Removal of Phosphate from Wastewater with a Recyclable La-Based Particulate Adsorbent in a Small-Scale Reactor
Abstract
:Highlights
- Tailored La-based particles (La-CZ) sorb phosphate in wastewater.
- The adsorption capacity of La-CZ particles was 18.2 mg g−1 (PO43−).
- The adsorbed phosphate mainly existed in a stable state.
- La-CZ particles were recovered easily in the small-scale reactor designed in this study.
- Effluent concentrations <0.1 mg/L PO43− were achieved in the reactor when treating river water.
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of Adsorbents
2.2. Characterization of the Adsorbents
2.3. Adsorption Batch Studies
2.4. Fractionation of the P-Saturated Absorbent
3. Results and Discussion
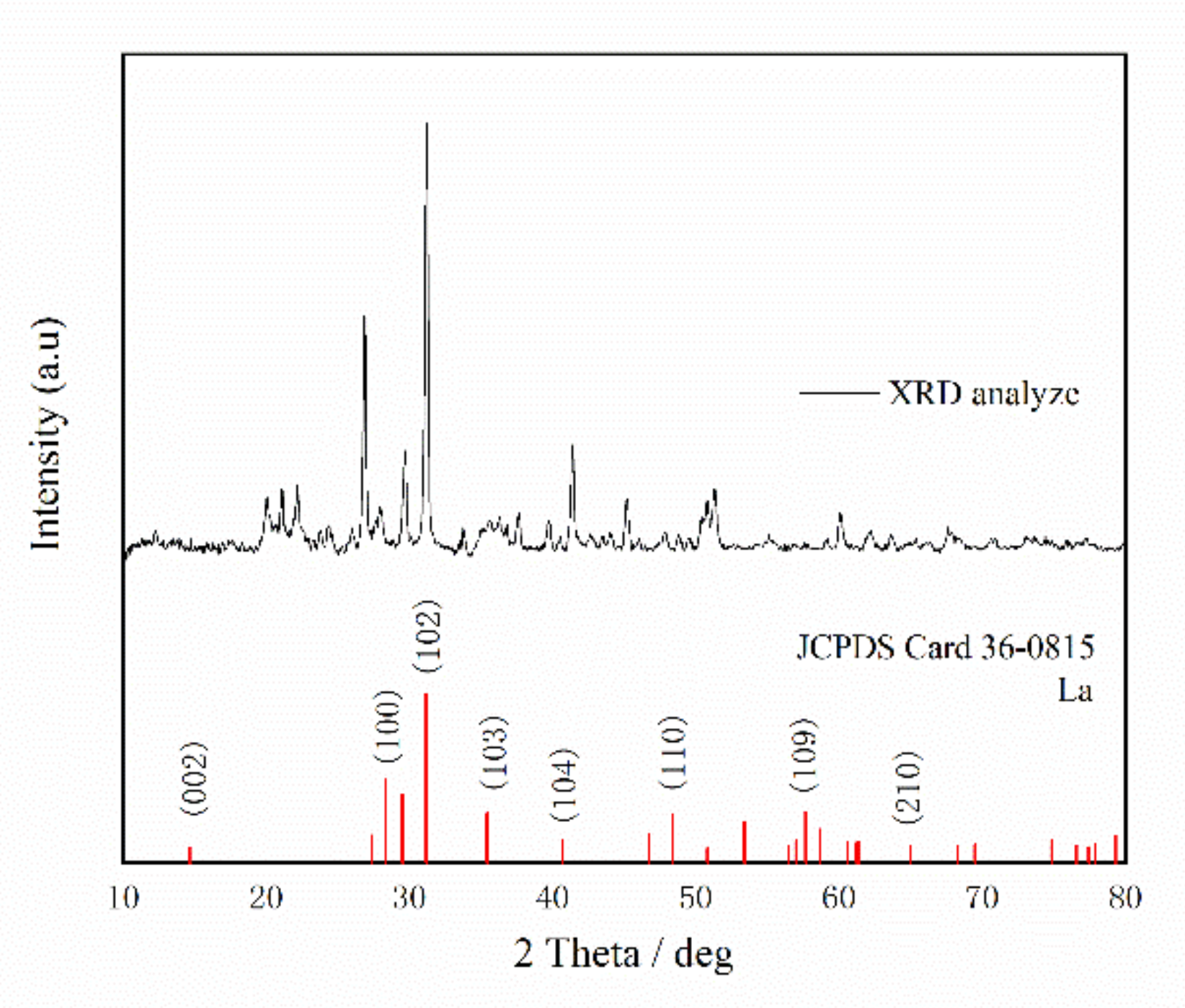
3.1. Absorbent Characterization
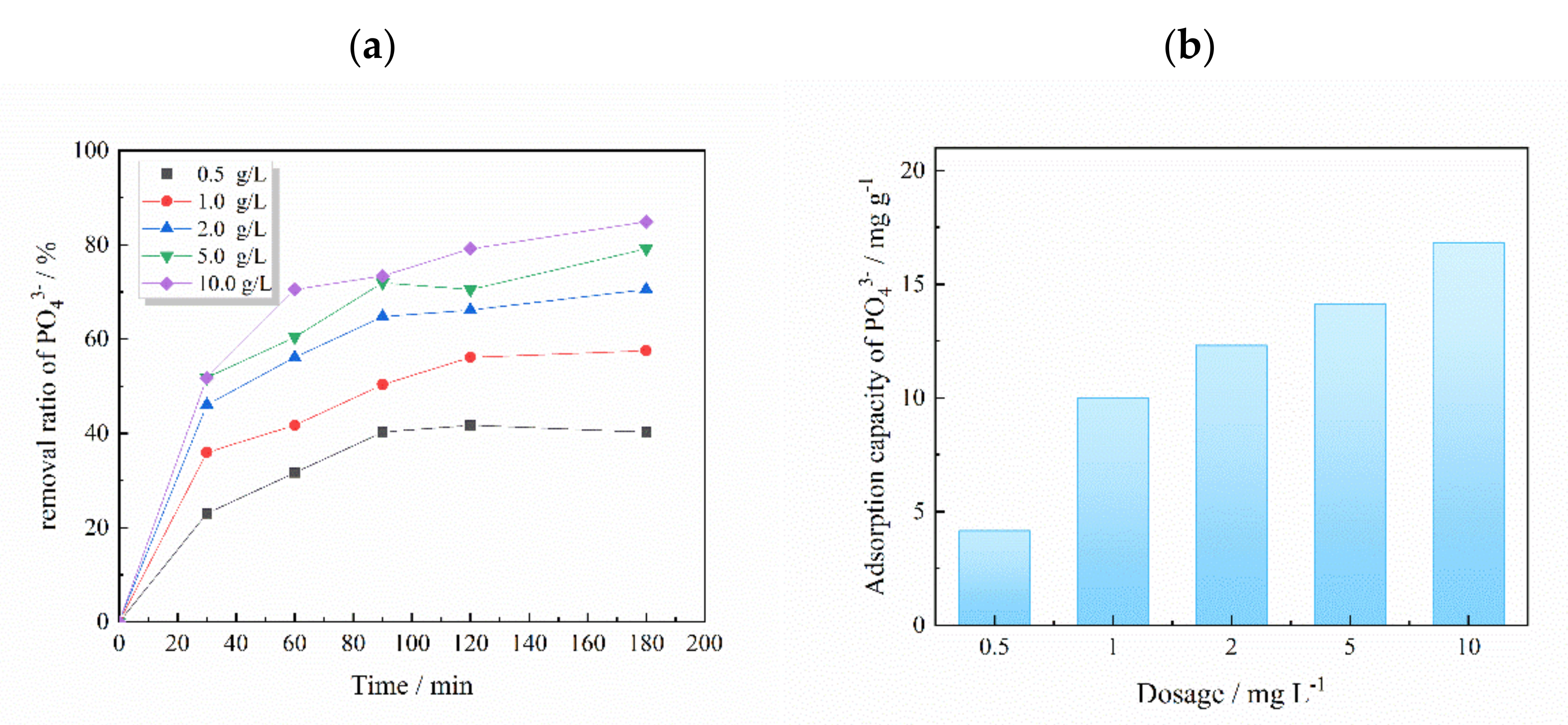
3.2. The Adsorption Capacity of La-CZ
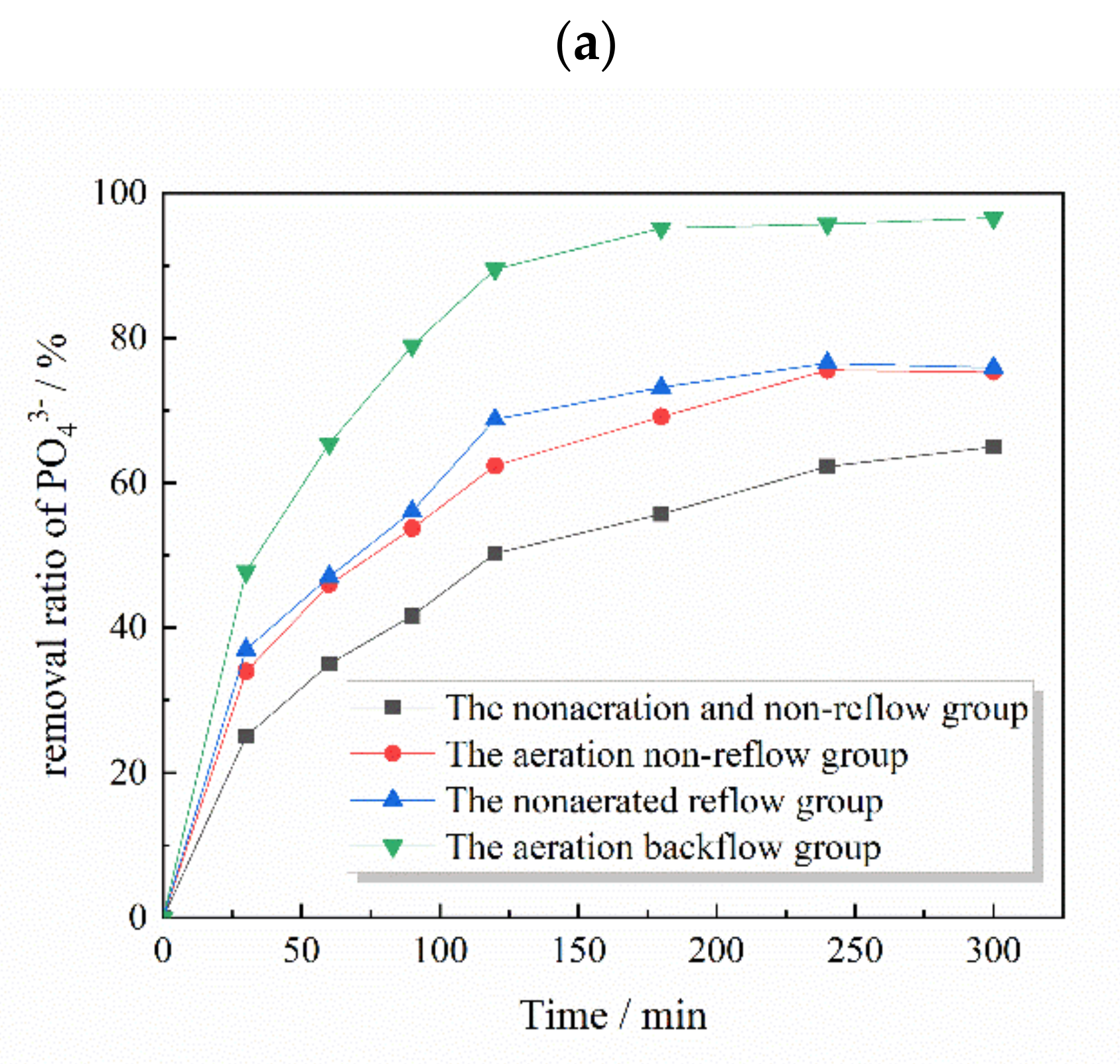
3.3. Matrix Effects on Phosphate Removal Capacity
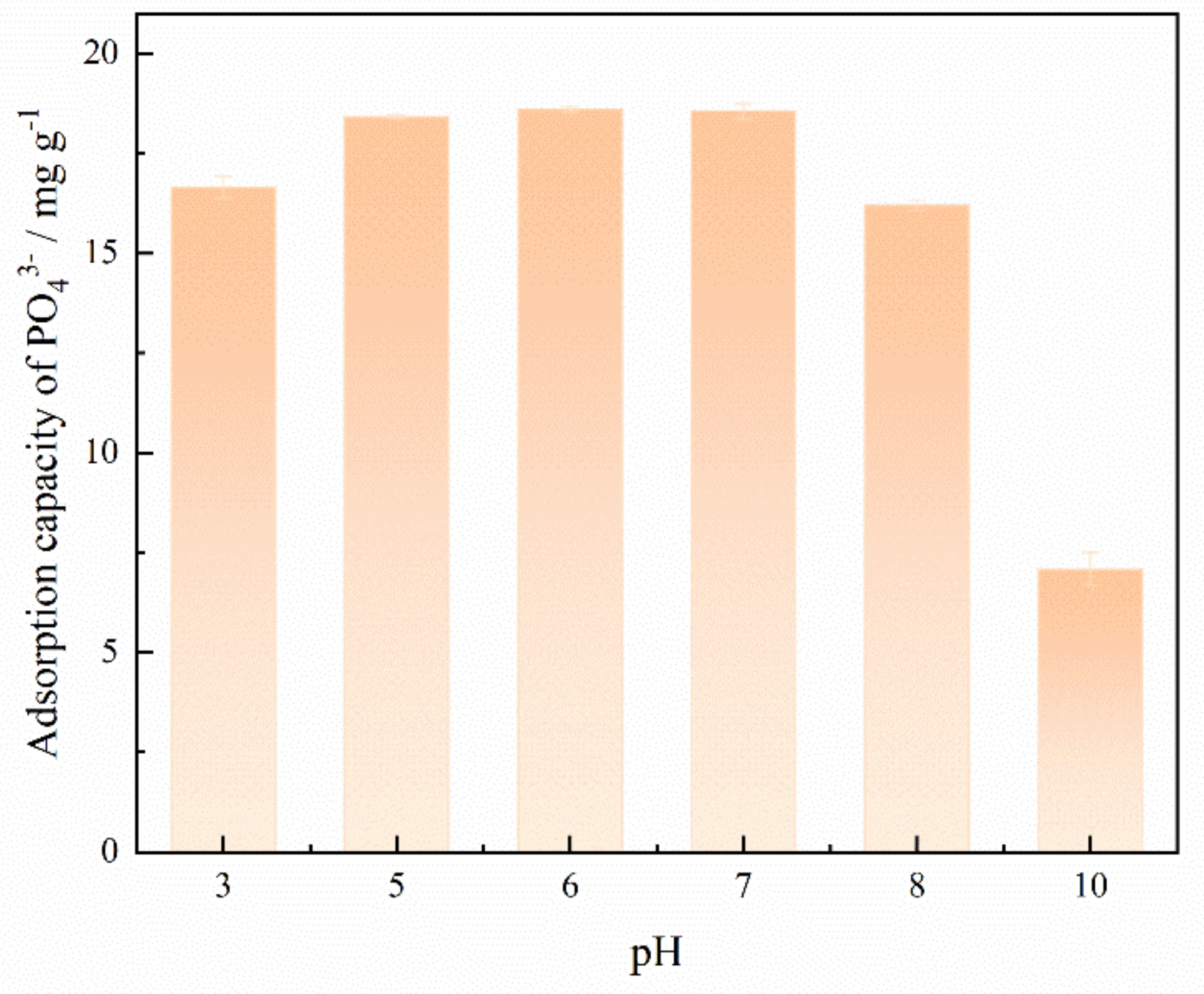
3.3.1. Effect of pH
3.3.2. Effect of Coexisting Anions
3.4. Phosphate Adsorption Mechanism
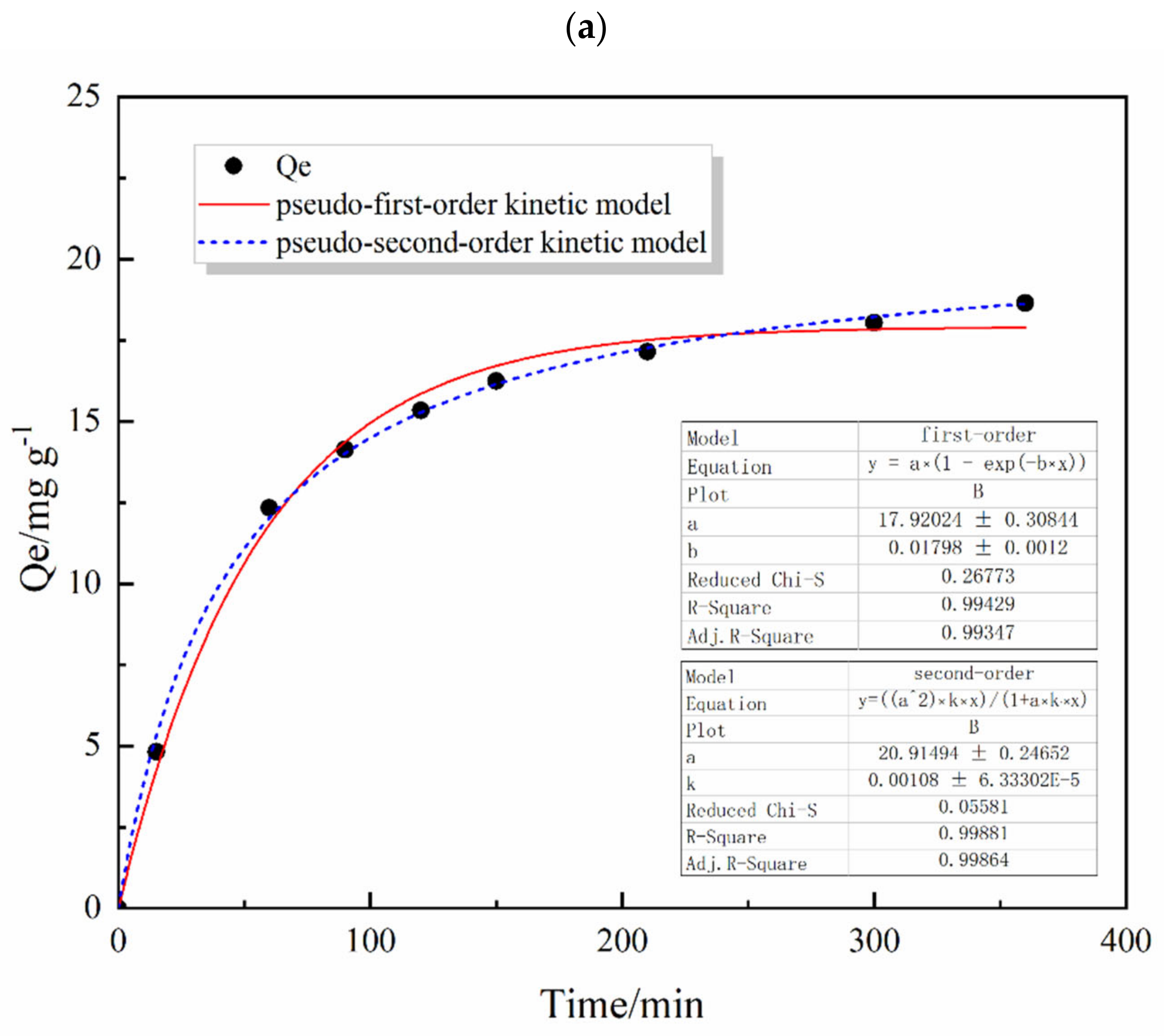
3.4.1. Sorption Kinetics
3.4.2. Sorption Isotherm
3.5. Fractionation of the P-Saturated Absorbent
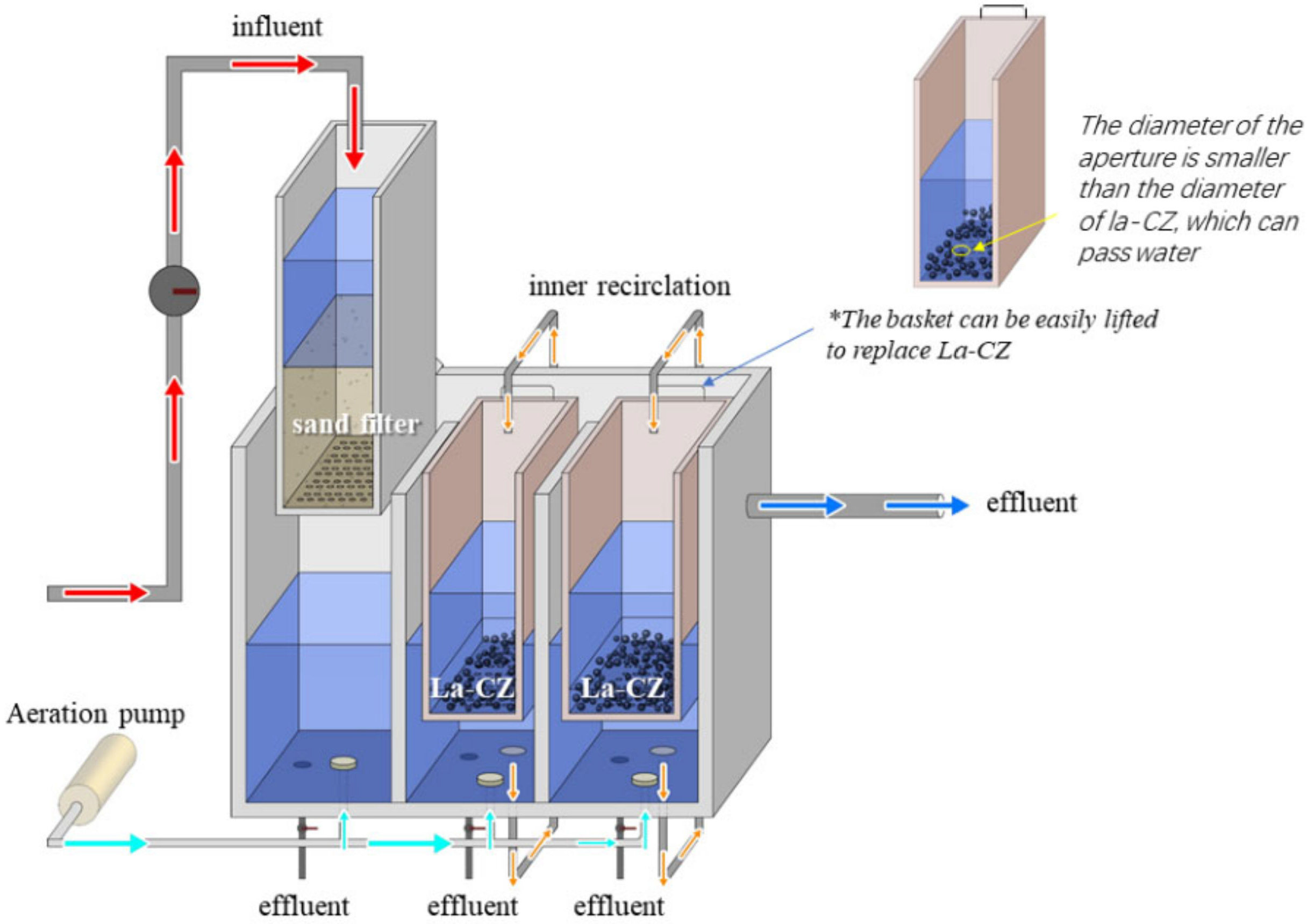
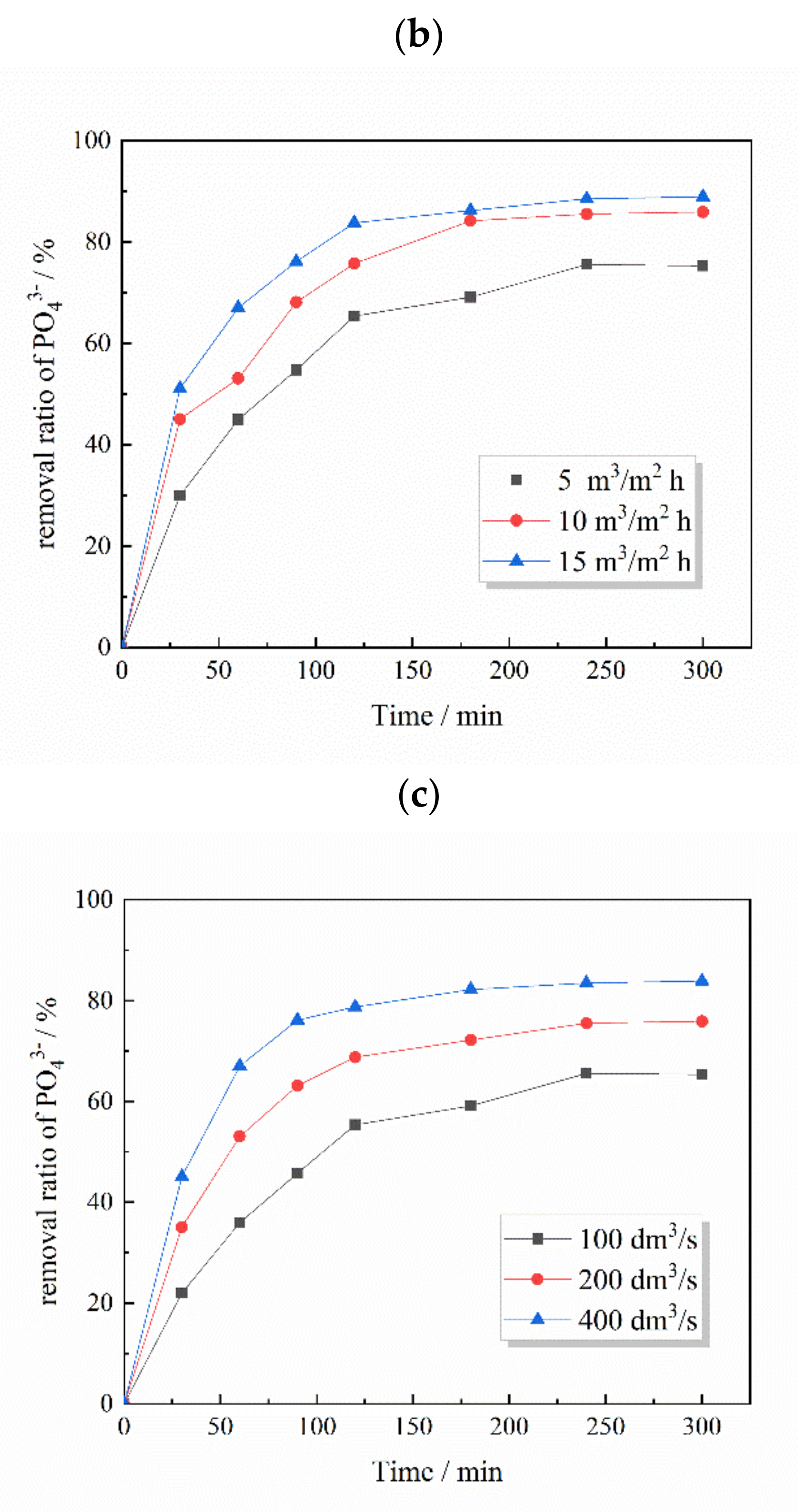
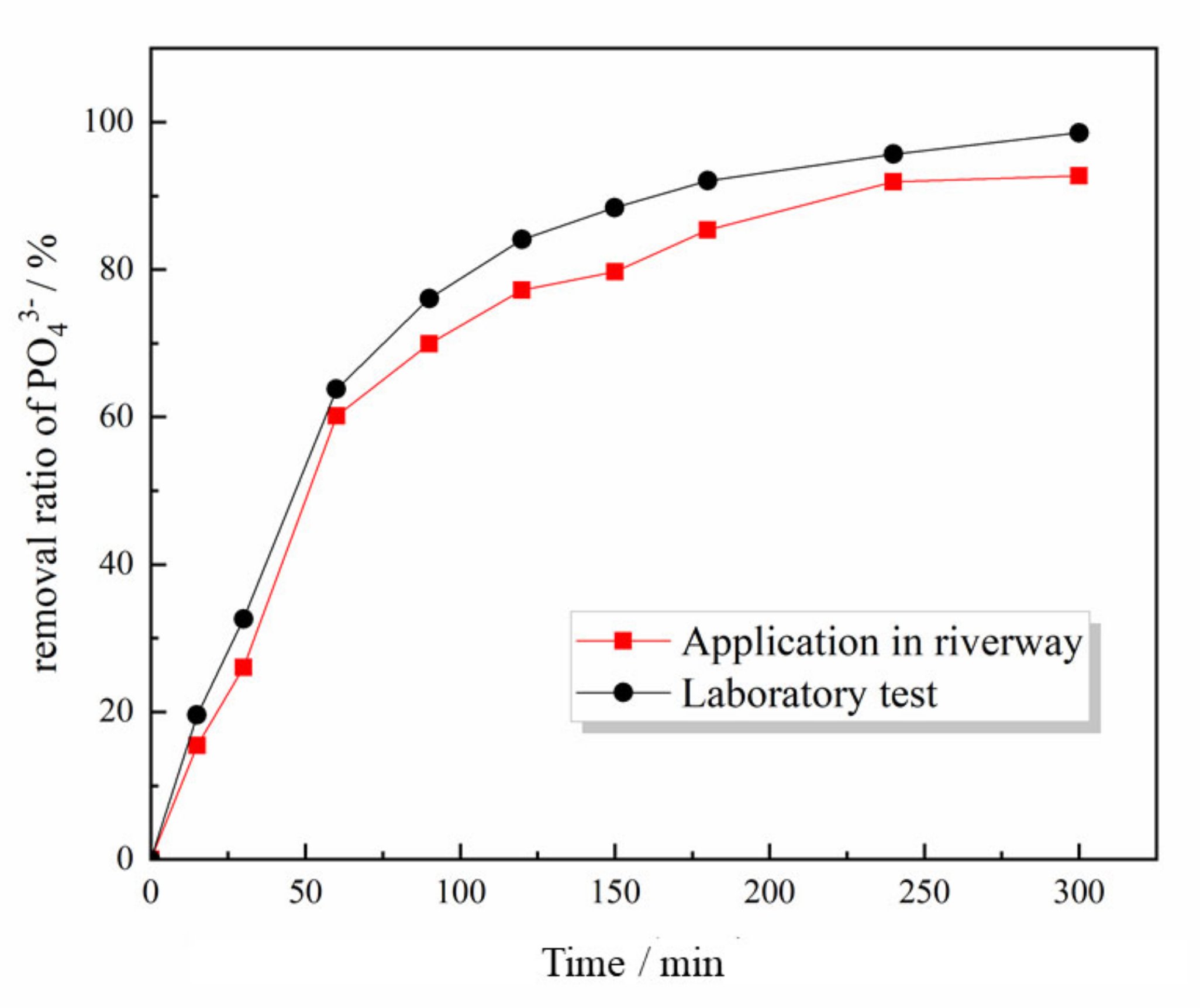
3.6. Application of the Phosphate-Removal Reactor
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Du, M.; Zhang, Y.; Wang, Z.; Lv, M.; Tang, A.; Yu, Y.; Qu, X.; Chen, Z.; Wen, Q.; Li, A. Insight into the synthesis and adsorption mechanism of adsorbents for efficient phosphate removal: Exploration from synthesis to modification. Chem. Eng. J. 2022, 442, 136147. [Google Scholar] [CrossRef]
- Bhatnagar, A.; Jain, A. A comparative adsorption study with different industrial wastes as adsorbents for the removal of cationic dyes from water. J. Colloid Interface Sci. 2005, 281, 49–55. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Wan, L.; Zhang, L.; Zhou, Q. Effect of pH, ionic strength, and temperature on the phosphate adsorption onto lanthanum-doped activated carbon fiber. J. Colloid Interface Sci. 2011, 364, 490–496. [Google Scholar] [CrossRef] [PubMed]
- Dai, L.; Wu, B.; Tan, F.; He, M.; Wang, W.; Qin, H.; Tang, X.; Zhu, Q.; Pan, K.; Hu, Q. Engineered hydrochar composites for phosphorus removal/recovery: Lanthanum doped hydrochar prepared by hydrothermal carbonization of lanthanum pretreated rice straw. Bioresour. Technol. 2014, 161, 327–332. [Google Scholar] [CrossRef] [PubMed]
- Drenkova-Tuhtan, A.; Schneider, M.; Franzreb, M.; Meyer, C.; Gellermann, C.; Sextl, G.; Mandel, K.; Steinmetz, H. Pilot-scale removal and recovery of dissolved phosphate from secondary wastewater effluents with reusable ZnFeZr adsorbent@ Fe3O4/SiO2 particles with magnetic harvesting. Water Res. 2017, 109, 77–87. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Gao, B.; Yue, Q.; Zhong, Q. Sorption of phosphate onto giant reed based adsorbent: FTIR, Raman spectrum analysis and dynamic sorption/desorption properties in filter bed. Bioresour. Technol. 2011, 102, 5278–5282. [Google Scholar] [CrossRef]
- Huang, X.; Sheng, X.; Guo, Y.; Sun, Y.; Fatehi, P.; Shi, H. Rice straw derived adsorbent for fast and efficient phosphate elimination from aqueous solution. Ind. Crops Prod. 2022, 184, 115105. [Google Scholar] [CrossRef]
- Sowmya, A.; Meenakshi, S. An efficient and regenerable quaternary amine modified chitosan beads for the removal of nitrate and phosphate anions. J. Environ. Chem. Eng. 2013, 1, 906–915. [Google Scholar] [CrossRef]
- Eltaweil, A.S.; Omer, A.M.; El-Aqapa, H.G.; Gaber, N.M.; Attia, N.F.; El-Subruiti, G.M.; Mohy-Eldin, M.S.; El-Monaem, E.M.A. Chitosan based adsorbents for the removal of phosphate and nitrate: A critical review. Carbohydr. Polym. 2021, 274, 118671. [Google Scholar] [CrossRef]
- Huang, H.; Liu, J.; Zhang, P.; Zhang, D.; Gao, F. Investigation on the simultaneous removal of fluoride, ammonia nitrogen and phosphate from semiconductor wastewater using chemical precipitation. Chem. Eng. J. 2017, 307, 696–706. [Google Scholar] [CrossRef]
- Wu, Y.; Chi, Y.; Bai, H.; Qian, G.; Cao, Y.; Zhou, J.; Xu, Y.; Liu, Q.; Xu, Z.P.; Qiao, S. Effective removal of selenate from aqueous solutions by the Friedel phase. J. Hazard. Mater. 2010, 176, 193–198. [Google Scholar] [CrossRef]
- Zhang, G.; Liu, H.; Liu, R.; Qu, J. Removal of phosphate from water by a Fe-Mn binary oxide adsorbent. J. Colloid Interface Sci. 2009, 335, 168–174. [Google Scholar] [CrossRef] [PubMed]
- Reardon, E.J.; Wang, Y. A limestone reactor for fluoride removal from wastewaters. Environ. Sci. Technol. 2000, 34, 3247–3253. [Google Scholar] [CrossRef]
- Song, L.; Huo, J.; Wang, X.; Yang, F.; He, J.; Li, C. Phosphate adsorption by a Cu (II)-loaded polyether sulfone-type metal affinity membrane with the presence of coexistent ions. Chem. Eng. J. 2016, 284, 182–193. [Google Scholar] [CrossRef]
- Debashan, L.E.; Bashan, Y. Recent advances in removing phosphorus from wastewater and its future use as fertilizer (1997–2003). Water Res. 2004, 38, 4222–4246. [Google Scholar] [CrossRef]
- He, Q.; Zhao, H.; Teng, Z.; Wang, Y.; Li, M.; Hoffmann, M.R. Phosphate removal and recovery by lanthanum-based adsorbents: A review for current advances. Chemosphere 2022, 303, 134987. [Google Scholar] [CrossRef]
- Adak, M.K.; Sen, A.; Mukherjee, A.; Sen, S.; Dhak, D. Removal of fluoride from drinking water using highly efficient nano-adsorbent, Al (III)-Fe (III)-La (III) trimetallic oxide prepared by chemical route. J. Alloy. Compd. 2017, 719, 460–469. [Google Scholar] [CrossRef]
- Zhang, J.; Shen, Z.; Shan, W.; Chen, Z.; Mei, Z.; Lei, Y.; Wang, W. Adsorption behavior of phosphate on lanthanum (III) doped mesoporous silicates material. J. Environ. Sci. 2010, 22, 507–511. [Google Scholar] [CrossRef]
- Zuo, Y.; Fu, X.; Chen, Y.; Cui, G.; Liu, M. Phosphorus removal from wastewater using a lanthanum oxide-loaded ceramic adsorbent. Adsorption 2016, 22, 1091–1098. [Google Scholar] [CrossRef]
- Elsergany, M.; Shanbleh, A. Exploratory study to assess the use of lanthanum-modified chitosan as a potential phosphorous adsorbent. Desalination Water Treat. 2018, 127, 171–177. [Google Scholar] [CrossRef]
- Wang, Z.; Shi, M.; Li, J.; Zheng, Z. Influence of moderate pre-oxidation treatment on the physical, chemical and phosphate adsorption properties of iron-containing activated carbon. J. Environ. Sci. 2014, 26, 519–528. [Google Scholar] [CrossRef]
- Yang, M.; Lin, J.; Zhan, Y.; Zhang, H. Adsorption of phosphate from water on lake sediments amended with zirconium-modified zeolites in batch mode. Ecol. Eng. 2014, 71, 223–233. [Google Scholar] [CrossRef]
- Luo, H.; Wang, Y.; Wen, X.; Cheng, S.; Li, J.; Lin, Q. Key roles of the crystal structures of MgO-biochar nanocomposites for enhancing phosphate adsorption. Sci. Total Environ. 2021, 766, 142618. [Google Scholar] [CrossRef] [PubMed]
- Liao, T.; Li, T.; Su, X.; Yu, X.; Song, H.; Zhu, Y.; Zhang, Y. La(OH)3-modified magnetic pineapple biochar as novel adsorbents for efficient phosphate removal. Bioresour. Technol. 2018, 263, 207–213. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Zhou, Q.; Chen, J.; Zhang, L.; Chang, N. Phosphate adsorption on hydroxyl–iron–lanthanum doped activated carbon fiber. Chem. Eng. J. 2013, 215, 859–867. [Google Scholar] [CrossRef]
- Zhang, J.M.; Zhao, M.X.; Li, H.J. The adsorption performance of ATP particulate adsorbent for phosphorus. Appl. Chem. Ind. 2017, 46, 1530–1535. [Google Scholar]
- Liu, D.; Zhu, H.; Wu, K.; Wang, F.; Zhao, X.; Liao, Q. Understanding the effect of particle size of waste concrete powder on phosphorus removal efficiency. Constr. Build. Mater. 2020, 236, 117526. [Google Scholar] [CrossRef]
- Zhang, L.; Zhou, Q.; Liu, J.; Chang, N.; Wan, L.; Chen, J. Phosphate adsorption on lanthanum hydroxide-doped activated carbon fiber. Chem. Eng. J. 2012, 185, 160–167. [Google Scholar] [CrossRef]
- Wang, Z.; Shen, D.; Shen, F.; Li, T. Phosphate adsorption on lanthanum loaded biochar. Chemosphere 2016, 150, 1–7. [Google Scholar] [CrossRef]
- Haghseresht, F.; Wang, S.; Do, D. A novel lanthanum-modified bentonite, Phoslock, for phosphate removal from wastewaters. Appl. Clay Sci. 2009, 46, 369–375. [Google Scholar] [CrossRef]
- Kuroki, V.; Bosco, G.E.; Fadini, P.S.; Mozeto, A.A.; Cestari, A.R.; Carvalho, W.A. Use of a La (III)-modified bentonite for effective phosphate removal from aqueous media. J. Hazard. Mater. 2014, 274, 124–131. [Google Scholar] [CrossRef] [PubMed]
- Ping, N.; Hans-Jörg, B.; Bing, L.; Xi, W.L.; Zhang, Y. Phosphate removal from wastewater by model-La (III) zeolite adsorbents. J. Environ. Sci. 2008, 20, 670–674. [Google Scholar]
- Fang, L.; Wu, B.; Chan, J.K.; Lo, I.M. Lanthanum oxide nanorods for enhanced phosphate removal from sewage: A response surface methodology study. Chemosphere 2018, 192, 209–216. [Google Scholar] [CrossRef]
- Huang, W.; Zhu, Y.; Tang, J.; Yu, X.; Wang, X.; Li, D.; Zhang, Y. Lanthanum-doped ordered mesoporous hollow silica spheres as novel adsorbents for efficient phosphate removal. J. Mater. Chem. A 2014, 2, 8839–8848. [Google Scholar] [CrossRef]
- Hamdi, N.; Srasra, E. Removal of phosphate ions from aqueous solution using Tunisian clays minerals and synthetic zeolite. J. Environ. Sci. 2012, 24, 617–623. [Google Scholar] [CrossRef]
- Jiang, D.; Amano, Y.; Machida, M. Removal and recovery of phosphate from water by a magnetic Fe3O4@ASC adsorbent. J. Environ. Chem. Eng. 2017, 5, 4229–4238. [Google Scholar] [CrossRef]
- Jiang, Y.H.; Li, A.Y.; Deng, H.; Ye, C.H.; Li, Y. Phosphate adsorption from wastewater using ZnAl-LDO-loaded modified banana straw biochar. Environ. Sci. Pollut. Res. 2019, 26, 18343–18353. [Google Scholar] [CrossRef]
- Wang, S.; Peng, Y. Natural zeolites as effective adsorbents in water and wastewater treatment. Chem. Eng. J. 2010, 156, 11–24. [Google Scholar] [CrossRef]
- Qin, K.; Li, F.; Xu, S.; Wang, T.; Liu, C. Sequential removal of phosphate and cesium by using zirconium oxide: A demonstration of designing sustainable adsorbents for green water treatment. Chem. Eng. J. 2017, 322, 275–280. [Google Scholar] [CrossRef]
- Shaila, K.; Nisha, D.; Pralhad, P.; Deepa, P. Zeolite synthesis strategies from coal fly ash: A comprehensive review of literature. Int. J. Environ. Res. 2015, 4, 93–98. [Google Scholar]
- Singh, R.P.; Gupta, A.K.; Ibrahim, M.H.; Mittal, A.K. Coal fly ash utilization in agriculture: Its potential benefits and risks. Rev. Environ. Sci. Bio/Technol. 2010, 9, 345–358. [Google Scholar] [CrossRef]
- Ji, X.; Zhang, M.; Wang, Y.; Song, Y.; Ke, Y.; Wang, Y. Immobilization of ammonium and phosphate in aqueous solution by zeolites synthesized from fly ashes with different compositions. J. Ind. Eng. Chem. 2015, 22, 1–7. [Google Scholar] [CrossRef]
- Wan, C.; Ding, S.; Zhang, C.; Tan, X.; Zou, W.; Liu, X. Simultaneous recovery of nitrogen and phosphorus from sludge fermentation liquid by zeolite adsorption: Mechanism and application. Sep. Purif. Technol. 2017, 180, 1–12. [Google Scholar] [CrossRef]
- Kaiserli, A.; Voutsa, D.; Samara, C. Phosphorus fractionation in lake sediments-Lakes Volvi and Koronia, N. Greece. Chemosphere 2002, 46, 1147–1155. [Google Scholar] [CrossRef]
- Dithmer, L.; Lipton, A.S.; Reitzel, K. Characterization of phosphate sequestration by a lanthanum modified bentonite clay: A solid-state NMR, EXAFS, and PXRD study. Environ. Sci. Technol. 2015, 49, 4559–4566. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Qiu, P.H.; Fan, H. Effects of insitu-chemical remediation on phosphorus and nitrogen in sediment-water system. Environ. Sci. Technol. 2016, 39, 271–276. [Google Scholar]
- Zhang, Q.Y.; Du, Y.X.; Luo, C.Y. Advances in researches on phosphorus immobilization by lanthanum modified bentonite in lakes and its ecological risk. J. Lake Sci. 2019, 31, 1499–1509. [Google Scholar]
- Durner, W.; Iden, S.C. The integral suspension pressure method (ISP) for precise particle-size analysis by gravitational sedimentation. Water Resour. Res. 2017, 53, 33–48. [Google Scholar] [CrossRef]
- Wu, D.Y.; Zhang, B.H.; Li, C.J.; Zhang, Z.J.; Kong, H.N. Simultaneous removal of ammonium and phosphate by zeolite synthesized from fly ash as influenced by salt treatment. J. Colloid Interface Sci. 2006, 304, 300–306. [Google Scholar] [CrossRef]




| Sample | BET Surface Area (m2·g−1) | Total Pore Volume (cm2·g−1) | Average Pore Diameter (nm) |
|---|---|---|---|
| CZ | 15.53 | 0.05 | 1.41 |
| La-CZ | 32.34 | 0.20 | 16.68 |
| Adsorbent | Initial Phosphorus Concentration/(mg·L−1) | Dosage/(g·L−1) | Particle Diameter/(mm) | qe/(mg·g−1) | Equilibration Time/(h) | De*/(mg) | References |
|---|---|---|---|---|---|---|---|
| ATP Clay particles | 20 | 8 | 3 | 2.8 | 14 | 6.96 | [26] |
| fine particles of waste concrete | 20 | 20 | 1 | 4.96 | 5 | 3.93 | [27] |
| Activated carbon fiber-LaOH | 30 | 2.5 | powdered form | 16.4 | 6 | 1.18 | [28] |
| La-doped activated carbon fiber | 20 | 5 | 2 | 9.41 | 4 | 2.07 | [29] |
| La-modified bentonite | 100 | 2.5 | 0.84 | 10.6 | 20 | 1.84 | [30] |
| NT-25La | 0.5 | 0.75 | powdered form | 14.4 | 2 | 1.35 | [31] |
| La-CZ | 20 | 1 | 5 | 18.1 | 2 | 1.07 | This study |
| Pseudo-First-Order Model | Pseudo-Second-Order Model | ||||
|---|---|---|---|---|---|
| qe (mg·g−1) | K1 (min−1) | R2 | qe (mg·g−1) | K2 (min−1) | R2 |
| 17.9202 ± 0.3084 | 0.0179 ± 0.0012 | 0.9942 | 20.9195 ± 0.2465 | 0.0011 ± 0.0001 | 0.9989 |
| Langmuir | Freundlich | ||||
|---|---|---|---|---|---|
| Qmax (mg·g−1) | KL (L·mg−1) | R2 | Ff (L·mg−1) | b | R2 |
| 14.6417 ± 0.732 | −0.1196 ± 0.0247 | 0.9669 | 20.7702 ± 0.3196 | 3.4686 ± 0.2201 | 0.9975 |
| Initial Concentration (mg L−1) | NH4Cl-P | BD-P | NaOH-P | HCl-P | ||||
|---|---|---|---|---|---|---|---|---|
| Concentration (μg g−1) | Percentage (%) | Concentration (μg g−1) | Percentage (%) | Concentration (μg g−1) | Percentage (%) | Concentration (mg g−1) | Percentage (%) | |
| 10.0 | 9.80 ± 0.21 | 6.24 | 0.46 ± 0.01 | 0.29 | 24.52 ± 0.06 | 15.61 | 122.29 ± 0.27 | 77.86 |
| 20.0 | 4.19 ± 0.37 | 2.22 | 1.00 ± 0.01 | 0.53 | 19.06 ± 0.03 | 10.09 | 166.37 ± 0.31 | 88.09 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Y.; Yang, K.; Fang, Y.; Ding, J.; Zhang, H. Removal of Phosphate from Wastewater with a Recyclable La-Based Particulate Adsorbent in a Small-Scale Reactor. Water 2022, 14, 2326. https://doi.org/10.3390/w14152326
Zhang Y, Yang K, Fang Y, Ding J, Zhang H. Removal of Phosphate from Wastewater with a Recyclable La-Based Particulate Adsorbent in a Small-Scale Reactor. Water. 2022; 14(15):2326. https://doi.org/10.3390/w14152326
Chicago/Turabian StyleZhang, Yinan, Kexin Yang, Yuxin Fang, Jiafeng Ding, and Hangjun Zhang. 2022. "Removal of Phosphate from Wastewater with a Recyclable La-Based Particulate Adsorbent in a Small-Scale Reactor" Water 14, no. 15: 2326. https://doi.org/10.3390/w14152326





