and growth of silver nanocubes upon nanosecond pulsed laser irradiation: Effects of laser intensity and irradiation time. Adv. Nanoparticles 2017, 6, 148–157. [Google Scholar] [CrossRef] [Green Version]
- Javaid, R.; Kawanami, H.; Chatterjee, M.; Ishizaka, T.; Suzuki, A.; Suzuki, T.M. Sonogashira C-C coupling reaction in water using tubular reactors with catalytic metallic inner surface. Chem. Eng. J. 2011, 167, 431–435. [Google Scholar] [CrossRef]
- Pan, L.; Cao, Y.; Zang, J.; Huang, Q.; Wang, L.; Zhang, Y.; Fan, S.; Tang, J.; Xie, Z. Preparation of iron-loaded granular activated carbon catalyst and its application in tetracycline antibiotic removal from aqueous solution. Int. J. Environ. Res. Public Health 2019, 16, 2270. [Google Scholar] [CrossRef] [Green Version]
- Javaid, R. Catalytic hydrogen production, storage and application. Catalysts 2021, 11, 836. [Google Scholar] [CrossRef]
- Afzal, A.; Abuilaiwi, F.A.; Javaid, R.; Ali, F.; Habib, A. Solid-state synthesis of heterogeneous Ni0.5Cu0.5-xZnxFe2O4 spinel oxides with controlled morphologyand tunable dielectric properties. J. Mater. Sci. Mater. Electron. 2020, 31, 14261–14270. [Google Scholar] [CrossRef]
- Tahir, N.; Tahir, F.; Qazi, U.Y.; Javaid, R.; Zhang, Q. Enhancing biohydrogen production from lignocellulosic biomass of paulownia waste by charge facilitation in Zn doped SnO2 nanocatalysts. Bioresour. Technol. 2022, 355, 127299. [Google Scholar] [CrossRef]
- Qazi, U.Y.; Javaid, R. A review on metal nanostructures: Preparation methods and their potential applications. Adv. Nanoparticles 2016, 5, 27–43. [Google Scholar] [CrossRef] [Green Version]


| Treatment Methods | Material | Removal Efficiency | Reference |
|---|---|---|---|
| Adsorption | Iron oxide-multiwalled carbon nanotube (Fe-MWCNT) | Adsorption capacity; As(III)1723 μg/g and As(V) 189 μg·/g | [23] |
| Adsorption on Membrane Material | PVDF/zirconia blend flat sheet membrane | 21.5 mg/g | [24] |
| Electrocoagulation | Fe and Al electrodes | Fe electrode (93.5%) and 12.5–15 min for Al electrode (95.7%) | [25] |
| Coagulation, Flocculation/ Dead-end ultrafiltration | FeCl3/polyetherimide-based membrane | 96.9% | [26] |
| Catalytic ozonation/Adsorption | Iron coated zeolite 4Å | 100% in 15 mins | [27] |
| Material | Surface Area BET (m2/g) | Pore Size (Å) | Iron Content [%] | Point of Zero Charges (pHpzc) |
|---|---|---|---|---|
| Fe-AC | 1100 | 9.3 | 5.28 | 8.8 ± 0.2 |
| AC | 1050 | 9.1 | - | 9 ± 0.1 |
| No. | Sample Name | Sample Code | pH | Arsenic (μg/L) | Turbidity (NTU) | Chlorides (mg/L) | Fecal Coliforms MPN/100 mL | Total Dissolved Solids (TDS) mg/L | Hardness as CaCO3 |
|---|---|---|---|---|---|---|---|---|---|
| 1 | Mughal Pura | MU 1 | 7.6 ± 0.2 | 58 ± 8 | 1.6 ± 0.2 | 48 ± 7 | 12 ± 4 | 750 ± 55 | 383 ± 25 |
| 2 | Mughal Pura | MU 2 | 7.8 ± 0.3 | 45 ± 4 | 2.6 ± 0.5 | 52 ± 11 | NIL | 648 ± 45 | 590 ± 50 |
| 3 | Mughal Pura | MU 3 | 8.0 ± 0.3 | 55 ± 10 | 1.5 ± 0.5 | 75 ± 15 | NIL | 591 ± 39 | 423 ± 21 |
| 4 | Mehmood Booti abad | ME 1 | 8.1 ± 0.3 | 75 ± 7 | 2.3 ± 0.2 | 32 ± 9 | 18 ± 7 | 715 ± 45 | 517 ± 39 |
| 5 | Mehmood Booti abad | ME 2 | 7.5 ± 0.2 | 66 ± 12 | 2.6 ± 0.2 | 88 ± 7 | 15 ± 5 | 880 ± 55 | 664 ± 22 |
| 6 | Mehmood Booti abad | ME 3 | 7.9 ± 0.1 | 68 ± 4 | 2.9 ± 0.5 | 52 ± 15 | NIL | 832 ± 35 | 465 ± 23 |
| 7 | Saggiyian pull | SG 1 | 8.3 ± 0.3 | 44 ± 5 | 2.6 ± 0.2 | 39 ± 7 | NIL | 694 ± 29 | 396 ± 21 |
| 8 | Saggiyian pull | SG 2 | 8.2 ± 0.1 | 41 ± 6 | 1.6 ± 0.2 | 48 ± 3 | NIL | 731 ± 55 | 473 ± 25 |
| 9 | Saggiyian pull | SG 3 | 8.2 ± 0.1 | 58 ± 8 | 0.6 ± 0.3 | 58 ± 5 | 10 ± 3 | 845 ± 55 | 511 ± 25 |
| WHO Guidelines/EPA/ European | 6.5–8.5 | 10 | <5 | 250 | NIL | 1000 | 500 | ||
| PEQS | 6.5–8.5 | 50 | <5 | 250 | NIL | 1000 | 500 | ||
| Sr# | Sample Name | Sample Code | Arsenic before Treatment (μg/L) | Arsenic after Treatment (μg/L) |
|---|---|---|---|---|
| 1 | Mughal Pura | MU 1 | 58 ± 8 | 5.8 ± 2 |
| 2 | Mughal Pura | MU 2 | 45 ± 4 | 3.5 ± 0.4 |
| 3 | Mughal Pura | MU 3 | 55 ± 10 | 4.1 ± 0.3 |
| 4 | Mehmood Booti abad | ME 1 | 75 ± 7 | 6.5 ± 2 |
| 5 | Mehmood Booti abad | ME 2 | 66 ± 12 | 6.9 ± 2 |
| 6 | Mehmood Booti abad | ME 3 | 68 ± 4 | 4.4 ± 1 |
| 7 | Saggiyian pull | SG 1 | 44 ± 5 | 2.7 ± 1 |
| 8 | Saggiyian pull | SG 2 | 41 ± 6 | 2.6 ± 1 |
| 9 | Saggiyian pull | SG 3 | 58 ± 8 | 5.6 ± 1 |
| WHO Guidelines/EPA/European | 10 | |||
| PEQS | 50 | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qazi, U.Y.; Javaid, R.; Ikhlaq, A.; Al-Sodani, K.A.A.; Rizvi, O.S.; Alazmi, A.; Asiri, A.M.; Ibn Shamsah, S.M. Synergistically Improved Catalytic Ozonation Process Using Iron-Loaded Activated Carbons for the Removal of Arsenic in Drinking Water. Water 2022, 14, 2406. https://doi.org/10.3390/w14152406
Qazi UY, Javaid R, Ikhlaq A, Al-Sodani KAA, Rizvi OS, Alazmi A, Asiri AM, Ibn Shamsah SM. Synergistically Improved Catalytic Ozonation Process Using Iron-Loaded Activated Carbons for the Removal of Arsenic in Drinking Water. Water. 2022; 14(15):2406. https://doi.org/10.3390/w14152406
Chicago/Turabian StyleQazi, Umair Yaqub, Rahat Javaid, Amir Ikhlaq, Khaled A. Alawi Al-Sodani, Osama Shaheen Rizvi, Amira Alazmi, Abdullah Mohamed Asiri, and Sami M. Ibn Shamsah. 2022. "Synergistically Improved Catalytic Ozonation Process Using Iron-Loaded Activated Carbons for the Removal of Arsenic in Drinking Water" Water 14, no. 15: 2406. https://doi.org/10.3390/w14152406











