High-Throughput Sequencing of Diatom Community, Its Spatial and Temporal Variation and Interrelationships with Physicochemical Factors in Danjiangkou Reservoir, China
Abstract
:1. Introduction
2. Materials and Methods
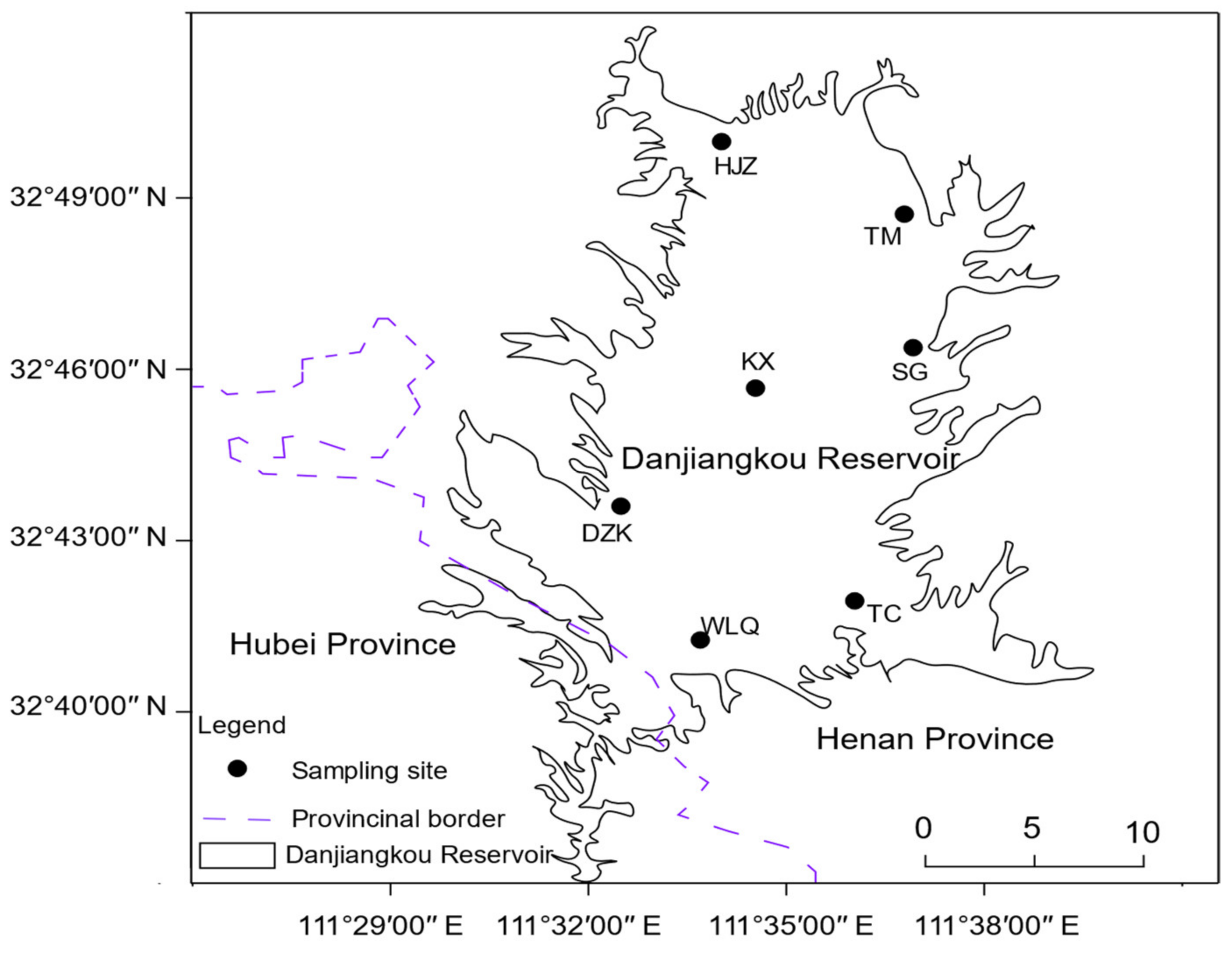
2.1. Study Area
2.2. Sampling Point Setting and Collection of Samples
2.3. DNA Extraction and Sequencing
2.4. Data Processing and Analysis
2.5. Nucleotide Sequence Accession Numbers
3. Results
3.1. Physicochemical Properties of Danjiangkou Reservoir Subsection
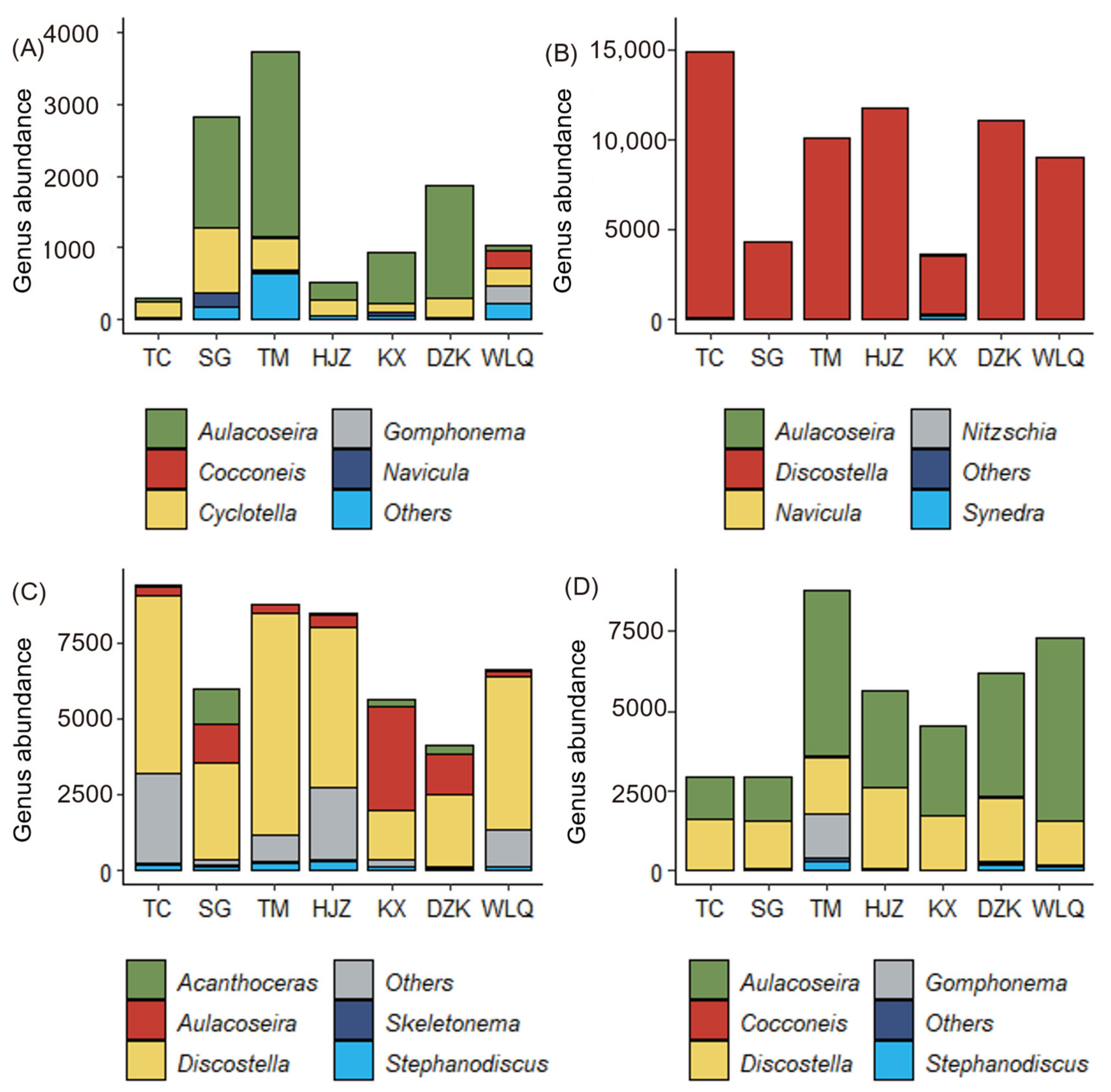
3.2. Seasonal Variation of Diatom Community Structure
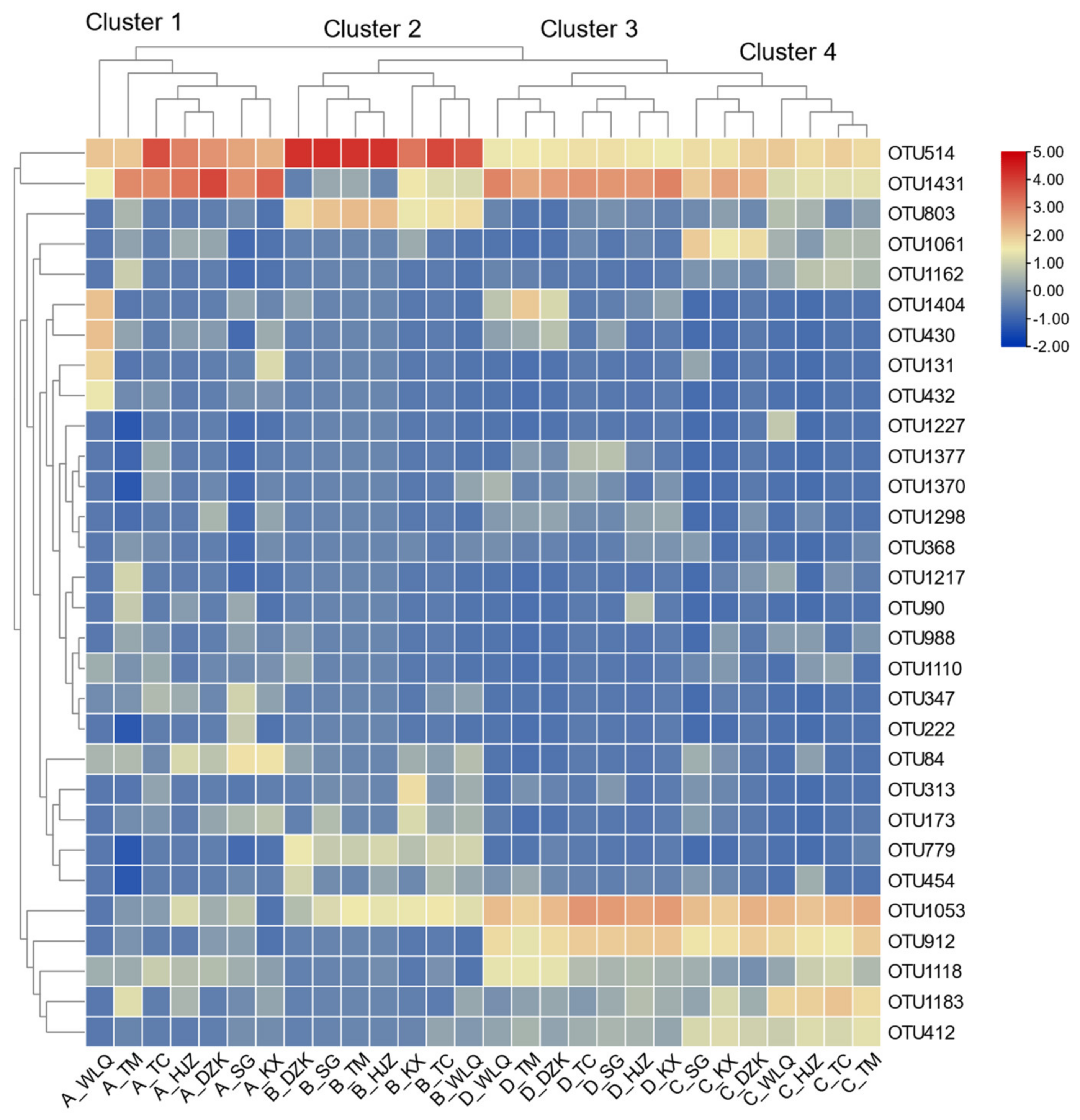
3.3. Spatial Variation of Diatom Community
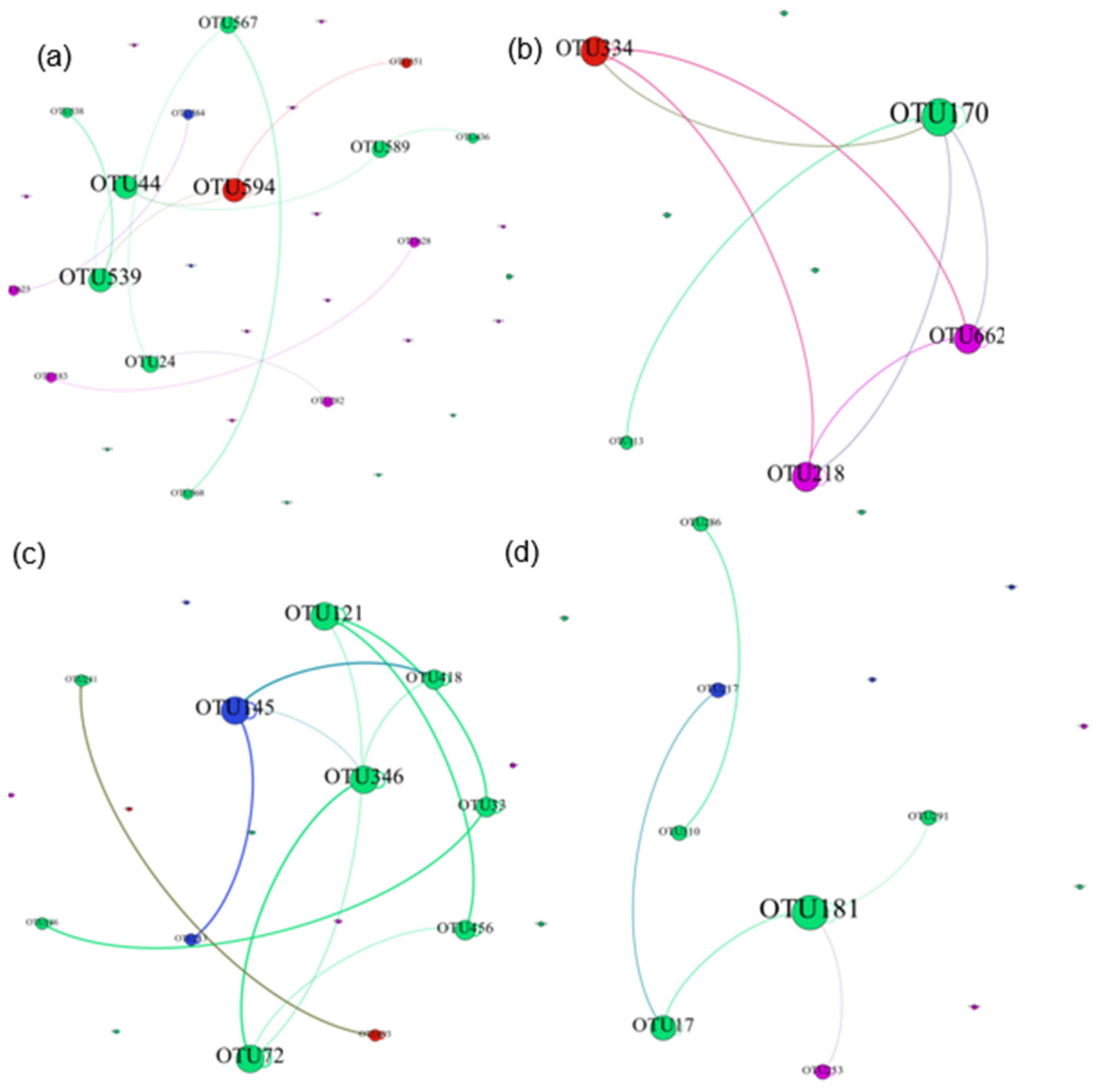
3.4. Network Relationship of Diatom Model Succession
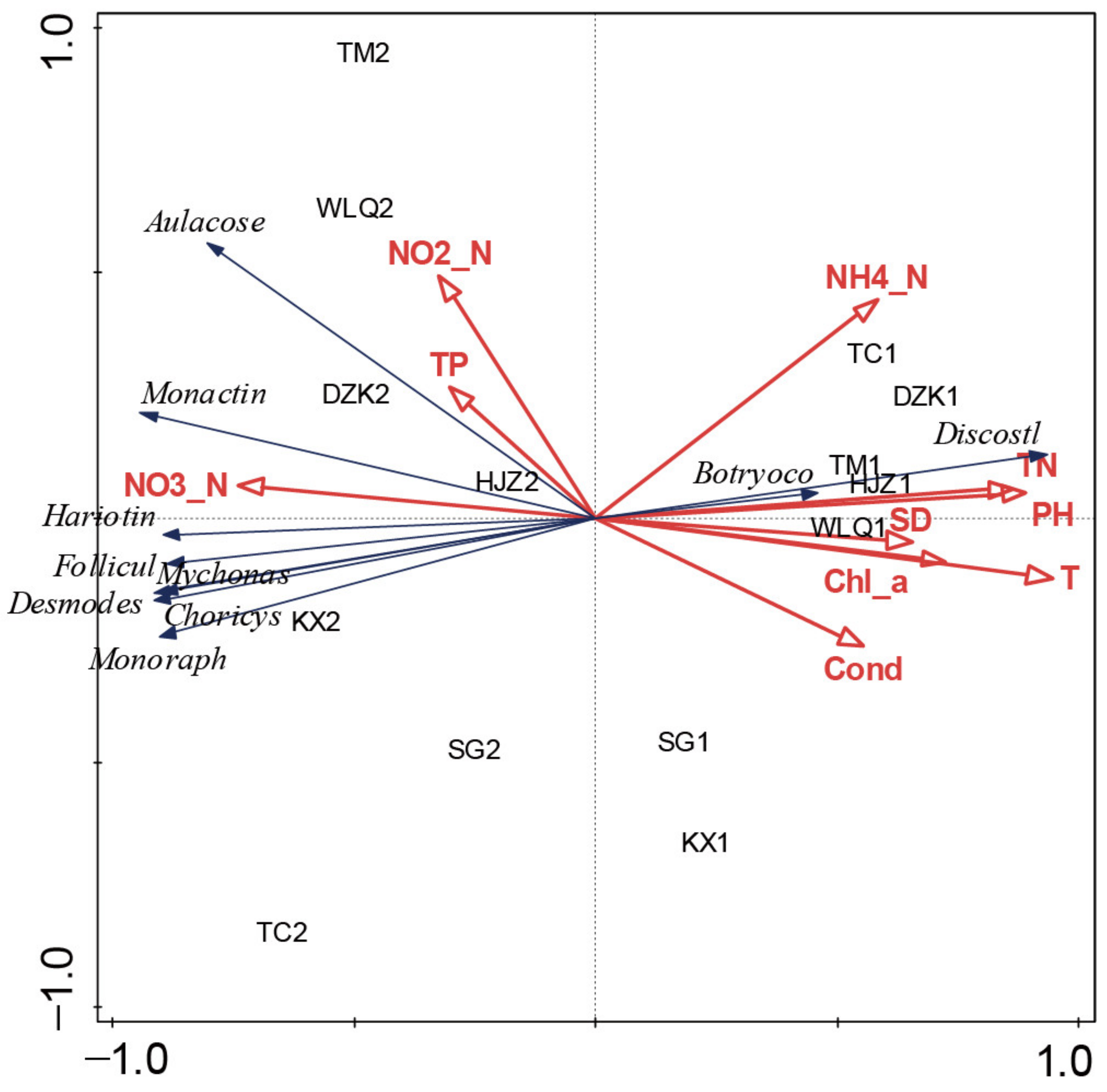
3.5. Environmental Factors Affecting Diatom Seasonal Variation
4. Discussion
4.1. Temporal and Spatial Variation of Diatom Community Structure in Danjiangkou Reservoir
4.2. Factors Affecting the Diatom Community in Danjiangkou Reservoir
4.3. The Effect of Detection Technology on Research Results on Diatoms in Aquatic Ecosystems
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wollschläger, J.; Wiltshire, K.H.; Petersen, W.; Metfies, K. Analysis of phytoplankton distribution and community structure in the German Bight with respect to the different size classes. J. Sea Res. 2015, 99, 83–96. [Google Scholar] [CrossRef] [Green Version]
- Tian, Y.; Jiang, Y.; Liu, Q.; Xu, D.; Song, J. The impacts of local and regional factors on the phytoplankton community dynamics in a temperate river, northern China. Ecol. Indic. 2021, 123, 107352. [Google Scholar] [CrossRef]
- Leynaert, A.; Fardel, C.; Beker, B.; Soler, C.; Delebecq, G.; Lemercier, A.; Pondaven, P.; Durand, P.E.; Heggarty, K. Diatom Frustules Nanostructure in Pelagic and Benthic Environments. Silicon 2018, 10, 2701–2709. [Google Scholar] [CrossRef]
- Saxena, A.; Tiwari, A.; Kaushik, R.; Iqbal, H.; Parra-Saldívar, R. Diatoms recovery from wastewater: Overview from an ecological and economic perspective. J. Water Process Eng. 2020, 39, 101705. [Google Scholar] [CrossRef]
- Lim, J.H.; Lee, C.W. Effects of eutrophication on diatom abundance, biovolume and diversity in tropical coastal waters. Environ. Monit. Assess. 2017, 189, 432. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.; Pan, K.; Wang, L.; Li, M.; Li, T.; Pang, B.; Kang, J.; Fu, J.; Lan, W. Anthropogenic Inputs Affect Phytoplankton Communities in a Subtropical Estuary. Water 2022, 14, 636. [Google Scholar] [CrossRef]
- Stonik, V.; Stonik, I. Low-Molecular-Weight Metabolites from Diatoms: Structures, Biological Roles and Biosynthesis. Mar. Drugs 2015, 13, 3672–3709. [Google Scholar] [CrossRef] [Green Version]
- Grimmett, M.R.; Lebkuecher, J.G. Composition of algae assemblages in middle Tennessee streams and correlations of composition to trophic state. J. Freshw. Ecol. 2017, 32, 363–389. [Google Scholar] [CrossRef]
- Battarbee, R.W. Diatom analysis and the acidification of lakes. Philos. Trans. R. Soc. B Biol. Sci. 1984, 305, 451–477. [Google Scholar] [CrossRef]
- Rühland, K.M.; Paterson, A.M.; Smol, J.P. Lake diatom responses to warming: Reviewing the evidence. J. Paleolimnol. 2015, 54, 1–35. [Google Scholar] [CrossRef]
- Kutlu, B.; Aydn, R.; Danabas, D.; Serdar, O. Temporal and seasonal variations in phytoplankton community structure in Uzuncayir Dam Lake (Tunceli, Turkey). Environ. Monit. Assess. 2020, 192, 105. [Google Scholar] [CrossRef] [PubMed]
- Chang, T.; Lu, X.; Pei, H.; Hu, W.; Xie, J. Seasonal dynamics of phytoplankton and its relationship with the environmental factors in Dongping Lake, China. Environ. Monit. Assess. 2013, 185, 2627–2645. [Google Scholar] [CrossRef]
- Moisan, J.R.; Moisan, T.A.; Abbott, M.R. Modelling the effect of temperature on the maximum growth rates of phytoplankton populations. Ecol. Model. 2002, 153, 197–215. [Google Scholar] [CrossRef]
- Boyd, P.W.; Rynearson, T.A.; Armstrong, E.A.; Fu, F.; Kendra, H.; Hu, Z.; Hutchins, D.A.; Kudela, R.M.; Elena, L.; Mulholland, M.R. Marine Phytoplankton Temperature versus Growth Responses from Polar to Tropical Waters—Outcome of a Scientific Community-Wide Study. PLoS ONE 2013, 8, e63091. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Paches, M.; Aguado, D.; Martínez-Guijarro, R.; Romero, I. Long-term study of seasonal changes in phytoplankton community structure in the western Mediterranean (Valencian Community). Environ. Sci. Pollut. Res. Int. 2019, 26, 14266–14276. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.R.; Yu, Z.D.; Ji, S.P.; Meng, J.; Liu, J. Diverse drivers of phytoplankton dynamics in different phyla across the annual cycle in a freshwater lake. J. Freshw. Ecol. 2021, 36, 13–29. [Google Scholar] [CrossRef]
- Reynolds, C.S.; Vera, H.; Carla, K.; Luigi, N.F.; Sergio, M. Towards a functional classification of the freshwater phytoplankton. J. Plankton Res. 2002, 24, 417–428. [Google Scholar] [CrossRef]
- Vajravelu, M.; Martin, Y.; Ayyappan, S.; Mayakrishnan, M. Seasonal influence of physico-chemical parameters on phytoplankton diversity, community structure and abundance at Parangipettai coastal waters, Bay of Bengal, South East Coast of India. Oceanologia 2018, 60, 114–127. [Google Scholar] [CrossRef]
- Xmab, C.; Swab, C.; Mlab, C.; Jie, M.; Jian, H.; Tlab, C.; Lcab, C. Changes in the phytoplankton community structure of the Backshore Wetland of Expo Garden, Shanghai from 2009 to 2010. Aquac. Fish. 2019, 4, 198–204. [Google Scholar] [CrossRef]
- Wu, Y.; Guo, P.; Su, H.; Zhang, Y.; Deng, J.; Wang, M.; Sun, Y.; Li, Y.; Zhang, X. Seasonal and spatial variations in the phytoplankton community and their correlation with environmental factors in the Jinjiang River Estuary in Quanzhou, China. Environ. Monit. Assess. 2021, 194, 44. [Google Scholar] [CrossRef]
- Huang, X.; Sun, M.; Xiang, L.; Zhang, E.; Grimm, E.C. The effect of diatoms on the grain size of lake sediments: A case study of the sediments of Lake Kanas. J. Paleolimnol. 2020, 63, 101–111. [Google Scholar] [CrossRef]
- Li, J.J.; Wang, L.; Cao, Q.; Rioual, P.; Lei, G.L.; Cai, B.G.; Zhang, J.; Zou, Y.F.; Yan, Y.; Wan, X.Q.; et al. Diatom Response to Global Warming in Douhu Lake, Southeast China. Acta Geol. Sin. 2021, 95, 638–647. [Google Scholar] [CrossRef]
- Liu, L.; Zhang, D.; Lv, H.; Yu, X.; Yang, J.U. Plankton communities along a subtropical urban river (Houxi River, southeast China) as revealed by morphological and molecular methods. J. Freshw. Ecol. 2013, 28, 99–112. [Google Scholar] [CrossRef]
- Gao, W.; Chen, Z.; Li, Y.; Pan, Y.; Zhu, J.; Guo, S.; Hu, L.; Huang, J. Bioassessment of a Drinking Water Reservoir Using Plankton: High Throughput Sequencing vs. Traditional Morphological Method. Water 2018, 10, 82. [Google Scholar] [CrossRef] [Green Version]
- Penna, A.; Casabianca, S.; Guerra, A.F.; Vernesi, C.; Scardi, M. Analysis of phytoplankton assemblage structure in the Mediterranean Sea based on high-throughput sequencing of partial 18S rRNA sequences. Mar. Genom. 2017, 36, 49–55. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.Y.; Gao, W.; Li, J.F.; Wen, Z.Z.; Liu, H.; Hu, L.Q.; Zhang, N.Q.; Cheng, X. Spatiotemporal distribution of phytoplankton and trophic status in the water resource area of the middle route of China’s South-to-North Water Transfer Project. Chin. J. Ecol. 2008, 27, 14–22. [Google Scholar] [CrossRef]
- Wang, Y.H.; Chen, L.; Niu, Y.; Yu, H.; Luo, M.K. Spatio-temporal variation in phytoplankton community and its influencing factors in Dan-jiangkou Reservoir. J. Lake Sci. 2016, 28, 1057–1065. [Google Scholar] [CrossRef] [Green Version]
- Yan, X.Y.; Zhang, Y.; Li, Y.Y.; Jiang, Y.Q.; Cui, Z.Z.; Gao, X.F.; Wu, N.C.; Nicola, F.; Han, X.M. Hydrologic and physicochemical factors co-drive seasonal changes of phytoplankton during dynamic water diversion processes in the Danjiangkou Reservoir. J. Lake Sci. 2021, 33, 1350–1363. [Google Scholar] [CrossRef]
- China Environmental Monitoring Station. Standard Practical Manual of Environmental Monitoring Methods; China Environmental Science Press: Beijing, China, 2013; Volume 1. [Google Scholar]
- Mora, D.; Abarca, N.; Proft, S.; Grau, J.H.; Zimmermann, J. Morphology and metabarcoding: A test with stream diatoms from Mexico highlights the complementarity of identification methods. Freshw. Sci. 2019, 38, 448–464. [Google Scholar] [CrossRef]
- Li, W.; Feng, Q.; Gao, B.; Chen, D.; Li, X. Distribution characteristics of microbial communities at a depth of 700 m level of Quantai coal mine in Xuzhou. Chin. J. Ecol. 2021, 40, 442–452. [Google Scholar] [CrossRef]
- Zhao, H.; Chen, P.; Tang, Q.; Chen, Y.; Li, J. Effects of in-situ biochar amendment on the microbial community structure of sediments in aquaculture ponds. J. Agro-Environ. Sci. 2021, 40, 2770–2778. [Google Scholar] [CrossRef]
- R Development Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2021; Available online: http://www.R-project.org (accessed on 10 August 2021).
- Hu, L.Q.; Feng, J.L.; Li, Y.F.; Sun, J.H. Pilot Study on Bio-monitoring in Danjiangkou Reservoir of the Water Source Area in the Middle Route of Chinas South to North Water Diversion Project. J. Henan Norm. Univ. (Nat. Sci. Ed.) 2014, 42, 100–104. [Google Scholar] [CrossRef]
- Jia, H.Y.; Xu, J.F.; Lei, J.S. Relationship of community structure of hytoplankton and environmental factors in Danjiangkou Reservoir bay. Yangtze River 2019, 50, 52–58. [Google Scholar] [CrossRef]
- Ampel, L.; Wohlfarth, B.; Risberg, J.; Veres, D.; Leng, M.J.; Tillman, P.K. Diatom assemblage dynamics during abrupt climate change: The response of lacustrine diatoms to Dansgaard–Oeschger cycles during the last glacial period. J. Paleolimnol. 2010, 44, 397–404. [Google Scholar] [CrossRef]
- Lei, Y.; Wang, Y.; Qin, F.; Liu, J.; Feng, P.; Luo, L.; Jordan, R.W.; Jiang, S. Diatom assemblage shift driven by nutrient dynamics in a large, subtropical reservoir in southern China. J. Clean. Prod. 2021, 317, 128435. [Google Scholar] [CrossRef]
- Youn, S.J.; Kim, H.N.; Im, J.K.; Kim, Y.J.; Yu, S.J. Effect of Environmental Factors on Phytoplankton Communities and Dominant Species Succession in Lake Cheongpyeong. J. Environ. Sci. Int. 2017, 26, 913–925. [Google Scholar] [CrossRef]
- Johnson, B.E.; Noble, P.J.; Heyvaert, A.C.; Chandra, S.; Karlin, R. Anthropogenic and climatic influences on the diatom flora within the Fallen Leaf Lake watershed, Lake Tahoe Basin, California over the last millennium. J. Paleolimnol. 2017, 59, 159–173. [Google Scholar] [CrossRef]
- Luo, W.; Li, J.; Lu, H.; Gu, Z.; Rioual, P.; Hao, Q.; Mackay, A.W.; Jiang, W.; Cai, B.; Xu, B.; et al. The East Asian winter monsoon over the last 15,000 years: Its links to high-latitudes and tropical climate systems and complex correlation to the summer monsoon. Quat. Sci. Rev. 2011, 32, 131–142. [Google Scholar] [CrossRef]
- Wang, L.; Lu, H.; Liu, J.; Gu, Z.; Mingram, J.; Chu, G.; Li, J.; Rioual, P.; Negendank, J.F.W.; Han, J.; et al. Diatom-based inference of variations in the strength of Asian winter monsoon winds between 17,500 and 6000 calendar years B.P. J. Geophys. Res. 2008, 113, D21101. [Google Scholar] [CrossRef] [Green Version]
- He, Y.X.; Zheng, Y.K.; Li, W.G.; Zhang, Z.C.; Ma, Y.X.; Zhao, T.X.; Ren, Y.F. Characteristics of eukaryotic phytoplankton community structure in early spring and its relationship with environmental factors in Danjiangkou Reservoir. Acta Sci. Circumst. 2021, 41, 2192–2200. [Google Scholar] [CrossRef]
- Byungkwan, J.; Yongjae, K.; Won, J.S.; Hakyoung, L.; Yongsik, S. Temporal Variation and Identification of a Centric Diatom, Stephanodiscus spp. during Winter-spring Blooms in the Yeongsan River. Korean J. Ecol. Environ. 2014, 47, 273–281. [Google Scholar] [CrossRef]
- Roberts, S.L.; Swann, G.E.A.; McGowan, S.; Panizzo, V.N.; Vologina, E.G.; Sturm, M.; Mackay, A.W. Diatom evidence of 20th century ecosystem change in Lake Baikal, Siberia. PLoS ONE 2018, 13, e0208765. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhu, C.; Zhang, J.; Nawaz, M.Z.; Mahboob, S.; Al-Ghanim, K.A.; Khan, I.A.; Lu, Z.; Chen, T. Seasonal succession and spatial distribution of bacterial community structure in a eutrophic freshwater Lake, Lake Taihu. Sci. Total Environ. 2019, 669, 29–40. [Google Scholar] [CrossRef]
- Liu, B.; Chen, S.; Liu, H.; Guan, Y. Changes in the ratio of benthic to planktonic diatoms to eutrophication status of Muskegon Lake through time: Implications for a valuable indicator on water quality. Ecol. Indic. 2020, 114, 106284. [Google Scholar] [CrossRef]
- Shahar, B.; Shpigel, M.; Barkan, R.; Masasa, M.; Neori, A.; Chernov, H.; Salomon, E.; Kiflawi, M.; Guttman, L. Changes in metabolism, growth and nutrient uptake of Ulva fasciata (Chlorophyta) in response to nitrogen source. Algal Res. 2020, 46, 101781. [Google Scholar] [CrossRef]
- Shou, W.; Zong, H.; Ding, P.; Hou, L. A modelling approach to assess the effects of atmospheric nitrogen deposition on the marine ecosystem in the Bohai Sea, China. Estuar. Coast. Shelf Sci. 2018, 208, 36–48. [Google Scholar] [CrossRef]
- Malik, H.I.; Northington, R.M.; Saros, J.E. Nutrient limitation status of Arctic lakes affects the responses of Cyclotella sensu lato diatom species to light: Implications for distribution patterns. Polar Biol. 2017, 40, 2445–2456. [Google Scholar] [CrossRef]
- Malik, H.I.; Warner, K.A.; Saros, J.E. Comparison of seasonal distribution patterns of Discostella stelligera and Lindavia bodanica in a boreal lake during two years with differing ice-off timing. Diatom Res. 2018, 33, 1–11. [Google Scholar] [CrossRef]
- Zhang, Q.; Dong, X.; Chen, Y.; Yang, X.; Xu, M.; Davidson, T.A.; Jeppesen, E. Hydrological alterations as the major driver on environmental change in a floodplain Lake Poyang (China): Evidence from monitoring and sediment records. J. Great Lakes Res. 2018, 44, 377–387. [Google Scholar] [CrossRef]
- Zhang, K.; Dong, X.; Yang, X.; Kattel, G.; Zhao, Y.; Wang, R. Ecological shift and resilience in China’s lake systems during the last two centuries. Glob. Planet. Change 2018, 165, 147–159. [Google Scholar] [CrossRef]
- Tuji, A.; Williams, D.M. The identity of Cyclotella glomerata Bachmann and Discostella nipponica (Skvortzov) Tuji et Williams comb. et stat. nov. (Bacillariophyceae) from Lake Kizaki, Japan. J. Clin. Microbiol. 2006, 41, 486–488. [Google Scholar] [CrossRef] [Green Version]
- Esteban, A.T.; Gunnar, B.; Jorn, B.; Agostino, M. Mechanisms shaping size structure and functional diversity of phytoplankton communities in the ocean. Sci. Rep. 2015, 5, 8918. [Google Scholar] [CrossRef]
- Acevedo-Trejos, E.; Brandt, G.; Merico, A.; Smith, S.L. Biogeographical patterns of phytoplankton community size structure in the oceans. Glob. Ecol. Biogeogr. 2013, 22, 1060–1070. [Google Scholar] [CrossRef]
- Borutsky, E.B.; Wu, X.; Bai, G.; Ge, M.; Wang, Q.; Wang, S.; Chen, S. Hydrobiological survey of the region of the projected dam-reservoir of tankiangkou, with propositions for fisheries management. Acta Hydrobiol. Sin. 1959, 1, 33–56. [Google Scholar]
- Wu, H.; Peng, J.; Han, D.; Jian, D.; Zhou, Q. Composition and ecological changes of Phytoplankton in Danjiangkou Reservoir. J. Lake Sci. 1996, 8, 43–50. [Google Scholar] [CrossRef]
- Banerji, A.; Bagley, M.; Elk, M.; Pilgrim, E.; Domingo, J.S. Spatial and temporal dynamics of a freshwater eukaryotic plankton community revealed via 18S rRNA gene metabarcoding. Hydrobiologia 2018, 818, 71–86. [Google Scholar] [CrossRef]
- Yu, L.; Zhang, W.; Liu, L.; Yang, J. Determining Microeukaryotic Plankton Community around Xiamen Island, Southeast China, Using Illumina MiSeq and PCR-DGGE Techniques. PLoS ONE 2017, 10, e0127721. [Google Scholar] [CrossRef] [Green Version]
- Nunes, S.; Latasa, M.; Delgado, M.; Emelianov, M.; Simó, R.; Estrada, M. Phytoplankton community structure in contrasting ecosystems of the Southern Ocean: South Georgia, South Orkneys and western Antarctic Peninsula. Deep-Sea Res. Part I 2019, 151, 103059. [Google Scholar] [CrossRef] [Green Version]
- Guerrero, J.M.; Echenique, R.O. Discostella taxa (Bacillariophyta) from the Río Limay basin (northwestern Patagonia, Argentina). Eur. J. Phycol. 2006, 41, 83–96. [Google Scholar] [CrossRef] [Green Version]

| Environmental Factor | Spring | Summer | Autumn | Winter |
|---|---|---|---|---|
| pH | 8.80 ± 0.05 | 9.15 ± 0.04 | 8.76 ± 0.05 | 8.86 ± 0.07 |
| Cond/ms·m−1 | 27.39 ± 0.37 | 28.21 ± 0.69 | 27.87 ± 0.41 | 27.39 ± 0.40 |
| WT/℃ | 23.07 ± 0.78 | 32.02 ± 0.90 | 21.66 ± 0.64 | 12.14 ± 0.57 |
| SD/m | 5.37 ± 1.03 | 3.94 ± 0.40 | 3.32 ± 0.32 | 3.41 ± 0.16 |
| TP/mg·L−1 | 0.03 ± 0.00 | 0.04 ± 0.01 | 0.04 ± 0.01 | 0.04 ± 0.02 |
| Chl-a/mg·L−1 | 0.002 ± 0.00 | 0.005 ± 0.00 | 0.003 ± 0.00 | 0.002 ± 0.00 |
| NH4+-N /mg·L−1 | 0.13 ± 0.03 | 0.17 ± 0.03 | 0.16 ± 0.03 | 0.13 ± 0.02 |
| NO3−-N/mg·L−1 | 0.68 ± 0.09 | 0.47 ± 0.07 | 0.95 ± 0.04 | 0.68 ± 0.10 |
| TN/mg·L−1 | 1.06 ± 0.07 | 1.26 ± 0.24 | 1.16 ± 0.07 | 1.19 ± 0.10 |
| NO2−-N/mg·L−1 | 0.003 ± 0.00 | 0.003 ± 0.00 | 0.004 ± 0.00 | 0.002 ± 0.00 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, C.; He, Y.; Li, W.; Guo, X.; Xiao, C.; Zhao, T. High-Throughput Sequencing of Diatom Community, Its Spatial and Temporal Variation and Interrelationships with Physicochemical Factors in Danjiangkou Reservoir, China. Water 2022, 14, 1609. https://doi.org/10.3390/w14101609
Zhang C, He Y, Li W, Guo X, Xiao C, Zhao T. High-Throughput Sequencing of Diatom Community, Its Spatial and Temporal Variation and Interrelationships with Physicochemical Factors in Danjiangkou Reservoir, China. Water. 2022; 14(10):1609. https://doi.org/10.3390/w14101609
Chicago/Turabian StyleZhang, Chunxia, Yuxiao He, Weiguo Li, Xiaoming Guo, Chunyan Xiao, and Tongqian Zhao. 2022. "High-Throughput Sequencing of Diatom Community, Its Spatial and Temporal Variation and Interrelationships with Physicochemical Factors in Danjiangkou Reservoir, China" Water 14, no. 10: 1609. https://doi.org/10.3390/w14101609
APA StyleZhang, C., He, Y., Li, W., Guo, X., Xiao, C., & Zhao, T. (2022). High-Throughput Sequencing of Diatom Community, Its Spatial and Temporal Variation and Interrelationships with Physicochemical Factors in Danjiangkou Reservoir, China. Water, 14(10), 1609. https://doi.org/10.3390/w14101609





