Chronic Toxic Effects of Waterborne Mercury on Silver Carp (Hypophthalmichthys molitrix) Larvae
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Animals and Test Chemicals
2.2. Experimental Design and Sample Collection
2.3. Biochemical Analysis
2.4. Integrated Biomarker Response
2.5. Statistical Analysis
3. Results
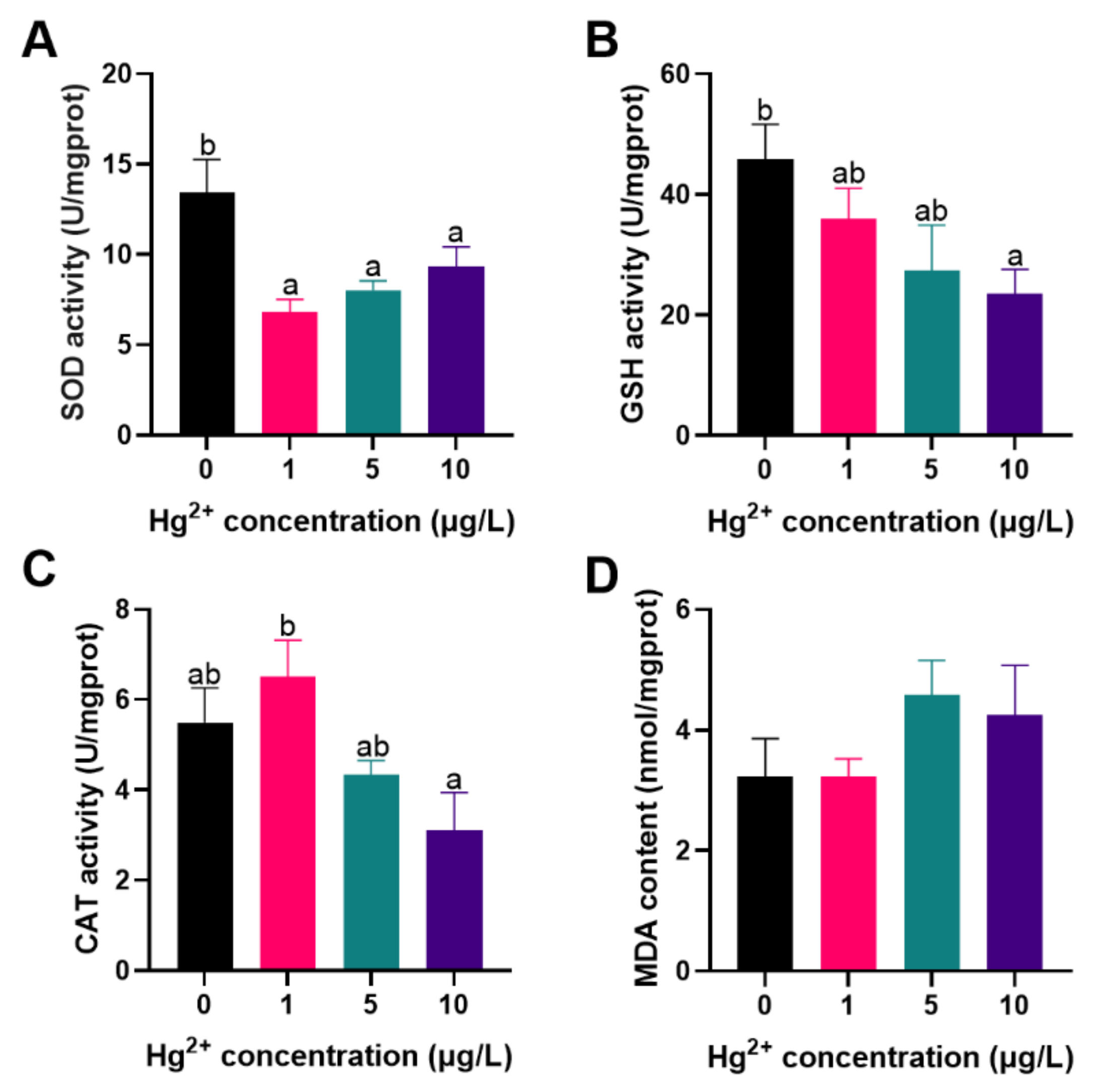
3.1. Antioxidant Ability
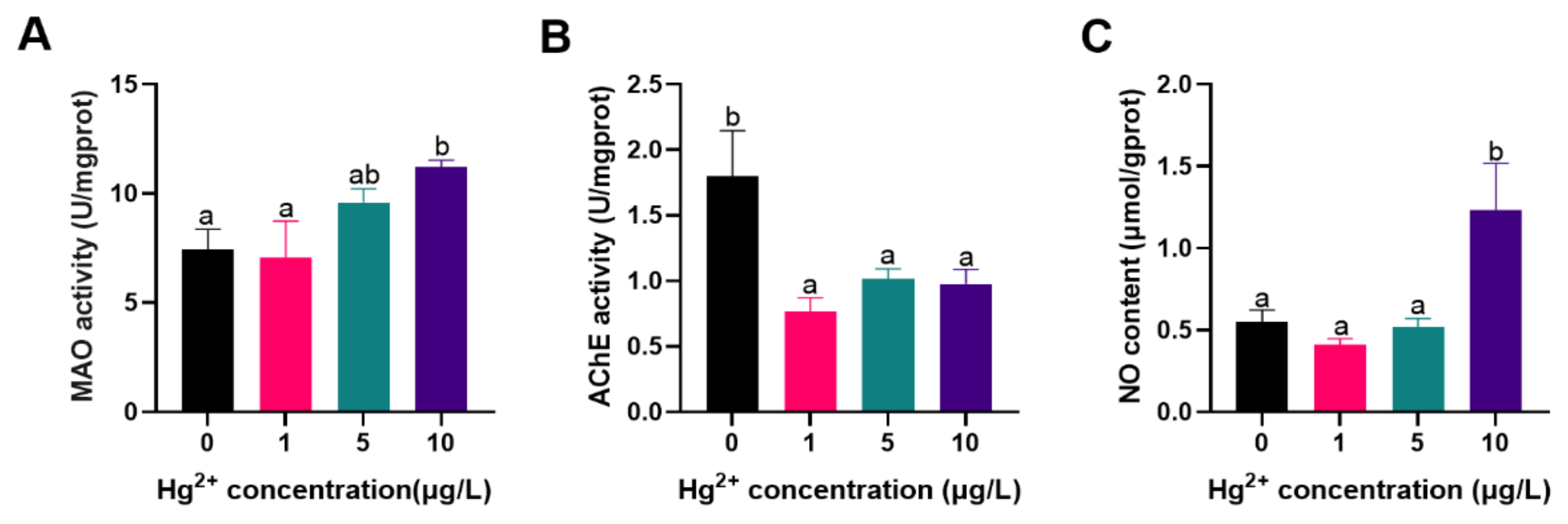
3.2. Neurotoxic Related Parameters
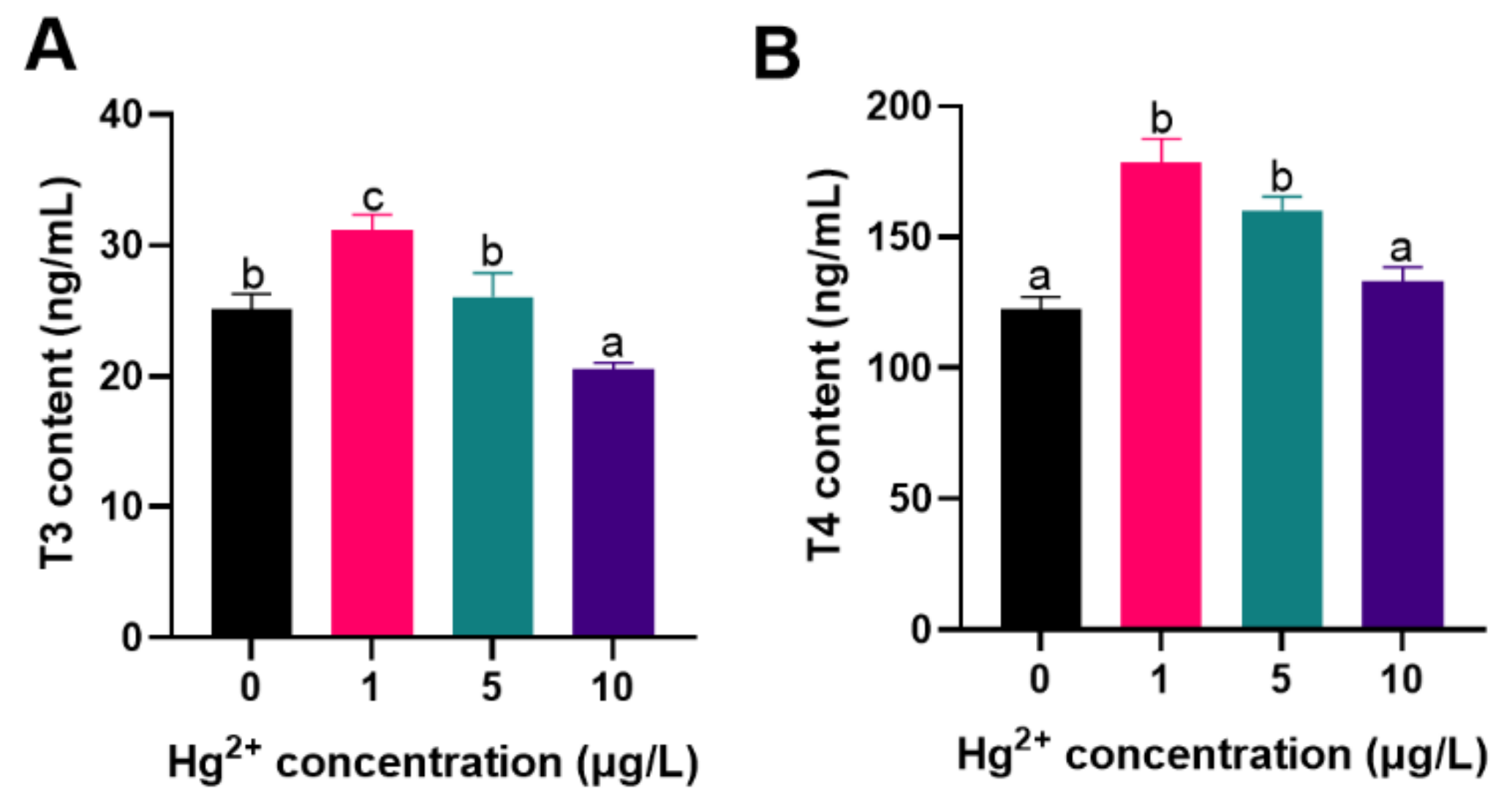
3.3. Thyroid Hormones (THs) Content
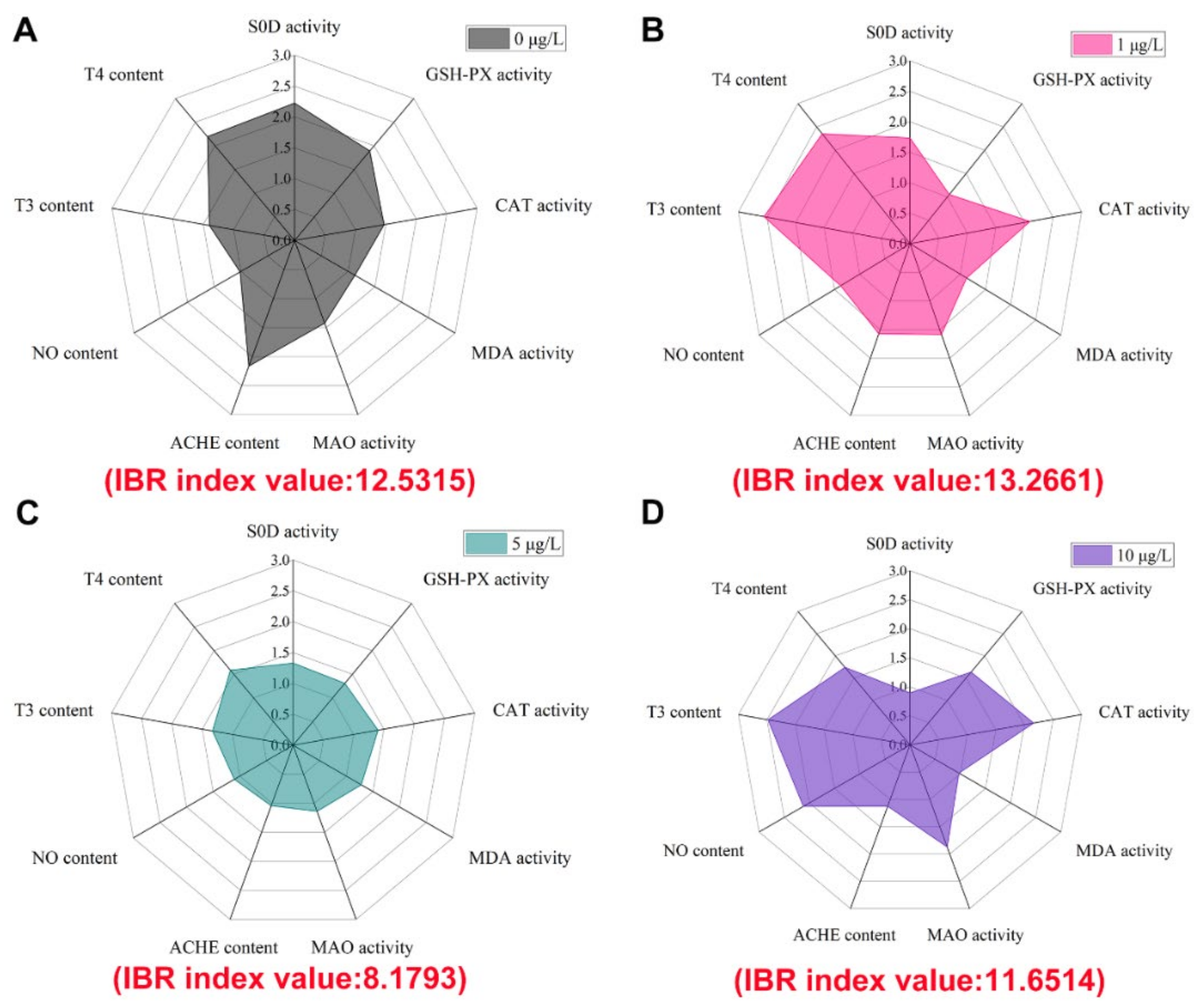
3.4. Integrated Biomarker Response
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mamdouh, A.-Z.; Zahran, E.; Mohamed, F.; Zaki, V. Nannochloropsis oculata feed additive alleviates mercuric chloride-induced toxicity in Nile tilapia (Oreochromis niloticus). Aquat. Toxicol. 2021, 238, 105936. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.H.; Chen, L.; Wu, Y.H.; Li, P.; Li, Y.F.; Ni, Z.H. Alteration of thyroid hormone levels and related gene expression in Chinese rare minnow larvae exposed to mercury chloride. Environ. Toxicol. Pharmacol. 2014, 38, 325–331. [Google Scholar] [CrossRef]
- Li, P.; Li, Z.H.; Wu, Y.H. Interactive effects of temperature and mercury exposure on the stress-related responses in the freshwater fish Ctenopharyngodon idella. Aquacult. Res. 2021, 52, 2070–2077. [Google Scholar] [CrossRef]
- Li, Z.-H.; Li, Z.-P.; Tang, X.; Hou, W.-H.; Li, P. Distribution and Risk Assessment of Toxic Pollutants in Surface Water of the Lower Yellow River, China. Water 2021, 13, 1582. [Google Scholar] [CrossRef]
- Islam, M.S.; Ahmed, M.K.; Raknuzzaman, M.; Habibullah-Al-Mamun, M.; Islam, M.K. Heavy metal pollution in surface water and sediment: A preliminary assessment of an urban river in a developing country. Ecol. Indic. 2015, 48, 282–291. [Google Scholar] [CrossRef]
- Li, Z.H.; Li, P.; Dzyuba, B.; Randak, T. Influence of environmental related concentrations of heavy metals on motility parameters and antioxidant responses in sturgeon sperm. Chem.-Biol. Interact. 2010, 188, 473–477. [Google Scholar] [CrossRef]
- Chen, L.; Zhang, X.; Cao, M.; Pan, Y.; Xiao, C.; Wang, P.; Liang, Y.; Liu, G.; Cai, Y. Release of legacy mercury and effect of aquaculture on mercury biogeochemical cycling in highly polluted Ya-Er Lake, China. Chemosphere 2021, 275, 130011. [Google Scholar] [CrossRef]
- Tomiyasu, T.; Matsuyama, A.; Eguchi, T.; Marumoto, K.; Oki, K.; Akagi, H. Speciation of mercury in water at the bottom of Minamata Bay, Japan. Mar. Chem. 2008, 112, 102–106. [Google Scholar] [CrossRef]
- Bravo, A.G.; Cosio, C. Biotic formation of methylmercury: A bio–physico–chemical conundrum. Limnol. Oceanogr. 2020, 65, 1010–1027. [Google Scholar] [CrossRef]
- Donadt, C.; Cooke, C.A.; Graydon, J.A.; Poesch, M.S. Mercury bioaccumulation in stream fish from an agriculturally-dominated watershed. Chemosphere 2021, 262, 128059. [Google Scholar] [CrossRef]
- Da Silva, J.P.; Do S Costa, M.; Campina, F.F.; Bezerra, C.F.; De Freitas, T.S.; Sousa, A.K.; Sobral Souza, C.E.; De Matos, Y.M.; Pereira-Junior, F.N.; Menezes, I.R. Evaluation of chelating and cytoprotective activity of vanillin against the toxic action of mercuric chloride as an alternative for phytoremediation. Environ. Geochem. Health 2021, 43, 1609–1616. [Google Scholar] [CrossRef] [PubMed]
- Nowosad, J.; Kucharczyk, D.; Łuczyńska, J. Changes in mercury concentration in muscles, ovaries and eggs of European eel during maturation under controlled conditions. Ecotoxicol. Environ. Saf. 2018, 148, 857–861. [Google Scholar] [CrossRef]
- Li, Z.H.; Li, P.; Wu, Y. Regulation of glutathione-dependent antioxidant defense system of grass carp Ctenopharyngodon idella under the combined stress of mercury and temperature. Env. Sci Pollut Res Int 2021, 28, 1689–1696. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Li, Y.; Rao, J.; Liu, Z.; Chen, Q. Effects of inorganic mercury exposure on histological structure, antioxidant status and immune response of immune organs in yellow catfish (Pelteobagrus fulvidraco). J. Appl. Toxicol. 2018, 38, 843–854. [Google Scholar] [CrossRef]
- Chen, Q.-L.; Sun, Y.-L.; Liu, Z.-H.; Li, Y.-W. Sex-dependent effects of subacute mercuric chloride exposure on histology, antioxidant status and immune-related gene expression in the liver of adult zebrafish (Danio rerio). Chemosphere 2017, 188, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, D.; Lin, L.; Wang, M. Quantitative proteomic analysis reveals proteins involved in the neurotoxicity of marine medaka Oryzias melastigma chronically exposed to inorganic mercury. Chemosphere 2015, 119, 1126–1133. [Google Scholar] [CrossRef]
- Morcillo, P.; Esteban, M.Á.; Cuesta, A. Heavy metals produce toxicity, oxidative stress and apoptosis in the marine teleost fish SAF-1 cell line. Chemosphere 2016, 144, 225–233. [Google Scholar] [CrossRef]
- Morcillo, P.; Romero, D.; Meseguer, J.; Esteban, M.; Cuesta, A. Cytotoxicity and alterations at transcriptional level caused by metals on fish erythrocytes in vitro. Environ. Sci. Pollut. Res. 2016, 23, 12312–12322. [Google Scholar] [CrossRef] [PubMed]
- Zheng, N.; Wang, S.; Dong, W.; Hua, X.; Li, Y.; Song, X.; Chu, Q.; Hou, S.; Li, Y. The toxicological effects of mercury exposure in marine fish. Bull. Environ. Contam. Toxicol. 2019, 102, 714–720. [Google Scholar] [CrossRef]
- Huang, W.; Cao, L.; Shan, X.; Lin, L.; Dou, S. Toxicity testing of waterborne mercury with red sea bream (Pagrus major) embryos and larvae. Bull. Environ. Contam. Toxicol. 2011, 86, 398–405. [Google Scholar] [CrossRef]
- Sha, H.; Luo, X.-Z.; Wang, D.; Li, X.-h.; Zou, G.-W.; Liang, H.-W. New insights to protection and utilization of silver carp (Hypophthalmichthys molitrix) in Yangtze River based on microsatellite analysis. Fish. Res. 2021, 241, 105997. [Google Scholar] [CrossRef]
- FAO. FAO Yearbook: The State of World Fisheries and Aquaculture 2020: Sustainability in Action; FAO: Rome, Italy, 2020. [Google Scholar]
- George, A.E.; Chapman, D.C. Aspects of embryonic and larval development in bighead carp Hypophthalmichthys nobilis and silver carp Hypophthalmichthys molitrix. PLoS ONE 2013, 8, e73829. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Szabó, T.; Urbányi, B.; Müller, T.; Szabó, R.; Horváth, L. Assessment of induced breeding of major Chinese carps at a large-scale hatchery in Hungary. Aquac. Rep. 2019, 14, 100193. [Google Scholar] [CrossRef]
- Houshmand, P.; Hedayati, A. Dietary Effect of Probiotic and Prebiotic on Some Mucus Indices of Silver Carp (Hypophthalmichthys molitrix) Exposed to Silver Nanoparticles. Biol. Trace Elem. Res. 2022, 1–9. [Google Scholar] [CrossRef]
- Meng, L.-J.; Zhang, Y.; Li, X.-X.; Liu, J.-H.; Wen, B.; Gao, J.-Z.; Chen, Z.-Z. Comparative analysis of bacterial communities of water and intestines of silver carp (Hypophthalmichthys molitrix) and bighead carp (H. nobilis) reared in aquaculture pond systems. Aquaculture 2021, 534, 736334. [Google Scholar] [CrossRef]
- Li, Z.-H.; Chen, L.; Wu, Y.-H.; Li, P.; Li, Y.-F.; Ni, Z.-H. Effects of waterborne cadmium on thyroid hormone levels and related gene expression in Chinese rare minnow larvae. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2014, 161, 53–57. [Google Scholar] [CrossRef]
- Chen, C.-Z.; Li, P.; Wang, W.-B.; Li, Z.-H. Response of growth performance, serum biochemical parameters, antioxidant capacity, and digestive enzyme activity to different feeding strategies in common carp (Cyprinus carpio) under high-temperature stress. Aquaculture 2022, 548, 737636. [Google Scholar] [CrossRef]
- Gu, P.; Li, Q.; Zhang, W.; Gao, Y.; Sun, K.; Zhou, L.; Zheng, Z. Biological toxicity of fresh and rotten algae on freshwater fish: LC50, organ damage and antioxidant response. J. Hazard. Mater. 2021, 407, 124620. [Google Scholar] [CrossRef]
- Kong, Y.; Li, M.; Shan, X.; Wang, G.; Han, G. Effects of deltamethrin subacute exposure in snakehead fish, Channa argus: Biochemicals, antioxidants and immune responses. Ecotoxicol. Environ. Saf. 2021, 209, 111821. [Google Scholar] [CrossRef]
- Mieiro, C.; Pereira, M.; Duarte, A.; Pacheco, M. Brain as a critical target of mercury in environmentally exposed fish (Dicentrarchus labrax)—bioaccumulation and oxidative stress profiles. Aquat. Toxicol. 2011, 103, 233–240. [Google Scholar] [CrossRef]
- Roos, D.H.; Puntel, R.L.; Santos, M.M.; Souza, D.O.; Farina, M.; Nogueira, C.W.; Aschner, M.; Burger, M.E.; Barbosa, N.B.; Rocha, J.B. Guanosine and synthetic organoselenium compounds modulate methylmercury-induced oxidative stress in rat brain cortical slices: Involvement of oxidative stress and glutamatergic system. Toxicol. Vitr. 2009, 23, 302–307. [Google Scholar] [CrossRef] [PubMed]
- Romeo, M.; Bennani, N.; Gnassia-Barelli, M.; Lafaurie, M.; Girard, J. Cadmium and copper display different responses towards oxidative stress in the kidney of the sea bass Dicentrarchus labrax. Aquat. Toxicol. 2000, 48, 185–194. [Google Scholar] [CrossRef]
- Li, Z.H.; Li, P.; Randak, T. Ecotoxocological effects of short-term exposure to a human pharmaceutical Verapamil in juvenile rainbow trout (Oncorhynchus mykiss). Comp. Biochem. Physiol. C 2010, 152, 385–391. [Google Scholar] [CrossRef]
- Li, Z.H.; Chen, L.; Wu, Y.H.; Li, P.; Li, Y.F.; Ni, Z.H. Effects of mercury on oxidative stress and gene expression of potential biomarkers in larvae of the Chinese rare minnow Gobiocypris rarus. Arch. Environ. Contam. Toxicol. 2014, 67, 245–251. [Google Scholar] [CrossRef] [PubMed]
- Gopi, N.; Vijayakumar, S.; Thaya, R.; Govindarajan, M.; Alharbi, N.S.; Kadaikunnan, S.; Khaled, J.M.; Al-Anbr, M.N.; Vaseeharan, B. Chronic exposure of Oreochromis niloticus to sub-lethal copper concentrations: Effects on growth, antioxidant, non-enzymatic antioxidant, oxidative stress and non-specific immune responses. J. Trace Elem. Med. Biol. 2019, 55, 170–179. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.H.; Zlabek, V.; Velisek, J.; Grabic, R.; Machova, J.; Kolarova, J.; Li, P.; Randak, T. Antioxidant responses and plasma biochemical characteristics in the freshwater rainbow trout, Oncorhynchus mykiss, after acute exposure to the fungicide propiconazole. Czech J. Anim. Sci. 2011, 56, 61–69. [Google Scholar] [CrossRef]
- Wu, F.; Huang, W.; Liu, Q.; Xu, X.; Zeng, J.; Cao, L.; Hu, J.; Xu, X.; Gao, Y.; Jia, S. Responses of antioxidant defense and immune gene expression in early life stages of large yellow croaker (Pseudosciaena crocea) under methyl mercury exposure. Front. Physiol. 2018, 9, 1436. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, Z.; Ren, T.; Han, Y.; Jiang, Z.; Hu, Y.; Bai, Z.; Wang, L.; Ding, J. The Effects of Sub-lethal Dietary Mercury on Growth Performance, Bioaccumulation, and Activities of Antioxidant Enzymes in Sea Cucumber, Apostichopus japonicus. Bull. Environ. Contam. Toxicol. 2018, 100, 683–689. [Google Scholar] [CrossRef]
- Barboza, L.G.A.; Vieira, L.R.; Branco, V.; Figueiredo, N.; Carvalho, F.; Carvalho, C.; Guilhermino, L. Microplastics cause neurotoxicity, oxidative damage and energy-related changes and interact with the bioaccumulation of mercury in the European seabass, Dicentrarchus labrax (Linnaeus, 1758). Aquat. Toxicol. 2018, 195, 49–57. [Google Scholar] [CrossRef]
- Pereira, P.; Puga, S.; Cardoso, V.; Pinto-Ribeiro, F.; Raimundo, J.; Barata, M.; Pousão-Ferreira, P.; Pacheco, M.; Almeida, A. Inorganic mercury accumulation in brain following waterborne exposure elicits a deficit on the number of brain cells and impairs swimming behavior in fish (white seabream—Diplodus sargus). Aquat. Toxicol. 2016, 170, 400–412. [Google Scholar] [CrossRef]
- Mukherjee, A.; Bhowmick, A.R.; Mukherjee, J.; Moniruzzaman, M. Physiological response of fish under variable acidic conditions: A molecular approach through the assessment of an eco-physiological marker in the brain. Environ. Sci. Pollut. Res. 2019, 26, 23442–23452. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.H.; Li, P.; Randak, T. Effect of a human pharmaceutical carbamazepine on antioxidant responses in brain of a model teleost in vitro: An efficient approach to biomonitoring. J. Appl. Toxicol. 2010, 30, 644–648. [Google Scholar] [CrossRef] [PubMed]
- Crump, K.L.; Trudeau, V.L. Mercury-induced reproductive impairment in fish. Environ. Toxicol. Chem. Int. J. 2009, 28, 895–907. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Li, Z.H.; Zhong, L.Q. Effects of low concentrations of triphenyltin on neurobehavior and the thyroid endocrine system in zebrafish. Ecotoxicol. Environ. Saf. 2019, 186, 109776. [Google Scholar] [CrossRef]
- Li, Z.-H.; Li, P.; Shi, Z.-C. Chronic exposure to tributyltin induces brain functional damage in juvenile common carp (Cyprinus carpio). PLoS ONE 2015, 10, e0123091. [Google Scholar] [CrossRef] [Green Version]
- Basha, D.C.; Rani, M.U.; Devi, C.B.; Kumar, M.R.; Reddy, G.R. Perinatal lead exposure alters postnatal cholinergic and aminergic system in rat brain: Reversal effect of calcium co-administration. Int. J. Dev. Neurosci. 2012, 30, 343–350. [Google Scholar] [CrossRef]
- Lee, J.-W.; Choi, H.; Hwang, U.-K.; Kang, J.-C.; Kang, Y.J.; Kim, K.I.; Kim, J.-H. Toxic effects of lead exposure on bioaccumulation, oxidative stress, neurotoxicity, and immune responses in fish: A review. Environ. Toxicol. Pharmacol. 2019, 68, 101–108. [Google Scholar] [CrossRef]
- Santos, D.; Luzio, A.; Matos, C.; Bellas, J.; Monteiro, S.M.; Félix, L. Microplastics alone or co-exposed with copper induce neurotoxicity and behavioral alterations on zebrafish larvae after a subchronic exposure. Aquat. Toxicol. 2021, 235, 105814. [Google Scholar] [CrossRef]
- Zhang, S.; Guo, X.; Lu, S.; Sang, N.; Li, G.; Xie, P.; Liu, C.; Zhang, L.; Xing, Y. Exposure to PFDoA causes disruption of the hypothalamus-pituitary-thyroid axis in zebrafish larvae. Environ. Pollut. 2018, 235, 974–982. [Google Scholar] [CrossRef]
- Liu, W.; Yang, J.; Li, J.; Zhang, J.; Zhao, J.; Yu, D.; Xu, Y.; He, X.; Zhang, X. Toxicokinetics and persistent thyroid hormone disrupting effects of chronic developmental exposure to chlorinated polyfluorinated ether sulfonate in Chinese rare minnow. Environ. Pollut. 2020, 263, 114491. [Google Scholar] [CrossRef]
- Li, P.; Li, Z.H. Environmental co-exposure to TBT and Cd caused neurotoxicity and thyroid endocrine disruption in zebrafish, a three-generation study in a simulated environment. Environ. Pollut. 2020, 259, 113868. [Google Scholar] [CrossRef] [PubMed]
- Deal, C.K.; Volkoff, H. The role of the thyroid axis in fish. Front. Endocrinol. 2020, 11, 861. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.H.; Li, P.; Wu, Y.H. Effects of temperature fluctuation on endocrine disturbance of grass carp Ctenopharyngodon idella under mercury chloride stress. Chemosphere 2021, 263, 128137. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Li, Y.; Liu, Z.; Chen, Q. Environmentally relevant concentrations of mercury exposure alter thyroid hormone levels and gene expression in the hypothalamic–pituitary–thyroid axis of zebrafish larvae. Fish Physiol. Biochem. 2018, 44, 1175–1183. [Google Scholar] [CrossRef]
- Kim, W.-K.; Lee, S.-K.; Park, J.-W.; Choi, K.; Cargo, J.; Schlenk, D.; Jung, J. Integration of multi-level biomarker responses to cadmium and benzo [k] fluoranthene in the pale chub (Zacco platypus). Ecotoxicol. Environ. Saf. 2014, 110, 121–128. [Google Scholar] [CrossRef]
- Li, Z.H.; Zlabek, V.; Turek, J.; Velisek, J.; Pulkrabova, J.; Kolarova, J.; Sudova, E.; Berankova, P.; Hradkova, P.; Hajslova, J.; et al. Evaluating environmental impact of STPs situated on streams in the Czech Republic: An integrated approach to biomonitoring the aquatic environment. Water Res. 2011, 45, 1403–1413. [Google Scholar] [CrossRef]
- Baudou, F.G.; Ossana, N.A.; Castañé, P.M.; Mastrángelo, M.M.; Núñez, A.A.G.; Palacio, M.J.; Ferrari, L. Use of integrated biomarker indexes for assessing the impact of receiving waters on a native neotropical teleost fish. Sci. Total Environ. 2019, 650, 1779–1786. [Google Scholar] [CrossRef]
- Biswal, A.; Srivastava, P.P.; Pal, P.; Gupta, S.; Varghese, T.; Jayant, M. A multi-biomarker approach to evaluate the effect of sodium chloride in alleviating the long-term transportation stress of Labeo rohita fingerlings. Aquaculture 2021, 531, 735979. [Google Scholar] [CrossRef]
- Devin, S.; Burgeot, T.; Giambérini, L.; Minguez, L.; Pain-Devin, S. The integrated biomarker response revisited: Optimization to avoid misuse. Environ. Sci. Pollut. Res. 2014, 21, 2448–2454. [Google Scholar] [CrossRef] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Y.-J.; Chen, C.-Z.; Li, P.; Liu, L.; Chai, Y.; Li, Z.-H. Chronic Toxic Effects of Waterborne Mercury on Silver Carp (Hypophthalmichthys molitrix) Larvae. Water 2022, 14, 1774. https://doi.org/10.3390/w14111774
Wang Y-J, Chen C-Z, Li P, Liu L, Chai Y, Li Z-H. Chronic Toxic Effects of Waterborne Mercury on Silver Carp (Hypophthalmichthys molitrix) Larvae. Water. 2022; 14(11):1774. https://doi.org/10.3390/w14111774
Chicago/Turabian StyleWang, Ya-Jun, Cheng-Zhuang Chen, Ping Li, Ling Liu, Yi Chai, and Zhi-Hua Li. 2022. "Chronic Toxic Effects of Waterborne Mercury on Silver Carp (Hypophthalmichthys molitrix) Larvae" Water 14, no. 11: 1774. https://doi.org/10.3390/w14111774
APA StyleWang, Y.-J., Chen, C.-Z., Li, P., Liu, L., Chai, Y., & Li, Z.-H. (2022). Chronic Toxic Effects of Waterborne Mercury on Silver Carp (Hypophthalmichthys molitrix) Larvae. Water, 14(11), 1774. https://doi.org/10.3390/w14111774